Potential Roles of n-3 PUFAs during Skeletal Muscle Growth and Regeneration
Abstract
1. Introduction
2. The Beneficial Effects of n-3 PUFAs in Skeletal Muscle
2.1. Reducing Inflammation
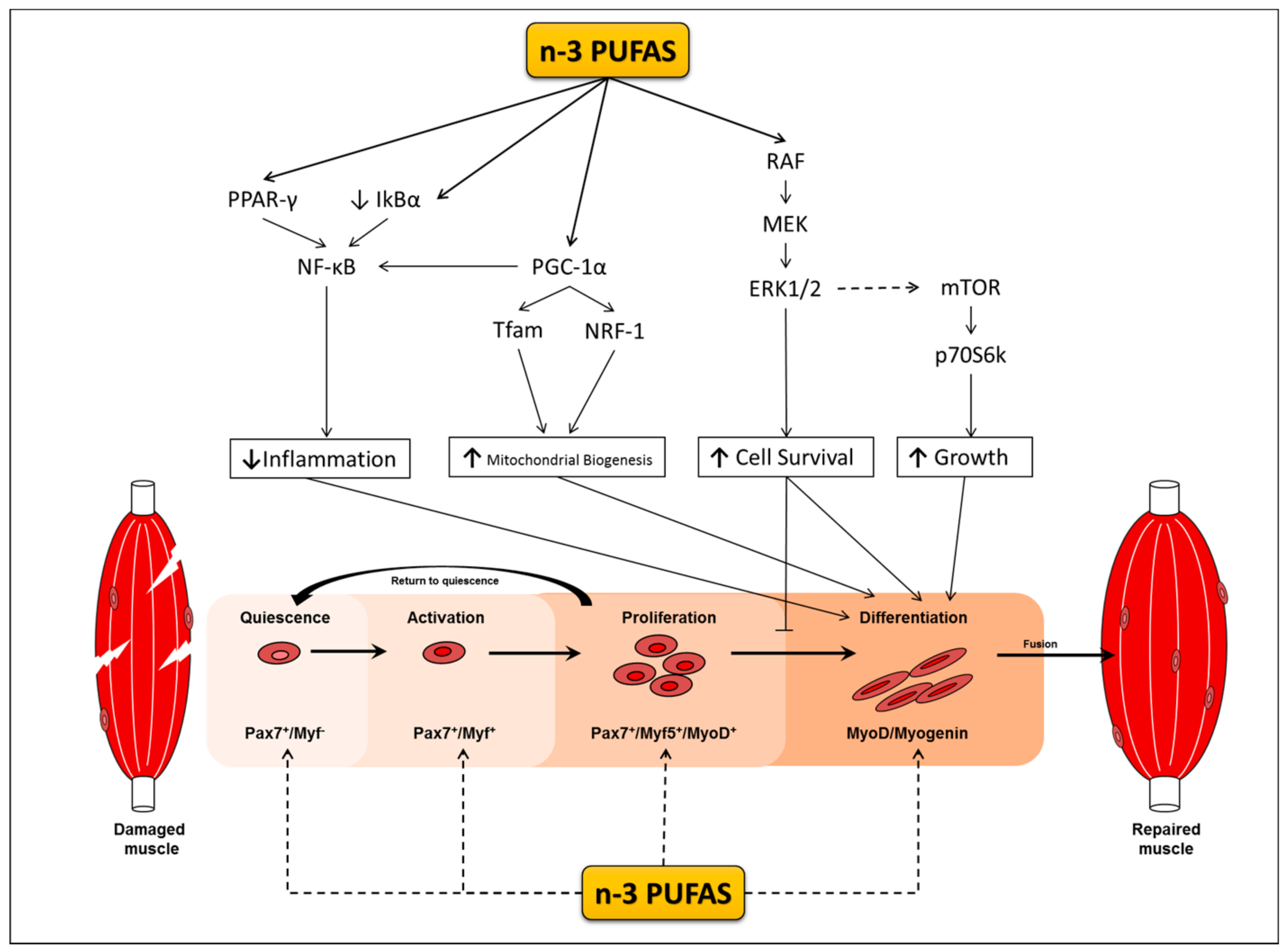
Mechanisms Underlying the Anti-Inflammatory Properties of n-3 PUFAs during Myogenesis
2.2. Protein Metabolism Regulation
2.3. Increasing Muscle Strength
2.4. Potential to Alter Satellite Cell Activation
The Mechanisms Underlying the Modulatory Effects of n-3 PUFAs on Protein Metabolism Signalling and Myogenesis
3. Limitations and Differences between In Vitro Cell Culture and In Vivo Human Studies
4. Future Directions
5. Summary and Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hawley, J.A.; Hargreaves, M.; Joyner, M.J.; Zierath, J.R. Integrative biology of exercise. Cell 2014, 159, 738–749. [Google Scholar] [CrossRef] [PubMed]
- Cuthbertson, D.; Smith, K.; Babraj, J.; Leese, G.; Waddell, T.; Atherton, P.; Wackerhage, H.; Taylor, P.M.; Rennie, M.J. Anabolic signaling deficits underlie amino acid resistance of wasting, aging muscle. FASEB J. 2005, 19, 422–424. [Google Scholar] [CrossRef] [PubMed]
- Wall, B.T.; Gorissen, S.H.; Pennings, B.; Koopman, R.; Groen, B.B.; Verdijk, L.B.; van Loon, L.J. Aging is accompanied by a blunted muscle protein synthetic response to protein ingestion. PLoS ONE 2015, 10, e0140903. [Google Scholar] [CrossRef] [PubMed]
- Brook, M.S.; Wilkinson, D.J.; Mitchell, W.K.; Lund, J.N.; Phillips, B.E.; Szewczyk, N.J.; Greenhaff, P.L.; Smith, K.; Atherton, P.J. Synchronous deficits in cumulative muscle protein synthesis and ribosomal biogenesis underlie age-related anabolic resistance to exercise in humans. J. Physiol. 2016, 594, 7399–7417. [Google Scholar] [CrossRef] [PubMed]
- Fry, C.S.; Drummond, M.J.; Glynn, E.L.; Dickinson, J.M.; Gundermann, D.M.; Timmerman, K.L.; Walker, D.K.; Dhanani, S.; Volpi, E.; Rasmussen, B.B. Aging impairs contraction-induced human skeletal muscle mtorc1 signaling and protein synthesis. Skelet Muscle 2011, 1, 11. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, I.H. Sarcopenia: Origins and clinical relevance. J. Nutr. 1997, 127, 990S–991S. [Google Scholar] [CrossRef] [PubMed]
- Volpi, E.; Mittendorfer, B.; Rasmussen, B.B.; Wolfe, R.R. The response of muscle protein anabolism to combined hyperaminoacidemia and glucose-induced hyperinsulinemia is impaired in the elderly. J. Clin. Endocrinol. Metab. 2000, 85, 4481–4490. [Google Scholar] [CrossRef] [PubMed]
- Janssen, H.C.; Samson, M.M.; Verhaar, H.J. Vitamin d deficiency, muscle function, and falls in elderly people. Am. J. Clin. Nutr. 2002, 75, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Szulc, P.; Duboeuf, F.; Marchand, F.; Delmas, P.D. Hormonal and lifestyle determinants of appendicular skeletal muscle mass in men: The minos study. Am. J. Clin. Nutr. 2004, 80, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Visser, M.; Pahor, M.; Taaffe, D.R.; Goodpaster, B.H.; Simonsick, E.M.; Newman, A.B.; Nevitt, M.; Harris, T.B. Relationship of interleukin-6 and tumor necrosis factor-alpha with muscle mass and muscle strength in elderly men and women: The health abc study. J. Gerontol. A Biol. Sci. Med. Sci. 2002, 57, M326–M332. [Google Scholar] [CrossRef] [PubMed]
- McKay, B.R.; Ogborn, D.I.; Baker, J.M.; Toth, K.G.; Tarnopolsky, M.A.; Parise, G. Elevated SOCS3 and altered IL-6 signaling is associated with age-related human muscle stem cell dysfunction. Am. J. Physiol. Cell Physiol. 2013, 304, C717–C728. [Google Scholar] [CrossRef] [PubMed]
- Vandervoort, A.A. Aging of the human neuromuscular system. Muscle Nerve 2002, 25, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Verdijk, L.B.; Snijders, T.; Drost, M.; Delhaas, T.; Kadi, F.; van Loon, L.J. Satellite cells in human skeletal muscle; from birth to old age. Age 2014, 36, 545–547. [Google Scholar] [CrossRef] [PubMed]
- Verdijk, L.B.; Koopman, R.; Schaart, G.; Meijer, K.; Savelberg, H.H.; van Loon, L.J. Satellite cell content is specifically reduced in type II skeletal muscle fibers in the elderly. Am. J. Physiol. Endocrinol. Metab. 2007, 292, E151–E157. [Google Scholar] [CrossRef] [PubMed]
- Larsson, L.; Sjodin, B.; Karlsson, J. Histochemical and biochemical changes in human skeletal muscle with age in sedentary males, age 22–65 years. Acta Physiol. Scand. 1978, 103, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Frontera, W.R.; Zayas, A.R.; Rodriguez, N. Aging of human muscle: Understanding sarcopenia at the single muscle cell level. Phys. Med. Rehabil. Clin. N. Am. 2012, 23, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Miljkovic, N.; Lim, J.Y.; Miljkovic, I.; Frontera, W.R. Aging of skeletal muscle fibers. Ann. Rehabil. Med. 2015, 39, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Relaix, F.; Marcelle, C. Muscle stem cells. Curr. Opin. Cell Biol. 2009, 21, 748–753. [Google Scholar] [CrossRef] [PubMed]
- Sambasivan, R.; Yao, R.; Kissenpfennig, A.; Van Wittenberghe, L.; Paldi, A.; Gayraud-Morel, B.; Guenou, H.; Malissen, B.; Tajbakhsh, S.; Galy, A. Pax7-expressing satellite cells are indispensable for adult skeletal muscle regeneration. Development 2011, 138, 3647–3656. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, J.J.; Mula, J.; Miyazaki, M.; Erfani, R.; Garrison, K.; Farooqui, A.B.; Srikuea, R.; Lawson, B.A.; Grimes, B.; Keller, C.; et al. Effective fiber hypertrophy in satellite cell-depleted skeletal muscle. Development 2011, 138, 3657–3666. [Google Scholar] [CrossRef] [PubMed]
- Olguin, H.C.; Olwin, B.B. Pax-7 up-regulation inhibits myogenesis and cell cycle progression in satellite cells: A potential mechanism for self-renewal. Dev. Biol. 2004, 275, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Venuti, J.M.; Morris, J.H.; Vivian, J.L.; Olson, E.N.; Klein, W.H. Myogenin is required for late but not early aspects of myogenesis during mouse development. J. Cell Biol. 1995, 128, 563–576. [Google Scholar] [CrossRef] [PubMed]
