Formula Milk Supplementation on the Postnatal Ward: A Cross-Sectional Analytical Study
Abstract
1. Introduction
2. Materials and Methods
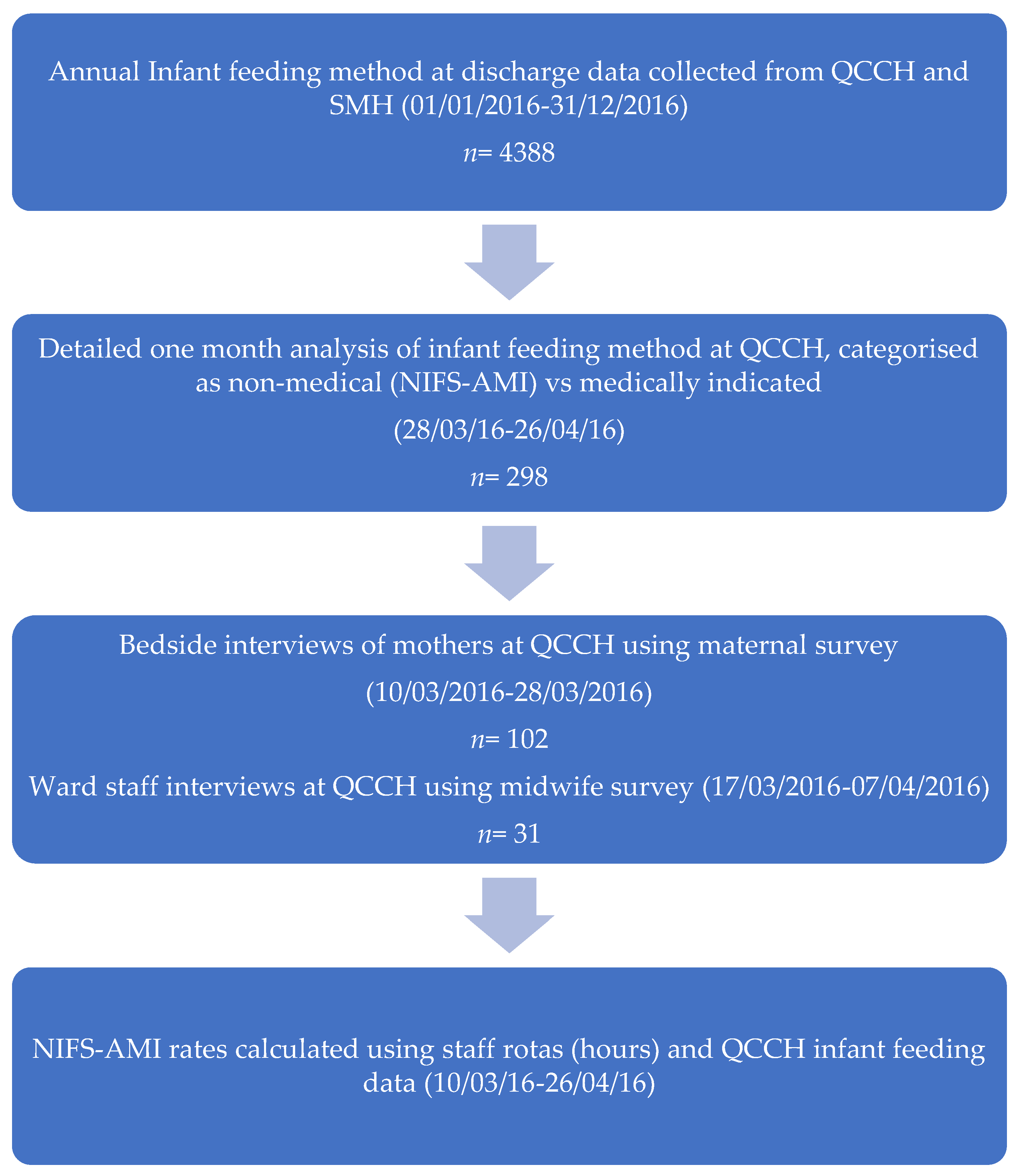
2.1. Study Design
2.2. Study Participants
2.3. Data Collection
2.3.1. Maternal Survey
2.3.2. Midwife Survey
2.4. Data Analysis
3. Results
3.1. Maternal Descriptive Characteristics
3.2. Maternal Characteristics and NIFS-AMI
3.3. Midwife Descriptive Characteristics
3.4. Midwife Attitudes and Recalled Infant Feeding Behaviour
3.5. Assessment of Real-Life BMS Prescription
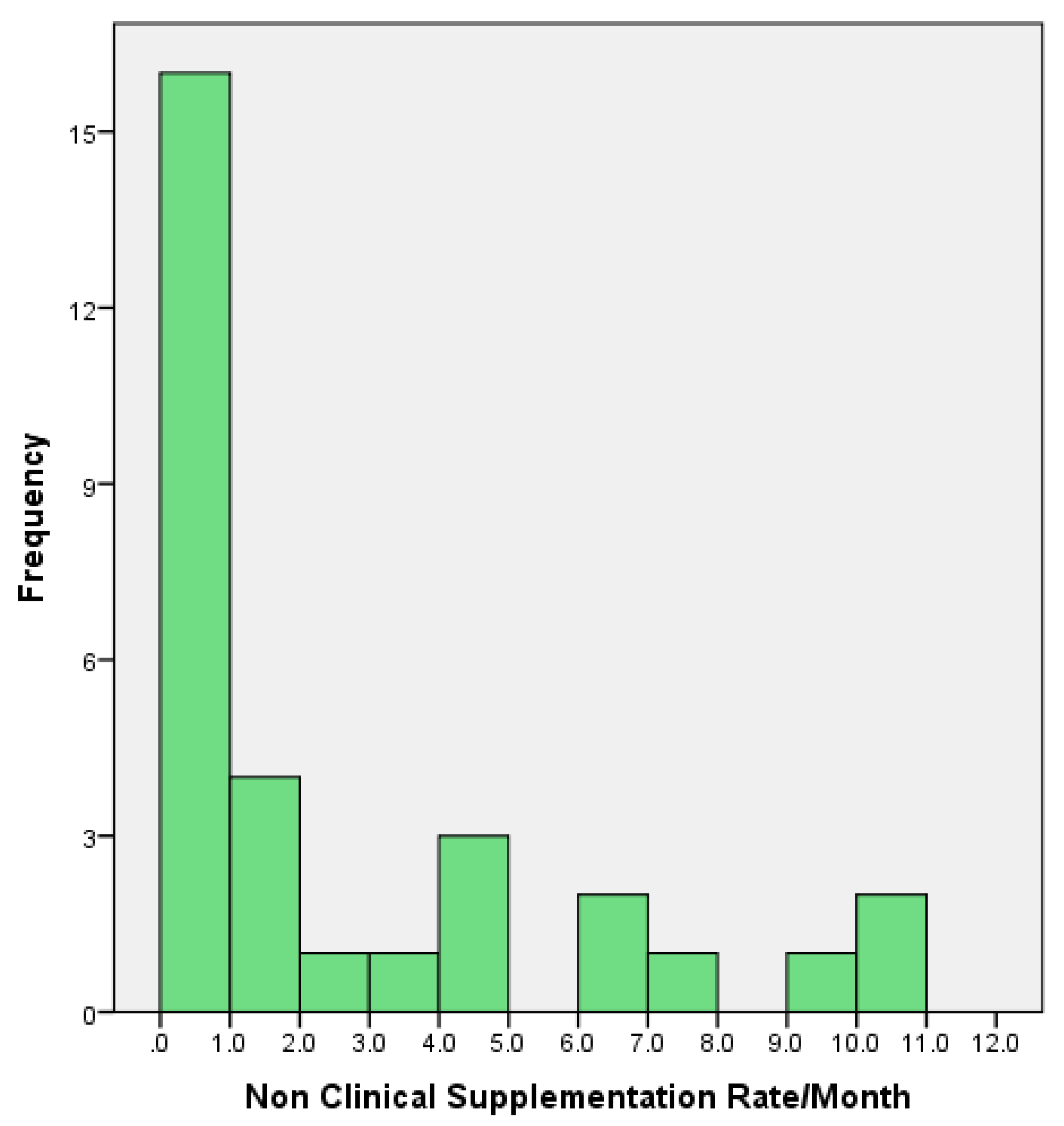
3.6. What Influences NIFS-AMI?
3.7. Perceived Barriers to Supporting Breastfeeding
“Sometimes I get depressed, it is a terrible thing not being able to help”.Midwife 4
“The training courses are pointless if you don’t have the time to use it”.Midwife 12
“It’s never them, the mothers want the support; it’s the time”.Midwife 30
“It has repercussions on our own health, not being able to help, as well as of course the baby and the mothers. The mothers get so depressed”.Midwife 23
“They [the mothers] genuinely don’t know how good it is! They underestimate the difference between breast and formula, it is such a shame”.Midwife 24
“The videos they show in antenatal classes give women unrealistic expectations of breastfeeding, it is harder than they make it look”.Midwife 7
“You cave in sometimes due to the workload, giving the bottle is so easy and that is why it is so sad”.Midwife 23
“I tell the women breastfeeding is hard and needs perseverance. One-to-one support is so important and 30 min isn’t enough”.Midwife 31
“It is exhausting helping women one-to-one to breastfeed all the time, especially when the ward is busy, it takes a lot of effort”.Midwife 18
4. Discussion
4.1. The Use of BMS Supplementation on the Postnatal Ward
4.2. Maternal Factors
4.3. Midwife Factors
4.4. Application of BFHI Policy into Clinical Practice
4.5. Study Limitations
5. Conclusions
- The use of in-hospital breastmilk substitute supplementation for breastfed infants is commonplace in the study hospital, as 95 (28%) of infants received a BMS supplement prior to discharge from maternity unit and 90% of BMS supplementation was given without medical indication.
- Maternal factors protective of neonatal infant-formula supplementation in the absence of medical indication included skin-to-skin and antenatal breastfeeding discussions.
- Although midwives were appropriately trained in accordance with BFHI protocols, they identified a number of workplace barriers in breastfeeding support, with lack of time and heavy workload being the major contributors.
- We found significant associations between the midwives’ decisions on non-medical infant-formula supplementation and educational status, which suggests a need to focus the existing BFHI training programme.
- In order to reduce unnecessary supplementation as well as address attitudes and perceived barriers, additional educational programmes should be developed, with a focus on one-to-one support for establishing lactation in the postnatal environment.
