Psychiatric Comorbidity in Children and Adults with Gluten-Related Disorders: A Narrative Review
Abstract
1. Introduction
2. Methodology
2.1. Search Strategy
2.2. Inclusion Criteria
2.3. Data Extraction
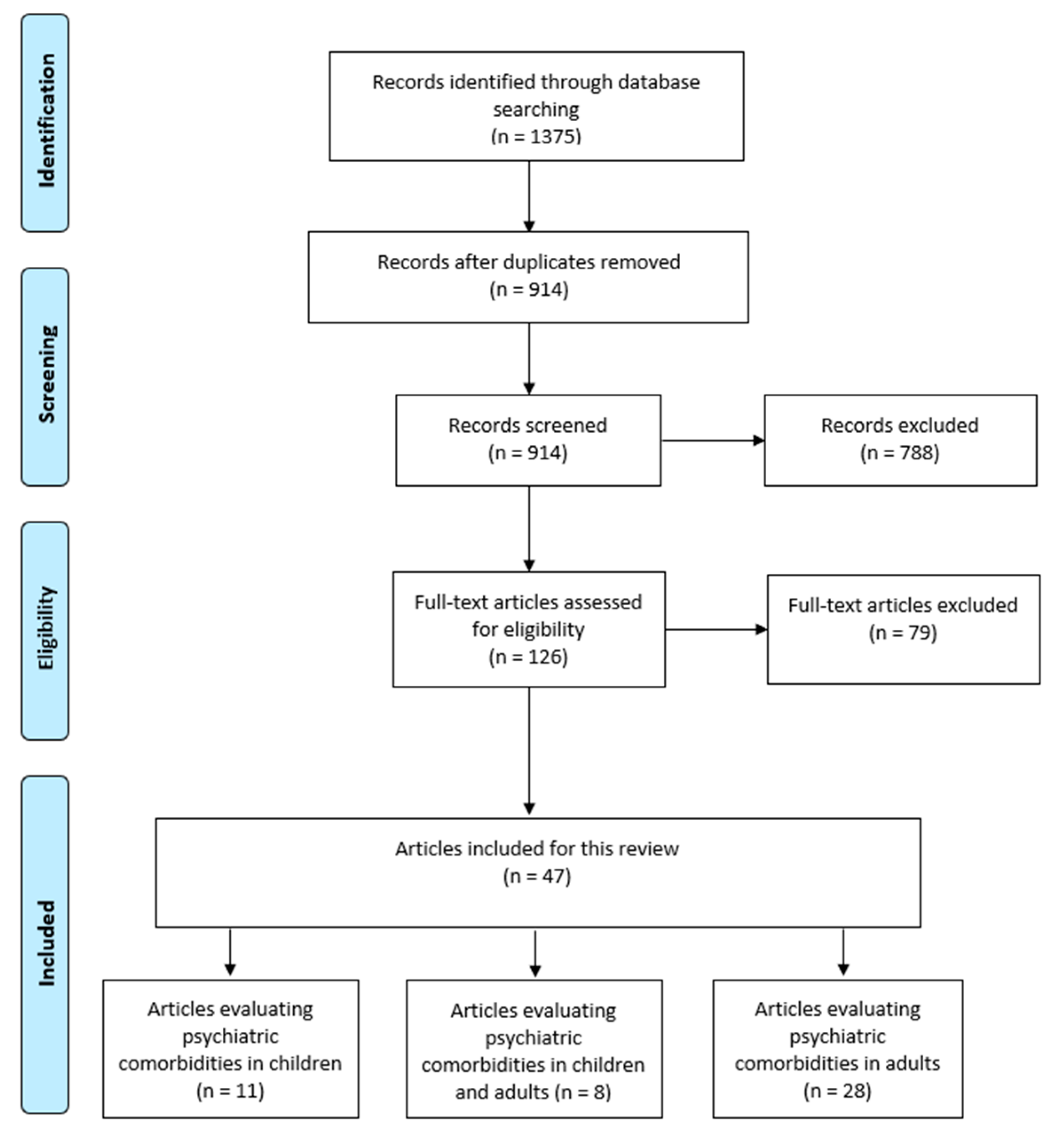
3. Results
3.1. Study Selection
3.2. Studies Conducted in Children with CD
3.3. Studies Conducted in Adults with CD
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sapone, A.; Bai, J.C.; Ciacci, C.; Dolinsek, J.; Green, P.H.; Hadjivassiliou, M.; Kaukinen, K.; Rostami, K.; Sanders, D.S.; Schumann, M.; et al. Spectrum of gluten-related disorders: Consensus on new nomenclature and classification. BMC Med. 2012, 10, 13. [Google Scholar] [CrossRef] [PubMed]
- Guandalini, S.; Assiri, A. Celiac disease: A review. JAMA Pediatr. 2014, 168, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Green, P.H.; Lebwohl, B.; Greywoode, R. Celiac disease. J. Allergy Clin. Immunol. 2015, 135, 1099–1106, quiz 1107. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Bai, J.C.; Biagi, F.; Card, T.R.; Ciacci, C.; Ciclitira, P.J.; Green, P.H.; Hadjivassiliou, M.; Holdoway, A.; van Heel, D.A.; et al. Diagnosis and management of adult coeliac disease: Guidelines from the british society of gastroenterology. Gut 2014, 63, 1210–1228. [Google Scholar] [CrossRef] [PubMed]
- Uibo, R.; Tian, Z.; Gershwin, M.E. Celiac disease: A model disease for gene-environment interaction. Cell. Mol. Immunol. 2011, 8, 93–95. [Google Scholar] [CrossRef] [PubMed]
- Szajewska, H.; Shamir, R.; Chmielewska, A.; Piescik-Lech, M.; Auricchio, R.; Ivarsson, A.; Kolacek, S.; Koletzko, S.; Korponay-Szabo, I.; Mearin, M.L.; et al. Systematic review with meta-analysis: Early infant feeding and coeliac disease—Update 2015. Aliment. Pharmacol. Ther. 2015, 41, 1038–1054. [Google Scholar] [CrossRef] [PubMed]
- Canova, C.; Zabeo, V.; Pitter, G.; Romor, P.; Baldovin, T.; Zanotti, R.; Simonato, L. Association of maternal education, early infections, and antibiotic use with celiac disease: A population-based birth cohort study in northeastern italy. Am. J. Epidemiol. 2014, 180, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Olivares, M.; Neef, A.; Castillejo, G.; Palma, G.D.; Varea, V.; Capilla, A.; Palau, F.; Nova, E.; Marcos, A.; Polanco, I.; et al. The hla-dq2 genotype selects for early intestinal microbiota composition in infants at high risk of developing coeliac disease. Gut 2015, 64, 406–417. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Gatti, S.; Lionetti, E. World perspective and celiac disease epidemiology. Dig. Dis. 2015, 33, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Arora, A.; Strand, T.A.; Leffler, D.A.; Catassi, C.; Green, P.H.; Kelly, C.P.; Ahuja, V.; Makharia, G.K. Global prevalence of celiac disease: Systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 2018, 16, 823–836.e2. [Google Scholar] [CrossRef] [PubMed]
- Lauret, E.; Rodrigo, L. Celiac disease and autoimmune-associated conditions. BioMed Res. Int. 2013, 2013, 127589. [Google Scholar] [CrossRef] [PubMed]
- Assa, A.; Frenkel-Nir, Y.; Tzur, D.; Katz, L.H.; Shamir, R. Large population study shows that adolescents with celiac disease have an increased risk of multiple autoimmune and nonautoimmune comorbidities. Acta Paediatr. 2017, 106, 967–972. [Google Scholar] [CrossRef] [PubMed]
- Viljamaa, M.; Kaukinen, K.; Pukkala, E.; Hervonen, K.; Reunala, T.; Collin, P. Malignancies and mortality in patients with coeliac disease and dermatitis herpetiformis: 30-year population-based study. Dig. Liver Dis. 2006, 38, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Ilus, T.; Kaukinen, K.; Virta, L.J.; Pukkala, E.; Collin, P. Incidence of malignancies in diagnosed celiac patients: A population-based estimate. Am. J. Gastroenterol. 2014, 109, 1471–1477. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.C.; Fried, M.; Corazza, G.R.; Schuppan, D.; Farthing, M.; Catassi, C.; Greco, L.; Cohen, H.; Ciacci, C.; Eliakim, R.; et al. World gastroenterology organisation global guidelines on celiac disease. J. Clin. Gastroenterol. 2013, 47, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Pavlovic, M.; Berenji, K.; Bukurov, M. Screening of celiac disease in down syndrome—Old and new dilemmas. World J. Clin. Cases 2017, 5, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, H.R.; Murch, S.H.; Beattie, R.M.; Coeliac Disease Working Group of British Society of Paediatric Gastroenterology, Hepatology and Nutrition. Diagnosing coeliac disease. Arch. Dis. Child. 2012, 97, 393–394. [Google Scholar] [CrossRef] [PubMed]
- Elli, L.; Branchi, F.; Tomba, C.; Villalta, D.; Norsa, L.; Ferretti, F.; Roncoroni, L.; Bardella, M.T. Diagnosis of gluten related disorders: Celiac disease, wheat allergy and non-celiac gluten sensitivity. World J. Gastroenterol. 2015, 21, 7110–7119. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Elli, L.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; Cellier, C.; Cristofori, F.; de Magistris, L.; Dolinsek, J.; et al. Diagnosis of non-celiac gluten sensitivity (ncgs): The salerno experts’ criteria. Nutrients 2015, 7, 4966–4977. [Google Scholar] [CrossRef] [PubMed]
- Nijeboer, P.; Bontkes, H.J.; Mulder, C.J.; Bouma, G. Non-celiac gluten sensitivity. Is it in the gluten or the grain? J. Gastrointest. Liver Dis. 2013, 22, 435–440. [Google Scholar]
- Cianferoni, A. Wheat allergy: Diagnosis and management. J. Asthma Allergy 2016, 9, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Pietzak, M. Celiac disease, wheat allergy, and gluten sensitivity: When gluten free is not a fad. JPEN J. Parenter. Enter. Nutr. 2012, 36, 68s–75s. [Google Scholar] [CrossRef] [PubMed]
- Jackson, J.R.; Eaton, W.W.; Cascella, N.G.; Fasano, A.; Kelly, D.L. Neurologic and psychiatric manifestations of celiac disease and gluten sensitivity. Psychiatr. Q. 2012, 83, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Zingone, F.; Swift, G.L.; Card, T.R.; Sanders, D.S.; Ludvigsson, J.F.; Bai, J.C. Psychological morbidity of celiac disease: A review of the literature. United Eur. Gastroenterol. J. 2015, 3, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Porcelli, B.; Verdino, V.; Bossini, L.; Terzuoli, L.; Fagiolini, A. Celiac and non-celiac gluten sensitivity: A review on the association with schizophrenia and mood disorders. Auto Immun. Highlights 2014, 5, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Cossu, G.; Carta, M.G.; Contu, F.; Mela, Q.; Demelia, L.; Elli, L.; Dell’Osso, B. Coeliac disease and psychiatric comorbidity: Epidemiology, pathophysiological mechanisms, quality-of-life, and gluten-free diet effects. Int. Rev. Psychiatry 2017, 29, 489–503. [Google Scholar] [CrossRef] [PubMed]
- Brietzke, E.; Cerqueira, R.O.; Mansur, R.B.; McIntyre, R.S. Gluten related illnesses and severe mental disorders: A comprehensive review. Neurosci. Biobehav. Rev. 2018, 84, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Da Silva Kotze, L.M.; David Paiva, A.D.; Roberto Kotze, L. Emotional disturbances in children and adolescents with celiac disease. Rev. Bras. Med. Psicossom. 2000, 4, 9–15. [Google Scholar]
- Horvath-Stolarczyk, A.; Sidor, K.; Dziechciarz, P.; Siemińska, J. Assessment of emotional status, selected personality traits and depression in young adults with celiac disease. Pediatr. Wspolcz. 2002, 4, 241–246. [Google Scholar]
- Pavone, L.; Fiumara, A.; Bottaro, G.; Mazzone, D.; Coleman, M. Autism and celiac disease: Failure to validate the hypothesis that a link might exist. Biol. Psychiatry 1997, 42, 72–75. [Google Scholar] [CrossRef]
