Dietary Iron Bioavailability: Agreement between Estimation Methods and Association with Serum Ferritin Concentrations in Women of Childbearing Age
Abstract
1. Introduction
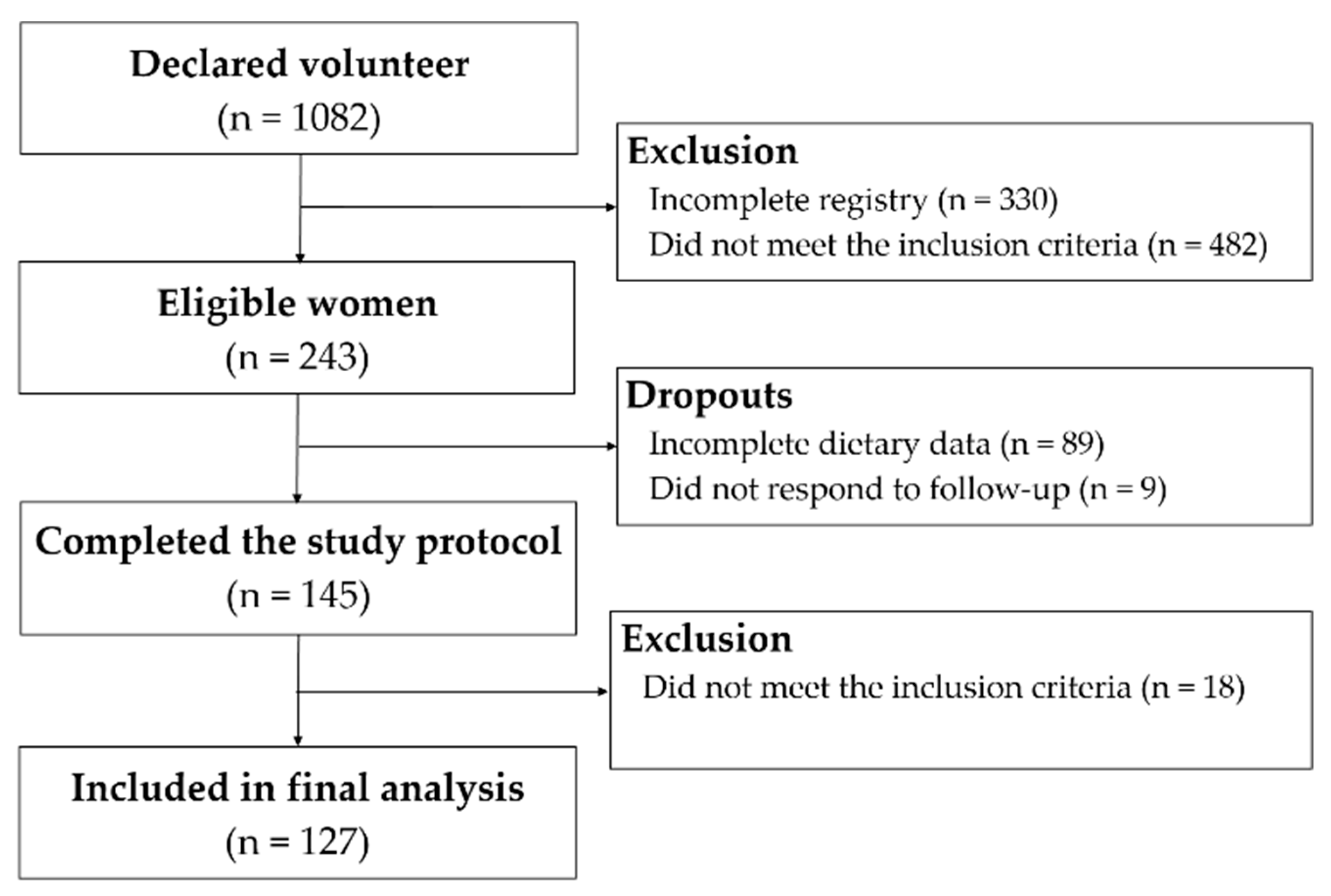
2. Materials and Methods
2.1. Participants
2.2. Dietary Data Acquisition
2.3. Dietary Data Analysis
2.3.1. Probabilistic Approach Estimate
2.3.2. Algorithm Estimates
2.4. Characterization Questionnaires and Anthropometry
2.5. Hematological and Biochemical Determinations
2.6. Statistical Analysis
3. Results
3.1. Participants’ Characteristics
3.2. Dietary Iron Bioavailability Estimates
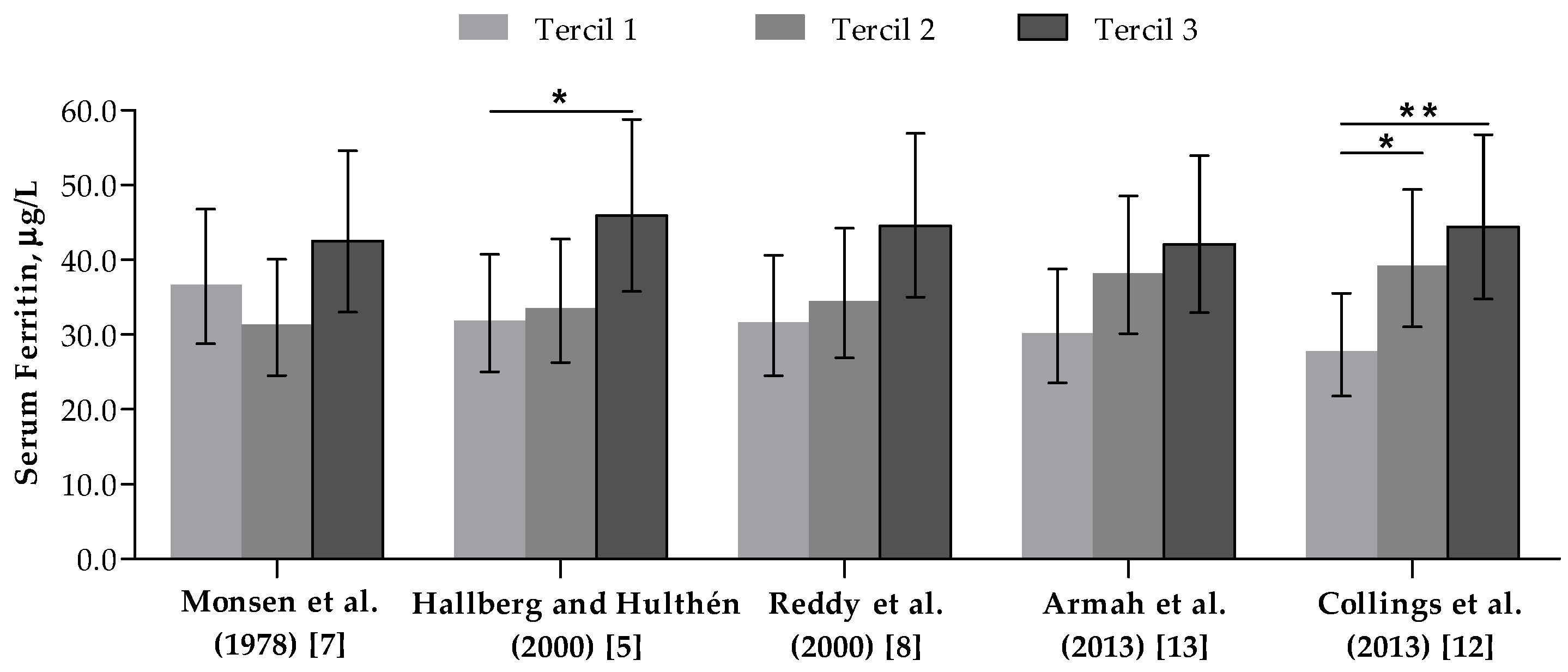
3.3. Correlation between Dietary Iron Bioavailability Estimates
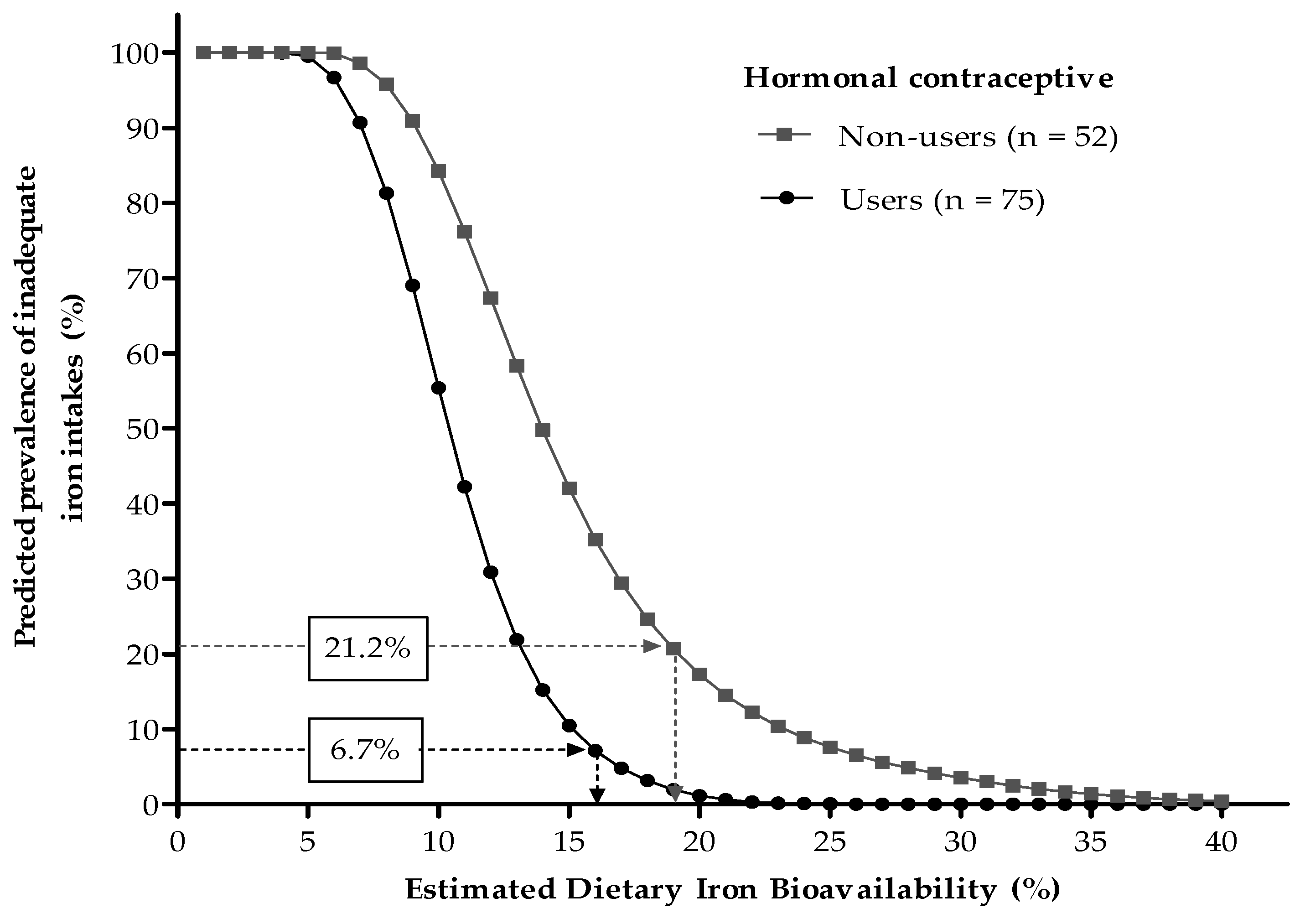
3.4. Association between Dietary Iron Bioavailability Estimates and Serum Ferritin Concentrations
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Nutritional Anaemias: Tools for Effective Prevention and Control; World Health Organization: Geneva, Switzerland, 2017; Available online: http://www.who.int/nutrition/publications/micronutrients/anaemias-tools-prevention-control/en (accessed on 30 March 2018).
- World Health Organization. Iron Deficiency Anaemia: Assessment, Prevention and Control. A Guide for Programme Managers; World Health Organization: Geneva, Switzerland, 2001; Available online: http://www.who.int/nutrition/publications/en/ida_assessment_prevention_control.pdf (accessed on 30 March 2018).
- Cook, J.D.; Dassenko, S.A.; Lynch, S.R. Assessment of the role of nonheme-iron availability in iron balance. Am. J. Clin. Nutr. 1991, 54, 717–722. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, M.; Hulthén, L.; Hallberg, L. The importance of bioavailability of dietary iron in relation to the expected effect from iron fortification. Eur. J. Clin. Nutr. 2008, 62, 761–769. [Google Scholar] [CrossRef] [PubMed]
- Hallberg, L.; Hulthén, L. Prediction of dietary iron absorption: An algorithm for calculating absorption and bioavailability of dietary iron. Am. J. Clin. Nutr. 2000, 71, 1147–1160. [Google Scholar] [CrossRef] [PubMed]