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Rollins, N.C.; Bhandari, N.; Hajeebhoy, N.; Horton, S.; Lutter, C.K.; Martines, J.C.; Piwoz, E.G.; Richter, L.M.; Victora, C.G. Why invest, and what it will take to improve breastfeeding practices? Lancet 2016, 387, 491–504. [Google Scholar] [CrossRef]
- Victora, C.G.; Bahl, R.; Barros, A.J.; França, G.V.; Horton, S.; Krasevec, J.; Murch, S.; Sankar, M.J.; Walker, N.; Rollins, N.C. Breastfeeding in the 21st century: Epidemiology, mechanisms, and lifelong effect. Lancet 2016, 387, 475–490. [Google Scholar] [CrossRef]
- Kramer, M.S.; Kakuma, R. Optimal duration of exclusive breastfeeding. Cochrane Database Syst. Rev. 2012, 15, CD003517. [Google Scholar] [CrossRef] [PubMed]
- Duijts, L.; Jaddoe, V.W.; Hofman, A.; Moll, H.A. Prolonged and exclusive breastfeeding reduces the risk of infectious diseases in infancy. Pediatrics 2010, 126, e18–e25. [Google Scholar] [CrossRef] [PubMed]
- Quigley, M.A.; Kelly, Y.J.; Sacker, A. Breastfeeding and hospitalization for diarrheal and respiratoryinfection in the United Kingdom Millennium Cohort Study. Pediatrics 2007, 119, e837–e842. [Google Scholar] [CrossRef] [PubMed]
- Klement, E.; Cohen, R.V.; Boxman, J.; Joseph, A.; Reif, S. Breastfeeding and risk of inflammatory bowel disease: A systematic review with meta-analysis. Am. J. Clin. Nutr. 2004, 80, 1342–1352. [Google Scholar] [CrossRef] [PubMed]
- Akobeng, A.K.; Ramanan, A.V.; Buchan, I.; Heller, R.F. Effect of breast-feeding on risk of coeliac disease: A systematic review and meta-analysis of observational studies. Arch. Dis. Child. 2006, 91, 39–43. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. Evidence on the Long-Term Effects of Breastfeeding, Systematic Reviews and Meta-Analysis, Department of Child and Adolescent Health and Development (CAH). 2007. Available online: http://apps.who.int/iris/bitstream/10665/43623/1/9789241595230_eng.pdf (accessed on 20 April 2018).
- WHO Collaborative Study Team on the Role of Breastfeeding on the Prevention of Infant Mortality. Effect of breastfeeding on infant and child mortality due to infectious diseases in less developed countries: A pooled analysis. Lancet 2000, 355, 451–455. [Google Scholar]
- Bachrach, V.R.; Schwarz, E.; Bachrach, L.R. Breastfeeding and the risk of hospitalization for respiratory disease in infancy: A meta-analysis. Arch. Pediatr. Adolesc. Med. 2003, 157, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Hauck, F.R.; Thompson, J.M.; Tanabe, K.O.; Moon, R.Y.; Vennemann, M.M. Breastfeeding and reduced risk of sudden infant death syndrome: A meta-analysis. Pediatrics 2011, 128, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Rothenbacher, D.; Weyermann, M.; Beermann, C.; Brenner, H. Breastfeeding, soluble CD14 concentration in breast milk and risk of atopic dermatitis and asthma in early childhood: Birth cohort study. Clin. Exp. Allergy 2005, 35, 1014–1021. [Google Scholar] [CrossRef] [PubMed]
- Greer, F.R.; Sicherer, S.H.; Burks, A.W.; American Academy of Pediatrics Committee on Nutrition; American Academy of Pediatrics Section on Allergy and Immunology. Effects of early nutritional interventions on the development of atopic disease in infants and children: The role of maternal dietary restriction, breastfeeding, timing of introduction of complementary foods, and hydrolyzed formulas. Pediatrics 2008, 121, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Horta, B.L.; Bahl, R.; Martines, J.C.; Victora, C.G. Evidence on the Long-Term Effects of Breastfeeding. Geneva: World Health Organization. 2007. Available online: http://apps.who.int/iris/bitstream/handle/10665/43623/9789241595230_eng.pdf;jsessionid=96C96746137B09E506E1051FAAA5CDDC?sequence=1 (accessed on 23 April 2018).
- Harder, T.; Bergmann, R.; Kallischnigg, G.; Plagemann, A. Duration of breastfeeding and risk of overweight: A meta-analysis. Am. J. Epidemiol. 2005, 162, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Arenz, S.; Ruckeri, R.; Koletzko, B.; von Kries, R. Breastfeeding and Childhood obesity: A systematic review. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 1247–1256. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.S.; Aboud, F.; Mironova, E.; Vanilovich, I.; Platt, R.W.; Matush, L.; Igumnov, S.; Fombonne, E.; Bogdanovich, N.; Ducruet, T.; et al. Breastfeeding and child cognitive development: New evidence from a large randomized trial. Arch. Gen. Psychiatry 2008, 65, 578–584. [Google Scholar] [CrossRef] [PubMed]
- Heikkilä, K.; Sacker, A.; Kelly, Y.; Renfrew, M.J.; Quigley, M.A. Breastfeeding and child behaviour in the Millennium Cohort Study. Arch. Dis. Child. 2011, 96, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Quigley, M.A.; Hockley, C.; Carson, C.; Kelly, Y.; Renfrew, M.J.; Sacker, A. Breastfeeding is Associated with Improved Child Cognitive Development: A Population-Based Cohort Study. J. Pediatr. 2012, 160, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.S.; Chalmers, B.; Hodnett, E.D.; Sevkovskaya, Z.; Dzikovich, I.; Shapiro, S.; Collet, J.P.; Vanilovich, I.; Mezen, I.; Ducruet, T.; et al. Promotion of Breastfeeding Intervention Trial (PROBIT). A randomized trial in the Republic of Belarus. JAMA 2001, 285, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Dewey, K.G.; Heinig, M.J.; Nommsen, L.A. Maternal weight-loss patterns during prolonged lactation. Am. J. Clin. Nutr. 1993, 58, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Short, R.V. Lactational infertility in family planning. Ann. Med. 1993, 25, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Collaborative Group on Hormonal Factors in Breast Cancer. Breast cancer and breastfeeding: Collaborative reanalysis of individual data from 47 epidemiological studies in 30 countries, including 50302 women with breast cancer and 96973 women without the disease. Lancet 2002, 360, 187–195. [Google Scholar]
