Mastication of Nuts under Realistic Eating Conditions: Implications for Energy Balance
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
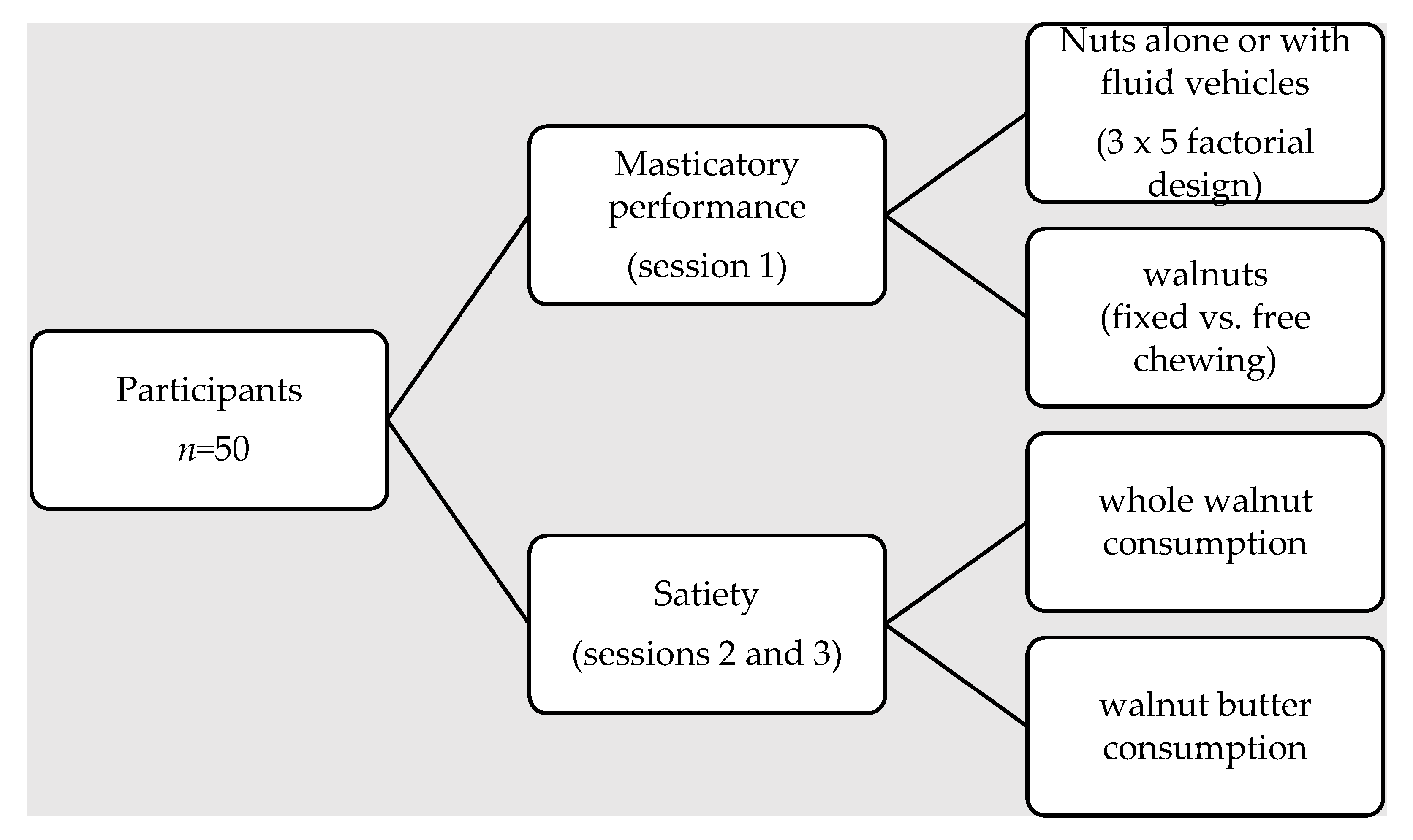
2.2. Experimental Design
2.3. Test Foods
2.4. Breaking Force
2.5. Masticatory Performance
2.6. Proportional Particle Size Distribution
2.7. Statistical Analysis
3. Results
3.1. Nut Breaking Force
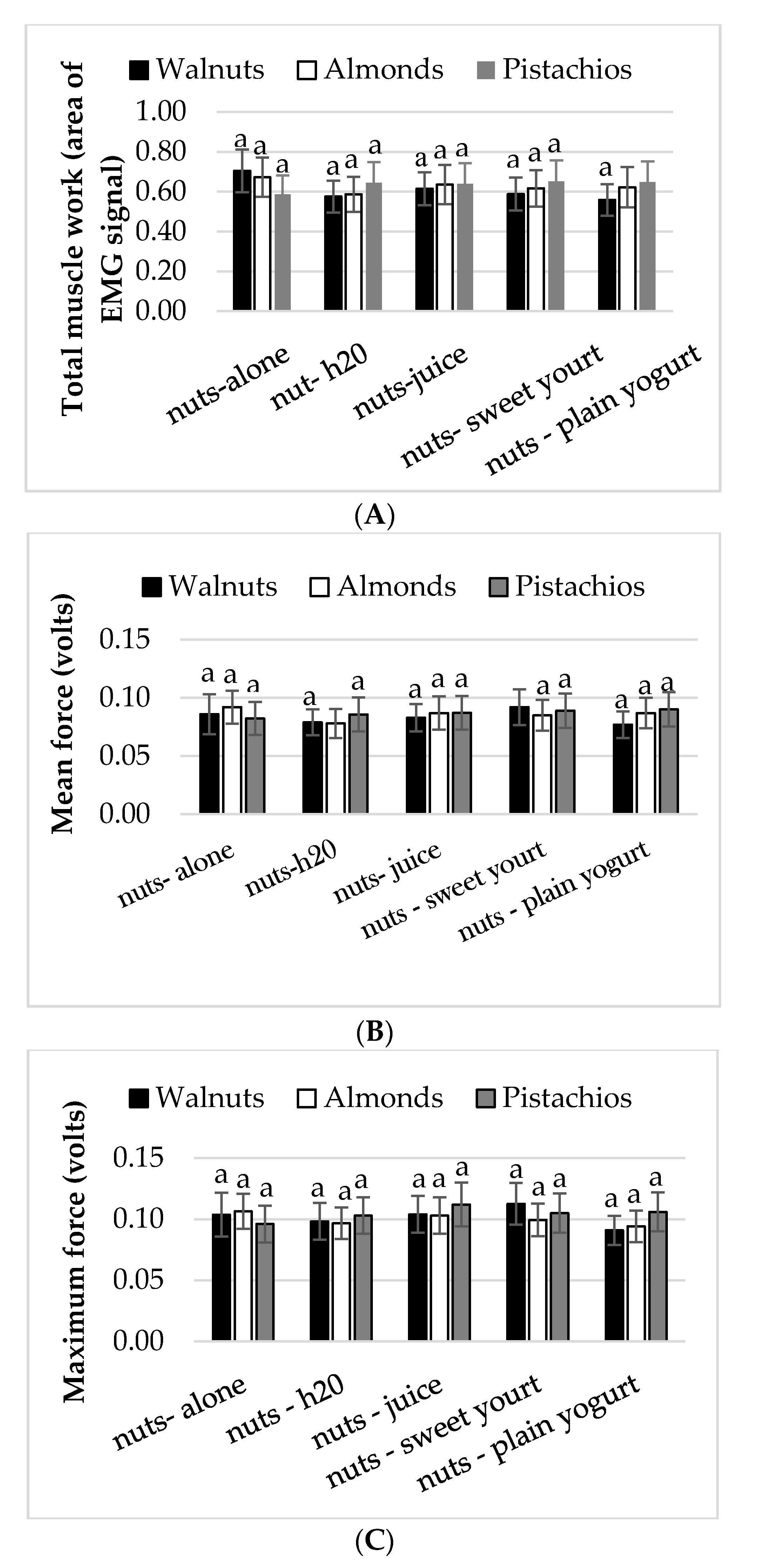
3.2. Mastication Parameters
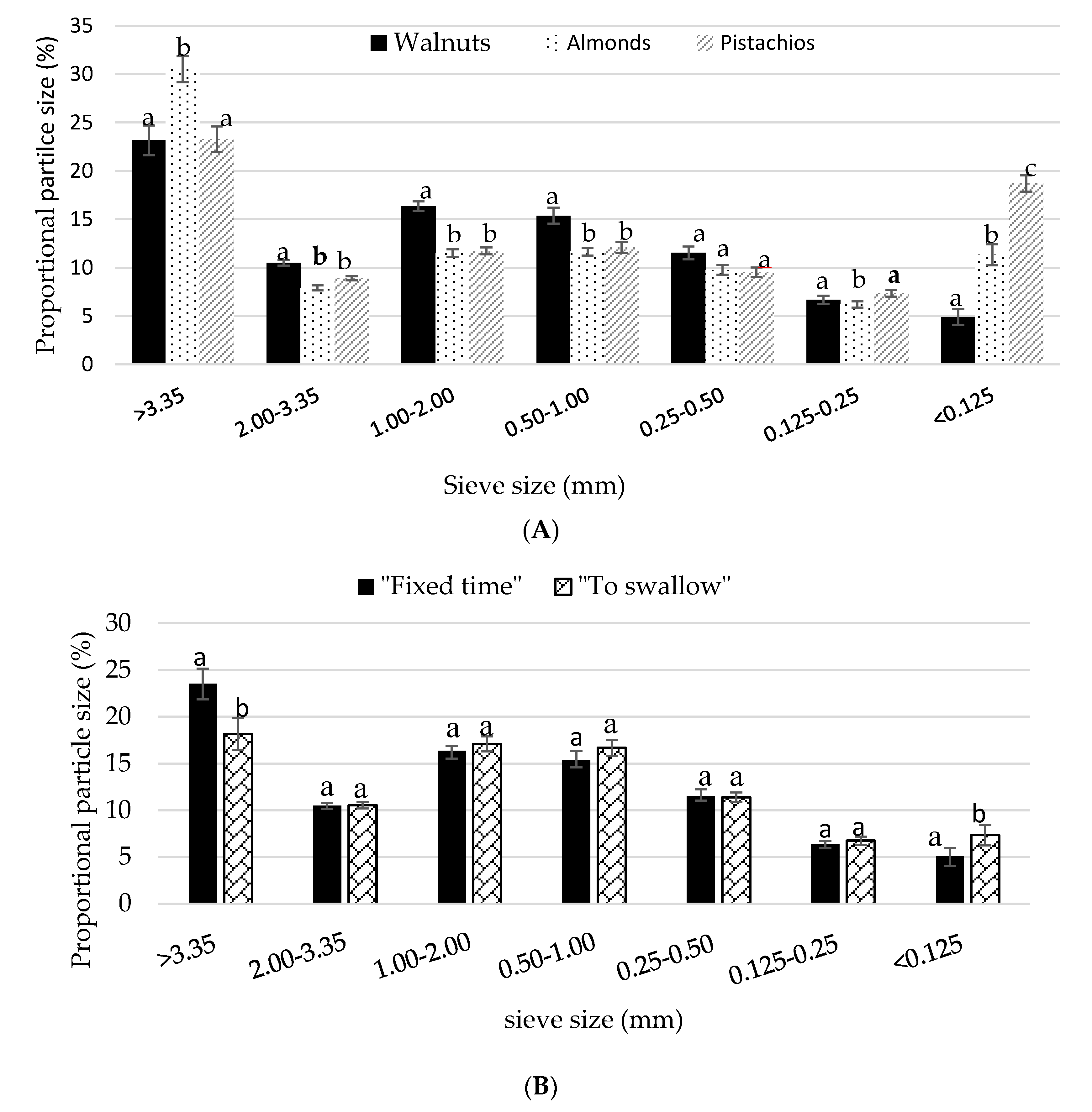
3.3. Particle Size
3.4. Palatability Ratings
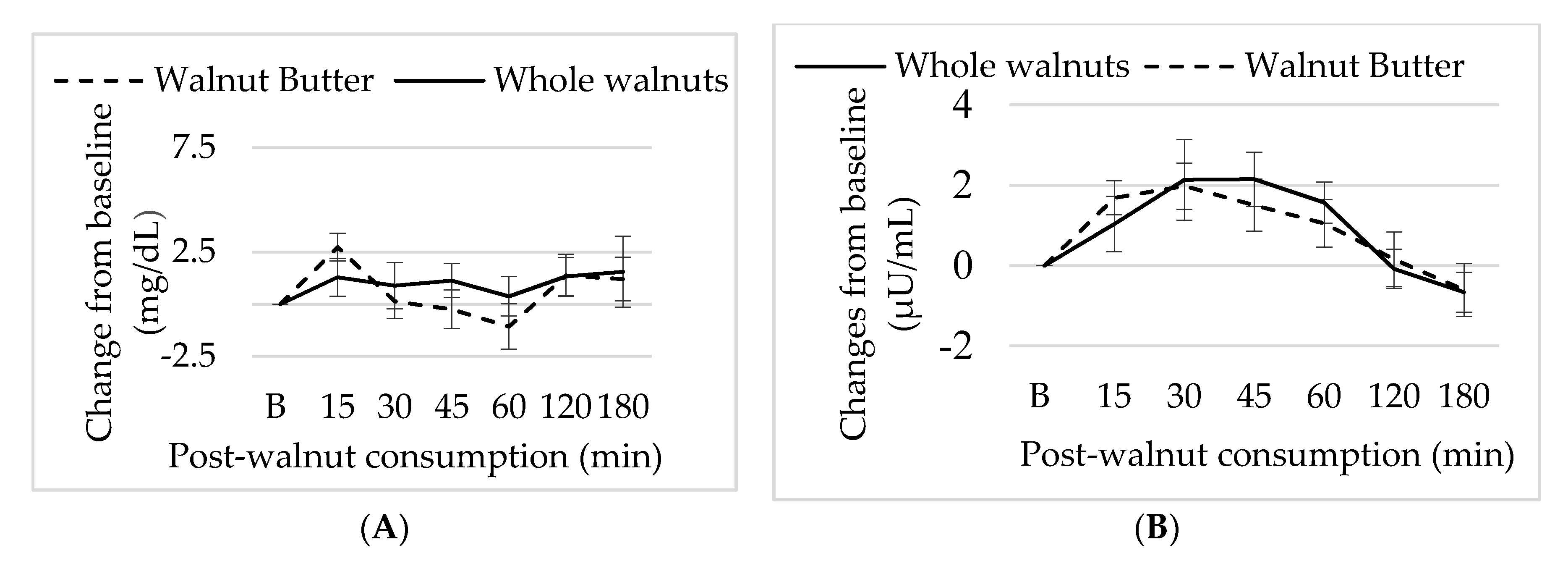
3.5. Effect of Walnut Consumption on Metabolic Measure
3.6. Appetitive Sensations
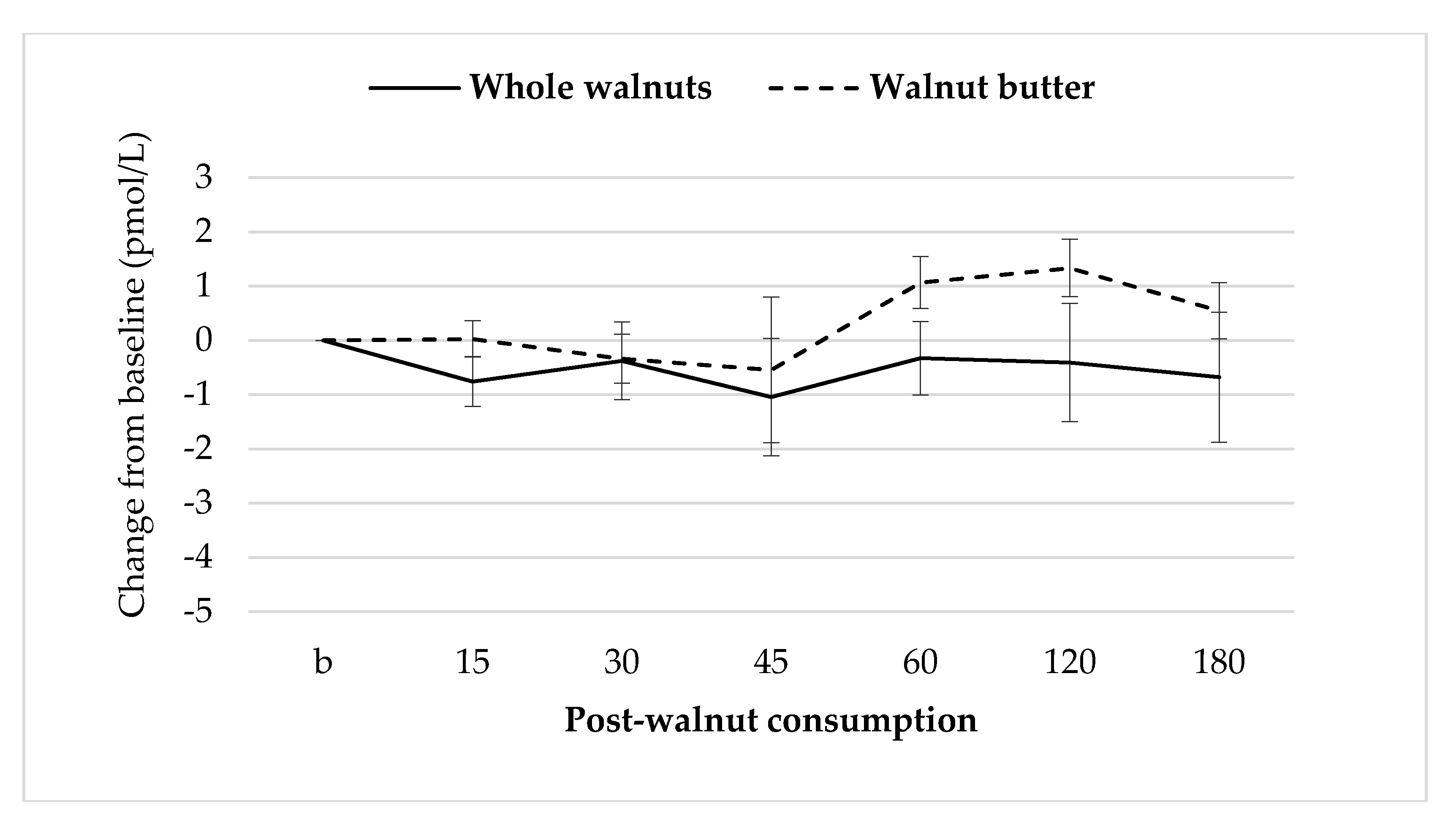
3.7. Gut Hormones
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Jackson, C.L.; Hu, F.B. Long-term associations of nut consumption with body weight and obesity. Am. J. Clin. Nutr. 2014, 100, 408S–411S. [Google Scholar] [CrossRef] [PubMed]
- Fraser, G.E.; Bennett, H.W.; Jaceldo, K.B.; Sabaté, J. Effect on body weight of a free 76 kilojoule (320 calorie) daily supplement of almonds for six months. J. Am. Coll. Nutr. 2002, 21, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Alper, C.; Mattes, R. Effects of chronic peanut consumption on energy balance and hedonics. Int. J. Obes. 2002, 26, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Sabaté, J.; Cordero-MacIntyre, Z.; Siapco, G.; Torabian, S.; Haddad, E. Does regular walnut consumption lead to weight gain? Br. J. Nutr. 2005, 94, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Bes-Rastrollo, M.; Wedick, N.M.; Martinez-Gonzalez, M.A.; Li, T.Y.; Sampson, L.; Hu, F.B. Prospective study of nut consumption, long-term weight change, and obesity risk in women. Am. J. Clin. Nutr. 2009, 89, 1913–1919. [Google Scholar] [CrossRef] [PubMed]
- Almario, R.U.; Vonghavaravat, V.; Wong, R.; Kasim-Karakas, S.E. Effects of walnut consumption on plasma fatty acids and lipoproteins in combined hyperlipidemia. Am. J. Clin. Nutr. 2001, 74, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Fraser, G.E.; Sabate, J.; Beeson, W.L.; Strahan, T.M. A possible protective effect of nut consumption on risk of coronary heart disease: The Adventist Health Study. Arch. Intern. Med. 1992, 152, 1416–1424. [Google Scholar] [CrossRef] [PubMed]
- Albert, C.M.; Gaziano, J.M.; Willett, W.C.; Manson, J.E. Nut consumption and decreased risk of sudden cardiac death in the Physicians’ Health Study. Arch. Intern. Med. 2002, 162, 1382–1387. [Google Scholar] [CrossRef] [PubMed]
- Morgan, W.A.; Clayshulte, B.J. Pecans lower low density lipoprotein cholesterol in people with normal lipid levels. J. Am. Diet. Assoc. 2000, 100, 312–318. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Hu, F.B.; Tapsell, L.C.; Josse, A.R.; Kendall, C.W. Possible benefit of nuts in type 2 diabetes. J. Nutr. 2008, 138, 1752S–1756S. [Google Scholar] [CrossRef] [PubMed]