- Hallberg, L.; Hulthén, L.; Gramatkovski, E. Iron absorption from the whole diet in men: How effective is the regulation of iron absorption? Am. J. Clin. Nutr. 1997, 66, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Monsen, E.R.; Hallberg, L.; Layrisse, M.; Hegsted, D.M.; Cook, J.D.; Mertz, W.; Finch, C.C. Estimation of available dietary iron. Am. J. Clin. Nutr. 1978, 31, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Reddy, M.B.; Hurrell, R.F.; Cook, J.D. Estimation of nonheme-iron bioavailability from meal composition. Am. J. Clin. Nutr. 2000, 71, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Chiplonkar, S.A.; Agte, V.V. Statistical model for predicting non-heme iron bioavailability from vegetarian meals. Int. J. Food Sci. Nutr. 2006, 57, 434–450. [Google Scholar] [CrossRef] [PubMed]
- Conway, R.E.; Powell, J.J.; Geissler, C.A. A food-group based algorithm to predict non-heme iron absorption. Int. J. Food Sci. Nutr. 2007, 58, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Rickard, A.P.; Chatfield, M.D.; Conway, R.E.; Stephen, A.M.; Powell, J.J. An algorithm to assess intestinal iron availability for use in dietary surveys. Br. J. Nutr. 2009, 102, 1678–1685. [Google Scholar] [CrossRef] [PubMed]
- Collings, R.; Harvey, L.J.; Hooper, L.; Hurst, R.; Brown, T.J.; Ansett, J.; King, M.; Fairweather-Tait, S.J. The absorption of iron from whole diets: A systematic review. Am. J. Clin. Nutr. 2013, 98, 65–81. [Google Scholar] [CrossRef] [PubMed]
- Armah, S.M.; Carriquiry, A.; Sullivan, D.; Cook, J.D.; Reddy, M.B. A Complete Diet-Based Algorithm for Predicting Nonheme Iron Absorption in Adults. J. Nutr. 2013, 143, 1136–1140. [Google Scholar] [CrossRef] [PubMed]
- Dainty, J.R.; Berry, R.; Lynch, S.R.; Harvey, L.J.; Fairweather-Tait, S.J. Estimation of Dietary Iron Bioavailability from Food Iron Intake and Iron Status. PLoS ONE 2014, 9, e111824. [Google Scholar] [CrossRef] [PubMed]