- Islami, F.; Liu, Y.; Jemal, A.; Zhou, J.; Weiderpass, E.; Colditz, G.; Boffetta, P.; Weiss, M. Breastfeeding and breast cancer risk by receptor status—A systematic review and meta-analysis. Ann. Oncol. 2015, 26, 2398–2407. [Google Scholar] [CrossRef] [PubMed]
- Tung, K.H.; Goodman, M.T.; Wu, A.H.; McDuffie, K.; Wilkens, L.R.; Kolonel, L.N.; Nomura, A.M.; Terada, K.Y.; Carney, M.E.; Sobin, L.H. Reproductive factors and epithelial ovarian cancer risk by histologic type:a multiethnic case-control study. Am. J. Epidemiol. 2003, 158, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Rosenblatt, K.A.; Thomas, D.B. Lactation and the risk of epithelial ovarian cancer—The WHO Collaborative Study of Neoplasia and Steroid Contraceptives. Int. J. Epidemiol. 1993, 22, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Bjornerem, A.; Ahmed, L.A.; Jorgensen, L.; Stormer, J.; Joakimsen, R.M. Breastfeeding protects against hip fracture in postmenopausal women: The Tromso study. J. Bone Miner. Res. 2011, 26, 2843–2850. [Google Scholar] [CrossRef] [PubMed]
- Stafford, M.; Kuh, D.L.; Gale, C.R.; Mishra, G.; Richards, M. Parent-child relationships and offspring’s positive mental wellbeing from adolescence to early older age. J. Posit. Psychol. 2016, 11, 326–337. [Google Scholar] [CrossRef] [PubMed]
- Puckering, C.; Allely, C.S.; Doolin, O.; Purves, D.; McConnachie, A.; Johnson, P.C.; Marwick, H.; Heron, J.; Golding, J.; Gillberg, C.; et al. Association between parent-infant interactions in infancy and disruptive behaviour disorders at age seven: A nested, case–control ALSPAC study. BMC Pediatr. 2014, 14, 223. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.; Rance, J.; Bennett, P. Understanding the relationship between breastfeeding and postnatal depression: The role of pain and physical difficulties. J. Adv. Nurs. 2016, 72, 273–282. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. Up to What Age Can a Baby Stay Well Nourished by Just Being Breastfed? 2015. Available online: http://www.who.int/features/qa/21/en/ (accessed on 24 April 2018).
- Kılcı, H.; Çoban, A. The correlation between breastfeeding success in the early postpartum period and the perception of self-efficacy in breastfeeding and breast problems in the late postpartum. Breastfeed. Med. 2016, 11, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Forster, D.A.; Johns, H.M.; McLachlan, H.L.; Moorhead, A.M.; McEgan, K.M.; Amir, L.H. Feeding infants directly at the breast during the postpartum hospital stay is associated with increased breastfeeding at 6 months postpartum: A prospective cohort study. BMJ Open 2015, 5, e007512. [Google Scholar] [CrossRef] [PubMed]
- National Infant Feeding Survey 2010. Chapter 2: Incidence Prevalence and Duration of Breastfeeding. 2012. Available online: http://www.hscic.gov.uk/catalogue/PUB08694/ifs-uk-2010-chap2-inc-prev-dur.pdf (accessed on 6 May 2016).
- O’Connor, M.; Allen, J.; Kelly, J.; Gao, Y.; Kildea, S. Predictors of breastfeeding exclusivity and duration in a hospital without Baby Friendly Hospital Initiative accreditation: A prospective cohort study. Women Birth 2017. [Google Scholar] [CrossRef] [PubMed]
- Chantry, C.J.; Dewey, K.G.; Peerson, J.M.; Wagner, E.A.; Nommsen-Rivers, L.A. In-hospital formula use increases early breastfeeding cessation among first-time mothers intending to exclusively breastfeed. J. Pediatr. 2014, 164, 1339–1345. [Google Scholar] [CrossRef] [PubMed]
- Gaufin, T.; Tobin, N.H.; Aldrovandi, G.M. The importance of the microbiome in pediatrics and pediatric infectious diseases. Curr. Opin. Pediatr. 2018, 30, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Bentley, J.P.; Nassar, N.; Porter, M.; de Vroome, M.; Yip, E.; Ampt, A.J. Formula supplementation in hospital and subsequent feeding at discharge among women who intended to exclusively breastfeed: An administrative data retrospective cohort study. Birth 2017, 44, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.; Dennison, B.A.; Fan, W.; Xu, C.; Birkhead, G.S. Variation in Formula Supplementation of Breastfed Newborn Infants in New York Hospitals. Pediatrics 2017, 140, E20170142. [Google Scholar] [CrossRef] [PubMed]
- Hunter, L.; Magill-Cuerden, J.; McCourt, C. ‘Oh no, no, no, we haven’t got time to be doing that’: Challenges encountered introducing a breast-feeding support intervention on a postnatal ward. Midwifery 2015, 31, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Cantrill, R.; Creedy, D.; Cooke, M. Midwives’ knowledge of newborn feeding ability and reported practice managing the first breastfeed. Breastfeed. Rev. 2004, 12, 25–33. [Google Scholar] [PubMed]
- Darwent, K.L.; Kempenaar, L.E. A comparison of breastfeeding women’s, peer supporters’ and student midwives’ breastfeeding knowledge and attitudes. Nurse Educ. Pract. 2014, 14, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Marks, D.; O’Connor, R. Breastfeeding support and promotion: The health professional’s perspective. J. Health Visit. 2015, 3, 38–46. [Google Scholar] [CrossRef]
- Marks, D.; O’Connor, R. Health professionals’ attitudes towards the promotion of breastfeeding. Br. J. Midwifery 2015, 23, 50–58. [Google Scholar] [CrossRef]
- World Health Organization. Acceptable Medical Reasons for Use of Breast Milk Substitutes; WHO: Geneva, Switzerland, 2009; Available online: http://apps.who.int/iris/bitstream/10665/69938/1/WHO_FCH_CAH_09.01_eng.pdf (accessed on 4 May 2016).