- Coelho, S.B.; de Sales, R.L.; Iyer, S.S.; Bressan, J.; Costa, N.M.B.; Lokko, P.; Mattes, R. Effects of peanut oil load on energy expenditure, body composition, lipid profile, and appetite in lean and overweight adults. Nutrition 2006, 22, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Alves, R.D.M.; Moreira, A.P.B.; Macedo, V.S.; Costa, N.M.B.; Alfenas, R.d.C.G.; Bressan, J. High-oleic peanuts increase diet-induced thermogenesis in overweight and obese men. Nutr. Hosp. 2014, 29, 1024–1032. [Google Scholar]
- Casas-Agustench, P.; López-Uriarte, P.; Bullo, M.; Ros, E.; Gómez-Flores, A.; Salas-Salvadó, J. Acute effects of three high-fat meals with different fat saturations on energy expenditure, substrate oxidation and satiety. Clin. Nutr. 2009, 28, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Pasman, W.J.; Heimerikx, J.; Rubingh, C.M.; van den Berg, R.; O’Shea, M.; Gambelli, L.; Hendriks, H.F.; Einerhand, A.W.; Scott, C.; Keizer, H.G. The effect of Korean pine nut oil on in vitro CCK release, on appetite sensations and on gut hormones in post-menopausal overweight women. Lipids Health Dis. 2008, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.Y.; Mattes, R. Appetitive, dietary and health effects of almonds consumed with meals or as snacks: A randomized, controlled trial. Eur. J. Clin. Nutr. 2013, 67, 1205–1214. [Google Scholar] [CrossRef] [PubMed]
- Reis, C.E.; Ribeiro, D.N.; Costa, N.M.; Bressan, J.; Alfenas, R.C.; Mattes, R.D. Acute and second-meal effects of peanuts on glycaemic response and appetite in obese women with high type 2 diabetes risk: A randomised cross-over clinical trial. Br. J. Nutr. 2013, 109, 2015–2023. [Google Scholar] [CrossRef] [PubMed]
- Barbour, J.A.; Howe, P.R.; Buckley, J.D.; Wright, G.C.; Bryan, J.; Coates, A.M. Lower energy intake following consumption of Hi-oleic and regular peanuts compared with iso-energetic consumption of potato crisps. Appetite 2014, 82, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Cassady, B.A.; Hollis, J.H.; Fulford, A.D.; Considine, R.V.; Mattes, R.D. Mastication of almonds: Effects of lipid bioaccessibility, appetite, and hormone response. Am. J. Clin. Nutr. 2009, 89, 794–800. [Google Scholar] [CrossRef] [PubMed]
- Brennan, A.M.; Sweeney, L.L.; Liu, X.; Mantzoros, C.S. Walnut Consumption Increases Satiation but Has No Effect on Insulin Resistance or the Metabolic Profile Over a 4-day Period. Obesity 2010, 18, 1176–1182. [Google Scholar] [CrossRef] [PubMed]
- Katz, D.L.; Davidhi, A.; Ma, Y.; Kavak, Y.; Bifulco, L.; Njike, V.Y. Effects of walnuts on endothelial function in overweight adults with visceral obesity: A randomized, controlled, crossover trial. J. Am. Coll. Nutr. 2012, 31, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Haddad, E.; Sabate, J. Effect of pecan consumption on stool fat. Proc. FASEB J. 2000, 14, 294. [Google Scholar]
- Zemaitis, J.; Sabaté, J. Effect of almond consumption on stool weight and stool fat. Proc. FASEB J. 2001, 15, A602. [Google Scholar]