- Fukada, S.I. The roles of muscle stem cells in muscle injury, atrophy, and hypertrophy. J. Biochem. 2018. [Google Scholar] [CrossRef] [PubMed]
- Murach, K.A.; Fry, C.S.; Kirby, T.J.; Jackson, J.R.; Lee, J.D.; White, S.H.; Dupont-Versteegden, E.E.; McCarthy, J.J.; Peterson, C.A. Starring or supporting role? Satellite cells and skeletal muscle fiber size regulation. Physiology 2018, 33, 26–38. [Google Scholar] [CrossRef] [PubMed]
- Murach, K.A.; White, S.H.; Wen, Y.; Ho, A.; Dupont-Versteegden, E.E.; McCarthy, J.J.; Peterson, C.A. Differential requirement for satellite cells during overload-induced muscle hypertrophy in growing versus mature mice. Skelet Muscle 2017, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Fry, C.S.; Lee, J.D.; Jackson, J.R.; Kirby, T.J.; Stasko, S.A.; Liu, H.; Dupont-Versteegden, E.E.; McCarthy, J.J.; Peterson, C.A. Regulation of the muscle fiber microenvironment by activated satellite cells during hypertrophy. FASEB J. 2014, 28, 1654–1665. [Google Scholar] [CrossRef] [PubMed]
- Egner, I.M.; Bruusgaard, J.C.; Gundersen, K. Satellite cell depletion prevents fiber hypertrophy in skeletal muscle. Development 2016, 143, 2898–2906. [Google Scholar] [CrossRef] [PubMed]
- Keefe, A.C.; Lawson, J.A.; Flygare, S.D.; Fox, Z.D.; Colasanto, M.P.; Mathew, S.J.; Yandell, M.; Kardon, G. Muscle stem cells contribute to myofibers in sedentary adult mice. Nat. Commun. 2015, 6, 7087. [Google Scholar] [CrossRef] [PubMed]
- Fry, C.S.; Lee, J.D.; Mula, J.; Kirby, T.J.; Jackson, J.R.; Liu, F.; Yang, L.; Mendias, C.L.; Dupont-Versteegden, E.E.; McCarthy, J.J.; et al. Inducible depletion of satellite cells in adult, sedentary mice impairs muscle regenerative capacity but does not contribute to sarcopenia. Nat. Med. 2015, 21, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Fry, C.S.; Kirby, T.J.; Kosmac, K.; McCarthy, J.J.; Peterson, C.A. Myogenic progenitor cells control extracellular matrix production by fibroblasts during skeletal muscle hypertrophy. Cell Stem Cell 2017, 20, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.M.; Lawson, J.A.; Mathew, S.J.; Hutcheson, D.A.; Kardon, G. Satellite cells, connective tissue fibroblasts and their interactions are crucial for muscle regeneration. Development 2011, 138, 3625–3637. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.I.; Julliand, S.; Reeds, D.N.; Sinacore, D.R.; Klein, S.; Mittendorfer, B. Fish oil-derived n-3 pufa therapy increases muscle mass and function in healthy older adults. Am. J. Clin. Nutr. 2015, 102, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.I.; Atherton, P.; Reeds, D.N.; Mohammed, B.S.; Rankin, D.; Rennie, M.J.; Mittendorfer, B. Omega-3 polyunsaturated fatty acids augment the muscle protein anabolic response to hyperinsulinaemia-hyperaminoacidaemia in healthy young and middle-aged men and women. Clin. Sci. 2011, 121, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Rodacki, C.L.; Rodacki, A.L.; Pereira, G.; Naliwaiko, K.; Coelho, I.; Pequito, D.; Fernandes, L.C. Fish-oil supplementation enhances the effects of strength training in elderly women. Am. J. Clin. Nutr. 2012, 95, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Human requirement for n-3 polyunsaturated fatty acids. Poult. Sci. 2000, 79, 961–970. [Google Scholar] [CrossRef] [PubMed]
- Baker, E.J.; Miles, E.A.; Burdge, G.C.; Yaqoob, P.; Calder, P.C. Metabolism and functional effects of plant-derived omega-3 fatty acids in humans. Prog. Lipid Res. 2016, 64, 30–56. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Sinclair, A.J.; Cameron-Smith, D.; Barr, D.P.; Molero-Navajas, J.C.; Konstantopoulos, N. Docosapentaenoic acid (22:5n-3) down-regulates the expression of genes involved in fat synthesis in liver cells. Prostaglandins Leukot Essent Fatty Acids 2011, 85, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Burdge, G.C.; Jones, A.E.; Wootton, S.A. Eicosapentaenoic and docosapentaenoic acids are the principal products of alpha-linolenic acid metabolism in young men. Br. J. Nutr. 2002, 88, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Burdge, G.C.; Wootton, S.A. Conversion of alpha-linolenic acid to eicosapentaenoic, docosapentaenoic and docosahexaenoic acids in young women. Br. J. Nutr. 2002, 88, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Arterburn, L.M.; Hall, E.B.; Oken, H. Distribution, interconversion, and dose response of n-3 fatty acids in humans. Am. J. Clin. Nutr. 2006, 83, 1467S. [Google Scholar] [CrossRef] [PubMed]
- Morin, C.; Charbonneau, L.; Ouellet, N.; Ouellet, H.; Blier, P.U.; Dufresne, F.; Fortin, S. Eicosapentaenoic acid monoglyceride resolves inflammation in an ex vivo model of human peripheral blood mononuclear cell. Eur. J. Pharmacol. 2017, 807, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Vedin, I.; Cederholm, T.; Freund Levi, Y.; Basun, H.; Garlind, A.; Faxen Irving, G.; Jonhagen, M.E.; Vessby, B.; Wahlund, L.O.; Palmblad, J. Effects of docosahexaenoic acid-rich n-3 fatty acid supplementation on cytokine release from blood mononuclear leukocytes: The omegad study. Am. J. Clin. Nutr. 2008, 87, 1616–1622. [Google Scholar] [CrossRef] [PubMed]
- Shindou, H.; Koso, H.; Sasaki, J.; Nakanishi, H.; Sagara, H.; Nakagawa, K.M.; Takahashi, Y.; Hishikawa, D.; Iizuka-Hishikawa, Y.; Tokumasu, F.; et al. Docosahexaenoic acid preserves visual function by maintaining correct disc morphology in retinal photoreceptor cells. J. Biol. Chem. 2017, 292, 12054–12064. [Google Scholar] [CrossRef] [PubMed]