- UNICEF. Tools and Forms to Help Support Baby Friendly. Available online: https://www.unicef.org.uk/babyfriendly/baby-friendly-resources/guidance-for-health-professionals/tools-and-forms-for-health-professionals/ (accessed on 4 May 2016).
- Office of National Statistics-Ethnicity Groups. Available online: https://www.google.co.uk/url?sa=t&rct=j&q=&esrc=s&source=web&cd=6&cad=rja&uact=8&ved=0ahUKEwjxqNqfxfzWAhXBPBoKHXa3AVQQFgg0MAU&url=https%3A%2F%2Fwww.ons.gov.uk%2Fons%2Fguide-method%2Fharmonisation%2Fprimary-set-of-harmonised-concepts-and-questions%2Fethnic-group.pdf&usg=AOvVaw3O-S-j3HqaDLWP0_wT-jhs (accessed on 4 May 2016).
- Tender, J.A.; Janakiram, J.; Arce, E.; Mason, R.; Jordan, T.; Marsh, J.; Kin, S.; He, J.; Moon, R.Y. Reasons for in-hospital formula supplementation of breastfed infants from low-income families. J. Hum. Lact. 2009, 25, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Good Food for London. 2015. Available online: http://www.sustainweb.org/londonfoodlink/goodfoodforlondon2015/ (accessed on 6 May 2016).
- National Maternity and Perinatal Audit 2017. Available online: http://www.maternityaudit.org.uk/downloads/RCOG%20NMPA%20Clinical%20Report(web).pdf (accessed on 13 February 2018).
- Flacking, R.; Nyqvist, K.H.; Ewald, U. Effects of socioeconomic status on breastfeeding duration in mothers of preterm and term infants. Eur. J. Public Health 2007, 17, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Moore, E.R.; Bergman, N.; Anderson, G.C.; Medley, N. Early skin-to-skin contact for mothers and their healthy newborn infants. Cochrane Database Syst. Rev. 2016, 11, CD003519. [Google Scholar] [CrossRef] [PubMed]
- DaMota, K.; Bañuelos, J.; Goldbronn, J.; Vera-Beccera, L.E.; Heinig, M.J. Maternal request for in-hospital supplementation of healthy breastfed infants among low-income women. J. Hum. Lact. 2012, 28, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J.S.; Hall, E.O.; Aagaard, H. Shattered expectations: When mothers’ confidence in breastfeeding is undermined--a metasynthesis. Scand. J. Caring Sci. 2008, 22, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Bailey, C.; Pain, R.H.; Aarvold, J.E. A ‘give it a go’ breast-feeding culture and early cessation among low-income mothers. Midwifery 2004, 20, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Trickey, H.; Newburn, M. Goals, dilemmas and assumptions in infant feeding education and support. Applying theory of constraints thinking tools to develop new priorities for action. Matern. Child Nutr. 2014, 10, 72–91. [Google Scholar] [CrossRef] [PubMed]
- Lumbiganon, P.; Martis, R.; Laopaiboon, M.; Festin, M.R.; Ho, J.J.; Hakimi, M. Antenatal breastfeeding education for increasing breastfeeding duration. Cochrane Database Syst. Rev. 2012, 9, CD006425. [Google Scholar] [CrossRef]
- Wong, K.L.; Fong, D.Y.; Lee, I.L.; Chu, S.; Tarrant, M. Antenatal education to increase exclusive breastfeeding: A randomized controlled trial. Obstet. Gynecol. 2014, 124, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Lavender, T.; Baker, L.; Smyth, R.; Collins, S.; Spofforth, A.; Dey, P. Breastfeeding expectations versus reality: A cluster randomised controlled trial. BJOG 2005, 112, 1047–1053. [Google Scholar] [CrossRef] [PubMed]
- Hauck, Y.L.; Irurita, V.F. Incompatible expectations: The dilemma of breastfeeding mothers. Health Care Women Int. 2003, 24, 62–78. [Google Scholar] [CrossRef] [PubMed]
- Fox, R.; McMullen, S.; Newburn, M. UK women’s experiences of breastfeeding and additional breastfeeding support: A qualitative study of Baby Café services. BMC Pregnancy Childbirth 2015, 15, 147. [Google Scholar] [CrossRef] [PubMed]
- Kmietowicz, Z. Staff shortages and lack of data continue to blight England’s NHS and care regulator. BMJ 2015, 351, h4000. [Google Scholar] [CrossRef] [PubMed]
- Triggle, N. Staff shortage puts neonatal services under strain. Nurs. Child. Young People 2015, 27, 7. [Google Scholar] [CrossRef] [PubMed]
- Furber, C.M.; Thomson, A.M. Midwives in the UK: An exploratory study of providing newborn feeding support for postpartum mothers in the hospital. J. Midwifery Womens Health 2007, 52, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Ockleford, E.M.; Berryman, J.C.; Hsu, R. Postnatal care: What new mothers say. Br. J. Midwifery 2004, 12, 166–170. [Google Scholar] [CrossRef]
- Sharp, D.; Entwistle, F. Why women stop breastfeeding in the early days. Pract. Midwife 2015, 18, 30–33. [Google Scholar] [PubMed]
- Oakley, L.L.; Henderson, J.; Redshaw, M.; Quigley, M.A. The role of support and other factors in early breastfeeding cessation: An analysis of data from a maternity survey in England. BMC Pregnancy Childbirth 2014, 14, 88. [Google Scholar] [CrossRef] [PubMed]
- Bowers, J.; Cheyne, H. Reducing the length of postnatal hospital stay: Implications for cost and quality of care. BMC Health Serv. Res. 2016, 16, 16. [Google Scholar] [CrossRef] [PubMed]
- UNICEF/BFI Supplementation Guidance: Maternity Standard 4. March 2016. Available online: https://www.unicef.org.uk/babyfriendly/wp-content/uploads/sites/2/2016/10/Supplementation-guidance.pdf (accessed on 17 April 2018).