- Reddy, M.B. Algorithms to assess non-Heme Iron Bioavailability. Int. J. Vitam. Nutr. Res. 2005, 75, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Lynch, S. The Precision of in vitro Methods and Algorithms for Predicting the Bioavailability of Dietary Iron. Int. J. Vitam. Nutr. Res. 2005, 75, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Hunt, J.R. Algorithms for Iron and Zinc Bioavailability: Are they Accurate? Int. J. Vitam. Nutr. Res. 2010, 80, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.B.; Chaouki, N.; Hurrell, R.F. Iron deficiency due to consumption of a habitual diet low in bioavailable iron: A longitudinal cohort study in Moroccan children. Am. J. Clin. Nutr. 2005, 81, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Beard, J.L.; Murray-Kolb, L.E.; Haas, J.D.; Lawrence, F. Iron Absorption Prediction Equations Lack Agreement and Underestimate Iron Absorption. J. Nutr. 2007, 137, 1741–1746. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.D.; Lipschitz, D.A.; Miles, L.E.M.; Finch, C.A. Serum ferritin as a measure of iron stores in normal subjects. Am. J. Clin. Nutr. 1974, 27, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Fairweather-Tait, S.J.; Jennings, A.; Harvey, L.J.; Berry, R.; Walton, J.; Dainty, J.R. Modeling tool for calculating dietary iron bioavailability in iron-sufficient adults. Am. J. Clin. Nutr. 2017, 105, 1408–1414. [Google Scholar] [CrossRef] [PubMed]
- Hultén, L.; Gramatkovski, E.; Gleerup, A.; Hallberg, L. Iron absorption from the whole diet. Relation to meal composition, iron requirements and iron stores. Eur. J. Clin. Nutr. 1995, 49, 794–808. [Google Scholar] [PubMed]
- Hallberg, L.; Hulthén, L.; Garby, L. Iron stores in man in relation to diet and iron requirements. Eur. J. Clin. Nutr. 1998, 52, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Hallberg, H.; Hulthén, L.; Garby, L. Iron stores and haemoglobin iron deficits in menstruating women. Calculations based on variations in iron requirements and bioavailability of dietary iron. Eur. J. Clin. Nutr. 2000, 54, 650–657. [Google Scholar] [CrossRef] [PubMed]
