Dietary Salt Restriction in Chronic Kidney Disease: A Meta-Analysis of Randomized Clinical Trials
Abstract
1. Introduction
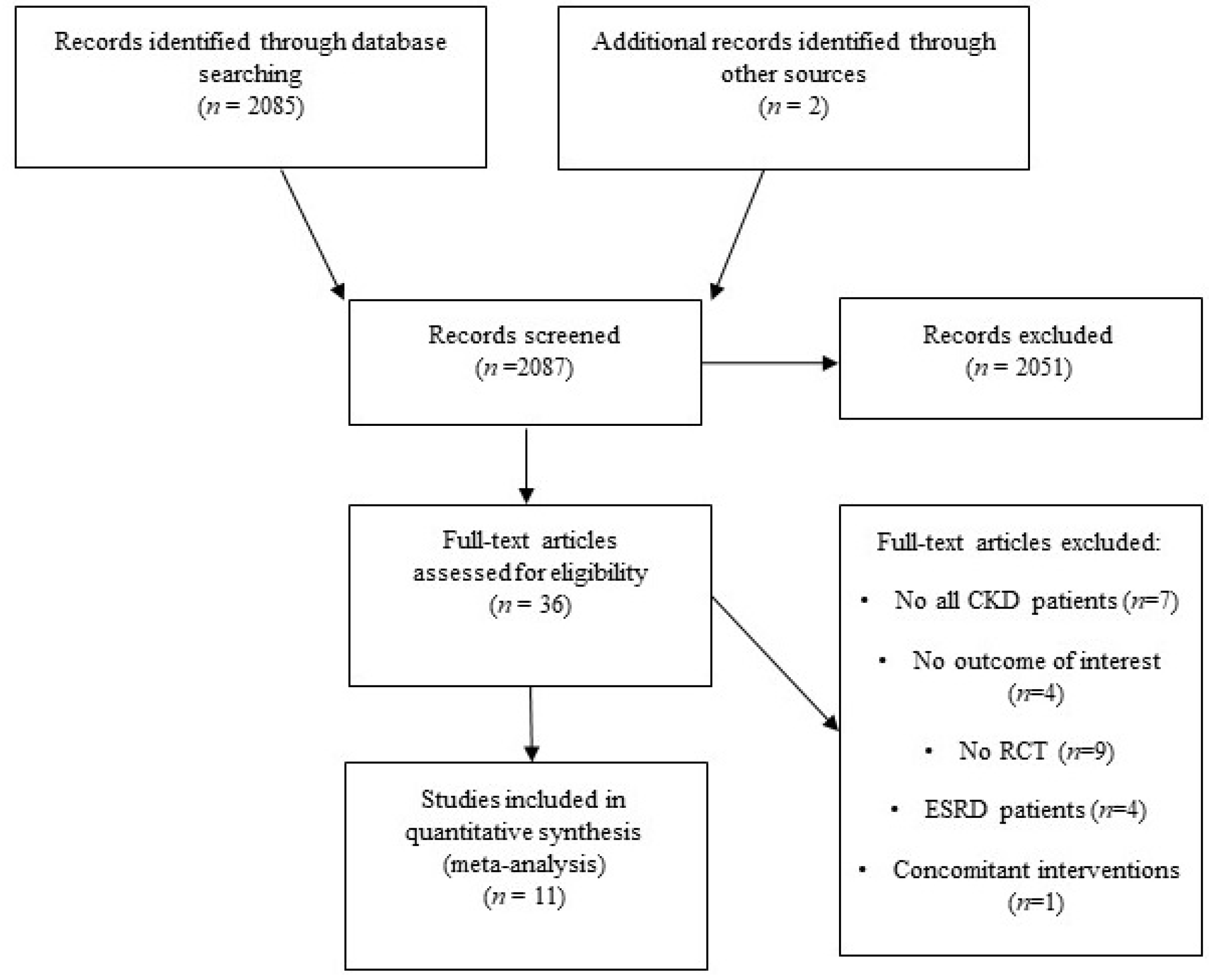
2. Materials and Methods
2.1. Search Strategies
2.2. Study Selection
2.3. Assessment of Risk of Bias in Included Studies
2.4. Statistical Analysis
3. Results
3.1. Characteristics of Studies
3.2. Patient Characteristics
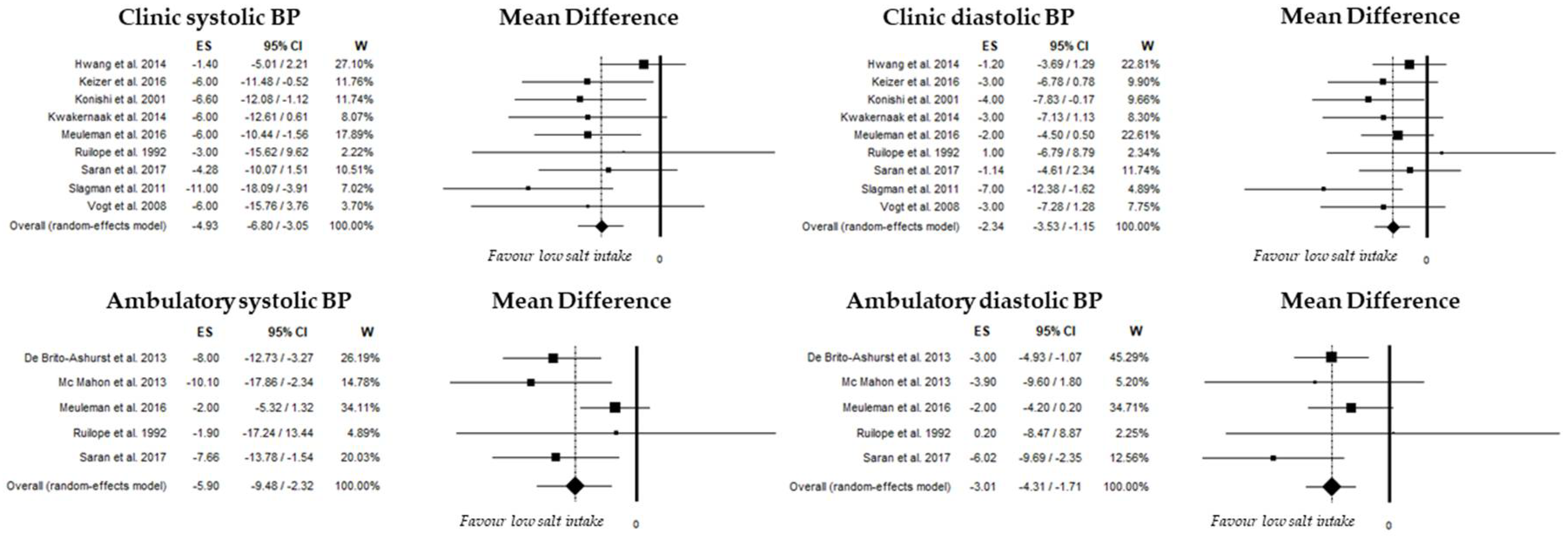
3.3. Outcomes
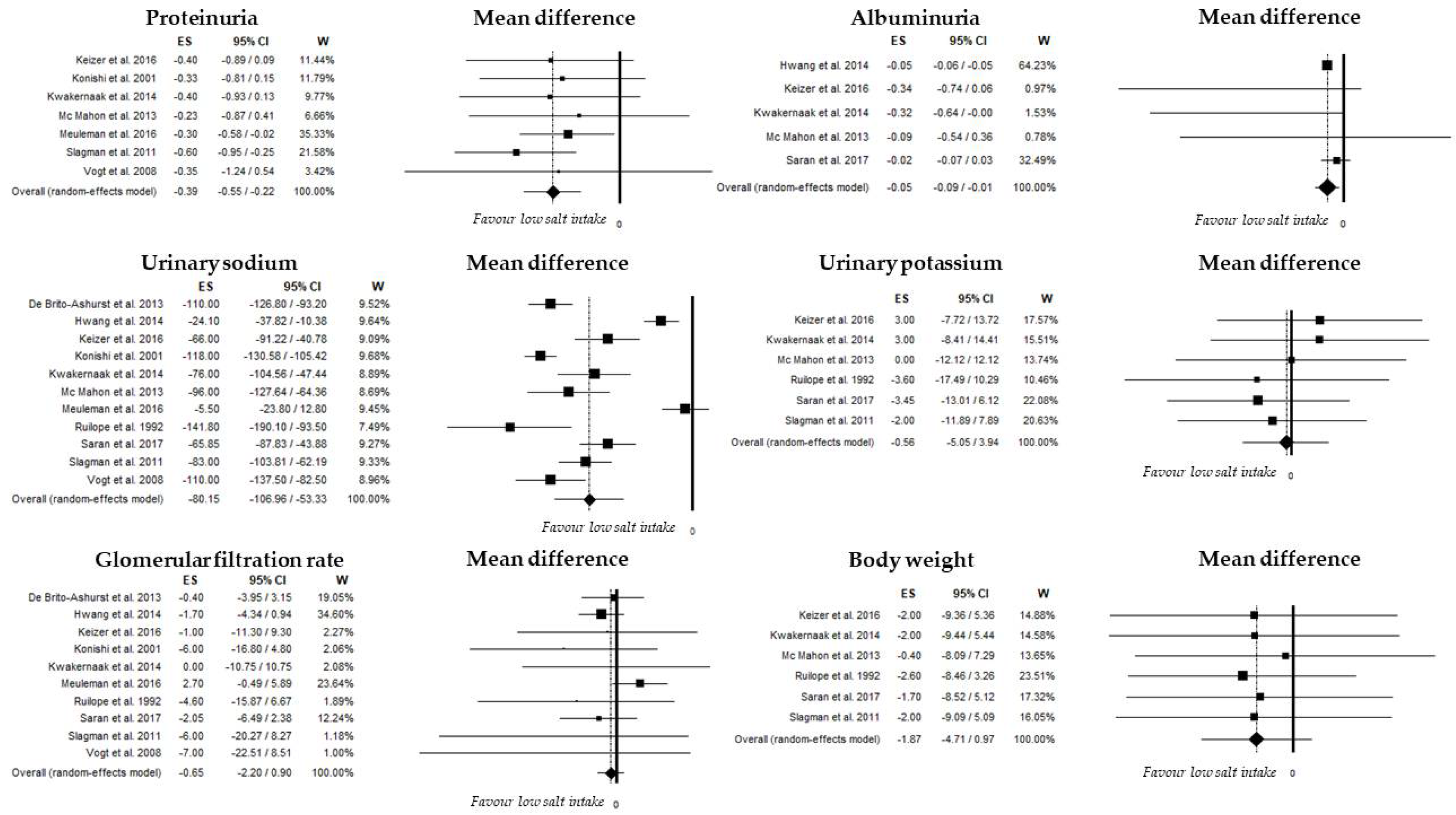
3.3.1. Urinary Sodium Excretion
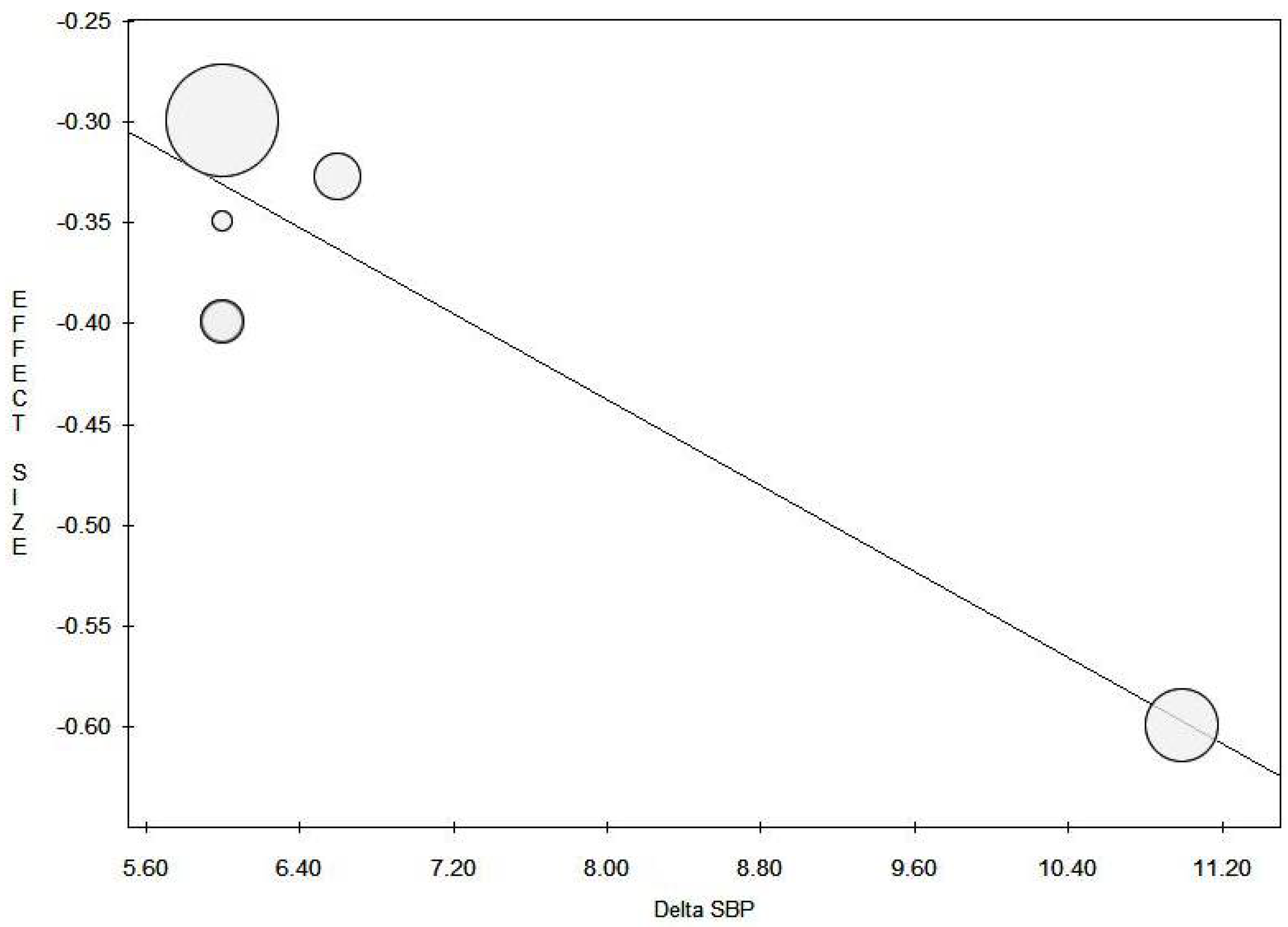
3.3.2. Systolic Blood Pressure
3.3.3. Diastolic Blood Pressure
3.3.4. Proteinuria and Albuminuria
3.3.5. Glomerular Filtration Rate
3.3.6. Additional Outcomes
3.3.7. Adverse Effects
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Appendix A
References
- Jha, V.; Garcia-Garcia, G.; Iseki, K.; Li, Z.; Naicker, S.; Plattner, B.; Saran, R.; Wang, A.Y.; Yang, C.W. Chronic kidney disease: Global dimension and perspectives. Lancet 2013, 382, 260–272. [Google Scholar] [CrossRef]
- Radhakrishnan, J.; Remuzzi, G.; Saran, R.; Williams, D.E.; Rios-Burrows, N.; Powe, N.; Brück, K.; Wanner, C.; Stel, V.S.; Venuthurupalli, S.K.; et al. Taming the chronic kidney disease epidemic: A global view of surveillance efforts. Kidney Int. 2014, 86, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Go, A.S.; Chertow, G.M.; Fan, D.; McCulloch, C.E.; Hsu, C.Y. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N. Engl. J. Med. 2004, 351, 1296–1305. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.C.; Maggo, J.K.; Campbell, K.L.; Craig, J.C.; Johnson, D.W.; Sutanto, B.; Ruospo, M.; Tong, A.; Strippoli, G.F. Dietary interventions for adults with chronic kidney disease. Cochrane Database Syst. Rev. 2017, 4, CD011998. [Google Scholar] [CrossRef] [PubMed]
- Ruggenenti, P.; Perticucci, E.; Cravedi, P.; Gambara, V.; Costantini, M.; Sharma, S.K.; Perna, A.; Remuzzi, G. Role of remission clinics in the longitudinal treatment of CKD. J. Am. Soc. Nephrol. 2008, 19, 1213–1224. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.L.; Ritz, E. Sodium chloride intake: Is lower always better? J. Am. Soc. Nephrol. 2012, 23, 1136–1139. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Iturbe, B.; Vaziri, N.D.; Herrera-Acosta, J.; Johnson, R.J. Oxidative stress, renal infiltration of immune cells, and salt-sensitive hypertension: All for one and one for all. Am. J. Physiol. Ren. Physiol. 2004, 286, F606–F616. [Google Scholar] [CrossRef] [PubMed]
