The Effect of Vitamins on Glaucoma: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
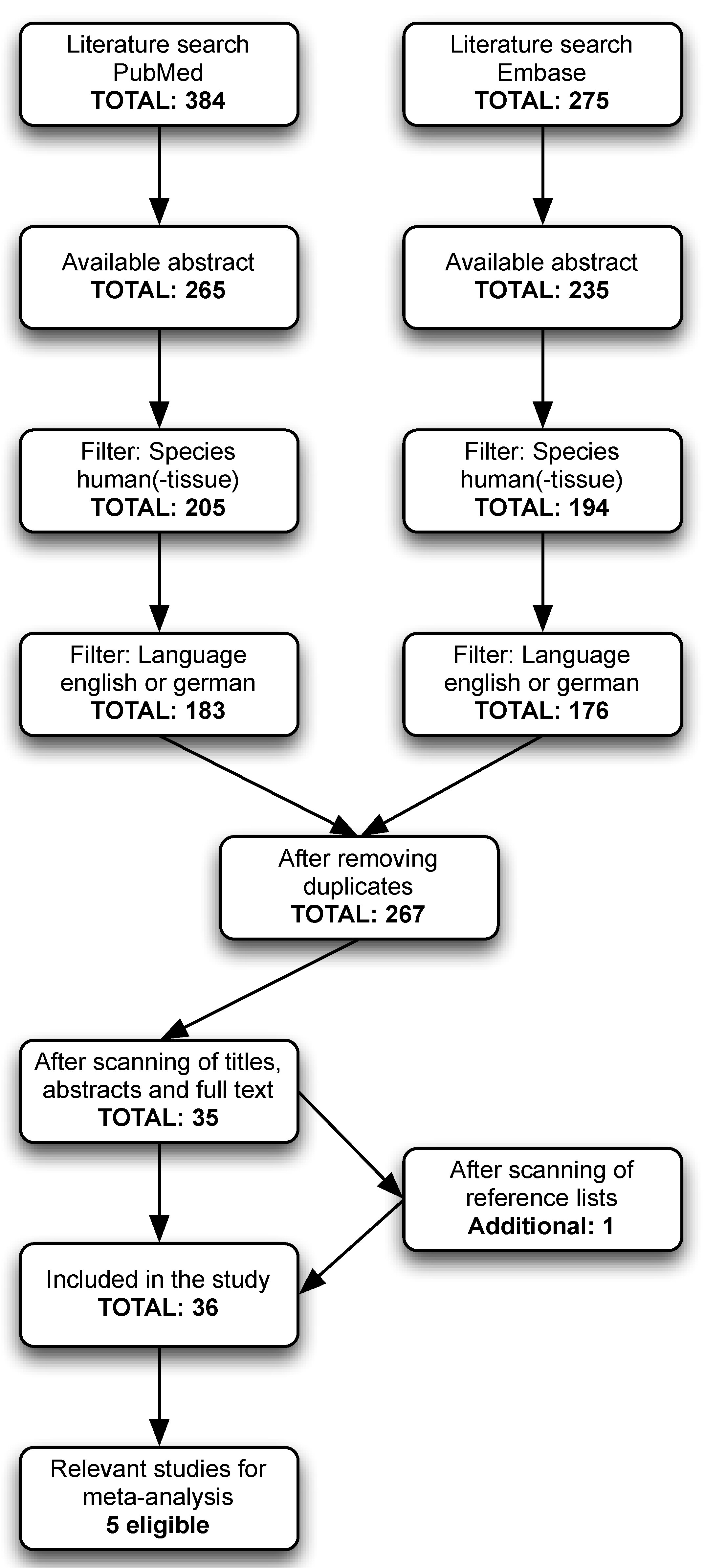
2.1. Search Strategy and Study Eligibility
2.2. Data Extraction and Quality Assessment
2.3. Data Synthesis and Analysis
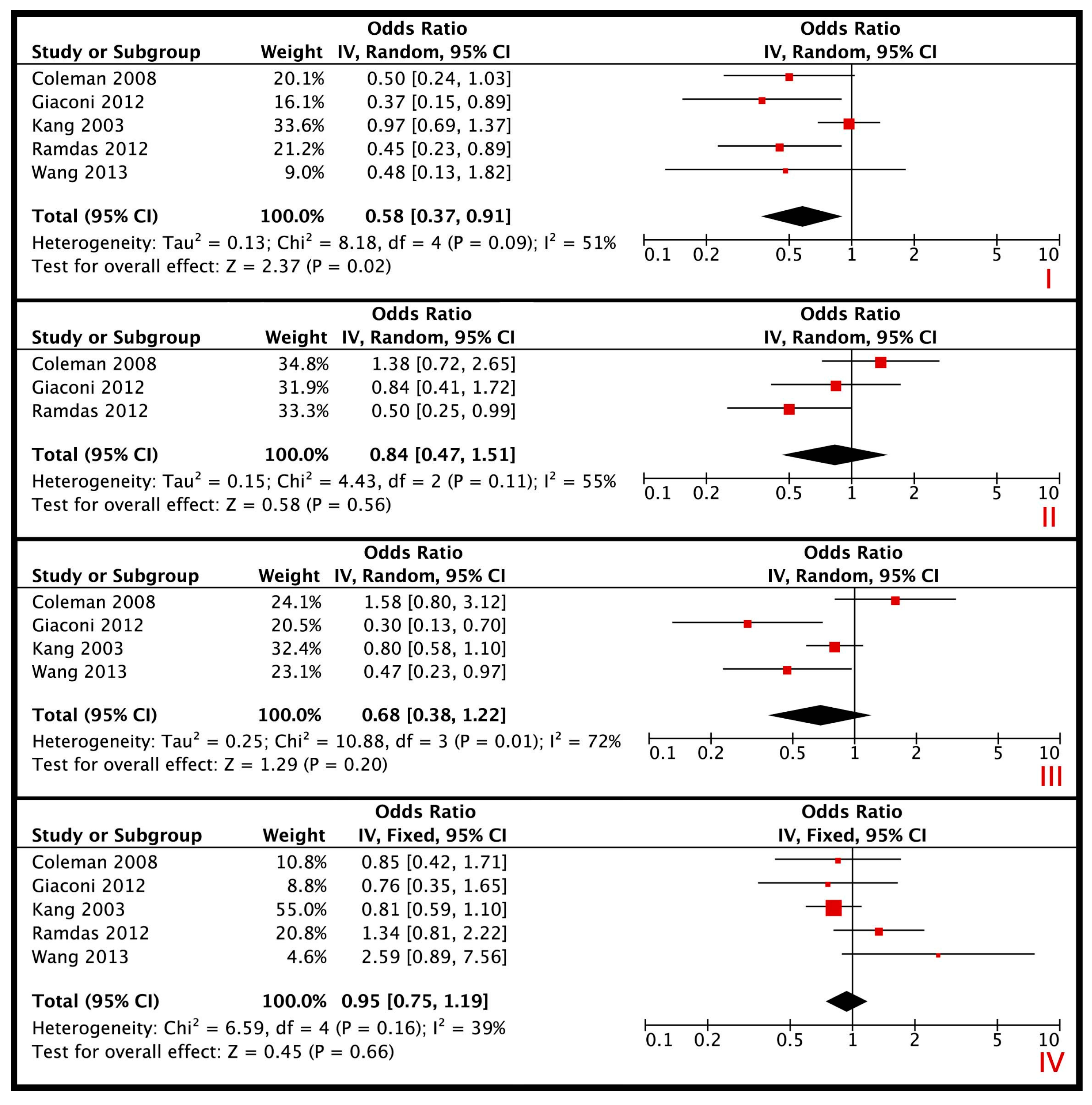
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Ramdas, W.D.; Wolfs, R.C.W.; Kiefte-de Jong, J.C.; Hofman, A.; de Jong, P.T.V.M.; Vingerling, J.R.; Jansonius, N.M. Nutrient intake and risk of open-angle glaucoma: The Rotterdam Study. Eur. J. Epidemiol. 2012, 27, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Wolfs, R.C.; Borger, P.H.; Ramrattan, R.S.; Klaver, C.C.; Hulsman, C.A.; Hofman, A.; Vingerling, J.R.; Hitchings, R.A.; de Jong, P.T. Changing views on open-angle glaucoma: Definitions and prevalences—The Rotterdam Study. Investig. Ophthalmol. Vis. Sci. 2000, 41, 3309–3321. [Google Scholar]
- Tham, Y.-C.; Li, X.; Wong, T.Y.; Quigley, H.A.; Aung, T.; Cheng, C.-Y. Global prevalence of glaucoma and projections of glaucoma burden through 2040: A systematic review and meta-analysis. Ophthalmology 2014, 121, 2081–2090. [Google Scholar] [CrossRef] [PubMed]
- Izzotti, A.; Saccà, S.C.; Cartiglia, C.; De Flora, S. Oxidative deoxyribonucleic acid damage in the eyes of glaucoma patients. Am. J. Med. 2003, 114, 638–646. [Google Scholar] [CrossRef]
- Saccà, S.C.; Pascotto, A.; Camicione, P.; Capris, P.; Izzotti, A. Oxidative DNA damage in the human trabecular meshwork: Clinical correlation in patients with primary open-angle glaucoma. Arch. Ophthalmol. 2005, 123, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Izzotti, A.; Bagnis, A.; Saccà, S.C. The role of oxidative stress in glaucoma. Mutat. Res. 2006, 612, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Nucci, C.; Russo, R.; Martucci, A.; Giannini, C.; Garaci, F.; Floris, R.; Bagetta, G.; Morrone, L.A. New strategies for neuroprotection in glaucoma, a disease that affects the central nervous system. Eur. J. Pharmacol. 2016, 787, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Tanito, M.; Kaidzu, S.; Takai, Y.; Ohira, A. Association between systemic oxidative stress and visual field damage in open-angle glaucoma. Sci. Rep. 2016, 6, 25792. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, S.; Tatt, I.D.; Higgins, J.P.T. Tools for assessing quality and susceptibility to bias in observational studies in epidemiology: A systematic review and annotated bibliography. Int. J. Epidemiol. 2007, 36, 666–676. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 17 January 2017).
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Tobias, A. Assessing the influence of a single study in the meta-analysis estimate. Stata Tech. Bull. 1999, 8, 15–17. [Google Scholar]
