Effects of Diets Differing in Composition of 18-C Fatty Acids on Adipose Tissue Thermogenic Gene Expression in Mice Fed High-Fat Diets
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Diets
2.2. Fatty Acid Composition of Diets
2.3. Indirect Calorimetry
2.4. Intraperitoneal Glucose Tolerance Test
2.5. Lipid, Glucose, and Insulin Concentrations
2.6. Quantitative Real-Time PCR
2.7. Statistical Analysis
3. Results
3.1. Body Weight, Calorie Intake, Growth Efficiency, and Tissue Weight
3.2. Indirect Calorimetry
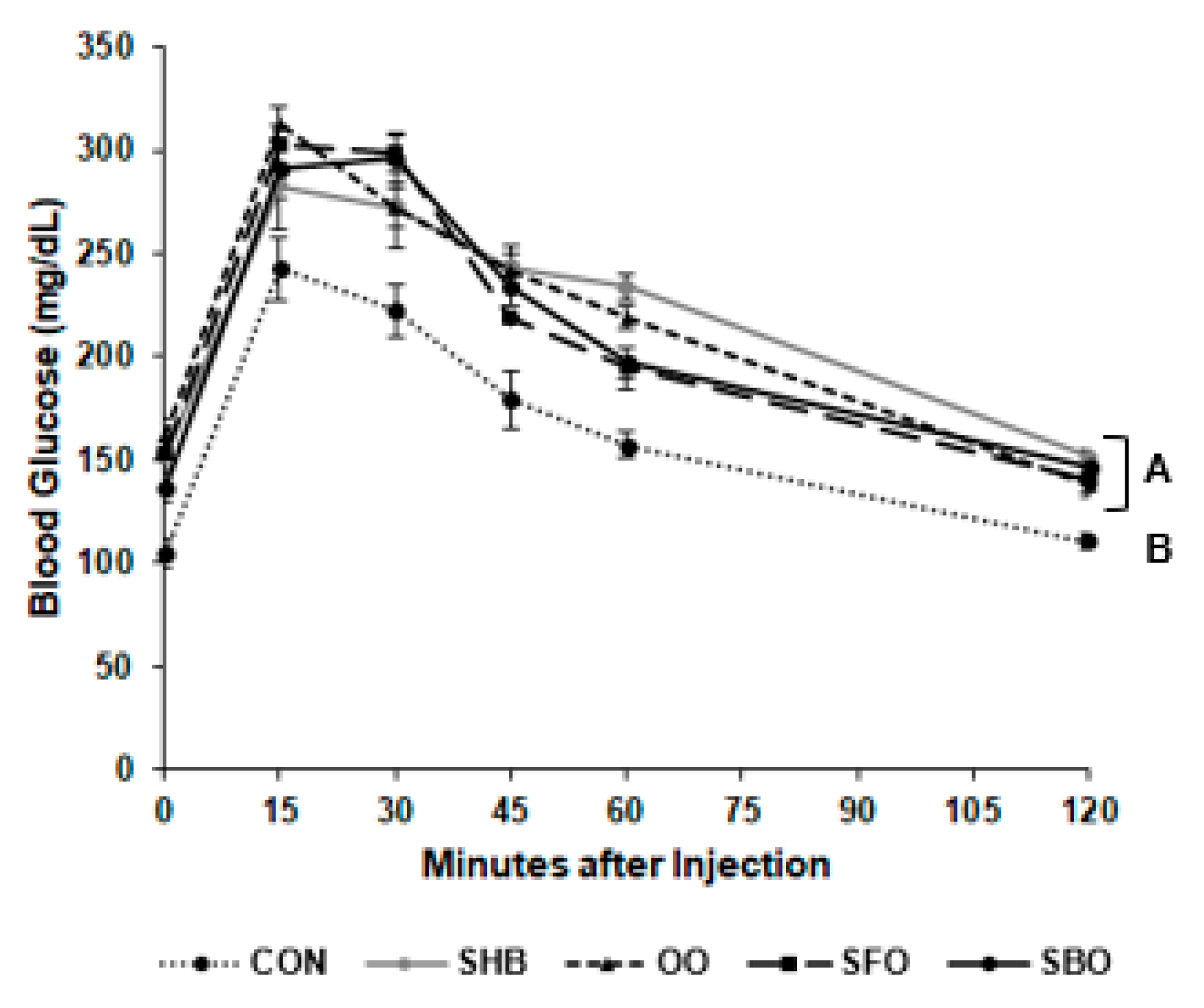
3.3. Intraperitoneal Glucose Tolerance Test
3.4. Lipid, Glucose, and Insulin Concentrations
3.5. Protein Expression
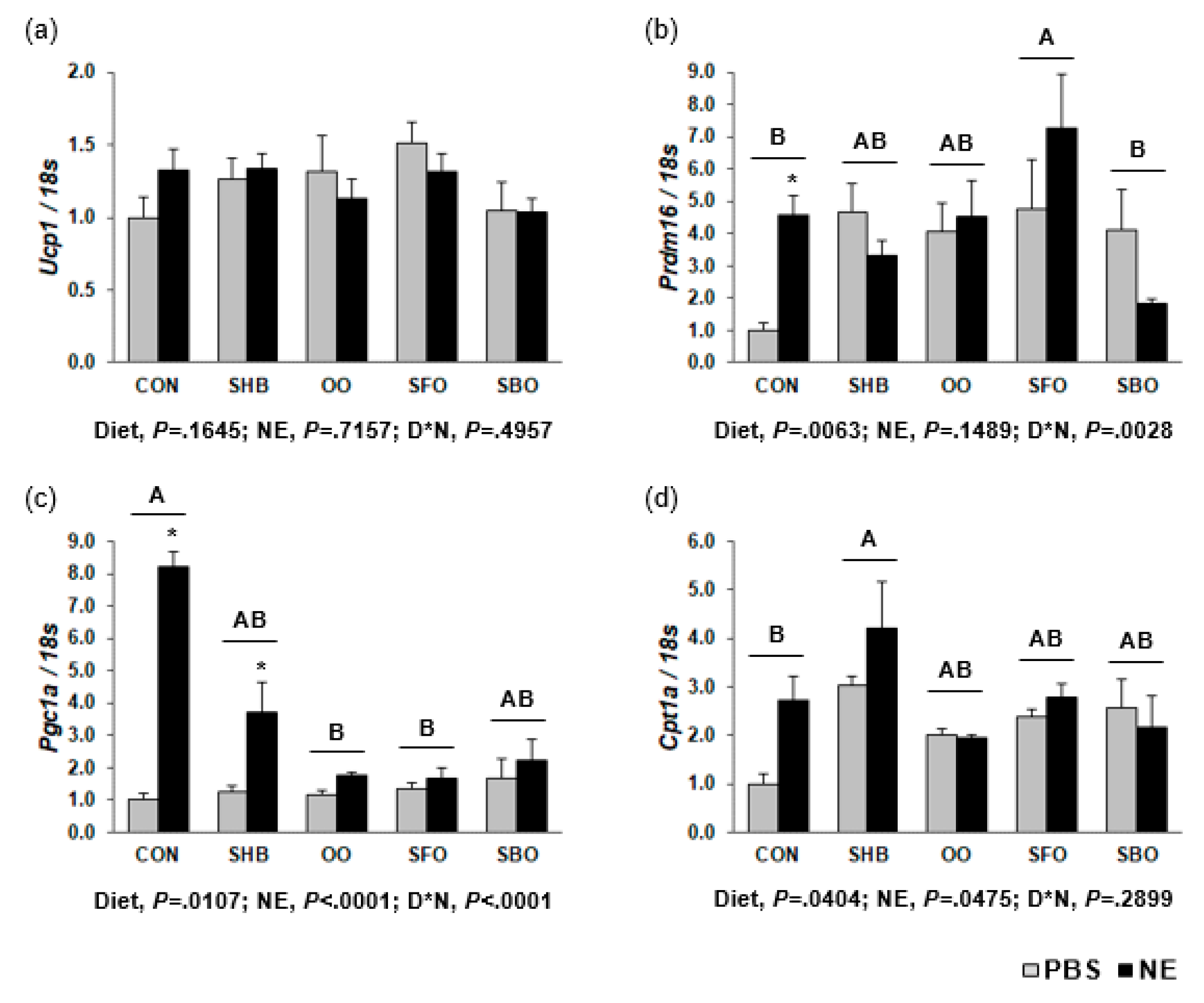
3.6. Regulation of Expression of Genes Involved in Thermogenesis in BAT
3.7. Effect of Diets on Thermogenic Gene Expression in sWAT
3.8. Effect of Diets on Thermogenic Gene Expression in eWAT
3.9. Dietary Effects on Expression of Genes Involved in Lipid Metabolism in the Liver
3.10. Regulation of Expression of Genes Involved in Lipid Metabolism in Hamstring Muscle
3.11. Expression of Neuropeptides and β-Adrenergic Receptors in Hypothalamus in Response to Diets
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Camp, H.S.; Ren, D.; Leff, T. Adipogenesis and fat-cell function in obesity and diabetes. Trends Mol. Med. 2002, 8, 442–447. [Google Scholar] [CrossRef]
- Must, A.; Spadano, J.; Coakley, E.H.; Field, A.E.; Colditz, G.; Dietz, W.H. The disease burden associated with overweight and obesity. JAMA 1999, 282, 1523–1529. [Google Scholar] [CrossRef] [PubMed]
- Madsen, L.; Petersen, R.K.; Kristiansen, K. Regulation of adipocyte differentiation and function by polyunsaturated fatty acids. Mol. Basis Dis. 2005, 1740, 266–286. [Google Scholar] [CrossRef] [PubMed]
- Forman, B.M.; Chen, J.; Evans, R.M. Hypolipidemic drugs, polyunsaturated fatty acids, and eicosanoids are ligands for peroxisome proliferator-activated receptors α and δ. Proc. Natl. Acad. Sci. USA 1997, 94, 4312–4317. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, T.; Shimano, H.; Yahagi, N.; Ide, T.; Amemiya-Kudo, M.; Matsuzaka, T.; Nakakuki, M.; Tomita, S.; Okazaki, H.; Tamura, Y. Polyunsaturated fatty acids suppress sterol regulatory element-binding protein 1c promoter activity by inhibition of liver X receptor (LXR) binding to LXR response elements. J. Biol. Chem. 2002, 277, 1705–1711. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Takahashi, M.; Ezaki, O. Fish oil feeding decreases mature sterol regulatory element-binding protein 1 (SREBP-1) by down-regulation of SREBP-1c mRNA in mouse liver a possible mechanism for down-regulation of lipogenic enzyme mRNAs. J. Biol. Chem. 1999, 274, 25892–25898. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Chen, X. Eicosapentaenoic acid promotes thermogenic and fatty acid storage capacity in mouse subcutaneous adipocytes. Biochem. Biophys. Res. Commun. 2014, 450, 1446–1451. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Okla, M.; Erickson, A.; Carr, T.; Natarajan, S.K.; Chung, S. Eicosapentaenoic acid potentiates brown thermogenesis through FFAR4-dependent up-regulation of miR-30b and miR-378. J. Biol. Chem. 2016, 291, 20551–20562. [Google Scholar] [CrossRef] [PubMed]
- Bargut, T.C.L.; Silva-e, A.C.A.G.; Souza-Mello, V.; Mandarim-de-Lacerda, C.A.; Aguila, M.B. Mice fed fish oil diet and upregulation of brown adipose tissue thermogenic markers. Eur. J. Nutr. 2016, 55, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Goto, T.; Yu, R.; Uchida, K.; Tominaga, M.; Kano, Y.; Takahashi, N.; Kawada, T. Fish oil intake induces UCP1 upregulation in brown and white adipose tissue via the sympathetic nervous system. Sci. Rep. 2015, 5, 18013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cannon, B.; Nedergaard, J. Brown adipose tissue: Function and physiological significance. Physiol. Rev. 2004, 84, 277–359. [Google Scholar] [CrossRef] [PubMed]
- Harms, M.; Seale, P. Brown and beige fat: Development, function and therapeutic potential. Nat. Med. 2013, 19, 1252–1263. [Google Scholar] [CrossRef] [PubMed]
- Wrolstad, R.E. Current Protocols in Food Analytical Chemistry; Wiley: Hoboken, NJ, USA, 2001. [Google Scholar]
- Rodríguez, V.M.; Portillo, M.P.; Picó, C.; Macarulla, M.T.; Palou, A. Olive oil feeding up-regulates uncoupling protein genes in rat brown adipose tissue and skeletal muscle. Am. J. Clin. Nutr. 2002, 75, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.J.; Jew, S.; AbuMweis, S. The effect of dietary oleic, linoleic, and linolenic acids on fat oxidation and energy expenditure in healthy men. Metabolism 2008, 57, 1198–1203. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Kastorini, C.-M.; Panagiotakos, D.B.; Giugliano, D. Mediterranean diet and weight loss: Meta-analysis of randomized controlled trials. Metab. Syndr. Relat. Disord. 2011, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.-H.; Gerhart-Hines, Z.; Dominy, J.E.; Lee, Y.; Kim, S.; Tabata, M.; Xiang, Y.K.; Puigserver, P. Oleic acid stimulates complete oxidation of fatty acids through protein kinase a-dependent activation of SIRT1-PGC1A complex. J. Biol. Chem. 2013, 288, 7117–7126. [Google Scholar] [CrossRef] [PubMed]
- Pisani, D.F.; Ghandour, R.A.; Beranger, G.E.; Le Faouder, P.; Chambard, J.-C.; Giroud, M.; Vegiopoulos, A.; Djedaini, M.; Bertrand-Michel, J.; Tauc, M. The ω6-fatty acid, arachidonic acid, regulates the conversion of white to brite adipocyte through a prostaglandin/calcium mediated pathway. Mol. Metab. 2014, 3, 834–847. [Google Scholar] [CrossRef] [PubMed]
