Growth Hormone Secretion Patterns in German Landrace (DL) Fetuses and Piglets Compared to DL Piglets with Inherited 1,25-Dihydroxyvitamin D3 Deficiency
Abstract
1. Introduction
2. Materials and Methods
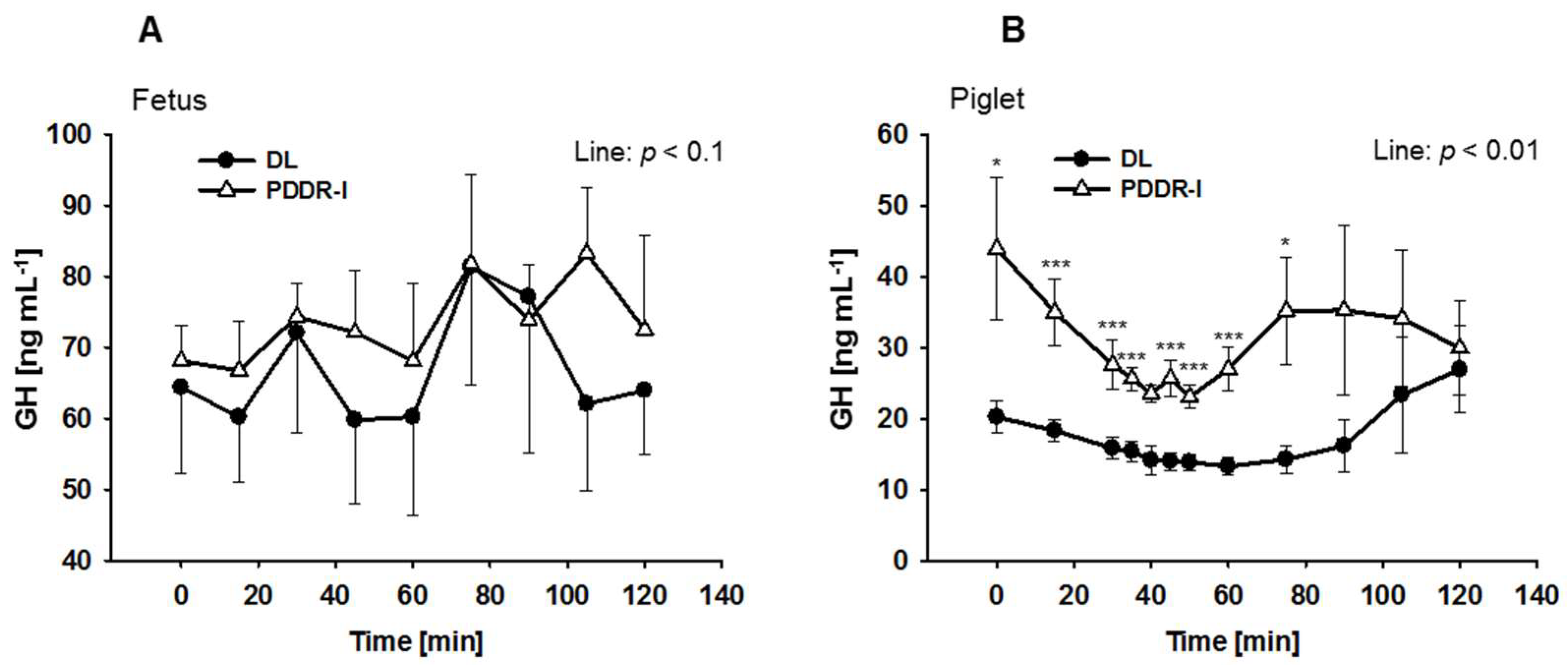
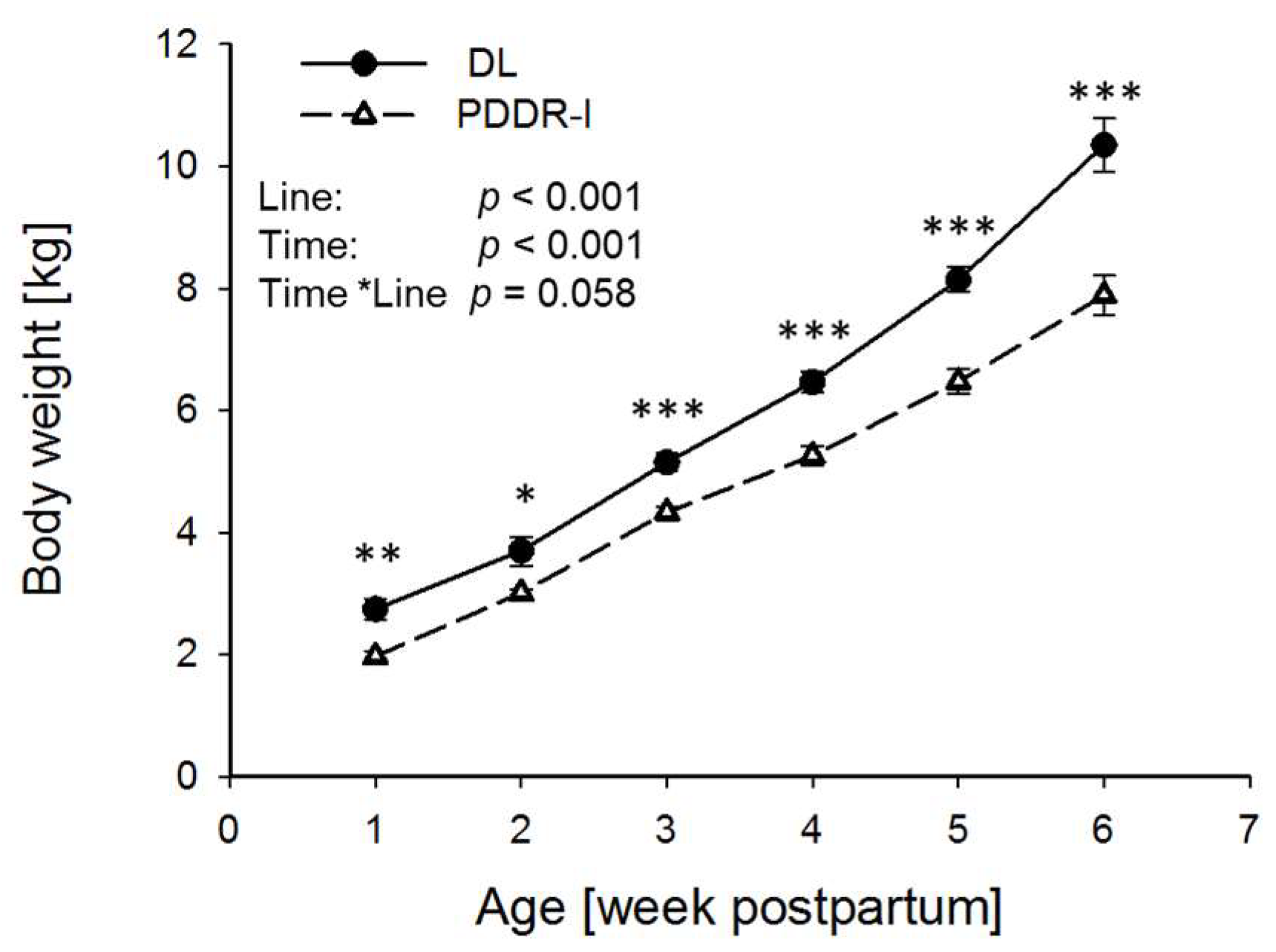
3. Results
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Holick, M.F. High prevalence of vitamin D inadequacy and implications for health. Mayo Clin. Proc. 2006, 81, 353–373. [Google Scholar] [CrossRef] [PubMed]
- Sowah, D.; Fan, X.; Dennett, L.; Hagtvedt, R.; Straube, S. Vitamin D levels and deficiency with different occupations: A systematic review. BMC Public Health 2017, 17, 519. [Google Scholar] [CrossRef] [PubMed]
- Leffelaar, E.R.; Vrijkotte, T.G.; van Eijsden, M. Maternal early pregnancy vitamin D status in relation to fetal and neonatal growth: Results of the multi-ethnic Amsterdam born children and their development cohort. Br. J. Nutr. 2010, 104, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhu, B.; Wu, X.; Li, S.; Tao, F. Association between maternal vitamin D deficiency and small for gestational age: Evidence from a meta-analysis of prospective cohort studies. BMJ Open 2017, 7, e016404. [Google Scholar] [CrossRef] [PubMed]
- Aghajafari, F.; Nagulesapillai, T.; Ronksley, P.E.; Tough, S.C.; O’Beirne, M.; Rabi, D.M. Association between maternal serum 25-hydroxyvitamin D level and pregnancy and neonatal outcomes: Systematic review and meta-analysis of observational studies. BMJ 2013, 346, f1169. [Google Scholar] [CrossRef] [PubMed]
- Ameri, P.; Giusti, A.; Boschetti, M.; Bovio, M.; Teti, C.; Leoncini, G.; Ferone, D.; Murialdo, G.; Minuto, F. Vitamin D increases circulating IGF1 in adults: Potential implication for the treatment of GH deficiency. Eur. J. Endocrinol. 2013, 169, 767–772. [Google Scholar] [CrossRef] [PubMed]
- Waters, M.J.; Kaye, P.L. The role of growth hormone in fetal development. Growth Horm. IGF Res. 2002, 12, 137–146. [Google Scholar] [CrossRef]
- Gluckman, P.D. Clinical review 68: The endocrine regulation of fetal growth in late gestation: The role of insulin-like growth factors. J. Clin. Endocrinol. Metab. 1995, 80, 1047–1050. [Google Scholar] [PubMed]
- Wester, T.J.; Fiorotto, M.L.; Klindt, J.; Burrin, D.G. Feeding colostrum increases circulating insulin-like growth factor I in newborn pigs independent of endogenous growth hormone secretion. J. Anim. Sci. 1998, 76, 3003–3009. [Google Scholar] [CrossRef] [PubMed]
