Effectiveness and Safety of a Probiotic-Mixture for the Treatment of Infantile Colic: A Double-Blind, Randomized, Placebo-Controlled Clinical Trial with Fecal Real-Time PCR and NMR-Based Metabolomics Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Patients
2.2. Analysis of Infant Colic Symptoms
2.3. Analysis of Infant Stool Samples
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| IBS | irritable bowel syndrome |
| CFU | colony forming units |
| NICU | Neonatal Intensive Care Unit |
| 1H-NMR | Proton Nuclear Magnetic Resonance |
| SCFAs | short chain fatty acids |
References
- Lucassen, P.L.; Assendelft, W.J.; van Eijk, J.T.; Gubbels, J.W.; Douwes, A.C.; van Geldrop, W.J. Systematic review of the occurrence of infantile colic in the community. Arch. Dis. Child. 2001, 84, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Pace, C.A. Infantile Colic: What to Know for the Primary Care Setting. Clin. Pediatr. (Phila.) 2017, 56, 616–618. [Google Scholar] [CrossRef] [PubMed]
- Hyman, P.E.; Milla, P.J.; Benninga, M.A.; Davidson, G.P.; Fleisher, D.F.; Taminiau, J. Childhood functional gastrointestinal disorders: Neonate/toddler. Gastroenterology 2006, 130, 1519–1526. [Google Scholar] [CrossRef] [PubMed]
- Halpern, R.; Coelho, R. Excessive crying in infants. J. Pediatr. (Rio J.) 2016, 92, S40–S45. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, M.; Park, S.-Y.; Scarpato, E.; Staiano, A. Exploring hypotheses and rationale for causes of infantile colic. Neurogastroenterol. Motil. 2017, 29, e12943. [Google Scholar] [CrossRef] [PubMed]
- Indrio, F.; Di Mauro, A.; Di Mauro, A.; Riezzo, G.; Panza, R.; Cavallo, L.; Francavilla, R. Prevention of functional gastrointestinal disorders in neonates: Clinical and socioeconomic impact. Benef. Microbes 2015, 6, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Savino, F.; Ceratto, S.; De Marco, A.; Cordero di Montezemolo, L. Looking for new treatments of Infantile Colic. Ital. J. Pediatr. 2014, 40, 53. [Google Scholar] [CrossRef] [PubMed]
- Barnes, D.; Yeh, A.M. Bugs and Guts: Practical Applications of Probiotics for Gastrointestinal Disorders in Children. Nutr. Clin. Pract. 2015, 30, 747–759. [Google Scholar] [CrossRef] [PubMed]
