Abstract
We propose curcumin as a preventive measure to avoid/manage periodontitis (PD), and as a natural immunosuppressant for rheumatoid arthritis (RA). PD, mainly caused by Porphyromonas gingivalis forming biofilm and leading to tooth decay, is a major public health issue and a risk factor for the development of RA in humans. P. gingivalis is able to trigger experimental autoimmune arthritis in animal models and in humans can induce citrullinated peptides, which not only are a source of anti-citrullinated antibodies (ACPAs), but also participate in autoreactive responses and disease development. Curcumin appears to have efficient anti-bacterial activity against P. gingivalis infection and biofilm formation. In addition to antibacterial, anti-oxidant, and anti-inflammatory action, curcumin exerts unique immunosuppressant properties via the inhibition of Th17 pro-inflammatory responses and promotion of regulatory T cells, thus suppressing autoimmunity. We introduce curcumin as a natural product for the management of both PD and RA-related autoreactivity, possibly also as a preventive measure in early RA or individuals at high risk to develop RA.
1. Introduction
Rheumatoid arthritis (RA) is an autoimmune rheumatic disease characterized by immune-mediated joint inflammation, which leads to joint destruction, loss of joint function, and disability, if left untreated [1,2,3]. In fact, joint erosion can develop early during the first three months of the disease in 25% of RA patients and mostly during the first two years of the disease [4].
The immunopathogenesis of RA has been extensively investigated and it is well established that genetic factors, predominantly HLA-DRB1 shared epitope (HLA-DRB1SE) alleles [5,6], epigenetic, and environmental factors are involved in the development of the disease [1,2,7,8,9,10]. In immunopathological terms, proinflammatory Th1 cells and B cells and proinflammatory soluble mediators (TNFα, IFNγ, IL-6) are involved in the pathogenesis of the disease [11]. A better understanding of the underlying mechanisms of the disease has led to its therapeutic management with synthetic disease-modifying antirheumatic drugs (sDMARD) and novel biological agents that target specific molecules involved in disease pathogenesis, which can prevent joint damage and disease progression, and improve disease prognosis [2,9,11,12,13]. Since these therapies can have potentially toxic side effects, it is very important for practicing physicians to diagnose the disease early and accurately, especially more aggressive forms, in order to select the appropriate treatment [14,15].
2. Curcumin as a Remedy for the Treatment of Rheumatoid Arthritis: Data from Animal Studies
Curcumin, a traditional remedy and major curcuminoid found in the spice turmeric, has been used for centuries for treating chronic inflammatory diseases [16,17,18,19]. Recent studies have demonstrated the decisive role of curcumin in treating autoimmune diseases [19,20,21,22], including experimental autoimmune arthritis, the animal model of rheumatoid arthritis (RA) [23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38]. How curcumin exerts its beneficial effect in experimental arthritis is under investigation [23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38]. It appears that curcumin has the ability to decrease pro-inflammatory Th1 and Th17 cells and increase regulatory T cells [23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39] (Figure 1). Recent evidence shows that reciprocal regulation of Th17/Treg cells (decreasing Th17 cells and increasing Tregs) by administration of IL-10-producing cells (Bregs), can suppress collagen-induced arthritis (CIA) and thus IL-10-producing Bregs can be considered an attractive therapeutic strategy for T cell-mediated autoimmune rheumatic diseases (ARDs) such as RA [40,41,42]. Currently, there is no available approved medication able to efficiently modulate the Th17/Treg/Breg balance in vivo in RA. Curcumin, as a bioactive immunomodulatory agent, has well-documented actions against proliferating lymphocytic populations [43], yet its role in modulating Th17/regulatory cell responses in ARDs remains largely unexplored. However, there are studies on murine models of experimental arthritis investigating the role of curcumin; these are summarized in Table 1.
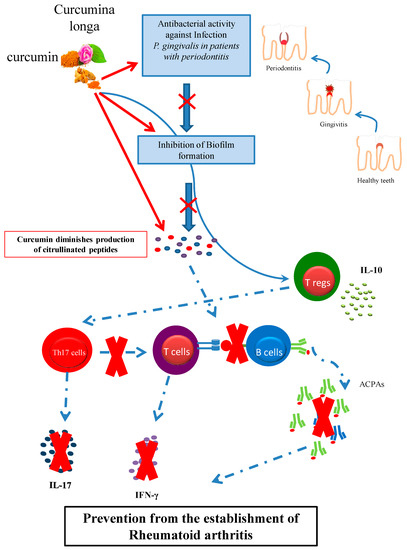
Figure 1.
Curcumin may play a significant role in preventing from rheumatoid arthritis through its antibacterial action against P. gingivalis infection and biofilm formation in patients with periodontitis; and modulation of the proinflammatory immune response, such as inhibition of Th17 cells and enhancement of IL-10 producing regulatory T cells.

Table 1.
Curcumin’s biological effects in animal models of experimental arthritis.
2.1. Experimental Arthritis
The CIA model is one of the best-characterized murine models for RA. In genetically susceptible murine strains, such as C57BL/6 mice, the administration of type II collagen (CII) causes a robust and sustained T-cell response to administered CII, leading to synovitis and erosion that histologically resemble RA [44]. It is also a significant model for studying Th17 responses [45] as symptoms of CIA are markedly suppressed in mice lacking IL-17 (IL-17−/−mice) [45,46]. Paradoxically, the effect of curcumin on IL-17 production in CIA induced in C57BL/6 mice has not been thoroughly investigated. Cong et al. [47] using C57BL/6 (B6) and B6.RAG-2−/−mice have demonstrated that curcumin could induce tolerogenic bone marrow-derived dendritic cells that promote the differentiation of intestinal Tregs [47]. Some evidence of Th17 cells was documented in C57BL/6 mice with a mutation in the Foxp3 gene scurfin (scurfy mice) that develop polyendocrinopathy and enteropathy X-linked (IPEX) syndrome, a lethal autoimmune disease [48]. Curcumin ameliorated IPEX syndrome by inhibiting Th1/Th2/Th17 responses [41]. Scurfy mice on a curcumin diet survived four times longer (92.5 days) compared to scurfy mice fed a normal diet (23 days), and in in vitro experiments curcumin decreased the production of cytokines IFN-γ (Th1 cytokine), IL-4 (Th2 cytokine), and IL-17A (Th17 cytokine) in CD4+ T cells [41].
Okamoto et al. [35] has reported that curcumin treatment inhibited IL-17 production in vitro and ameliorated CIA in DBA/1 J mice. A significant anti-arthritic effect of curcumin has also been previously demonstrated in CIA induced in DBA/1 J mice but no information has been given on the effects on Th17 cells. Huang et al. [37] injected mice intra-peritoneally with curcumin and found decreased levels of B cell-activating factor (BAFF), IFN-γ, and IL-6 in serum and their production by spleen cells but gave no information on IL-17 production [37]. DBA/1 mice immunized with CII and treated with curcumin every other day for two weeks also demonstrated reduced clinical arthritis scores, associated with reduced expression of TNF-alpha and IL-1beta in the ankle joint, and decreased levels of IgG2a in serum [36]. Orally administrated curcumin also suppressed the production of matrix metalloproteinase (MMP)-1 and MMP-3 and ameliorated CIA [38]. Curcumin can also attenuate collagen-induced inflammatory responses through the “gut–brain axis” by modulating the function of the cholinergic system [23].
Funk et al. [34] were the first to report the anti-arthritic effect of complex turmeric extracts containing curcuminoids in experimental arthritis induced in Lewis rats [34]. The anti-inflammatory effect of the tetramethylpyrazine, resveratrol, and curcumin (TRC) combination in acute and chronic inflammation was reported in vivo in CIA induced in rats [28]. The TRC combination could inhibit the production of TNF-α, IL-1β, and IL-6 in the serum but, again, no information was available for IL-17 levels [28]. In addition, curcumin administered via the intravenous (IV) or oral route had a therapeutic effect similar to methotrexate on adjuvant-induced arthritis in rats, an effect associated with decreased levels of TNF-α and interleukin-1β in both synovial fluid and serum [25].
2.2. Experimental Periodontitis
In experimental periodontitis (PD), an effect of curcumin on systemic Th17 responses, gingival IL-17A expression, Retinoic Acid Receptor-Related Orphan Receptor γt, and alveolar bone loss has recently been described [49].