- Scanes, C.G.; Lazarus, D.; Bowen, S.; Buonomo, F.C.; Gilbreath, R.L. Postnatal changes in circulating concentrations of growth hormone, somatomedin C and thyroid hormones in pigs. Domest. Anim. Endocrinol. 1987, 4, 253–257. [Google Scholar] [CrossRef]
- Dauncey, M.J.; Burton, K.A.; White, P.; Harrison, A.P.; Gilmour, R.S.; Duchamp, C.; Cattaneo, D. Nutritional regulation of growth hormone receptor gene expression. FASEB J. 1994, 8, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Harrell, R.J.; Thomas, M.J.; Boyd, R.D.; Czerwinski, S.M.; Steele, N.C.; Bauman, D.E. Ontogenic maturation of the somatotropin/insulin-like growth factor axis. J. Anim. Sci. 1999, 77, 2934–2941. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schroeder, B.; Dahl, M.R.; Breves, G. Duodenal Ca2+ absorption is not stimulated by calcitriol during early postnatal development of pigs. Am. J. Physiol. 1998, 275, G305–G313. [Google Scholar] [CrossRef] [PubMed]
- Chavez, L.S.; Serda, R.; Choe, S.; Davidi, L.; Harmeyer, J.; Omdahl, J.L. Molecular basis for pseudo vitamin D-deficiency rickets in the Hannover pig. J. Nutr. Biochem. 2003, 14, 378–385. [Google Scholar] [CrossRef]
- Plonait, H. Hereditary rickets in suckling pigs: Pathogenesis and therapy. Zentralblatt fur Veterinarmedizin. Reihe A 1969, 16, 271–288. [Google Scholar] [CrossRef] [PubMed]
- Lachenmaier-Currle, U.; Harmeyer, J. Placental transport of calcium and phosphorus in pigs. J. Perinat. Med. 1989, 17, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Eyles, D.W.; Feron, F.; Cui, X.; Kesby, J.P.; Harms, L.H.; Ko, P.; McGrath, J.J.; Burne, T.H. Developmental vitamin D deficiency causes abnormal brain development. Psychoneuroendocrinology 2009, 34 (Suppl. S1), S247–S257. [Google Scholar] [CrossRef] [PubMed]
- Eyles, D.; Burne, T.; McGrath, J. Vitamin D in fetal brain development. Semin. Cell Dev. Biol. 2011, 22, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Pertile, R.A.; Cui, X.; Eyles, D.W. Vitamin D signaling and the differentiation of developing dopamine systems. Neuroscience 2016, 333, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Muller, E.E.; Locatelli, V.; Cocchi, D. Neuroendocrine control of growth hormone secretion. Physiol. Rev. 1999, 79, 511–607. [Google Scholar] [CrossRef] [PubMed]