- Cardile, S.; Alterio, T.; Arrigo, T.; Salpietro, C. Role of prebiotics and probiotics in pediatric diseases. Minerva Pediatr. 2016, 68, 487–497. [Google Scholar] [PubMed]
- Xu, M.; Wang, J.; Wang, N.; Sun, F.; Wang, L.; Liu, X.-H. The Efficacy and Safety of the Probiotic Bacterium Lactobacillus reuteri DSM 17938 for Infantile Colic: A Meta-Analysis of Randomized Controlled Trials. PLoS ONE 2015, 10, e0141445. [Google Scholar] [CrossRef] [PubMed]
- Sung, V.; D’Amico, F.; Cabana, M.D.; Chau, K.; Koren, G.; Savino, F.; Szajewska, H.; Deshpande, G.; Dupont, C.; Indrio, F.; et al. Lactobacillus reuteri to Treat Infant Colic: A Meta-analysis. Pediatrics 2018, 141, e20171811. [Google Scholar] [CrossRef] [PubMed]
- Schreck Bird, A.; Gregory, P.J.; Jalloh, M.A.; Risoldi Cochrane, Z.; Hein, D.J. Probiotics for the Treatment of Infantile Colic: A Systematic Review. J. Pharm. Pract. 2017, 30, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Szajewska, H.; Dryl, R. Probiotics for the Management of Infantile Colic. J. Pediatr. Gastroenterol. Nutr. 2016, 63 (Suppl. 1), S22–S24. [Google Scholar] [CrossRef] [PubMed]
- Guandalini, S.; Magazzù, G.; Chiaro, A.; La Balestra, V.; Di Nardo, G.; Gopalan, S.; Sibal, A.; Romano, C.; Canani, R.B.; Lionetti, P.; et al. VSL#3 Improves Symptoms in Children With Irritable Bowel Syndrome: A Multicenter, Randomized, Placebo-Controlled, Double-Blind, Crossover Study. J. Pediatr. Gastroenterol. Nutr. 2010, 51, 24–30. [Google Scholar] [CrossRef]
- Baldassarre, M.E.; Di Mauro, A.; Mastromarino, P.; Fanelli, M.; Martinelli, D.; Urbano, F.; Capobianco, D.; Laforgia, N. Administration of a Multi-Strain Probiotic Product to Women in the Perinatal Period Differentially Affects the Breast Milk Cytokine Profile and May Have Beneficial Effects on Neonatal Gastrointestinal Functional Symptoms. A Randomized Clinical Trial. Nutrients 2016, 8, 677. [Google Scholar] [CrossRef] [PubMed]
- Wessel, M.A.; Cobb, J.C.; Jackson, E.B.; Harris, G.S.; Detwiler, A.C. Paroxysmal fussing in infancy, sometimes called colic. Pediatrics 1954, 14, 421–435. [Google Scholar] [PubMed]
- Lust, K.D.; Brown, J.E.; Thomas, W. Maternal intake of cruciferous vegetables and other foods and colic symptoms in exclusively breast-fed infants. J. Am. Diet. Assoc. 1996, 96, 46–48. [Google Scholar] [CrossRef]
