Eicosapentaenoic Acid (EPA) and Docosahexaneoic Acid (DHA) in Muscle Damage and Function
Abstract
:1. Introduction
2. EPA and DHA for Muscle and Nerve Damage
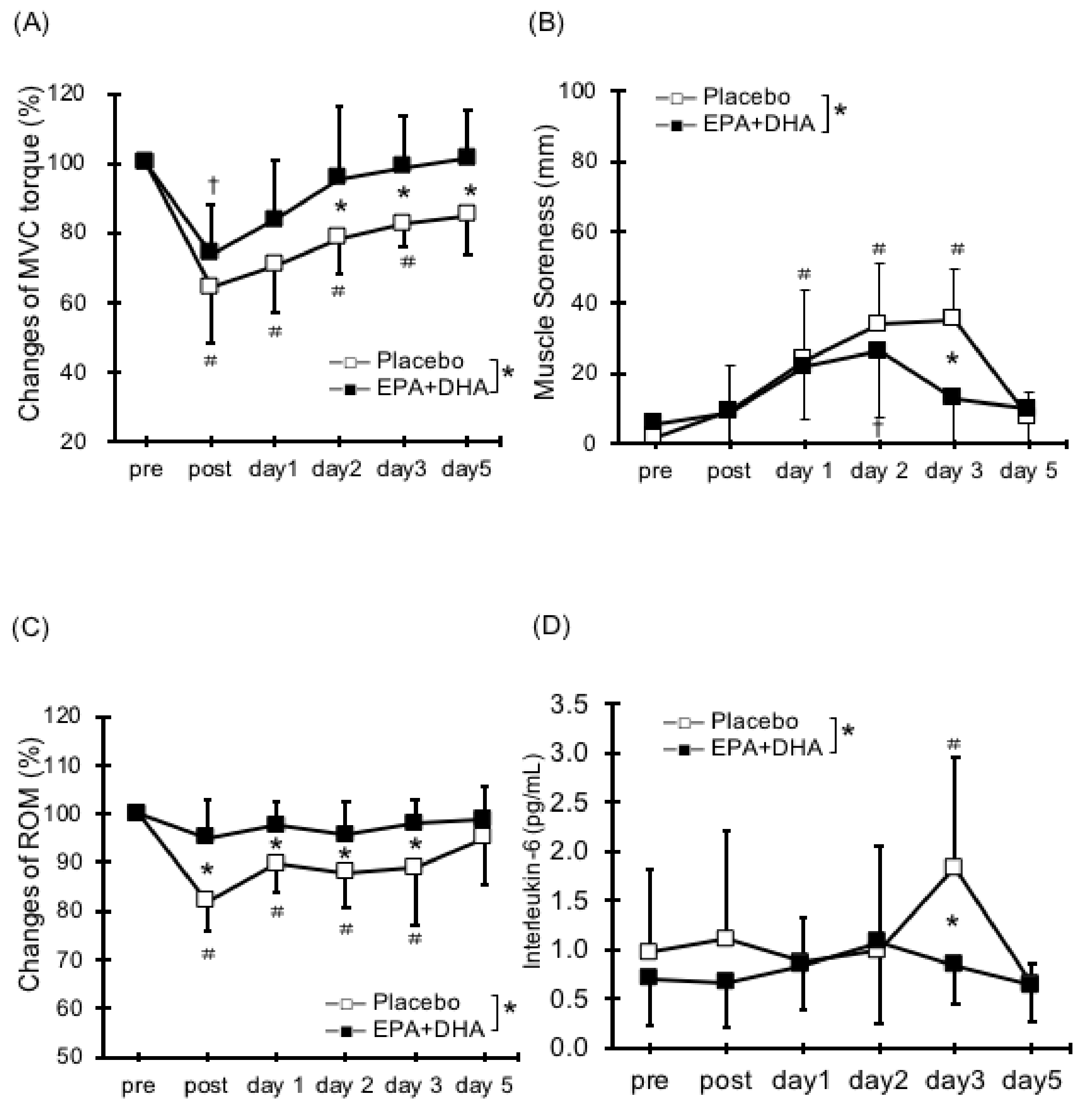
2.1. Muscle Strength Deficit
2.2. Delayed Onset Muscle Soreness (DOMS)
2.3. Range of Motion
2.4. Swelling (Circumference and Cross-Sectional Area)
2.5. Serum Cytokines and Muscle Damage Markers
2.6. Neuromuscular Damage
3. EPA and DHA for Muscle Mass and Function
3.1. Muscle Mass
3.2. Muscle Function
4. Summary and Future Directions
- Some positive effects of EPA and DHA have been observed on ECC-induced nerve and muscle damage (muscle strength deficit, DOMS, reduced ROM, and muscle swelling), while some results are not consistent.