- Hallberg, L. New tools in studies on iron nutrition. Principles, applications and consequences. Scand. J. Food Nutr. 2000, 44, 150–154. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. Human Vitamin and Mineral Requirements; World Health Organization: Rome, Italy, 2001; Available online: http://www.fao.org/docrep/004/y2809e/y2809e00.htm (accessed on 30 March 2018).
- Institute of Medicine (US) Panel on Micronutrients. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; National Academies Press: Washington, DC, USA, 2001; Available online: https://www.nap.edu/read/10026/chapter/1 (accessed on 30 March 2018).
- Galante, A. Desenvolvimento e validação de um método computadorizado para avaliação do consumo alimentar, preenchido por indivíduos adultos utilizando a Web. Ph.D. Thesis, University of São Paulo (USP), São Paulo, Brazil, 12 May 2007. Available online: http://www.teses.usp.br/teses/disponiveis/89/89131/tde-11032013-155020/pt-br.php (accessed on 30 March 2018).
- Fisberg, R.M.; Villar, B.S. Manual de Receitas e Medidas Caseiras para Cálculo de Inquéritos Alimentares, 1st ed.; Signus: São Paulo, Brazil, 2002. [Google Scholar]
- Pinheiro, A.B.V. Tabela para Avaliação de Consumo Alimentar em Medidas Caseiras, 5th ed; Atheneu: São Paulo, Brazil, 2008. [Google Scholar]
- NEPA—UNICAMP. Tabela Brasileira de Composição de Alimentos, 4th ed.; NEPA—UNICAMP: Campinas, Brazil, 2011; Available online: http://www.nepa.unicamp.br/taco/tabela.php?ativo=tabela (accessed on 31 March 2018).
- Universidade de São Paulo. Faculdade de Ciências Farmacêuticas, Departamento de Alimentos e Nutrição Experimental/BRASILFOODS. Tabela Brasileira de Composição de Alimentos-USP, 5th ed.; Universidade de São Paulo: São Paulo, Brazil, 1998; Available online: http://www.intranet.fcf.usp.br/tabela/ (accessed on 31 March 2018).