2.3. Experimental Autoimmune Encephalitis
More data on curcumin and Th17 responses are available from studies in experimental autoimmune encephalomyelitis (EAE), a model for multiple sclerosis (MS). In EAE, strong evidence suggests that IL-17-producing T cells play a dominant pathogenic role. In EAE induced by the MOG-peptide 35–55 in C57BL/6 mice, CD4+ Th17 cells are present both in the periphery and in the inflamed central nervous system [50] and amelioration of EAE by curcumin treatment was through inhibition of IL-17 production [39]. Further studies indicated that dietary curcumin inhibited the differentiation of pro-inflammatory Th1/Th17 cells in vivo during encephalomyelitis and instead promoted Th2 cells [51]. Curcumin appeared to silence IL-23/Th17-mediated pathology in EAE by enhancing HO-1/STAT3 interaction in dendritic cells (DCs) [51].
3. Human Rheumatoid Arthritis: Data from Clinical Trials
Clinical data in human RA have started to emerge. Though limited, the findings from two small clinical trials are encouraging [52,53]. The first small randomized clinical trial [52] enrolled 45 patients with active RA into three groups, receiving curcumin (500 mg) alone, diclofenac sodium (50 mg) alone, or curcumin and diclofenac in combination. The primary endpoint was good or moderate Disease Activity Score (DAS) 28 response, and the secondary end point was C-reactive protein (CRP) levels at week 8 post-treatment. Curcumin administration was safe and no adverse events were reported. Patients in all three treatment groups showed statistically significant changes in their DAS28 scores. DAS28 was reduced from 6.40 at baseline to 3.55 in the curcumin-alone group, and from 6.72 to 3.89 in the diclofenac-alone group. However, CRP levels were reduced by 52% in the curcumin-alone group but were not reduced in the diclofenac-alone group [52]. Another small randomized, double-blind, placebo-controlled study [53] evaluated the efficacy of curcumin compared with that of a placebo in RA patients with medium disease activity and rather low CRP levels. A novel curcumin matrix formulation, with 10-fold bioavailability compared to unformulated 95% curcumin, was given to all participants. Twelve patients in each group received the placebo, 250 or 500 mg of the curcumin product twice daily for 90 days. Curcumin was well tolerated without adverse effects. At the end of the study, there was a significant DAS28 improvement and ACR20 response in a high percentage of patients who received curcumin at either the 250 or 500 mg dose. In the curcumin groups, the disease improvement was accompanied by a significant decrease in markers of inflammation (CRP and erythrocyte sedimentation rate, ESR), and rheumatoid factor (RF) levels [53]. These results are encouraging and should be confirmed in larger trials, which will also address the safety profile of long-term usage of curcumin or a supplement’s tolerance. In addition, the reduction of RF levels to be found in such a short time post-treatment is unexpected and requires confirmation. There are no reports on the effect of curcumin on ACPA levels. Nevertheless, these findings support the notion that curcumin may indeed have beneficial effects not only in experimental models of arthritis but also in patients with RA. The work performed in animals has hinted at the pathophysiological pathways that could account for curcumin’s advantageous effects. Another point to be considered is the long-term safety profile of curcumin at pharmacological dosing, which is not known at present.
5. Rheumatoid Arthritis, Anti-Citrullinated Peptide Antibodies, Citrullination, and Periodontitis
The serological marker of RA is the presence of high-titer autoantibodies (autoabs), namely rheumatoid factor (RF) and abs against citrullinated peptides (anti-CCP abs, ACPAs) [1,64,65,66]. ACPAs appear years before the clinical onset of RA. Of clinical importance, the presence of peptide-specific ACPAs predict the future development of the disease in patients with undifferentiated arthritis [4,65,67,68], raising the question of whether such autoantibodies or ACPA-producing plasmocytes play a pathogenic role in the development of RA [69,70,71]. Along these lines, several studies have clearly demonstrated that ACPA is the most reliable prognosticator of radiographic progression in RA [72].
Prompt therapeutic intervention at very early disease stages, a period known as the “window of opportunity,” can slow disease onset and progression [14,15]. Of relevance, there is an ongoing debate among investigators as to how we can manage and whether we can/must intervene in anti-CCP (ACPA) seropositive patients with no recognizable clinical arthritis [73].
Nevertheless, upon the discovery of ACPAs, a better understanding of the immunopathogenesis of RA has been gained [65,74]. ACPAs recognize few citrullinated peptides in early preclinical disease states, but over time the number of recognized citrullinated peptides increases; such an increase is accompanied by elevated levels of pro-inflammatory cytokines and finally clinical disease [70,75]. The fact that the presence of ACPAs, particularly those against specific citrullinated peptides, is associated with more aggressive/severe disease supports the notion that such peptides are indeed makers of the disease rather than markers of it [11,65,73,76].
Citrullination is a post-translational modification of proteins caused by peptidyl arginine deiminases (PADs) [77,78]. At the experimental level, it has been shown that citrullination can form neoantigens that activate T cells. Such T helper (Th) cells can offer antigen-specific help to B cells to differentiate into ACPA-producing plasma cells [8,79,80]. Convincing data have shown that citrullination amplifies the affinity of peptides to HLA-DRB1SE alleles [81,82]. Of interest, T cells can also recognize PAD. Such an ability of T cells has a clear implication, namely the capacity of T cells to help plasmocytes produce Abs against hapten/carrier citrullinated proteins bound to PAD [83].
The extent to which pathogens play a role in the development of RA started to unfold when it became clear that citrullination induced by Porphyromonas (P. gingivalis), a pathogen of PD, can be a source of citrullinated peptides (such as neoantigens) that can then break immunological tolerance [63,84,85,86,87]. P. gingivalis produces gingipains, proteases that cleave proteins at peptidyl arginine, and PAD (PPAD) that preferentially citrullinates C-terminal arginine, thus creating neoantigens [88]. There is evidence of P. gingivalis infection years before the onset of clinical RA [87,89].
There is an association between P. gingivalis infection and ACPA-positive RA [8,63,90,91]. Furthermore, in ACPA-positive RA an interaction between markers of P. gingivalis infection, smoking, and HLA-DRB1SE was reported [92]. ACPA to citrullinated α-enolase peptide 1 (CEP1), an immunodominant peptide in RA, showed high homology with α-enolase from P. gingivalis and cross-reacted with citrullinated recombinant P. gingivalis enolase [93]. P. gingivalis inoculation of mice caused PPAD-dependent exacerbation of CIA [94]. In addition, oral inoculation of P. gingivalis in HLA-DR1 transgenic mice transiently increased Th17 cells in regional lymph nodes and peripheral blood, induced a massive increase in proinflammatory cytokines, and exacerbated CIA [95]. P. gingivalis can also affect inflammation through gut microbiota (discussed later) [96,97].
Another causative agent of PD, Aggregatibacter actinomycetemcomitans (A. actinomycetemcomitans), also causes citrullination of human proteins through the production of leukotoxin A [84,98]. Furthermore, HLA-DRB1SE is associated with ACPA only in RA patients exposed to A.actinomycetemcomitans [98]. Epstein–Barr virus (EBV), which infects epithelial cells and B cells, also causes ACPA production. ACPAs against peptides derived for EBV nuclear antigen (EBNA)1 (PCV1) and EBNA2 (PCV2) cross-react with human citrullinated proteins [99]. In addition, ACPAs against PCV1 and PCV2 and ACPAs against histone-4-derived citrullinated peptide (HCP1) and HCP2 appear years before the onset of clinical RA and predict with a high risk ratio (OR = 8 to 19) the subsequent development of RA [90]. EBV is also a cause of PD [100].
Newly diagnosed, untreated RA patients show a high prevalence of PD and significant changes to their oral microbiome, compared to those without RA [101]. Also, microbiome changes seen in saliva and dental samples appear to be restored, at least in part, after efficient treatment of the underlying disease [102].
P. gingivalis can cause arthritis through changes of gut microbiota. Orally administered P. gingivalis changed the gut microbiome with a decrease in Bacteriodetes phylum, increased Th17 cells in mesenteric lymphocytes, increased intestinal permeability, and aggravated CIA [96,97,103]. P. gingivalis DNA was also detected in synovial tissue from RA patients. More interestingly, P. gingivalis DNA in synovial tissue was detected more frequently in HLA-DRB1*04-positive than HLA-DRB1*04-negative RA patients [104]. These findings suggest that molecular mimicry between dysbiotic bacteria and humans may operate in RA as well. The proiflammatory milieu of periodontitis exacerbates arthritis as DNA from periodontopathogenic bacteria stimulates macrophage IL-6 and TNFa production [105].
