Buckwheat and CVD Risk Markers: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources and Literature Search
2.2. Data Extraction
2.3. Statistical Analysis
3. Results
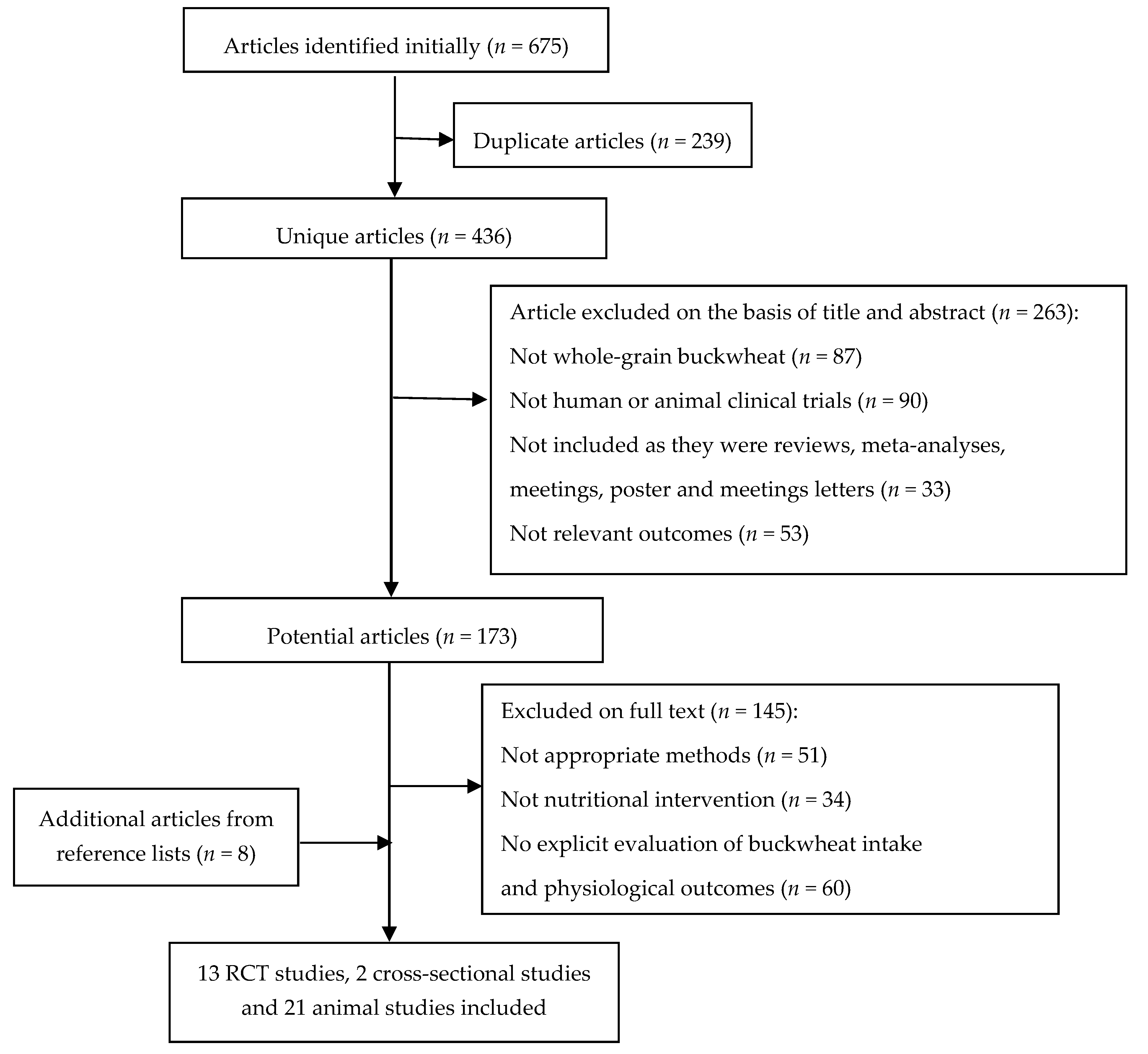
3.1. Study Selection
3.2. Characteristics of Studies
3.3. Human Studies
3.3.1. Effects on Body Weight and BMI
3.3.2. Effects on Blood Pressure
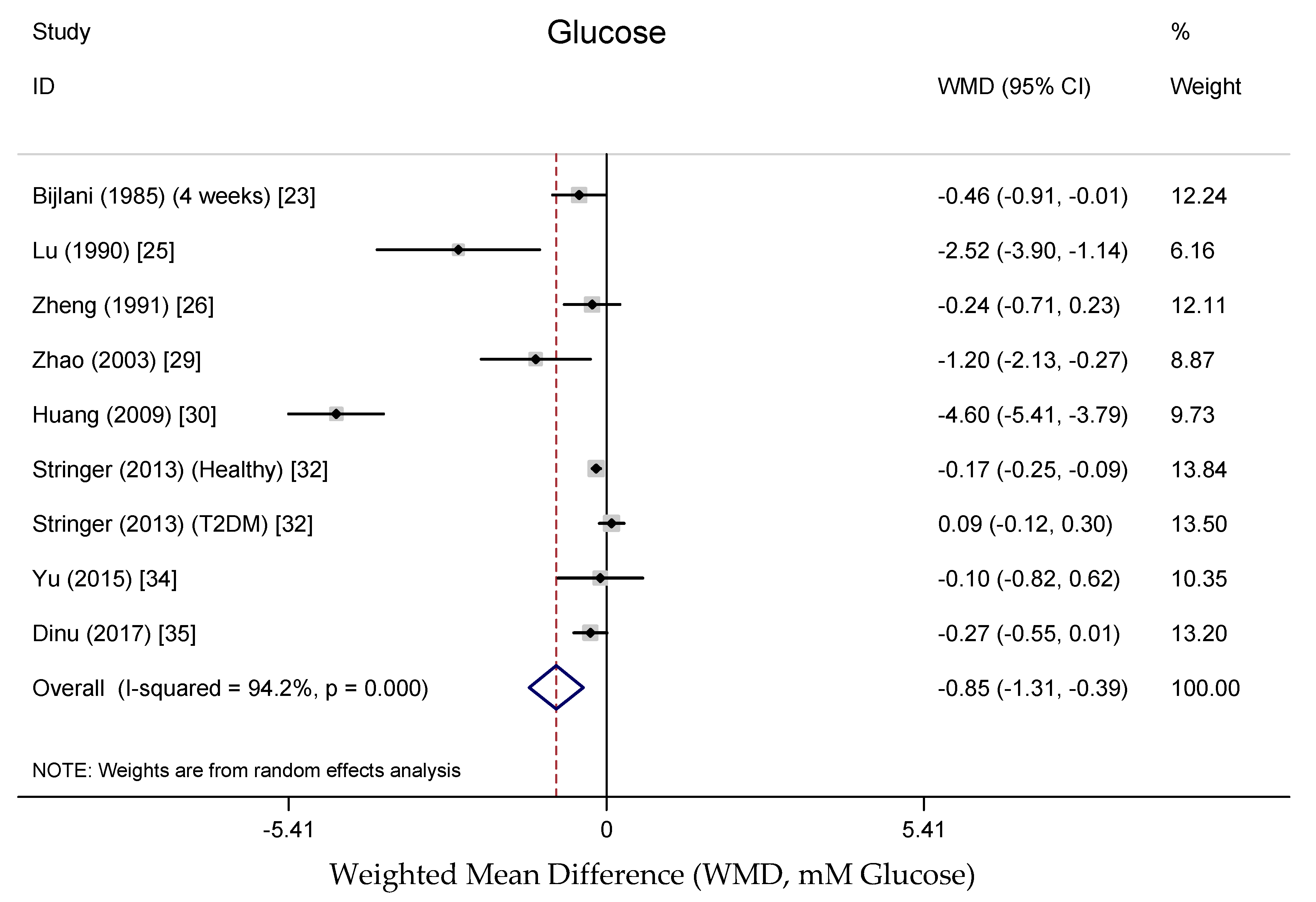
3.3.3. Effects on Blood Glucose and Insulin
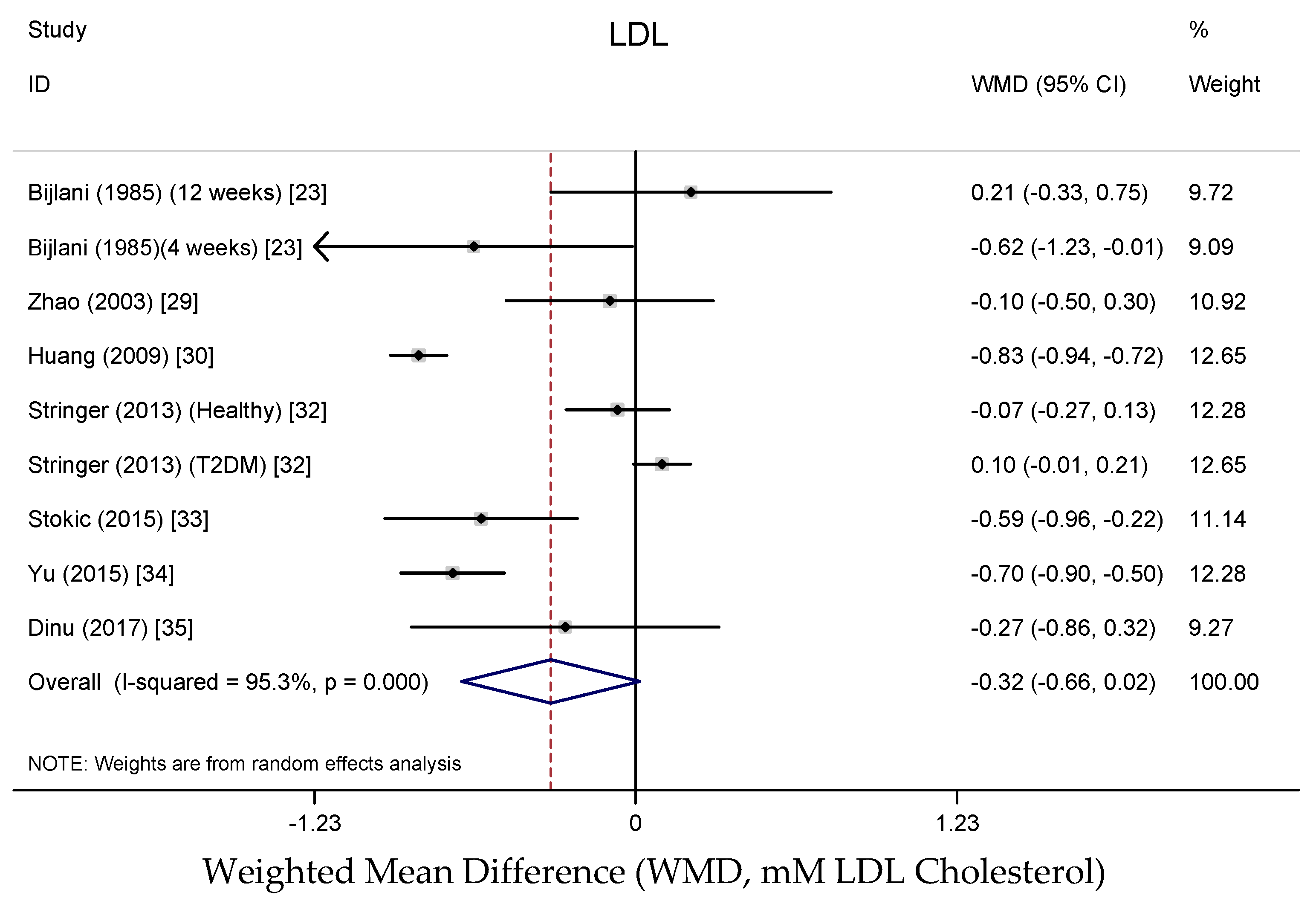
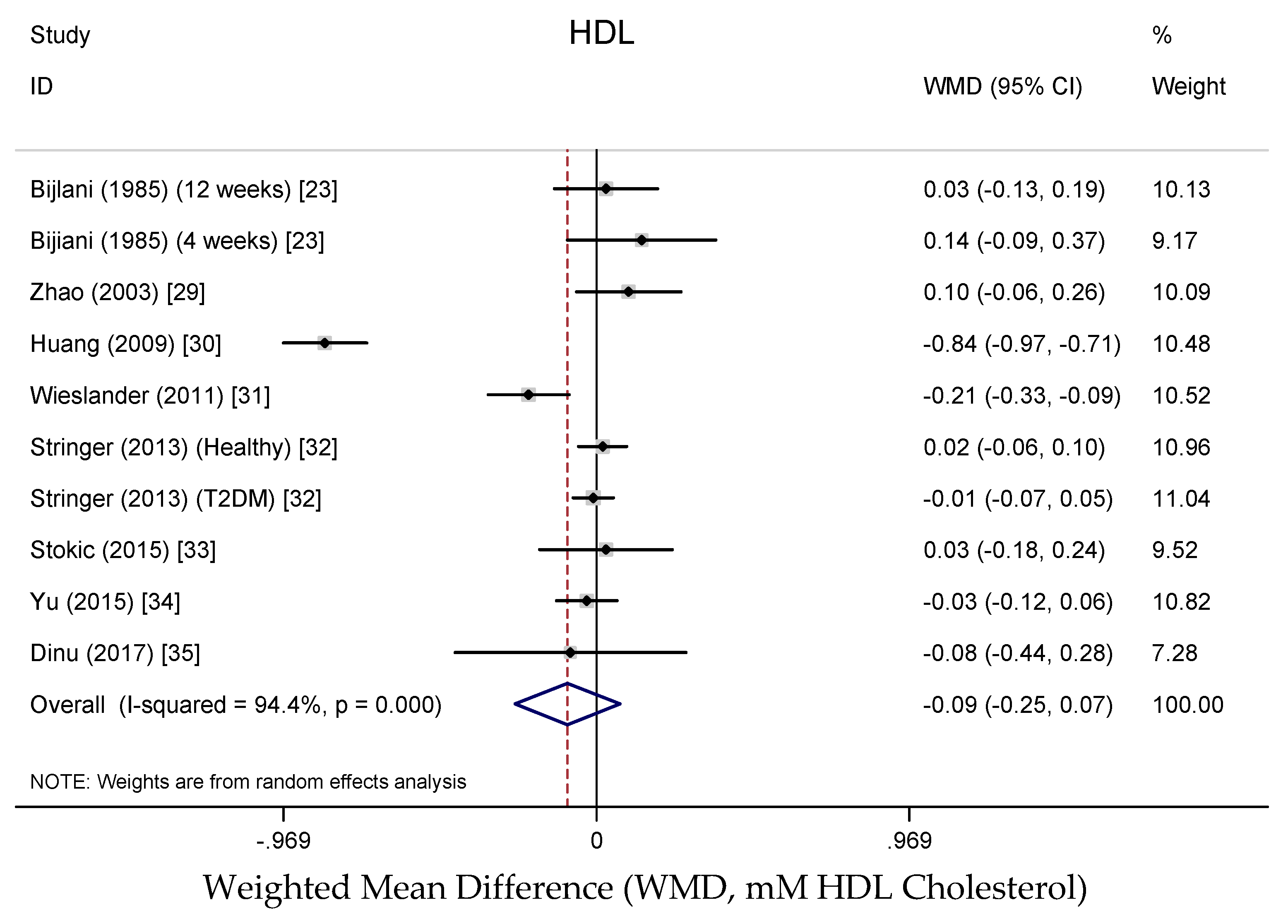
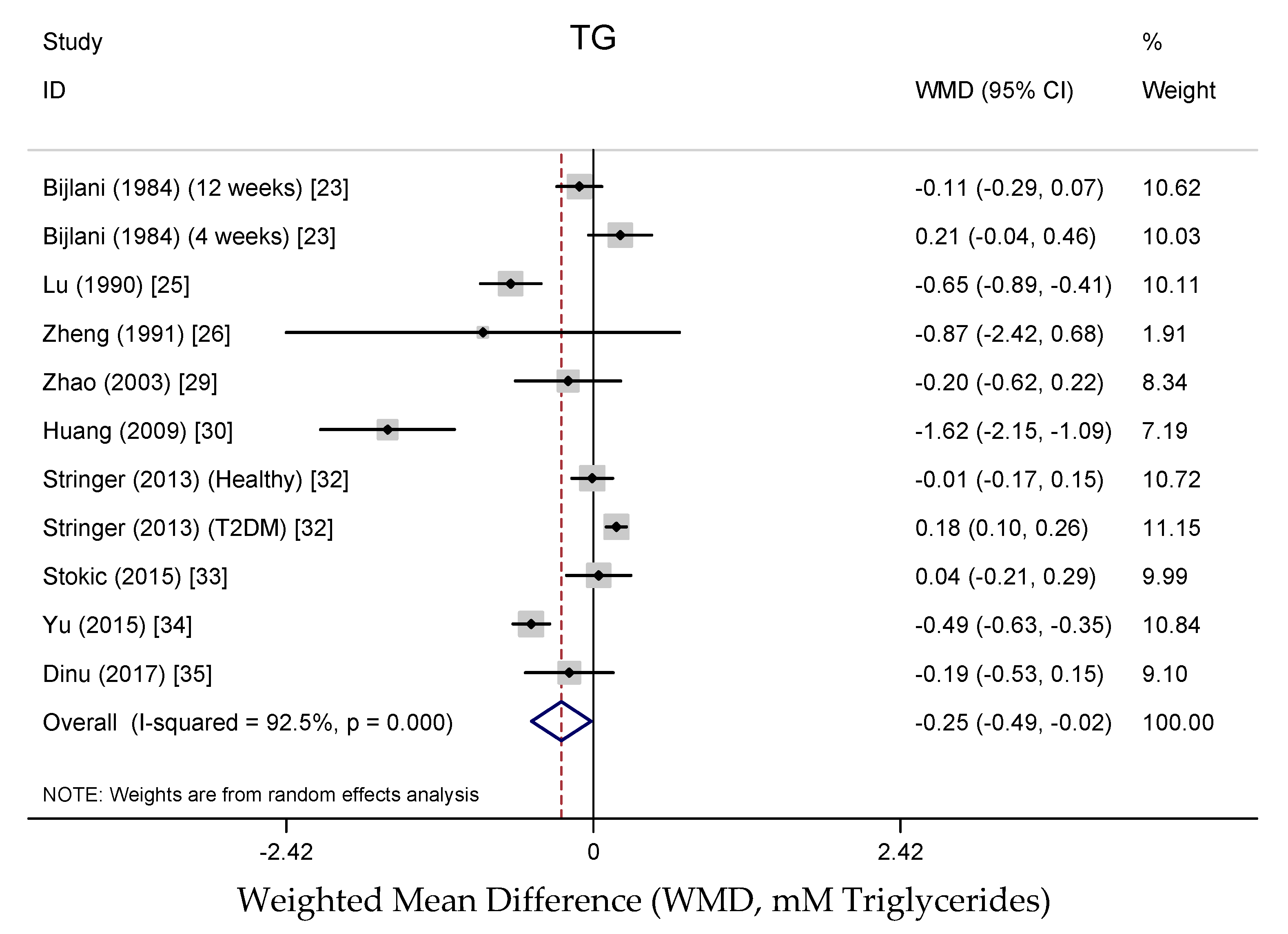
3.3.4. Effects on Lipid Profile
3.3.5. Sensitivity Analyses and Subgroups Analyses
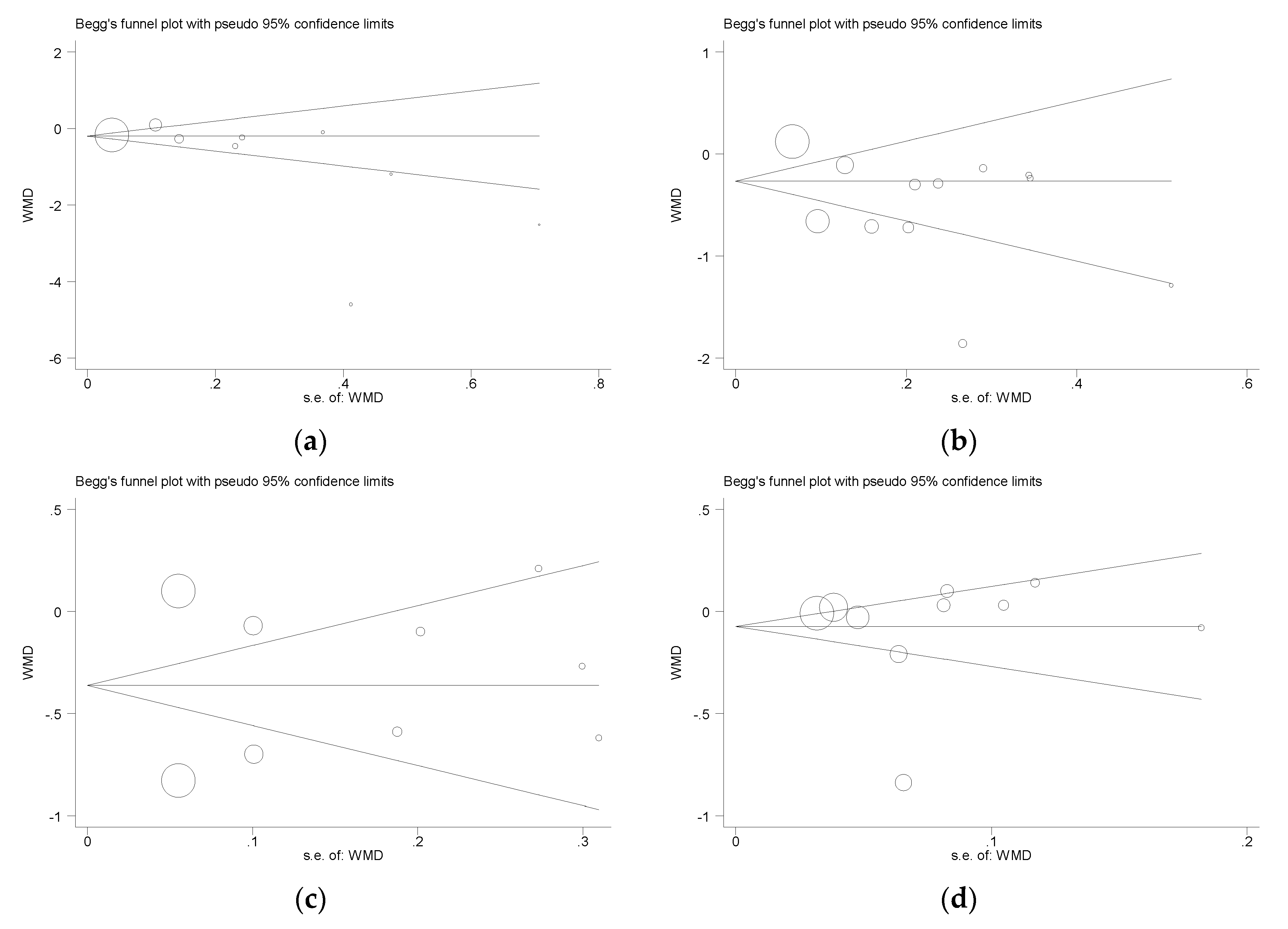
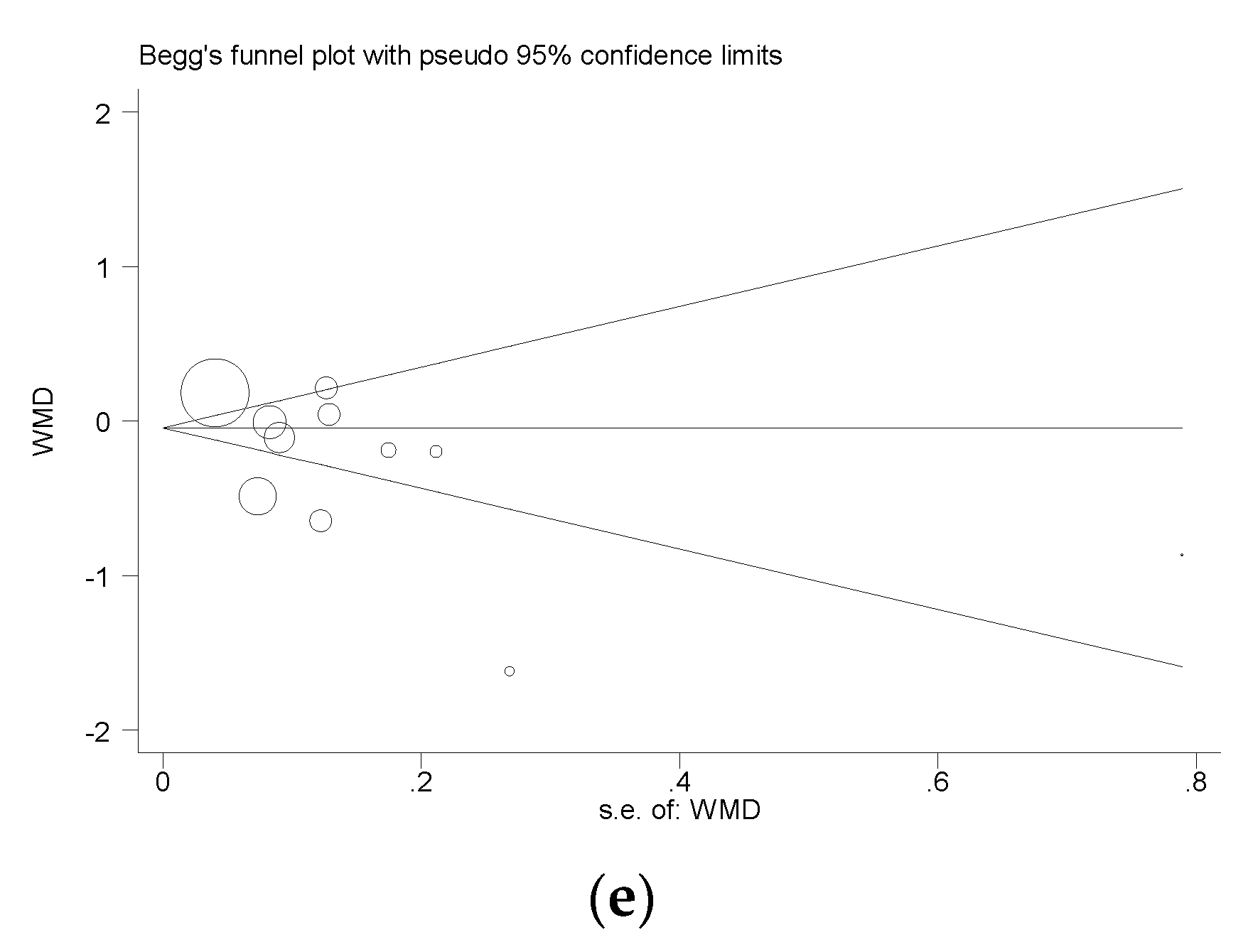
3.3.6. Publication Bias
3.4. Animal Studies
3.4.1. Effects on Weight Gain and Food Intake
3.4.2. Effects on Blood Glucose and Insulin
3.4.3. Effects on Lipid Profile
3.4.4. Other Outcomes
4. Discussion
4.1. Effects on Body Weight
4.2. Effects on Blood Pressure
4.3. Effects on Blood Glucose and Insulin
4.4. Effects on Lipid Profile
4.5. Buckwheat Intake Levels
4.6. Bioactive Compounds Responsible for Lipid-Lowering Activity
4.7. Sensitivity Analysis
4.8. Limitations
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- WHO. Diet, Nutrition and the Prevention of Chronic Diseases. Joint Who/FAO Expert Consultation; WHO Technical Report Series no. 916; WHO: Geneva, Switzerland, 2003. [Google Scholar]
- Liu, S.; Stampfer, M.J.; Hu, F.B.; Giovannucci, E.; Rimm, E.; Manson, J.E.; Hennekens, C.H.; Willett, W.C. Whole-grain consumption and risk of coronary heart disease: Results from the nurses’ health study. Am. J. Clin. Nutr. 1999, 70, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.R.; Pereira, M.A.; Kushi, L.H.; Meyer, K.A. Fiber from whole grains, but not refined grains, is inversely associated with all-cause mortality in older women: The iowa women’s health study. J. Am. Coll. Nutr. 2000, 19, 326S–330S. [Google Scholar] [CrossRef] [PubMed]
- Mellen, P.B.; Walsh, T.F.; Herrington, D.M. Whole grain intake and cardiovascular disease: A meta-analysis. Nutr. Metab. Cardiovasc. Dis. 2008, 18, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Whole grain consumption and risk of cardiovascular disease, cancer, and all cause and cause specific mortality: Systematic review and dose-response meta-analysis of prospective studies. BMJ 2016, 353, i2716. [Google Scholar] [CrossRef] [PubMed]
- Ge, K. The transition of chinese dietary guidelines and the food guide pagoda. Asia Pac. J. Clin. Nutr. 2011, 20, 439–446. [Google Scholar] [PubMed]
- Critchley, J.; Liu, J.; Zhao, D.; Wei, W.; Capewell, S. Explaining the increase in coronary heart disease mortality in Beijing between 1984 and 1999. Circulation 2004, 110, 1236–1244. [Google Scholar] [CrossRef] [PubMed]
- Van der Kamp, J.W.; Poutanen, K.; Seal, C.J.; Richardson, D.P. The healthgrain definition of ‘whole grain’. Food Nutr. Res. 2014, 58, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Li, S.Q.; Zhang, Q.H. Advances in the development of functional foods from buckwheat. Crit. Rev. Food Sci. 2001, 41, 451–464. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.; Gan, R.; Dai, S.; Cai, Y.Z.; Corke, H.; Zhu, F. Buckwheat and millet affect thermal, rheological, and gelling properties of wheat flour. J. Food Sci. 2016, 81, E627–E636. [Google Scholar] [CrossRef] [PubMed]
- Bonafaccia, G.; Marocchini, M.; Kreft, I. Composition and technological properties of the, flour and bran from common and Tartary buckwheat. Food Chem. 2003, 80, 9–15. [Google Scholar] [CrossRef]
- Gimenez-Bastida, J.A.; Piskula, M.K.; Zielinski, H. Recent advances in processing and development of buckwheat derived bakery and non-bakery products—A review. Pol. J. Food Nutr. Sci. 2015, 65, 9–20. [Google Scholar] [CrossRef]
- Krkošková, B.; Mrázová, Z. Prophylactic components of buckwheat. Food Res. Int. 2005, 38, 561–568. [Google Scholar] [CrossRef]
- Saturni, L.; Ferretti, G.; Bacchetti, T. The gluten-free diet: Safety and nutritional quality. Nutrients 2010, 2, 16–34. [Google Scholar] [CrossRef] [PubMed]
- Gimenez-Bastida, J.A.; Zielinski, H. Buckwheat as a functional food and its effects on health. J. Agric. Food Chem. 2015, 63, 7896–7913. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. Br. Med. J. 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.W.; Zhang, Y.H.; Lu, M.J.; Tong, W.J.; Cao, G.W. Comparison of hypertension, dyslipidaemia and Hyperglycaemia between buckwheat seed-consuming and non-consuming Mongolian-Chinese populations in Inner Mongolia, China. Clin. Exp. Pharmacol. Physiol. 2007, 34, 838–844. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ao, Y.; Tong, W.; Zhao, Y.; Zhang, H.; Yang, T.; Li, J.; Bao, J.; Wang, J.; Wang, F. Relationship between Tartary buckwheat intake and hypertension and high serum lipids in Mongolian people. Chin. J. Hypertens. 2001, 9, 330–332. [Google Scholar]
- Zhang, H.; Zhang, Y.; Lu, M.; Tong, W.; Ao, Y.; Zhao, Y.; Li, J.; Bao, J. Effects of buckwheat-eating on blood sugars and serum lipids levels and blood pressure. J. Environ. Occup. Med. 2003, 20, 120–122. [Google Scholar]
- Lu, M.; Zhang, H.; Zhang, Y.; Tong, W.; Zhao, Y.; Shan, S.; Liu, H. An epidemiological study on relationship between buckwheat in diet and the prevalence rate of diabetes mellitus as well as blood glucose concentration. Mod. Prev. Med. 2002, 29, 326–327. [Google Scholar]
