1. Introduction
In conventional approaches to complementary feeding, parents typically spoon-feed their infant puréed foods, with gradual introduction of a wider variety of tastes and textures as their experience with solid foods develops. It is not until around 12 months of age that the infant is generally expected to be consuming the same foods as the rest of the family [
1,
2]. Baby-Led Weaning (BLW) is an alternative method for introducing solids where the infant feeds themselves all their foods from the start of the complementary feeding period. This self-feeding necessitates the use of handheld or finger foods as children of this age do not have the manual dexterity to use utensils to feed themselves puréed food [
3]. Parents are also encouraged to include their infant in family meals [
4]. As a result, BLW might be expected to result in differences in food, and therefore nutrient, intake at least in the early months of complementary feeding, and perhaps into childhood.
There is growing international interest in BLW with many parents using the approach and yet, surprisingly, we know almost nothing about the food and nutrient intake of infants who are following a baby-led approach to infant feeding. Retrospective questionnaire data indicate that different foods are offered as first foods. Infants following BLW are more likely to be offered fruit, vegetables, bread and rusks, whereas infant cereal (which would usually be fortified) is the main first food for conventionally fed infants [
3,
5,
6,
7]. In addition, concern has been expressed that energy and iron intakes may be inadequate and sodium intakes high [
8]. However, only one study appears to have examined food intake using more rigorous prospective tools. Morison et al. [
9] analyzed weighed diet records collected in a small sample of 6–8 month old infants following BLW (
n = 25) or traditional spoon-feeding (
n = 26). Despite similar energy intakes, the BLW infants were consuming less protein, carbohydrate and dietary fiber, and more fat (including saturated fat). Lower intakes of iron, zinc, calcium, vitamin B12 and vitamin C were also observed, although no difference was apparent in sodium intakes. Whether these differences persist at older ages has not yet been determined.
In response to concerns from health professionals that unmodified BLW might be associated with increased risk of growth faltering, iron deficiency and choking [
10], the Baby-Led Introduction to SolidS (BLISS) randomized controlled trial investigated the impact of a modified form of BLW on these outcomes. We have shown that this modified version of BLW was associated with a number of benefits including: a reduction in food fussiness compared to traditional spoon-feeding [
11]; higher iron [
12] and zinc [
13] intakes than have been reported for unmodified Baby-Led Weaning [
9]; and no evidence of an increased risk of iron deficiency [
12], choking [