PD and smoking, the two known environmental risk factors for RA, argue in support of the concept that an immune response mounted at mucosal sites provides the impetus for the initiation of a series of events that culminate in losing immune tolerance and the perpetuation of autoreactive responses leading to RA [106,107,108]. An excellent Swedish study in twins (from the Swedish Twin Registry) has shown that among 12,590 twins, 350 (2.8%) had ACPA (including 124 (1% of the total or 35.2% of the ACPA positive)) [109]. A clear association was found between smoking and HLADRB1SE and the ACPAs’ presence [109]. The authors concluded that environmental and lifestyle (e.g., smoking, PD) rather than genetic factors may play a more important role in the production of ACPAs [109].
Gingival grevicular fluid collected from the space between the tooth and gingival mucosa from patients with PD, not only shows marked citrullination, but also mirrors the hypercitrullination in RA joints; on the other hand, it shows minimal citrullination in healthy individuals without PD [98].
PD is highly prevalent among adults in most developed countries around the globe [110]. It is a chronic disease associated with the development of a pathogenic bacterial biofilm of dental plaque. This bacterial biofilm is one of the most diverse microbial ecosystems within the human body, involving approximately 700 different bacterial species, the most prominent of which are Porphyromonas gingivalis, Prevotella intermedia, and A. actinomycetemcomitans [111]. The encounter of the host’s immune system with these pathogenic bacteria is responsible for the initiation of an immune-mediated inflammation, which ultimately leads to the destruction of periodontal tissues and the tooth. PD is a major public health issue affecting approximately 15% of the adult population worldwide. The socioeconomic impact of the disease is huge and attempts to efficiently manage the disease are still lacking [110]. Surgical intervention, scaling, and root planing accompanied by antibiotic therapy are widely used measures to handle PD. However, their effect is limited, short-term, and may have adverse side effects, the most profound being antibiotic resistance in those undergoing long-term antibiotic treatment [112]. Thus, there is an agreement among investigators that the most efficient means must be non-invasive, preventive measures to avoid the establishment of PD and its deleterious effects via control of the infection and the development and biofilm formation. Towards this, several dietary compounds/supplements have been suggested as efficient and safe, such as plant-originated polyphenol extracts [113], which exert antibacterial (and anti-oxidant or anti-inflammatory), in particular anti-P. gingivalis activity, such as cranberries, gallic acid, quercetin, naringin, and, more recently, curcumin.
6. Prevention of Early RA: Fighting Periodontitis
Since directly or indirectly citrullinated peptides, ACPAs, and autoreactive lymphocytes specific for such peptides are involved in the pathogenesis of RA, preventive measures must be taken to control or, even better, prevent the development of the vicious circle that places citrullinated peptides as the key player [14,15,64,65,73,114].
If PD induced by P. gingivalis or other pathogens is a key factor participating in the loss of tolerance that characterizes early RA [85,89,91,94,104,107,115,116], a key preventive measure would be efficient treatment of PD, i.e., infection by P. gingivalis or other PD-related oral pathogens (Figure 1). To this end, lessons must be learned from the work performed on this topic by researchers in the past [117,118,119,120,121,122,123,124,125,126,127,128,129].
Curcumin and Oral Hygiene: Curcumin Inhibits the Growth of P. gingivalis and Prevents Periodontitis
Following the above argument, an obvious intervention to reduce the risk of ACPA production is to maintain good oral hygiene and treat PD. The rationality of this approach is to stop feeding autoantigens. It becomes apparent that remedies, such as curcumin, could be used as part of preventive measures, especially if they have an effect on the management of PD. Indeed, investigators have studied the effect of curcumin in PD, mainly its ability to inhibit P. gingivalis [130]. The lipopolysaccharide (LPS) of P. gingivalis stimulates cytokine secretion in immune cells, and has been considered a major cause of inflammation in PD. Macrophages are prominent cell subsets at periodontal sites of inflammation and the effect of curcumin on macrophages stimulated with P. gingivalis LPS has been studied [117]. TNF-alpha and IL-1beta expression was inhibited in a dose-dependent manner when the murine macrophage RAW264.7 cell line was pre-treated with various concentrations of curcumin and stimulated by P. gingivalis LPS [117]. Also, curcumin can inhibit P. gingivalis LPS-induced COX-2 expression, mainly due to the inhibition of the NF-κB pathway [119]. Curcumin also downregulated NF-κB and NF-κB-regulated genes (vascular endothelial growth factor, matrix metalloproteinase 9, cyclo-oxygenase-2) in an orthotopic mouse model of pancreatic cancer [131].
Curcumin can exert antibacterial activity on a series of periodontopathic bacteria, including P. gingivalis, Prevotella intermedia, Fusobacterium nucleatum, and Treponema denticola. Bacterial growth was inhibited even at very low concentrations of curcumin, as 20 μg/mL curcumin inhibited the growth of P. gingivalis biofilm formations by >80%, suggesting that, at least in vitro, this nutrient can be a potent agent for preventing periodontal diseases [130]. Amongst polyphenols, both curcumin and quercetin are able to alter the architecture of mature multi-species biofilms; however, only curcumin-treated biofilms display significantly reduced metabolic activity [132]. Local curcumin was clinically effective and reduced the microbial load in patients with chronic PD [124].
7. Unresolved Issues
Curcumin, the most bioactive curcuminoid of turmeric, has relatively poor oral bioavailability in humans due to low solubility in aqueous solvents, poor stability and absorption, and rapid elimination from the systemic circulation [133]. Such disadvantages may limit its therapeutic application, including fighting or preventing the development of RA. Multiple heterogeneous approaches have been applied to overcome such obstacles, involving the development of curcumin-based nanoparticle formulas, curcumin structure modifications, metabolism inhibitors-based administration, and the development of curcumin prodrugs to improve bioactivity and bioavailability and to optimize its antimicrobial, anti-oxidant, and anti-inflammatory potential [134,135]. This may assist efforts to fight PD, as well as prevent RA, especially in individuals at high risk, such as those who carry susceptible genes (i.e., the HLA–DRB1SE genes). The data are still limited and safe conclusions are difficult to draw. An increasing number of reports, mainly on the Internet, suggest that turmeric (alone or in combination with coconut oil) as a surplus for efficient whitening of teeth, appears attractive, but the data are still lacking. Nevertheless, natural compounds such as turmeric’s curcumin, which convincingly show both RA-related anti-bacterial activity and immunosuppressant/regulatory action, are ideal candidates to focus on in the years to come.