- EPA and DHA may have positive effects on muscle mass under wasting condition, but it is unclear with regard to training.
- EPA and DHA have positive effects on muscle function, especially for neuromuscular adaptation.
Author Contributions
Conflicts of Interest
References
- Dyerberg, J.; Bang, H.O.; Hjorne, N. Fatty acid composition of the plasma lipids in greenland eskimos. Am. J. Clin. Nutr. 1975, 28, 958–966. [Google Scholar] [CrossRef] [PubMed]
- Yamori, Y.; Nara, Y.; Iritani, N.; Workman, R.J.; Inagami, T. Comparison of serum phospholipid fatty acids among fishing and farming japanese populations and american inlanders. J. Nutr. Sci. Vitaminol. (Tokyo) 1985, 31, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Paschos, G.K.; Magkos, F.; Panagiotakos, D.B.; Votteas, V.; Zampelas, A. Dietary supplementation with flaxseed oil lowers blood pressure in dyslipidaemic patients. Eur. J. Clin. Nutr. 2007, 61, 1201–1206. [Google Scholar] [CrossRef] [PubMed]
- Jiao, J.; Li, Q.; Chu, J.; Zeng, W.; Yang, M.; Zhu, S. Effect of n-3 pufa supplementation on cognitive function throughout the life span from infancy to old age: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2014, 100, 1422–1436. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Functional roles of fatty acids and their effects on human health. JPEN J. Parenter. Enter. Nutr. 2015, 39, 18s–32s. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J.; McDonald, K.; Caldarella, S.M.; Chung, H.Y.; Troen, A.M.; Snodderly, D.M. Cognitive findings of an exploratory trial of docosahexaenoic acid and lutein supplementation in older women. Nutr. Neurosci. 2008, 11, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Albert, C.M.; Campos, H.; Stampfer, M.J.; Ridker, P.M.; Manson, J.E.; Willett, W.C.; Ma, J. Blood levels of long-chain n-3 fatty acids and the risk of sudden death. N. Engl. J. Med. 2002, 346, 1113–1118. [Google Scholar] [CrossRef] [PubMed]
- Oomen, C.M.; Feskens, E.J.; Rasanen, L.; Fidanza, F.; Nissinen, A.M.; Menotti, A.; Kok, F.J.; Kromhout, D. Fish consumption and coronary heart disease mortality in finland, italy, and the netherlands. Am. J. Epidemiol. 2000, 151, 999–1006. [Google Scholar] [CrossRef] [PubMed]
- Buckley, J.D.; Burgess, S.; Murphy, K.J.; Howe, P.R. Dha-rich fish oil lowers heart rate during submaximal exercise in elite australian rules footballers. J. Sci. Med. Sport 2009, 12, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.A.; Beilin, L.J.; Burke, V.; Morris, J.; Ritchie, J. Interactions between dietary fat, fish, and fish oils and their effects on platelet function in men at risk of cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Macartney, M.J.; Hingley, L.; Brown, M.A.; Peoples, G.E.; McLennan, P.L. Intrinsic heart rate recovery after dynamic exercise is improved with an increased omega-3 index in healthy males. Br. J. Nutr. 2014, 112, 1984–1992. [Google Scholar] [CrossRef] [PubMed]
- Zebrowska, A.; Mizia-Stec, K.; Mizia, M.; Gasior, Z.; Poprzecki, S. Omega-3 fatty acids supplementation improves endothelial function and maximal oxygen uptake in endurance-trained athletes. Eur. J. Sport Sci. 2015, 15, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Ochi, E.; Tsuchiya, Y.; Nosaka, K. Differences in post-exercise t2 relaxation time changes between eccentric and concentric contractions of the elbow flexors. Eur. J. Appl. Physiol. 2016, 116, 2145–2154. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.J.; Qin, Z.; Wang, P.Y.; Sun, Y.; Liu, X. Muscle fatigue: General understanding and treatment. Exp. Mol. Med. 2017, 49, e384. [Google Scholar] [CrossRef] [PubMed]
- Capostagno, B.; Lambert, M.I.; Lamberts, R.P. Systematic review of submaximal cycle tests to predict, monitor and optimize cycling performance. Int. J. Sports Physiol. Perform. 2016. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, P.M.; Hubal, M.J. Exercise-induced muscle damage in humans. Am. J. Phys. Med. Rehabil. 2002, 81, S52–S69. [Google Scholar] [CrossRef] [PubMed]