- Baillie, R.; Takada, R.; Nakamura, M.; Clarke, S. Coordinate induction of peroxisomal acyl-CoA oxidase and UCP-3 by dietary fish oil: A mechanism for decreased body fat deposition. Prostaglandins Leukot. Essent. Fat. Acids 1999, 60, 351–356. [Google Scholar] [CrossRef]
- Nuernberg, K.; Breier, B.H.; Jayasinghe, S.N.; Bergmann, H.; Thompson, N.; Nuernberg, G.; Dannenberger, D.; Schneider, F.; Renne, U.; Langhammer, M. Metabolic responses to high-fat diets rich in n-3 or n-6 long-chain polyunsaturated fatty acids in mice selected for either high body weight or leanness explain different health outcomes. Nutr. Metab. 2011, 8, 56. [Google Scholar] [CrossRef] [PubMed]
- Alvheim, A.R.; Torstensen, B.E.; Lin, Y.H.; Lillefosse, H.H.; Lock, E.-J.; Madsen, L.; Hibbeln, J.R.; Malde, M.K. Dietary linoleic acid elevates endogenous 2-arachidonoylglycerol and anandamide in atlantic salmon (Salmo salar L.) and mice, and induces weight gain and inflammation in mice. Br. J. Nutr. 2013, 109, 1508–1517. [Google Scholar] [CrossRef] [PubMed]
- Massiera, F.; Saint-Marc, P.; Seydoux, J.; Murata, T.; Kobayashi, T.; Narumiya, S.; Guesnet, P.; Amri, E.-Z.; Negrel, R.; Ailhaud, G. Arachidonic acid and prostacyclin signaling promote adipose tissue development a human health concern? J. Lipid Res. 2003, 44, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Alvheim, A.R.; Torstensen, B.E.; Lin, Y.H.; Lillefosse, H.H.; Lock, E.-J.; Madsen, L.; Frøyland, L.; Hibbeln, J.R.; Malde, M.K. Dietary linoleic acid elevates the endocannabinoids 2-AG and anandamide and promotes weight gain in mice fed a low fat diet. Lipids 2014, 49, 59–69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plourde, M.; Cunnane, S.C. Extremely limited synthesis of long chain polyunsaturates in adults: Implications for their dietary essentiality and use as supplements. Appl. Physiol. Nutr. Metab. 2007, 32, 619–634. [Google Scholar] [CrossRef] [PubMed]
- Talahalli, R.R.; Vallikannan, B.; Sambaiah, K.; Lokesh, B.R. Lower efficacy in the utilization of dietary ALA as compared to preformed EPA+ DHA on long chain n-3 PUFA levels in rats. Lipids 2010, 45, 799–808. [Google Scholar] [CrossRef] [PubMed]
- Bachman, E.S.; Dhillon, H.; Zhang, C.-Y.; Cinti, S.; Bianco, A.C.; Kobilka, B.K.; Lowell, B.B. βAR signaling required for diet-induced thermogenesis and obesity resistance. Science 2002, 297, 843–845. [Google Scholar] [CrossRef] [PubMed]
- Nonogaki, K. New insights into sympathetic regulation of glucose and fat metabolism. Diabetologia 2000, 43, 533–549. [Google Scholar] [CrossRef] [PubMed]
