A Comparative Study on Carbohydrate Estimation: GoCARB vs. Dietitians
Abstract
1. Introduction
2. Materials and Methods
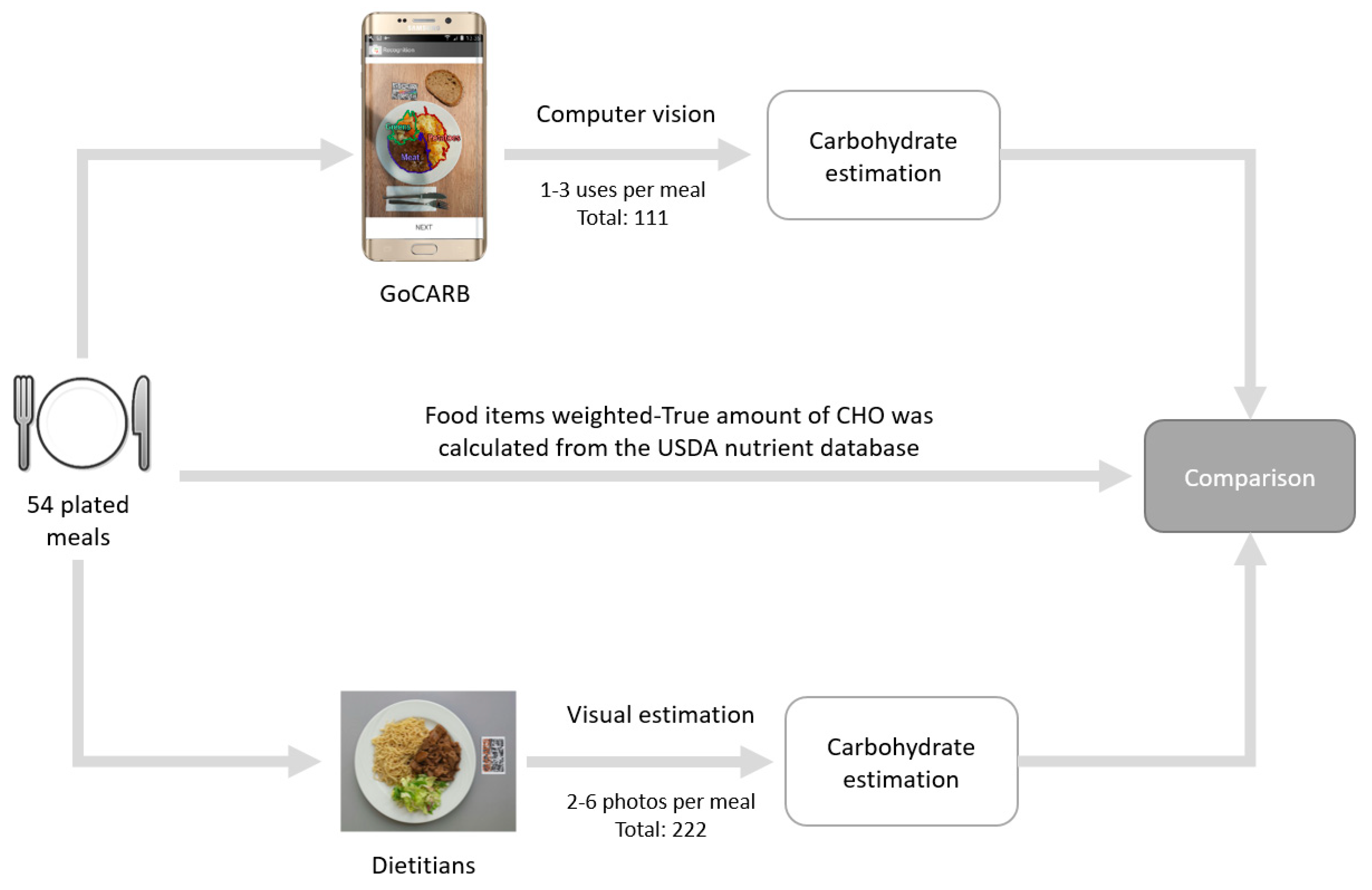
2.1. Study Design
2.2. Participants
2.3. Data Analysis
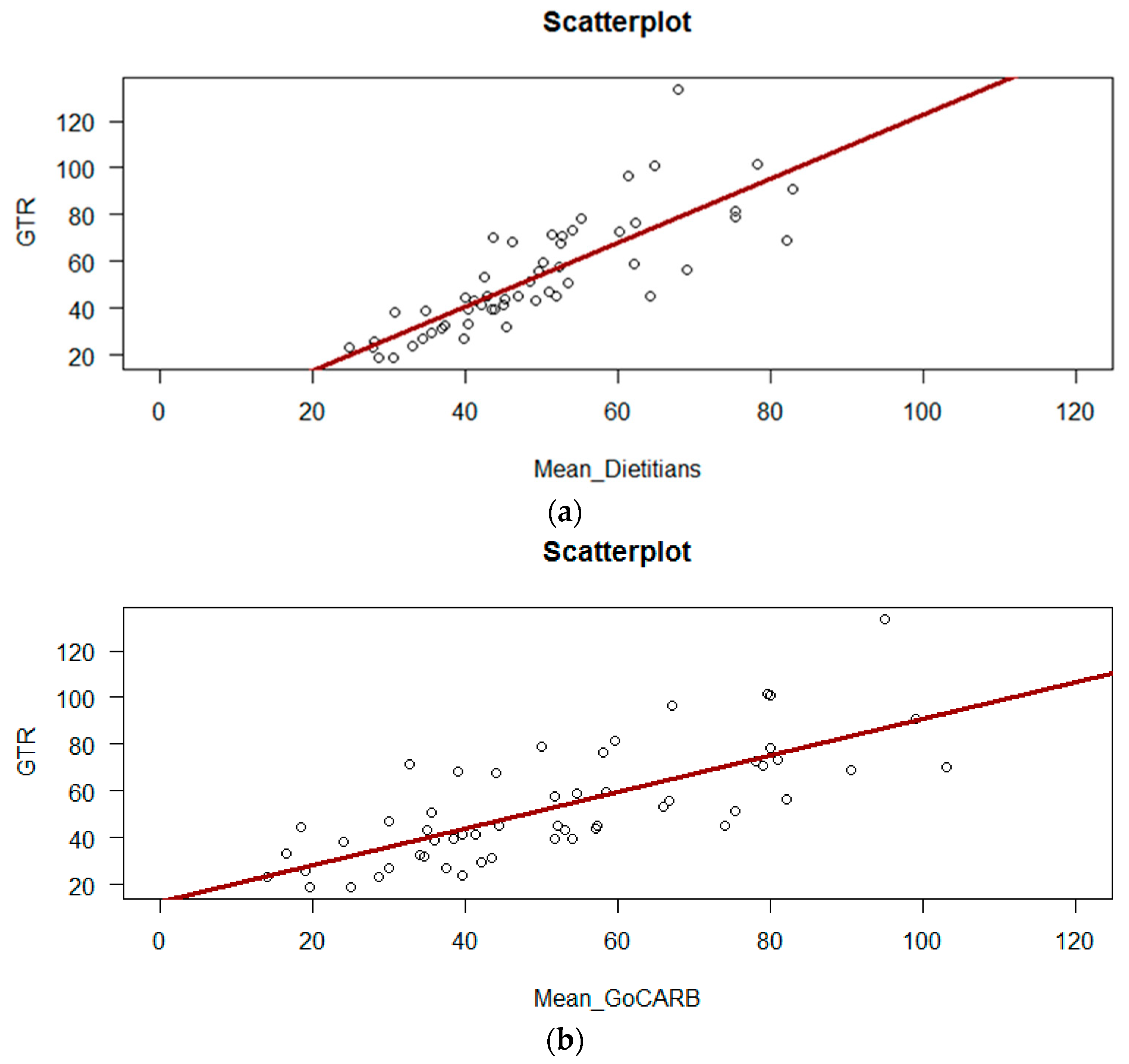
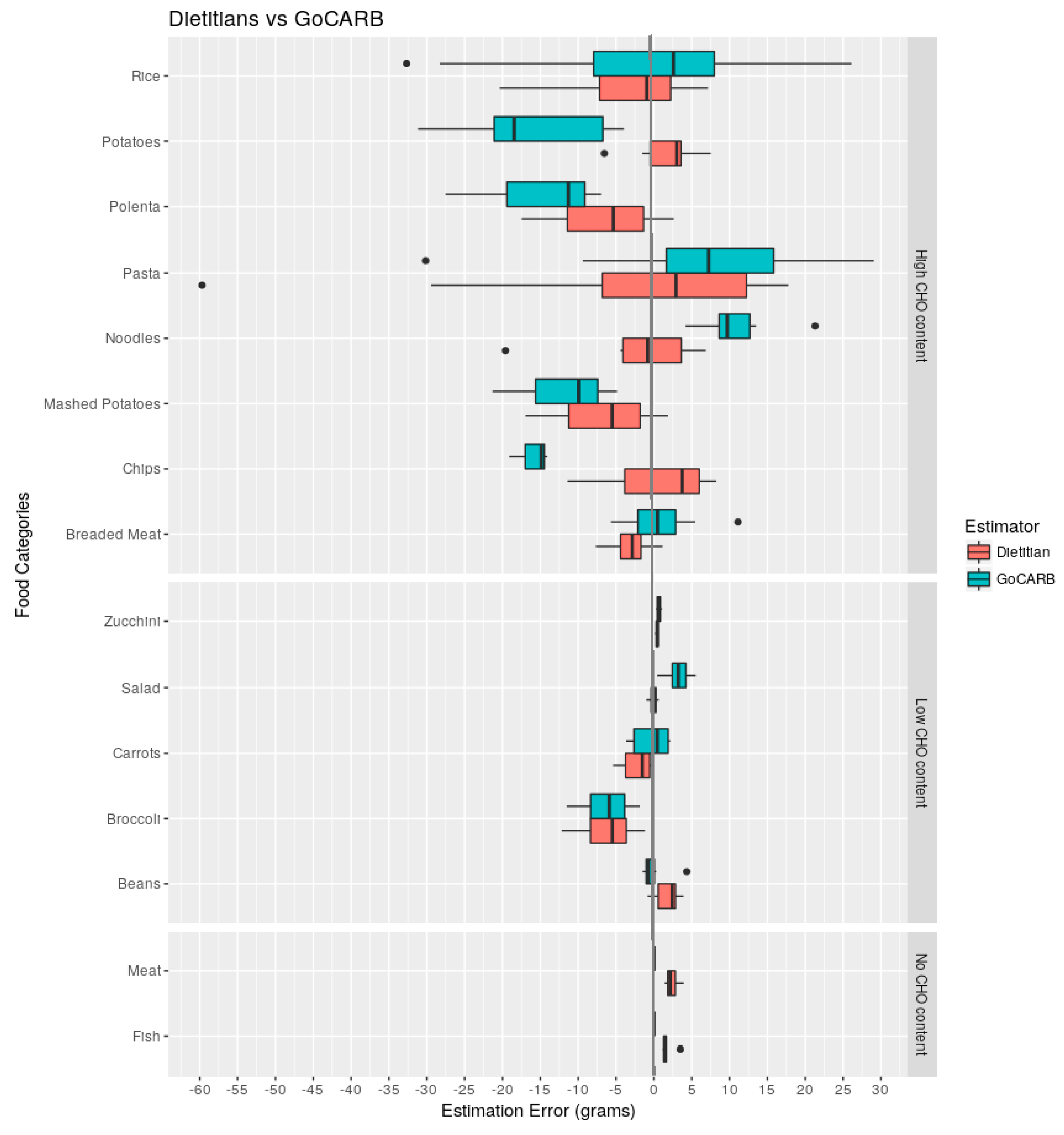
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| T1D | Type 1 diabetes |
| App | Application |
| CHO | Carbohydrate |
| GTR | Ground truth |
| ICC | Intraclass correlation coefficients |
References
- IDF. IDF Diabetes Atlas; International Diabetes Federation: Brussels, Belgium, 2015; pp. 1–163. [Google Scholar]
- American Diabetes Association (ADA). Standard of Medical Care in Diabetes—2017 (s4–s5, s33–s43). Available online: https://professional.diabetes.org/sites/professional.diabetes.org/files/media/standardofcare2017fulldeckfinal_0.pdf (accessed on 3 May 2018).