- Tong, W.; Wang, J.; Bu, R.B.T.; Shi, T.; Chen, W.; Feng, H.; Li, Y.; Qing, G.L.T.; Bao, J.; Zhang, Y.; et al. Epidemiological study on relationship between taking sweet buckwheat and human’s blood-lipid. China Public Health 2002, 18, 1370–1371. [Google Scholar]
- Yu, F. Observation of the hypolipidemic effects of Tartary buckwheat tea. J. Today Health 2015, 4, 283. [Google Scholar]
- Bijlani, R.L.; Gandhi, B.M.; Gupta, M.C.; Manocha, S.; Tandon, B.N. Effect of whole buckwheat (Fagopyrum esculentum) flour supplementation on lipid profile & glucose tolerance. Indian J. Med. Res. 1985, 81, 162–168. [Google Scholar] [PubMed]
- Bijlani, R.L.; Sud, S.; Sahi, A.; Gandhi, B.M.; Tandon, B.N. Effect of sieved buckwheat (Fagopyrum esculentum) flour supplementation on lipid profile and glucose tolerance. Indian J. Physiol. Pharmacol. 1984, 29, 69–74. [Google Scholar]
- Lu, C.; Xu, J.; Zhao, P. Clinical application and therapeutic effect of composite tartary buckwheat flour on hyperglycemia and hyperlipidemia. Food Sci. 1990, 7, 45–46. [Google Scholar]
- Zheng, G.; Pan, X.; An, Z.; Wang, Y. Preliminary observation of lipid-decreasing effects of compound Tartary buckwheat flour on niddm patients. Beijing Med. J. 1991, 5, 280. [Google Scholar]
- Liu, X.; Fu, X. Randomized clinical trial of Tartary buckwheat’s curing effect on elderly hyperlipidemia. Acad. Pap. Complies Chin. Nutr. Soc. Conf. 1996, 7, 184–185. [Google Scholar]
- Lin, R.; Jia, W.; Ren, J. Research and utilization of Tartary buckwheat. In Proceeding from the International Conference Asian Product Development; Corke, H., Lin, R., Eds.; Science Press: Shanxi, Taiyuan, China, 1998; pp. 187–197. [Google Scholar]
- Zhao, J.; Guan, L. Effects of buckwheat flour eating for 8 weeks on blood glucose, serum lipids levels and blood pressure. Chin. J. Clin. Rehabil. 2003, 7, 2231. [Google Scholar]
- Huang, G.; Huang, M.; Chen, W.; Huang, Y.; Yang, Z.; You, Y. Clinical effects of tartary buckwheat mixture on the treatment of early diabetic and nephropathy. J. Chin. Med. Mater. 2009, 32, 1932–1935. [Google Scholar]
- Wieslander, G.; Fabjan, N.; Vogrincic, M.; Kreft, I.; Janson, C.; Spetz-Nystrom, U.; Vombergar, B.; Tagesson, C.; Leanderson, P.; Norback, D. Eating buckwheat cookies is associated with the reduction in serum levels of myeloperoxidase and cholesterol: A double blind crossover study in day-care centre staffs. Tohoku J. Exp. Med. 2011, 225, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Stringer, D.M.; Taylor, C.G.; Appah, P.; Blewett, H.; Zahradka, P. Consumption of buckwheat modulates the post-prandial response of selected gastrointestinal satiety hormones in individuals with type 2 diabetes mellitus. Metabolism 2013, 62, 1021–1031. [Google Scholar] [CrossRef] [PubMed]
- Stokić, E.; Mandic, A.; Sakac, M.; Misan, A.; Pestoric, M.; Simurina, O.; Jambrec, D.; Jovanov, P.; Nedeljkovic, N.; Milovanovic, I.; et al. Quality of buckwheat-enriched wheat bread and its antihyperlipidemic effect in statin treated patients. LWT Food Sci. Technol. 2015, 63, 556–561. [Google Scholar] [CrossRef]
- Dinu, M.; Ghiselli, L.; Whittaker, A.; Pagliai, G.; Cesari, F.; Fiorillo, C.; Becatti, M.; Marcucci, R.; Benedettelli, S.; Sofi, F. Consumption of buckwheat products and cardiovascular risk profile: A randomized, single-blinded crossover trial. Nutr. Metab. Cardiovasc. Dis. 2017, 27, e20–e21. [Google Scholar] [CrossRef]
- He, J.; Klag, M.J.; Whelton, P.K.; Mo, J.P.; Chen, J.Y.; Qian, M.C.; Mo, P.S.; He, G.Q. Oats and buckwheat intakes and cardiovascular-disease risk-factors in an ethnic-minority of china. Am. J. Clin. Nutr. 1995, 61, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Son, B.K.; Kim, J.Y.; Lee, S.S. Effect of adlay, buckwheat and barley on lipid metabolism and aorta histopathology in rats fed an obesogenic diet. Ann. Nutr. Metab. 2008, 52, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Li, Y.M.; Zhang, K.S.; Jiao, R.; Ma, K.Y.; Zhang, R.; Ren, G.X.; Chen, Z.Y. Hypocholesterolemic activity of buckwheat flour is mediated by increasing sterol excretion and down-regulation of intestinal NPC1L1 and ACAT2. J. Funct. Foods 2014, 6, 311–318. [Google Scholar] [CrossRef]
- Prestamo, G.; Pedrazuela, A.; Penas, E.; Lasuncion, M.A.; Arroyo, G. Role of buckwheat diet on rats as prebiotic and healthy food. Nutr. Res. 2003, 23, 803–814. [Google Scholar] [CrossRef]
- Orzel, D.; Zmijewski, M.; Bronkowska, M. Impact of products from ground buckwheat added to balanced diets on biochemical blood markers in wistar rats. Roczniki Państwowego Zakładu Higieny 2015, 66, 239–244. [Google Scholar] [PubMed]
- Tomotake, H.; Yamamoto, N.; Yanaka, N.; Ohinata, H.; Yamazaki, R.; Kayashita, J.; Kato, N. High protein buckwheat flour suppresses hypercholesterolemia in rats and gallstone formation in mice by hypercholesterolemic diet and body fat in rats because of its low protein digestibility. Nutrition 2006, 22, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Magdy, A.-A.M.S.; Sayed, A.E.-K.S.F.; Marwa, M.E.S.A.E.G. Protective effects of wheat bran and buckwheat hull extracts against hypercholesterolemia in male rats. Int. J. Adv. Res. 2014, 2, 724–736. [Google Scholar]
- Wang, M.; Liu, J.R.; Gao, J.M.; Parry, J.W.; Wei, Y.M. Antioxidant activity of tartary buckwheat bran extract and its effect on the lipid profile of hyperlipidemic rats. J. Agric. Food Chem. 2009, 57, 5106–5112. [Google Scholar] [CrossRef] [PubMed]