- Cianciaruso, B.; Bellizzi, V.; Minutolo, R.; Colucci, G.; Bisesti, V.; Russo, D.; Conte, G.; De Nicola, L. Renal adaptation to dietary sodium restriction in moderate renal failure resulting from chronic glomerular disease. J. Am. Soc. Nephrol. 1996, 7, 306–313. [Google Scholar] [PubMed]
- Cianciaruso, B.; Bellizzi, V.; Minutolo, R.; Tavera, A.; Capuano, A.; Conte, G.; De Nicola, L. Salt intake and renal outcome in patients with progressive renal disease. Miner. Electrolyte Metab. 1998, 24, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Esnault, V.L.; Ekhlas, A.; Delcroix, C.; Moutel, M.G.; Nguyen, J.M. Diuretic and enhanced sodium restriction results in improved antiproteinuric response to RAS blocking agents. J. Am. Soc. Nephrol. 2005, 16, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Vogt, L.; Waanders, F.; Boomsma, F.; de Zeeuw, D.; Navis, G. Effects of dietary sodium and hydrochlorothiazide on the antiproteinuric efficacy of losartan. J. Am. Soc. Nephrol. 2008, 19, 999–1007. [Google Scholar] [CrossRef] [PubMed]
- Lambers Heerspink, H.J.; Holtkamp, F.A.; Parving, H.H.; Navis, G.J.; Lewis, J.B.; Ritz, E.; de Graeff, P.A.; de Zeeuw, D. Moderation of dietary sodium potentiates the renal and cardiovascular protective effects of angiotensin receptor blockers. Kidney Int. 2012, 82, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Torres, V.E.; Abebe, K.Z.; Schrier, R.W.; Perrone, R.D.; Chapman, A.B.; Yu, A.S.; Braun, W.E.; Steinman, T.I.; Brosnahan, G.; Hogan, M.C.; et al. Dietary salt restriction is beneficial to the management of autosomal dominant polycystic kidney disease. Kidney Int. 2017, 91, 493–500. [Google Scholar] [CrossRef] [PubMed]
- McMahon, E.J.; Campbell, K.L.; Bauer, J.D.; Mudge, D.W. Altered dietary salt intake for people with chronic kidney disease. Cochrane Database Syst. Rev. 2015, CD010070. [Google Scholar] [CrossRef] [PubMed]
- Bellizzi, V.; Conte, G.; Borrelli, S.; Cupisti, A.; De Nicola, L.; Di Iorio, B.R.; Cabiddu, G.; Mandreoli, M.; Paoletti, E.; Piccoli, G.B.; et al. Controversial issues in CKD clinical practice: Position statement of the CKD-treatment working group of the Italian Society of Nephrology. J. Nephrol. 2017, 30, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Decreased, G.F.R. Chapter 1: Definition and classification of CKD. Kidney Int. Suppl. (2011) 2013, 3, 19–62. [Google Scholar]
- Higgins, J.P.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Tobias, A. Assessing the influence of a single study in the meta-analysis estimate. Stata Tech. Bull. 1999, 8, 15–17. [Google Scholar]