- Ludvigsson, J.F.; Osby, U.; Ekbom, A.; Montgomery, S.M. Coeliac disease and risk of schizophrenia and other psychosis: A general population cohort study. Scand. J. Gastroenterol. 2007, 42, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Pynnönen, P.A.; Isometsä, E.T.; Aronen, E.T.; Verkasalo, M.A.; Savilahti, E.; Aalberg, V.A. Mental disorders in adolescents with celiac disease. Psychosomatics 2004, 45, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Accomando, S.; Fragapane, M.L.; Montaperto, D.; Trizzino, A.; Amato, G.M.; Calderone, F.; Accomando, I. Coeliac disease and depression: Two related entities? Dig. Liver Dis. 2005, 37, 298–299. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Reutfors, J.; Osby, U.; Ekbom, A.; Montgomery, S.M. Coeliac disease and risk of mood disorders—A general population-based cohort study. J. Affect. Disord. 2007, 99, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Fidan, T.; Ertekin, V.; Karabag, K. Depression-anxiety levels and the quality of life among children and adolescents with coeliac disease. Dusunen Adam 2013, 26, 232–238. [Google Scholar] [CrossRef]
- Esenyel, S.; Unal, F.; Vural, P. Depression and anxiety in child and adolescents with follow-up celiac disease and in their families. Turk. J. Gastroenterol. 2014, 25, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Simsek, S.; Baysoy, G.; Gencoglan, S.; Uluca, U. Effects of gluten-free diet on quality of life and depression in children with celiac disease. J. Pediatr. Gastroenterol. Nutr. 2015, 61, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.B.; Lynch, K.F.; Kurppa, K.; Koletzko, S.; Krischer, J.; Liu, E.; Johnson, S.B.; Agardh, D.; Rewers, M.; Bautista, K.; et al. Psychological manifestations of celiac disease autoimmunity in young children. Pediatrics 2017, 139, e20162848. [Google Scholar] [CrossRef] [PubMed]
- Wagner, G.; Zeiler, M.; Berger, G.; Huber, W.D.; Favaro, A.; Santonastaso, P.; Karwautz, A. Eating disorders in adolescents with celiac disease: Influence of personality characteristics and coping. Eur. Eat. Disord. Rev. 2015, 23, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Babio, N.; Alcázar, M.; Castillejo, G.; Recasens, M.; Martínez-Cerezo, F.; Gutiérrez-Pensado, V.; Vaqué, C.; Vila-Martí, A.; Torres-Moreno, M.; Sánchez, E.; et al. Risk of eating disorders in patients with celiac disease. J. Pediatr. Gastroenterol. Nutr. 2018, 66, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Terrone, G.; Parente, I.; Romano, A.; Auricchio, R.; Greco, L.; Del Giudice, E. The pediatric symptom checklist as screening tool for neurological and psychosocial problems in a paediatric cohort of patients with coeliac disease. Acta Paediatr. Int. J. Paediatr. 2013, 102, e325–e328. [Google Scholar] [CrossRef] [PubMed]
- Ruggieri, M.; Incorpora, G.; Polizzi, A.; Parano, E.; Spina, M.; Pavone, P. Low prevalence of neurologic and psychiatric manifestations in children with gluten sensitivity. J. Pediatr. 2008, 152, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Mazzone, L.; Reale, L.; Spina, M.; Guarnera, M.; Lionetti, E.; Martorana, S.; Mazzone, D. Compliant gluten-free children with celiac disease: An evaluation of psychological distress. BMC Pediatr. 2011, 11, 46. [Google Scholar] [CrossRef] [PubMed]
- Butwicka, A.; Lichtenstein, P.; Frisen, L.; Almqvist, C.; Larsson, H.; Ludvigsson, J.F. Celiac disease is associated with childhood psychiatric disorders: A population-based study. J. Pediatr. 2017, 184, 87–93.e81. [Google Scholar] [CrossRef] [PubMed]
- Zelnik, N.; Pacht, A.; Obeid, R.; Lerner, A. Range of neurologic disorders in patients with celiac disease. Pediatrics 2004, 113, 1672–1676. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Reichenberg, A.; Hultman, C.M.; Murray, J.A. A nationwide study of the association between celiac disease and the risk of autistic spectrum disorders. JAMA Psychiatry 2013, 70, 1224–1230. [Google Scholar] [CrossRef] [PubMed]
- West, J.; Logan, R.F.; Hubbard, R.B.; Card, T.R. Risk of schizophrenia in people with coeliac disease, ulcerative colitis and crohn’s disease: A general population-based study. Aliment. Pharmacol. Ther. 2006, 23, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Eaton, W.W.; Byrne, M.; Ewald, H.; Mors, O.; Chen, C.Y.; Agerbo, E.; Mortensen, P.B. Association of schizophrenia and autoimmune diseases: Linkage of danish national registers. Am. J. Psychiatry 2006, 163, 521–528. [Google Scholar] [CrossRef] [PubMed]
- Benros, M.E.; Nielsen, P.R.; Nordentoft, M.; Eaton, W.W.; Dalton, S.O.; Mortensen, P.B. Autoimmune diseases and severe infections as risk factors for schizophrenia: A 30-year population-based register study. Am. J. Psychiatry 2011, 168, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Wijarnpreecha, K.; Jaruvongvanich, V.; Cheungpasitporn, W.; Ungprasert, P. Association between celiac disease and schizophrenia: A meta-analysis. Eur. J. Gastroenterol. Hepatol. 2018, 30, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Hallert, C.; Aström, J. Psychic disturbances in adult coeliac disease. II. Psychological findings. Scand. J. Gastroenterol. 1982, 17, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Hallert, C.; Aström, J.; Walan, A. Reversal of psychopathology in adult coeliac disease with the aid of pyridoxine (vitamin b6). Scand. J. Gastroenterol. 1983, 18, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Addolorato, G.; Stefanini, G.F.; Capristo, E.; Caputo, F.; Gasbarrini, A.; Gasbarrini, G. Anxiety and depression in adult untreated celiac subjects and in patients affected by inflammatory bowel disease: A personality “trait” or a reactive illness? Hepato-Gastroenterology 1996, 43, 1513–1517. [Google Scholar] [PubMed]
- Ciacci, C.; Iavarone, A.; Mazzacca, G.; De Rosa, A. Depressive symptoms in adult coeliac disease. Scand. J. Gastroenterol. 1998, 33, 247–250. [Google Scholar] [CrossRef] [PubMed]
- Addolorato, G.; Capristo, E.; Ghittoni, G.; Valeri, C.; Masciana, R.; Ancona, C.; Gasbarrini, G. Anxiety but not depression decreases in coeliac patients after one-year gluten-free diet: A longitudinal study. Scand. J. Gastroenterol. 2001, 36, 502–506. [Google Scholar] [CrossRef] [PubMed]
- Cicarelli, G.; Della Rocca, G.; Amboni, M.; Ciacci, C.; Mazzacca, G.; Filla, A.; Barone, P. Clinical and neurological abnormalities in adult celiac disease. Neurol. Sci. 2003, 24, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Carta, M.G.; Hardoy, M.C.; Boi, M.F.; Mariotti, S.; Carpiniello, B.; Usai, P. Association between panic disorder, major depressive disorder and celiac disease: A possible role of thyroid autoimmunity. J. Psychosom. Res. 2002, 53, 789–793. [Google Scholar] [CrossRef]
- Carta, M.G.; Hardoy, M.C.; Usai, P.; Carpiniello, B.; Angst, J. Recurrent brief depression in celiac disease. J. Psychosom. Res. 2003, 55, 573–574. [Google Scholar] [CrossRef]