Author Contributions
The work is part of the MSc thesis of E.A., supervised by D.P.B. E.A., A.M., A.G., and D.P.B. wrote the first draft. C.K. and L.I.S. revised the draft and rewrote significant parts of the manuscript. T.M. prepared the table. D.P.B. and A.G. prepared the artwork. D.P.B. and L.I.S. had the original idea. All authors significantly contributed to the work and approved the final version of the paper.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- McInnes, I.B.; Schett, G. The pathogenesis of rheumatoid arthritis. N. Engl. J. Med. 2011, 365, 2205–2219. [Google Scholar] [CrossRef] [PubMed]
- Choy, E.H.; Panayi, G.S. Cytokine pathways and joint inflammation in rheumatoid arthritis. N. Eng. J. Med. 2001, 344, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, L.I.; Xu, B.; Artlett, C.M.; Lu, S.; Jimenez, S.A.; Platsoucas, C.D. Oligoclonal T cell expansion in the skin of patients with systemic sclerosis. J. Immunol. 2002, 168, 3649–3659. [Google Scholar] [CrossRef] [PubMed]
- Barouta, G.; Katsiari, C.G.; Alexiou, I.; Liaskos, C.; Varna, A.; Bogdanos, D.P.; Germenis, A.E.; Sakkas, L.I. Anti-mcv antibodies predict radiographic progression in Greek patients with very early (<3 months duration) rheumatoid arthritis. Clin. Rheumatol. 2017, 36, 885–894. [Google Scholar] [PubMed]
- Wordsworth, B.P.; Lanchbury, J.S.; Sakkas, L.I.; Welsh, K.I.; Panayi, G.S.; Bell, J.I. Hla-dr4 subtype frequencies in rheumatoid arthritis indicate that DRB 1 is the major susceptibility locus within the HLA class ii region. Proc. Natl. Acad. Sci. USA 1989, 86, 10049–10053. [Google Scholar] [CrossRef] [PubMed]
- Gregersen, P.K.; Silver, J.; Winchester, R.J. The shared epitope hypothesis. An approach to understanding the molecular genetics of susceptibility to rheumatoid arthritis. Arthritis Rheum. 1987, 30, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Glant, T.T.; Mikecz, K.; Rauch, T.A. Epigenetics in the pathogenesis of rheumatoid arthritis. BMC Med. 2014, 12, 35. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, L.I.; Daoussis, D.; Liossis, S.-N.; Bogdanos, D.P. The infectious basis of ACPA-positive rheumatoid arthritis. Front. Microbiol. 2017, 8, 1853. [Google Scholar] [CrossRef] [PubMed]
- McInnes, I.B.; Schett, G. Pathogenetic insights from the treatment of rheumatoid arthritis. Lancet 2017, 389, 2328–2337. [Google Scholar] [CrossRef]
- Kudaeva, F.M.; Speechley, M.R.; Pope, J.E. A systematic review of viral exposures as a risk for rheumatoid arthritis. Semin. Arthritis Rheum. 2018. [Google Scholar] [CrossRef] [PubMed]
- Scherer, H.U.; Huizinga, T.W.J.; Kronke, G.; Schett, G.; Toes, R.E.M. The B cell response to citrullinated antigens in the development of rheumatoid arthritis. Nat. Rev. Rheumatol. 2018, 14, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Kuijper, T.M.; Folmer, R.; Stolk, E.A.; Hazes, J.M.W.; Luime, J.J. Doctors’ preferences in de-escalating dmards in rheumatoid arthritis: A discrete choice experiment. Arthritis Res. Ther. 2017, 19, 78. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y. Rheumatoid arthritis: Dmard de-escalation—Let the patient guide you. Nat. Rev. Rheum. 2017, 13, 637–638. [Google Scholar] [CrossRef] [PubMed]
- Van Nies, J.A.; Krabben, A.; Schoones, J.W.; Huizinga, T.W.; Kloppenburg, M.; van der Helm-van Mil, A.H. What is the evidence for the presence of a therapeutic window of opportunity in rheumatoid arthritis? A systematic literature review. Ann. Rheum. Dis. 2014, 73, 861–870. [Google Scholar] [CrossRef] [PubMed]
- O’Dell, J.R. Treating rheumatoid arthritis early: A window of opportunity? Arthritis Rheum. 2002, 46, 283–285. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, A.H.; Alsahli, M.A.; Aly, S.M.; Khan, M.A.; Aldebasi, Y.H. Role of curcumin in disease prevention and treatment. Adv. Biomed. Res. 2018, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, B.B.; Sundaram, C.; Malani, N.; Ichikawa, H. Curcumin: The Indian solid gold. Adv. Exp. Med. Biol. 2007, 595, 1–75. [Google Scholar] [PubMed]
- Asher, G.N.; Spelman, K. Clinical utility of curcumin extract. Altern. Ther. Health Med. 2013, 19, 20–22. [Google Scholar] [PubMed]
- Bright, J.J. Curcumin and autoimmune disease. Adv. Exp. Med. Biol. 2007, 595, 425–451. [Google Scholar] [PubMed]
- Momtazi-Borojeni, A.A.; Haftcheshmeh, S.M.; Esmaeili, S.A.; Johnston, T.P.; Abdollahi, E.; Sahebkar, A. Curcumin: A natural modulator of immune cells in systemic lupus erythematosus. Autoimmun. Rev. 2018, 17, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Park, M.J.; Moon, S.J.; Lee, S.H.; Yang, E.J.; Min, J.K.; Cho, S.G.; Yang, C.W.; Park, S.H.; Kim, H.Y.; Cho, M.L. Curcumin attenuates acute graft-versus-host disease severity via in vivo regulations on th1, th17 and regulatory t cells. PLoS ONE 2013, 8, e67171. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Li, X.K.; Takahara, S. Curcumin has bright prospects for the treatment of multiple sclerosis. Int. Immunopharmacol. 2011, 11, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Dou, Y.; Luo, J.; Wu, X.; Wei, Z.; Tong, B.; Yu, J.; Wang, T.; Zhang, X.; Yang, Y.; Yuan, X.L.; et al. Curcumin attenuates collagen-induced inflammatory response through the “gut-brain axis”. J. Neuroinflamm. 2018, 15, 6. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wu, X.; Wei, Z.; Dou, Y.; Zhao, D.; Wang, T.; Bian, D.; Tong, B.; Xia, Y.; Xia, Y.; et al. Oral curcumin has anti-arthritic efficacy through somatostatin generation via CAMP/PKA and Ca(2+)/CaMKII signaling pathways in the small intestine. Pharmacol. Res. 2015, 95–96, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Sun, Y.; Liu, Z.; Zhang, M.; Li, C.; Cai, H. The effect of curcumin and its nanoformulation on adjuvant-induced arthritis in rats. Drug Des. Dev. Ther. 2015, 9, 4931–4942. [Google Scholar]
- Kuncha, M.; Naidu, V.G.; Sahu, B.D.; Gadepalli, S.G.; Sistla, R. Curcumin potentiates the anti-arthritic effect of prednisolone in freund’s complete adjuvant-induced arthritic rats. J. Pharm. Pharmacol. 2014, 66, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Banji, D.; Pinnapureddy, J.; Banji, O.J.; Saidulu, A.; Hayath, M.S. Synergistic activity of curcumin with methotrexate in ameliorating freund’s complete adjuvant induced arthritis with reduced hepatotoxicity in experimental animals. Eur. J. Pharmacol. 2011, 668, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Liu, T.; Wang, Q.; Liu, J. Anti-inflammatory effect of combined tetramethylpyrazine, resveratrol and curcumin in vivo. BMC Complement. Altern. Med. 2017, 17, 233. [Google Scholar] [CrossRef] [PubMed]
- Sumeet, G.; Rachna, K.; Samrat, C.; Ipshita, C.; Vikas, J.; Manu, S. Anti inflammatory and anti arthritic activity of different milk based formulation of curcumin in rat model. Curr. Drug Deliv. 2018, 15, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Jeengar, M.K.; Shrivastava, S.; Mouli Veeravalli, S.C.; Naidu, V.G.; Sistla, R. Amelioration of FCA induced arthritis on topical application of curcumin in combination with emu oil. Nutrition 2016, 32, 955–964. [Google Scholar] [CrossRef] [PubMed]
- Kamarudin, T.A.; Othman, F.; Mohd Ramli, E.S.; Md Isa, N.; Das, S. Protective effect of curcumin on experimentally induced arthritic rats: Detailed histopathological study of the joints and white blood cell count. EXCLI J. 2012, 11, 226–236. [Google Scholar] [PubMed]