- Van Houwelingen, H.C.; Arends, L.R.; Stijnen, T. Advanced methods in meta-analysis: Multivariate approach and meta-regression. Stat. Med. 2002, 21, 589–624. [Google Scholar] [CrossRef] [PubMed]
- Begg, C.B.; Mazumdar, M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994, 50, 1088–1101. [Google Scholar] [CrossRef] [PubMed]
- Ruilope, L.M.; Casal, M.C.; Guerrero, L.; Alcázar, J.M.; Férnandez, M.L.; Lahera, V.; Rodicio, J.L. Sodium intake does not influence the effect of verapamil in hypertensive patients with mild renal insufficiency. Drugs 1992, 44 (Suppl. 1), 94–98. [Google Scholar] [CrossRef] [PubMed]
- Konishi, Y.; Okada, N.; Okamura, M.; Morikawa, T.; Okumura, M.; Yoshioka, K.; Imanishi, M. Sodium sensitivity of blood pressure appearing before hypertension and related to histological damage in immunoglobulin a nephropathy. Hypertension 2001, 38, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Slagman, M.C.; Waanders, F.; Hemmelder, M.H.; Woittiez, A.J.; Janssen, W.M.; Lambers Heerspink, H.J.; Navis, G.; Laverman, G.D.; Holland Nephrology Study (HONEST) Group. Moderate dietary sodium restriction added to angiotensin converting enzyme inhibition compared with dual blockade in lowering proteinuria and blood pressure: Randomised controlled trial. BMJ 2011, 343, d4366. [Google Scholar] [CrossRef] [PubMed]
- De Brito-Ashurst, I.; Perry, L.; Sanders, T.A.; Thomas, J.E.; Dobbie, H.; Varagunam, M.; Yaqoob, M.M. The role of salt intake and salt sensitivity in the management of hypertension in South Asian people with chronic kidney disease: A randomised controlled trial. Heart 2013, 99, 1256–1260. [Google Scholar] [CrossRef] [PubMed]
- McMahon, E.J.; Bauer, J.D.; Hawley, C.M.; Isbel, N.M.; Stowasser, M.; Johnson, D.W.; Campbell, K.L. A randomized trial of dietary sodium restriction in CKD. J. Am. Soc. Nephrol. 2013, 24, 2096–2103. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.H.; Chin, H.J.; Kim, S.; Kim, D.K.; Kim, S.; Park, J.H.; Shin, S.J.; Lee, S.H.; Choi, B.S.; Lim, C.S. Effects of intensive low-salt diet education on albuminuria among nondiabetic patients with hypertension treated with olmesartan: A single-blinded randomized, controlled trial. Clin. J. Am. Soc. Nephrol. 2014, 9, 2059–2069. [Google Scholar] [CrossRef] [PubMed]
- Kwakernaak, A.J.; Krikken, J.A.; Binnenmars, S.H.; Visser, F.W.; Hemmelder, M.H.; Woittiez, A.J.; Groen, H.; Laverman, G.D.; Navis, G.; Holland Nephrology Study (HONEST) Group. Effects of sodium restriction and hydrochlorothiazide on RAAS blockade efficacy in diabetic nephropathy: A randomised clinical trial. Lancet Diabetes Endocrinol. 2014, 2, 385–395. [Google Scholar] [CrossRef]