- Higgins, J.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions. Available online: http://handbook.cochrane.org (accessed on 13 April 2017).
- Zhang, J.; Yu, K.F. What’s the relative risk? A method of correcting the odds ratio in cohort studies of common outcomes. JAMA 1998, 280, 1690–1691. [Google Scholar] [CrossRef] [PubMed]
- Belmonte, A.; Tormo, C.; Lopez, N.; Villalba, C.; Fernandez, C.; Hernandez, F. Vitamins A, E, B12 and folate levels in different types of glaucoma. Clin. Chem. Lab. Med. 2011, 49, S816. [Google Scholar]
- Engin, K.N.; Yemişci, B.; Yiğit, U.; Ağaçhan, A.; Coşkun, C. Variability of serum oxidative stress biomarkers relative to biochemical data and clinical parameters of glaucoma patients. Mol. Vis. 2010, 16, 1260–1271. [Google Scholar] [PubMed]
- Yuki, K.; Murat, D.; Kimura, I.; Ohtake, Y.; Tsubota, K. Reduced-serum vitamin C and increased uric acid levels in normal-tension glaucoma. Graefes Arch. Clin. Exp. Ophthalmol. 2010, 248, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Riquelme, N.; Villalba, C.; Tormo, C.; Belmonte, A.; Fernandez, C.; Torralba, G.; Hernandez, F. Endothelin-1 levels and biomarkers of oxidative stress in glaucoma patients. Int. Ophthalmol. 2014, 35, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Singh, K.; Lin, S.C. Glaucoma and vitamins A, C, and E supplement intake and serum levels in a population-based sample of the United States. Eye 2013, 27, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Giaconi, J.A.; Yu, F.; Stone, K.L.; Pedula, K.L.; Ensrud, K.E.; Cauley, J.A.; Hochberg, M.C.; Coleman, A.L. The association of consumption of fruits/vegetables with decreased risk of glaucoma among older African-American women in the study of osteoporotic fractures. Am. J. Ophthalmol. 2012, 154, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Coleman, A.L.; Stone, K.L.; Kodjebacheva, G.; Yu, F.; Pedula, K.L.; Ensrud, K.E.; Cauley, J.A.; Hochberg, M.C.; Topouzis, F.; Badala, F.; et al. Glaucoma Risk and the Consumption of Fruits and Vegetables Among Older Women in the Study of Osteoporotic Fractures. Am. J. Ophthalmol. 2008, 145, 1081–1089. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Pasquale, L.R.; Willett, W.; Rosner, B.; Egan, K.M.; Faberowski, N.; Hankinson, S.E. Antioxidant intake and primary open-angle glaucoma: A prospective study. Am. J. Epidemiol. 2003, 158, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Asregadoo, E.R. Blood levels of thiamine and ascorbic acid in chronic open-angle glaucoma. Ann. Ophthalmol. 1979, 11, 1095–1100. [Google Scholar] [PubMed]