- Collins, S.; Daniel, K.W.; Petro, A.E.; Surwit, R.S. Strain-specific response to β 3-adrenergic receptor agonist treatment of diet-induced obesity in mice. Endocrinology 1997, 138, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Ahren, B.; Bengtsson, H.; Hedner, P. Effects of norepinephrine on basal and thyrotropin-stimulated thyroid hormone secretion in the mouse. Endocrinology 1986, 119, 1058–1062. [Google Scholar] [CrossRef] [PubMed]
- Rubio, A.; Raasmaja, A.; Silva, J.E. Thyroid hormone and norepinephrine signaling in brown adipose tissue. Ii: Differential effects of thyroid hormone on β 3-adrenergic receptors in brown and white adipose tissue. Endocrinology 1995, 136, 3277–3284. [Google Scholar] [CrossRef] [PubMed]
- Carpéné, C.; Galitzky, J.; Collon, P.; Esclapez, F.; Dauzats, M.; Lafontan, M. Desensitization of β-1 and β-2, but not β-3, adrenoceptor-mediated lipolytic responses of adipocytes after long-term norepinephrine infusion. J. Pharmacol. Exp. Ther. 1993, 265, 237–247. [Google Scholar] [PubMed]
- Feve, B.; Elhadri, K.; Quignard-Boulange, A.; Pairault, J. Transcriptional down-regulation by insulin of the beta 3-adrenergic receptor expression in 3T3-F442A adipocytes: A mechanism for repressing the camp signaling pathway. Proc. Natl. Acad. Sci. USA 1994, 91, 5677–5681. [Google Scholar] [CrossRef] [PubMed]
- Feve, B.; Baude, B.; Krief, S.; Strosberg, A.; Pairault, J.; Emorine, L. Inhibition by dexamethasone of beta 3-adrenergic receptor responsiveness in 3T3-F442A adipocytes. Evidence for a transcriptional mechanism. J. Biol. Chem. 1992, 267, 15909–15915. [Google Scholar] [PubMed]
- Bakopanos, E.; Silva, J.E. Thiazolidinediones inhibit the expression of beta3-adrenergic receptors at a transcriptional level. Diabetes 2000, 49, 2108–2115. [Google Scholar] [CrossRef] [PubMed]
- Cousin, B.; Cinti, S.; Morroni, M.; Raimbault, S.; Ricquier, D.; Penicaud, L.; Casteilla, L. Occurrence of brown adipocytes in rat white adipose tissue: Molecular and morphological characterization. J. Cell Sci. 1992, 103, 931–942. [Google Scholar] [PubMed]
- Wu, J.; Boström, P.; Sparks, L.M.; Ye, L.; Choi, J.H.; Giang, A.-H.; Khandekar, M.; Virtanen, K.A.; Nuutila, P.; Schaart, G. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012, 150, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-H.; Petkova, A.P.; Mottillo, E.P.; Granneman, J.G. In vivo identification of bipotential adipocyte progenitors recruited by β3-adrenoceptor activation and high-fat feeding. Cell Metab. 2012, 15, 480–491. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.V.; Bikopoulos, G.; Hung, S.; Ceddia, R.B. Thermogenic capacity is antagonistically regulated in classical brown and white subcutaneous fat depots by high fat diet and endurance training in rats impact on whole-body energy expenditure. J. Biol. Chem. 2014, 289, 34129–34140. [Google Scholar] [CrossRef] [PubMed]