- Hosaka, T.; Sasaga, S.; Yamasaka, Y.; Nii, Y.; Edazawa, K.; Tsutsumi, R.; Shuto, E.; Okahisa, N.; Iwata, S.; Tomotake, H.; et al. Treatment with buckwheat bran extract prevents the elevation of serum triglyceride levels and fatty liver in kk-a(y) mice. J. Med. Investig. 2014, 61, 345–352. [Google Scholar] [CrossRef][Green Version]
- Yao, Y.; Shan, F.; Bian, J.; Chen, F.; Wang, M.; Ren, G. d-chiro-inositol-enriched tartary buckwheat bran extract lowers the blood glucose level in KK-Ay mice. J. Agric. Food Chem. 2008, 56, 10027–10031. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.Y.; Zhao, Y.; Ren, D.Y.; Guo, J.J.; Luo, Y.Y.; Yang, X.B. Hypoglycemic and hepatoprotective effects of d-chiro-inositol-enriched Tartary buckwheat extract in high fructose-fed mice. Food Funct. 2015, 6, 3760–3769. [Google Scholar] [CrossRef] [PubMed]
- Tomotake, H.; Shimaoka, I.; Kayashita, J.; Yokoyama, F.; Nakajoh, M.; Kato, N. A buckwheat protein product suppresses gallstone formation and plasma cholesterol more strongly than soy protein isolate in hamsters. J. Nutr. 2000, 130, 1670–1674. [Google Scholar] [CrossRef] [PubMed]
- Tomotake, H.; Yamamoto, N.; Kitabayashi, H.; Kwakami, A.; Kayashita, J.; Ohinata, H.; Karasawa, H.; Kato, N. Preparation of tartary buckwheat protein product and its improving effect on cholesterol metabolism in rats and mice fed cholesterol-enriched diet. J. Food Sci. 2007, 72, S528–S533. [Google Scholar] [CrossRef] [PubMed]
- Tomotake, H.; Shimaoka, I.; Kayashita, J.; Yokoyama, F.; Nakajoh, M.; Kato, N. Stronger suppression of plasma cholesterol and enhancement of the fecal excretion of steroids by a buckwheat protein product than by a soy protein isolate in rats fed on a cholesterol-free diet. Biosci. Biotechnol. Biochem. 2001, 65, 1412–1414. [Google Scholar] [CrossRef] [PubMed]
- Kayashita, J.; Shimaoka, I.; Nakajoh, M.; Yamazaki, M.; Kato, N. Consumption of buckwheat protein lowers plasma cholesterol and raises fecal neutral sterols in cholesterol-fed rats because of its low digestibility. J. Nutr. 1997, 127, 1395–1400. [Google Scholar] [CrossRef] [PubMed]
- Kayashita, J.; Shimaoka, I.; Nakajoh, M.; Arachi, Y.; Kato, N. Feeding of buckwheat protein extract reduces body fat content in rats. In Current Advances in Buckwheat Research, Proceedings of the 6th International Symposium on Buckwheat, Shinshu, Japan, 24–29 August 1995; Matano, T., Ujihara, A., Eds.; Shinshu University Press: Shinshu, China, 1995; pp. 935–940. [Google Scholar]
- Kayashita, J.; Shimaoka, I.; Nakajoh, M.; Kato, N. Feeding of buckwheat protein extract reduces hepatic triglyceride concentration, adipose tissue weight, and hepatic lipogenesis in rats. J. Nutr. Biochem. 1996, 7, 555–559. [Google Scholar] [CrossRef]
- Kayashita, J.; Shimaoka, I.; Nakajyoh, M. Hypocholesterolemic effect of buckwheat protein extract in rats fed cholesterol-enriched diets. Nutr. Res. 1995, 15, 691–698. [Google Scholar] [CrossRef]
- Hu, Y.Y.; Zhao, Y.; Yuan, L.; Yang, X.B. Protective effects of tartary buckwheat flavonoids on high tmao diet-induced vascular dysfunction and liver injury in mice. Food Funct. 2015, 6, 3359–3372. [Google Scholar] [CrossRef] [PubMed]
- Han, S.Y.; Lu, H.; Zhu, L.S.; Liu, S.M.; Jia, X.R.; La, W.Y.; Liang, Y.P. Effect of total flavones of buckwheat seed onlowering serum lipid, glucose and anti-lipid peroxidation. Chin. Pharmacol. Bull. 2001, 17, 694–696. [Google Scholar]
- Qu, Y.; Yasuda, T.; Nakajima, K.; Hiwatashi, A.; Moroi, C.; Sanada, H.; Egashira, Y. Effect of rutin in buckwheat noodle on lipid metabolism in rats. Food Sci. Technol. Res. 2013, 19, 1011–1018. [Google Scholar] [CrossRef][Green Version]
- Zhang, C.; Zhang, R.; Li, Y.M.; Liang, N.; Zhao, Y.; Zhu, H.; He, Z.; Liu, J.; Hao, W.; Jiao, R.; et al. Cholesterol-lowering activity of Tartary buckwheat protein. J. Agric. Food Chem. 2017, 65, 1900–1906. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Green, S. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions; Version 5.1.0 [Updated March 2011]; The Cochrane Collaboration. Available online: http://handbook-5-1.cochrane.org/ (accessed on 1 May 2018).
- Goldberg, I.J.; Kako, Y.; Lutz, E.P. Responses to eating: Lipoproteins, lipolytic products and atherosclerosis. Curr. Opin. Lipidol. 2000, 11, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.O.; Peters, J.C. Environmental contributions to the obesity epidemic. Science 1998, 280, 1371–1374. [Google Scholar] [CrossRef] [PubMed]
- Kahn, B.B.; Flier, J.S. Obesity and insulin resistance. J. Clin. Investig. 2000, 106, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Williams, G. Obesity and type 2 diabetes: A conflict of interests? Int. J. Obes. Relat. Metab. Disord. 1999, 23 (Suppl. 7), S2–S4. [Google Scholar] [CrossRef] [PubMed]
- Lewington, S.; Clarke, R.; Qizilbash, N.; Peto, R.; Collins, R.; Collaboration, P.S. Age-specific relevance of usual blood pressure to vascular mortality: A meta-analysis of individual data for one million adults in 61 prospective studies. Lancet 2002, 360, 1903–1913. [Google Scholar] [CrossRef]
- Banach, M.; Aronow, W.S. Hypertension therapy in the older adults-do we know the answers to all the questions? The status after publication of the accf/aha 2011 expert consensus document on hypertension in the elderly. J. Hum. Hypertens. 2012, 26, 641–643. [Google Scholar] [CrossRef] [PubMed]