- Coban, D.T.; Ariturk, N.; Elmacioglu, F.; Ulus, C.A. The relationship between glaucoma and serum vitamin B12, folic acid levels and nutrition. Acta Med. Mediterr. 2015, 31, 281–286. [Google Scholar]
- Cumurcu, T.; Sahin, S.; Aydin, E. Serum homocysteine, vitamin B 12 and folic acid levels in different types of glaucoma. BMC Ophthalmol. 2006, 6, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Zhao, X.; Zeng, S.-M.; Li, L.; Zhong, H.-B.; Li, M. Homocysteine, B vitamins, methylenetetrahydrofolate reductase gene, and risk of primary open-angle glaucoma: A meta-analysis. Ophthalmology 2012, 119, 2493–2499. [Google Scholar] [CrossRef] [PubMed]
- Roedl, J.B.; Bleich, S.; Reulbach, U.; von Ahsen, N.; Schlötzer-Schrehardt, U.; Rejdak, R.; Naumann, G.O.H.; Kruse, F.E.; Kornhuber, J.; Jünemann, A.G.M. Homocysteine levels in aqueous humor and plasma of patients with primary open-angle glaucoma. J. Neural Transm. 2007, 114, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xu, F.; Zeng, R.; Gong, H.; Lan, Y. Plasma Homocysteine, Serum Folic Acid, Serum Vitamin B12, Serum Vitamin B6, MTHFR, and Risk of Normal-Tension Glaucoma. J. Glaucoma 2016, 25, e94–e98. [Google Scholar] [CrossRef] [PubMed]
- Leibovitzh, H.; Cohen, E.; Levi, A.; Kramer, M.; Shochat, T.; Goldberg, E.; Krause, I. Relationship between homocysteine and intraocular pressure in men and women: A population-based study. Medicine 2016, 95, e4858. [Google Scholar] [CrossRef] [PubMed]
- Rössler, C.W.; Baleanu, D.; Reulbach, U.; Lewczuk, P.; Bleich, S.; Kruse, F.E.; Kornhuber, J.; Schlötzer-Schrehardt, U.; Juenemann, A.G.M. Plasma homocysteine levels in patients with normal tension glaucoma. J. Glaucoma 2010, 19, 576–580. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Zhang, L.; Li, M. Plasma homocysteine, serum folic acid, serum vitamin B12, serum vitamin B6, MTHFR and risk of pseudoexfoliation glaucoma: A meta-analysis. Graefes Arch. Clin. Exp. Ophthalmol. 2012, 250, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Roedl, J.B.; Bleich, S.; Reulbach, U.; Rejdak, R.; Naumann, G.O.H.; Kruse, F.E.; Schlötzer-Schrehardt, U.; Kornhuber, J.; Jünemann, A.G.M. Vitamin deficiency and hyperhomocysteinemia in pseudoexfoliation glaucoma. J. Neural Transm. 2007, 114, 571–575. [Google Scholar] [CrossRef] [PubMed]