- Keyzer, C.A.; van Breda, G.F.; Vervloet, M.G.; de Jong, M.A.; Laverman, G.D.; Hemmelder, M.H.; Janssen, W.M.; Lambers Heerspink, H.J.; Kwakernaak, A.J.; Bakker, S.J.; et al. Effects of Vitamin D Receptor Activation and Dietary Sodium Restriction on Residual Albuminuria in CKD: The ViRTUE-CKD Trial. J. Am. Soc. Nephrol. 2017, 28, 1296–1305. [Google Scholar] [CrossRef] [PubMed]
- Meuleman, Y.; Hoekstra, T.; Dekker, F.W.; Navis, G.; Vogt, L.; van der Boog, P.J.M.; Bos, W.J.W.; van Montfrans, G.A.; van Dijk, S.; ESMO Study Group. Sodium Restriction in Patients with CKD: A Randomized Controlled Trial of Self-management Support. Am. J. Kidney Dis. 2017, 69, 576–586. [Google Scholar] [CrossRef] [PubMed]
- Saran, R.; Padilla, R.L.; Gillespie, B.W.; Heung, M.; Hummel, S.L.; Derebail, V.K.; Pitt, B.; Levin, N.W.; Zhu, F.; Abbas, S.R.; et al. A Randomized Crossover Trial of Dietary Sodium Restriction in Stage 3–4 CKD. Clin. J. Am. Soc. Nephrol. 2017, 12, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Cheung, A.K.; Rahman, M.; Reboussin, D.M.; Craven, T.E.; Greene, T.; Kimmel, P.L.; Cushman, W.C.; Hawfield, A.T.; Johnson, K.C.; Lewis, C.E.; et al. Effects of Intensive BP Control in CKD. J. Am. Soc. Nephrol. 2017, 28, 2812–2823. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, R.; Andersen, M.J. Prognostic importance of ambulatory blood pressure recordings in patients with chronic kidney disease. Kidney Int. 2006, 69, 1175–1180. [Google Scholar] [CrossRef] [PubMed]
- De Nicola, L.; Borrelli, S.; Gabbai, F.B.; Chiodini, P.; Zamboli, P.; Iodice, C.; Vitiello, S.; Conte, G.; Minutolo, R. Burden of resistant hypertension in hypertensive patients with non-dialysis chronic kidney disease. Kidney Blood Press. Res. 2011, 34, 58–67. [Google Scholar] [CrossRef] [PubMed]
- De Nicola, L.; Gabbai, F.B.; Agarwal, R.; Chiodini, P.; Borrelli, S.; Bellizzi, V.; Nappi, F.; Conte, G.; Minutolo, R. Prevalence and prognostic role of resistant hypertension in chronic kidney disease patients. J. Am. Coll. Cardiol. 2013, 61, 2461–2467. [Google Scholar] [CrossRef] [PubMed]
- Minutolo, R.; Gabbai, F.B.; Agarwal, R.; Chiodini, P.; Borrelli, S.; Bellizzi, V.; Nappi, F.; Stanzione, G.; Conte, G.; De Nicola, L. Assessment of achieved clinic and ambulatory blood pressure recordings and outcomes during treatment in hypertensive patients with CKD: A multicenter prospective cohort study. Am. J. Kidney Dis. 2014, 64, 744–752. [Google Scholar] [CrossRef] [PubMed]