- Tighe, P.; Duthie, G.; Vaughan, N.; Brittenden, J.; Simpson, W.G.; Duthie, S.; Mutch, W.; Wahle, K.; Horgan, G.; Thies, F. Effect of increased consumption of whole-grain foods on blood pressure and other cardiovascular risk markers in healthy middle-aged persons: A randomized controlled trial. Am. J. Clin. Nutr. 2010, 92, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Seal, C.J.; Brownlee, I.A. Whole-grain foods and chronic disease: Evidence from epidemiological and intervention studies. Proc. Nutr. Soc. 2015, 74, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, M.; Gerstein, H.; Wang, Y.; Yusuf, S. The relationship between glucose and incident cardiovascular events: A metaregression analysis of published data from 20 studies of 95,783 individuals followed for 12.4 years. Diabetes Care 1999, 22, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Uwaifo, G.I.; Ratner, R.E. The roles of insulin resistance, hyperinsulinemia, and thiazolidinediones in cardiovascular disease. Am. J. Med. 2003, 115, 12–19. [Google Scholar] [CrossRef]
- Hallfrisch, J.; Behall, K.M. Mechanisms of the effects of grains on insulin and glucose responses. J. Am. Coll. Nutr. 2000, 19, 320s–325s. [Google Scholar] [CrossRef] [PubMed]
- Liese, A.D.; Roach, A.K.; Sparks, K.C.; Marquart, L.; D’Agostino, R.B.; Mayer-Davis, E.J. Whole-grain intake and insulin sensitivity: The insulin resistance atherosclerosis study. Am. J. Clin. Nutr. 2003, 78, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Steffen, L.M.; Jacobs, D.R.; Murtaugh, M.A.; Moran, A.; Steinberger, J.; Hong, C.P.; Sinaiko, A.R. Whole grain intake is associated with lower body mass and greater insulin sensitivity among adolescents. Am. J. Epidemiol. 2003, 158, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Sloth, B.; Krog-Mikkelsen, I.; Flint, A.; Tetens, I.; Bjorck, I.; Vinoy, S.; Elmstahl, H.; Astrup, A.; Lang, V.; Raben, A. No difference in body weight decrease between a low-glycemic-index and a high-glycemic-index diet but reduced LDL cholesterol after 10-wk ad libitum intake of the low-glycemic-index diet. Am. J. Clin. Nutr. 2004, 80, 337–347. [Google Scholar] [CrossRef] [PubMed]
- De Rougemont, A.; Normand, S.; Nazare, J.A.; Skilton, M.R.; Sothier, M.; Vinoy, S.; Laville, M. Beneficial effects of a 5-week low-glycaemic index regimen on weight control and cardiovascular risk factors in overweight non-diabetic subjects. Br. J. Nutr. 2007, 98, 1288–1298. [Google Scholar] [CrossRef] [PubMed]
- Tovar, J.; Nilsson, A.; Johansson, M.; Bjorck, I. Combining functional features of whole-grain barley and legumes for dietary reduction of cardiometabolic risk: A randomised cross- over intervention in mature women. Br. J. Nutr. 2014, 111, 706–714. [Google Scholar] [CrossRef] [PubMed]
- Fonteles, M.C.; Almeida, M.Q.; Larner, J. Antihyperglycemic effects of 3-O-methyl-d-chiro-inositol and d-chiro-inositol associated with manganese in streptozotocin diabetic rats. Horm. Metab. Res. 2000, 32, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Johnston, K.; Sharp, P.; Clifford, M.; Morgan, L. Dietary polyphenols decrease glucose uptake by human intestinal caco-2 cells. FEBS Lett. 2005, 579, 1653–1657. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-L.; Zhou, M.-L.; Tang, Y.; Li, F.-L.; Tang, Y.-X.; Shao, J.-R.; Xue, W.-T.; Wu, Y.-M. Bioactive compounds in functional buckwheat food. Food Res. Int. 2012, 49, 389–395. [Google Scholar] [CrossRef]
- Kwon, O.; Eck, P.; Chen, S.L.; Corpe, C.P.; Lee, J.H.; Kruhlak, M.; Levine, M. Inhibition of the intestinal glucose transporter glut2 by flavonoids. FASEB J. 2007, 21, 366–377. [Google Scholar] [CrossRef] [PubMed]
- Skrabanja, V.; Elmstahl, H.G.M.L.; Kreft, I.; Bjorck, I.M.E. Nutritional properties of starch in buckwheat products: Studies in vitro and in vivo. J. Agric. Food Chem. 2001, 49, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, S.; Hawken, S.; Ounpuu, S.; Dans, T.; Avezum, A.; Lanas, F.; McQueen, M.; Budaj, A.; Pais, P.; Varigos, J.; et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the interheart study): Case-control study. Lancet 2004, 364, 937–952. [Google Scholar] [CrossRef]
- Lloyd-Jones, D.; Adams, R.J.; Brown, T.M.; Carnethon, M.; Dai, S.; De Simone, G.; Ferguson, T.B.; Ford, E.; Furie, K.; Gillespie, C.; et al. Heart disease and stroke statistics-2010 update a report from the american heart association. Circulation 2010, 121, E46–E215. [Google Scholar] [PubMed]
- Cholesterol Treatment Trialists Collaboration. Efficacy and safety of more intensive lowering of LDL cholesterol: A meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet 2010, 376, 1670–1681. [Google Scholar]
- Ripsin, C.M.; Keenan, J.M.; Jacobs, D.R., Jr.; Elmer, P.J.; Welch, R.R.; Horn, L.V.; Liu, K.; Turnbull, W.H.; Thye, F.W.; Kestin, M.; et al. Oat products and lipid lowering: A meta-analysis. JAMA 1992, 267, 3317–3325. [Google Scholar] [CrossRef] [PubMed]
- Christa, K.; Soral-Smietana, M. Buckwheat grains and buckwheat products—Nutritional and prophylactic value of their components—A review. Czech J. Food Sci. 2008, 26, 153–162. [Google Scholar] [CrossRef]
- Dziedzic, K.; Górecka, D.; Kobus-Cisowska, J.; Jeszka, M. Opportunities of using buckwheat in production of functional food. Nauka Przyroda Technologie 2010, 4, 2–6. [Google Scholar]
- Judd, P.A.; Truswell, A.S. The effect of rolled oats on blood-lipids and fecal steroid-excretion in man. Am. J. Clin. Nutr. 1981, 34, 2061–2067. [Google Scholar] [CrossRef] [PubMed]