- Carey, A.L.; Vorlander, C.; Reddy-Luthmoodoo, M.; Natoli, A.K.; Formosa, M.F.; Bertovic, D.A.; Anderson, M.J.; Duffy, S.J.; Kingwell, B.A. Reduced UCP-1 content in in vitro differentiated beige/brite adipocytes derived from preadipocytes of human subcutaneous white adipose tissues in obesity. PLoS ONE 2014, 9, e91997. [Google Scholar] [CrossRef] [PubMed]
- Carey, A.L.; Formosa, M.F.; Van Every, B.; Bertovic, D.; Eikelis, N.; Lambert, G.W.; Kalff, V.; Duffy, S.J.; Cherk, M.H.; Kingwell, B.A. Ephedrine activates brown adipose tissue in lean but not obese humans. Diabetologia 2013, 56, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Orava, J.; Nuutila, P.; Noponen, T.; Parkkola, R.; Viljanen, T.; Enerbäck, S.; Rissanen, A.; Pietiläinen, K.H.; Virtanen, K.A. Blunted metabolic responses to cold and insulin stimulation in brown adipose tissue of obese humans. Obesity 2013, 21, 2279–2287. [Google Scholar] [CrossRef] [PubMed]
- Kurylowicz, A.; Jonas, M.; Lisik, W.; Jonas, M.; Wicik, Z.A.; Wierzbicki, Z.; Chmura, A.; Puzianowska-Kuznicka, M. Obesity is associated with a decrease in expression but not with the hypermethylation of thermogenesis-related genes in adipose tissues. J. Transl. Med. 2015, 13, 31. [Google Scholar] [CrossRef] [PubMed]
- Collins, S.; Surwit, R.S. The beta-adrenergic receptors and the control of adipose tissue metabolism and thermogenesis. Recent Prog. Horm. Res. 2001, 56, 309–328. [Google Scholar] [CrossRef] [PubMed]
- Kazak, L.; Chouchani, E.T.; Jedrychowski, M.P.; Erickson, B.K.; Shinoda, K.; Cohen, P.; Vetrivelan, R.; Lu, G.Z.; Laznik-Bogoslavski, D.; Hasenfuss, S.C. A creatine-driven substrate cycle enhances energy expenditure and thermogenesis in beige fat. Cell 2015, 163, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, K.; Kang, Q.; Yoneshiro, T.; Camporez, J.P.; Maki, H.; Homma, M.; Shinoda, K.; Chen, Y.; Lu, X.; Maretich, P. UCP1-independent signaling involving SERCA2b-mediated calcium cycling regulates beige fat thermogenesis and systemic glucose homeostasis. Nat. Med. 2017, 23, 1454. [Google Scholar] [CrossRef] [PubMed]
- Sunny, N.E.; Parks, E.J.; Browning, J.D.; Burgess, S.C. Excessive hepatic mitochondrial TCA cycle and gluconeogenesis in humans with nonalcoholic fatty liver disease. Cell Metab. 2011, 14, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Iozzo, P.; Bucci, M.; Roivainen, A.; Någren, K.; Järvisalo, M.J.; Kiss, J.; Guiducci, L.; Fielding, B.; Naum, A.G.; Borra, R. Fatty acid metabolism in the liver, measured by positron emission tomography, is increased in obese individuals. Gastroenterology 2010, 139, 846–856. [Google Scholar] [CrossRef] [PubMed]