14] or growth faltering [
11]. However, it is not known whether BLISS has beneficial or detrimental impacts on infant food and nutrient intake.
The aim of this paper was to determine the impact of a modified version of BLW, Baby-Led Introduction to SolidS (BLISS), on food and nutrient intake in infants from 7 to 24 months of age.
3. Results
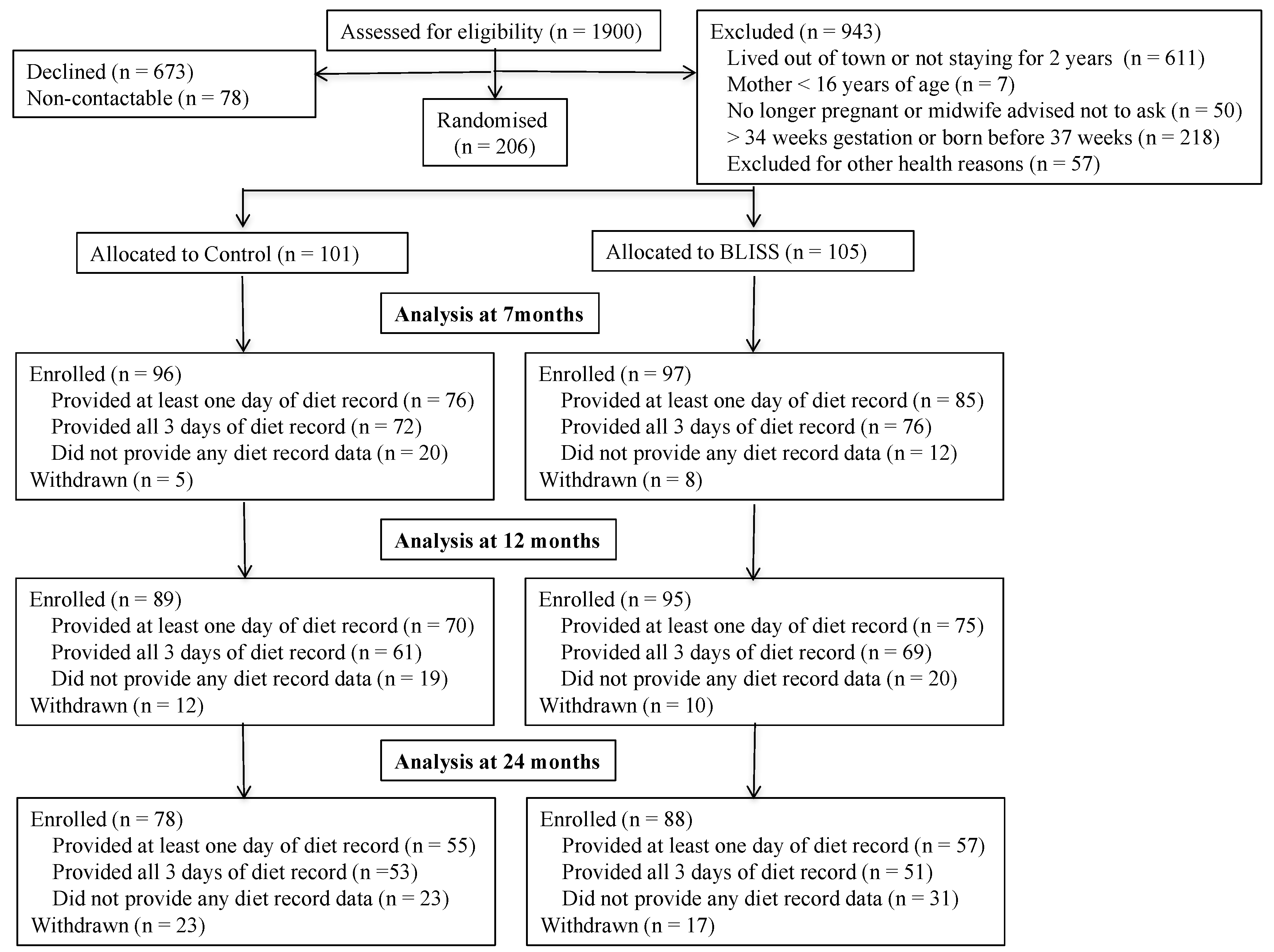
In total, 206 mother–infant pairs were recruited into the study (24% of those eligible and contactable) (
Figure 1). The mothers in the study were predominantly European (82%) and a high proportion (49%) were university educated (
Table 1). Although they were less likely to be from deprived households (
p = 0.07) than the mothers who were not recruited, they reported similar ethnicity and parity (
p ≥ 0.12). Dietary data were provided by 161, 145, and 112 (83%, 79%, and 67% of enrolled) mother–infant pairs at 7 months, 12 months and 24 months of age, respectively. Women who provided at least one diet record for analysis (
n = 165, 80%) were significantly older than those who did not (mean (SD): 32.1 (SD 5.2) compared with 28.2 (SD 6.0) years,
p < 0.001), and were more likely to be University educated (52% compared with 34%,
p = 0.039), but were similar in terms of maternal body mass index (BMI) (
p = 0.426) and household deprivation (
p = 0.945), and did not differ by study group (
p = 0.508).
Infants in the BLISS group were exclusively breastfed for substantially longer (median 21.7 weeks; 95% CI: 13.0 to 23.8 weeks) than those in the Control group (median 17.3 weeks; 95% CI: 6.0 to 21.7 weeks;
p = 0.002) [
11]. In keeping with this, BLISS infants were introduced to solid foods later than Control infants (median 24.6 weeks compared to 22.6 weeks,
p < 0.001) [
12] and, therefore, fewer of them started solids before six months (26 weeks) of age (35% compared to 82%,
p < 0.001).
At seven months of age, as a group, BLISS infants were feeding themselves on average 40% (25th, 75th percentile: 27%, 51%) of their food compared with Control infants who were feeding themselves on average only 9% (0%, 31%) of their food (
p < 0.001) (
Table 2). At 12 months of age, foods were slightly, but significantly, more likely to be fed by an adult in the Control group than in the BLISS group (7% vs. 0% of food;
p = 0.027), but infants in both groups were feeding themselves virtually all of their food by 24 months of age. Infant involvement in family meals also differed between groups (
Table 3). At seven months of age, BLISS infants were 2–4 times as likely to eat meals with their family (79% to 88% of meals compared to 61% to 75%), and to consume the same foods as the family (27% to 42% of foods compared to 9% to 22%), as Control infants. At 12 months of age, BLISS infants were still twice as likely to be eating the same foods as their family at lunch and evening meals (50% to 69% of foods compared to 22% to 55%).