- Levine, A.S.; Silvis, S.E. Absorption of whole peanuts, peanut oil, and peanut butter. N. Engl. J. Med. 1980, 303, 917–918. [Google Scholar] [CrossRef] [PubMed]
- Traoret, C.; Lokko, P.; Cruz, A.; Oliveira, C.; Costa, N.; Bressan, J.; Alfenas, R.; Mattes, R. Peanut digestion and energy balance. Int. J. Obes. 2008, 32, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Levine, J.; Baukol, P.; Pavlidis, I. The energy expended in chewing gum. N. Engl. J. Med. 1999, 341, 2100. [Google Scholar] [CrossRef] [PubMed]
- Kresge, D.L.; Melanson, K. Chewing gum increases energy expenditure before and after controlled breakfasts. Appl. Physiol. Nutr. Metab. 2014, 40, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Paphangkorakit, J.; Leelayuwat, N.; Boonyawat, N.; Parniangtong, A.; Sripratoom, J. Effect of chewing speed on energy expenditure in healthy subjects. Acta Odontol. Scand. 2014, 72, 424–427. [Google Scholar] [CrossRef] [PubMed]
- Komai, N.; Motokubota, N.; Suzuki, M.; Hayashi, I.; Moritani, T.; Nagai, N. Thorough Mastication Prior to Swallowing Increases Postprandial Satiety and the Thermic Effect of a Meal in Young Women. J. Nutr. Sci. Vitaminol. 2016, 62, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Hollis, J.; Mattes, R. Effect of chronic consumption of almonds on body weight in healthy humans. Br. J. Nutr. 2007, 98, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Brown, R.; Gray, A.; Chisholm, A.; Delahunty, C. Nuts improve diet quality compared to other energy-dense snacks while maintaining body weight. J. Nutr. Metab. 2011, 2011, 357350. [Google Scholar] [CrossRef] [PubMed]
- Stull, A.J.; Apolzan, J.W.; Thalacker-Mercer, A.E.; Iglay, H.B.; Campbell, W.W. Liquid and Solid Meal Replacement Products Differentially Affect Postprandial Appetite and Food Intake in Older Adults. J. Am. Diet. Assoc. 2008, 108, 1226–1230. [Google Scholar] [CrossRef] [PubMed]
- Mattes, R. Soup and satiety. Physiol. Behav. 2005, 83, 739–747. [Google Scholar] [CrossRef] [PubMed]
- DiMeglio, D.P.; Mattes, R.D. Liquid versus solid carbohydrate: Effects on food intake and body weight. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 794–800. [Google Scholar] [CrossRef] [PubMed]
- Lasschuijt, M.P.; Mars, M.; Stieger, M.; Miquel-Kergoat, S.; de Graaf, C.; Smeets, P. Comparison of oro-sensory exposure duration and intensity manipulations on satiation. Physiol. Behav. 2017, 176, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Houchins, J.A.; Tan, S.-Y.; Campbell, W.W.; Mattes, R.D. Effects of fruit and vegetable, consumed in solid vs. beverage forms on acute and chronic appetitive responses in lean and obese adults. Int. J. Obes. 2013, 37, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Kirkmeyer, S.; Mattes, R. Effects of food attributes on hunger and food intake. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 1167–1175. [Google Scholar] [CrossRef] [PubMed]