- Ryan, M.C.; Itsiopoulos, C.; Thodis, T.; Ward, G.; Trost, N.; Hofferberth, S.; O’Dea, K.; Desmond, P.V.; Johnson, N.A.; Wilson, A.M. The mediterranean diet improves hepatic steatosis and insulin sensitivity in individuals with non-alcoholic fatty liver disease. J. Hepatol. 2013, 59, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Araya, J.; Rodrigo, R.; Videla, L.A.; Thielemann, L.; Orellana, M.; Pettinelli, P.; Poniachik, J. Increase in long-chain polyunsaturated fatty acid n-6/n-3 ratio in relation to hepatic steatosis in patients with non-alcoholic fatty liver disease. Clin. Sci. 2004, 106, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Storlien, L.H.; Jenkins, A.B.; Chisholm, D.J.; Pascoe, W.S.; Khouri, S.; Kraegen, E.W. Influence of dietary fat composition on development of insulin resistance in rats: Relationship to muscle triglyceride and ω-3 fatty acids in muscle phospholipid. Diabetes 1991, 40, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Bi, S.; Li, L. Browning of white adipose tissue: Role of hypothalamic signaling. Ann. N. Y. Acad. Sci. 2013, 1302, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Hammamieh, R.; Chakraborty, N.; Gautam, A.; Miller, S.-A.; Muhie, S.; Meyerhoff, J.; Jett, M. Transcriptomic analysis of the effects of a fish oil enriched diet on murine brains. PLoS ONE 2014, 9, e90425. [Google Scholar] [CrossRef] [PubMed]
- Begg, D.P.; Puskás, L.G.; Kitajka, K.; Ménesi, D.; Allen, A.M.; Li, D.; Mathai, M.L.; Shi, J.R.; Sinclair, A.J.; Weisinger, R.S. Hypothalamic gene expression in ω-3 pufa-deficient male rats before, and following, development of hypertension. Hypertens. Res. 2012, 35, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Richard, J.E.; López-Ferreras, L.; Chanclón, B.; Eerola, K.; Micallef, P.; Skibicka, K.P.; Asterholm, I.W. Cns β3-adrenergic receptor activation regulates feeding behavior, white fat browning and body weight. Am. J. Physiol. 2017, 313, E344–E358. [Google Scholar] [CrossRef] [PubMed]



| 10% Fat | 45% Fat | ||||
|---|---|---|---|---|---|
| CON | SHB | OO | SFO | SBO | |
| Myristic acid (C14:0) | 1.046 | 0.728 | 0.681 | 0.709 | 0.685 |
| Palmitic acid (C16:0) | 17.148 | 13.287 | 18.595 | 13.887 | 15.740 |
| Palmitoleic acid (C16:1, ∆9) | 0.976 | 0.998 | 1.863 | 0.963 | 0.944 |
| Stearic acid (C18:0) | 8.604 | 23.781 | 7.503 | 7.144 | 7.808 |
| Oleic acid (C18:1, ∆9) | 27.793 | 38.828 | 44.971 | 27.233 | 28.916 |
| Linoleic acid (C18:2, ∆9,12) | 32.187 | 15.201 | 18.772 | 43.238 | 35.580 |
| α-Linolenic acid (C18:3, ∆9,12,15) | 3.374 | 0.858 | 1.023 | 0.817 | 3.731 |
| Arachidonic acid (C20:4, ∆5,8,11,14) | 0.142 | 0.159 | 0.148 | 0.149 | 0.154 |
| Eicosapentaenoic acid (C20:5, ∆5,8,11,14,17) | 0.100 | 0.011 | 0.022 | 0.066 | 0.032 |