- Dimauro, I.; Mercatelli, N.; Caporossi, D. Exercise-induced ros in heat shock proteins response. Free Radic. Biol. Med. 2016, 98, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.H.; Hoover, R.L.; Williams, J.D.; Sperling, R.I.; Ravalese, J., 3rd; Spur, B.W.; Robinson, D.R.; Corey, E.J.; Lewis, R.A.; Austen, K.F. Effect of dietary enrichment with eicosapentaenoic and docosahexaenoic acids on in vitro neutrophil and monocyte leukotriene generation and neutrophil function. N. Engl. J. Med. 1985, 312, 1217–1224. [Google Scholar] [CrossRef] [PubMed]
- Mickleborough, T.D. Omega-3 polyunsaturated fatty acids in physical performance optimization. Int. J. Sport Nutr. Exerc. Metab. 2013, 23, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, Y.; Yanagimoto, K.; Nakazato, K.; Hayamizu, K.; Ochi, E. Eicosapentaenoic and docosahexaenoic acids-rich fish oil supplementation attenuates strength loss and limited joint range of motion after eccentric contractions: A randomized, double-blind, placebo-controlled, parallel-group trial. Eur. J. Appl. Physiol. 2016, 116, 1179–1188. [Google Scholar] [CrossRef] [PubMed]
- Da Boit, M.; Hunter, A.M.; Gray, S.R. Fit with good fat? The role of n-3 polyunsaturated fatty acids on exercise performance. Metabolism 2017, 66, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, Y.; Kikuchi, N.; Shirato, M.; Ochi, E. Differences of activation pattern and damage in elbow flexor muscle after isokinetic eccentric contractions. Isokinet. Exerc. Sci. 2015, 23, 169–175. [Google Scholar] [CrossRef]
- Armstrong, R.B.; Warren, G.L.; Warren, J.A. Mechanisms of exercise-induced muscle fibre injury. Sports Med. 1991, 12, 184–207. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.; Nosaka, K.; Suzuki, K. Characterization of inflammatory responses to eccentric exercise in humans. Exerc. Immunol. Rev. 2005, 11, 64–85. [Google Scholar] [PubMed]
- Kouzaki, K.; Nosaka, K.; Ochi, E.; Nakazato, K. Increases in m-wave latency of biceps brachii after elbow flexor eccentric contractions in women. Eur. J. Appl. Physiol. 2016, 116, 939–946. [Google Scholar] [CrossRef] [PubMed]
- Bloomer, R.J.; Larson, D.E.; Fisher-Wellman, K.H.; Galpin, A.J.; Schilling, B.K. Effect of eicosapentaenoic and docosahexaenoic acid on resting and exercise-induced inflammatory and oxidative stress biomarkers: A randomized, placebo controlled, cross-over study. Lipids Health Dis. 2009, 8, 36. [Google Scholar] [CrossRef] [PubMed]
- Jouris, K.B.; McDaniel, J.L.; Weiss, E.P. The effect of omega-3 fatty acid supplementation on the inflammatory response to eccentric strength exercise. J. Sports Sci. Med. 2011, 10, 432–438. [Google Scholar] [PubMed]
- Lembke, P.; Capodice, J.; Hebert, K.; Swenson, T. Influence of omega-3 (n3) index on performance and wellbeing in young adults after heavy eccentric exercise. J. Sports Sci. Med. 2014, 13, 151–156. [Google Scholar] [PubMed]
- Ochi, E.; Tsuchiya, Y.; Yanagimoto, K. Effect of eicosapentaenoic acids-rich fish oil supplementation on motor nerve function after eccentric contractions. J. Int. Soc. Sports Nutr. 2017, 14, 23. [Google Scholar] [CrossRef] [PubMed]
- Phillips, T.; Childs, A.C.; Dreon, D.M.; Phinney, S.; Leeuwenburgh, C. A dietary supplement attenuates il-6 and crp after eccentric exercise in untrained males. Med. Sci. Sports Exerc. 2003, 35, 2032–2037. [Google Scholar] [CrossRef] [PubMed]
- Tartibian, B.; Maleki, B.H.; Abbasi, A. The effects of ingestion of omega-3 fatty acids on perceived pain and external symptoms of delayed onset muscle soreness in untrained men. Clin. J. Sport Med. 2009, 19, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Tartibian, B.; Maleki, B.H.; Abbasi, A. Omega-3 fatty acids supplementation attenuates inflammatory markers after eccentric exercise in untrained men. Clin. J. Sport Med. 2011, 21, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Tinsley, G.M.; Gann, J.J.; Huber, S.R.; Andre, T.L.; La Bounty, P.M.; Bowden, R.G.; Gordon, P.M.; Grandjean, P.W. Effects of fish oil supplementation on postresistance exercise muscle soreness. J. Diet. Suppl. 2016, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Houghton, D.; Onambele, G.L. Can a standard dose of eicosapentaenoic acid (epa) supplementation reduce the symptoms of delayed onset of muscle soreness? J. Int. Soc. Sports Nutr. 2012, 9, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DiLorenzo, F.M.; Drager, C.J.; Rankin, J.W. Docosahexaenoic acid affects markers of inflammation and muscle damage after eccentric exercise. J. Strength Cond. Res. 2014, 28, 2768–2774. [Google Scholar] [CrossRef] [PubMed]