- Lane, M.M.; Czyzewski, D.I.; Chumpitazi, B.P.; Shulman, R.J. Reliability and validity of a modified Bristol Stool Form Scale for children. J. Pediatr. 2011, 159, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Szajewska, H.; Gyrczuk, E.; Horvath, A. Lactobacillus reuteri DSM 17938 for the Management of Infantile Colic in Breastfed Infants: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Pediatr. 2013, 162, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Mastromarino, P.; Capobianco, D.; Miccheli, A.; Praticò, G.; Campagna, G.; Laforgia, N.; Capursi, T.; Baldassarre, M.E. Administration of a multistrain probiotic product (VSL#3) to women in the perinatal period differentially affects breast milk beneficial microbiota in relation to mode of delivery. Pharmacol. Res. 2015, 95, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Ventrella, D.; Laghi, L.; Barone, F.; Elmi, A.; Romagnoli, N.; Bacci, M.L. Age-Related 1H NMR Characterization of Cerebrospinal Fluid in Newborn and Young Healthy Piglets. PLoS ONE 2016, 11, e0157623. [Google Scholar] [CrossRef] [PubMed]
- Simmler, C.; Napolitano, J.G.; McAlpine, J.B.; Chen, S.-N.; Pauli, G.F. Universal quantitative NMR analysis of complex natural samples. Curr. Opin. Biotechnol. 2014, 25, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Kneen, M.A.; Annegarn, H.J. Algorithm for fitting xRF, SeM and PIxe x-ray spectra backgrounds. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 1996, 109, 209–213. [Google Scholar] [CrossRef]
- Liland, K.H.; Almøy, T.; Mevik, B.-H. Optimal choice of baseline correction for multivariate calibration of spectra. Appl. Spectrosc. 2010, 64, 1007–1016. [Google Scholar] [CrossRef] [PubMed]
- Barbara, G.; Scaioli, E.; Barbaro, M.R.; Biagi, E.; Laghi, L.; Cremon, C.; Marasco, G.; Colecchia, A.; Picone, G.; Salfi, N.; et al. Gut microbiota, metabolome and immune signatures in patients with uncomplicated diverticular disease. Gut 2017, 66, 1252–1261. [Google Scholar] [CrossRef] [PubMed]
- Dieterle, F.; Ross, A.; Schlotterbeck, G.; Senn, H. Probabilistic quotient normalization as robust method to account for dilution of complex biological mixtures. Application in 1H NMR metabonomics. Anal. Chem. 2006, 78, 4281–4290. [Google Scholar] [CrossRef] [PubMed]