- Hummel, S.L.; Seymour, E.M.; Brook, R.D.; Sheth, S.S.; Ghosh, E.; Zhu, S.; Weder, A.B.; Kovács, S.J.; Kolias, T.J. Low-sodium DASH diet improves diastolic function and ventricular-arterial coupling in hypertensive heart failure with preserved ejection fraction. Circ. Heart Fail. 2013, 6, 1165–1171. [Google Scholar] [CrossRef] [PubMed]
- Oberleithner, H.; Riethmüller, C.; Schillers, H.; MacGregor, G.A.; de Wardener, H.E.; Hausberg, M. Plasma sodium stiffens vascular endothelium and reduces nitric oxide release. Proc. Natl. Acad. Sci. USA 2007, 104, 16281–16286. [Google Scholar] [CrossRef] [PubMed]
- Al-Solaiman, Y.; Jesri, A.; Zhao, Y.; Morrow, J.D.; Egan, B.M. Low-Sodium DASH reduces oxidative stress and improves vascular function in salt-sensitive humans. J. Hum. Hypertens. 2009, 23, 826–835. [Google Scholar] [CrossRef] [PubMed]
- Rugale, C.; Delbosc, S.; Cristol, J.P.; Mimran, A.; Jover, B. Sodium restriction prevents cardiac hypertrophy and oxidative stress in angiotensin II hypertension. Am. J. Physiol. Heart Circ. Physiol. 2003, 284, H1744–H1750. [Google Scholar] [CrossRef] [PubMed]
- De Nicola, L.; Conte, G.; Minutolo, R. Prediabetes as a Precursor to Diabetic Kidney Disease. Am. J. Kidney Dis. 2016, 67, 817–819. [Google Scholar] [CrossRef] [PubMed]
- Yoon, C.Y.; Noh, J.; Lee, J.; Kee, Y.K.; Seo, C.; Lee, M.; Cha, M.U.; Kim, H.; Park, S.; Yun, H.R.; et al. High and low sodium intakes are associated with incident chronic kidney disease in patients with normal renal function and hypertension. Kidney Int. 2018, 93, 921–931. [Google Scholar] [CrossRef] [PubMed]
- Kotchen, T.A.; Cowley, A.W., Jr.; Frohlich, E.D. Salt in health and disease—A delicate balance. N. Engl. J. Med. 2013, 368, 1229–1237. [Google Scholar] [CrossRef] [PubMed]
- Verbeke, F.; Lindley, E.; Van Bortel, L.; Vanholder, R.; London, G.; Cochat, P.; Wiecek, A.; Fouque, D.; Van Biesen, W. A European Renal Best Practice (ERBP) position statement on the Kidney Disease: Improving Global Outcomes (KDIGO) clinical practice guideline for the management of blood pressure in non-dialysis-dependent chronic kidney disease: An endorsement with some caveats for real-life application. Nephrol. Dial. Transplant. 2014, 29, 490–496. [Google Scholar] [PubMed]
- Sloan, P.J.; Beevers, G.; Baxter, F.E. The Quantab strip in the measurement of urinary chloride and sodium concentrations. Clin. Chem. 1984, 30, 1705–1707. [Google Scholar] [PubMed]