There were no significant group differences in estimated intake of breast milk at 7 (difference BLISS relative to Controls, 95% CI: 0.0 g, −5.1 to 5.1;
p = 1.00 [
12]), 12 (−0.0 g, −0.1 to 0.1;
p = 0.94 [
12]) or 24 (−6 g, −32 to 20;
p = 0.648) months of age. Corresponding differences (95% CI) in infant formula intake were 216 g (−97.2 to 530 g;
p = 0.17) at 7 months and −85 g (−277 to 107 g;
p = 0.38) at 12 months of age [
12]. No infants were consuming infant formula at 24 months of age. BLISS and Control infants also consumed a similar weight of complementary food at each age (mean (SD) in BLISS vs. Control: 7 months 199 g (SD 177) vs. 233 g (SD 177),
p = 0.293; 12 months 627 g (SD 339) vs. 588 g (SD 280),
p = 0.393; 24 months 975 g (SD 348) vs. 920 g (SD 372),
p = 0.367).
There were very few differences in nutrient intake between the BLISS and Control infants (
Table 4 and
Table 5). At seven months of age, BLISS infants ate significantly more total fat (percent difference, 95% CI: 6% more, 1% to 11% more) and sodium (35% more, 19% to 54% more) than Controls, but intakes of all other nutrients were similar. At 12 months, BLISS infants consumed significantly less saturated fat as a percentage of energy (7% less, 14% to 0.4% less) than Control infants. Nutrient intakes were similar for all nutrients at 24 months of age. There was no evidence of differences in nutrient adequacy between groups (data not shown): at seven months of age, intake appeared to be adequate for both groups for all nutrients measured (except total carbohydrate for which no conclusion can be made as the group mean intake was less than the AI [
27], and dietary fiber for which no AI is defined). Although at 12 months of age 19% of infants had inadequate vitamin B12 intakes, and 15% had inadequate calcium intakes, by 24 months the intakes of just 4% and 6% of children, respectively, were inadequate.
As a group, the prevalence of excessive intakes of sodium and added sugars increased with age. Nine percent of infants had sodium intakes greater than the UL at 12 months (3% (n = 2) of Controls, 15% (n = 11) of BLISS; p = 0.018), but by 24 months, two-thirds (68%) of the children had excessive sodium intakes (66% (n = 37) of Controls, 70% (n = 40) of BLISS; p = 0.458). At seven months, 57% of infants consumed at least one sweet snack during the three days of diet recording, and 7% were already consuming 5% or more of their energy as added sugars (12% (n = 9) of Controls, 2% (n = 2) of BLISS; p = 0.013). By 12 months, 31% of children (39% (n = 27) of Controls, 24% (n = 18) of BLISS; p = 0.068) had intakes of added sugars that were higher than recommended, and this had increased to 75% by 24 months of age (80% (n = 45) of Controls, 70% (n = 40) of BLISS; p = 0.229).