- Gordon, T.; Castelli, W.P.; Hjortland, M.C.; Kannel, W.B.; Dawber, T.R. High-density lipoprotein as a protective factor against coronary heart-disease—Framingham study. Am. J. Med. 1977, 62, 707–714. [Google Scholar] [CrossRef]
- Shinnick, F.L.; Ink, S.L.; Marlett, J.A. Dose response to a dietary oat bran fraction in cholesterol-fed rats. J. Nutr. 1990, 120, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Story, J.A. Dietary fiber and lipid-metabolism. Proc. Soc. Exp. Biol. Med. 1985, 180, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.W.; Siesel, A.E. Hypocholesterolemic effects of oat products. Adv. Exp. Med. Biol. 1990, 270, 17–36. [Google Scholar] [PubMed]
- Slavin, J.L.; Martini, M.C.; Jacobs, D.R.; Marquart, L. Plausible mechanisms for the protectiveness of whole grains. Am. J. Clin. Nutr. 1999, 70, 459s–463s. [Google Scholar] [CrossRef] [PubMed]
- Escudero, N.L.; Zirulnik, F.; Gomez, N.N.; Mucciarelli, S.I.; Gimenez, M.S. Influence of a protein concentrate from Amaranthus cruentus seeds on lipid metabolism. Exp. Biol. Med. 2006, 231, 50–59. [Google Scholar] [CrossRef]
- Liu, S.; Manson, J.; Stampfer, M.; Holmes, M.; Hu, F.; Hankinson, S.; Willett, W. Dietary glycemic load assessed by food-frequency questionnaire in relation to plasma high-density-lipoprotein cholesterol and fasting plasma triacylglycerols in postmenopausal women. Am. J. Clin. Nutr. 2001, 73, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Carroll, K.K.; Hamilton, R.M. Effects of dietary-protein and carbohydrate on plasma cholesterol levels in relation to atherosclerosis. J. Food Sci. 1975, 40, 18–23. [Google Scholar] [CrossRef]
- Carroll, K.K. Hypercholesterolemia and atherosclerosis—Effects of dietary-protein. Fed. Proc. 1982, 41, 2792–2796. [Google Scholar] [PubMed]
- Terpstra, A.H.; Hermus, R.J.; West, C.E. The role of dietary protein in cholesterol metabolism. World Rev. Nutr. Dietetics 1983, 42, 1–55. [Google Scholar]
- Pomeranz, Y. Buckwheat—Structure, composition, and utilization. Crit. Rev. Food Sci. 1983, 19, 213–258. [Google Scholar] [CrossRef] [PubMed]
- Pomeranz, Y.; Robbins, G.S. Amino-acid composition of buckwheat. J. Agric. Food Chem. 1972, 20, 270–274. [Google Scholar] [CrossRef]
- Ikeda, K.; Kishida, M. Digestibility of proteins in buckwheat seed. Fagopyrum 1993, 13, 21–24. [Google Scholar]
- Eastwood, M.A. The physiological effect of dietary fiber—An update. Annu. Rev. Nutr. 1992, 12, 19–35. [Google Scholar] [CrossRef] [PubMed]
- Kritchevsky, D. Dietary fiber. Annu. Rev. Nutr. 1988, 8, 301–328. [Google Scholar] [CrossRef] [PubMed]
- Yiming, Z.; Hong, W.; Linlin, C.; Xiaoli, Z.; Wen, T.; Xinli, S. Evolution of nutrient ingredients in tartary buckwheat seeds during germination. Food Chem. 2015, 186, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Kararli, T.T. Comparison of the gastrointestinal anatomy, physiology, and biochemistry of humans and commonly used laboratory-animals. Biopharm. Drug Dispos. 1995, 16, 351–380. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Xiong, Y.L. Antioxidant and bile acid binding activity of buckwheat protein in vitro digests. J. Agric. Food Chem. 2009, 57, 4372–4380. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.L.; Huang, L.; Tang, W.; Zhou, Y.M.; Wang, Q.; Li, Z.J. A novel buckwheat protein with a beneficial effect in atherosclerosis was purified from Fagopyrum tataricum (L.) gaertn. Arch. Biol. Sci. 2013, 65, 767–772. [Google Scholar] [CrossRef]
- Holasova, M.; Fiedlerova, V.; Smrcinova, H.; Orsak, M.; Lachman, J.; Vavreinova, S. Buckwheat—The source of antioxidant activity in functional foods. Food Res. Int. 2002, 35, 207–211. [Google Scholar] [CrossRef]
- Fabjan, N.; Rode, J.; Kosir, I.J.; Wang, Z.; Zhang, Z.; Kreft, I. Tartary buckwheat (Fagopyrum tataricum gaertn.) as a source of dietary rutin and quercitrin. J. Agric. Food Chem. 2003, 51, 6452–6455. [Google Scholar] [CrossRef] [PubMed]
- Vogrincic, M.; Timoracka, M.; Melichacova, S.; Vollmannova, A.; Kreft, I. Degradation of rutin and polyphenols during the preparation of tartary buckwheat bread. J. Agric. Food Chem. 2010, 58, 4883–4887. [Google Scholar] [CrossRef] [PubMed]
- KuwABARA, T.; Han, K.H.; Hashimoto, N.; Yamauchi, H.; Shimada, K.I.; Sekikawa, M.; Fukushima, M. Tartary buckwheat sprout powder lowers plasma cholesterol level in rats. J. Nutr. Sci. Vitaminol. 2007, 53, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Bok, S.H.; Jeon, S.M.; Park, Y.B.; Lee, S.J.; Jeong, T.S.; Choi, M.S. Effect of rutin and tannic acid supplements on cholesterol metabolism in rats. Nutr. Res. 2002, 22, 283–295. [Google Scholar] [CrossRef]
- Panchal, S.K.; Poudyal, H.; Arumugam, T.V.; Brown, L. Rutin attenuates metabolic changes, nonalcoholic steatohepatitis, and cardiovascular remodeling in high-carbohydrate, high-fat diet-fed rats. J. Nutr. 2011, 141, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- Choi, I.; Park, Y.; Choi, H.; Lee, E.H. Anti-adipogenic activity of rutin in 3T3-L1 cells and mice fed with high-fat diet. Biofactors 2006, 26, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Kitabayashi, H.; Ujihara, A.; Hirose, T.; Minami, M. Varietal differences and heritability for rutin content in common buckwheat, Fagopyrum esculentum moench. Breed. Sci. 1995, 45, 75–79. [Google Scholar] [CrossRef]