- Chen, J.J.; Dai, L.; Zhao, L.X.; Zhu, X.; Cao, S.; Gao, Y.J. Intrathecal curcumin attenuates pain hypersensitivity and decreases spinal neuroinflammation in rat model of monoarthritis. Sci. Rep. 2015, 5, 10278. [Google Scholar] [CrossRef] [PubMed]
- Arora, R.; Kuhad, A.; Kaur, I.P.; Chopra, K. Curcumin loaded solid lipid nanoparticles ameliorate adjuvant-induced arthritis in rats. Eur. J. Pain 2015, 19, 940–952. [Google Scholar] [CrossRef] [PubMed]
- Funk, J.L.; Oyarzo, J.N.; Frye, J.B.; Chen, G.; Lantz, R.C.; Jolad, S.D.; Solyom, A.M.; Timmermann, B.N. Turmeric extracts containing curcuminoids prevent experimental rheumatoid arthritis. J. Nat. Prod. 2006, 69, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Tanaka, M.; Fukui, T.; Masuzawa, T. Inhibition of interleukin 17 production by curcumin in mice with collagen-induced arthritis. Biomed. Res. 2011, 22, 299–304. [Google Scholar]
- Moon, D.O.; Kim, M.O.; Choi, Y.H.; Park, Y.M.; Kim, G.Y. Curcumin attenuates inflammatory response in il-1beta-induced human synovial fibroblasts and collagen-induced arthritis in mouse model. Int. Immunopharmacol. 2010, 10, 605–610. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Xu, Z.; Huang, Y.; Duan, X.; Gong, W.; Zhang, Y.; Fan, J.; He, F. Curcumin protects against collagen-induced arthritis via suppression of baff production. J. Clin. Immunol. 2013, 33, 550–557. [Google Scholar] [CrossRef] [PubMed]
- Mun, S.H.; Kim, H.S.; Kim, J.W.; Ko, N.Y.; Kim, D.K.; Lee, B.Y.; Kim, B.; Won, H.S.; Shin, H.S.; Han, J.W.; et al. Oral administration of curcumin suppresses production of matrix metalloproteinase (MMP)-1 and MMP-3 to ameliorate collagen-induced arthritis: Inhibition of the PKCδ/JNK/c-jun pathway. J. Pharmacol. Sci. 2009, 111, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Li, X.K.; Funeshima-Fuji, N.; Kimura, H.; Matsumoto, Y.; Isaka, Y.; Takahara, S. Amelioration of experimental autoimmune encephalomyelitis by curcumin treatment through inhibition of il-17 production. Int. Immunopharmacol. 2009, 9, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Park, M.J.; Lee, S.H.; Kim, E.K.; Lee, E.J.; Baek, J.A.; Park, S.H.; Kwok, S.K.; Cho, M.L. Interleukin-10 produced by myeloid-derived suppressor cells is critical for the induction of tregs and attenuation of rheumatoid inflammation in mice. Sci. Rep. 2018, 8, 3753. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.; Chung, H.S.; Lee, K.; Lee, H.; Kim, M.; Bae, H. Curcumin attenuates the scurfy-induced immune disorder, a model of ipex syndrome, with inhibiting TH1/TH2/TH17 responses in mice. Phytomedicine 2017, 33, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Mollazadeh, H.; Cicero, A.F.G.; Blesso, C.N.; Pirro, M.; Majeed, M.; Sahebkar, A. Immune modulation by curcumin: The role of interleukin-10. Crit. Rev. Food Sci. Nutr. 2017, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Rogers, N.M.; Kireta, S.; Coates, P.T. Curcumin induces maturation-arrested dendritic cells that expand regulatory t cells in vitro and in vivo. Clin. Exp. Immunol. 2010, 162, 460–473. [Google Scholar] [CrossRef] [PubMed]
- Inglis, J.J.; Notley, C.A.; Essex, D.; Wilson, A.W.; Feldmann, M.; Anand, P.; Williams, R. Collagen-induced arthritis as a model of hyperalgesia: Functional and cellular analysis of the analgesic actions of tumor necrosis factor blockade. Arthritis Rheum. 2007, 56, 4015–4023. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M. Th17 in animal models of rheumatoid arthritis. J. Clin. Med. 2017, 6, 73. [Google Scholar] [CrossRef] [PubMed]
- Nakae, S.; Nambu, A.; Sudo, K.; Iwakura, Y. Suppression of immune induction of collagen-induced arthritis in il-17-deficient mice. J. Immunol. 2003, 171, 6173–6177. [Google Scholar] [CrossRef] [PubMed]
- Cong, Y.; Wang, L.; Konrad, A.; Schoeb, T.; Elson, C.O. Curcumin induces the tolerogenic dendritic cell that promotes differentiation of intestine-protective regulatory t cells. Eur. J. Immunol. 2009, 39, 3134–3146. [Google Scholar] [CrossRef] [PubMed]
- Wildin, R.S.; Ramsdell, F.; Peake, J.; Faravelli, F.; Casanova, J.L.; Buist, N.; Levy-Lahad, E.; Mazzella, M.; Goulet, O.; Perroni, L.; et al. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nat. Genet. 2001, 27, 18–20. [Google Scholar] [CrossRef] [PubMed]
- Bakir, B.; Yetkin Ay, Z.; Buyukbayram, H.I.; Kumbul Doguc, D.; Bayram, D.; Candan, I.A.; Uskun, E. Effect of curcumin on systemic T helper 17 cell response; gingival expressions of interleukin-17 and retinoic acid receptor-related orphan receptor gammat; and alveolar bone loss in experimental periodontitis. J. Periodontol. 2016, 87, e183–e191. [Google Scholar] [CrossRef] [PubMed]
- Hofstetter, H.H.; Toyka, K.V.; Tary-Lehmann, M.; Lehmann, P.V. Kinetics and organ distribution of il-17-producing cd4 cells in proteolipid protein 139-151 peptide-induced experimental autoimmune encephalomyelitis of SJL mice. J. Immunol. 2007, 178, 1372–1378. [Google Scholar] [CrossRef] [PubMed]
- Bruck, J.; Holstein, J.; Glocova, I.; Seidel, U.; Geisel, J.; Kanno, T.; Kumagai, J.; Mato, N.; Sudowe, S.; Widmaier, K.; et al. Nutritional control of IL-23/TH17-mediated autoimmune disease through ho-1/stat3 activation. Sci. Rep. 2017, 7, 44482. [Google Scholar] [CrossRef] [PubMed]
- Chandran, B.; Goel, A. A randomized, pilot study to assess the efficacy and safety of curcumin in patients with active rheumatoid arthritis. Phytother. Res. PTR 2012, 26, 1719–1725. [Google Scholar] [CrossRef] [PubMed]
- Amalraj, A.; Varma, K.; Jacob, J.; Divya, C.; Kunnumakkara, A.B.; Stohs, S.J.; Gopi, S. A novel highly bioavailable curcumin formulation improves symptoms and diagnostic indicators in rheumatoid arthritis patients: A randomized, double-blind, placebo-controlled, two-dose, three-arm, and parallel-group study. J. Med. Food 2017, 20, 1022–1030. [Google Scholar] [CrossRef] [PubMed]
- Zorofchian Moghadamtousi, S.; Abdul Kadir, H.; Hassandarvish, P.; Tajik, H.; Abubakar, S.; Zandi, K. A review on antibacterial, antiviral, and antifungal activity of curcumin. BioMed Res. Int. 2014, 2014, 186864. [Google Scholar] [CrossRef] [PubMed]
- Rai, D.; Singh, J.K.; Roy, N.; Panda, D. Curcumin inhibits FTsZ assembly: An attractive mechanism for its antibacterial activity. Biochem. J. 2008, 410, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; De, R.; Mukhopadhyay, A.K. Curcumin as a potential therapeutic candidate for helicobacter pylori associated diseases. World J. Gastroenterol. 2016, 22, 2736–2748. [Google Scholar] [CrossRef] [PubMed]
- De, R.; Kundu, P.; Swarnakar, S.; Ramamurthy, T.; Chowdhury, A.; Nair, G.B.; Mukhopadhyay, A.K. Antimicrobial activity of curcumin against helicobacter pylori isolates from India and during infections in mice. Antimicrob. Agents Chemother. 2009, 53, 1592–1597. [Google Scholar] [CrossRef] [PubMed]
- Zandi, K.; Ramedani, E.; Mohammadi, K.; Tajbakhsh, S.; Deilami, I.; Rastian, Z.; Fouladvand, M.; Yousefi, F.; Farshadpour, F. Evaluation of antiviral activities of curcumin derivatives against HSV-1 in vero cell line. Nat. Prod. Commun. 2010, 5, 1935–1938. [Google Scholar] [PubMed]