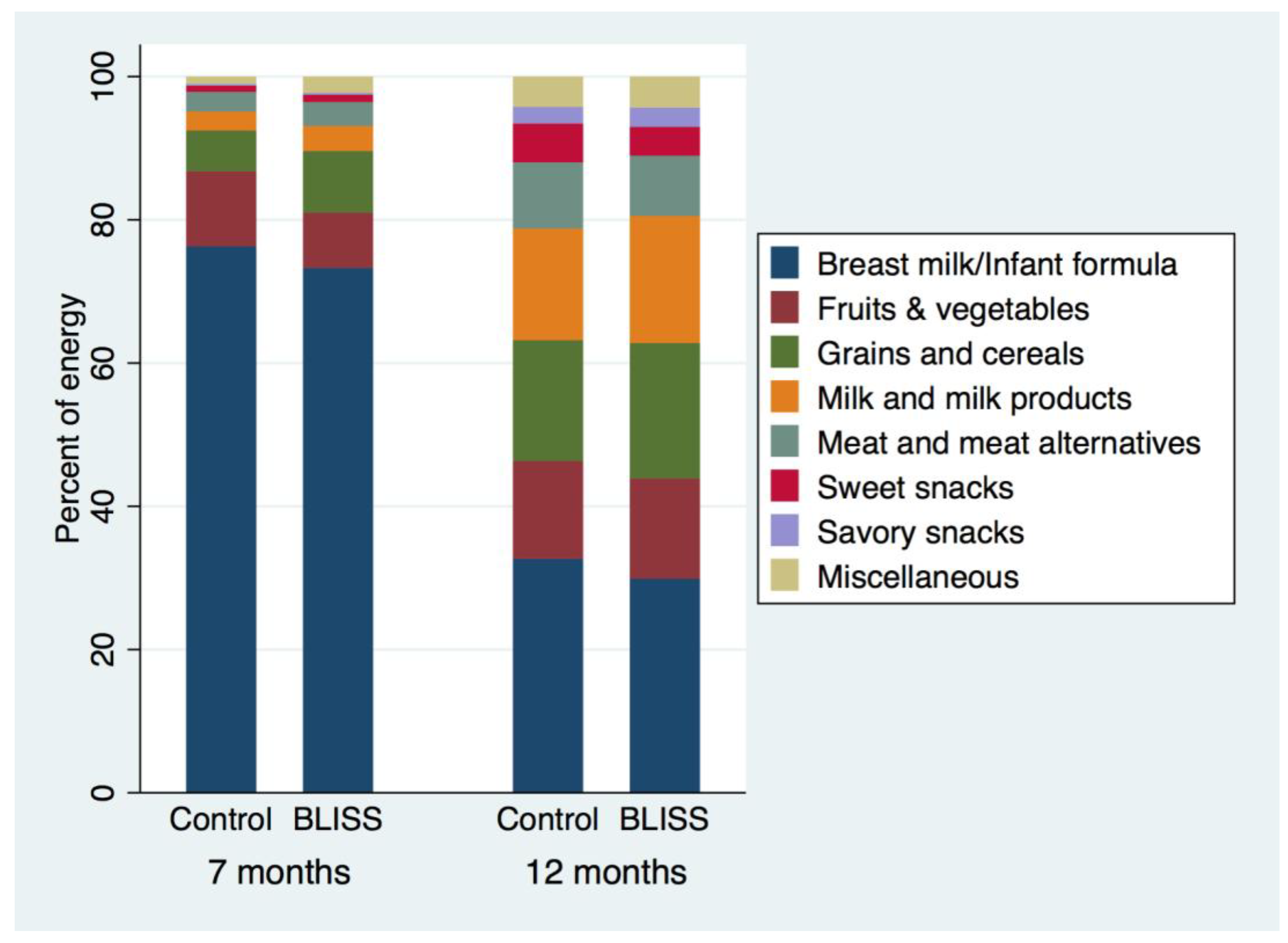
Despite similar energy intakes, the sources of energy differed between BLISS and Control infants at seven months of age (
Figure 3): “grains and cereals” contributed more energy to the diet in BLISS infants (difference, 95% CI: 127 kJ, 62 to 192 kJ), as did “meat and meat alternatives” (42 kJ, 8 to 77 kJ), “milk and milk products” (49 kJ, 17 to 80 kJ), and “miscellaneous foods” (32 kJ, 15 to 49 kJ,
Table 6). By contrast, no significant differences in energy sources were observed at 12 months of age (
Table 7).
4. Discussion
This randomized controlled trial demonstrates that a baby-led approach to introducing solids can meet nutrient requirements at seven months of age, although sodium intakes were higher in infants following Baby-Led Introduction to SolidS than in Controls. Total fat intake was slightly higher at 7 months, and percent energy from saturated fat slightly lower at 12 months, but by 24 months no differences were apparent for any of the nutrients measured. Similarly, intakes of “grains and cereals”, “meat and meat alternatives”, “milk and milk products”, and “miscellaneous foods” were significantly higher in the BLISS infants at 7 months, but by 12 months of age there was no evidence of differences in the food group intakes between infants following a baby-led approach and those following traditional spoon-feeding. Of concern, however, were the high intakes of sodium and added sugars in both groups by two years of age.