- US Department of Agriculture, Agricultural Research Service, Nutrient Data Laboratory. USDA National Nutrient Database for Standard Reference, 28th ed.; Nutrient Data Laboratory: Beltsville, MD, USA, 2015. Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 31 March 2018).
- Kongkachuichai, R.; Napatthalung, P.; Charoensiri, R. Heme and Nonheme Iron Content of Animal Products Commonly Consumed in Thailand. J. Food Compost. Anal. 2002, 15, 389–398. [Google Scholar] [CrossRef]
- Schönfeldt, H.C.; Hall, N.G.H. Determining iron bio-availability with a constant heme iron value. J. Food Compost. Anal. 2011, 24, 738–740. [Google Scholar] [CrossRef]
- Mistura, L.P.F. Cinética de ruptura do ferro heme em carne bovina (coxão mole—semi membranosus) submetida a diferentes tratamentos térmicos. Ph.D. Thesis, University of São Paulo, São Paulo, Brazil, 8 December 2006. Available online: http://www.teses.usp.br/teses/disponiveis/9/9132/tde-04042007-122100/pt-br.php (accessed on 31 March 2018).
- Harland, B.F.; Oberleas, D. Phytate contents of foods. In CRC Handbook of Dietary Fiber in Human Nutrition, 3rd ed.; Spiller, G.A., Ed.; CRC Press: Boca Raton, FL, USA, 1993; p. 673. [Google Scholar]
- Mccrory, M.A.; Hajduk, C.L.; Roberts, S.B. Procedures for screening out inaccurate reports of dietary energy intake. Public Health Nutr. 2006, 5, 873–882. [Google Scholar] [CrossRef] [PubMed]
- Vinken, A.G.; Bathalon, G.P.; Sawaya, A.L.; Dallal, G.E.; Tucker, K.L.; Roberts, S.B. Equations for predicting the energy requirements of healthy adults aged 18–81 y. Am. J. Clin. Nutr. 1999, 69, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Fisberg, R.M.; Colucci, A.C.A.; Morimoto, J.M.; Marchioni, D.M.L. Food frequency questionnaire for adults from a population-based study. Rev. Saude Publica 2008, 42, 1–4. [Google Scholar]
- Haubrock, J.; Nöthlings, U.; Volatier, J.L.; Dekkers, A.; Ocké, M.; Harttig, U.; Illner, A.K.; Knüppel, S.; Andersen, L.F.; Boeing, H.; et al. Estimating Usual Food Intake Distributions by Using the Multiple Source Method in the EPIC-Potsdam Calibration Study. J. Nutr. 2011, 141, 914–920. [Google Scholar] [CrossRef] [PubMed]
- Harttig, U.; Haubrock, J.; Knuppel, S.; Boeing, H.; EFCOVAL Consortium. The MSM program: Web-based statistics package for estimating usual dietary intake using the Multiple Source Method. Eur. J. Clin. Nutr. 2011, 65, S87–S91. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.C.; Howe, G.R.; Kushi, L.H. Adjustment for total energy intake in epidemiologic studies. Am. J. Clin. Nutr. 1997, 65, 1220S–1228S. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013; Available online: http://www.R-project.org (accessed on 31 March 2018).
- World Health Organization. Serum Ferritin Concentrations for the Assessment of Iron Status and Iron Deficiency in Populations; World Health Organization: Geneva, Switzerland, 2011; Available online: http://www.who.int/vmnis/indicators/serum_ferritin.pdf (accessed on 31 March 2018).
- Agência Nacional de Vigilância Sanitária (ANVISA). Resolução 344/2002: Regulamento Técnico para a Fortificação das Farinhas de Trigo e das Farinhas de Milho com Ferro e Ácido Fólico; ANVISA: Brasília, Brazil, 2002. Available online: http://portal.anvisa.gov.br/documents/10181/2718376/RDC_344_2002_COMP.pdf/b4d87885-dcb9-4fe3-870d-db57921cf73f (accessed on 31 March 2018).
- Allen, L.H.; De Benoist, B.; Omar, D.; Hurrell, R. Guidelines on Food Fortification with Micronutrients; World Health Organization: Geneva, Switzerland, 2006; Available online: http://www.who.int/nutrition/publications/micronutrients/9241594012/en (accessed on 31 March 2018).
- Assunção, M.C.F.; Santos, I.S.; Barro, A.J.; Gigante, D.P.; Victora, C.G. Flour fortification with iron has no impact on anaemia in urban Brazilian children. Public Health Nutr. 2012, 15, 1796–1801. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, M.; Sjöberg, A.; Hallberg, L.; Hulthén, L. Iron status in Swedish teenage girls: Impact of low dietary iron bioavailability. Nutrition 2008, 24, 638–645. [Google Scholar] [CrossRef] [PubMed]
- Associação Brasileira de Empresas de Pesquisa (ABEP). Critério Brasil 2014; ABEP: São Paulo, Brazil, 2014; Available online: http://www.abep.org/criterio-brasil (accessed on 31 March 2018).