- Cecil, J.; Francis, J.; Read, N. Relative contributions of intestinal, gastric, oro-sensory influences and information to changes in appetite induced by the same liquid meal. Appetite 1998, 31, 377–390. [Google Scholar] [CrossRef] [PubMed]
- Mourao, D.; Bressan, J.; Campbell, W.; Mattes, R. Effects of food form on appetite and energy intake in lean and obese young adults. Int. J. Obes. 2007, 31, 1688–1695. [Google Scholar] [CrossRef] [PubMed]
- Sakata, T. Histamine receptor and its regulation of energy metabolism. Obesity 1995, 3, 541S–548S. [Google Scholar] [CrossRef]
- Sakata, T.; Yoshimatsu, H.; Masaki, T.; Tsuda, K. Anti-obesity actions of mastication driven by histamine neurons in rats. Exp. Biol. Med. 2003, 228, 1106–1110. [Google Scholar] [CrossRef]
- Arosio, M.; Ronchi, C.L.; Beck-Peccoz, P.; Gebbia, C.; Giavoli, C.; Cappiello, V.; Conte, D.; Peracchi, M. Effects of modified sham feeding on ghrelin levels in healthy human subjects. J. Clin. Endocrinol. Metab. 2004, 89, 5101–5104. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Hsu, W.H.; Hollis, J.H. Modified sham feeding of foods with different macronutrient compositions differentially influences cephalic change of insulin, ghrelin, and NMR-based metabolomic profiles. Physiol. Behav. 2014, 135, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, J.L.; Paton, C.M.; Cooper, J.A. Hunger and satiety responses to high-fat meals after a high-polyunsaturated fat diet: A randomized trial. Nutrition 2017, 41, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Baer, D.J.; Gebauer, S.K.; Novotny, J.A. Walnuts Consumed by Healthy Adults Provide Less Available Energy than Predicted by the Atwater Factors. J. Nutr. 2015, 146, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Baer, D.J.; Gebauer, S.K.; Novotny, J.A. Measured energy value of pistachios in the human diet. Br. J. Nutr. 2012, 107, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Novotny, J.A.; Gebauer, S.K.; Baer, D.J. Discrepancy between the Atwater factor predicted and empirically measured energy values of almonds in human diets. Am. J. Clin. Nutr. 2012, 96, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Ellis, P.R.; Kendall, C.W.; Ren, Y.; Parker, C.; Pacy, J.F.; Waldron, K.W.; Jenkins, D.J. Role of cell walls in the bioaccessibility of lipids in almond seeds. Am. J. Clin. Nutr. 2004, 80, 604–613. [Google Scholar] [CrossRef] [PubMed]
- Miquel-Kergoat, S.; Azais-Braesco, V.; Burton-Freeman, B.; Hetherington, M.M. Effects of chewing on appetite, food intake and gut hormones: A systematic review and meta-analysis. Physiol. Behav. 