- Fu, S.; Li, L.; Deng, S.; Zan, L.; Liu, Z. Effectiveness of advanced carbohydrate counting in type 1 diabetes mellitus: A systematic review and meta-analysis. Sci. Rep. 2016, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bell, K.J.; Barclay, A.W.; Petocz, P.; Colagiuri, S.; Brand-Miller, J.C. Efficacy of carbohydrate counting in type 1 diabetes: A systematic review and meta-analysis. Lancet Diabet. Endocrinol. 2014, 2, 133–140. [Google Scholar] [CrossRef]
- Brazeau, A.S.; Mircescu, H.; Desjardins, K.; Leroux, C.; Strychar, I.; Ekoé, J.M.; Rabasa-Lhoret, R. Carbohydrate counting accuracy and blood glucose variability in adults with type 1 diabetes. Diabet. Res. Clin. Pract. 2013, 99, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Meade, L.T.; Rushton, W.E. Accuracy of carbohydrate counting in adults. Clin. Diabet. 2016, 34, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, T.; Takamura, C.; Hirose, M.; Hashimoto, T.; Higashide, T.; Kashihara, Y.; Hashimura, K.; Shintaku, H. The factors affecting on estimation of carbohydrate content of meals in carbohydrate counting. Clin. Pediatr. Endocrinol. 2015, 24, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Bishop, F.K.; Maahs, D.M.; Spiegel, G.; Owen, D.; Klingensmith, G.J.; Bortsov, A.; Thomas, J.; Mayer-Davis, E.J. The carbohydrate counting in adolescents with type 1 diabetes (CCAT) study. Diabet. Spectr. 2009, 22, 56–62. [Google Scholar] [CrossRef]
- Smart, C.E.; Ross, K.; Edge, J.A.; King, B.R.; McElduff, P.; Collins, C.E. Can children with type 1 diabetes and their caregivers estimate the carbohydrate content of meals and snacks? Diabet. Med. 2010, 27, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Smart, C.E.; King, B.R.; Mcelduff, P.; Collins, C.E. In children using intensive insulin therapy, a 20-g variation in carbohydrate amount significantly impacts on postprandial glycaemia. Diabet. Med. 2012, 29, 21–24. [Google Scholar] [CrossRef] [PubMed]
- El-Gayar, O.; Timsina, P.; Nawar, N.; Eid, W.; Elizabeth, S. Mobile applications for diabetes self-management: Status and potential. J. Diabet. Sci. Technol. 2013, 7, 247–262. [Google Scholar] [CrossRef] [PubMed]
- Kawano, Y.; Yanai, K. FoodCam: A real-time food recognition system on a smartphone. Multimed. Tools Appl. 2015, 74, 5263–5287. [Google Scholar] [CrossRef]
- Kong, F.; Tan, J. DietCam: Automatic dietary assessment with mobile camera phones. Pervasive Mob. Comput. 2012, 8, 147–163. [Google Scholar] [CrossRef]
- Puri, M.; Zhu, Z.; Yu, Q.; Divakaran, A.; Sawhney, H. Recognition and volume estimation of food intake using a mobile device. In Proceedings of the 2009 IEEE Workshop on Applications of Computer Vision (WACV), Snowbird, UT, USA, 7–8 December 2009; pp. 1–8. [Google Scholar]
- Zhu, F.; Bosch, M.; Khanna, N.; Boushey, C.J.; Delp, E.J. Multiple Hypotheses Image Segmentation and Classification with Application to Dietary Assessment. IEEE J. Biomed. Health Inform. 2015, 19, 377–388. [Google Scholar] [CrossRef] [PubMed]