- Ferreira, V.H.; Nazli, A.; Dizzell, S.E.; Mueller, K.; Kaushic, C. The anti-inflammatory activity of curcumin protects the genital mucosal epithelial barrier from disruption and blocks replication of HIV-1 and HSV-2. PLoS ONE 2015, 10, e0124903. [Google Scholar] [CrossRef] [PubMed]
- Kutluay, S.B.; Doroghazi, J.; Roemer, M.E.; Triezenberg, S.J. Curcumin inhibits herpes simplex virus immediate-early gene expression by a mechanism independent of p300/CBP histone acetyltransferase activity. Virology 2008, 373, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Dhamgaye, S.; Maurya, I.K.; Singh, A.; Sharma, M.; Prasad, R. Curcumin targets cell wall integrity via calcineurin-mediated signaling in Candida albicans. Antimicrob. Agents Chem. 2014, 58, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Goel, A.; Kunnumakkara, A.B.; Aggarwal, B.B. Curcumin as “curecumin”: From kitchen to clinic. Biochem. Pharmacol. 2008, 75, 787–809. [Google Scholar] [CrossRef] [PubMed]
- Bogdanos, D.P.; Sakkas, L.I. From microbiome to infectome in autoimmunity. Curr. Opin. Rheumatol. 2017, 29, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Willemze, A.; Trouw, L.A.; Toes, R.E.; Huizinga, T.W. The influence of ACPA status and characteristics on the course of RA. Nat. Res. Rheumatol. 2012, 8, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, L.I.; Bogdanos, D.P.; Katsiari, C.; Platsoucas, C.D. Anti-citrullinated peptides as autoantigens in rheumatoid arthritis-relevance to treatment. Autoimmun. Res. 2014, 13, 1114–1120. [Google Scholar] [CrossRef] [PubMed]
- Van Venrooij, W.J.; van Beers, J.J.; Pruijn, G.J. Anti-CCP antibodies: The past, the present and the future. Nat. Res. Rheumatol. 2011, 7, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Jilani, A.A.; Mackworth-Young, C.G. The role of citrullinated protein antibodies in predicting erosive disease in rheumatoid arthritis: A systematic literature review and meta-analysis. Int. J. Rheumatol. 2015, 2015, 728610. [Google Scholar] [CrossRef] [PubMed]
- Syversen, S.W.; Gaarder, P.I.; Goll, G.L.; Odegard, S.; Haavardsholm, E.A.; Mowinckel, P.; van der Heijde, D.; Landewe, R.; Kvien, T.K. High anti-cyclic citrullinated peptide levels and an algorithm of four variables predict radiographic progression in patients with rheumatoid arthritis: Results from a 10-year longitudinal study. Ann. Rheum. Dis. 2008, 67, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Nielen, M.M.; van Schaardenburg, D.; Reesink, H.W.; van de Stadt, R.J.; van der Horst-Bruinsma, I.E.; de Koning, M.H.; Habibuw, M.R.; Vandenbroucke, J.P.; Dijkmans, B.A. Specific autoantibodies precede the symptoms of rheumatoid arthritis: A study of serial measurements in blood donors. Arthritis Rheum. 2004, 50, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Van der Woude, D.; Rantapaa-Dahlqvist, S.; Ioan-Facsinay, A.; Onnekink, C.; Schwarte, C.M.; Verpoort, K.N.; Drijfhout, J.W.; Huizinga, T.W.; Toes, R.E.; Pruijn, G.J. Epitope spreading of the anti-citrullinated protein antibody response occurs before disease onset and is associated with the disease course of early arthritis. Ann. Rheum. Dis. 2010, 69, 1554–1561. [Google Scholar] [CrossRef] [PubMed]
- Brink, M.; Hansson, M.; Mathsson, L.; Jakobsson, P.J.; Holmdahl, R.; Hallmans, G.; Stenlund, H.; Ronnelid, J.; Klareskog, L.; Rantapaa-Dahlqvist, S. Multiplex analyses of antibodies against citrullinated peptides in individuals prior to development of rheumatoid arthritis. Arthritis Rheum. 2013, 65, 899–910. [Google Scholar] [CrossRef] [PubMed]
- Koga, T.; Okada, A.; Fukuda, T.; Hidaka, T.; Ishii, T.; Ueki, Y.; Kodera, T.; Nakashima, M.; Takahashi, Y.; Honda, S.; et al. Anti-citrullinated peptide antibodies are the strongest predictor of clinically relevant radiographic progression in rheumatoid arthritis patients achieving remission or low disease activity: A post hoc analysis of a nationwide cohort in japan. PLoS ONE 2017, 12, e0175281. [Google Scholar] [CrossRef] [PubMed]
- Alivernini, S.; Galeazzi, M.; Peleg, H.; Tolusso, B.; Gremese, E.; Ferraccioli, G.; Naparstek, Y. Is ACPA positivity the main driver for rheumatoid arthritis treatment? Pros and cons. Autoimmun. Rev. 2017, 16, 1096–1102. [Google Scholar] [CrossRef] [PubMed]
- McHugh, J. Rheumatoid arthritis: IFNγ drives synovial tissue remodelling. Nat. Rev. Rheumatol. 2018, 14, 63. [Google Scholar] [CrossRef] [PubMed]
- Sokolove, J.; Bromberg, R.; Deane, K.D.; Lahey, L.J.; Derber, L.A.; Chandra, P.E.; Edison, J.D.; Gilliland, W.R.; Tibshirani, R.J.; Norris, J.M.; et al. Autoantibody epitope spreading in the pre-clinical phase predicts progression to rheumatoid arthritis. PLoS ONE 2012, 7, e35296. [Google Scholar] [CrossRef]
- England, B.R.; Thiele, G.M.; Mikuls, T.R. Anticitrullinated protein antibodies: Origin and role in the pathogenesis of rheumatoid arthritis. Curr. Opin. Rheumatol. 2017, 29, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Foulquier, C.; Sebbag, M.; Clavel, C.; Chapuy-Regaud, S.; Al Badine, R.; Mechin, M.C.; Vincent, C.; Nachat, R.; Yamada, M.; Takahara, H.; et al. Peptidyl arginine deiminase type 2 (PAD-2) and PAD-4 but not PAD-1, PAD-3, and PAD-6 are expressed in rheumatoid arthritis synovium in close association with tissue inflammation. Arthritis Rheum. 2007, 56, 3541–3553. [Google Scholar] [CrossRef] [PubMed]
- Anzilotti, C.; Pratesi, F.; Tommasi, C.; Migliorini, P. Peptidylarginine deiminase 4 and citrullination in health and disease. Autoimmun. Rev. 2010, 9, 158–160. [Google Scholar] [CrossRef] [PubMed]
- Kidd, B.A.; Ho, P.P.; Sharpe, O.; Zhao, X.; Tomooka, B.H.; Kanter, J.L.; Steinman, L.; Robinson, W.H. Epitope spreading to citrullinated antigens in mouse models of autoimmune arthritis and demyelination. Arthritis Res. Ther. 2008, 10, R119. [Google Scholar] [CrossRef] [PubMed]
- Konig, M.F.; Paracha, A.S.; Moni, M.; Bingham, C.O., 3rd; Andrade, F. Defining the role of Porphyromonas gingivalis peptidylarginine deiminase (PPAD) in rheumatoid arthritis through the study of PPAD biology. Ann. Rheum. Dis. 2015, 74, 2054–2061. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.A.; Southwood, S.; Sette, A.; Jevnikar, A.M.; Bell, D.A.; Cairns, E. Cutting edge: The conversion of arginine to citrulline allows for a high-affinity peptide interaction with the rheumatoid arthritis-associated HLA-DRB1*0401 MHC class II molecule. J. Immunol. 2003, 171, 538–541. [Google Scholar] [CrossRef] [PubMed]
- Scally, S.W.; Petersen, J.; Law, S.C.; Dudek, N.L.; Nel, H.J.; Loh, K.L.; Wijeyewickrema, L.C.; Eckle, S.B.; van Heemst, J.; Pike, R.N.; et al. A molecular basis for the association of the HLA-DRB1 locus, citrullination, and rheumatoid arthritis. J. Exp. Med. 2013, 210, 2569–2582. [Google Scholar] [CrossRef] [PubMed]
- Arnoux, F.; Mariot, C.; Peen, E.; Lambert, N.C.; Balandraud, N.; Roudier, J.; Auger, I. Peptidyl arginine deiminase immunization induces anticitrullinated protein antibodies in mice with particular MHC types. Proc. Natl. Acad. Sci. USA 2017, 114, E10169–E10177. [Google Scholar] [CrossRef] [PubMed]