- Forster, D.A.; McLachlan, H.L.; Yelland, J.; Rayner, J.; Lumley, J.; Davey, M.A. Staffing in postnatal units: Is it adequate for the provision of quality care? Staff perspectives from a state-widereview of postnatal care in Victoria, Australia. BMC Health Serv. Res. 2006, 6, 83. [Google Scholar] [CrossRef] [PubMed]
- Odom, E.C.; Li, R.; Scanlon, K.S.; Perrine, C.G.; Grummer-Strawn, L. Association of family and health care provider opinion on infant feeding with mother’s breastfeeding decision. J. Acad. Nutr. Diet. 2014, 114, 1203–1207. [Google Scholar] [CrossRef] [PubMed]
- Swanson, V.; Power, K.G. Initiation and continuation of breastfeeding: Theory of planned behaviour. J. Adv. Nurs. 2005, 50, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Radzyminski, S.; Callister, L.C. Mother’s beliefs, attitudes, and decision making related to infant feeding choices. J. Perinat. Educ. 2016, 25, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.M.; Hutchings, M. Using video narratives of women’s lived experience of breastfeeding in midwifery education: Exploring its impact on midwives’ attitudes to breastfeeding. Matern. Child Nutr. 2012, 8, 88–102. [Google Scholar] [CrossRef] [PubMed]
- DiGirolamo, A.M.; Grummer-Strawn, L.M.; Fein, S.B. Do perceived attitudes of physicians and hospital staff affect breastfeeding decisions? Birth 2003, 30, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Creedy, D.K.; Cantrill, R.M.; Cooke, M. Assessing midwives’ breastfeeding knowledge: Properties of the Newborn Feeding Ability questionnaire and Breastfeeding Initiation Practices scale. Int. Breastfeed. J. 2008, 3, 7. [Google Scholar] [CrossRef] [PubMed]
- Cantrill, R.M.; Creedy, D.K.; Cooke, M. An Australian study of midwives’ breast-feeding knowledge. Midwifery 2003, 19, 310–317. [Google Scholar] [CrossRef]
- Pierro, J.; Abulaimoun, B.; Roth, P.; Blau, J. Factors associated with supplemental formula feeding of breastfeeding infants during postpartum hospital stay. Breastfeed. Med. 2016, 11, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Atchan, M.; Davis, D.; Foureur, M. Applying a knowledge translation model to the uptake of the Baby Friendly Health Initiative in the Australian health care system. Women Birth. 2014, 27, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Glasziou, P.; Haynes, B. The paths from research to improved health outcomes. Evid. Based Nurs. 2005, 8, 36–38. [Google Scholar] [CrossRef] [PubMed]
- Furber, C.M.; Thomson, A.M. The emotions of integrating breastfeeding knowledge into practice for English midwives: A qualitative study. Int. J. Nurs. Stud. 2008, 45, 286–297. [Google Scholar] [CrossRef] [PubMed]
- Crano, W.D.; Prislin, R. (Eds.) Attitudes and Attitude Change; Psychology Press, Taylor and Francis Group: New York, NY, USA, 2008; p. 11. Available online: https://books.google.co.uk/books?hl=en&lr=&id=HpF5AgAAQBAJ&oi=fnd&pg=PP1&dq=change+attitudes+&ots=dyeEeCvkgp&sig=6rxaq_rrILyrOBbwYRkpRw4izOs#v=onepage&q=behavior&f=false (accessed on 14 May 2016).
- Schwarz, J.; Gaito, P.; Lennick, D. That’s the way we (used to) do things around here. Strategy Bus. 2011, 62, 45–53. [Google Scholar]
- Fazio, R.H.; Zanna, M.P.; Cooper, J. Direct experience and attitude-behavior consistency: An information processing analysis. Pers. Soc. Psychol. Bull. 1978, 4, 48–51. [Google Scholar] [CrossRef]
- Russell, K.; Walsh, D. Can the use of behavioural intervention studies support change in professional practice behaviours? Evid. Based Midwifery 2009, 7, 54–59. [Google Scholar]
- Salvador, A.; Dumas, L.; Davies, B.; Emard, M.J.; Lortie, K. A dozen strategies along the ten steps Baby-Friendly Initiative journey. Healthc. Q. 2016, 18, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M. Baby Friendly: A way to accreditation. Part 2. Br. J. Midwifery 2003, 11, 657–662. [Google Scholar] [CrossRef]
- Reddin, E.; Pincombe, J.; Darbyshire, P. Passive resistance: Early experiences of midwifery students/graduates and the Baby Friendly Health Initiative 10 steps to successful breastfeeding. Women Birth 2007, 20, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Levin, K.A. Study design III: Cross-sectional studies. Evid. Based Dent. 2006, 7, 24–25. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Pomar, E.; Blubaugh, R. The Baby Friendly Hospital Initiative and the ten steps for successful breastfeeding. A critical review of the literature. J. Perinatol. 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Howe-Heyman, A.; Lutenbacher, M. The Baby-Friendly Hospital Initiative as an Intervention to Improve Breastfeeding Rates: A Review of the Literature. J. Midwifery Womens Health 2016, 61, 77–102. [Google Scholar] [CrossRef] [PubMed]