- Turgut, B.; Kaya, M.; Arslan, S.; Demir, T.; Güler, M.; Kaya, M.K. Levels of circulating homocysteine, vitamin B6, vitamin B12, and folate in different types of open-angle glaucoma. Clin. Interv. Aging 2010, 5, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Loomis, S.J.; Wiggs, J.L.; Willett, W.C.; Pasquale, L.R. A prospective study of folate, vitamin B6, and vitamin B12 intake in relation to exfoliation glaucoma or suspected exfoliation glaucoma. JAMA Ophthalmol. 2014, 132, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Türkyılmaz, K.; Öner, V.; Türkyılmaz, A.K.; Kırbaş, A.; Kırbaş, S.; Şekeryapan, B. Evaluation of peripapillary retinal nerve fiber layer thickness in patients with vitamin B12 deficiency using spectral domain optical coherence tomography. Curr. Eye Res. 2013, 38, 680–684. [Google Scholar] [CrossRef] [PubMed]
- Zanon-Moreno, V.; Asensio-Marquez, E.M.; Ciancotti-Oliver, L.; Garcia-Medina, J.J.; Sanz, P.; Ortega-Azorin, C.; Pinazo-Duran, M.D.; Ordovás, J.M.; Corella, D. Effects of polymorphisms in vitamin E-, vitamin C-, and glutathione peroxidase-related genes on serum biomarkers and associations with glaucoma. Mol. Vis. 2013, 19, 231–242. [Google Scholar] [PubMed]
- Lee, P.-F.; Lam, K.W.; Lai, M.-M. Aqueous humor ascorbate concentration and open-angle glaucoma. Arch. Ophthalmol. 1977, 95, 308–310. [Google Scholar] [CrossRef] [PubMed]
- Aleksidze, A.T.; Beradze, I.N.; Golovachev, O.G. Effect of the ascorbic acid of the aqueous humor on the lipid peroxidation process in the eye in primary open-angle glaucoma. Oftalmol. Zh. 1989, 2, 114–116. [Google Scholar]
- Goyal, A.; Srivastava, A.; Sihota, R.; Kaur, J. Evaluation of oxidative stress markers in aqueous humor of primary open angle glaucoma and primary angle closure glaucoma patients. Curr. Eye Res. 2014, 39, 823–829. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, Z.; Midgley, J.M.; Watson, D.G.; Karditsas, S.D.; Dutton, G.N.; Wilson, W.S. Effect of oral administration of vitamin C on human aqueous humor ascorbate concentration. Acta Pharmacol. Sin. 1999, 20, 879–883. [Google Scholar]
- Moïse, M.M.; Benjamin, L.-M.; Doris, T.M.; Dalida, K.N.; Augustin, N.O. Role of Mediterranean diet, tropical vegetables rich in antioxidants, and sunlight exposure in blindness, cataract and glaucoma among African type 2 diabetics. Int. J. Ophthalmol. 2012, 5, 231–237. [Google Scholar] [PubMed]
- Kang, J.H.; Willett, W.C.; Rosner, B.A.; Buys, E.; Wiggs, J.L.; Pasquale, L.R. Association of Dietary Nitrate Intake With Primary Open-Angle Glaucoma: A Prospective Analysis From the Nurses’ Health Study and Health Professionals Follow-up Study. JAMA Ophthalmol. 2016, 134, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, A.; Milea, D.; Gohier, P.; Jallet, G.; Leruez, S.; Baskaran, M.; Aung, T.; Annweiler, C. Serum Vitamin D status is associated with the presence but not the severity of primary open angle glaucoma. Maturitas 2015, 81, 470–474. [Google Scholar] [CrossRef] [PubMed]