- Lenn, J.; Uhl, T.; Mattacola, C.; Boissonneault, G.; Yates, J.; Ibrahim, W.; Bruckner, G. The effects of fish oil and isoflavones on delayed onset muscle soreness. Med. Sci. Sports Exerc. 2002, 34, 1605–1613. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, R.G.; James, M.J.; Gibson, R.A.; Edwards, J.R.; Stubberfield, J.; Stuklis, R.; Roberts-Thomson, K.; Young, G.D.; Cleland, L.G. Effects of fish-oil supplementation on myocardial fatty acids in humans. Am. J. Clin. Nutr. 2007, 85, 1222–1228. [Google Scholar] [CrossRef] [PubMed]
- Gray, P.; Chappell, A.; Jenkinson, A.M.; Thies, F.; Gray, S.R. Fish oil supplementation reduces markers of oxidative stress but not muscle soreness after eccentric exercise. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 206–214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jakeman, J.R.; Lambrick, D.M.; Wooley, B.; Babraj, J.A.; Faulkner, J.A. Effect of an acute dose of omega-3 fish oil following exercise-induced muscle damage. Eur. J. Appl. Physiol. 2017, 117, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Chleboun, G.S.; Howell, J.N.; Conatser, R.R.; Giesey, J.J. Relationship between muscle swelling and stiffness after eccentric exercise. Med. Sci. Sports Exerc. 1998, 30, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Kouzaki, K.; Kobayashi, M.; Nakamura, K.I.; Ohta, K.; Nakazato, K. Repeated bouts of fast eccentric contraction produce sciatic nerve damage in rats. Muscle Nerve 2016, 54, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Gerbi, A.; Maixent, J.M.; Ansaldi, J.L.; Pierlovisi, M.; Coste, T.; Pelissier, J.F.; Vague, P.; Raccah, D. Fish oil supplementation prevents diabetes-induced nerve conduction velocity and neuroanatomical changes in rats. J. Nutr. 1999, 129, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Ochi, E.; Hirose, T.; Hiranuma, K.; Min, S.K.; Ishii, N.; Nakazato, K. Elevation of myostatin and foxos in prolonged muscular impairment induced by eccentric contractions in rat medial gastrocnemius muscle. J. Appl. Physiol. (1985) 2010, 108, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Morton, R.W.; Murphy, K.T.; McKellar, S.R.; Schoenfeld, B.J.; Henselmans, M.; Helms, E.; Aragon, A.A.; Devries, M.C.; Banfield, L.; Krieger, J.W.; et al. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br. J. Sports Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M. The impact of protein quality on the promotion of resistance exercise-induced changes in muscle mass. Nutr. Metab. (Lond.) 2016, 13, 64. [Google Scholar] [CrossRef] [PubMed]
- McGlory, C.; Phillips, S.M. Exercise and the regulation of skeletal muscle hypertrophy. Prog. Mol. Biol. Transl. Sci. 2015, 135, 153–173. [Google Scholar] [CrossRef] [PubMed]
- Tanimoto, M.; Ishii, N. Effects of low-intensity resistance exercise with slow movement and tonic force generation on muscular function in young men. J. Appl. Physiol. (1985) 2006, 100, 1150–1157. [Google Scholar] [CrossRef] [PubMed]
- Madarame, H.; Neya, M.; Ochi, E.; Nakazato, K.; Sato, Y.; Ishii, N. Cross-transfer effects of resistance training with blood flow restriction. Med. Sci. Sports Exerc. 2008, 40, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Gingras, A.A.; White, P.J.; Chouinard, P.Y.; Julien, P.; Davis, T.A.; Dombrowski, L.; Couture, Y.; Dubreuil, P.; Myre, A.; Bergeron, K.; et al. Long-chain omega-3 fatty acids regulate bovine whole-body protein metabolism by promoting muscle insulin signalling to the akt-mtor-s6k1 pathway and insulin sensitivity. J. Physiol. 2007, 579, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.K.; Deng, Z.; Jiang, S.Z.; Song, T.X.; Zhou, Y.F.; Peng, J.; Tao, Y.X. Eicosapentaenoic acid abolishes inhibition of insulin-induced mtor phosphorylation by lps via ptp1b downregulation in skeletal muscle. Mol. Cell. Endocrinol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.K.; Zhou, Y.; Jiang, S.; Tao, Y.X.; Sun, H.; Peng, J.; Jiang, S. Feeding a dha-enriched diet increases skeletal muscle protein synthesis in growing pigs: Association with increased skeletal muscle insulin action and local mrna expression of insulin-like growth factor 1. Br. J. Nutr. 2013, 110, 671–680. [Google Scholar] [CrossRef] [PubMed]