It is difficult to compare our findings with the literature given the scarcity of studies investigating food and nutrient intake in infants following a baby-led approach to complementary feeding. Only one small study (
n = 51) has investigated the
nutrient intake of infants following baby-led rather than traditional spoon-feeding approaches to complementary feeding, and only at 6–8 months of age [
9]. Unlike the current study, Morison et al. [
9] observed multiple differences in micronutrient intake between infants following BLW and their traditionally spoon-fed counterparts. Baby-Led Weaned infants consumed less iron (mean percent difference 59%), zinc (21%), calcium (19%), vitamin C (30%) and vitamin B12 (60%) than traditionally spoon-fed infants, although sodium intakes were comparable. In response to concern from health professionals that BLW might be associated with greater risk of growth faltering, iron deficiency, and choking [
10], we made specific modifications to the diet recommended in our BLISS approach [
15]. Presumably as a result of these modifications, BLISS infants appeared to have higher intakes of iron [
12], and possibly zinc [
13], vitamin C, vitamin B12 and calcium than have been reported for infants following unmodified BLW [
9]. Importantly, intakes of iron [
12], zinc [
13], vitamin C, vitamin B12 and calcium were no lower than those of Controls (see discussion of the impact on sodium intake in the next paragraph). The nutritional adequacy of the BLISS approach to infant feeding was further supported by similar growth rates in BLISS and Control infants, and the lack of evidence of an increased risk of growth faltering in those following BLISS [
11]. The earlier study of infants following unmodified BLW also reported significantly lower intakes of protein, carbohydrate, and dietary fiber, and higher intakes of total fat and saturated fat at 6–8 months of age [
9]. Although the current study showed a similarly higher total fat intake as a gram amount, the contribution of fat to total energy intake was not significantly different (unmodified BLW infants consumed 15% more of their energy as fat than Controls (
p < 0.01) [
9], vs. BLISS infants consuming a non-statistically significant 1% (95% CI: −2% to 5%) more of their energy as fat than Controls). The only other macronutrient that appeared to be statistically significantly altered by BLISS was saturated fat (as a percentage of energy but not as a gram amount) which was slightly (7%) lower at 12 months of age. To date, no study has attempted to measure and report
food intake in infants following BLW, although one small observational study reported that, somewhat surprisingly, infants following BLW were exposed to vegetables, fruit, carbohydrates, protein, “meals” and sweet foods less often than infants who were being spoon-fed [
29]. Overall, BLISS appears to result in a diet that is as nutritionally adequate as traditional spoon-feeding, and may address some concerns about the nutritional adequacy of unmodified BLW.
However, sodium intake was significantly higher in infants following BLISS than in Controls at seven months of age. Dietary sodium intake is positively associated with blood pressure in adults [
30] and children [
31]. Furthermore, it has been suggested that if children develop a taste for salty food, it may carry on into adulthood leading to lifelong high sodium intakes [
32]. Concern has been expressed that salt intake may be high in infants following unmodified BLW [
8]—presumably because consuming family meals will lead to infants consuming additional sodium if the family food is salted—although the only other study that has attempted to measure sodium intake in infants following a baby-led approach to complementary feeding saw no difference in sodium intake between BLW infants and age- and sex-matched traditionally spoon-fed controls at 6–8 months of age [
9]. The increased sodium intake of BLISS infants in the current study at seven months of age may be because parents were encouraged to use toast “fingers” as a vehicle for spreads such as iron-fortified infant cereal that they could not otherwise pick up themselves (to improve iron intakes), and also to offer cheese as an energy rich and palatable food (to minimize risk of growth faltering). Both foods are comparatively high in sodium. This suggestion is supported by the significantly higher intake of “grains and cereals” and “milk and milk products” in the BLISS group at seven months. However, the magnitude of the sodium difference at 7 months was not large (it was equivalent to around a ¼ slice of bread), and was not apparent at 12 and 24 months of age. This is in itself interesting as it suggests that, at least for these BLISS infants, even though they had higher intakes of sodium at 7 months, it did not result in them developing a greater preference for salty foods than Controls at 24 months. However, it is concerning that both groups showed a substantial increase in sodium intake with increasing age. In fact, although 9% of the study participants had sodium intakes at 12 months that exceeded the recommended UL (1000 mg/day), by 24 months of age this had increased to 68%. Few studies to date have reported the trajectory of sodium intakes prospectively in such a young cohort.