2015, 151, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, N.; Hu, L.; Li, Z.; Li, R.; Li, C.; Wang, S. Improvement in chewing activity reduces energy intake in one meal and modulates plasma gut hormone concentrations in obese and lean young Chinese men. Am. J. Clin. Nutr. 2011, 94, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Hsu, W.H.; Hollis, J.H. Increasing the number of masticatory cycles is associated with reduced appetite and altered postprandial plasma concentrations of gut hormones, insulin and glucose. Br. J. Nutr. 2013, 110, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Frecka, J.M.; Hollis, J.H.; Mattes, R.D. Effects of appetite, BMI, food form and flavor on mastication: Almonds as a test food. Eur. J. Clin. Nutr. 2007, 62, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- McKiernan, F.; Mattes, R.D. Effects of peanut processing on masticatory performance during variable appetitive states. J. Nutr. Metab. 2009, 2010, 487301. [Google Scholar] [CrossRef] [PubMed]
- King, J.C.; Blumberg, J.; Ingwersen, L.; Jenab, M.; Tucker, K.L. Tree nuts and peanuts as components of a healthy diet. J. Nutr. 2008, 138, 1736S–1740S. [Google Scholar] [CrossRef] [PubMed]
- Mandalari, G.; Bisignano, C.; Filocamo, A.; Chessa, S.; Sarò, M.; Torre, G.; Faulks, R.M.; Dugo, P. Bioaccessibility of pistachio polyphenols, xanthophylls, and tocopherols during simulated human digestion. Nutrition 2013, 29, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Hutchings, S.C.; Foster, K.D.; Bronlund, J.E.; Lentle, R.G.; Jones, J.R.; Morgenstern, M.P. Mastication of heterogeneous foods: Peanuts inside two different food matrices. Food Qual. Preference 2011, 22, 332–339. [Google Scholar] [CrossRef]
- Engelen, L.; Fontijn-Tekamp, A.; van der Bilt, A. The influence of product and oral characteristics on swallowing. Arch. Oral Biol. 2005, 50, 739–746. [Google Scholar] [CrossRef] [PubMed]
- Prinz, J.F.; Lucas, P.W. Swallow thresholds in human mastication. Arch. Oral Biol. 1995, 40, 401–403. [Google Scholar] [CrossRef]
- Bellisle, F.; Guy-Grand, B.; Le Magnen, J. Chewing and swallowing as indices of the stimulation to eat during meals in humans: Effects revealed by the edogram method and video recordings. Neurosci. Biobehav. Rev. 2000, 24, 223–228. [Google Scholar] [CrossRef]
- Guy-Grand, B.; Lehnert, V.; Doassans, M.; Bellisle, F. Type of Test-meal Affects Palatability and Eating Style in Humans. Appetite 1994, 22, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Rolls, B.J.; Wood, R.J.; Stevens, R.M. Palatability and body fluid homeostasis. Physiol. Behav. 1978, 20, 15–19. [Google Scholar] [CrossRef]
- Mosca, A.C.; van de Velde, F.; Bult, J.H.F.; van Boekel, M.A.J.S.; Stieger, M. Taste enhancement in food gels: Effect of fracture properties on oral breakdown, bolus formation and sweetness intensity. Food Hydrocoll. 2015, 43, 794–802. [Google Scholar] [CrossRef]
- Pereira, L.J.; Duarte Gaviao, M.B.; Van Der Bilt, A. Influence of oral characteristics and food products on masticatory function. Acta Odontol. Scand. 2006, 64, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Singh, H.; Ye, A.; Ferrua, M.J. Aspects of food structures in the digestive tract. Curr. Opin. Food Sci. 2015, 3, 85–93. [Google Scholar] [CrossRef]