- Addolorato, G.; Mirijello, A.; D’Angelo, C.; Leggio, L.; Ferrulli, A.; Vonghia, L.; Cardone, S.; Leso, V.; Miceli, A.; Gasbarrini, G. Social phobia in coeliac disease. Scand. J. Gastroenterol. 2008, 43, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Garud, S.; Leffler, D.; Dennis, M.; Edwards-George, J.; Saryan, D.; Sheth, S.; Schuppan, D.; Jamma, S.; Kelly, C.P. Interaction between psychiatric and autoimmune disorders in coeliac disease patients in the northeastern united states. Aliment. Pharmacol. Ther. 2009, 29, 898–905. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.F.; Gerdes, L.U. Meta-analysis on anxiety and depression in adult celiac disease. Acta Psychiatr. Scand. 2012, 125, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.L.; Biesiekierski, J.R.; Yelland, G.W.; Muir, J.G.; Gibson, P.R. Randomised clinical trial: Gluten may cause depression in subjects with non-coeliac gluten sensitivity—An exploratory clinical study. Aliment. Pharmacol. Ther. 2014, 39, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Carta, M.G.; Conti, A.; Lecca, F.; Sancassiani, F.; Cossu, G.; Carruxi, R.; Boccone, A.; Cadoni, M.; Pisanu, A.; Moro, M.F.; et al. The burden of depressive and bipolar disorders in celiac disease. Clin. Pract. Epidemiol. Ment. Health 2015, 11, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Di Sabatino, A.; Volta, U.; Salvatore, C.; Biancheri, P.; Caio, G.; De Giorgio, R.; Di Stefano, M.; Corazza, G.R. Small amounts of gluten in subjects with suspected nonceliac gluten sensitivity: A randomized, double-blind, placebo-controlled, cross-over trial. Clin. Gastroenterol. Hepatol. 2015, 13, 1604–1612.e1603. [Google Scholar] [CrossRef] [PubMed]
- Tortora, R.; Imperatore, N.; Ciacci, C.; Zingone, F.; Capone, P.; Leo, M.; Pellegrini, L.; De Stefano, G.; Caporaso, N.; Rispo, A. High prevalence of post-partum depression in coeliac women. Dig. Liver Dis. 2013, 45, S120. [Google Scholar] [CrossRef]
- Sainsbury, K.; Marques, M.M. The relationship between gluten free diet adherence and depressive symptoms in adults with coeliac disease: A systematic review with meta-analysis. Appetite 2018, 120, 578–588. [Google Scholar] [CrossRef] [PubMed]
- Passananti, V.; Siniscalchi, M.; Zingone, F.; Bucci, C.; Tortora, R.; Iovino, P.; Ciacci, C. Prevalence of eating disorders in adults with celiac disease. Gastroenterol. Res. Pract. 2013, 2013, 491657. [Google Scholar] [CrossRef] [PubMed]
- Satherley, R.M.; Howard, R.; Higgs, S. The prevalence and predictors of disordered eating in women with coeliac disease. Appetite 2016, 107, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Marild, K.; Størdal, K.; Bulik, C.M.; Rewers, M.; Ekbom, A.; Liu, E.; Ludvigsson, J.F. Celiac disease and anorexia nervosa: A nationwide study. Pediatrics 2017, 139, e20164367. [Google Scholar] [CrossRef] [PubMed]
- Marild, K.; Morgenthaler, T.I.; Somers, V.K.; Kotagal, S.; Murray, J.A.; Ludvigsson, J.F. Increased use of hypnotics in individuals with celiac disease: A nationwide case-control study. BMC Gastroenterol. 2015, 15, 10. [Google Scholar] [CrossRef] [PubMed]
- Roos, S.; Karner, A.; Hallert, C. Psychological well-being of adult coeliac patients treated for 10 years. Dig. Liver Dis. 2006, 38, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Gili, M.; Béjar, L.; Ramirez, G.; Lopez, J.; Cabanillas, J.L.; Sharp, B. Celiac disease and alcohol use disorders: Increased length of hospital stay, overexpenditures and attributable mortality. Rev. Esp. Enferm. Dig. 2013, 105, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Lebwohl, B.; Luchsinger, J.A.; Freedberg, D.E.; Green, P.H.; Ludvigsson, J.F. Risk of dementia in patients with celiac disease: A population-based cohort study. J. Alzheimer’s Dis. 2016, 49, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Fera, T.; Cascio, B.; Angelini, G.; Martini, S.; Guidetti, C.S. Affective disorders and quality of life in adult coeliac disease patients on a gluten-free diet. Eur. J. Gastroenterol. Hepatol. 2003, 15, 1287–1292. [Google Scholar] [CrossRef] [PubMed]
- Sainsbury, K.; Mullan, B.; Sharpe, L. Reduced quality of life in coeliac disease is more strongly associated with depression than gastrointestinal symptoms. J. Psychosom. Res. 2013, 75, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Zylberberg, H.M.; Demmer, R.T.; Murray, J.A.; Green, P.H.R.; Lebwohl, B. Depression and insomnia among individuals with celiac disease or on a gluten-free diet in the United States: Results from the national health and nutrition examination survey (nhanes) 2009–2014. Gastroenterology 2017, 152, S482–S483. [Google Scholar] [CrossRef]
- Dickson, B.C.; Streutker, C.J.; Chetty, R. Coeliac disease: An update for pathologists. J. Clin. Pathol. 2006, 59, 1008–1016. [Google Scholar] [CrossRef] [PubMed]
- Leffler, D.A.; Green, P.H.; Fasano, A. Extraintestinal manifestations of coeliac disease. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.G.; Mihalopoulos, C.; Erskine, H.E.; Roberts, J.; Rahman, A. Childhood mental and developmental disorders. In Mental, Neurological, and Substance Use Disorders: Disease Control Priorities, 3rd ed.; Patel, V., Chisholm, D., Dua, T., Laxminarayan, R., Medina-Mora, M.E., Eds.; The World Bank: Washington, DC, USA, 2016; Volume 4. [Google Scholar]
- Egede, L.E. Major depression in individuals with chronic medical disorders: Prevalence, correlates and association with health resource utilization, lost productivity and functional disability. Gen. Hosp. Psychiatry 2007, 29, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Katon, W.J. Epidemiology and treatment of depression in patients with chronic medical illness. Dialog. Clin. Neurosci. 2011, 13, 7–23. [Google Scholar]
- Kang, H.J.; Kim, S.Y.; Bae, K.Y.; Kim, S.W.; Shin, I.S.; Yoon, J.S.; Kim, J.M. Comorbidity of depression with physical disorders: Research and clinical implications. Chonnam Med. J. 2015, 51, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Karwautz, A.; Wagner, G.; Berger, G.; Sinnreich, U.; Grylli, V.; Huber, W.D. Eating pathology in adolescents with celiac disease. Psychosomatics 2008, 49, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Hudson, J.I.; Hiripi, E.; Pope, H.G., Jr.; Kessler, R.C. The prevalence and correlates of eating disorders in the national comorbidity survey replication. Biol. Psychiatry 2007, 61, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.J.; Chao, Y.L.; Chen, C.Y.; Chang, C.M.; Wu, E.C.; Wu, C.S.; Yeh, H.H.; Chen, C.H.; Tsai, H.J. Prevalence of autoimmune diseases in in-patients with schizophrenia: Nationwide population-based study. Br. J. Psychiatry 2012, 200, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Ergun, C.; Urhan, M.; Ayer, A. A review on the relationship between gluten and schizophrenia: Is gluten the cause? Nutr. Neurosci. 2017. [Google Scholar] [CrossRef] [PubMed]
- Cascella, N.G.; Kryszak, D.; Bhatti, B.; Gregory, P.; Kelly, D.L.; Mc Evoy, J.P.; Fasano, A.; Eaton, W.W. Prevalence of celiac disease and gluten sensitivity in the united states clinical antipsychotic trials of intervention effectiveness study population. Schizophr. Bull. 2011, 37, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Kalaydjian, A.E.; Eaton, W.; Cascella, N.; Fasano, A. The gluten connection: The association between schizophrenia and celiac disease. Acta Psychiatr. Scand. 2006, 113, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, B.J.; Crawford, S.G.; Field, C.J.; Simpson, J.S. Vitamins, minerals, and mood. Psychol. Bull. 2007, 133, 747–760. [Google Scholar] [CrossRef] [PubMed]
- Wierdsma, N.J.; van Bokhorst-de van der Schueren, M.A.; Berkenpas, M.; Mulder, C.J.; van Bodegraven, A.A. Vitamin and mineral deficiencies are highly prevalent in newly diagnosed celiac disease patients. Nutrients 2013, 5, 3975–3992. [Google Scholar] [CrossRef] [PubMed]
- Najjar, S.; Pearlman, D.M.; Alper, K.; Najjar, A.; Devinsky, O. Neuroinflammation and psychiatric illness. J. Neuroinflam. 2013, 10, 43. [Google Scholar] [CrossRef] [PubMed]
- Grenham, S.; Clarke, G.; Cryan, J.F.; Dinan, T.G. Brain-gut-microbe communication in health and disease. Front. Physiol. 2011, 2, 94. [Google Scholar] [CrossRef] [PubMed]
- Heyman, M.; Abed, J.; Lebreton, C.; Cerf-Bensussan, N. Intestinal permeability in coeliac disease: Insight into mechanisms and relevance to pathogenesis. Gut 2012, 61, 1355–1364. [Google Scholar] [CrossRef] [PubMed]