- Litman, B.J.; Niu, S.L.; Polozova, A.; Mitchell, D.C. The role of docosahexaenoic acid containing phospholipids in modulating G protein-coupled signaling pathways: Visual transduction. J. Mol. Neurosci. 2001, 16. [Google Scholar] [CrossRef]
- Albert, B.B.; Derraik, J.G.; Brennan, C.M.; Biggs, J.B.; Smith, G.C.; Garg, M.L.; Cameron-Smith, D.; Hofman, P.L.; Cutfield, W.S. Higher omega-3 index is associated with increased insulin sensitivity and more favourable metabolic profile in middle-aged overweight men. Sci. Rep. 2014, 4, 6697. [Google Scholar] [CrossRef] [PubMed]
- McGlory, C.; Wardle, S.L.; Macnaughton, L.S.; Witard, O.C.; Scott, F.; Dick, J.; Bell, J.G.; Phillips, S.M.; Galloway, S.D.; Hamilton, D.L.; et al. Fish oil supplementation suppresses resistance exercise and feeding-induced increases in anabolic signaling without affecting myofibrillar protein synthesis in young men. Physiol. Rep. 2016, 4, e12715. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.I.; Atherton, P.; Reeds, D.N.; Mohammed, B.S.; Rankin, D.; Rennie, M.J.; Mittendorfer, B. Dietary omega-3 fatty acid supplementation increases the rate of muscle protein synthesis in older adults: A randomized controlled trial. Am. J. Clin. Nutr. 2011, 93, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Bhullar, A.S.; Putman, C.T.; Mazurak, V.C. Potential role of omega-3 fatty acids on the myogenic program of satellite cells. Nutr. Metab. Insights 2016, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kalinkovich, A.; Livshits, G. Sarcopenic obesity or obese sarcopenia: A cross talk between age-associated adipose tissue and skeletal muscle inflammation as a main mechanism of the pathogenesis. Ageing Res. Rev. 2017, 35, 200–221. [Google Scholar] [CrossRef] [PubMed]
- Millward, D.J. Nutrition and sarcopenia: Evidence for an interaction. Proc. Nutr. Soc. 2012, 71, 566–575. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M. Nutritional supplements in support of resistance exercise to counter age-related sarcopenia. Adv. Nutr. 2015, 6, 452–460. [Google Scholar] [CrossRef] [PubMed]
- Soysal, P.; Stubbs, B.; Lucato, P.; Luchini, C.; Solmi, M.; Peluso, R.; Sergi, G.; Isik, A.T.; Manzato, E.; Maggi, S.; et al. Inflammation and frailty in the elderly: A systematic review and meta-analysis. Ageing Res. Rev. 2016, 31, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bano, G.; Trevisan, C.; Carraro, S.; Solmi, M.; Luchini, C.; Stubbs, B.; Manzato, E.; Sergi, G.; Veronese, N. Inflammation and sarcopenia: A systematic review and meta-analysis. Maturitas 2017, 96, 10–15. [Google Scholar] [CrossRef] [PubMed]
- McKay, B.R.; De Lisio, M.; Johnston, A.P.; O’Reilly, C.E.; Phillips, S.M.; Tarnopolsky, M.A.; Parise, G. Association of interleukin-6 signalling with the muscle stem cell response following muscle-lengthening contractions in humans. PLoS ONE 2009, 4, e6027. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Li, Y.; Wu, Y.; Wang, L.; Wang, X.; Du, J. Interleukin-6/signal transducer and activator of transcription 3 (STAT3) pathway is essential for macrophage infiltration and myoblast proliferation during muscle regeneration. J. Biol. Chem. 2013, 288, 1489–1499. [Google Scholar] [CrossRef] [PubMed]
- Sindhu, S.; Thomas, R.; Shihab, P.; Sriraman, D.; Behbehani, K.; Ahmad, R. Obesity is a positive modulator of IL-6R and IL-6 expression in the subcutaneous adipose tissue: Significance for metabolic inflammation. PLoS ONE 2015, 10, e0133494. [Google Scholar] [CrossRef] [PubMed]
- Feinstein, R.; Kanety, H.; Papa, M.Z.; Lunenfeld, B.; Karasik, A. Tumor necrosis factor-alpha suppresses insulin-induced tyrosine phosphorylation of insulin receptor and its substrates. J. Biol. Chem. 1993, 268, 26055–26058. [Google Scholar] [PubMed]
- Hotamisligil, G.S.; Shargill, N.S.; Spiegelman, B.M. Adipose expression of tumor necrosis factor-alpha: Direct role in obesity-linked insulin resistance. Science 1993, 259, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Merritt, E.K.; Stec, M.J.; Thalacker-Mercer, A.; Windham, S.T.; Cross, J.M.; Shelley, D.P.; Craig Tuggle, S.; Kosek, D.J.; Kim, J.S.; Bamman, M.M. Heightened muscle inflammation susceptibility may impair regenerative capacity in aging humans. J. Appl. Physiol. 2013, 115, 937–948. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Long-chain fatty acids and inflammation. Proc. Nutr. Soc. 2012, 71, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. N-3 fatty acids, inflammation and immunity: New mechanisms to explain old actions. Proc. Nutr. Soc. 2013, 72, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Wada, M.; DeLong, C.J.; Hong, Y.H.; Rieke, C.J.; Song, I.; Sidhu, R.S.; Yuan, C.; Warnock, M.; Schmaier, A.H.; Yokoyama, C.; et al. Enzymes and receptors of prostaglandin pathways with arachidonic acid-derived versus eicosapentaenoic acid-derived substrates and products. J. Biol. Chem. 2007, 282, 22254–22266. [Google Scholar] [CrossRef] [PubMed]
- Kelley, D.S.; Taylor, P.C.; Nelson, G.J.; Schmidt, P.C.; Mackey, B.E.; Kyle, D. Effects of dietary arachidonic acid on human immune response. Lipids 1997, 32, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Kelley, D.S.; Taylor, P.C.; Nelson, G.J.; Mackey, B.E. Arachidonic acid supplementation enhances synthesis of eicosanoids without suppressing immune functions in young healthy men. Lipids 1998, 33, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Phang, M.; Garg, M.L.; Sinclair, A.J. Inhibition of platelet aggregation by omega-3 polyunsaturated fatty acids is gender specific-redefining platelet response to fish oils. Prostaglandins Leukot Essent Fatty Acids 2009, 81, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Bartram, H.P.; Gostner, A.; Scheppach, W.; Reddy, B.S.; Rao, C.V.; Dusel, G.; Richter, F.; Richter, A.; Kasper, H. Effects of fish oil on rectal cell proliferation, mucosal fatty acids, and prostaglandin e2 release in healthy subjects. Gastroenterology 1993, 105, 1317–1322. [Google Scholar] [CrossRef]