- Yoo, T.K.; Oh, E.; Hong, S. Is vitamin D status associated with open-angle glaucoma? A cross-sectional study from South Korea. Public Health Nutr. 2014, 17, 833–843. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Yao, Q.; Ma, W.; Liu, H.; Ji, J.; Li, X. Associations of vitamin D deficiency and vitamin D receptor (Cdx-2, Fok I, Bsm I and Taq I) polymorphisms with the risk of primary open-angle glaucoma. BMC Ophthalmol. 2016, 16, 116. [Google Scholar] [CrossRef] [PubMed]
- Arar, Ž.V.; Praveček, M.K.; Miškić, B.; Vatavuk, Z.; Rodriguez, J.V.; Sekelj, S. Association between serum vitamin D level and glaucoma in women. Acta Clin. Croat. 2016, 55, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Gye, H.J.; Kim, J.M.; Yoo, C.; Shim, S.H.; Won, Y.S.; Sung, K.C.; Lee, M.Y.; Park, K.H. Relationship between high serum ferritin level and glaucoma in a South Korean population: The Kangbuk Samsung health study. Br. J. Ophthalmol. 2016, 100, 1703–1707. [Google Scholar] [CrossRef] [PubMed]
- Krefting, E.A.; Jorde, R.; Christoffersen, T.; Grimnes, G. Vitamin D and intraocular pressure—Results from a case-control and an intervention study. Acta Ophthalmol. 2014, 92, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Burgess, L.G.; Uppal, K.; Walker, D.I.; Roberson, R.M.; Tran, V.; Parks, M.B.; Wade, E.A.; May, A.T.; Umfress, A.C.; Jarrell, K.L.; et al. Metabolome-Wide Association Study of Primary Open Angle Glaucoma. Investig. Ophthalmol. Vis. Sci. 2015, 56, 5020–5028. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.; Magulike, N.; Ghebremeskel, K.; Crawford, M. Primary open-angle glaucoma patients have reduced levels of blood docosahexaenoic and eicosapentaenoic acids. Prostaglandins Leukot. Essent. Fat. Acids 2006, 74, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Mares, J.A. High-dose antioxidant supplementation and cataract risk. Nutr. Rev. 2004, 62, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Evans, J. Antioxidant supplements to prevent or slow down the progression of AMD: A systematic review and meta-analysis. Eye 2008, 22, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.A.; Harder, J.M.; John, S.W.M. Glaucoma as a Metabolic Optic Neuropathy: Making the Case for Nicotinamide Treatment in Glaucoma. J. Glaucoma 2017, 26, 1161–1168. [Google Scholar] [CrossRef] [PubMed]
- Engin, K.N.; Engin, G.; Kucuksahin, H.; Oncu, M.; Engin, G.; Guvener, B. Clinical evaluation of the neuroprotective effect of alpha-tocopherol against glaucomatous damage. Eur. J. Ophthalmol. 2007, 17, 528–533. [Google Scholar] [CrossRef] [PubMed]
- Saccà, S.C.; Gandolfi, S.; Bagnis, A.; Manni, G.; Damonte, G.; Traverso, C.E.; Izzotti, A. The Outflow Pathway: A Tissue with Morphological and Functional Unity. J. Cell. Physiol. 2016, 231, 1876–1893. [Google Scholar] [CrossRef] [PubMed]