Parents have also been discouraged from adding any sugar to infants’ diets for many years, both because it is unnecessary and because it may increase liking of sweet foods [
2]. More recently, the World Health Organization has recommended that free sugars should be less than 5% of energy intake in children and adults, in response to increasing international concern about the dose–response relationship between free sugars intake and dental caries (even in populations with water fluoridation) [
28]. In the current study, BLISS and Control infants as a group had similar intakes of added sugars (similar to free sugars but excluding sugars from non-concentrated fruits in juices, jams, jellies, preserves and spreads) at 7, 12 and 24 months of age, and sweet snacks were being consumed in very small amounts by both groups at 7 and 12 months. However, 57% of infants had consumed at least one sweet snack during the three days of diet recording at seven months of age, so foods containing added sugars were being offered to many infants, despite Ministry of Health recommendations [
2]. In addition, 7% were already consuming 5% or more of their total energy intake energy as added sugars. By 12 months, the number with higher than recommended intakes had increased to one in three children, and by 24 months, to three in four. Although BLISS infants were less likely than Controls to have excessive intakes of added sugars at 7 months, with a similar tendency at 12 months, the absolute numbers were 9 Controls and 2 BLISS infants at 7 months, and the difference was no longer apparent by 24 months. To our knowledge, these are the first longitudinal data published on added sugars intakes in infants and toddlers. They show a concerning trend with children already consuming an average of two teaspoons of added sugars a day (7 g in Controls) by one year and four teaspoons (17 g) by two years of age.
In addition to differences in nutrient and food intake between the two groups, particularly at 7 months of age, there were substantial differences in the way in which foods were fed. Infants in the BLISS group were substantially more likely to feed themselves (they self-fed 40% of their foods, compared to 9% for Controls), and were considerably more likely both to eat meals with their family, and to eat the same foods as their family. The only other study to collect data on individual foods and meals at the time of consumption reported even higher levels of self-feeding in 6–8-month old infants following unmodified BLW (77% of foods self-fed) [
9], although the inclusion of eight month old infants in that study at least partially explains the higher rate of self-feeding. In keeping with our findings, high rates of family meal consumption have been widely reported in infants following BLW [
5,
7,
9]. In fact, parents commonly report that the family being able to eat together is one of the advantages of BLW [
6,
10,
33,
34]. It is not clear what the health and social effects of self-feeding and participating in the family meal in infancy might be, but family meal involvement in children and adolescents is associated with consumption of healthier diets, and a reduced risk of overweight [
35].
This study has several strengths including the randomized controlled design, rigorous assessment of dietary intake at multiple time points, and high levels of adherence. We took care to teach parents the importance of accurately measuring foods that were offered but not eaten (i.e., “leftovers”), given this is a particular challenge in this age group. We believe that parents did indeed record actual intake and uneaten foods well, a belief supported by median energy intakes reflecting current Estimated Energy Requirements [
26]. We carefully assessed adherence to ensure that any lack of effect (if observed) was not a result of poor adherence given the randomized trial design. Who fed the infant was assessed for each individual food and drink item consumed over the three days of diet recording, and parents detailed whether each meal was the same as, or almost the same as, that consumed by the rest of the family, and whether meals were eaten together; all important components of a baby-led approach to feeding [
4]. Indeed, these data revealed that substantially more foods were self-fed by infants in the BLISS group, and that BLISS infants were considerably more likely to consume meals with their families, and eat the same foods, than Controls.
The study also has some limitations. The sample was relatively small and as such should be treated as exploratory. We have reported confidence intervals to demonstrate the range of plausible values for the differences. Power calculations for energy and nutrient intake were not performed as the primary outcome of the study was to determine differences in child growth [
11]. Not all parents completed diet records (20% provided none) and these participants were significantly younger and less well educated than parents who did provide dietary data, although the degree of household deprivation and maternal BMI were comparable. Overall, our BLISS study recruited a relatively socio-economically advantaged group, meaning that our findings may not apply to other groups, although 21% came from households with a high level of deprivation. Finally, we had to estimate breast milk volume which will have introduced some error, and future studies should endeavor to use more accurate measures of intake such as the deuterium oxide dilution “dose-to-mother” technique [
36] where feasible. There is, however, no reason to believe that the estimates of breast milk intake differentially influenced the BLISS intake data because, although the BLISS group breastfed exclusively for longer, they were no more likely to be breastfeeding than Controls at 7 months of age, and there was no evidence of a difference between groups in the amount of complementary foods eaten.
In conclusion, BLISS appears to result in a diet that is as nutritionally adequate as traditional spoon-feeding, and may address some concerns about the nutritional adequacy of unmodified BLW. However, BLISS and Control infants both had high intakes of sodium and added sugars by 24 months that are concerning.