- Calder, P.C. Polyunsaturated fatty acids and inflammatory processes: New twists in an old tale. Biochimie 2009, 91, 791–795. [Google Scholar] [CrossRef] [PubMed]
- Cai, D.; Frantz, J.D.; Tawa, N.E., Jr.; Melendez, P.A.; Oh, B.C.; Lidov, H.G.; Hasselgren, P.O.; Frontera, W.R.; Lee, J.; Glass, D.J.; et al. Ikkbeta/nf-kappab activation causes severe muscle wasting in mice. Cell 2004, 119, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lin, Q.W.; Zheng, P.P.; Zhang, J.S.; Huang, F.R. Dha inhibits protein degradation more efficiently than epa by regulating the ppargamma/nfkappab pathway in c2c12 myotubes. Biomed. Res. Int. 2013, 2013, 318981. [Google Scholar] [PubMed]
- Camandola, S.; Leonarduzzi, G.; Musso, T.; Varesio, L.; Carini, R.; Scavazza, A.; Chiarpotto, E.; Baeuerle, P.A.; Poli, G. Nuclear factor kb is activated by arachidonic acid but not by eicosapentaenoic acid. Biochem. Biophys. Res. Commun. 1996, 229, 643–647. [Google Scholar] [CrossRef] [PubMed]
- Williams-Bey, Y.; Boularan, C.; Vural, A.; Huang, N.N.; Hwang, I.Y.; Shan-Shi, C.; Kehrl, J.H. Omega-3 free fatty acids suppress macrophage inflammasome activation by inhibiting nf-κb activation and enhancing autophagy. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Wei, H.; Luo, H.; Jiang, S.; Peng, J. EPA inhibits the inhibitor of kappabalpha (IκBα)/NF-κB/muscle ring finger 1 pathway in C2C12 myotubes in a PPARγ-dependent manner. Br. J. Nutr. 2011, 105, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Serrano, A.L.; Baeza-Raja, B.; Perdiguero, E.; Jardi, M.; Munoz-Canoves, P. Interleukin-6 is an essential regulator of satellite cell-mediated skeletal muscle hypertrophy. Cell Metab. 2008, 7, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.E.; Gerken, E.; Zhang, Y.; Zhan, M.; Mohan, R.K.; Li, A.S.; Reid, M.B.; Li, Y.P. Role of tnf-{alpha} signaling in regeneration of cardiotoxin-injured muscle. Am. J. Physiol. Cell Physiol. 2005, 289, C1179–C1187. [Google Scholar] [CrossRef] [PubMed]
- Langen, R.C.; Van Der Velden, J.L.; Schols, A.M.; Kelders, M.C.; Wouters, E.F.; Janssen-Heininger, Y.M. Tumor necrosis factor-alpha inhibits myogenic differentiation through myod protein destabilization. FASEB J. 2004, 18, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Domingues-Faria, C.; Vasson, M.P.; Goncalves-Mendes, N.; Boirie, Y.; Walrand, S. Skeletal muscle regeneration and impact of aging and nutrition. Ageing Res. Rev. 2016, 26, 22–36. [Google Scholar] [CrossRef] [PubMed]
- Wahlin-Larsson, B.; Carnac, G.; Kadi, F. The influence of systemic inflammation on skeletal muscle in physically active elderly women. Age 2014, 36, 9718. [Google Scholar] [CrossRef] [PubMed]
- Da Boit, M.; Sibson, R.; Sivasubramaniam, S.; Meakin, J.R.; Greig, C.A.; Aspden, R.M.; Thies, F.; Jeromson, S.; Hamilton, D.L.; Speakman, J.R.; et al. Sex differences in the effect of fish-oil supplementation on the adaptive response to resistance exercise training in older people: A randomized controlled trial. Am. J. Clin. Nutr. 2017, 105, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Briolay, A.; Jaafar, R.; Nemoz, G.; Bessueille, L. Myogenic differentiation and lipid-raft composition of l6 skeletal muscle cells are modulated by pufas. Biochim. Biophys. Acta 2013, 1828, 602–613. [Google Scholar] [CrossRef] [PubMed]
- Jeromson, S.; Mackenzie, I.; Doherty, M.K.; Whitfield, P.D.; Bell, G.; Dick, J.; Shaw, A.; Rao, F.; Ashcroft, S.; Philp, A.; et al. Lipid remodelling and an altered membrane proteome may drive the effects of epa and dha treatment on skeletal muscle glucose uptake and protein accretion. Am. J. Physiol. Endocrinol. Metab. 2017. [Google Scholar] [CrossRef] [PubMed]
- Mukai, A.; Kurisaki, T.; Sato, S.B.; Kobayashi, T.; Kondoh, G.; Hashimoto, N. Dynamic clustering and dispersion of lipid rafts contribute to fusion competence of myogenic cells. Exp. Cell Res. 2009, 315, 3052–3063. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, P.A. Regulatory role of peroxisome proliferator-activated receptor delta (ppar delta) in muscle metabolism. A new target for metabolic syndrome treatment? Biochimie 2005, 87, 5–8. [Google Scholar] [CrossRef] [PubMed]
- Chandrashekar, P.; Manickam, R.; Ge, X.; Bonala, S.; McFarlane, C.; Sharma, M.; Wahli, W.; Kambadur, R. Inactivation of pparbeta/delta adversely affects satellite cells and reduces postnatal myogenesis. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E122–E131. [Google Scholar] [CrossRef] [PubMed]
- Deng, B.; Zhang, F.; Chen, K.; Wen, J.; Huang, H.; Liu, W.; Ye, S.; Wang, L.; Yang, Y.; Gong, P.; et al. Myod promotes porcine ppargamma gene expression through an e-box and a myod-binding site in the ppargamma promoter region. Cell Tissue Res. 2016, 365, 381–391. [Google Scholar] [CrossRef] [PubMed]
- Varga, T.; Czimmerer, Z.; Nagy, L. Ppars are a unique set of fatty acid regulated transcription factors controlling both lipid metabolism and inflammation. Biochim. Biophys. Acta 2011, 1812, 1007–1022. [Google Scholar] [CrossRef] [PubMed]
- Fritsche, K.L. The science of fatty acids and inflammation. Adv. Nutr. 2015, 6, 293S–301S. [Google Scholar] [CrossRef] [PubMed]
- Magee, P.; Pearson, S.; Allen, J. The omega-3 fatty acid, eicosapentaenoic acid (EPA), prevents the damaging effects of tumour necrosis factor (TNF)-alpha during murine skeletal muscle cell differentiation. Lipids Health Dis. 2008, 7, 24. [Google Scholar] [CrossRef] [PubMed]
- Magee, P.; Pearson, S.; Whittingham-Dowd, J.; Allen, J. Ppargamma as a molecular target of epa anti-inflammatory activity during tnf-alpha-impaired skeletal muscle cell differentiation. J. Nutr. Biochem. 2012, 23, 1440–1448. [Google Scholar] [CrossRef] [PubMed]