- Kaune, R.; Harmeyer, J. An inherited disturbance of vitamin-D metabolism in pigs—Pseudo vitamin-D deficiency ricket—Type-1. Berliner und Münchener Tierärztliche Wochenschrift 1987, 100, 6–13. [Google Scholar] [PubMed]
- Kaune, R.; Harmeyer, J. Vitamin-D3 metabolism in a pig strain with pseudo vitamin-D-deficiency rickets, type-I. Acta Endocrinol. 1987, 115, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Bauer, M.; Parvizi, N. Pulsatile and diurnal secretion of GH and IGF-I in the chronically catheterized pig fetus. J. Endocrinol. 1996, 149, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Ellendorff, F.; Parvizi, N.; Elsaesser, F.; Smidt, D. The miniature pig as an animal model in endocrine and neuroendocrine studies of reproduction. Lab. Anim. Sci. 1977, 27, 822–830. [Google Scholar] [PubMed]
- Dixit, V.D.; Mielenz, M.; Taub, D.D.; Parvizi, N. Leptin induces growth hormone secretion from peripheral blood mononuclear cells via a protein kinase C- and nitric oxide-dependent mechanism. Endocrinology 2003, 144, 5595–5603. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Holick, M.F. Benefits and requirements of vitamin D for optimal health: A review. Altern. Med. Rev. 2005, 10, 94–111. [Google Scholar] [PubMed]
- Zentilin, L.; Giacca, M. Competitive PCR for precise nucleic acid quantification. Nat. Protoc. 2007, 2, 2092–2104. [Google Scholar] [CrossRef] [PubMed]
- Schröder, B.; Hattenhauer, O.; Breves, G. Phosphate transport in pig proximal small intestines during postnatal development: Lack of modulation by calcitriol. Endocrinology 1998, 139, 1500–1507. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Holick, M.F. Resurrection of vitamin D deficiency and rickets. J. Clin. Investig. 2006, 116, 2062–2072. [Google Scholar] [CrossRef] [PubMed]
- Klindt, J.; Stone, R.T. Porcine growth hormone and prolactin: Concentrations in the fetus and scretory patterns in the growing pig. Growth 1983, 48, 1–15. [Google Scholar]
- Buonomo, F.C.; Klindt, J. Ontogeny of growth hormone (GH), insulin-like growth factors (IGF-I and IGF-II) and IGF binding protein-2 (IGFBP-2) in genetically lean and obese swine. Domest. Anim. Endocrinol. 1993, 10, 257–265. [Google Scholar] [CrossRef]