- Hayes, D.; Angove, M.J.; Tucci, J.; Dennis, C. Walnuts (Juglans regia) chemical composition and research in human health. Crit. Rev. Food Sci. Nutr. 2016, 56, 1231–1241. [Google Scholar] [CrossRef] [PubMed]
- Yeh, L.; Kim, K.; Chompreeda, P.; Rimkeeree, H.; Yau, N.; Lundahl, D. Comparison in use of the 9-point hedonic scale between Americans, Chinese, Koreans, and Thai. Food Qual. Preference 1998, 9, 413–419. [Google Scholar] [CrossRef]
- Hill, A.J.; Blundell, J.E. Nutrients and behaviour: Research strategies for the investigation of taste characteristics, food preferences, hunger sensations and eating patterns in man. J. Psychiatr. Res. 1982, 17, 203–212. [Google Scholar] [CrossRef]
- Ow, R.K.K.; Carlsson, G.E.; Karlsson, S. Relationship of masticatory mandibular movements to masticatory performance of dentate adults: A method study. J. Oral Rehabil. 1998, 25, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Grundy, M.M.L.; Lapsley, K.; Ellis, P.R. A review of the impact of processing on nutrient bioaccessibility and digestion of almonds. Int. J. Food Sci. Technol. 2016, 51, 1937–1946. [Google Scholar] [CrossRef] [PubMed]
- Grundy, M.M.L.; Grassby, T.; Mandalari, G.; Waldron, K.W.; Butterworth, P.J.; Berry, S.E.E.; Ellis, P.R. Effect of mastication on lipid bioaccessibility of almonds in a randomized human study and its implications for digestion kinetics, metabolizable energy, and postprandial lipemia. Am. J. Clin. Nutr. 2015, 101, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Chen, J. Food oral processing—A review. Food Hydrocoll. 2009, 23, 1–25. [Google Scholar] [CrossRef]
- Bornhorst, G.M.; Singh, R.P. Bolus formation and disintegration during digestion of food carbohydrates. Compr. Rev. Food Sci. Food Saf. 2012, 11, 101–118. [Google Scholar] [CrossRef]
- Mioche, L.; Bourdiol, P.; Monier, S. Chewing behaviour and bolus formation during mastication of meat with different textures. Arch. Oral Biol. 2003, 48, 193–200. [Google Scholar] [CrossRef]
- Moongngarm, A.; Bronlund, J.E.; Grigg, N.; Sriwai, N. Chewing behavior and bolus properties as affected by different rice types. Int. J. Med. Biol. Sci. 2012, 6, 51–56. [Google Scholar]
- De Lavergne, M.D.; van de Velde, F.; Stieger, M. Bolus matters: The influence of food oral breakdown on dynamic texture perception. Food Funct. 2017, 8, 464–480. [Google Scholar] [CrossRef] [PubMed]
- Jalabert-Malbos, M.-L.; Mishellany-Dutour, A.; Woda, A.; Peyron, M.-A. Particle size distribution in the food bolus after mastication of natural foods. Food Qual. Preference 2007, 18, 803–812. [Google Scholar] [CrossRef]
- Foster, K.D.; Grigor, J.; Cheong, J.N.; Yoo, M.J.; Bronlund, J.E.; Morgenstern, M.P. The role of oral processing in dynamic sensory perception. J. Food Sci. 2011, 76, R49–R61. [Google Scholar] [CrossRef] [PubMed]