- Angione, A.R.; Jiang, C.; Pan, D.; Wang, Y.-X.; Kuang, S. Pparδ regulates satellite cell proliferation and skeletal muscle regeneration. Skeletal Muscle 2011, 1, 33. [Google Scholar] [CrossRef] [PubMed]
- Eisele, P.S.; Salatino, S.; Sobek, J.; Hottiger, M.O.; Handschin, C. The peroxisome proliferator-activated receptor gamma coactivator 1α/β (PGC-1) coactivators repress the transcriptional activity of NF-κB in skeletal muscle cells. J. Biol. Chem. 2013, 288, 2246–2260. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Shin, Y.; Moon, S.; Kim, S.; Kim, Y. Effects of eicosapentaenoic acid and docosahexaenoic acid on mitochondrial DNA replication and PGC-1α gene expression in C(2)C(12) muscle cells. Prev. Nutr. Food Sci. 2016, 21, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Virbasius, J.V.; Scarpulla, R.C. Activation of the human mitochondrial transcription. Proc. Natl. Acad. Sci. USA 1994, 91, 1309–1313. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Zhao, Y.; Li, R.; Gong, J.; Zheng, Y.; Wang, Y. Pgc-1α is associated with c2c12 myoblast differentiation. Cent. Eur. J. Biol. 2014, 9, 1030–1036. [Google Scholar] [CrossRef]
- Hancock, C.R.; Han, D.H.; Chen, M.; Terada, S.; Yasuda, T.; Wright, D.C.; Holloszy, J.O. High-fat diets cause insulin resistance despite an increase in muscle mitochondria. Proc. Natl. Acad. Sci. USA 2008, 105, 7815–7820. [Google Scholar] [CrossRef] [PubMed]
- Pinel, A.; Rigaudiere, J.P.; Laillet, B.; Pouyet, C.; Malpuech-Brugere, C.; Prip-Buus, C.; Morio, B.; Capel, F. N-3pufa differentially modulate palmitate-induced lipotoxicity through alterations of its metabolism in C2C12 muscle cells. Biochim. Biophys. Acta 2016, 1861, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Bryner, R.W.; Woodworth-Hobbs, M.E.; Williamson, D.L.; Alway, S.E. Docosahexaenoic acid protects muscle cells from palmitate-induced atrophy. ISRN Obes. 2012, 2012, 647348. [Google Scholar] [CrossRef] [PubMed]
- Endres, S.; Ghorbani, R.; Kelley, V.E.; Georgilis, K.; Lonnemann, G.; van der Meer, J.W.; Cannon, J.G.; Rogers, T.S.; Klempner, M.S.; Weber, P.C.; et al. The effect of dietary supplementation with n-3 polyunsaturated fatty acids on the synthesis of interleukin-1 and tumor necrosis factor by mononuclear cells. N. Engl. J. Med. 1989, 320, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.Y.; Talukdar, S.; Bae, E.J.; Imamura, T.; Morinaga, H.; Fan, W.; Li, P.; Lu, W.J.; Watkins, S.M.; Olefsky, J.M. GPR120 is an omega-3 fatty acid receptor mediating potent anti-inflammatory and insulin-sensitizing effects. Cell 2010, 142, 687–698. [Google Scholar] [CrossRef] [PubMed]
- Rieu, I.; Magne, H.; Savary-Auzeloux, I.; Averous, J.; Bos, C.; Peyron, M.A.; Combaret, L.; Dardevet, D. Reduction of low grade inflammation restores blunting of postprandial muscle anabolism and limits sarcopenia in old rats. J. Physiol. 2009, 587, 5483–5492. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.R.; Robinson, M.J.; Fry, J.L.; Tang, J.E.; Glover, E.I.; Wilkinson, S.B.; Prior, T.; Tarnopolsky, M.A.; Phillips, S.M. Ingested protein dose response of muscle and albumin protein synthesis after resistance exercise in young men. Am. J. Clin. Nutr. 2009, 89, 161–168. [Google Scholar] [CrossRef] [PubMed]
- MacDougall, J.D.; Gibala, M.J.; Tarnopolsky, M.A.; MacDonald, J.R.; Interisano, S.A.; Yarasheski, K.E. The time course for elevated muscle protein synthesis following heavy resistance exercise. Can. J. Appl. Physiol. 1995, 20, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Witard, O.C.; Jackman, S.R.; Breen, L.; Smith, K.; Selby, A.; Tipton, K.D. Myofibrillar muscle protein synthesis rates subsequent to a meal in response to increasing doses of whey protein at rest and after resistance exercise. Am. J. Clin. Nutr. 2014, 99, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Hulmi, J.J.; Kovanen, V.; Lisko, I.; Selanne, H.; Mero, A.A. The effects of whey protein on myostatin and cell cycle-related gene expression responses to a single heavy resistance exercise bout in trained older men. Eur J. Appl. Physiol. 2008, 102, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Farup, J.; Rahbek, S.K.; Knudsen, I.S.; de Paoli, F.; Mackey, A.L.; Vissing, K. Whey protein supplementation accelerates satellite cell proliferation during recovery from eccentric exercise. Amino Acids 2014, 46, 2503–2516. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Selby, A.; Rankin, D.; Patel, R.; Atherton, P.; Hildebrandt, W.; Williams, J.; Smith, K.; Seynnes, O.; Hiscock, N.; et al. Age-related differences in the dose-response relationship of muscle protein synthesis to resistance exercise in young and old men. J. Physiol. 2009, 587, 211–217. [Google Scholar] [CrossRef] [PubMed]
- McGlory, C.; Galloway, S.D.; Hamilton, D.L.; McClintock, C.; Breen, L.; Dick, J.R.; Bell, J.G.; Tipton, K.D. Temporal changes in human skeletal muscle and blood lipid composition with fish oil supplementation. Prostaglandins Leukot Essent Fatty Acids 2014, 90, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Lauretani, F.; Bandinelli, S.; Bartali, B.; Cherubini, A.; Iorio, A.D.; Ble, A.; Giacomini, V.; Corsi, A.M.; Guralnik, J.M.; Ferrucci, L. Omega-6 and omega-3 fatty acids predict accelerated decline of peripheral nerve function in older persons. Eur J. Neurol. 2007, 14, 801–808. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.X.; Leaf, A. Effects of long-chain polyunsaturated fatty acids on the contraction of neonatal rat cardiac myocytes. Proc. Natl. Acad. Sci. USA 1994, 91, 9886–9890. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, S.C.; Perez, M.R.; Hammond, K.E.; Sheader, E.A.; Negretti, N. Direct and indirect modulation of rat cardiac sarcoplasmic reticulum function by n-3 polyunsaturated fatty acids. J. Physiol. 2002, 538, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.J.; Radonic, P.W.; Wolever, T.M.; Wells, G.D. 21 Days of mammalian omega-3 fatty acid supplementation improves aspects of neuromuscular function and performance in male athletes compared to olive oil placebo. J. Int. Soc. Sports Nutr. 2015, 12, 28. [Google Scholar] [CrossRef] [PubMed]