- Himori, N.; Kunikata, H.; Shiga, Y.; Omodaka, K.; Maruyama, K.; Takahashi, H.; Nakazawa, T. The association between systemic oxidative stress and ocular blood flow in patients with normal-tension glaucoma. Graefes Arch. Clin. Exp. Ophthalmol. 2016, 254, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Nathanson, J.A.; McKee, M. Alterations of ocular nitric oxide synthase in human glaucoma. Investig. Ophthalmol. Vis. Sci. 1995, 36, 1774–1784. [Google Scholar]
- Cui, X.; Xiang, J.; Zhu, W.; Wei, A.; Le, Q.; Xu, J.; Zhou, X. Vitamin A Palmitate and Carbomer Gel Protects the Conjunctiva of Patients with Long-term Prostaglandin Analogs Application. J. Glaucoma 2016, 25, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Jenab, M.; Slimani, N.; Bictash, M.; Ferrari, P.; Bingham, S.A. Biomarkers in nutritional epidemiology: Applications, needs and new horizons. Hum. Genet. 2009, 125, 507–525. [Google Scholar] [CrossRef] [PubMed]
- Zanon-Moreno, V.; Ciancotti-Olivares, L.; Asencio, J.; Sanz, P.; Ortega-Azorin, C.; Pinazo-Duran, M.D.; Corella, D. Association between a SLC23A2 gene variation, plasma vitamin C levels, and risk of glaucoma in a Mediterranean population. Mol. Vis. 2011, 17, 2997–3004. [Google Scholar] [PubMed]
- Dehghan, M.; Akhtar-Danesh, N.; McMillan, C.R.; Thabane, L. Is plasma vitamin C an appropriate biomarker of vitamin C intake? A systematic review and meta-analysis. Nutr. J. 2007, 6, 41. [Google Scholar] [CrossRef] [PubMed]
- Quigley, H.A. Number of people with glaucoma worldwide. Br. J. Ophthalmol. 1996, 80, 389–393. [Google Scholar] [CrossRef] [PubMed]
| Vitamin | Vitamer Chemical Name | Remarks | Relevance to the Eye | Main Sources | Rich Food Sources |
|---|---|---|---|---|---|
| A | Retinol (A1) | Preformed retinols (or active form)—is immediately available to the body | Important for rhodopsin found in the retina for visual phototransduction, and protects against free radicals i.e., acts as an antioxidant | Animal products | Liver, eggs, cheese, butter, dark green vegetables (spinach) and carrots |
| Precursors (or provitamins)—has to be metabolized into an active form | Important for rhodopsin found in the retina for visual phototransduction, and protects against free radicals i.e., acts as an antioxidant | Plant products | Vegetables and fruits with orange pigments (known as carotenoids) | ||
| B-complex | Thiamin (B1) | Some flavonoids may antagonize vitamin B1 | Serve as coenzymes in catabolism of amino acids, carboxylase, cell division and growth, and DNA synthesis and repair in every cell of the body | Animal/plant products | Yeast extract, cereal grains (especially in the outer layer of the grain and in the germ), meat (pork), nuts, peas, and beans |