| Survey | QCCH | SMH | Both Sites | |
|---|---|---|---|---|
| n = 102 | n = 486 | n = 329 | n = 815 | |
| Maternal age (years) | 32 (28, 36) | 32 (29, 36) | 33 (28, 36) | 33 (29, 36) |
| Feeding status | n = 220 * | n = 117 * | n = 337 * | |
| Exclusively BF | 55 (54) | 138 (63) | 75 (64) | 213 (63) |
| Supplemented with medical justification | 3 (3) | 67 (30) | 28 (24) | 95 (28) |
| Supplemented without medical justification | ||||
| NIFS-AMI | 38 (37) | |||
| Exclusive BMS | 6 (6) | 15 (7) | 14 (12) | 29 (9) |
| Baby age (days) | 1 (1, 2) | |||
| Length of admission (days) | 2 (1, 3) | 2 (1, 4) | 2 (1, 3) | |
| Age left education (years) | 23 (21, 24) | |||
| Highest level of education: University degree | 66 (65) | |||
| Ethnicity: White Caucasian | 53/88 (60) | 149/295 (51) | 106/182 (58) | 255/477 (53) |
| Vaginal delivery | 64 (63) | 357 (73) | 201 (61) | 558 (68) |
| Laboured <2 h | 82 (80) | 410 (84) | 280 (85) | 690 (85) |
| Primiparous | 59 (58) | 83 (17) | 42 (13) | 125 (15) |
| Weight of infant | 3.2 (2.9, 3.6) | 3.2 (2.9, 3.5) | 3.3 (3.0, 3.6) | 3.3 (2.9, 3.6) |
| Low (<2.5 kg) | 8 (8) | 41 (8) | 20 (7) | 61 (8) |
| Gestational age (weeks) | 39 (38, 40) | 39 (38, 40) | 39 (39, 40) | 39 (38, 40) |
| If multiparous, EBF previous infants | 26/43 (61) | |||
| Ward type | ||||
| Postnatal | 82 (80) | 406 (84) | 216 (66) | 622 (76) |
| Private Ward | 9 (9) | 0 (0) | 70 (21) | 70 (9) |
| Birth Centre | 11 (11) | 80 (16) | 43 (13) | 123 (15) |
| Private room | 25 (25) | |||
| Infant remained with mother in hospital | 92 (90) | |||
| Partner present at night | 42 (41) | |||
| Any skin-to-skin after birth | 84 (82) | |||
| Skin-to-skin for 1 h after birth | 50 (49) | |||
| Supported to BF after birth | 68 (67) | |||
| Assisted with positioning and attachment | 82 (80) | |||
| Reports having received education about signs that infant is receiving enough milk | 50 (49) | |||
| Reports having received education about hand expression | 39 (38) | |||
| Reports having antenatal BF discussion with HCP | 39 (38) |
| Number of Infants Receiving NIFS-AMI | Number of Infants EBF + Medically Justified Supplementation | p Value | |
|---|---|---|---|
| n = 38 | n = 55 + 3 | ||
| Maternal age (years) | 29 (25, 37) | 34 (29, 36) | 0.13 |
| Baby age (days) | 2 (1, 3) | 1 (1, 1) | 0.01 |
| Age left education (years) | 22 (19, 24) | 23 (21, 24) | 0.19 |
| Highest level of education: University degree | 18 (47) | 44 (76) | <0.01 |
| Employed prior to pregnancy | 21 (55) | 43 (74) | 0.08 |
| Ethnicity: White Caucasian | 23 (61) | 33 (57) | 0.29 |
| Vaginal delivery | 23 (61) | 38 (66) | 0.62 |
| Laboured >2 h | 31 (82) | 45 (78) | 0.64 |
| Primiparous | 24 (63) | 32 (55) | 0.44 |
| Weight of infant | 3.3 (2.8, 3.6) | 3.3 (3.0, 3.5) | |
| Gestational age (weeks) | 39 (38, 40) | 40 (39, 41) | 0.07 |
| If multiparous, EBF previous infants | 6/14 (43) | 19/26 (73) | 0.06 |
| BF duration with previous infants | 12 (5, 24) | 7 (6, 9) | 0.39 |
| Ward type | 0.04 | ||
| Postnatal | 34 (45) | 42 (55) | |
| Birth Centre/Private Ward | 4 (25) | 16 (75) | |
| Private room | 7 (18) | 18 (31) | 0.17 |
| Infant remained with mother in hospital | 36 (95) | 52 (90) | 0.47 |
| Partner present at night | 18 (47) | 20 (35) | 0.13 |
| Any skin-to-skin after birth | 26 (68) | 54 (93) | <0.01 |
| Skin-to-skin for 1 h after birth | 13 (34) | 36 (62) | 0.01 |
| Supported to BF after birth | 27 (71) | 41 (71) | 0.97 |
| Assisted with positioning and attachment | 33 (87) | 47 (81) | 0.46 |
| Mother reported antenatal education about signs that infant is receiving enough milk | 22 (58) | 25 (43) | 0.16 |
| Mother reported antenatal education on responsive feeding | 10 (26) | 33 (57) | 0.10 |
| Mother reported antenatal education about hand expression | 23 (61) | 15 (26) | <0.01 |
| Mother reported antenatal education on benefits of developing relationship with infant | 14 (37) | 32 (55) | 0.01 |
| Informed of support available within the community | 17 (45) | 24 (41) | 0.75 |
| Attended BF antenatal class at QCCH | 3 (8) | 7 (12) | 0.74 |
| Attended BF antenatal class at another site | 7 (18) | 16 (28) | 0.30 |
| Antenatal BF discussion | 9 (24) | 29 (50) | 0.01 |
| Mother reports having been EBF as infant | 16 (43) | 23 (40) | 0.99 |
| Duration EBF as infant | 12 (12, 24) | 7 (6, 12) | 0.05 |
| BF confidence on ward (1–10) | 7 (6, 8) | 7 (6, 9) | 0.28 |
| BF confidence before birth (1–10) | 8 (6, 10) | 6 (5, 9) | 0.09 |