- Ciocca, G.; Napoletano, P.; Schettini, R. Food Recognition and Leftover Estimation for Daily Diet Monitoring. In Proceedings of the ICIAP 2015 18th International Conference on Image Analysis and Processing, Genoa, Italy, 7–11 September 2015; Volume 9281, pp. 334–341. [Google Scholar]
- Anthimopoulos, M.; Dehais, J.; Shevchik, S.; Ransford, B.H.; Duke, D.; Diem, P.; Mougiakakou, S. Computer vision-based carbohydrate estimation for type 1 patients with diabetes using smartphones. J. Diabet. Sci. Technol. 2015, 9, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, K.; Yamasaki, T.; Aizawa, K. FoodLog: Capture, Analysis and Retrieval of Personal Food Images via Web. In Proceedings of the ACM Multimedia 2009 Workshop on Multimedia for Cooking and Eating Activities, Beijing, China, 23 October 2009. [Google Scholar]
- Beijbom, O.; Joshi, N.; Morris, D.; Saponas, S.; Khullar, S. Menu-match: Restaurant-specific food logging from images. In Proceedings of the 2015 IEEE Winter Conference on Applications of Computer Vision, WACV, Big Island, HI, USA, 6–9 January 2015; pp. 844–851. [Google Scholar]
- Rhyner, D.; Loher, H.; Dehais, J.; Anthimopoulos, M.; Shevchik, S.; Botwey, R.H.; Duke, D.; Stettler, C.; Diem, P.; Mougiakakou, S. Carbohydrate estimation by a mobile phone-based system versus self-estimations of individuals with type 1 diabetes mellitus: A comparative study. J. Med. Internet Res. 2016, 18, e10. [Google Scholar] [CrossRef] [PubMed]
- Bally, L.; Dehais, J.; Nakas, C.T.; Anthimopoulos, M.; Laimer, M.; Rhyner, D.; Rosenberg, G.; Zueger, T.; Diem, P.; Mougiakakou, S.; et al. Carbohydrate estimation supported by the GoCARB system in individuals with type 1 diabetes: A randomized prospective pilot study. Diabet. Care 2017, 40, e6–e7. [Google Scholar] [CrossRef] [PubMed]
- Dehais, J.; Anthimopoulos, M.; Shevchik, S.; Mougiakakou, S. Two-view 3D reconstruction for food volume estimation. IEEE Trans. Multimed. 2017, 19, 1090–1099. [Google Scholar] [CrossRef]
- Wheeler, M.L.; Daly, A.; Evert, A.; Franz, M.J.; Geil, P.; Holzmeister, L.A.; Kulkarni, K.; Loghmani, E.; Ross, T.A.; Woolf, P. Choose Your Foods: Exchange Lists for Diabetes, Sixth Edition, 2008: Description and Guidelines for Use. J. Am. Diet. Assoc. 2008, 108, 883–888. [Google Scholar] [CrossRef]
- Howes, E.; Boushey, C.J.; Kerr, D.A.; Tomayko, E.J.; Cluskey, M. Image-based dietary assessment ability of dietetics students and interns. Nutrients 2017, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Williamson, D.; Allen, H.; Martin, P.; Alfonso, A.; Gerald, B.; Hunt, A. Comparison of digital photography to weight visual estimation of portion sizes. J. Am. Diet. Assoc. 2003, 103, 1139–1145. [Google Scholar] [CrossRef]
- Japur, C.C.; Diez-Garcia, R.W. Food energy content influences food portion size estimation by nutrition students. J. Hum. Nutr. Diet. 2010, 23, 272–276. [Google Scholar] [CrossRef] [PubMed]