- Matsudo, S.; Araújo, T.; Matsudo, V.; Andrade, D.; Andrade, E.; Oliveira, L.C.; Braggion, G. International physical activity questionnaire (IPAQ): Study of validity and reability in Brazil. Rev. Bras. Ativ. Fís. Saúde 2001, 6, 5–18. [Google Scholar]
- World Health Organization. Physical Status: The Use of and Interpretation of Anthropometry; World Health Organization: Geneva, Switzerland, 1995; Available online: http://www.who.int/childgrowth/publications/physical_status/en (accessed on 31 March 2018).
- World Health Organization. Waist Circumference and Waist–Hip Ratio; World Health Organization: Geneva, Switzerland, 2011; Available online: http://www.who.int/nutrition/publications/obesity/WHO_report_waistcircumference_and_waisthip_ratio/en (accessed on 31 March 2018).
- Thurnham, D.I.; McCabe, G.P. Influence of infection and inflammation on biomarkers of nutritional status with an emphasis on vitamin A and iron. In Report: Priorities in the Assessment of Vitamin A and Iron Status in Populations, Panama City, Panama, 15–17 September 2010; World Health Organization: Geneva, Switzerland, 2012; Available online: http://www.who.int/nutrition/publications/micronutrients/background_paper4_report_assessment_vitAandIron_status.pdf (accessed on 31 March 2018).
- Armah, S.M.; Carriquiry, A.L.; Reddy, M.B. Total Iron Bioavailability from the US Diet Is Lower Than the Current Estimate. J. Nutr. 2015, 145, 2617–2621. [Google Scholar] [CrossRef] [PubMed]
- Perignon, M.; Barré, T.; Gazan, R.; Amiot, M.J.; Darmon, N. The bioavailability of iron, zinc, protein and vitamin A is highly variable in French individual diets: Impact on nutrient inadequacy assessment and relation with the animal-to-plant ratio of diets. Food Chem. 2018, 238, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Subar, A.F.; Freedman, L.S.; Tooze, J.A.; Kirkpatrick, S.I.; Boushey, C.; Neuhouser, M.L.; Thompson, F.E.; Potischman, N.; Guenther, P.M.; Tarasuk, V.; et al. Addressing Current Criticism Regarding the Value of Self-Report Dietary Data. J. Nutr. 2015, 145, 2639–2645. [Google Scholar] [CrossRef] [PubMed]
- Instituto Brasileiro de Geografia e Estatística (IBGE). Pesquisa de Orçamentos Familiares 2008–2009: Análise do Consumo Alimentar Pessoal No Brasil; IBGE: Rio de Janeiro, Brazil, 2011. Available online: https://biblioteca.ibge.gov.br/visualizacao/livros/liv50063.pdf (accessed on 31 March 2018).
- Dos Santos, Q.; Nilson, E.A.; Verly, E.J.; Sichieri, R. An evaluation of the effectiveness of the flour iron fortification programme in Brazil. Public Health Nutr. 2015, 18, 1670–1674. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, A.M.; César, C.L.; Fisberg, R.M.; Marchioni, D.M. Meat Consumption in Sao Paulo—Brazil: Trend in the Last Decade. PLoS ONE 2014, 9, e96667. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, M.A.; Cominetti, C.; Kakazu, M.H.; Sarkis, J.E.; Dainty, J.; Fox, T.E.; Cozzolino, S.M. Zinc absorption in Brazilian subjects fed a healthy meal. J. Hum. Nutr. Diet. 2014, 27, 313–320. [Google Scholar] [CrossRef] [PubMed]
| Algorithm Model | Calculus Basis | Dietary Factors Adjusting Non-Heme Iron Absorption | Dietary Factors Adjusting Heme Iron Absorption | Adjustment Made for Individual’s Body Iron Status |
|---|---|---|---|---|
| Monsen et al. (1978) [7] | Meals | Cooked animal tissues Vitamin C | None | Heme and nonheme iron absorption are corrected for the body iron stores of 0, 250, 500, and 1000 mg, using specific correction factors. |
| Hallberg and Hulthén (2000) [5] | Meals | Raw animal tissues Vitamin C Alcohol Phytate phosphorous Calcium Polyphenols Soy Protein Eggs | Calcium | Heme and nonheme iron absorptions are corrected according to serum ferritin concentration, using two independent equations. |