- You, J.S.; Park, M.N.; Song, W.; Lee, Y.S. Dietary fish oil alleviates soleus atrophy during immobilization in association with akt signaling to p70s6k and e3 ubiquitin ligases in rats. Appl. Physiol. Nutr. Metab. 2010, 35, 310–318. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, S.C.; Hindi, S.M.; Kumar, A.; Marques, M.J. Effects of omega-3 on matrix metalloproteinase-9, myoblast transplantation and satellite cell activation in dystrophin-deficient muscle fibers. Cell Tissue Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.I.; Atherton, P.; Reeds, D.N.; Mohammed, B.S.; Rankin, D.; Rennie, M.J.; Mittendorfer, B. Omega-3 polyunsaturated fatty acids augment the muscle protein anabolic response to hyperinsulinaemia-hyperaminoacidaemia in healthy young and middle-aged men and women. Clin. Sci. (Lond.) 2011, 121, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.I.; Julliand, S.; Reeds, D.N.; Sinacore, D.R.; Klein, S.; Mittendorfer, B. Fish oil-derived n-3 pufa therapy increases muscle mass and function in healthy older adults. Am. J. Clin. Nutr. 2015, 102, 115–122. [Google Scholar] [CrossRef] [PubMed]
- McGlory, C.; Wardle, S.L.; Macnaughton, L.S.; Witard, O.C.; Scott, F.; Dick, J.; Bell, J.G.; Phillips, S.M.; Galloway, S.D.; Hamilton, D.L.; et al. Fish oil supplementation suppresses resistance exercise and feeding-induced increases in anabolic signaling without affecting myofibrillar protein synthesis in young men. Physiol. Rep. 2016, 4. [Google Scholar] [CrossRef] [PubMed]
- Da Boit, M.; Sibson, R.; Sivasubramaniam, S.; Meakin, J.R.; Greig, C.A.; Aspden, R.M.; Thies, F.; Jeromson, S.; Hamilton, D.L.; Speakman, J.R.; et al. Sex differences in the effect of fish-oil supplementation on the adaptive response to resistance exercise training in older people: A randomized controlled trial. Am. J. Clin. Nutr. 2017, 105, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.J.; Radonic, P.W.; Wolever, T.M.; Wells, G.D. 21 days of mammalian omega-3 fatty acid supplementation improves aspects of neuromuscular function and performance in male athletes compared to olive oil placebo. J. Int. Soc. Sports Nutr. 2015, 12, 28. [Google Scholar] [CrossRef] [PubMed]
- Rodacki, C.L.; Rodacki, A.L.; Pereira, G.; Naliwaiko, K.; Coelho, I.; Pequito, D.; Fernandes, L.C. Fish-oil supplementation enhances the effects of strength training in elderly women. Am. J. Clin. Nutr. 2012, 95, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Bourre, J.M. Nature, origin and role of fatty acids of the nervous system: An essential fatty acid, an alpha-linolenic acid, changing the structure and the cerebral function. Bull. Acad. Natl. Med. 1989, 173, 1137–1148, discussion 1148–1151. [Google Scholar] [PubMed]
- Lauritzen, L.; Hansen, H.S.; Jorgensen, M.H.; Michaelsen, K.F. The essentiality of long chain n-3 fatty acids in relation to development and function of the brain and retina. Prog. Lipid Res. 2001, 40, 1–94. [Google Scholar] [CrossRef]
- Salem, N., Jr.; Litman, B.; Kim, H.Y.; Gawrisch, K. Mechanisms of action of docosahexaenoic acid in the nervous system. Lipids 2001, 36, 945–959. [Google Scholar] [CrossRef] [PubMed]
- Solomon, S.A.; Cartwright, I.; Pockley, G.; Greaves, M.; Preston, F.E.; Ramsay, L.E.; Waller, P.C. A placebo-controlled, double-blind study of eicosapentaenoic acid-rich fish oil in patients with stable angina pectoris. Curr. Med. Res. Opin. 1990, 12, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Gans, R.O.; Bilo, H.J.; Weersink, E.G.; Rauwerda, J.A.; Fonk, T.; Popp-Snijders, C.; Donker, A.J. Fish oil supplementation in patients with stable claudication. Am. J. Surg. 1990, 160, 490–495. [Google Scholar] [CrossRef]
- U. S. Food and Drug Administration. Letter Regarding Dietary Supplement Health Claim for Omega-3 Fatty Acids and Coro-Nary Heart Disease. Docket No. 91 N-0103; 2000. Available online: https://pdfs.semanticscholar.org/5c90/42de155b3c273a5d70c7ed88acb7bb39b2b7.pdf (accessed on 14 March 2018).