- Leech, T.; Bartold, P.M. The association between rheumatoid arthritis and periodontitis. Best Pract. Res. Clin. Rheumatol. 2015, 29, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Yusof, Z.; Porter, S.R.; Greenman, J.; Scully, C. Levels of serum igg against Porphyromonas gingivalis in patients with rapidly progressive periodontitis, rheumatoid arthritis and adult periodontitis. J. Nihon Univ. Sch. Dent. 1995, 37, 197–200. [Google Scholar] [CrossRef] [PubMed]
- Koziel, J.; Mydel, P.; Potempa, J. The link between periodontal disease and rheumatoid arthritis: An updated review. Curr. Rheumatol. Rep. 2014, 16, 408. [Google Scholar] [CrossRef] [PubMed]
- Wegner, N.; Lundberg, K.; Kinloch, A.; Fisher, B.; Malmstrom, V.; Feldmann, M.; Venables, P.J. Autoimmunity to specific citrullinated proteins gives the first clues to the etiology of rheumatoid arthritis. Immunol. Rev. 2010, 233, 34–54. [Google Scholar] [CrossRef] [PubMed]
- Janssen, K.M.; Vissink, A.; de Smit, M.J.; Westra, J.; Brouwer, E. Lessons to be learned from periodontitis. Curr. Opin. Rheumatol. 2013, 25, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Yamazaki, T.; Hamaguchi, M.; Morimoto, T.; Yamori, M.; Asai, K.; Isobe, Y.; Furu, M.; Ito, H.; Fujii, T.; et al. Periodontitis and Porphyromonas gingivalis in preclinical stage of arthritis patients. PLoS ONE 2015, 10, e0122121. [Google Scholar] [CrossRef] [PubMed]
- Johansson, L.; Sherina, N.; Kharlamova, N.; Potempa, B.; Larsson, B.; Israelsson, L.; Potempa, J.; Rantapaa-Dahlqvist, S.; Lundberg, K. Concentration of antibodies against Porphyromonas gingivalis is increased before the onset of symptoms of rheumatoid arthritis. Arthritis Res. Ther. 2016, 18, 201. [Google Scholar] [CrossRef] [PubMed]
- Lange, L.; Thiele, G.M.; McCracken, C.; Wang, G.; Ponder, L.A.; Angeles-Han, S.T.; Rouster-Stevens, K.A.; Hersh, A.O.; Vogler, L.B.; Bohnsack, J.F.; et al. Symptoms of periodontitis and antibody responses to Porphyromonas gingivalis in juvenile idiopathic arthritis. Pediatr. Rheumatol. Online J. 2016, 14, 8. [Google Scholar] [CrossRef] [PubMed]
- Kharlamova, N.; Jiang, X.; Sherina, N.; Potempa, B.; Israelsson, L.; Quirke, A.M.; Eriksson, K.; Yucel-Lindberg, T.; Venables, P.J.; Potempa, J.; et al. Antibodies to Porphyromonas gingivalis indicate interaction between oral infection, smoking, and risk genes in rheumatoid arthritis etiology. Arthritis Rheumatol. 2016, 68, 604–613. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, K.; Kinloch, A.; Fisher, B.A.; Wegner, N.; Wait, R.; Charles, P.; Mikuls, T.R.; Venables, P.J. Antibodies to citrullinated alpha-enolase peptide 1 are specific for rheumatoid arthritis and cross-react with bacterial enolase. Arthritis Rheum. 2008, 58, 3009–3019. [Google Scholar] [CrossRef] [PubMed]
- Maresz, K.J.; Hellvard, A.; Sroka, A.; Adamowicz, K.; Bielecka, E.; Koziel, J.; Gawron, K.; Mizgalska, D.; Marcinska, K.A.; Benedyk, M.; et al. Porphyromonas gingivalis facilitates the development and progression of destructive arthritis through its unique bacterial peptidylarginine deiminase (PAD). PLoS Pathog. 2013, 9, e1003627. [Google Scholar] [CrossRef] [PubMed]
- Sandal, I.; Karydis, A.; Luo, J.; Prislovsky, A.; Whittington, K.B.; Rosloniec, E.F.; Dong, C.; Novack, D.V.; Mydel, P.; Zheng, S.G.; et al. Bone loss and aggravated autoimmune arthritis in HLA-DRβ1-bearing humanized mice following oral challenge with Porphyromonas gingivalis. Arthritis Res. Ther. 2016, 18, 249. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, M.; Arimatsu, K.; Kato, T.; Matsuda, Y.; Minagawa, T.; Takahashi, N.; Ohno, H.; Yamazaki, K. Oral administration of P. gingivalis induces dysbiosis of gut microbiota and impaired barrier function leading to dissemination of enterobacteria to the liver. PLoS ONE 2015, 10, e0134234. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Takahashi, N.; Kato, T.; Matsuda, Y.; Yokoji, M.; Yamada, M.; Nakajima, T.; Kondo, N.; Endo, N.; Yamamoto, R.; et al. Aggravation of collagen-induced arthritis by orally administered Porphyromonas gingivalis through modulation of the gut microbiota and gut immune system. Sci. Rep. 2017, 7, 6955. [Google Scholar] [CrossRef] [PubMed]
- Konig, M.F.; Abusleme, L.; Reinholdt, J.; Palmer, R.J.; Teles, R.P.; Sampson, K.; Rosen, A.; Nigrovic, P.A.; Sokolove, J.; Giles, J.T.; et al. Aggregatibacter actinomycetemcomitans-induced hypercitrullination links periodontal infection to autoimmunity in rheumatoid arthritis. Sci. Transl. Med. 2016, 8, 369ra176. [Google Scholar] [CrossRef] [PubMed]
- Pratesi, F.; Tommasi, C.; Anzilotti, C.; Puxeddu, I.; Sardano, E.; Di Colo, G.; Migliorini, P. Antibodies to a new viral citrullinated peptide, VCP2: Fine specificity and correlation with anti-cyclic citrullinated peptide (CCP) and anti-VCP1 antibodies. Clin. Exp. Immunol. 2011, 164, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Li, F.; Wong, M.C.; Feng, X.P.; Lu, H.X.; Xu, W. Association between herpesviruses and chronic periodontitis: A meta-analysis based on case-control studies. PLoS ONE 2015, 10, e0144319. [Google Scholar] [CrossRef] [PubMed]
- Scher, J.U.; Ubeda, C.; Equinda, M.; Khanin, R.; Buischi, Y.; Viale, A.; Lipuma, L.; Attur, M.; Pillinger, M.H.; Weissmann, G.; et al. Periodontal disease and the oral microbiota in new-onset rheumatoid arthritis. Arthritis Rheum. 2012, 64, 3083–3094. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, D.; Jia, H.; Feng, Q.; Wang, D.; Liang, D.; Wu, X.; Li, J.; Tang, L.; Li, Y.; et al. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat. Med. 2015, 21, 895–905. [Google Scholar] [CrossRef] [PubMed]
- Arimatsu, K.; Yamada, H.; Miyazawa, H.; Minagawa, T.; Nakajima, M.; Ryder, M.I.; Gotoh, K.; Motooka, D.; Nakamura, S.; Iida, T.; et al. Oral pathobiont induces systemic inflammation and metabolic changes associated with alteration of gut microbiota. Sci. Rep. 2014, 4, 4828. [Google Scholar] [CrossRef] [PubMed]
- Totaro, M.C.; Cattani, P.; Ria, F.; Tolusso, B.; Gremese, E.; Fedele, A.L.; D’Onghia, S.; Marchetti, S.; Di Sante, G.; Canestri, S.; et al. Porphyromonas gingivalis and the pathogenesis of rheumatoid arthritis: Analysis of various compartments including the synovial tissue. Arthritis Res. Ther. 2013, 15, R66. [Google Scholar] [CrossRef] [PubMed]
- Nonnenmacher, C.; Dalpke, A.; Zimmermann, S.; Flores-De-Jacoby, L.; Mutters, R.; Heeg, K. DNA from periodontopathogenic bacteria is immunostimulatory for mouse and human immune cells. Infect. Immun. 2003, 71, 850–856. [Google Scholar] [CrossRef] [PubMed]
- Klareskog, L.; Stolt, P.; Lundberg, K.; Kallberg, H.; Bengtsson, C.; Grunewald, J.; Ronnelid, J.; Harris, H.E.; Ulfgren, A.K.; Rantapaa-Dahlqvist, S.; et al. A new model for an etiology of rheumatoid arthritis: Smoking may trigger HLA-DR (shared epitope)-restricted immune reactions to autoantigens modified by citrullination. Arthritis Rheum. 2006, 54, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Mikuls, T.R.; Payne, J.B.; Yu, F.; Thiele, G.M.; Reynolds, R.J.; Cannon, G.W.; Markt, J.; McGowan, D.; Kerr, G.S.; Redman, R.S.; et al. Periodontitis and Porphyromonas gingivalis in patients with rheumatoid arthritis. Arthritis Rheumatol. 2014, 66, 1090–1100. [Google Scholar] [CrossRef] [PubMed]
- Fuggle, N.R.; Smith, T.O.; Kaul, A.; Sofat, N. Hand to mouth: A systematic review and meta-analysis of the association between rheumatoid arthritis and periodontitis. Front. Immunol. 2016, 7, 80. [Google Scholar] [CrossRef] [PubMed]