| Author/Country (Year) | Nr. Patients Low/High Salt Intake | Study Design | Intervention Duration (Weeks) | Mean Age (Years) | Male Gender (%) | Diabetes (%) | Cardiovascular Disease (%) | Anti-RAS Inhibitors Use (%) | CKD Stage | Interventions | Salt Restriction Modalities | Outcomes |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ruilope/Spain (1992) | 14/14 | Cross-over RCT | 1 | NA | NA | 0 | 0 | 0 | 1–3 | Low salt: 4 g/day High salt: 11 g/day | Salt supplementation in high salt | cSBP, cDBP, aSBP, aDBP, GFR, UNaV, UKV, body weight |
| Konishi/Japan (2001) | 41/41 | Cross-over RCT | 1 | 45 ± 15 | 34.0 | 0 | 0 | 0 | 1–2 | Low salt: 5 g/day High salt: 12 g/day | Replacing sodium-rich products with a low-sodium products | cSBP, cDBP, GFR, proteinuria, UNaV |
| Vogt/Netherlands (2008) | 33/33 | Cross-over RCT | 18 | 50 ± 12 | 75.7 | 0 | 0 | 0 | 1–3 | Low sodium: 50 mmol/day High sodium: 200 mmol/day | Replacing sodium-rich products with a low-sodium product of the same product group | cSBP, cDBP, GFR, proteinuria, UNaV |
| Slagman/Netherlands (2011) | 52/52 | Cross-over RCT | 6 | 51 | 82.7 | 0 | 0 | 100 | 1–3 | Low sodium: 50 mmol/day High sodium: 200 mmol/day | Dietary counseling in low salt | cSBP, cDBP, eGFR, proteinuria, UNaV, UKV, body weight |
| De Brito-Ashurst/UKV (2011) | 25/23 | Parallel RCT | 24 | 58 | 58.3 | 64.6 | NA | NA | 3–4 | Low salt: educational intervention High salt: regular care | Practical cooking andeducational sessions with dietitian | aSBP, aDBP, eGFR, UNaV, body weight, TBV |
| McMahon/Australia (2013) | 20/20 | Cross-over RCT | 2 | 68.5 ± 11 | 75.0 | 40 | NA | 30 | 3-4 | Low salt: 60–80 mmol/day High salt: 180–200 mmol/day | Salt supplementation in high salt | aSBP, aDBP, proteinuria, albuminuria, UNaV, UKV, body weight, TBV |
| Hwang/Korea (2014) | 119/126 | Parallel RCT | 8 | 49.5 ± 13.3 | 49.8 | 0 | 0 | 100 | 1–3 | Low salt: <100 mmol/day High salt: ≥25% reduction from baseline | Intensive and conventional education groups | cSBP, cDBP, GFR, albuminuria, UNaV |
| Kwakernaak/Netherlands (2014) | 45/45 | Cross-over RCT | 6 | 65 ± 9 | 84.0 | 100 | 47 | 100 | 1–3 | Low sodium: 50 mmol/day High sodium: 200 mmol/day | Counselling session with dietitians | SBP, DBP, body weight, GFR, albuminuria, proteinuria, UNaV, UKV |
| Keizer/Netherlands (2016) | 43/44 | Cross-over RCT | 8 | 63.2 | 43.1 | 0 | 0 | 100 | 1–3 | Low sodium: 1.2 g/day High sodium: 4.8 g/day | Replacing sodium-rich products with a low-sodium products | cSBP, cDBP, GFR, proteinuria, albuminuria, UNaV, UKV, body weight |