| Riboflavin (B2) | Primary coenzyme form of vitamin B6 | Used in the treatment of corneal ectasia for strengthening of corneal collagen tissue. It can also be used as a prophylaxis for migraine: a potential risk factor for glaucoma | Animal/plant products | Milk, cheese, eggs, green leafy vegetables, almonds, and mushrooms | |
| Niacin (B3) | May be converted to nicotinamide, which is another form of vitamin B3 | Involved in vasodilatation of among others retinal arterioles. It improves endothelial dysfunction | Animal/plant products | Fish, dates, nuts, and cereal grains | |
| Pyridoxin (B6) | Dietary vitamin B6 cannot be used without vitamin B2 (Riboflavin) | Involved in neurotransmitter synthesis. Plays a role in the treatment of gyrate atrophy of the choroid and retina | Animal/plant products | Meat (pork), bananas, chickpeas, potatoes, and pistachios | |
| Folate (B9) | Synthetic form, used in supplements and food processing, is called Folic acid | When combined with vitamin B12 it reduces homocysteine levels, which induces vascular injury, alterations in extracellular matrix remodeling, and neuronal cell death | Plant products | Cereal grains (especially in the outer layer of the grain and in the germ), dark green leafy vegetables, nuts, peas, and beans | |
| Cobalamin (B12) | Should be in balance with folic acid and iron | Coenzyme involved in the metabolism of every cell of the body. Deficiency can results in elevated homocysteine levels, optic neuropathy, and irreversible damage to nervous system | Animal products | Fish, red meat, eggs, and cheese | |
| C | Ascorbic acid | Oxidized form of vitamin C is reduced by glutathione, which helps maintain vitamin C in a reduced (active) form | Enzymatic cofactor for collagen synthesis and very effective in scavenging of reactive oxygen species, i.e., vitamin C is an important non-enzymatic antioxidant | Plant products | Peppers, green leafy vegetables, and in many fruits (kiwis, strawberries, oranges, guavas, and papaya) |
| D | Cholecalciferol | Most important subtype in humans is vitamin D3. In the liver vitamin D is converted to 25-hydroxyvitamin D, which is used as a biomarker | Responsible for intestinal absorption of several minerals including calcium, iron, magnesium, and zinc | Sunlight exposure | Mushrooms, cod liver oil, fish, and cereals |