- Leffler, D.A.; Edwards-George, J.; Dennis, M.; Schuppan, D.; Cook, F.; Franko, D.L.; Blom-Hoffman, J.; Kelly, C.P. Factors that influence adherence to a gluten-free diet in adults with celiac disease. Dig. Dis. Sci. 2008, 53, 1573–1581. [Google Scholar] [CrossRef] [PubMed]
- Lebwohl, B.; Ludvigsson, J.F.; Green, P.H. Celiac disease and non-celiac gluten sensitivity. BMJ 2015, 351, h4347. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Sellgren, C.; Runeson, B.; Langstrom, N.; Lichtenstein, P. Increased suicide risk in coeliac disease—A Swedish nationwide cohort study. Dig. Liver Dis. 2011, 43, 616–622. [Google Scholar] [CrossRef] [PubMed]
| Author (Year) | Country | Primary Objective | Design ‡ | Study Setting | Psychiatric Comorbidity Assessment | Celiac Disease Diagnostic Criteria |
|---|---|---|---|---|---|---|
| Autism spectrum disorders | ||||||
| Pavone (1997) [30] | Italy | To evaluate behavioral problems and autistic features in children with CD | Cross-sectional | Clinical | DSM-III-R | Biopsy |
| Schizophrenia Spectrum | ||||||
| Ludvigsson (2007) * [31] | Sweden | To determine the risk of non-affective psychosis in patients with CD in a national general population cohort | Population-based cohort | Community | ICD | ICD |
| Bipolar, depressive and anxiety disorders | ||||||
| Pynnonen (2004) [32] | Finland | To compare the prevalence of current and lifetime mental disorders in adolescents with CD and controls | Cross-sectional | Clinical | K-SADS-PL Youth Self-Report BDI and BAI HDRS and HARS | Biopsy |
| Accomando (2005) * [33] | Italy | To investigate the relationship between CD and depression | Cross-sectional | Clinical | CDQ (adults) CDS (children) | NR |
| Ludvigsson (2007) * [34] | Sweden | To investigate the risk of subsequent depression and bipolar in patients with CD | Population-based cohort | Community | ICD | NR |
| Fidan (2013) [35] | Turkey | To investigate the depression and anxiety levels of children and adolescents with celiac disease and the impact of these on quality of life | Cross-sectional | Clinical | CDI STAIC | NR |
| Esenyel (2014) [36] | Turkey | To explore the diet compliance and depression and anxiety levels of pediatric celiac children and their families after a GFD | Cross-sectional | Clinical | CDI SCARED | ESPGHAN criteria |
| Simsek (2015) [37] | Turkey | To evaluate depressive symptoms at time of CD diagnosis and 6 months following GFD initiation | Phase 1: Cross-sectional Phase 2: Case-series | Clinical | CDI HRQOL (Kid-KINDL) | Biopsy |
| Smith (2017) [38] | USA, Finland, Germany, and Sweden | To assess mother’s report of psychological functioning in children with CDA | Cohort | Community | CBCL | Serology and optional biopsy |
| Feeding and eating disorders | ||||||
| Wagner (2015) [39] | Austria | To assess the determinants of eating disorders in female adolescents with CD | Cross-sectional | Clinical | EDI-2 EDE DSM-IV for subclinical eating disorders CDI (total score ≥ 18) | Both |
| Babio (2018) * [40] | Spain | To assess the risk of eating disorders in individuals between 10 and 23 years old diagnosed with CD | Cross-sectional | Clinical | CEAT EAT-26 SCFF BITE BSQ | Both |
| Overall psychological status | ||||||
| Terrone (2013) [41] | Italy | To screen for neurological and behavioral disorders in children with CD | Phase 1: cross-sectional Phase 2: cohort | Clinical | PSC (total score ≥ 28) | ESPGHAN criteria |
| Various psychiatric conditions | ||||||
| Ruggieri (2008) [42] | Italy | To determine the prevalence of neurologic symptoms in children with gluten sensitivity enteropathy | Cross-sectional | Clinical | NR | Both |
| Mazzone (2011) [43] | Italy | To identify psychological features in children with CD following strict GFD | Cross-sectional | Clinical | MASC CBCL CDI DSM-IV-TR criteria to assess autistic disorders | ESPGHAN criteria |
| Butwicka (2017) [44] | Sweden | To examine the risk of psychiatric disorders in children with a biopsy-verified diagnosis of CD and to examine the prevalence of psychiatric disorders before CD is diagnosed in children | Population-based cohort | Community | ICD | Biopsy |
| Author (Year) | Design | Sample Size and Demographic Characteristics | Summary of Outcomes | Associated Factors with Psychiatric Comorbidities and Other Relevant Information |
|---|---|---|---|---|
| Autism spectrum disorders | ||||
| Pavone (1997) [30] | Cross-sectional | CD, n = 120 (mean age 9.6 years, 48% females) Recently-diagnosed CD, n = 27 CD on strict GFD, n = 70 GFD non-adherent CD, n = 23 Controls, n = 20 (mean age 9.6 years, 48% females) | - Autism diagnosis: none of the recently-diagnosed CD - Language delay: Two subjects in GFD-compliant, one subject in the non-adherent group - Differences were not statistically significant compared to controls | NR |
| Schizophrenia Spectrum | ||||
| Ludvigsson (2007) [31] | Population-based cohort study | CD, n = 14,003 (age at diagnosis, 0–15 years 66% & ≥16 years 34%; 59% females) Controls, n = 68,125 (matched age and gender) | - Likelihood of psychosis in CD vs. controls using a Cox regression model stratified for gender, age, year of study entry and county: Any non-affective psychosis (schizophrenia and other psychoses) HR = 1.55 (95% CI: 1.16–2.06) Non-schizophrenic non-affective psychosis HR = 1.61 (95% CI: 1.19–2.20) Schizophrenia HR = 1.43 (95% CI: 0.77–2.67) | NR |
| Bipolar, depressive and anxiety disorders | ||||
| Pynnonen (2004) [32] | Cross-sectional | CD, n = 29 (mean age 14.2 years, 55% females) Controls, n = 29 (mean age 14.4 years, 55% females) | - Lifetime prevalence of major depression disorder (CD vs. controls): 31% vs. 7%, p <0.05. OR = 6.06 (95% CI: 1.18–31.23). - Disruptive behavior disorders (CD vs. controls): 28% vs. 3%, p <0.05. OR = 10.67 (95% CI: 1.24–92). - Lifetime prevalence of anxiety disorders (CD vs. controls): 21% vs. 24%, p = NS - Differences in the prevalence of current depressive, anxiety, or disruptive behavior disorders between the two groups were non-significant | - History of parental depressive disorder was more common in CD patients with depressive symptomatology compared to CD without depressive symptomatology - Parental educational level, divorce of parents, poor weight or height gain, and somatic symptoms were not associated with mental disorders |
| Accomando (2005) [33] | Cross-sectional | CD, n = 42 (17 adults and 25 children) HC, n = 42 | Prevalence of depression (CD vs. HC): 26.2% vs. 30.9%, p = NS | - Females predominated in CD patients with depression (not reaching statistical significance) - Depression was more common in CD with functional comorbid conditions (specific conditions not specified) |
| Ludvigsson (2007) [34] | Population-based cohort study | CD, n = 13,776 (median age at diagnosis 2 years, 58.6% females) Controls, n = 66,815 (median age at diagnosis 2 years, 58.7% females) | - CD was associated with an increased risk of subsequent depression (HR = 1.8, 95% CI: 1.6–2.2) - No significant association between CD and bipolar disorder was reported (HR = 1.1, 95% CI: 0.7–1.7) | - Socioeconomic index didn’t have any confounding effect on the later schizophrenia diagnosis in CD |
| Fidan (2013) [35] | Cross-sectional | CD, n =30 (mean age 12.4 ± 3.1 years, 57% females). HC, n = 30 (mean age NR, 57% females) | - CD vs. HC: CDI: 10.8 ± 7.4 vs. 8.8 ± 6.8, p=0.28 STAIC-State Anxiety: 34.6 ± 6.1 vs. 32.8 + 7.2, p = 0.30 STAIC-Trait Anxiety: 33.7 ± 6.5 vs. 33 ± 6.3, p =0.64 | - Data on the impact of depression and anxiety on HRQOL NR |