| Meuleman/Netherlands (2016) | 67/71 | Parallel RCT | 24 | 55.1 | 81.8 | 25 | 38 | 100 | 1–4 | Low salt: self -management intervention High salt: regular care | Nutrition counseling by a dietician and psychologists, point-of-care chip-device | cSBP, cDBP, aSBP, aDBP, GFR, proteinuria, UNaV |
| Saran/US (2017) | 58/58 | Cross-over RCT | 4 | 56.5 | 52.0 | 38 | 21 | NA | 3–4 | Low sodium: <2g/day High sodium: 10 g/day | dietary counseling with training in motivational interviewing techniques | aSBP, aDBP, cSBP, cDBP, GFR, albuminuria, UNaV, UKV, body weight, TBV |
| Author | Final GFR(ml/min) | Final Uprot (g/day) | Final Ualb (g/day) | Final Urinary Na (mmol/day) | Final cSBP (mmHg) | Final cDBP (mmHg) | Final aSBP (mmHg) | Final aDBP (mmHg) | |
|---|---|---|---|---|---|---|---|---|---|
| Ruilope et al. | Low High | 62.7 ± 10.8 * 67.3 ± 18.6 * | NA | NA | 72.6 ± 39.1 214.4 ± 83.5 | 151.6 ± 17.9 154.7 ± 15.8 | 95.6 ± 10.2 94.9 ± 11.3 | 146.1 ± 20.2 148.0 ± 21.2 | 90.3 ± 11.3 90.1 ± 12.1 |
| Konishi et al. | Low High | 108 ± 23 * 114 ± 25 * | 0.55 ± 0.76 0.88 ± 1.30 | NA | 48 ± 14 166 ± 37 | 115 ± 11.2 121.6 ± 13.1 | 75 ± 8 79 ± 9 | NA | NA |
| Vogt et al. | Low High | 82 ± 35 * 89 ± 29 * | 2.10 ± 2.10 ^ 2.45 ± 1.55 ^ | NA | 90 ± 57 200 ± 57 | 137 ± 17 143 ± 23 | 83 ± 6 86 ± 11 | NA | NA |
| Slagman et al. | Low High | 66 ± 34 * 72 ± 40 * | 0.6 ± 0.7 ^ 1.2 ± 1.1 ^ | NA | 106 ± 50 189 ± 58 | 123 ± 14 134 ± 22 | 73 ± 14 80 ± 14 | NA | NA |
| De Brito-Ashurst et al. | Low High | NA | NA | NA | NA | NA | NA | NA | NA |
| McMahon et al. | Low High | NA | 0.64 ± 0.93 0.87 ± 1.12 | 0.35 ± 0.69 0.44 ± 0.76 | 82 ± 43 178 ± 58 | NA | NA | 144.9 ± 13.1 154.6 ± 11.9 | 79.4 ± 9.4 83.3 ± 9.0 |
| Hwang et al. | Low High | 63.4 ± 10.9 # 65.1 ± 10.1 # | NA | 0.18 ± 0.01 0.23 ± 0.01 | 122.2 ± 54.5 146.3 ± 55 | 121.2 ± 14.2 122.6 ± 14.6 | 73.6 ± 9.8 74.8 ± 10.1 | NA | NA |
| Kwakernaak et al. | Low High | 65 ±27 ° 65± 25 ° | 0.9 ± 1.0 1.3 ± 1.5 | 0.39 ± 0.57 0.71 ± 0.93 | 148 ± 65 224 ± 73 | 141 ± 16 147 ± 16 | 79 ± 10 82 ± 10 | NA | NA |
| Keizer et al. | Low High | 67 ± 24 # 68 ± 25 # | 1.0 ± 1.0 1.4 ± 1.3 | 0.72 ± 0.8 1.06 ± 1.09 | 104 ± 59 170 ± 61 | 123 ± 12 129 ± 14 | 74 ± 9 77 ± 9 | NA | NA |
| Meuleman et al. | Low High | 49.6 ± 9.0 # 46.9 ± 10.1 # | 1.1 ± 0.82 1.4 ± 0.84 | NA | 157.0 ± 52.4 162.5 ± 57.3 | 133 ± 13.1 139 ± 13.5 | 81 ± 7.4 83 ± 7.6 | 128 ± 9.8 130 ± 10.1 | 75 ± 6.5 77 ± 6.7 |
| Saran et al. | Low High | 35.6 # 37.6 # | NA | 0.10 0.16 | 104.8 170.2 | 127.3 131.4 | 69.4 70.7 | 133.5 141.4 | 71.5 77.3 |