| E | Tocopherol | Excessive vitamin E may lead to vitamin K * deficiency resulting in bleedings | Plays a role in the oxidation of low-density lipoproteins (LDL) and prevents the production of damaging free radicals. Therefore vitamin E is seen as an important antioxidant. Deficiency might result in peripheral neuropathy and retinopathy | Plant products | Nuts (especially almonds), sunflower oil and seeds, avocados, and dark green leafy vegetables |
| Study | Study Design | Age (Years) | Race | N (Patients) | Prevalence (%) | Definition of Glaucoma | Adjusted Covariates | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Glaucoma (Undefined) | OAG | N (Total) | Cases/Total | |||||||
| Coleman 2008 [23] | cross-sectional | cohort | >65 | Caucasian/African | 95 | 1155 | 8.23 | Based on ONH photographs and VF | Study sites, age, race and ethnicity, education, smoking, alcohol, walking for exercise, BMI, self-rated health status, self-reported diabetes, self-reported hypertension, and age-related macular degeneration. | |
| Giaconi 2012 [22] | cross-sectional | case-control | >65 | African | 77 | 584 | 13.18 | Based on ONH photographs and VF | Study sites, age, education, smoking, alcohol, walking for exercise, BMI, self-rated health status, self-reported diabetes, and self-reported hypertension. | |
| Kang 2003 [24] | prospective | cohort | >40 | Caucasian/African | 474 | 116,484 | 0.41 | Self-reported glaucoma, confirmed with medical records | African heritage, diabetes, hypertension, BMI, physical activity, alcohol intake, and smoking. | |
| Ramdas 2012 [1] | prospective | cohort | ≥55 | Caucasian | 91 | 3502 | 2.60 | Based on slitlamp, ONH (photographs and scans), and VF | Age, gender, IOP, IOP-lowering treatment, BMI, beta-carotene, vitamin B1, B12, E, and magnesium. | |
| Wang 2013 [21] | cross-sectional | cohort | ≥40 | Multiracial | 203 | 2912 | 6.97 | Self-reported glaucoma | Age, demographic variables, smoking, alcohol, self-reported general health condition, and self-reported diabetic retinopathy. | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramdas, W.D.; Schouten, J.S.A.G.; Webers, C.A.B. The Effect of Vitamins on Glaucoma: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 359. https://doi.org/10.3390/nu10030359
Ramdas WD, Schouten JSAG, Webers CAB. The Effect of Vitamins on Glaucoma: A Systematic Review and Meta-Analysis. Nutrients. 2018; 10(3):359. https://doi.org/10.3390/nu10030359
Chicago/Turabian StyleRamdas, Wishal D., Jan S. A. G. Schouten, and Carroll A. B. Webers. 2018. "The Effect of Vitamins on Glaucoma: A Systematic Review and Meta-Analysis" Nutrients 10, no. 3: 359. https://doi.org/10.3390/nu10030359
APA StyleRamdas, W. D., Schouten, J. S. A. G., & Webers, C. A. B. (2018). The Effect of Vitamins on Glaucoma: A Systematic Review and Meta-Analysis. Nutrients, 10(3), 359. https://doi.org/10.3390/nu10030359