- Mizunoya, W.; Iwamoto, Y.; Shirouchi, B.; Sato, M.; Komiya, Y.; Razin, F.R.; Tatsumi, R.; Sato, Y.; Nakamura, M.; Ikeuchi, Y. Dietary fat influences the expression of contractile and metabolic genes in rat skeletal muscle. PLoS ONE 2013, 8, e80152. [Google Scholar] [CrossRef] [PubMed]
- Leenders, M.; Verdijk, L.B.; van der Hoeven, L.; van Kranenburg, J.; Nilwik, R.; van Loon, L.J. Elderly men and women benefit equally from prolonged resistance-type exercise training. J. Gerontol. A Biol. Sci. Med. Sci. 2013, 68, 769–779. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Lee, M.; Park, Y. Synergistic attenuation of ovariectomy-induced bone loss by combined use of fish oil and 17beta-oestradiol. Br. J. Nutr. 2017, 117, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.I.; Mittendorfer, B. Sexual dimorphism in skeletal muscle protein turnover. J. Appl. Physiol. 2016, 120, 674–682. [Google Scholar] [CrossRef] [PubMed]
- Egerman, M.A.; Cadena, S.M.; Gilbert, J.A.; Meyer, A.; Nelson, H.N.; Swalley, S.E.; Mallozzi, C.; Jacobi, C.; Jennings, L.L.; Clay, I.; et al. Gdf11 increases with age and inhibits skeletal muscle regeneration. Cell Metab. 2015, 22, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Conboy, I.M.; Conboy, M.J.; Wagers, A.J.; Girma, E.R.; Weissman, I.L.; Rando, T.A. Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature 2005, 433, 760–764. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Bosco, N.; Bourdet-Sicard, R.; Capuron, L.; Delzenne, N.; Dore, J.; Franceschi, C.; Lehtinen, M.J.; Recker, T.; Salvioli, S.; et al. Health relevance of the modification of low grade inflammation in ageing (inflammageing) and the role of nutrition. Ageing Res. Rev. 2017, 40, 95–119. [Google Scholar] [CrossRef] [PubMed]
- Apolinario, L.M.; De Carvalho, S.C.; Santo Neto, H.; Marques, M.J. Long-term therapy with omega-3 ameliorates myonecrosis and benefits skeletal muscle regeneration in mdx mice. Anat. Rec. 2015, 298, 1589–1596. [Google Scholar] [CrossRef] [PubMed]
- Mackey, A.L.; Rasmussen, L.K.; Kadi, F.; Schjerling, P.; Helmark, I.C.; Ponsot, E.; Aagaard, P.; Durigan, J.L.; Kjaer, M. Activation of satellite cells and the regeneration of human skeletal muscle are expedited by ingestion of nonsteroidal anti-inflammatory medication. FASEB J. 2016, 30, 2266–2281. [Google Scholar] [CrossRef] [PubMed]
- Landi, F.; Marzetti, E.; Liperoti, R.; Pahor, M.; Russo, A.; Martone, A.M.; Colloca, G.; Capoluongo, E.; Bernabei, R. Nonsteroidal anti-inflammatory drug (NSAID) use and sarcopenia in older people: Results from the ilsirente study. J. Am. Med. Direct. Assoc. 2013, 14, 626. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Liang, X.; Shan, T.; Jiang, Q.; Deng, C.; Zheng, R.; Kuang, S. Mtor is necessary for proper satellite cell activity and skeletal muscle regeneration. Biochem. Biophys. Res. Commun. 2015, 463, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Kamolrat, T.; Gray, S.R. The effect of eicosapentaenoic and docosahexaenoic acid on protein synthesis and breakdown in murine C2C12 myotubes. Biochem. Biophys. Res. Commun. 2013, 432, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Bodine, S.C.; Stitt, T.N.; Gonzalez, M.; Kline, W.O.; Stover, G.L.; Bauerlein, R.; Zlotchenko, E.; Scrimgeour, A.; Lawrence, J.C.; Glass, D.J.; et al. Akt/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy in vivo. Nat. Cell Biol. 2001, 3, 1014–1019. [Google Scholar] [CrossRef] [PubMed]
- Martin, T.D.; Dennis, M.D.; Gordon, B.S.; Kimball, S.R.; Jefferson, L.S. Mtorc1 and jnk coordinate phosphorylation of the p70s6k1 autoinhibitory domain in skeletal muscle following functional overloading. Am. J. Physiol. Endocrinol. Metab. 2014, 306, E1397–E1405. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.M.; Tonks, N.K. Regulation of distinct stages of skeletal muscle differentiation by mitogen-activated protein kinases. Science 1997, 278, 1288–1291. [Google Scholar] [CrossRef] [PubMed]
- Kramer, H.F.; Goodyear, L.J. Exercise, mapk, and nf-kappab signaling in skeletal muscle. J. Appl. Physiol. 2007, 103, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Volonte, D.; Liu, Y.; Galbiati, F. The modulation of caveolin-1 expression controls satellite cell activation during muscle repair. FASEB J. 2005, 19, 237–239. [Google Scholar] [CrossRef] [PubMed]
- Kook, S.H.; Son, Y.O.; Choi, K.C.; Lee, H.J.; Chung, W.T.; Hwang, I.H.; Lee, J.C. Cyclic mechanical stress suppresses myogenic differentiation of adult bovine satellite cells through activation of extracellular signal-regulated kinase. Mol. Cell. Biochem. 2008, 309, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.Y.; Yao, K.; Bode, A.M.; Bergen, H.R., III; Madden, B.J.; Oh, S.M.; Ermakova, S.; Kang, B.S.; Choi, H.S.; Shim, J.H.; et al. RSK2 mediates muscle cell differentiation through regulation of NFAT3. J. Biol. Chem. 2007, 282, 8380–8392. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Johnson, S.E. ERK2 is required for efficient terminal differentiation of skeletal myoblasts. Biochem. Biophys. Res. Commun. 2006, 345, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Zheng, Y.; Zhang, Y.; Zhao, J.; Chang, F.; Lu, T.; Zhang, R.; Li, Q.; Hu, X.; Li, N. Different effects of omega-3 fatty acids on the cell cycle in c2c12 myoblast proliferation. Mol. Cell. Biochem. 2012, 367, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Day, K.; Shefer, G.; Shearer, A.; Yablonka-Reuveni, Z. The depletion of skeletal muscle satellite cells with age is concomitant with reduced capacity of single progenitors to produce reserve progeny. Dev. Biol. 2010, 340, 330–343. [Google Scholar] [CrossRef] [PubMed]