- Simopoulos, A.P. Omega-3 fatty acids and athletics. Curr. Sports Med. Rep. 2007, 6, 230–236. [Google Scholar] [PubMed]
| Reference (year) | Population (Age) | Dose (Per Day) | Duration | Exercise | Outcome |
|---|---|---|---|---|---|
| Houghton and Onambele (2012) [34] | 17 healthy females (20.4 ± 2.3 years) | 0.36 g EPA | 3 weeks | Resistance exercise (leg flexions, leg extensions, straight leg dead lifts, walking lunges; 3 set of 10 repetitions at 70%1RM) | Ineffective |
| DiLorenzo et al. (2014) [35] | 41 healthy, untrained males (21.8 ± 2.7 years) | 2.0 g DHA | 4 weeks | Elbow flexor eccentric contractions (6 sets of 10 repetitions at 140%1RM using dumbbell) | Ineffective |
| Lenn et al. (2002) [36] | 13 males (22.7 ± 3.9 years) and 9 females (24.5 ± 5.5 years) | 0.287 g EPA and 0.194 g DHA | 30 days | Elbow flexor eccentric contractions (50 maximal effort at a 90 °/s using the Kin-Com dynamometer) | Ineffective |
| Gray et al. (2014) [38] | 20 healthy, untrained males (23.0 ± 2.3 years) | 1.30 g EPA and 0.30 g DHA | 6 weeks | Knee extensor eccentric contractions (20 sets of 10 repetitions at a 0.52 rads/s using the Biodex isokinetic dynamometer) | Ineffective |
| Tsuchiya et al. (2016) [20] | 24 healthy, untrained males (19.5 ± 0.8 years) | 0.60 g EPA and 0.26 g DHA | 8 weeks | Elbow flexor eccentric contractions (6 sets of maximal 5 repetitions at a 30 °/s using the Biodex isokinetic dynamometer) | Effective |
| Ochi et al. (2017) [29] | 21 healthy, untrained males (21.0 ± 0.8 years) | 0.60 g EPA and 0.26 g DHA | 8 weeks | Elbow flexor eccentric contractions (6 sets of 10 repetitions at 40%1RM, 30 °/s using dumbbell) | Effective |
| Reference (Year) | Population (Age) | Dose (Per Day) | Duration | Exercise | Outcome |
|---|---|---|---|---|---|
| Houghton and Onambele (2012) [34] | 17 healthy females (20.4 ± 2.3 years) | 0.36 g EPA | 3 weeks | Resistance exercise (leg flexions, leg extensions, straight leg dead lifts, walking lunges; 3 set of 10 repetitions at 70%1RM) | Ineffective |
| Lembke et al. (2014) [28] | 64 healthy, untrained males and females (over the age of 18 years) | 2.70 g EPA and DHA | 30 days | Elbow flexor eccentric contractions (2 sets of 30 maximal efforts using the Cybex isokinetic dynamometer) | Effective |
| DiLorenzo et al. (2014) [35] | 41 healthy, untrained males (21.8 ± 2.7 years) | 2.0 g DHA | 4 weeks | Elbow flexor eccentric contractions (6 sets of 10 repetitions at 140%1RM using dumbbell) | Ineffective |
| Lenn et al. (2002) [36] | 13 males (22.7 ± 3.9 years) and 9 females (24.5 ± 5.5 years) | 0.287 g EPA and 0.194 g DHA | 30 days | Elbow flexor eccentric contractions (50 maximal efforts at a 90 °/s using the Kin-Com dynamometer) | Ineffective |
| Gray et al. (2014) [38] | 20 healthy, untrained males (23.0 ± 2.3 years) | 1.30 g EPA and 0.30 g DHA | 6 weeks | Knee extensor eccentric contractions (20 sets of 10 repetitions at a 0.52 rads/s using the Biodex isokinetic dynamometer) | Ineffective |
| Tartibian et al. (2009) [31] | 27 healthy males (33.4 ± 4.2 years) | 0.324 g EPA and 0.216 g DHA | 30 days | 40-minite bench stepping (knee height step-50 cm on average-at a rate of 15 steps per minute) | Effective |
| Jouris et al. (2011) [27] | 3 males and 8 females (18 to 60 years) | 2.0 g EPA and 1.0 g DHA | 2 weeks | Elbow flexor eccentric contractions (2 sets to failure at 120%1RM using dumbbell) | Effective |