| Esenyel (2014) [36] | Cross-sectional | CD, n = 30 (mean age 11.9 ± 2 years, 70% females) HC, n =20 (mean age 12 ± 2 years, 55% females) | - CD vs. HC: CDI points: 8.73 ± 5.51 vs. 8.3 ± 4.02, p = 0.921 SCARED points: 24.5 ± 14.41 vs. 17.85 ± 9.12, p = 0.120 - There were no differences in depression and anxiety scores between patients with CD compliant or non-compliant with a GFD | NR |
| Simsek (2015) [37] | Phase 1: Cross-sectional Phase 2: Case-series | CD, n = 25 (mean age 11.8 years, 72% females) Controls, n = 25 (mean age 12.2 years, 64%) | - At the time of diagnosis (CD vs. controls): CDI scores: 9 vs. 6, p = NS - 6 months following GFD initiation: CDI scores in CD: 9 before diet vs. 9.5 after diet, p = NS | - Total scores of HRQOL were significantly lower in CD patients (p <0.05) |
| Smith (2017) [38] | Cohort | Aware-CDA, n = 440 (58% females) Unware-CDA, n = 66 (50% females) No CDA, n = 3651 (NR) | - At 3.5 years of age, unaware-CDA mothers reported more anxious/depressed symptoms, aggressive behavior, and externalizing composite score compared to aware-CDA group (p <0.05) or without CDA (p <0.05) - At 3.5 years of age, Aware-CDA mothers reported significantly fewer problems on the anxious/depressed subscale compared to No CDA group (p = 0.03) - At 4.5 years, there were no significant differences | NR |
| Feeding and eating disorders | ||||
| Wagner (2015) [39] | Cross-sectional | CD, n = 206 (mean age NR) CD with ED, n = 32 (mean age 16.4 yeas) CD without ED, n = 174 (mean age 14.5 years) Controls, n = 53 (mean age 14.7 years) | - Lifetime prevalence of EDs: 5.3% of girls with CD: anorexia nervosa (n = 1), bulimia nervosa (n = 4), and EDs not otherwise specified (n = 6); 3.9% suffered from current ED - Criteria for lifetime subclinical EDs: 21 girls (10.2%) with CD - Higher BMI and self-directedness were predictors of greater risk of ED - Higher ill-being and lower joy in life were reported by patients with CD with ED compared with patients without EDs, even when controlling for age and depression levels | - No differences between patients (with CD) with and without EDs in coping strategies were found - Higher BMI and lower self-directedness were linked to higher risk of ED in CD |
| Babio (2018) [40] | Cross-sectional | CD, n = 98 (mean age 15 years, 60% females) Controls, n = 98 (mean age 15 years, 60% females) | - No significant differences in the median scores of the screening tools for EDs between CD and HC - CD vs. HC: β coefficient = 2.15 (1.04); p = 0.04 in a multiple linear regression model for EAT after adjusting for several factors | - Only significant results for one out of the 4 models (one for each screening test) - Age > 13 years old was positively associated with an increase in the score on the EAT |
| Overall psychological status | ||||
| Terrone (2013) [41] | Phase 1: cross-sectional Phase 2: cohort | CD, n = 139 (mean age 10 years, 64.7% females): Group A (n =40): newly diagnosed CD Group B (n = 54): CD in remission on GFD > 1 year Group C (n = 45): potential CD | - Comparison of mean PSC scores using ANOVA: Group A, 14.8 ± 4.2 (one pathological score) vs. Group B, 12.3 ± 6.4 (one pathological score) vs. Group C, 7.6 ± 6 (p <0.0001) | NR |
| Various psychiatric conditions | ||||
| Ruggieri (2008) [42] | Cross-sectional | GS, n = 835 (demographic characteristics NR) Controls, n = 300 (demographic characteristics NR) | - 3 out of 835 children had bipolar disorders - None of the controls had psychiatric disorders | NR |
| Mazzone (2011) [43] | Cross-sectional | CD, n = 100 (mean age 10.4 years, 65% females) HC, n = 100 (mean age 11.5 years, 58% females) | - MASC scores: CD children showed significantly higher scores (50 ± 8.3 vs. 42.9 ± 6.6, p <0.01) - CDI scores: CD children showed significantly higher scores (8.1 ± 5.7 vs. 5.6 ± 3.4, p <0.01) - No significant differences were found in CBCL analysis - Two children in the CD group were classified within the spectrum of autistic disorders | - CD males showed significantly higher scores for total CBCL - CD females showed an increased rate of anxiety and depression symptoms, as indicated by significantly higher MASC and CDI scores |
| Butwicka (2017) [44] | Population-based cohort study | CD, n = 10,903 (median age 3 years, 62% females) Controls, n = 1,042,072 (age NR but matched, 61% females) | - HRs from a Multivariate Cox regression adjusted for maternal/paternal age at child’s birth, maternal/paternal country of birth, level of education of higher-educated parent, gestational age, birth weight, birth cohort, Apgar score, and history of psychiatric disorders before recruitment: Any psychiatric disorder 1.4 (95% CI: 1.3–1.4) Psychotic disorders 1.9 (95% CI: 1.0–3.5) Mood disorders 1.2 (95% CI: 1.0–1.4) Anxiety disorders 1.2 (95% CI: 1.0–1.4) EDs 1.4 (95% CI: 1.1–1.8) Substance misuse 1.0 (95% CI: 0.9–1.3) Behavioral disorders 1.4 (95% CI: 1.2–1.6) ADHD 1.2 (95% CI: 1.0–1.4) Autism spectrum disorder 1.3 (95% CI: 1.1–1.7) Intellectual disability 1.7 (95% CI: 1.4–2.1) | NR |
| Author (Year) | Country | Primary Objective | Design ‡ | Study Setting | Psychiatric Comorbidity Assessment | Celiac Disease Diagnostic Criteria |
|---|---|---|---|---|---|---|
| Attention-Deficit/Hyperactivity Disorder | ||||||
| Zelnik (2004) * [45] | Israel | To evaluate neurologic disorders including ADHD in CD | Cross-sectional | Clinical | DSM criteria for ADHD | Both |
| Autism spectrum disorders | ||||||
| Ludvigsson (2013) * [46] | Sweden | To examine the association between autistic spectrum disorder and CD | Cohort study | Community | ICD | Group 1: villous atrophy, Marsh stage 3 Group 2: villous atrophy, Marsh stages 1–2 Group 3: normal mucosa and positive serologic findings |
| Schizophrenia Spectrum | ||||||
| West (2006) [47] | UK | To compare the risk of schizophrenia in patients with CD, ulcerative colitis, Crohn’s disease with the general population | Population-based cross-sectional | Community | NR | NR |
| Eaton (2006) [48] | Denmark | To estimate the association of schizophrenia with autoimmune disorders | Cross-sectional | Community | ICD | ICD |
| Benros (2011) [49] | Denmark | To investigate whether autoimmune diseases are associated with increased risk of schizophrenia | Population-based retrospective cohort | Community | ICD | NR |
| Wijarnpreecha (2018) [50] | USA | To evaluate the risk of developing schizophrenia among patients with CD | Meta-analysis | NA | NR | NR |
| Bipolar, depressive or anxiety disorders | ||||||
| Hallert (1982) [51] Hallert (1983) [52] | Sweden | To compare the prevalence of psychiatric illness among patients with CD vs. controls and to assess the effects of gluten withdrawal and vitamin B6 supplement on depressive symptoms | Phase 1: cross-sectional Phase 2: case-series | Clinical | MMPI | Both (serological and biopsy) combined with morphological improvement with GFD |
| Addolorato (1996) [53] | Italy | To conduct psychometric evaluation in patients with CD or IBD compared to healthy controls | Cross-sectional | Clinical | STAI IDSQ | Both |
| Ciacci (1998) [54] | Italy | To explore the relevance of depressive symptoms in a large series of adult celiacs | Cross-sectional | Clinical | SRDS | Both |
| Addolorato (2001) [55] | Italy | To evaluate state and trait anxiety and depression in adult CD patients before and after 1 year of GFD | Phase 1: Cross-sectional Phase 2: Case-series | Clinical | STAI SRDS | Both |