- He, W.A.; Berardi, E.; Cardillo, V.M.; Acharyya, S.; Aulino, P.; Thomas-Ahner, J.; Wang, J.; Bloomston, M.; Muscarella, P.; Nau, P.; et al. Nf-kappab-mediated pax7 dysregulation in the muscle microenvironment promotes cancer cachexia. J. Clin. Investig. 2013, 123, 4821–4835. [Google Scholar] [CrossRef] [PubMed]
- Murphy, L.O.; Blenis, J. Mapk signal specificity: The right place at the right time. Trends Biochem. Sci. 2006, 31, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Saini, A.; Sharples, A.P.; Al-Shanti, N.; Stewart, C.E. Omega-3 fatty acid epa improves regenerative capacity of mouse skeletal muscle cells exposed to saturated fat and inflammation. Biogerontology 2017, 18, 109–129. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, M.A.; Le Grand, F.; Scimè, A.; Kuang, S.; von Maltzahn, J.; Seale, V.; Cuenda, A.; Ranish, J.A.; Rudnicki, M.A. P38-γ–dependent gene silencing restricts entry into the myogenic differentiation program. J. Cell Biol. 2009, 187, 991–1005. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Yang, R.; Martinod, K.; Kasuga, K.; Pillai, P.S.; Porter, T.F.; Oh, S.F.; Spite, M. Maresins: Novel macrophage mediators with potent antiinflammatory and proresolving actions. J. Exp. Med. 2009, 206, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Gura, K.M.; Kim, S.; Arsenault, D.A.; Bistrian, B.R.; Puder, M. Current clinical applications of omega-6 and omega-3 fatty acids. Nutr. Clin. Pract. 2006, 21, 323–341. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Zhou, Y.; Hu, X.; Peng, X.; Wei, H.; Peng, J.; Jiang, S. Activation of PPARγ2 by ppargamma1 through a functional ppre in transdifferentiation of myoblasts to adipocytes induced by EPA. Cell Cycle 2015, 14, 1830–1841. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Tachibana, H.; Morinaga, Y.; Fujimura, Y.; Yamada, K. Modulation of proliferation and differentiation of c2c12 skeletal muscle cells by fatty acids. Life Sci. 2009, 84, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.J.; Lorite, M.J.; Tisdale, M.J. Effect of a cancer cachectic factor on protein synthesis/degradation in murine C2C12 myoblasts: Modulation by eicosapentaenoic acid. Cancer Res. 1999, 59, 5507–5513. [Google Scholar] [PubMed]
- Lee, M.S.; Kim, I.H.; Kim, Y. Effects of eicosapentaenoic acid and docosahexaenoic acid on uncoupling protein 3 gene expression in C(2)C(12) muscle cells. Nutrients 2013, 5, 1660–1671. [Google Scholar] [CrossRef] [PubMed]
- Woodworth-Hobbs, M.E.; Hudson, M.B.; Rahnert, J.A.; Zheng, B.; Franch, H.A.; Price, S.R. Docosahexaenoic acid prevents palmitate-induced activation of proteolytic systems in c2c12 myotubes. J. Nutr. Biochem. 2014, 25, 868–874. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.C.; Chen, P.Y.; Wu, Y.L.; Chen, C.W.; Chen, H.W.; Lii, C.K.; Sun, H.L.; Liu, K.L. Long-chain polyunsaturated fatty acids amend palmitate-induced inflammation and insulin resistance in mouse C2C12 myotubes. Food Funct. 2016, 7, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Lalia, A.Z.; Dasari, S.; Robinson, M.M.; Abid, H.; Morse, D.M.; Klaus, K.A.; Lanza, I.R. Influence of omega-3 fatty acids on skeletal muscle protein metabolism and mitochondrial bioenergetics in older adults. Aging 2017, 9, 1096–1115. [Google Scholar] [CrossRef] [PubMed]
- Schiaffino, S.; Bormioli, S.P.; Aloisi, M. The fate of newly formed satellite cells during compensatory muscle hypertrophy. Virchows Arch. B Cell Pathol. 1976, 21, 113–118. [Google Scholar] [PubMed]
- Moss, F.P.; Leblond, C.P. Nature of dividing nuclei in skeletal muscle of growing rats. J. Cell Biol. 1970, 44, 459–461. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, J.; Smith, G.I.; Kelly, S.C.; Julliand, S.; Reeds, D.N.; Mittendorfer, B. Effect of dietary n-3 pufa supplementation on the muscle transcriptome in older adults. Physiol. Rep. 2016, 4, e12785. [Google Scholar] [CrossRef] [PubMed]
- Pawar, A.; Jump, D.B. Unsaturated fatty acid regulation of peroxisome proliferator-activated receptor alpha activity in rat primary hepatocytes. J. Biol. Chem. 2003, 278, 35931–35939. [Google Scholar] [CrossRef] [PubMed]
- Kishida, E.; Tajiri, M.; Masuzawa, Y. Docosahexaenoic acid enrichment can reduce l929 cell necrosis induced by tumor necrosis factor. Biochim. Biophys. Acta 2006, 1761, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Lo, C.J.; Chiu, K.C.; Fu, M.; Chu, A.; Helton, S. Fish oil modulates macrophage p44/p42 mitogen-activated protein kinase activity induced by lipopolysaccharide. JPEN J. Parenter Enter. Nutr. 2000, 24, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Weldon, S.M.; Mullen, A.C.; Loscher, C.E.; Hurley, L.A.; Roche, H.M. Docosahexaenoic acid induces an anti-inflammatory profile in lipopolysaccharide-stimulated human thp-1 macrophages more effectively than eicosapentaenoic acid. J. Nutr. Biochem. 2007, 18, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Mullen, A.; Loscher, C.E.; Roche, H.M. Anti-inflammatory effects of epa and dha are dependent upon time and dose-response elements associated with lps stimulation in thp-1-derived macrophages. J. Nutr. Biochem. 2010, 21, 444–450. [Google Scholar] [CrossRef] [PubMed]
| Source | Treatment | Cell Line | Outcome |
|---|---|---|---|
| Smith, et al. [141] | 50 μM EPA 1–10 nM PIF | C2C12 | EPA ↓ protein degradation in PIF treated myoblasts |
| Magee, et al. [87] | 50 μM EPA 20 ng/mL TNF-α | C2C12 | EPA ↓ the negative effects TNF-α via ↓ NF-κB expression. EPA ↑ PPARγ expression |