| Reddy et al. (2000) [8] | Meals | Cooked animal tissues Vitamin C Phytic acid | None | Nonheme iron absorption is corrected according to serum ferritin concentration, using an equation proposed by Cook [3]. Lacks a correction term for heme iron absorption. A fixed factor of 25% of heme iron absorption was assumed, as proposed by the Institute of Medicine [27]. |
| Armah et al. (2013) [13] | Complete diets | Cooked animal tissues Vitamin C Phytic acid Calcium Tea and coffee equivalents Non-heme iron | None | Nonheme iron absorption is corrected according to serum ferritin concentration, using an equation incorporate in the same model of adjustments for dietary factors. Lacks a correction term for heme iron absorption. A fixed factor of 25% of heme iron absorption was assumed, as proposed by the Institute of Medicine [27]. |
| Collings et al. (2013) [12] | Complete diets | Diets are categorized into 3 types:
| None | Nonheme iron absorption is corrected according to serum ferritin concentration, using an equation incorporate in the same model of adjustments for dietary factors. Lacks a correction term for heme iron absorption. A fixed factor of 25% of heme iron absorption was assumed, as proposed by the Institute of Medicine [27]. |
| Selected Characteristics | Overall Sample | Hormonal Contraceptives | |
|---|---|---|---|
| Non-users | Users | ||
| (n = 127) | (n = 52) | (n = 75) | |
| Sociodemographic and lifestyle characterization | |||
| Age (years) | 27.4 (5.1) [18.0–42.0] | 27.8 (6.0) | 27.2 (4.3) |
| >30 year, n (%) | 34 (26.8) | 19 (36.5) | 15 (20.0) # |
| Body Mass Index (kg/m2) | 22.1 (2.4) [17.0–29.1] | 21.8 (2.4) | 22.2 (2.4) |
| >25 Kg/m2, n (%) | 14 (11.0) | 5 (9.6) | 9 (12.0) |
| Waist circumference (cm) | 75.4 (7.1) [60.5–99.3] | 75.9 (7.1) | 75.5 (7.1) |
| >80 cm, n (%) | 33 (26.0) | 15 (28.8) | 18 (24.0) |
| Self-declared skin color/race, n (%) | |||
| White | 95 (74.8) | 36 (69.2) | 59 (78.7) |
| Black or brown/mixed | 24 (18.9) | 11 (21.2) | 13 (17.3) |
| Yellow/Asian | 8 (6.3) | 5 (9.6) | 3 (4.0) |
| Socioeconomic level, n (%) * | |||
| Class A | 15 (12.0) | 6 (11.5) | 9 (12.3) |
| Class B | 86 (68.8) | 30 (57.7) | 56 (76.7) # |
| Classes C or D | 24 (19.2) | 16 (30.8) | 8 (11.0) # |
| Self-declared diet, n (%) | |||
| Omnivorous | 109 (85.8) | 43 (82.7) | 66 (88.0) |
| Vegetarians or meat restrictors | 18 (14.2) | 9 (17.3) | 9 (12.0) |
| Physical Activity Level, n (%) | |||
| Active | 62 (48.8) | 22 (42.3) | 40 (53.3) |
| Very active | 16 (12.6) | 7 (13.5) | 9 (12.0) |
| Insufficiently active | 49 (38.6) | 23 (44.2) | 26 (34.7) |
| Menstrual flow characterization | |||
| Periods duration, n (%) | |||
| 2–3 days | 20 (15.7) | 6 (11.5) | 14 (18.7) |
| 4–5 days | 89 (70.1) | 37 (71.2) | 52 (69.3) |
| 6–7 days | 18 (14.2) | 9 (17.3) | 9 (12.0) |
| Menstrual flow intensity score, n (%) | |||
| Tertile 1 (≤13 units) | 44 (34.6) | 14 (26.9) | 30 (40.0) ## |
| Tertile 2 (14–21 units) | 39 (30.7) | 12 (23.1) | 27 (36.0) ## |
| Tertile 3 (≥22 units) | 44 (34.6) | 26 (50.0) | 18 (24.0) ## |
| Hematological and biochemical characterization | |||
| Hemoglobin (g/dL) | 13.4 (0.8) [11.0–15.4] | 13.4 (0.9) | 13.4 (0.8) |
| <12 g/dL, n (%) | 4 (3.1) | 3 (5.8) | 1 (1.3) |
| Transferrin saturation (%) | 29.9 (11.4) [6.4–59.7] | 30.1 (12.8) | 29.8 (10.3) |
| <16%, n (%) | 14 (11.0) | 7 (13.5) | 7 (9.3) |
| Ferritin (µg/L) ** | 36.6 (31.4; 42.6) [4.0–198.0] | 31.0 (23.6; 40.7) | 41.0 (34.3; 49.0) |
| <15 µg/L, n (%) | 16 (12.6) | 11 (21.2) | 5 (6.7) # |