| Docosahexaenoic acid (C22:6, ∆4,7,10,13,16,19) | 0.072 | 0.044 | 0.037 | 0.060 | 0.062 |
| Total saturated fatty acids | 27.552 | 38.422 | 27.498 | 22.465 | 24.986 |
| Total monounsaturated fatty acids | 30.803 | 41.411 | 49.470 | 29.937 | 31.784 |
| Total polyunsaturated fatty acids | 37.663 | 18.221 | 21.519 | 45.818 | 41.364 |
| Total n-6 fatty acids | 32.976 | 16.031 | 19.549 | 44.059 | 36.412 |
| Total n-3 fatty acids | 3.822 | 1.622 | 1.397 | 1.213 | 4.097 |
| n-6:n-3 ratio | 8.628 | 9.883 | 13.994 | 36.322 | 8.887 |
| 10% Fat | 45% Fat | P-Value | ||||
|---|---|---|---|---|---|---|
| CON | SHB | OO | SFO | SBO | ||
| Body weight at 0 week (g) | 17.88 ± 0.61 | 17.73 ± 0.51 | 17.83 ± 0.50 | 17.89 ± 0.45 | 17.88 ± 0.62 | 0.9931 |
| Body weight at 12 week (g) | 26.00 ± 0.69 C | 35.79 ± 0.81 A | 29.33 ± 0.66 BC | 30.41 ± 0.90 BC | 31.95 ± 1.93 AB | <0.0001 |
| Body weight gain (g) | 8.13 ± 0.59 C | 18.06 ± 0.71 A | 11.50 ± 0.73 BC | 12.33 ± 0.93 B | 14.08 ± 1.47 B | <0.0001 |
| Calorie intake (kcal/day) | 9.27 ± 0.28 B | 12.97 ± 0.02 A | 10.68 ± 0.09 AB | 10.43 ± 0.19 B | 11.14 ± 0.04 AB | 0.0119 |
| Growth efficiency (mg/kcal) 1 | 10.39 ± 0.57 B | 16.58 ± 0.65 A | 12.82 ± 0.82 AB | 14.01 ± 0.89 AB | 15.03 ± 1.57 A | 0.0011 |
| Subcutaneous WAT (g/100 g body weight) | 0.59 ± 0.09 B | 2.60 ± 0.46 A | 1.25 ± 0.17 AB | 1.88 ± 0.45 AB | 2.43 ± 0.64 A | 0.0081 |
| Epidydimal WAT (g/100 g body weight) | 1.38 ± 0.18 C | 3.80 ± 0.42 A | 2.12 ± 0.21 BC | 2.79 ± 0.51 ABC | 3.18 ± 0.48 AB | 0.0010 |
| Brown adipose tissue (g/100 g body weight) | 0.34 ± 0.02 AB | 0.38 ± 0.04 A | 0.20 ± 0.02 C | 0.24 ± 0.03 BC | 0.36 ± 0.05 AB | 0.0009 |
| Liver (g/100 g body weight) | 3.72 ± 0.18 | 3.36 ± 0.12 | 3.33 ± 0.06 | 3.22 ± 0.11 | 3.21 ± 0.19 | 0.0956 |
| Thigh (g/100 g body weight) | 0.86 ± 0.04 | 0.80 ± 0.05 | 1.05 ± 0.06 | 0.95 ± 0.08 | 1.06 ± 0.14 | 0.1160 |
| TRT 1 | 10% Fat | 45% Fat | TRT Mean 3 | P-Value 4 | ||||
|---|---|---|---|---|---|---|---|---|
| CON | SHB | OO | SFO | SBO | ||||
| Serum TAG (mg/mL) | PBS | 0.87 ± 0.05 | 0.69 ± 0.08 | 0.98 ± 0.20 | 1.00 ± 0.12 | 0.85 ± 0.09 | 0.87 ± 0.05 | D; 0.9803 |
| NE | 1.01 ± 0.17 | 1.21 ± 0.08 | 0.97 ± 0.20 | 0.98 ± 0.10 | 1.07 ± 0.12 | 1.05 ± 0.06 * | N; 0.0374 | |
| Diet mean 2 | 0.94 ± 0.09 | 0.95 ± 0.11 | 0.97 ± 0.13 | 0.99 ± 0.07 | 0.96 ± 0.08 | D*N; 0.1894 | ||
| Liver TAG (mg/mg protein) | PBS | 0.19 ± 0.04 | 0.22 ± 0.01 | 0.17 ± 0.04 | 0.21 ± 0.05 | 0.21 ± 0.07 | 0.20 ± 0.02 | D; 0.5666 |
| NE | 0.28 ± 0.04 | 0.36 ± 0.03 | 0.30 ± 0.01 | 0.35 ± 0.05 | 0.39 ± 0.10 | 0.34 ± 0.02 * | N; 0.0002 | |
| Diet mean 2 | 0.23 ± 0.03 | 0.29 ± 0.03 | 0.24 ± 0.03 | 0.28 ± 0.04 | 0.30 ± 0.07 | D*N; 0.9033 | ||