| Cicarelli (2003) [56] | Italy | To evaluate the prevalence of headache, mood disorders, epilepsy, ataxia and peripheral neuropathy in adult celiac patients | Cross-sectional | Clinical | DSM-IV | Both |
| Carta (2002) [57] Carta (2003) [58] | Italy | To evaluate the association between celiac disease and specific anxiety and depressive disorders | Cross-sectional | Clinical | CIDI-DSM-IV | Both |
| Addolorato (2008) [59] | Italy | To evaluate social phobia in CD patients | Cross-sectional | Clinical | LSAS total > 30 SRDS > 49 | Both |
| Garud (2009) [60] | US | To determine the prevalence of psychiatric and autoimmune disorders in patients with CD in the US compared with control groups | Cross-sectional | Community | Clinical charts | Biopsy |
| Smith (2012) [61] | Denmark | To investigate whether CD is reliably linked with anxiety and/or depression | Meta-analysis | NA | NA | NA |
| Peters (2014) [62] | Australia | To investigate the effect of gluten on mental state among patients with NCGS | Randomized, double-blind, cross-over trial | Clinical | STPI | Challenging with varying amounts of gluten |
| Carta (2015) [63] | Italy | To measure the association between CD and affective disorders | Cross-sectional | Clinical | DSM-IV | NR |
| Di Sabatino (2015) [64] | Italy | To assess the effects of gluten administration on intestinal and extraintestinal symptoms in subjects with NCGS | Randomized, double-blind, placebo-controlled cross-over trial | Clinical | Extraintestinal symptoms, including depression, were self-reported by patients as absent or present | Self-reported persistence of relevant intestinal and extraintestinal symptoms at low gluten doses |
| Tortora (2013) [65] | Italy | To evaluate the prevalence of post-partum depression in CD | Cross-sectional | Clinical | EPDS (Total score > 10 possible PPD) | Both |
| Sainsbury (2018) [66] | UK | To synthesize the evidence on the relationship between depression and degree of adherence to GFD in patients with CD | Meta-analysis | NA | NA | NA |
| Feeding and eating disorders | ||||||
| Passananti (2013) [67] | Italy | To investigate the prevalence of eating disorders in patients with celiac disease | Cross-sectional | Clinical | Structured psychological assessment using: BES (Total score ≥ 17) EAT-26 (Total score ≥ 20) EDI-2 M-SDS (Total score > 44) SCL-90 | Both |
| Satherley (2016) [68] | United Kingdom | To examine the prevalence of eating disorders in women with CD | Cross-sectional | Clinical | EAT-26 (Total score > 20) BES (Moderate bingeing, score > 17; severe bingeing, score > 27) DASS-21 | Self-reported a biopsy-confirmed diagnosis |
| Mårild (2017) * [69] | Sweden | To determine whether women with CD are at increased risk of diagnosis of anorexia nervosa | Register-based cohort study | Community | ICD | Group 1: villous atrophy, Marsh stage 3 Group 2: villous atrophy, Marsh stages 1–2 Group 3: normal mucosa and positive serologic findings |
| Sleep-Wake disorders | ||||||
| Marild (2015) * [70] | Sweden | To estimate the risk of repeated use of hypnotics among individuals with CD as a proxy measure for poor sleep | Population-based cohort study | Community | Prescribed Drug Register in Sweden—Use of hypnotics | Biopsy |
| Substance-related and addictive disorders | ||||||
| Roos (2006) [71] | Sweden | To assess psychological well-being in adults with CD with proven remission (treated for 10 years) | Cross-sectional | Clinical | PGWB | Remission was ascertained with a return of villous structure at repeat biopsy (82%) or negative serology (18%) |
| Gili (2013) [72] | Spain | To study the impact of alcohol disorders on length of hospital stays, over-expenditures during hospital stays, and excess mortality in CD patients | Cross-sectional | Clinical | ICD | ICD |
| Neurocognitive Disorders | ||||||
| Lebwohl (2016) [73] | Sweden | To determine whether patients with CD have an increased risk of dementia | Population-based cohort | Community | ICD | Biopsy |
| Various psychiatric conditions | ||||||
| Fera (2003) [74] | Italy | To estimate the incidence of psychiatric disorders in celiac disease patients on gluten withdrawal | Cross-sectional | Clinical | DSM-IV criteria | Biopsy & Clinical history |
| Sainsbury (2013) [75] | Australia | To compare the relevant impact of psychological symptoms to known negative impacts of gastrointestinal symptoms and adherence to the GFD on quality of life | Study 1: Cross-sectional Study 2: Cross-sectional | Clinical | DASS EDI-3 CISS | Biopsy |
| Zylberberg (2017) [76] | US | To assess the prevalence of depression and insomnia among patients with CD, both diagnosed and undiagnosed, and people without CD who avoid gluten | Population-based cross-sectional | Community | PHQ-9 (Total score on questions 1–9 ≥ 10) SDQ | Diagnosed CD: self-reported diagnosis Undiagnosed CD: serology |
| Author (Year) | Design | Sample Size and Demographic Characteristics | Summary of Outcomes | Associated Factors with Psychiatric Comorbidities and other Relevant Information |
|---|---|---|---|---|
| Attention-Deficit/Hyperactivity Disorder | ||||
| Zelnik (2004) [45] | Cross-sectional | CD, n = 111 (mean age 20.1 years, 57.7% females) Controls, n = 211 (mean age 20.1 years, 59.7% females) | - ADHD diagnosis: 20.7% in CD vs. 10.5% in controls (p <0.01) CD, 20.3% of female patients and 21.2% of male patients Controls, 8.7% of females and 12.9% males | -No gender differences were found in the prevalence of ADHD in patients with CD -Differences in ADHD were not different among CD patients presenting with infantile form of CD or late-onset symptoms |
| Autism spectrum disorders | ||||
| Ludvigsson (2013) [46] | Cohort study | Group 1, n = 26,995 (age at diagnosis, 0–19 years 40.4%, >20 years 59.6%; 62.1% females); Controls, n = 134,076 (matched age and gender) Group 2, n = 12,304 (age at diagnosis, 0–19 years 8.9%, >20 years 91.1%; 56.9% females); Controls, n = 60,654 (matched age and gender) Group 3, n = 3719 (age at diagnosis, 0–19 years 25.3%, >20 years 74.7%; 62.1% females; 62.1% females); Controls, n = 18,478 (matched age and gender) | - Risk of later ASD diagnosis: Group 1: HR = 1.39 (95% CI: 1.13–1.71) Group 2: HR =2.01 (95% CI: 1.29–3.13) Group 3: HR = 3.09 (95% CI: 1.99–4.8) | NR |
| Schizophrenia Spectrum | ||||
| West (2006) [47] | Population-based case-control | CD, n = 4732; matched controls, n = 23,620 Crohn’s disease, n = 5961; matched controls, n = 29,843 Ulcerative colitis, n = 8301; matched controls, n = 41,589 Demographics NR | - Prevalence of schizophrenia 0.25% in CD, 0.27% in Crohn’s disease and 0.24% in ulcerative colitis, 0.37% in general population - ORs for schizophrenia compared to controls adjusted for smoking status: 0.76 (95% CI: 0.4-1.4) in CD, 0.74 (95% CI: 0.4–1.3) in Crohn’s disease, 0.71 (95% CI 0.4–1.1) in ulcerative colitis | NR |
| Eaton (2006) [48] | Cross-sectional | Schizophrenia, n = 7704, 25 controls for each case. Demographics NR | - Prior CD diagnosis in subjects with schizophrenia: Crude incidence rate: 3.8 (95% CI: 1.3–11) Adjusted incidence rate: 3.6 (95% CI: 1.2–10.6) | NR |
| Benros (2011) [49] | Population-based cohort | Schizophrenia, n = 39,076: Prior diagnosis of autoimmune disease, n = 927, autoimmune disease and infections, n = 444, without autoimmune disease, n = 37,705 Demographics NR | - The risk of schizophrenia among individuals with CD was increased: CD without infection: Incidence rate ratio = 2.11 (95% CI: 1.09–3.61) CD with infections: Incidence rate ratio = 2.47 (95% CI: 1.13–4.61) | NR |
| Wijarnpreecha (2018) [50] | Meta-analysis | Four studies were included | - Higher risk of schizophrenia among patients with CD was found; pooled OR = 2.03 (95% CI: 1.45–2.86) | NR |