| Alfa1-Acid Glycoprotein (mg/dL) | 61.4 (18.9) [26.0–133.0] | 65.6 (16.4) | 71.9 (20.8) |
| ≥100 mg/dL, n (%) | 4 (3.1) | 2 (3.8) | 2 (2.7) |
| High-sensitive C-Reactive Protein (mg/L) | 1.1 (0.9; 1.3) [0.2–9.8] | 0.5 (0.4; 0.7) | 1.7 (1.4; 2.3) ## |
| ≥5 mg/L, n (%) | 15 (11.8) | 1 (1.9) | 14 (18.7) ## |
| Dietetic characterization | |||
| Total energy (kcal/day) | 2032.5 (436.5) [958.0–3531.9] | 1984.6 (465.5) | 2065.7 (415.1) |
| Total iron (mg/day) | 10.9 (2.2) [6.1–19.5] | 10.3 (2.2) | 11.2 (2.2) # |
| Non-heme iron (mg/day) | 10.2 (2.2) [5.8–18.1] | 9.6 (2.1) | 10.5 (2.2) # |
| Fortification iron (mg/day) | 3.2 (1.2) [0.4–6.3] | 2.9 (1.3) | 3.4 (1.1) # |
| Heme iron (mg/day) | 0.7 (0.4) [0.0–2.1] | 0.7 (0.5) | 0.7 (0.4) |
| Animal tissues (g/day) 1 | 169.9 (76.8) [0.0–325.2] | 172.1 (79.6) | 168.4 (75.2) |
| Vitamin C (mg/day) | 129.0 (81.9) [20.5–434.1] | 121.1 (65.8) | 134.5 (91.5) |
| Calcium (mg/day) | 845.1 (285.8) [210.3–1872.1] | 844.6 (2851) | 845.5 (288.2) |
| Phytate (mg/day) 2 | 260.2 (84.1) [82.0–563.5] | 246.7 (77.7) | 269.5 (87.5) |
| Polyphenols (mg/day) 3 | 223.3 (82.7) [56.4–563.0] | 234.1 (94.1) | 215.8 (73.4) |
| Tea and coffee equivalents (cups/day) 4 | 0.3 (0.3; 0.4) [0.0–2.0] | 0.4 (0.3; 0.5) | 0.3 (0.2; 0.4) |
| Alcohol (g/day) | 1.1 (1.0; 1.3) [0.0–11.3] | 1.1 (0.9; 1.4) | 1.1 (1.0; 1.3) |
| Algorithm Models | Absolute FeBio (mg/day) | Relative FeBio (%) |
|---|---|---|
| Monsen et al. (1978) [7] | 1.26 (0.36) [0.45–2.44] a | 11.57 (2.21) [6.54–17.95] a |
| Hallberg and Hulthén (2000) [5] | 1.30 (0.42) [0.44–2.88] a | 12.02 (3.34) [4.10–24.90] a |
| Reddy et al. (2000) [8] | 1.38 (0.48) [0.40–3.41] a | 12.80 (3.87) [3.61–25.07] a |
| Armah et al. (2013) [13] | 0.96 (0.21) [0.49–1.67] b | 8.91 (1.36) [5.62–13.33] b |
| Collings et al. (2013) [12] | 0.92 (0.23) [0.49–1.84] b | 8.51 (1.04) [4.61–11.44] b |
| Algorithm Models | Monsen et al. (1978) [7] | Hallberg and Hulthén (2000) [5] | Reddy et al. (2000) [8] | Armah et al. (2013) [13] |
|---|---|---|---|---|
| Hallberg and Hulthén (2000) [5] | r = 0.823 y = 1.5x − 0.8 | |||
| Reddy et al. (2000) [8] | r = 0.718 y = 1.7x − 0.9 | r = 0.851 y = 1.2x + 0.0 | ||
| Armah et al. (2013) [13] | r = 0.850 y = 0.6x + 0.1 | r = 0.836 y = 0.4x + 0.4 | r = 0.749 y = 0.3x + 0.4 | |
| Collings et al. (2013) [12] | r = 0.793 y = 0.5x + 0.1 | r = 0.764 y = 0.3x + 0.4 | r = 0.694 y = 0.3x + 0.4 | r = 0.809 y = 0.8x + 0.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Carli, E.; Dias, G.C.; Morimoto, J.M.; Marchioni, D.M.L.; Colli, C. Dietary Iron Bioavailability: Agreement between Estimation Methods and Association with Serum Ferritin Concentrations in Women of Childbearing Age. Nutrients 2018, 10, 650. https://doi.org/10.3390/nu10050650
De Carli E, Dias GC, Morimoto JM, Marchioni DML, Colli C. Dietary Iron Bioavailability: Agreement between Estimation Methods and Association with Serum Ferritin Concentrations in Women of Childbearing Age. Nutrients. 2018; 10(5):650. https://doi.org/10.3390/nu10050650
Chicago/Turabian StyleDe Carli, Eduardo, Gisele Cristina Dias, Juliana Massami Morimoto, Dirce Maria Lobo Marchioni, and Célia Colli. 2018. "Dietary Iron Bioavailability: Agreement between Estimation Methods and Association with Serum Ferritin Concentrations in Women of Childbearing Age" Nutrients 10, no. 5: 650. https://doi.org/10.3390/nu10050650
APA StyleDe Carli, E., Dias, G. C., Morimoto, J. M., Marchioni, D. M. L., & Colli, C. (2018). Dietary Iron Bioavailability: Agreement between Estimation Methods and Association with Serum Ferritin Concentrations in Women of Childbearing Age. Nutrients, 10(5), 650. https://doi.org/10.3390/nu10050650