- Engmann, J.; Burbidge, A.S. Fluid mechanics of eating, swallowing and digestion–overview and perspectives. Food Funct. 2013, 4, 443–447. [Google Scholar] [CrossRef] [PubMed]
- Fontijn-Tekamp, F.A.; van der Bilt, A.; Abbink, J.H.; Bosman, F. Swallowing threshold and masticatory performance in dentate adults. Physiol. Behav. 2004, 83, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Mishellany, A.; Woda, A.; Labas, R.; Peyron, M.-A. The challenge of mastication: Preparing a bolus suitable for deglutition. Dysphagia 2006, 21, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L.J.; de Wijk, R.A.; Gavião, M.B.D.; van der Bilt, A. Effects of added fluids on the perception of solid food. Physiol. Behav. 2006, 88, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Van Der Bilt, A.; Engelen, L.; Abbink, J.; Pereira, L.J. Effects of adding fluids to solid foods on muscle activity and number of chewing cycles. Eur. J. Oral Sci. 2007, 115, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Derks, J.; De Wijk, R.; De Graaf, C.; Stieger, M. Influence of stimulus properties and sensory task instructions on oral processing behavior of liquid stimuli. J. Texture Stud. 2016, 47, 49–57. [Google Scholar] [CrossRef]
- De Wijk, R.A.; Prinz, J.F. Mechanisms underlying the role of friction in oral texture. J. Texture Stud. 2006, 37, 413–427. [Google Scholar] [CrossRef]
- De Wijk, R.; Zijlstra, N.; Mars, M.; De Graaf, C.; Prinz, J. The effects of food viscosity on bite size, bite effort and food intake. Physiol. Behav. 2008, 95, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Engelen, L.; de Wijk, R.A.; van der Bilt, A.; Prinz, J.F.; Janssen, A.M.; Bosman, F. Relating particles and texture perception. Physiol. Behav. 2005, 86, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Lucey, J. Formation and physical properties of yogurt. Asian-Australas. J. Anim. Sci. 2010, 23, 1127–1136. [Google Scholar] [CrossRef]
- Kim, E.H.J.; Jakobsen, V.B.; Wilson, A.J.; Waters, I.R.; Motoi, L.; Hedderley, D.I.; Morgenstern, M.P. Oral processing of mixtures of food particles. J. Texture Stud. 2015, 46, 487–498. [Google Scholar] [CrossRef]
- Loret, C.; Walter, M.; Pineau, N.; Peyron, M.; Hartmann, C.; Martin, N. Physical and related sensory properties of a swallowable bolus. Physiol. Behav. 2011, 104, 855–864. [Google Scholar] [CrossRef] [PubMed]
- De Wijk, R.A.; Janssen, A.M.; Prinz, J.F. Oral movements and the perception of semi-solid foods. Physiol. Behav. 2011, 104, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Bobroff, E.M.; Kissileff, H.R. Effects of changes in palatability on food intake and the cumulative food intake curve in man. Appetite 1986, 7, 85–96. [Google Scholar] [CrossRef]
- Liu, D.; Deng, Y.; Sha, L.; Hashem, M.A.; Gai, S. Impact of oral processing on texture attributes and taste perception. J. Food Sci. Technol. 2017, 54, 2585–2593. [Google Scholar] [CrossRef] [PubMed]