| BMS first introduced (hours) | 6 (4, 24) | 5 (3, 39) | 0.94 |
| Positive attitude to BF in social/cultural background | 29 (76) | 46 (79) | 0.73 |
| Midwife Attitude and Knowledge of Breastfeeding | n (IQR) |
|---|---|
| Perceived importance of role in BF (1–10) * | 10 (10, 10) |
| Perceived impact of BMS supplements (1–10) * | 8 (6, 10) |
| Perceived time satisfaction (1–10) * | 5 (1, 8) |
| Confidence in FF (1–10) * | 8 (4, 10) |
| n (%) | |
| Positive attitudes to BF in social/cultural background | 24 (77) |
| Staff satisfied with time for FF | 10 (32) |
| Received training on FF | 19 (61) |
| Supplementation practice violates WHO criteria | 20 (65) |
| One of three examples violated criteria | 6 (19) |
| Two of three examples violated criteria | 10 (32) |
| All examples described violated criteria | 4 (13) |
| Percentage of time allocation to feeding support versus other tasks during a typical day | % (IQR) |
| Specific feeding support | 12 (10, 20) |
| General patient care | 25 (20, 40) |
| Computer/paperwork | 50 (30, 60) |
| Taking bedside observations | 10 (10, 15) |
| Descriptive Characteristics | Median/IQR | r | p Value |
|---|---|---|---|
| Staff age | 0.2 | 0.28 | |
| Years’ experience as a midwife | 0.03 | 0.89 | |
| Ward Type | |||
| Postnatal | 1.9 (0.7, 4.2) | <0.01 | |
| Birth Centre/Private Ward | 0.0 (0.0, 0.4) | ||
| Ethnic Background | |||
| White Caucasian | 0.4 (0.0, 0.7) | 0.26 | |
| Non-Caucasian | 1.9 (0.6, 3.0) | ||
| Highest level of education | |||
| Degree | 0.4 (0.0, 1.9) | 0.07 | |
| Non-degree | 3.4 (2.2, 4.7) | ||
| Personal experiences | |||
| Attitudes to BF in social/cultural background | |||
| Positive | 0.7 (0.0, 2.7) | 0.84 | |
| Negative or mixed | 0.4 (0.0, 2.7) | ||
| Parity | |||
| Primiparous/multiparous | 0.7 (0.4, 4.2) | 0.22 | |
| Nulliparous | 0.4 (0.0, 1.8) | ||
| Infant feeding method | |||
| EBF | 0.6 (0.0, 1.9) | 0.57 | |
| Non-EBF | 2.5 (0.5, 4.2) | ||
| Longest BF duration (months) | −0.29 | 0.32 | |
| Earliest age child received non-breastmilk (months) | −0.79 | 0.58 | |
| Midwife’s reported feeding method as infant | 0.62 | ||
| EBF | 0.6 (0.0, 1.8) | ||
| Mon-EBF | 0.8 (0.0, 2.7) | ||
| Duration of BF as infant (months) | 0.43 | 0.11 | |
| Staff perceptions and professional experiences | |||
| Perceived impact of BMS supplementation (1–10) | −0.33 | 0.07 | |
| Happiness with time for infant feeding support (1–10) | −0.47 | 0.01 | |
| Attended training sponsored by a formula company | |||
| Yes | 0.8 (0.3, 2.7) | 0.62 | |
| No | 0.5 (0.0, 2.6) | ||
| Received training on finger feeding | |||
| Yes | 0.4 (0.0, 3.0) | 0.86 | |
| No | 0.9 (0.0, 2.7) | ||
| Satisfied with time available for finger feeding | |||
| Yes | 0.0 (0.0, 0.6) | 0.43 | 0.02 |
| No | 1.2 (0.3, 4.2) | ||
| Confidence in finger feeding (1–10) | −0.40 | 0.03 | |
| Correctly identify three correct reasons for supplementing | |||
| Yes | 0.6 (0.0, 4.6) | 0.07 | |
| No | 0.6 (0.0, 2.1) | ||
| Percentage of time allocation to feeding support versus other tasks during a typical day (%) | |||
| Infant feeding support | 0.56 | 0.77 | |
| General patient care | −0.01 | 0.96 | |
| Computer/paperwork | −0.01 | 0.95 | |
| Taking bedside observations | 0.14 | 0.47 | |
| Parameter | B | p Value | 95% Confidence Interval | |
|---|---|---|---|---|
| Lower Bound | Upper Bound | |||
| Education | 2.53 | 0.01 | 0.95 | 4.12 |
| Breastmilk substitute impact (1–10) | −0.19 | 0.22 | −0.48 | 0.07 |
| Ability to provide three correct scenarios (100%) | 1.01 | 0.18 | −2.47 | 0.23 |
| Ward | −1.63 | 0.01 | −2.50 | −0.66 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Biggs, K.V.; Hurrell, K.; Matthews, E.; Khaleva, E.; Munblit, D.; Boyle, R.J. Formula Milk Supplementation on the Postnatal Ward: A Cross-Sectional Analytical Study. Nutrients 2018, 10, 608. https://doi.org/10.3390/nu10050608
Biggs KV, Hurrell K, Matthews E, Khaleva E, Munblit D, Boyle RJ. Formula Milk Supplementation on the Postnatal Ward: A Cross-Sectional Analytical Study. Nutrients. 2018; 10(5):608. https://doi.org/10.3390/nu10050608
Chicago/Turabian StyleBiggs, Kirsty V., Katherine Hurrell, Eleanor Matthews, Ekaterina Khaleva, Daniel Munblit, and Robert J. Boyle. 2018. "Formula Milk Supplementation on the Postnatal Ward: A Cross-Sectional Analytical Study" Nutrients 10, no. 5: 608. https://doi.org/10.3390/nu10050608
APA StyleBiggs, K. V., Hurrell, K., Matthews, E., Khaleva, E., Munblit, D., & Boyle, R. J. (2018). Formula Milk Supplementation on the Postnatal Ward: A Cross-Sectional Analytical Study. Nutrients, 10(5), 608. https://doi.org/10.3390/nu10050608