- Igarashi, K.; Ohmuma, M. Effects of isorhamnetin, rhamnetin, and quercetin on the concentrations of cholesterol and lipoperoxide in the serum and liver and on the blood and liver antioxidative enzyme-activities of rats. Biosci. Biotechnol. Biochem. 1995, 59, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Juzwiak, S.; Wojcicki, J.; Mokrzycki, K.; Marchlewicz, M.; Bialecka, M.; Wenda-Rozewicka, L.; Gawronska-Szklarz, B.; Drozdzik, M. Effect of quercetin on experimental hyperlipidemia and atherosclerosis in rabbits. Pharmacol. Rep. 2005, 57, 604–609. [Google Scholar] [PubMed]
- Odbayar, T.O.; Badamhand, D.; Kimura, T.; Takahashi, Y.; Tsushida, T.; Ide, T. Comparative studies of some phenolic compounds (quercetin, rutin, and ferulic acid) affecting hepatic fatty acid synthesis in mice. J. Agric. Food Chem. 2006, 54, 8261–8265. [Google Scholar] [CrossRef] [PubMed]
- Yugarani, T.; Tan, B.K.; Teh, M.; Das, N.P. Effects of polyphenolic natural products on the lipid profiles of rats fed high fat diets. Lipids 1992, 27, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Hayek, T.; Fuhrman, B.; Vaya, J.; Rosenblat, M.; Belinky, P.; Coleman, R.; Elis, A.; Aviram, M. Reduced progression of atherosclerosis in apolipoprotein E-deficient mice following consumption of red wine, or its polyphenols quercetin or catechin, is associated with reduced susceptibility of LDL to oxidation and aggregation. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 2744–2752. [Google Scholar] [CrossRef] [PubMed]
- Lauridsen, S.T.; Mortensen, A. Probucol selectively increases oxidation of atherogenic lipoproteins in cholesterol-fed mice and in Watanabe heritable hyperlipidemic rabbits. Atherosclerosis 1999, 142, 169–178. [Google Scholar] [CrossRef]
- Enkhmaa, B.; Shiwaku, K.; Katsube, T.; Kitajima, K.; Anuurad, E.; Yamasaki, M.; Yamane, Y. Mulberry (Morus alba L.) leaves and their major flavonol quercetin 3-(6-malonylglucoside) attenuate atherosclerotic lesion development in ldl receptor-deficient mice. J. Nutr. 2005, 729–734. [Google Scholar]
- Glasser, G.; Graefe, E.U.; Struck, F.; Veit, M.; Gebhardt, R. Comparison of antioxidative capacities and inhibitory effects on cholesterol biosynthesis of quercetin and potential metabolites. Phytomedicine 2002, 9, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Pincemail, J.; Deby, C.; Thirion, A.; de Bruyn-Dister, M.; Goutier, R. Human myeloperoxidase activity is inhibited in vitro by quercetin. Comparison with three related compounds. Experientia 1988, 44, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, S.J.; Hazen, S.L. Myeloperoxidase, modified lipoproteins, and atherogenesis. J. Lipid Res. 2009, 50, S346–S351. [Google Scholar] [CrossRef] [PubMed]

| Human Studies | ||||||
|---|---|---|---|---|---|---|
| Source | Study Population | Foodstuff; Intake | Duration | Outcomes 95% CI | ||
| Significant | Insignificant | |||||
| Bijlani et al. (1985) [24] | healthy (n = 8♂) | 100 g of whole BW flour | 12 weeks | serum: VLDL | ↓ | body weight |
| serum: TC | ||||||
| serum: LDL | ||||||
| serum: HDL | ||||||
| serum: HDL/TC | ||||||
| serum: LDLTG | ||||||
| serum: VLDLTG | ||||||
| serum: HDLTG | ||||||
| serum: TG | ||||||
| healthy (n = 9♂) | 100 g of whole BW flour | 4 weeks | serum: HDL/TC | ↑ | body weight | |
| serum: LDLTG | ↑ | fasting blood glucose | ||||
| serum: VLDLTG | ↑ | glucose | ||||
| serum: HDLTG | ↓ | serum: TC | ||||
| serum: LDL | ||||||
| serum: VLDL | ||||||
| serum: HDL | ||||||
| serum: TG | ||||||
| Bijlani et al. (1984) [25] | healthy (n = 12♂) | 100 g of sieved BW preparation | 4 weeks | serum: HDL | ↑ | blood glucose |
| serum: HDL/TC | ↑ | serum: TC | ||||
| serum: LDL | ||||||
| serum: VLDL | ||||||
| Lu et al. (1990) [26] | patients with diabetes and hyperlipidemia (n = 23, 13 and 18) | BW flour | 1 month | fasting blood sugar | ↓ | |
| BW flour | 1 month | serum: TC | ↓ | |||
| BW flour | 1 month | serum: TG | ↓ | |||
| Zheng et al. (1991) [27] | NIDDM patients (n = 10♂, 9♀) | Tartary BW flour; 50 g | 3 months | serum: TG | ↓ | Blood glucose |
| insulin | ||||||
| serum: TC | ||||||
| Liu and Fu (1996) [28] | patients (n = 60) | Tartary BW flour; 40 g/day | 4 weeks | body weight | ↓ | |
| systolic BP | ↓ | |||||
| diastolic BP | ↓ | |||||
| serum: TC | ↓ | |||||
| serum: LDL | ↓ | |||||
| serum: HDL | ↑ | |||||
| serum: TG | ↓ | |||||
| Lin et al. (1998) [29] | Type 2 diabetes (T2DM) (n = 32) | 100 g of Tartary BW flour | 5 weeks | fasting blood | ↓ | serum: TC |
| glucose | ||||||
| serum: TG | ↓ | |||||
| Zhao and Guan (2003) [30] | T2DM (n = 30♂, 30♀) | BW flour | 8 weeks | blood glucose | ↓ | body weight |
| systolic BP | ↓ | diastolic BP | ||||
| serum: TC | ↓ | serum: LDL | ||||
| serum: HDL | ↓ | serum: TG | ||||
| Huang et al. (2009) [31] | patients with diabtes (n = 18♂, 17♀) | Tartary BW mixture | 2 months | blood glucose | ↓ | |
| HbA1 c/% | ↓ | |||||
| serum: TC | ↓ | |||||
| serum: LDL | ↓ | |||||
| serum: HDL | ↓ | |||||
| serum: TG | ↓ | |||||
| Wieslander et al. (2011) [32] | healthy (n = 62♀) | group 1: four common BW cookies (daily). group 2: four Tartary BW cookies (daily). (after 2 weeks wash-out, the groups switch type of cookies) 100 g of sieved BW preparation | 6 weeks | serum: TC | ↓ | sPLA2 |
| serum: HDL | ↓ | |||||
| Stringer et al. (2013) [33] | healthy (n = 23) | BW cracker; 76 g | 7 days | plasma glucose | ||
| plasma: TC | ||||||
| plasma: LDL | ||||||
| plasma: HDL | ||||||
| plasma: TG | ||||||
| liver enzyme AST | ||||||
| liver enzyme ALT | ||||||
| T2DM (n = 24) | BW cracker; 76 g | 7 days | plasma glucose | |||
| plasma: TC | ||||||
| plasma: LDL | ||||||