| Bipolar, depressive and anxiety disorders | ||||
| Hallert (1982) [51] Hallert (1983) [52] | Phase 1: cross-sectional Phase 2: case-series | CD, n = 12 (mean age 47 years, 67% females) Controls undergoing cholecystectomy, n = 12 (mean age 47 years, 67% females) | - MMPI depression subscale: Significantly higher scores in CD vs. controls (70.3 ± 12.5 vs. 59.2 ± 9.3, p <0.01) - MMPI sores: Post-remission in small intestinal mucosa following GFD in CD: no improvement in mood (70 ± 12.5 at point 0 vs. 68 ± 14 at year 1, p = NS) - Post-cholecystectomy in controls: No change in MMPI scores - Supplementation with Vitamin B6 80 mg/day for 6 months: Significant decrease in depressive symptoms (68 ± 14.0 to 56 ± 8.5, p <0.01) | -In patients with CD, significant correlation was found between depression scores and degree of steatorrhea -No correlation was found between abdominal complaints (diarrhea and pain) and depression scores |
| Addolorato (1996) [53] | Case-control | CD, n = 20 (mean age 37 years, 56% females) IBD, n = 16 (mean age 32 years, 56% females) Controls, n = 16 (mean age 35 years, 56% females) | - Prevalence of State anxiety: 62.5% in CD, 50% in IBD, and 31.3% in controls (p = NS). - Prevalence of depression: 68.7% in CD, 37.5% in IBD, and 18.8% in controls (p <0.01 for CD vs. controls only) | NR |
| Ciacci (1998) [54] | Cross-sectional | CD, n = 92 (mean age 29.4 years, 70% females) CPH, n = 48 (mean age 31.8 years, 34% females) Controls, n =100 (mean age 30 years, 71% females) | - Mean scores of the M-SDS: CD: 31.81 ± 7.84 CPH: 28.73 ± 7.09 (p = 0.038 vs. CD) Controls: 27.14 ± 5.26 (p <0.0001 vs. CD) | - Demographic characteristics did not influence M-SDS scores -Depressive symptoms are present to a similar extent in patients with childhood- and adulthood-diagnosed CD |
| Addolorato (2001) [55] | Phase 1: Cross-sectional Phase 2: Case-series | CD, n = 35 (mean age 29.8 years, 60% females) Controls, n = 59 (mean age 31.7 years, 54% females) | Before diet: - Prevalence of high levels of state anxiety: CD vs. control: 71.4% versus 23.7% (p <0.0001) - Prevalence of high levels of trait anxiety: CD vs. controls: 25.7% versus 15.2% (p = NS) - Prevalence of depression CD vs. controls: 57.1% versus 9.6% (p <0.0001) After 1 year of GFD (T0 vs. T1) - Prevalence of high levels of state anxiety: T0 71.4% versus T1: 25.7% (p <0.001) - Prevalence of high levels of trait anxiety: T0: 25.7% versus T1: 17.1% (p = NS) - Prevalence of depression T0: 57.1% versus T1:45.7 (p = NS) | NR |
| Cicarelli (2003) [56] | Cross-sectional | CD, n = 176 (mean age 30.9 years, 75% females) Controls, n = 52 (mean age 31.7 years, 65% females) | - Prevalence of mood disorders (CD vs. controls): Mood disorders 50 (29%) vs. 9 (17%), p = NS Depression episodes 24 (14%) vs. 7 (13%), p = NS Dysthymia 26 (15%) vs. 2 (4%), p <0.05 | - Adherence to a strict gluten-free diet was associated with a significant reduction of dysthymia |
| Carta (2002) [57] Carta (2003) [58] | Cross-sectional | CD, n = 36 (mean age 41.1 years, 75% females) Controls, n = 144 (mean age 41.3 years, 75% females) | - Lifetime prevalence of psychiatric disorders (cases vs. controls): Major depressive disorder 15 (41.7%) vs. 30 (20.8%), p = 0.01 Dysthymic disorder 3 (8.3%) vs. 2 (1.4%), p = 0.05 Adjustment disorders 11 (30.5%) vs. 11 (7.6%), p = 0.001 Generalized anxiety disorder 10 (27.7%) vs. 23 (16%), p = NS Panic disorder 5 (13.9%) vs. 3 (2.1%), p = 0.001 Specific phobia 1 (2.7%) vs. 6 (4.2%), p = NS Social phobia 3 (8.3%) vs. 10 (6.9%), p = NS Recurrent brief depression 36.1% versus 6.9% (OR = 7.6; 95% CI: 3.2–17.8) | - Earlier onset of CD was linked to higher prevalence of major depressive disorder - Subclinical thyroid disease appears to represent a significant risk factor for these psychiatric disorders |
| Addolorato (2008) [59] | Cross-sectional | CD, n = 40 (mean age 38 years, 86% females) HC, n = 50 (mean age 36 years, 80% females) | - Prevalence of social phobia: 70% in CD vs. 16% in HC (p <0.0001) - Prevalence of depression: 53% in CD vs. 8% in HC (p <0.0001) | - The prevalence of social phobia or depression in patients with CD did not differ among subjects newly diagnosed with CD and those already on GFD |
| Garud (2009) [60] | Cross-sectional | CD, n = 600 (mean age 54 males & 49 females, 75% females) IBS, n = 200 (mean age 48 males & 45 females, 75% females) Controls, n = 200 (mean age 52 males & 47 females, 75% females) | Prevalence of depression: 17.2% in CD vs. 18.5% in IBS (p = 0.74 vs. CD) and 16% in controls (p = 0.79 vs. CD) | - Among CD patients, type I diabetes mellitus was identified as a significant risk factor for depression (p <0.01) with 37% of patients with both CD and type I DM having clinical depression |
| Smith (2012) [61] | Meta-analysis | Eleven studies on depression and eight studies on anxiety were included | - Depression is more common and severe in CD than in healthy adults with an overall effect size of 0.97 - Anxiety did not differ significantly between CD and healthy adults - No differences in depression or anxiety in CD vs. other medical disorders | - Other medical conditions included: Crohn’s disease, DM, IBD, lactose intolerance, surgery patients, CPH |
| Peters (2014) [62] | Randomized, double-blind, cross-over trial | NCGS, n = 22 (median age 48 years, 77% females) | - Gluten ingestion effect on STPI depression scores: Significantly higher scores in CD vs. controls (mean difference = 2.03, 95% CI: 0.55–3.51, p = 0.01) - No differences in other STPI state indices or for any STPI trait measures | NR |
| Carta (2015) [63] | Cross-sectional | CD, n = 46 (mean age 41 years, 83% females) Controls, n = 240 (mean age 41 years, 83% females) | - Prevalence of depression: 30.0% in CD vs. 8.3% in controls, p <0.0001 - Prevalence of panic disorder: 18.3% in CD vs. 5.4% in controls, p <0.001 - Prevalence of bipolar disorder: 4.3% in CD vs. 0.4% in controls, p <0.005 | - Patients with CD but without comorbidity with major depression, panic disorder, or bipolar disorder do not show worse QOL than controls |
| Di Sabatino (2015) [64] | Randomized, double-blind, placebo-controlled cross-over trial | NCGS, n = 61 (mean age 39 years, 87% females) randomly assigned to: Gluten 4.375 mg/day for 1 week Placebo 4.375 g/day rice starch for 1 week Wash-out period: 1 week | - Depression was significantly worsened by gluten ingestion (p = 0.02) | NR |
| Tortora (2013) [65] | Cross-sectional | CD, n = 70 (mean age 33 years) Controls, n = 70 (mean age 32 years) | - EPDS scores in CD women vs. controls: 9.9 ± 5.9 vs. 6.7 ± 3.7, p <0.01 - EPDS > 10: 47% in CD vs. 14% in controls (OR = 3.3, p <0.01) - PPD diagnosis: 41% of CD women with vs. 11% in controls (p <0.01) | - A significant association was observed between the onset of PPD and a previous menstrual disorder in women suffering from CD - QOL scores were significantly higher in women with CD |
| Sainsbury (2018) [66] | Meta-analysis | Eight studies were included in quantitative analysis (total n = 1644, mean age ranged from 39 to 57 years, % of females ranged from 76.6% to 100%) | - Moderate association between poor adherence to GFD and greater depressive symptoms (r = 0.398, 95% CI: 0.32–0.47) with marked heterogeneity in effects (I2 = 66.8%) - Exclusion of studies with high or unclear risk of bias did not alter the results | - Poorer QOL was correlated with a higher incidence of psychological and gastrointestinal symptoms, greater reliance on maladaptive coping strategies, and poorer GFD adherence |