- Kesby, J.P.; Turner, K.M.; Alexander, S.; Eyles, D.W.; McGrath, J.J.; Burne, T.H.J. Developmental vitamin D deficiency alters multiple neurotransmitter systems in the neonatal rat brain. Int. J. Dev. Neurosci. 2017, 62, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Bauer, R.; Walter, B.; Brust, P.; Fuchtner, F.; Zwiener, U. Impact of asymmetric intrauterine growth restriction on organ function in newborn piglets. Eur. J. Obstet. Gynecol. Reprod. Biol. 2003, 110 (Suppl. S1), S40–S49. [Google Scholar] [CrossRef]
- Vance, M.L.; Kaiser, D.L.; Frohman, L.A.; Rivier, J.; Vale, W.W.; Thorner, M.O. Role of dopamine in the regulation of growth hormone secretion: Dopamine and bromocriptine augment growth hormone (GH)-releasing hormone-stimulated GH secretion in normal man. J. Clin. Endocrinol. Metab. 1987, 64, 1136–1141. [Google Scholar] [CrossRef] [PubMed]
- Eyles, D.; Feldon, J.; Meyer, U. Schizophrenia: Do all roads lead to dopamine or is this where they start? Evidence from two epidemiologically informed developmental rodent models. Transl. Psychiatry 2012, 2, e81. [Google Scholar] [CrossRef] [PubMed]
- Tekes, K.; Gyenge, M.; Hantos, M.; Csaba, G. Transgenerational hormonal imprinting caused by vitamin A and vitamin D treatment of newborn rats. Alterations in the biogenic amine contents of the adult brain. Brain Dev. 2009, 31, 666–670. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mielenz, M.; Pfaffl, M.W.; Schlumbohm, C.; Harmeyer, J.; Parvizi, N. Growth Hormone Secretion Patterns in German Landrace (DL) Fetuses and Piglets Compared to DL Piglets with Inherited 1,25-Dihydroxyvitamin D3 Deficiency. Nutrients 2018, 10, 617. https://doi.org/10.3390/nu10050617
Mielenz M, Pfaffl MW, Schlumbohm C, Harmeyer J, Parvizi N. Growth Hormone Secretion Patterns in German Landrace (DL) Fetuses and Piglets Compared to DL Piglets with Inherited 1,25-Dihydroxyvitamin D3 Deficiency. Nutrients. 2018; 10(5):617. https://doi.org/10.3390/nu10050617
Chicago/Turabian StyleMielenz, Manfred, Michael W. Pfaffl, Christina Schlumbohm, Johein Harmeyer, and Nahid Parvizi. 2018. "Growth Hormone Secretion Patterns in German Landrace (DL) Fetuses and Piglets Compared to DL Piglets with Inherited 1,25-Dihydroxyvitamin D3 Deficiency" Nutrients 10, no. 5: 617. https://doi.org/10.3390/nu10050617
APA StyleMielenz, M., Pfaffl, M. W., Schlumbohm, C., Harmeyer, J., & Parvizi, N. (2018). Growth Hormone Secretion Patterns in German Landrace (DL) Fetuses and Piglets Compared to DL Piglets with Inherited 1,25-Dihydroxyvitamin D3 Deficiency. Nutrients, 10(5), 617. https://doi.org/10.3390/nu10050617