- Hensvold, A.H.; Magnusson, P.K.; Joshua, V.; Hansson, M.; Israelsson, L.; Ferreira, R.; Jakobsson, P.J.; Holmdahl, R.; Hammarstrom, L.; Malmstrom, V.; et al. Environmental and genetic factors in the development of anticitrullinated protein antibodies (ACPAs) and ACPA-positive rheumatoid arthritis: An epidemiological investigation in twins. Ann. Rheum. Dis. 2015, 74, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Petersen, P.E.; Ogawa, H. Strengthening the prevention of periodontal disease: The WHO approach. J. Periodontol. 2005, 76, 2187–2193. [Google Scholar] [CrossRef] [PubMed]
- Teles, R.; Teles, F.; Frias-Lopez, J.; Paster, B.; Haffajee, A. Lessons learned and unlearned in periodontal microbiology. Periodontology 2000 2013, 62, 95–162. [Google Scholar] [CrossRef] [PubMed]
- Ardila, C.M.; Granada, M.I.; Guzman, I.C. Antibiotic resistance of subgingival species in chronic periodontitis patients. J. Periodontal Res. 2010, 45, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Petti, S.; Scully, C. Polyphenols, oral health and disease: A review. J. Dent. 2009, 37, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Hensvold, A.H.; Frisell, T.; Magnusson, P.K.; Holmdahl, R.; Askling, J.; Catrina, A.I. How well do ACPA discriminate and predict RA in the general population: A study based on 12 590 population-representative swedish twins. Ann. Rheum. Dis. 2017, 76, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Lappin, D.F.; Apatzidou, D.; Quirke, A.M.; Oliver-Bell, J.; Butcher, J.P.; Kinane, D.F.; Riggio, M.P.; Venables, P.; McInnes, I.B.; Culshaw, S. Influence of periodontal disease, Porphyromonas gingivalis and cigarette smoking on systemic anti-citrullinated peptide antibody titres. J. Clin. Periodontol. 2013, 40, 907–915. [Google Scholar] [CrossRef] [PubMed]
- Marchesan, J.T.; Gerow, E.A.; Schaff, R.; Taut, A.D.; Shin, S.Y.; Sugai, J.; Brand, D.; Burberry, A.; Jorns, J.; Lundy, S.K.; et al. Porphyromonas gingivalis oral infection exacerbates the development and severity of collagen-induced arthritis. Arthritis Res. Ther. 2013, 15, R186. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Nie, M.; Fan, M.W.; Bian, Z. Anti-inflammatory activity of curcumin in macrophages stimulated by lipopolysaccharides from Porphyromonas gingivalis. Pharmacology 2008, 82, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Chen, D.; Li, Q.; Sun, X.; Song, Y.; Wang, C. Curcumin inhibits inflammatory response and bone loss during experimental periodontitis in rats. Acta Odontol. Scand. 2013, 71, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.; Huang, P.; Chen, M.W. Curcumin attenuates cyclooxygenase-2 expression via inhibition of the nf-kappab pathway in lipopolysaccharide-stimulated human gingival fibroblasts. Cell Biol. Int. 2013, 37, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Gottumukkala, S.N.; Koneru, S.; Mannem, S.; Mandalapu, N. Effectiveness of sub gingival irrigation of an indigenous 1% curcumin solution on clinical and microbiological parameters in chronic periodontitis patients: A pilot randomized clinical trial. Cont. Clin. Dent. 2013, 4, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Lee, H.M.; Napolitano, N.; Clemens, M.; Zhang, Y.; Sorsa, T.; Zhang, Y.; Johnson, F.; Golub, L.M. 4-methoxycarbonyl curcumin: A unique inhibitor of both inflammatory mediators and periodontal inflammation. Med. Inflamm. 2013, 2013, 329740. [Google Scholar] [CrossRef] [PubMed]
- Anuradha, B.R.; Bai, Y.D.; Sailaja, S.; Sudhakar, J.; Priyanka, M.; Deepika, V. Evaluation of anti-inflammatory effects of curcumin gel as an adjunct to scaling and root planing: A clinical study. J. Int. Oral Health JIOH 2015, 7, 90–93. [Google Scholar] [PubMed]
- Nagasri, M.; Madhulatha, M.; Musalaiah, S.V.; Kumar, P.A.; Krishna, C.H.; Kumar, P.M. Efficacy of curcumin as an adjunct to scaling and root planning in chronic periodontitis patients: A clinical and microbiological study. J. Pharm. Bioallied Sci. 2015, 7, S554–S558. [Google Scholar] [CrossRef] [PubMed]
- Anitha, V.; Rajesh, P.; Shanmugam, M.; Priya, B.M.; Prabhu, S.; Shivakumar, V. Comparative evaluation of natural curcumin and synthetic chlorhexidine in the management of chronic periodontitis as a local drug delivery: A clinical and microbiological study. Indian J. Dent. Res. 2015, 26, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Sreedhar, A.; Sarkar, I.; Rajan, P.; Pai, J.; Malagi, S.; Kamath, V.; Barmappa, R. Comparative evaluation of the efficacy of curcumin gel with and without photo activation as an adjunct to scaling and root planing in the treatment of chronic periodontitis: A split mouth clinical and microbiological study. J. Nat. Sci. Biol. Med. 2015, 6, 1021. [Google Scholar] [CrossRef] [PubMed]
- Hugar, S.S.; Patil, S.; Metgud, R.; Nanjwade, B.; Hugar, S.M. Influence of application of chlorhexidine gel and curcumin gel as an adjunct to scaling and root planing: A interventional study. J. Nat. Sci. Biol. Med. 2016, 7, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Elavarasu, S.; Suthanthiran, T.; Thangavelu, A.; Alex, S.; Palanisamy, V.K.; Kumar, T.S. Evaluation of superoxide dismutase levels in local drug delivery system containing 0.2% curcumin strip as an adjunct to scaling and root planing in chronic periodontitis: A clinical and biochemical study. J. Pharm. Bioallied Sci. 2016, 8, S48–S52. [Google Scholar] [PubMed]
- Kaur, H.; Grover, V.; Malhotra, R.; Gupta, M. Evaluation of curcumin gel as adjunct to scaling & root planing in management of periodontitis-randomized clinical & biochemical investigation. Inf. Disord. Drug Targets 2018. [Google Scholar] [CrossRef]
- Ravishankar, P.L.; Kumar, Y.P.; Anila, E.N.; Chakraborty, P.; Malakar, M.; Mahalakshmi, R. Effect of local application of curcumin and ornidazole gel in chronic periodontitis patients. Int. J. Pharm. Investig. 2017, 7, 188–192. [Google Scholar] [CrossRef] [PubMed]
- Izui, S.; Sekine, S.; Maeda, K.; Kuboniwa, M.; Takada, A.; Amano, A.; Nagata, H. Antibacterial activity of curcumin against periodontopathic bacteria. J. Periodontol. 2016, 87, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Bimonte, S.; Barbieri, A.; Palma, G.; Luciano, A.; Rea, D.; Arra, C. Curcumin inhibits tumor growth and angiogenesis in an orthotopic mouse model of human pancreatic cancer. Biomed. Res. Int. 2013, 2013, 810423. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, M.; Millhouse, E.; Culshaw, S.; Edwards, C.A.; Ramage, G.; Combet, E. Selected dietary (poly)phenols inhibit periodontal pathogen growth and biofilm formation. Food Funct. 2015, 6, 719–729. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Naksuriya, O.; Okonogi, S.; Schiffelers, R.M.; Hennink, W.E. Curcumin nanoformulations: A review of pharmaceutical properties and preclinical studies and clinical data related to cancer treatment. Biomaterials 2014, 35, 3365–3383. [Google Scholar] [CrossRef] [PubMed]
- Joung, H.J.; Choi, M.J.; Kim, J.T.; Park, S.H.; Park, H.J.; Shin, G.H. Development of food-grade curcumin nanoemulsion and its potential application to food beverage system: Antioxidant property and in vitro digestion. J. Food Sci. 2016, 81, N745–N753. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).