- Salles, C.; Chagnon, M.-C.; Feron, G.; Guichard, E.; Laboure, H.; Morzel, M.; Semon, E.; Tarrega, A.; Yven, C. In-mouth mechanisms leading to flavor release and perception. Crit. Rev. Food Sci. Nutr. 2010, 51, 67–90. [Google Scholar] [CrossRef] [PubMed]
- Bellisle, F.; Le Magnen, J. The analysis of human feeding patterns: The edogram. Appetite 1980, 1, 141–150. [Google Scholar] [CrossRef]
- Rock, C.L.; Flatt, S.W.; Barkai, H.-S.; Pakiz, B.; Heath, D.D. A walnut-containing meal had similar effects on early satiety, CCK, and PYY, but attenuated the postprandial GLP-1 and insulin response compared to a nut-free control meal. Appetite 2017, 117, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Rock, C.L.; Flatt, S.W.; Barkai, H.-S.; Pakiz, B.; Heath, D.D. Walnut consumption in a weight reduction intervention: Effects on body weight, biological measures, blood pressure and satiety. Nutr. J. 2017, 16, 76. [Google Scholar] [CrossRef] [PubMed]
- Mattes, R.D.; Considine, R.V. Oral processing effort, appetite and acute energy intake in lean and obese adults. Physiol. Behav. 2013, 120, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Burton-Freeman, B. Sex and cognitive dietary restraint influence cholecystokinin release and satiety in response to preloads varying in fatty acid composition and content. J. Nutr. 2005, 135, 1407–1414. [Google Scholar] [CrossRef] [PubMed]
- Farr, O.M.; Tuccinardi, D.; Upadhyay, J.; Oussaada, S.M.; Mantzoros, C.S. Walnut consumption increases activation of the insula to highly desirable food cues: A randomized, double-blind, placebo-controlled, cross-over fMRI study. Diabetes Obes. Metab. 2018, 20, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Prinz, J.F.; Lucas, P.W. An optimization model for mastication and swallowing in mammals. Proc. R. Soc. B Biol. Sci. 1997, 264, 1715–1721. [Google Scholar] [CrossRef] [PubMed]
- Krop, E.M.; Hetherington, M.M.; Nekitsing, C.; Miquel, S.; Postelnicu, L.; Sarkar, A. Influence of oral processing on appetite and food intake—A systematic review and meta-analysis. Appetite 2018, 125, 253–269. [Google Scholar] [CrossRef] [PubMed]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McArthur, B.M.; Considine, R.V.; Mattes, R.D. Mastication of Nuts under Realistic Eating Conditions: Implications for Energy Balance. Nutrients 2018, 10, 710. https://doi.org/10.3390/nu10060710
McArthur BM, Considine RV, Mattes RD. Mastication of Nuts under Realistic Eating Conditions: Implications for Energy Balance. Nutrients. 2018; 10(6):710. https://doi.org/10.3390/nu10060710
Chicago/Turabian StyleMcArthur, Breanna M., Robert V. Considine, and Richard D. Mattes. 2018. "Mastication of Nuts under Realistic Eating Conditions: Implications for Energy Balance" Nutrients 10, no. 6: 710. https://doi.org/10.3390/nu10060710
APA StyleMcArthur, B. M., Considine, R. V., & Mattes, R. D. (2018). Mastication of Nuts under Realistic Eating Conditions: Implications for Energy Balance. Nutrients, 10(6), 710. https://doi.org/10.3390/nu10060710