| Feeding and eating disorders | ||||
| Passananti (2013) [67] | Cross-sectional | CD, n = 100 (mean age 29 years, 72% females) HC, n = 100 (mean age 30 years, 68% females) | - BES ≥ 17: 6% in CD vs. 0% controls (p = NS) - Women with CD had significantly higher scores in pulse thinness, social insecurity, perfectionism, inadequacy, ascetisim, and interpersonal diffidence compared to HC women of the Eating Disorder Inventory - EAT-26 ≥ 20: 16% in CD vs. 4% in HC (p = 0.01) - SRDS > 44: 39% in CD vs. 6% in controls (p <0.001) - SCL-90 pathological scores: 42% in CD vs. 6% in HC (p <0.0001) | - EAT-26 demonstrated association between indices of diet-related disorders in both CD and the female gender after controlling for anxiety and depression |
| Satherley (2016) [68] | Cross-sectional | CD, n = 157 (mean age 38 years, sex NR) IBD, n = 116 (mean age 36 years, sex NR) DM-type 2, n = 88 (mean age 47 years, sex NR) HC, n = 142 (mean age 33 years, sex NR) | - EAT-26 > 20: 15.7% in CD vs. 8.8% in DM and 3.8% in HC (p <0.05) - BES > 17: 19.4% in CD vs. 2.3% in controls (p <0.05) - Mean EAT-26 and BES scores: 11.1 in CD vs. 7.7 in controls (p <0.05) and 11.2 in CD vs. 3.9 in controls (p <0.05), respectively - Significant associations between EAT-26 and BES scores with DASS-21 scores were reported (p <0.008) | - Dietary-management and gastrointestinal symptoms were significantly associated with EAT scores in CD |
| Mårild (2017) [69] | Population-based cohort study | Group 1, n = 17,959 (median age 28 years); Matched controls, n = 89,379 Group 2, n = 7455 (median age 46 years); Matched controls, n = 36,940 Group 3, n = 2307 (median age 38 years); Matched controls, n = 11,499 | - Risk of developing anorexia nervosa: Group 1: HR= 1.46 (95% CI: 1.08–1.98) Group 2: HR=2.12 (95% CI: 0.97–4.67) Group 3: HR=2.45 (95% CI: 1.10–5.45) - Adjustment for education level, socioeconomic status, and type 1 DM didn’t affect conclusions in all groups | - There was no significantly increased risk for subsequent anorexia nervosa among males with CD |
| Sleep-Wake disorders | ||||
| Mårild (2015) [70] | Population-based cohort study | CD, n = 2933 (median age 28 years, 61.2% females) Controls, n = 14,571 (median age 28 years, 61.3 females) | - Poor sleep in CD vs. controls: 12.5% vs. 9.8% (HR = 1.36, 95% CI: 1.30–1.41) - Individuals with CD had a similar increased risk irrespective of age at CD diagnosis, sex and type of hypnotic used | - Overall, poor sleep was more prevalent in females than in males. However, differences in risk estimates for poor sleep were small between females and males with CD - Adjustment for sleep apnea and restless leg syndrome did not influence the risk of poor sleep in CD |
| Substance-related and addictive disorders | ||||
| Roos (2006) [71] | Cross-sectional | CD, n = 51 (age 45–64 years, 59% females) Controls, n = 182 (age 45–64 years, 57% females) | - PGWB index scores: 103 (95% CI: 99–107) in CD vs. 103 (95% CI: 100–106) in controls (p = NS) | - Males with CD tended to score higher on the PGWB domains than the male controls - CD women scored somewhat lower in the PGWB domains than the female controls - CD men tended to score higher than the CD women in all six domains of the PGWB |
| Gili 2013 [72] | Cross-sectional | CD, n = 3327 (mean age 49 years and 70% females). Controls, n = 5,471,988) (mean age 58 years and 54% females). | - Prevalence of alcohol disorders: 4.9% in CD vs. 6.3% in controls (p = 0.0009) | - The presence of alcohol disorders in CD increased the length of stay, costs and had an excess of mortality |
| Neurocognitive Disorders | ||||
| Lebwohl (2016) [73] | Population-based cohort | CD, n = 8846 (mean age 64 years and 56% females). Control, n = 43,474 (mean age 64 years and 56% females). | - In a median follow-up period of 8.4 years: 4.3% of CD patients and 4.4% of controls had a diagnosis of dementia (HR 1.07; 95% CI 0.95–1.20) - A subgroup analysis showed an increased risk of vascular dementia (HR 1.28; 95% CI 1.00–1.64) | - A significant association between CD and dementia among the age group 60–69 was found, which was not present in the younger or older age groups - Increased risk of dementia was found in the first year following CD diagnosis |
| Various psychiatric conditions | ||||
| Fera (2003) [74] | Cross-sectional | CD, n = 100 (mean age 40 years, 75% females) DM, n = 100 (mean age 53 years, 74% females) HC, n = 100 (mean age 41 years, 68% females) | - CD, prevalence of OCD 28%, depressive disorder/dysthymia 19% - DM, prevalence of OCD 0%, depressive disorder/dysthymia 10% HC, anxiety and depression in 10% of subjects | - QOL was poorer in both CD and diabetic patients than in healthy controls and significantly correlated with anxiety |
| Sainsbury (2013) [75] | Study 1: cross-sectional | n = 390 (mean age 44 years, 82.8% females) | - Severe gastrointestinal symptoms at CD diagnosis were associated with: increased depression (r = 0.28, p <0.001), anxiety (r = 0.29, p <0.001), stress (r = 0.28, p <0.001), eating disorder (r = 0.15, p <0.01), and emotion-oriented coping (r = 0.17, p <0.01) | - Poorer QOL was significantly associated with a greater number and longer duration of CD symptoms prior to diagnosis - Higher number of symptoms was associated with poorer QOL - There were no gender differences in QOL, although females reported a greater number of symptoms - More severe gastrointestinal symptoms at diagnosis were also associated with increased psychological manifestations |
| Study 2: cross-sectional | n = 189 (mean age 46.5 years, 87.3% females) | - Hierarchical regression analyses: Current psychological distress significantly contributed to poor QOL (accounting for 23.8% of the variance in QOL) | ||
| Zylberberg (2017) [76] | Population-based cross-sectional | Diagnosed CD, n = 27 (age NR, 78% females) Undiagnosed CD, n = 79 (age NR, 58% females) PWAG; n = 213 (age NR, 55% females) Controls; n = 14,769 (demographic characteristics NR) | - Prevalence of depression: 8.2% of controls vs. 3.9% in CD (p = 0.18) and 2.9% in PWAGs (0.002) - Prevalence of sleep difficulty: 37.3% in CD, 34.1% in PWAGs vs. 27.4% in controls (p = NS) - Multivariate analysis adjusted for race/ethnicity, annual household income, number of healthcare visits: PWAGS, significantly lower odds of depression (OR = 0.25, 95% CI: 0.12–0.5, p = 0.0001) CD, OR = 0.30; 95% CI: 0.08–1.19, p = 0.09 | - QOL: The presence of physical, mental, and emotional limitations was reported in 2.9% of controls vs. 13.8% diagnosed CD (p =0.004), 9.6% with undiagnosed CD (p = 0.02), and 5.1% in PWAGs (p = 0.18) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Slim, M.; Rico-Villademoros, F.; Calandre, E.P. Psychiatric Comorbidity in Children and Adults with Gluten-Related Disorders: A Narrative Review. Nutrients 2018, 10, 875. https://doi.org/10.3390/nu10070875
Slim M, Rico-Villademoros F, Calandre EP. Psychiatric Comorbidity in Children and Adults with Gluten-Related Disorders: A Narrative Review. Nutrients. 2018; 10(7):875. https://doi.org/10.3390/nu10070875
Chicago/Turabian StyleSlim, Mahmoud, Fernando Rico-Villademoros, and Elena P. Calandre. 2018. "Psychiatric Comorbidity in Children and Adults with Gluten-Related Disorders: A Narrative Review" Nutrients 10, no. 7: 875. https://doi.org/10.3390/nu10070875
APA StyleSlim, M., Rico-Villademoros, F., & Calandre, E. P. (2018). Psychiatric Comorbidity in Children and Adults with Gluten-Related Disorders: A Narrative Review. Nutrients, 10(7), 875. https://doi.org/10.3390/nu10070875