| Tinsley et al. (2016) [33] | 19 healthy, untrained females (22.5 ± 1.8 years) | 3.60 g EPA and DHA | 2 weeks | Elbow flexor and leg extensor eccentric contractions (10 sets to failure at 50%1RM using the elbow flexions and leg extensions machines) | Effective |
| Tsuchiya et al. (2016) [20] | 24 healthy, untrained males (19.5 ± 0.8 years) | 0.60 g EPA and 0.26 g DHA | 8 weeks | Elbow flexor eccentric contractions (6 sets of maximal 5 repetitions at a 30 °/s using the Biodex isokinetic dynamometer) | Effective |
| Ochi et al. (2017) [29] | 21 healthy, untrained males (21.0 ± 0.8 years) | 0.60 g EPA and 0.26 g DHA | 8 weeks | Elbow flexor eccentric contractions (6 sets of 10 repetitions at 40%1RM, 30 °/s using dumbbell) | Effective |
| Phillips et al. (2003) [30] | 40 healthy, untrained males (18–35 years) | 0.80 g DHA | 2 weeks | Elbow flexor eccentric contractions (3 sets of 10 repetitions using 80% using the arm curl machine) | Ineffectiv |
| Bloomer et al. (2009) [26] | 14 recreational males (25.5 ± 4.8 years) | 2.224 g EPA and 2.208 g DHA | 6 weeks | 60-min treadmill climb using a weighted pack (weight equal to 25% of body mass) | Ineffective |
| Reference (Year) | Population (Age) | Dose (Per Day) | Duration | Exercise | Outcome |
|---|---|---|---|---|---|
| Lembke et al. (2014) [28] | 64 healthy, untrained males and females (over the age of 18 years) | 2.70 g EPA and DHA | 30 days | Elbow flexor eccentric contractions (2 sets of 30 maximal efforts using the Cybex isokinetic dynamometer) | Ineffective |
| Lenn et al. (2002) [36] | 13 males (22.7 ± 3.9 years) and 9 females (24.5 ± 5.5 years) | 0.287 g EPA and 0.194 g DHA | 30 days | Elbow flexor eccentric contractions (50 maximal efforts at a 90 °/s using the Kin-Com dynamometer) | Ineffective |
| Tartibian et al. (2009) [31] | 27 healthy males (33.4 ± 4.2 years) | 0.324 g EPA and 0.216 g DHA | 30 days | 40-min bench stepping (knee height step-50 cm on average-at a rate of 15 steps per minute) | Effective |
| Tsuchiya et al. (2016) [20] | 24 healthy, untrained males (19.5 ± 0.8 years) | 0.60 g EPA and 0.26 g DHA | 8 weeks | Elbow flexor eccentric contractions (6 sets of maximal 5 repetitions at a 30 °/s using the Biodex isokinetic dynamometer) | Effective |
| Ochi et al. (2017) [29] | 21 healthy, untrained males (21.0 ± 0.8 years) | 0.60 g EPA and 0.26 g DHA | 8 weeks | Elbow flexor eccentric contractions (6 sets of 10 repetitions at 40%1RM, 30 °/s using dumbbell) | Effective |
| Reference (Year) | Population (Age) | Dose (Per Day) | Duration | Exercise | Outcome |
|---|---|---|---|---|---|
| Tartibian et al. (2009) [31] | 27 healthy males (33.4 ± 4.2 years) | 0.324 g EPA and 0.216 g DHA | 30 days | 40-minute bench stepping (knee height step-50 cm on average-at a rate of 15 steps per minute) | Circumference; Effective |
| Jouris et al. (2011) [27] | 3 males and 8 females (18 to 60 years) | 0.20 g EPA and 0.10 g DHA | 2 weeks | Elbow flexor eccentric contractions (2 sets to failure at 120%1RM using dumbbell) | Circumference; Ineffective |
| Tinsley et al. (2016) [33] | 19 healthy, untrained females (22.5 ± 1.8 years) | 3.60 g EPA and DHA | 2 weeks | Elbow flexor and leg extensor eccentric contractions (10 sets to failure at 50%1RM using the elbow flexion and leg extension machines) | Circumference; Ineffective |
| Tsuchiya et al. (2016) [20] | 24 healthy, untrained males (19.5 ± 0.8 years) | 0.60 g EPA and 0.26 g DHA | 8 weeks | Elbow flexor eccentric contractions (6 sets of maximal 5 repetitions at a 30 °/s using the Biodex isokinetic dynamometer) | Circumference; Ineffective |