- Wishart, D.S.; Tzur, D.; Knox, C.; Eisner, R.; Guo, A.C.; Young, N.; Cheng, D.; Jewell, K.; Arndt, D.; Sawhney, S.; et al. HMDB: The Human Metabolome Database. Nucleic Acids Res. 2007, 35, D521–D526. [Google Scholar] [CrossRef] [PubMed]
- Hubert, M.; Rousseeuw, P.J.; Branden, K.V. ROBPCA: A new approach to robust principal components analysis. Technometrics 2005, 47, 64–79. [Google Scholar] [CrossRef]
- Ndagijimana, M.; Laghi, L.; Vitali, B.; Placucci, G.; Brigidi, P.; Guerzoni, M.E. Effect of a synbiotic food consumption on human gut metabolic profiles evaluated by (1)H Nuclear Magnetic Resonance spectroscopy. Int. J. Food Microbiol. 2009, 134, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Di Mauro, A.; Neu, J.; Riezzo, G.; Raimondi, F.; Martinelli, D.; Francavilla, R.; Indrio, F. Gastrointestinal function development and microbiota. Ital. J. Pediatr. 2013, 39, 15. [Google Scholar] [CrossRef] [PubMed]
- Shamir, R.; St James-Roberts, I.; Di Lorenzo, C.; Burns, A.J.; Thapar, N.; Indrio, F.; Riezzo, G.; Raimondi, F.; Di Mauro, A.; Francavilla, R.; et al. Infant crying, colic, and gastrointestinal discomfort in early childhood: A review of the evidence and most plausible mechanisms. J. Pediatr. Gastroenterol. Nutr. 2013, 57 (Suppl. 1), S1–S45. [Google Scholar] [CrossRef] [PubMed]
- Roos, S.; Dicksved, J.; Tarasco, V.; Locatelli, E.; Ricceri, F.; Grandin, U.; Savino, F. 454 pyrosequencing analysis on faecal samples from a randomized DBPC trial of colicky infants treated with Lactobacillus reuteri DSM 17938. PLoS ONE 2013, 8, e56710. [Google Scholar] [CrossRef] [PubMed]
- Kianifar, H.; Ahanchian, H.; Grover, Z.; Jafari, S.; Noorbakhsh, Z.; Khakshour, A.; Sedaghat, M.; Kiani, M. Synbiotic in the management of infantile colic: A randomised controlled trial: Synbiotic in infantile colic. J. Paediatr. Child Health 2014, 50, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Chapman, C.M.C.; Gibson, G.R.; Rowland, I. In vitro evaluation of single- and multi-strain probiotics: Inter-species inhibition between probiotic strains, and inhibition of pathogens. Anaerobe 2012, 18, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Indrio, F.; Di Mauro, A.; Riezzo, G.; Cavallo, L.; Francavilla, R. Infantile colic, regurgitation, and constipation: An early traumatic insult in the development of functional gastrointestinal disorders in children? Eur. J. Pediatr. 2015, 174, 841–842. [Google Scholar] [CrossRef] [PubMed]