| plasma: HDL | ||||||
| plasma: TG | ||||||
| liver enzyme AST | ||||||
| liver enzyme ALT | ||||||
| Stokić et al. (2015) [34] | patients (n = 7♂, 13♀) | BW-enriched wheat bread; 300 g/day | 1 month | serum: TC | ↓ | BMI |
| serum: LDL | ↓ | systolic BP | ||||
| liver enzyme AST | ↑ | diastolic BP | ||||
| liver enzyme ALT | ↓ | serum: HDL | ||||
| serum: TG | ||||||
| Yu (2015) [23] | patients with hyperlipidemia (n = 36♂, 24♀) | Tartary BW tea, 15 g | 60 days | serum: TC | ↓ | systolic BP |
| serum: LDL | ↓ | diastolic BP | ||||
| serum: TG | ↓ | blood glucose | ||||
| serum: HDL | ||||||
| Dinu et al. (2017) [35] | participants with high CVD risk (n = 10♂, 11♀) | group 1: BW products (daily); group 2: control products (daily) (after 8 weeks wash-out, the groups switch type of products) | 24 weeks | fasting blood glucose | ↓ | body weight |
| glucose | insulin | |||||
| serum: TC | ↓ | serum: HDL | ||||
| serum: LDL | ↓ | |||||
| serum: TG | ↓ | |||||
| He et al. (1995) [36] | healthy (n = 857♂) | BW; group 1 (n = 319), 0 g/day group 2 (n = 207), <40 g/day group 3 (n = 161), 40–200 g/day group 4 (n = 163), >200 g/day | cross-sectional study | systolic BP | ↓ | BMI |
| diastolic BP | ↓ | serum: HDL | ||||
| serum: TC | ↑ | serum: TG | ||||
| serum: LDL | ↓ | |||||
| serum: HDL/TC | ↓ | |||||
| Zhang et al. (2007) [18] | healthy (n = 491♂, 470♀) | BW; not stated | cross-sectional study | BMI | ↑ | systolic BP |
| blood glucose | ↓ | diastolic BP | ||||
| serum: TC | ↓ | serum: TG | ||||
| serum: LDL | ↓ | |||||
| serum: HDL | ↑ | |||||
| Animal Studies | ||||||
|---|---|---|---|---|---|---|
| Source | Model | Assay Product; Dose | Duration | Outcomes 95% CI | ||
| Significant | Insignificant | |||||
| Son et al. (2008) [37] | ♂Sprague–Dawley rats | BW powder; 50% in the diets (diet with 1% cholesterol) | 4 weeks | plasma: TC | ↓ | food intake |
| plasma: LDL | ↓ | body weight gain | ||||
| plasma: HDL | ↑ | food efficiency ratio | ||||
| plasma: TG | ↓ | transit time | ||||
| area of lumen | ↑ | wall thickness | ||||
| Yang et al. (2014) [38] | ♂Syrian Golden hamster | Tartary BW flour; 24% in diet (fed cholesterol diet) | 6 weeks | serum: TC | ↓ | food intake |
| serum: non-HDL | ↓ | body weight gain | ||||
| liver cholesterol | ↓ | serum: HDL | ||||
| feces: neutral sterols | ↑ | serum: TG | ||||
| feces: acidic sterols | ||||||
| Prestamo (1985) et al. [39] | ♀Wistar Hannover rats | cooked BW | 30 days | body weight | ↓ | blood glucose |
| serum: TC | ↓ | serum: LDL | ||||
| serum: HDL | ↓ | serum: TG | ||||
| HDL phospholipids | ↓ | liver weight | ||||
| uric acids | ||||||
| Orzel et al. (2015) [40] | ♂Wistar rats | buckwheat flour, meal and bran; 200 g/kg (normal diet) | 4 weeks | body weight gain | ↑ | food intake |
| serum: LDL | ↓ | glucose | ||||
| serum: TG | ↓ | hemoglobin | ||||
| serum: TC | ||||||
| serum: HDL | ||||||
| Tomotake et al. (1985) [41] | ♂Sprague–Dawley rats and ♂ddY mice | 30.7% of BWP extract in the diet (rats fed a normal or high-cholesterol diet); 54.8% of PBF (mice fed a high-cholesterol diet) | 10 or 27 days | serum: TC | ↓ | food intake |
| serum: TG | ↓ | body weight gain | ||||
| serum: phospholipids | ↓ | |||||
| liver weight | ↓ | |||||
| liver cholesterol (PBF) | ↓ | |||||
| feces: dry weight (PBF) | ↑ | |||||
| feces: neutral steroids | ↑ | |||||
| feces: bile acids (PBF) | ↑ | |||||
| Magdy et al. (2014) [42] | ♂albino rats | BW hull extracts; 1000 mg/kg b. wt/day in diet (hypercholesterolemia-induced diet) | 8 weeks | blood glucose | ↓ | plasma: HDL |
| plasma: TC | ↓ | |||||
| plasma: LDL | ↓ | |||||
| plasma: TG | ↓ | |||||
| plasma: AST | ↓ | |||||
| plasma: ALT | ↓ | |||||
| Wang et al. (2009) [43] | ♂pathogen-free Wistar rat | Tartary BW bran extract; 0.2–1 g/kg body weight (high-fat diet) | 6 weeks | serum: TC | ↓ | body weight gain |
| serum: HDL (low dose) | ↑ | serum: LDL | ||||
| serum: TG | ↓ | |||||
| hepatic: TC | ↓ | |||||
| hepatic: TG | ↓ | |||||
| Hosaka et al. (2014) [44] | KK-Ay mice | common BW bran powder; 0.05 mg/g body weight | 6 weeks | body weight gain | ↓ | food intake |
| serum: TG | ↓ | fasting blood glucose | ||||
| liver weight | ↓ | insulin resistance | ||||
| serum: TC | ||||||
| Yao et al. (2008) [45] | ♂C57BL/6 control mice and diabetic KK-Ay mice | d-Chiro-Inositol (DCI) enriched Tartary BW bran extract (TBBE); 45–182 mg of TBBE/kg in diet | 5 weeks | fasting blood glucose level | ↓ | body weight gain |
| plasma: TG (high dose) | ↓ | plasma: TC | ||||
| Insulin immunoreactivity | ↑ | |||||
| immunoreactivity | ||||||
| Hu et al. (2015) [46] | ♂Kunming mice | d-Chiro-Inositol (DCI) enriched Tartary BW extract (DTBE); 40, 80 and 160 mg per kg body weight/day (high-fructose water) | 8 weeks | body weight gain | ↓ | all parameters in the group of 40 mg per kg body weight/day showed on significant effect except serum AST activity |
| serum: glucose | ↓ | |||||
| serum: insulin level | ↓ | |||||
| serum: TC | ↓ | |||||
| serum: LDL | ↓ | |||||
| serum: HDL | ↑ | |||||
| serum: TG | ↓ | |||||
| liver weight | ↓ | |||||
| serum AST activity | ↓ | |||||
| serum ALT activity | ↓ | |||||
| Tomotake et al. (2000) [47] | ♂Golden Syrian hamster | BWP extract; 381 g/kg (high-cholesterol diet) | 2 weeks | food intake | ↑ | body weight gain |
| plasma: TC | ↓ | hepatic TG | ||||
| plasma: HDL | ↓ | hepatic phospholipids | ||||
| plasma: HDL/TC | ↑ | |||||
| plasma: TG | ↓ | |||||