| Ochi et al. (2017) [29] | 21 healthy, untrained males (21.0 ± 0.8 years) | 0.60 g EPA and 0.26 g DHA | 8 weeks | Elbow flexor eccentric contractions (6 sets of 10 repetitions at 40%1RM, 30 °/s using dumbbell) | Circumference; Ineffective Cross-sectional area; ineffective |
| Reference (Year) | Population (Age) | Dose (Per Day) | Duration | Exercise | Outcome |
|---|---|---|---|---|---|
| Houghton and Onambele (2012) [34] | 17 healthy females (20.4 ± 2.3 years) | 0.36 g EPA | 3 weeks | Resistance exercise (leg flexions, leg extensions, straight leg dead lifts, walking lunges; 3 set of 10 repetitions at 70%1RM) | CK; Ineffective IL-6; Effective |
| DiLorenzo et al. (2014) [35] | 41 healthy, untrained males (21.8 ± 2.7 years) | 2.0 g DHA | 4 weeks | Elbow flexor eccentric contractions (6 sets of 10 repetitions at 140%1RM using dumbbell) | CK; Effective IL-6; Effective |
| Gray et al. (2014) [38] | 20 healthy, untrained males (23.0 ± 2.3 years) | 1.30 g EPA and 0.30 g DHA | 6 weeks | Knee extensor eccentric contractions (20 sets of 10 repetitions at a 0.52 rads/s using the Biodex isokinetic dynamometer) | CK; Ineffective |
| Tartibian et al. (2011) [32] | 45 healthy, untrained males (29.7 ± 6.6 years) | 0.324 g EPA and 0.216 g DHA | 30 days | 40-min bench stepping (knee height step-50 cm on average-at a rate of 15 steps per minute) | CK; Effective Mb; Effective IL-6; Effective TNF-α; Effective |
| Jakeman et al. (2017) [39] | 27 physically active males (26 ± 4 years) | High EPA group; EPA 0.75 g, DHA 0.05 g Low EPA group; EPA 0.15 g, DHA 0.10 g | One dose upon completion of the plyometric protocol | 10 sets of 10 repetitions of squat jump performance and countermovement jump performance | CK; Ineffective IL-6; Ineffective |
| Tsuchiya et al. (2016) [20] | 24 healthy, untrained males (19.5 ± 0.8 years) | 0.60 g EPA and 0.26 g DHA | 8 weeks | Elbow flexor eccentric contractions (6 sets of maximal 5 repetitions at a 30 °/s using the Biodex isokinetic dynamometer) | CK; Ineffective Mb; Ineffective IL-6; Effective TNF-α; Ineffective |
| Phillips et al. (2003) [30] | 40 healthy, untrained males (18–35 years) | 0.80 g DHA | 2 weeks | Elbow flexor eccentric contractions (3 sets of 10 repetitions using 80% using the arm curl machine) | CK; Ineffective IL-6; Effective |
| Bloomer et al. (2009) [26] | 14 recreational males (25.5 ± 4.8 years) | 2.224 g EPA and 2.208 g DHA | 6 weeks | 60-min treadmill climb using a weighted pack (weight equal to 25% of body mass) | CK; Ineffective TNF-α; Effective |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ochi, E.; Tsuchiya, Y. Eicosapentaenoic Acid (EPA) and Docosahexaneoic Acid (DHA) in Muscle Damage and Function. Nutrients 2018, 10, 552. https://doi.org/10.3390/nu10050552
Ochi E, Tsuchiya Y. Eicosapentaenoic Acid (EPA) and Docosahexaneoic Acid (DHA) in Muscle Damage and Function. Nutrients. 2018; 10(5):552. https://doi.org/10.3390/nu10050552
Chicago/Turabian StyleOchi, Eisuke, and Yosuke Tsuchiya. 2018. "Eicosapentaenoic Acid (EPA) and Docosahexaneoic Acid (DHA) in Muscle Damage and Function" Nutrients 10, no. 5: 552. https://doi.org/10.3390/nu10050552
APA StyleOchi, E., & Tsuchiya, Y. (2018). Eicosapentaenoic Acid (EPA) and Docosahexaneoic Acid (DHA) in Muscle Damage and Function. Nutrients, 10(5), 552. https://doi.org/10.3390/nu10050552