| Muscle TAG (mg/mg protein) | PBS | 0.21 ± 0.03 | 0.16 ± 0.02 | 0.20 ± 0.02 | 0.24 ± 0.03 | 0.22 ± 0.02 | 0.20 ± 0.01 | D; 0.0012 |
| NE | 0.16 ± 0.01 | 0.16 ± 0.02 | 0.24 ± 0.02 | 0.34 ± 0.06 | 0.19 ± 0.03 | 0.22 ± 0.02 | N; 0.8868 | |
| Diet mean 2 | 0.18 ± 0.02 B | 0.16 ± 0.01 B | 0.22 ± 0.02 AB | 0.30 ± 0.04 A | 0.20 ± 0.02 AB | D*N; 0.1315 | ||
| Serum NEFA (mM) | PBS | 0.077 ± 0.003 | 0.058 ± 0.006 | 0.057 ± 0.007 | 0.062 ± 0.004 | 0.066 ± 0.006 | 0.064 ± 0.003 | D; 0.0146 |
| NE | 0.087 ± 0.009 | 0.067 ± 0.005 | 0.070 ± 0.005 | 0.074 ± 0.007 | 0.079 ± 0.003 | 0.075 ± 0.003 * | N; 0.0044 | |
| Diet mean 2 | 0.082 ± 0.005 A | 0.062 ± 0.004 B | 0.063 ± 0.005 B | 0.068 ± 0.004 AB | 0.073 ± 0.004 AB | D*N; 0.9844 | ||
| Serum Glucose (mg/dL) | PBS | 118.3 ± 27.2 | 280.2 ± 41.3 | 252.5 ± 50.8 | 340.7 ± 41.6 | 162.7 ± 9.7 | 230.9 ± 23.6 | D; 0.0003 |
| NE | 101.0 ± 11.0 | 179.5 ± 49.7 | 114.5 ± 40.0 | 194.7 ± 22.2 | 104.0 ± 13.8 | 138.7 ± 15.4 * | N; 0.0002 | |
| Diet mean 2 | 109.7 ± 14.0 C | 229.9 ± 35.4 AB | 183.5 ± 39.7 ABC | 267.7 ± 35.2 A | 133.4 ± 13.6 BC | D*N; 0.3867 | ||
| Serum Insulin (ng/mL) | PBS | 0.20 ± 0.02 | 2.12 ± 0.86 | 0.99 ± 0.23 | 1.19 ± 0.51 | 1.12 ± 0.38 | 1.12 ± 0.25 | D; 0.0036 |
| NE | 0.38 ± 0.03 | 1.24 ± 0.29 | 0.35 ± 0.17 | 0.91 ± 0.28 | 0.93 ± 0.37 | 0.76 ± 0.13 | N; 0.2604 | |
| Diet mean 2 | 0.30 ± 0.04 B | 1.62 ± 0.40 A | 0.62 ± 0.16 AB | 1.03 ± 0.25 A | 1.01 ± 0.25 AB | D*N; 0.2493 | ||
| Glucose: Insulin ratio | PBS | 6.07 ± 1.19 | 2.65 ± 1.55 | 3.22 ± 1.19 | 3.94 ± 1.52 | 2.10 ± 1.07 | 3.60 ± 0.62 | D; 0.1321 |
| NE | 2.68 ± 0.35 | 1.46 ± 0.17 | 3.34 ± 1.67 | 2.69 ± 0.74 | 2.10 ± 0.86 | 2.45 ± 0.31 | N; 0.3044 | |
| Diet mean 2 | 4.13 ± 0.84 | 1.97 ± 0.64 | 3.29 ± 0.69 | 3.22 ± 0.74 | 2.10 ± 0.61 | D*N; 0.7640 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shin, S.; Ajuwon, K.M. Effects of Diets Differing in Composition of 18-C Fatty Acids on Adipose Tissue Thermogenic Gene Expression in Mice Fed High-Fat Diets. Nutrients 2018, 10, 256. https://doi.org/10.3390/nu10020256
Shin S, Ajuwon KM. Effects of Diets Differing in Composition of 18-C Fatty Acids on Adipose Tissue Thermogenic Gene Expression in Mice Fed High-Fat Diets. Nutrients. 2018; 10(2):256. https://doi.org/10.3390/nu10020256
Chicago/Turabian StyleShin, Sunhye, and Kolapo M. Ajuwon. 2018. "Effects of Diets Differing in Composition of 18-C Fatty Acids on Adipose Tissue Thermogenic Gene Expression in Mice Fed High-Fat Diets" Nutrients 10, no. 2: 256. https://doi.org/10.3390/nu10020256
APA StyleShin, S., & Ajuwon, K. M. (2018). Effects of Diets Differing in Composition of 18-C Fatty Acids on Adipose Tissue Thermogenic Gene Expression in Mice Fed High-Fat Diets. Nutrients, 10(2), 256. https://doi.org/10.3390/nu10020256