| plasma: phospholipids | ↓ | |||||
| liver weight | ↑ | |||||
| hepatic cholesterol | ↓ | |||||
| fecal dry weight | ↑ | |||||
| feces: neutral steroids | ↑ | |||||
| feces: acidic steroids | ↑ | |||||
| Tomotake et al. (2007) [48] | ♂Sprague–Dawley rats | Tartary BW flour protein and common BWP extract; 30.7% of common BWP and 43.7% of Tartary BWP in the diet (high-cholesterol diet) | 27 days | serum: TC | ↓ | body weight gain |
| liver weight | ↓ | food intake | ||||
| hepatic cholesterol | ↓ | |||||
| fecal dry weight | ↑ | |||||
| fecal excretion: nitrogen | ↑ | |||||
| feces: neutral steroids | ↑ | |||||
| feces: bile acids | ↑ | |||||
| protein digestibility | ↓ | |||||
| Tomotake et al. (2001) [49] | ♂Sprague–Dawley rats | BWP extract; 307 g/kg (normal diet) | 8 weeks | plasma: TC | ↓ | body weight gain |
| plasma: HDL | ↓ | food intake | ||||
| feces: neutral steroids | ↑ | plasma: TG | ||||
| feces: acidic steroids | ↑ | plasma: phospholipid | ||||
| feces dry weight | ||||||
| Kayashita et al. (1997) [50] | ♂Sprague–Dawley rats | BWP extract; 381 g/kg (high-Cholesterol diet) | 3 weeks | plasma: TC | ↓ | body weight gain |
| plasma: HDL/TC | ↑ | food intake | ||||
| plasma: TG | ↓ | plasma: HDL | ||||
| plasma: phospholipids | ↑ | hepatic: TG | ||||
| plasma: bile acids | ↓ | feces: acidic steroids | ||||
| liver weight | ↓ | |||||
| hepatic cholesterol | ↓ | |||||
| hepatic: phospholipids | ↓ | |||||
| feces dry weight | ↑ | |||||
| feces: neutral steroids | ↑ | |||||
| protein digestibility | ↓ | |||||
| Kayashita et al. [51] | ♂Sprague–Dawley rats | BWP extract; 38.1% | 3 weeks | plasma: TC | ↓ | body weight gain |
| plasma: HDL/TC | ↑ | food intake | ||||
| plasma: TG | ↓ | plasma: HDL | ||||
| plasma: free fatty acid | ↓ | hepatic cholesterol | ||||
| plasma: phospholipids | ↓ | hepatic TG | ||||
| liver weight | ↓ | hepatic phospholipids | ||||
| fat pad weights | ↓ | |||||
| Kayashita et al. [52] | ♂Sprague–Dawley rats | BWP extract; 381 g/kg | 3 weeks | plasma: TC | ↓ | body weight gain |
| hepatic TG | ↓ | food intake | ||||
| fecal dry weight | ↑ | insulin | ||||
| fat pad weights | ↓ | plasma: TG | ||||
| plasma: free fatty acid | ||||||
| plasma: phospholipids | ||||||
| liver weight | ||||||
| hepatic TC | ||||||
| hepatic phospholipids | ||||||
| Kayashita et al. [53] | ♂Sprague–Dawley rats | BWP extract; 323.1 g/kg (high-Cholesterol diet) | 3 weeks | plasma: TC | ↓ | body weight gain |
| hepatic: weight | ↓ | food intake | ||||
| hepatic TC | ↓ | serum: TG | ||||
| hepatic TG | ↑ | serum: free fatty acids | ||||
| serum: glucose | ||||||
| Hu et al. [54] | ♂Kunming mice | Tartary buckwheat flavonoid fraction; 200, 400 and 800 mg per kg bw in diet (high trimethylamine-N-oxide diet) | 8 weeks | body weight gain | ↓ | food intake |
| serum: TC | ↓ | water intake | ||||
| serum: LDL | ↓ | |||||
| serum: HDL | ↑ | |||||
| serum: TG | ↓ | |||||
| liver weight | ↓ | |||||
| hepatosomatic index | ↓ | |||||
| Han et al. [55] | Wister mice | total flavones of buckwheat seeds; 2 g/kg/day (high-fat diet) | 10 days | serum: TC | ↓ | fasting blood glucose |
| serum: TG | ↓ | |||||
| Qu et al. [56] | ♂Sprague–Dawley rats | high rutin in BW noodles; 980 mg/kg in diet (high-fat, high-sucrose diet) | 4 weeks | serum: TC | ↓ | body weight gain |
| liver lipid | ↑ | feed efficiency | ||||
| serum: HDL | ||||||
| serum: TG | ||||||
| serum: free fatty acids | ||||||
| liver TC | ||||||
| dry weight of feces | ||||||
| fecal total lipid | ||||||
| Zhang et al. [57] | ♂Golden Syrian Hypercholesterolemia hamster | Tartary BWP extract; 353 g/kg in diet | 6 weeks | plasma: TC | ↓ | body weight |
| plasma: non-HDL | ↓ | fatty streak (%) | ||||
| plasma: HDL | ↓ | |||||
| plasma: TG | ↓ | |||||
| liver cholesterol | ↓ | |||||
| total neutral sterols | ↑ | |||||
| acidic sterols | ↑ | |||||
| Number of Studies | |||
|---|---|---|---|
| Significantly higher in Buckwheat Treatment | No Effect | Significantly Lower in Buckwheat Treatment | |
| Human Studies | |||
| Body weight gain or BMI | 1 | 5 | 1 |
| Blood pressure | — | 3 | 3 |
| Blood glucose | — | 5 | 6 |
| Blood insulin | — | 2 | — |
| Total-Cholesterol | — | 5 | 10 |
| LDL-Cholesterol | — | 4 | 7 |
| HDL-Cholesterol | 3 | 6 | 3 |
| Triglycerides | — | 6 | 7 |
| Animal Studies | |||
| Body weight gain | 1 | 14 | 4 |
| Food intake | 1 | 12 | — |
| Blood glucose | — | 4 | 3 |
| Blood insulin | 1 | 2 | 1 |
| Total-Cholesterol | — | 2 | 19 |
| LDL-Cholesterol | — | 2 | 5 |
| HDL-Cholesterol | 4 | 6 | 4 |
| Triglycerides | — | 6 | 14 |
| Liver weight | 1 | 2 | 8 |
| Liver Total-Cholesterol | — | 3 | 8 |
| Fecal weight | 5 | 2 | — |
| Fecal neutral steroids | 6 | — | — |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.; Lietz, G.; Seal, C. Buckwheat and CVD Risk Markers: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 619. https://doi.org/10.3390/nu10050619
Li L, Lietz G, Seal C. Buckwheat and CVD Risk Markers: A Systematic Review and Meta-Analysis. Nutrients. 2018; 10(5):619. https://doi.org/10.3390/nu10050619
Chicago/Turabian StyleLi, Liangkui, Georg Lietz, and Chris Seal. 2018. "Buckwheat and CVD Risk Markers: A Systematic Review and Meta-Analysis" Nutrients 10, no. 5: 619. https://doi.org/10.3390/nu10050619
APA StyleLi, L., Lietz, G., & Seal, C. (2018). Buckwheat and CVD Risk Markers: A Systematic Review and Meta-Analysis. Nutrients, 10(5), 619. https://doi.org/10.3390/nu10050619