| Lee, et al. [140] | 1–10 μM DHA 48 h 1–10 μM EPA 48 h | C2C12 | DHA ↑ cell proliferation at 10µM concentration only. EPA has no effect on proliferation between 0 and10 µM. EPA and DHA were no investigated in the context of differentiation in this study |
| Magee, et al. [88] | 50 μM EPA 24 h 20 TNF-α ng/mL 24 h | C2C12 | EPA ↓ the negative effects TNFα has on cell death, apoptosis and myotube formation by ↑ PPARγ expression via the NfKB signaling pathway. |
| Peng, et al. [131] | 10–100 μM DHA 24 h 10–100 μM EPA 24 h | C2C12 | EPA and DHA ↓ cell proliferation in dose dependent manner DHA and EPA ↓ protein levels of Cyclin D and Cyclin E and CDK2. EPA and DHA ↓ ERK1/2 protein expression |
| Briolay, et al. [79] | 20 μM DHA 72 h 20 μM EPA 72 h | L6 | Treating cells with EPA and DHA ↑ fusion index. This is accompanied by ↑ in p70S6K1, and membrane bund protein caveolin-3 that is associated with fusion. |
| Luo, et al. [139] | 400–600 μM EPA 10 days | C2C12 | EPA in high doses of 400–600 µM induces transdifferentiation of myoblasts to adipocytes by ↓ Wnt/β-catanin signalling through PPARγ |
| Saini, et al. [135] | 50 μM EPA 36 h & 72 h 750 μM PAL 36 h & 72 h | C2 | EPA ↓ palmitate induced cell death via affecting the MAPK pathway. EPA can protect cells from MAPK induced cell death and myogenic suppression but not against of Id3/caspase induced apoptosis EPA ↑ MyoD |
| Publication | Treatment | Outcome |
|---|---|---|
| Bryner, et al. [96] | 100 μM DHA 500 μM PAL | DHA attenuates ↓ tube size by PAL |
| Wang, et.al [69] | 300–700 μM DHA 300–700 μM EPA | EPA and DHA ↓ total protein degradation in a dose response manner DHA and EPA ↓ protein degradation by regulating the IkBα/NFκB signalling DHA is more effective in preventing degradation than EPA |
| Kamolrat and Gray [122] | 50 μM DHA 50 μM EPA | EPA ↑ MPS and ↓ MPB DHA has no effect on MPS or MPB at 50 μM |
| Lee, et al. [142] | 1–50 μM DHA, 1–50 μM EPA | DHA and EPA both ↑ UCP3 AICAR synergistically ↑ UCP3 activity when co-treated with DHA or EPA |
| Woodworth-Hobbs, et al. [143] | 100 μM DHA 500 μM PAL | DHA ↓ proteolysis by palmitate in a time dependent manner and counteracts the effects of PAL by restoring Akt activity, ↓ FOXO activity ↓ and atrogin expression. |
| Lee, et al. [91] | 1–50 μM DHA 1–50 μM EPA | EPA and DHA ↑ PGC-1α, NRF-1 and Tfam gene expression and ↑ PGC-1α promoter activity |
| Chen, et al. [144] | 50 μM DHA 50 μM EPA, 50 μM AA 750 μM PAL | The negative effects of PAL on AMPK phosphorylation GLUT4 mRNA expression and basal glucose uptake were ↓ by AA, DHA and EPA. The expression of genes associated with protein degradation was ↓ by EPA, DHA and AA |
| Pinel, et al. [95] | 50 μM DHA 50 μM EPA 50 μM ALA 500 μM PAL | EPA and DHA ↑ membrane fluidity which may improve glucose uptake.EPA and DHA ↑ key gene involved in β-oxidation. |
| Publication | Dose | Duration | Age | Sex | N | REX | Protein | Outcomes |
|---|---|---|---|---|---|---|---|---|
| Smith, et al. [33] | 1.86 g/day EPA 1.5 g/day DHA | 8 weeks | 39.7 ± 1.7 | M | 5 | - | Insulin clamp Amino Acid clamp | Hyperaminoacidemia-hyperinsulinemia induced MPS ↑ after supplementation and ↑ p-mTORSer2448 p-p70S6k1Thr389. |
| F | 4 | |||||||
| Smith, et al. [47] | 1.86 g/day EPA 1.5 g/day DHA | 8 weeks | 71 ± 1 | M | 5 | - | Insulin clamp Amino Acid clamp | Hyperaminoacidemia-hyperinsulinemia induced MPS ↑ after supplementation and ↑ p-mTORSer2448 p-p70S6k1Thr389. |
| F | 3 | |||||||
| Rodacki, et al. [34] | 0.4 g/day EPA 0.3 g/day DHA | 90 days | 64 ± 1.4 | F | 45 | 3x/week | - | Fish Oil supplementation and strength training results in ↑ improvements in peak torque and rate of torque development than strength training alone. |
| 150 days | 64 ± 1.4 | F | 45 | 3x/week | ||||
| McGlory, et al. [106] | 3.5 g/ay EPA 0.9 g/day DHA | 4 weeks | 21.3 ± 3 | M | 10 | - | - | EPA and DHA in the blood ↑ from week 0 to 2 and ↔ from 2 to 4 weeks. Levels of EPA and DHA in skeletal muscle are ↑ from 0 to 2 weeks and further ↑ 2 to 4 weeks. |
| Smith, et al. [32] | 1.86 g/day EPA 1.5 g/day DHA | 26 weeks | 68 ± 5 | M | 10 | - | - | ↑ thigh muscle volume, ↑ hand grip strength and ↑1RM |
| F | 19 | |||||||
| McGlory, et al. [46] | 3.5 g/day EPA 0.9 g/day DHA 0.1 g/day DPA | 8 weeks | 24 ± 0 | M | 9 | Acute bout | 30 g Whey | Compared to placebo no change in myofibrillar MPS following an acute bout of REX and protein ingestion ↓ p70S6K1 and Akt anabolic kinases following REX and protein ingestion. |
| Da Boit, et al. [78] | 2.1 g/day EPA 0.6 g/day DHA | 18 weeks | 70.6 ± 4.5 | M | 27 | 2x/week | - | ↑ Maximal isometric torque and ↑ muscle quality in women after exercise training in n-3 PUFA group than in the placebo group, with no such differences in men |
| 70.7 ± 3.3 | F | 23 | ||||||
| Lalia, et al. [145] | 0.675 g/day EPA 0.3 g/day DHA | 16 weeks | 27 ± 5 | M/F | 12 | Acute bout | - | Myofibrillar MPS ↑ in both young and old after exercise and supplementation whereas mitoMPS and sarcoMPS ↑ in the older participants |
| 76 ± 5 | M/F | 12 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tachtsis, B.; Camera, D.; Lacham-Kaplan, O. Potential Roles of n-3 PUFAs during Skeletal Muscle Growth and Regeneration. Nutrients 2018, 10, 309. https://doi.org/10.3390/nu10030309
Tachtsis B, Camera D, Lacham-Kaplan O. Potential Roles of n-3 PUFAs during Skeletal Muscle Growth and Regeneration. Nutrients. 2018; 10(3):309. https://doi.org/10.3390/nu10030309
Chicago/Turabian StyleTachtsis, Bill, Donny Camera, and Orly Lacham-Kaplan. 2018. "Potential Roles of n-3 PUFAs during Skeletal Muscle Growth and Regeneration" Nutrients 10, no. 3: 309. https://doi.org/10.3390/nu10030309
APA StyleTachtsis, B., Camera, D., & Lacham-Kaplan, O. (2018). Potential Roles of n-3 PUFAs during Skeletal Muscle Growth and Regeneration. Nutrients, 10(3), 309. https://doi.org/10.3390/nu10030309