| Study | Random Sequence Generation | Allocation Concealment | Blinding of Participants | Blinding of Outcome Assessment | Free of Incomplete Outcome Data | Free of Selective Reporting | Free of Carry Over Effect | Free of bias from Confounders | Free of Others Bias |
|---|---|---|---|---|---|---|---|---|---|
| Ruilope et al. | Unclear | Unclear | Unclear | Unclear | Unclear | Unclear | No | Unclear | Unclear |
| Konishi et al. | Unclear | Unclear | Unclear | Yes | Unclear | Unclear | No | Unclear | Unclear |
| Vogt et al. | Yes | Unclear | Unclear | Unclear | Yes | Yes | Yes | No | No |
| Slagman et al. | Yes | Unclear | Unclear | Unclear | Yes | Yes | Yes | No | Unclear |
| De Brito-Ashurst et al. | Yes | Unclear | Unclear | Unclear | Yes | Unclear | Yes | Unclear | Yes |
| McMahon et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Unclear | Yes |
| Hwang et al. | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Unclear | Unclear |
| Kwakernaak et al. | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes |
| Keizer et al. | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes |
| Meuleman et al. | Yes | Yes | Unclear | Unclear | Unclear | Unclear | Yes | Unclear | Yes |
| Saran et al. | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garofalo, C.; Borrelli, S.; Provenzano, M.; De Stefano, T.; Vita, C.; Chiodini, P.; Minutolo, R.; Nicola, L.D.; Conte, G. Dietary Salt Restriction in Chronic Kidney Disease: A Meta-Analysis of Randomized Clinical Trials. Nutrients 2018, 10, 732. https://doi.org/10.3390/nu10060732
Garofalo C, Borrelli S, Provenzano M, De Stefano T, Vita C, Chiodini P, Minutolo R, Nicola LD, Conte G. Dietary Salt Restriction in Chronic Kidney Disease: A Meta-Analysis of Randomized Clinical Trials. Nutrients. 2018; 10(6):732. https://doi.org/10.3390/nu10060732
Chicago/Turabian StyleGarofalo, Carlo, Silvio Borrelli, Michele Provenzano, Toni De Stefano, Carlo Vita, Paolo Chiodini, Roberto Minutolo, Luca De Nicola, and Giuseppe Conte. 2018. "Dietary Salt Restriction in Chronic Kidney Disease: A Meta-Analysis of Randomized Clinical Trials" Nutrients 10, no. 6: 732. https://doi.org/10.3390/nu10060732
APA StyleGarofalo, C., Borrelli, S., Provenzano, M., De Stefano, T., Vita, C., Chiodini, P., Minutolo, R., Nicola, L. D., & Conte, G. (2018). Dietary Salt Restriction in Chronic Kidney Disease: A Meta-Analysis of Randomized Clinical Trials. Nutrients, 10(6), 732. https://doi.org/10.3390/nu10060732