- Timon, C.M.; Cooper, S.E.; Barker, M.E.; Astell, A.J.; Adlam, T.; Hwang, F.; Williams, E.A. A comparison of food portion size estimation by older adults, young adults and nutritionists. J. Nutr. Health Aging 2018, 22, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Byrd-Bredbenner, C.; Schwartz, J. The effect of practical portion size measurement aids on the accuracy of portion size estimates made by young adults. J. Hum. Nutr. Diet. 2004, 17, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Gittelsohn, J.; Shankar, A.V.; Pokhrel, R.P.; West, K.P. Accuracy of estimating food intake by observation. J. Am. Diet. Assoc. 1994, 94, 1273–1277. [Google Scholar] [CrossRef]
- Lee, C.D.; Chae, J.; Schap, T.; Kerr, D.A.; Delp, E.J.; Ebert, D.S.; Boushey, C.J. Comparison of Known Food Weights with Image-Based Portion-Size Automated Estimation and Adolescents’ Self-Reported Portion Size. J. Diabet. Sci. Technol. 2012, 6, 428–434. [Google Scholar] [CrossRef] [PubMed]
- US Department of Agriculture, Agricultural Research Service, Nutrient Data Laboratory. USDA National Nutrient Database for Standard Reference, Release 28. Version Current: September 2015, Slightly Revised May 2016. Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 3 May 2018).

| Study Demographics | Dietitians (n = 6) |
|---|---|
| Age (mean, SD) | 38.7 years (10.8) |
| Years of experience | 12.2 (9) |
| Exhibiting T1D | 1/6 |
| Meal Size | Absolute Error (grams), Mean ± SD | p-Value | |
|---|---|---|---|
| Dietitians | GoCARB | ||
| Small (n = 16) | 5.9 ± 3.5 | 8.5 ± 5.6 | 0.18 |
| Medium (n = 16) | 7.6 ± 6.3 | 11.3 ± 8.9 | 0.27 |
| Large (n = 16) | 19.4 ± 15.2 | 20.7 ± 11.6 | 0.41 |
| Meal Size | SD (grams) of Dietitians Errors | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |
| Small (n = 14) | 3.8 | 4.7 | 3.0 | 7.9 | 3.6 | 0.1 |
| Medium (n = 14) | 4.8 | 7.5 | 3.4 | 5.7 | 3.7 | 1.7 |
| Large (n = 14) | 5.3 | 6.9 | 3.5 | 8.6 | 6.1 | 2.7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasiloglou, M.F.; Mougiakakou, S.; Aubry, E.; Bokelmann, A.; Fricker, R.; Gomes, F.; Guntermann, C.; Meyer, A.; Studerus, D.; Stanga, Z. A Comparative Study on Carbohydrate Estimation: GoCARB vs. Dietitians. Nutrients 2018, 10, 741. https://doi.org/10.3390/nu10060741
Vasiloglou MF, Mougiakakou S, Aubry E, Bokelmann A, Fricker R, Gomes F, Guntermann C, Meyer A, Studerus D, Stanga Z. A Comparative Study on Carbohydrate Estimation: GoCARB vs. Dietitians. Nutrients. 2018; 10(6):741. https://doi.org/10.3390/nu10060741
Chicago/Turabian StyleVasiloglou, Maria F., Stavroula Mougiakakou, Emilie Aubry, Anika Bokelmann, Rita Fricker, Filomena Gomes, Cathrin Guntermann, Alexa Meyer, Diana Studerus, and Zeno Stanga. 2018. "A Comparative Study on Carbohydrate Estimation: GoCARB vs. Dietitians" Nutrients 10, no. 6: 741. https://doi.org/10.3390/nu10060741
APA StyleVasiloglou, M. F., Mougiakakou, S., Aubry, E., Bokelmann, A., Fricker, R., Gomes, F., Guntermann, C., Meyer, A., Studerus, D., & Stanga, Z. (2018). A Comparative Study on Carbohydrate Estimation: GoCARB vs. Dietitians. Nutrients, 10(6), 741. https://doi.org/10.3390/nu10060741