- Partty, A.; Kalliomaki, M.; Salminen, S.; Isolauri, E. Infant Distress and Development of Functional Gastrointestinal Disorders in Childhood: Is There a Connection? JAMA Pediatr. 2013, 167, 977. [Google Scholar] [CrossRef] [PubMed]
- Ponnusamy, K.; Choi, J.N.; Kim, J.; Lee, S.-Y.; Lee, C.H. Microbial community and metabolomic comparison of irritable bowel syndrome faeces. J. Med. Microbiol. 2011, 60, 817–827. [Google Scholar] [CrossRef] [PubMed]
- Kawase, T.; Nagasawa, M.; Ikeda, H.; Yasuo, S.; Koga, Y.; Furuse, M. Gut microbiota of mice putatively modifies amino acid metabolism in the host brain. Br. J. Nutr. 2017, 117, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.H.; Pomare, E.W.; Branch, W.J.; Naylor, C.P.; Macfarlane, G.T. Short chain fatty acids in human large intestine, portal, hepatic and venous blood. Gut 1987, 28, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Tana, C.; Umesaki, Y.; Imaoka, A.; Handa, T.; Kanazawa, M.; Fukudo, S. Altered profiles of intestinal microbiota and organic acids may be the origin of symptoms in irritable bowel syndrome. Neurogastroenterol. Motil. Off. J. Eur. Gastrointest. Motil. Soc. 2010, 22, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Chow, J.; Panasevich, M.R.; Alexander, D.; Vester Boler, B.M.; Rossoni Serao, M.C.; Faber, T.A.; Bauer, L.L.; Fahey, G.C. Fecal metabolomics of healthy breast-fed versus formula-fed infants before and during in vitro batch culture fermentation. J. Proteome Res. 2014, 13, 2534–2542. [Google Scholar] [CrossRef] [PubMed]
- González, R.; Klaassens, E.S.; Malinen, E.; de Vos, W.M.; Vaughan, E.E. Differential transcriptional response of Bifidobacterium longum to human milk, formula milk, and galactooligosaccharide. Appl. Environ. Microbiol. 2008, 74, 4686–4694. [Google Scholar] [CrossRef] [PubMed]
- Morshed, K.M.; Desjeux, J.F.; Nagpaul, J.P.; Majumdar, S.; Amma, M.K. The effect of propane-diols on the intestinal uptake of nutrients and brush border membrane enzymes in the rat. Biochem. Med. Metab. Biol. 1991, 45, 161–170. [Google Scholar] [CrossRef]
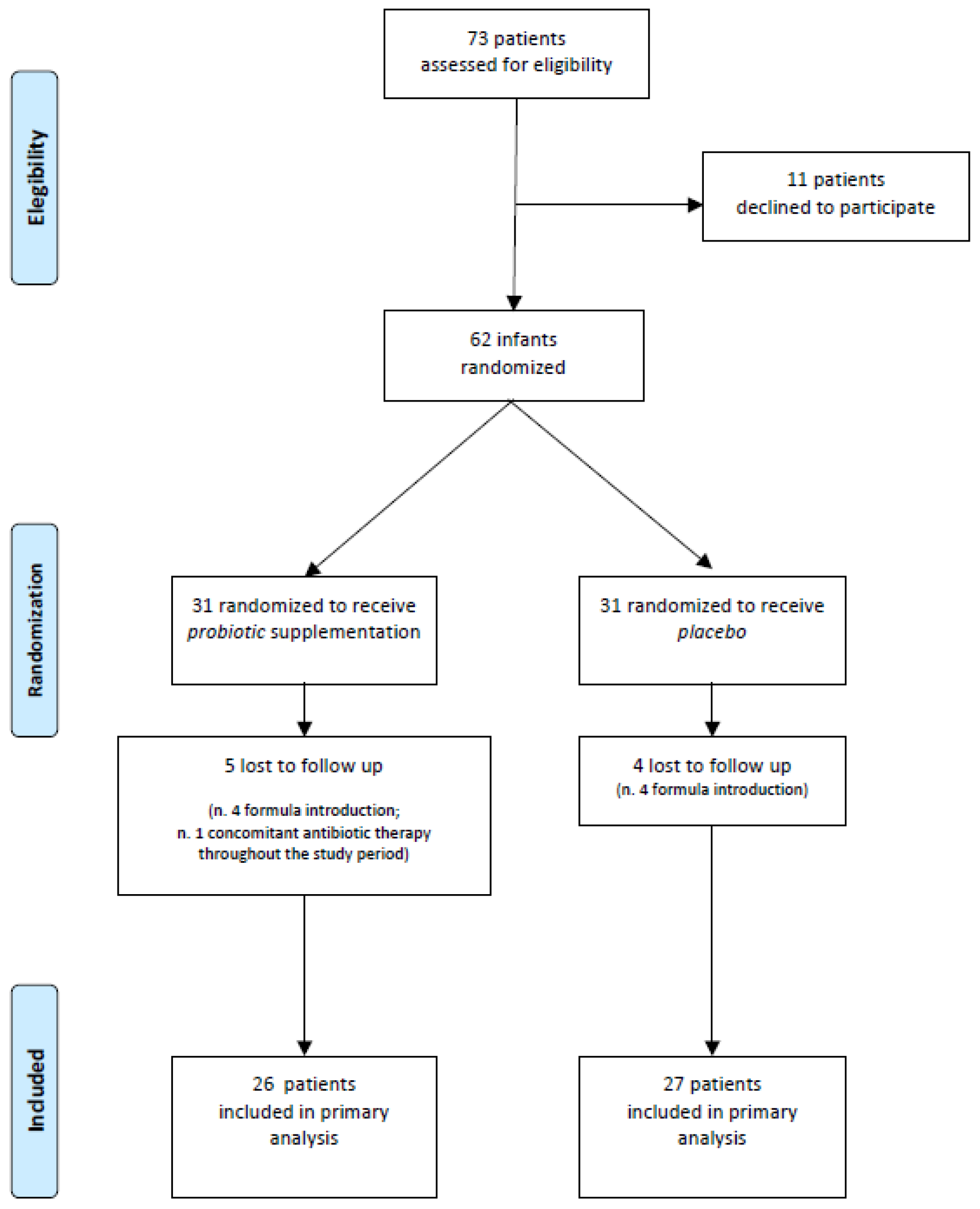
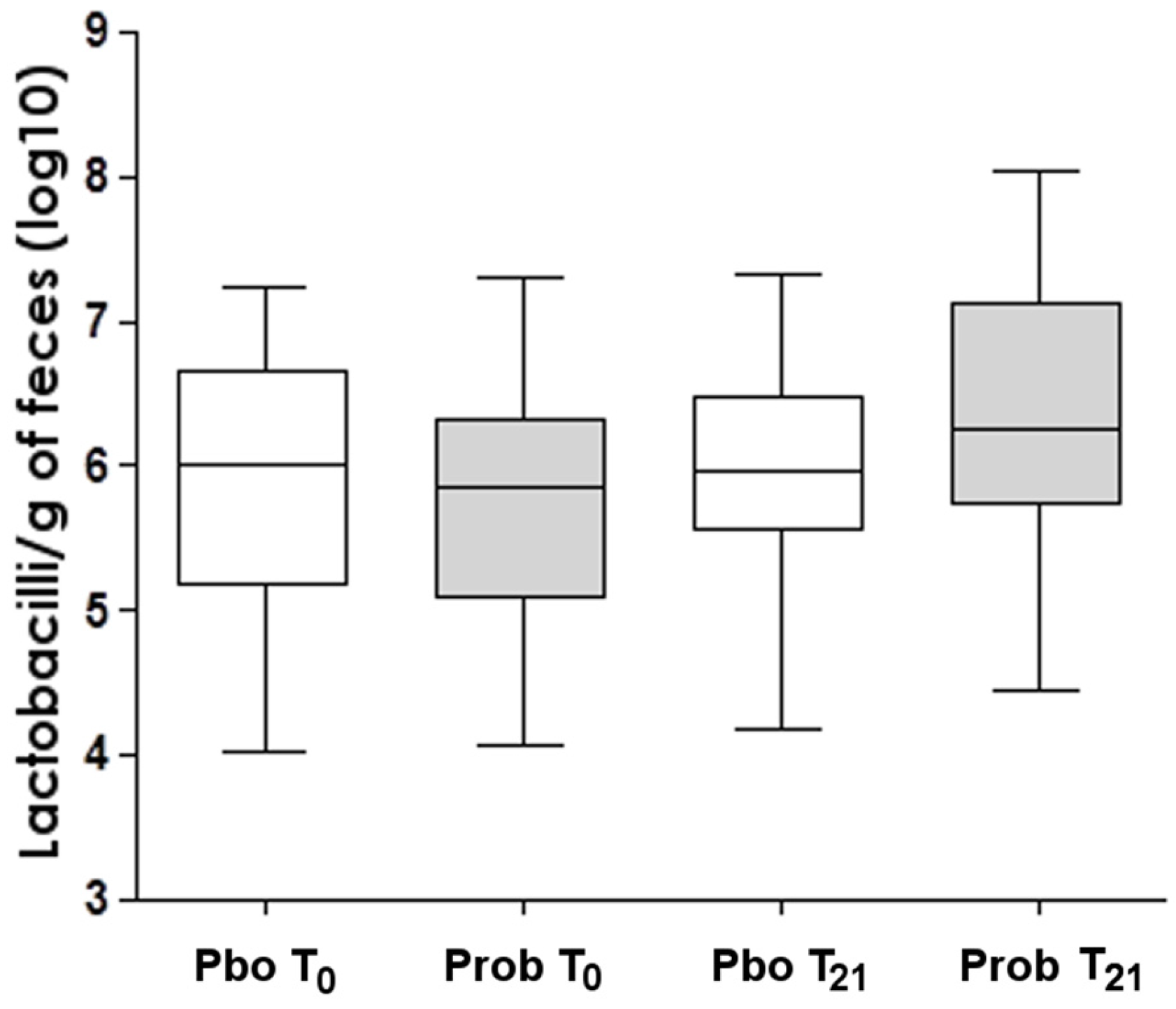
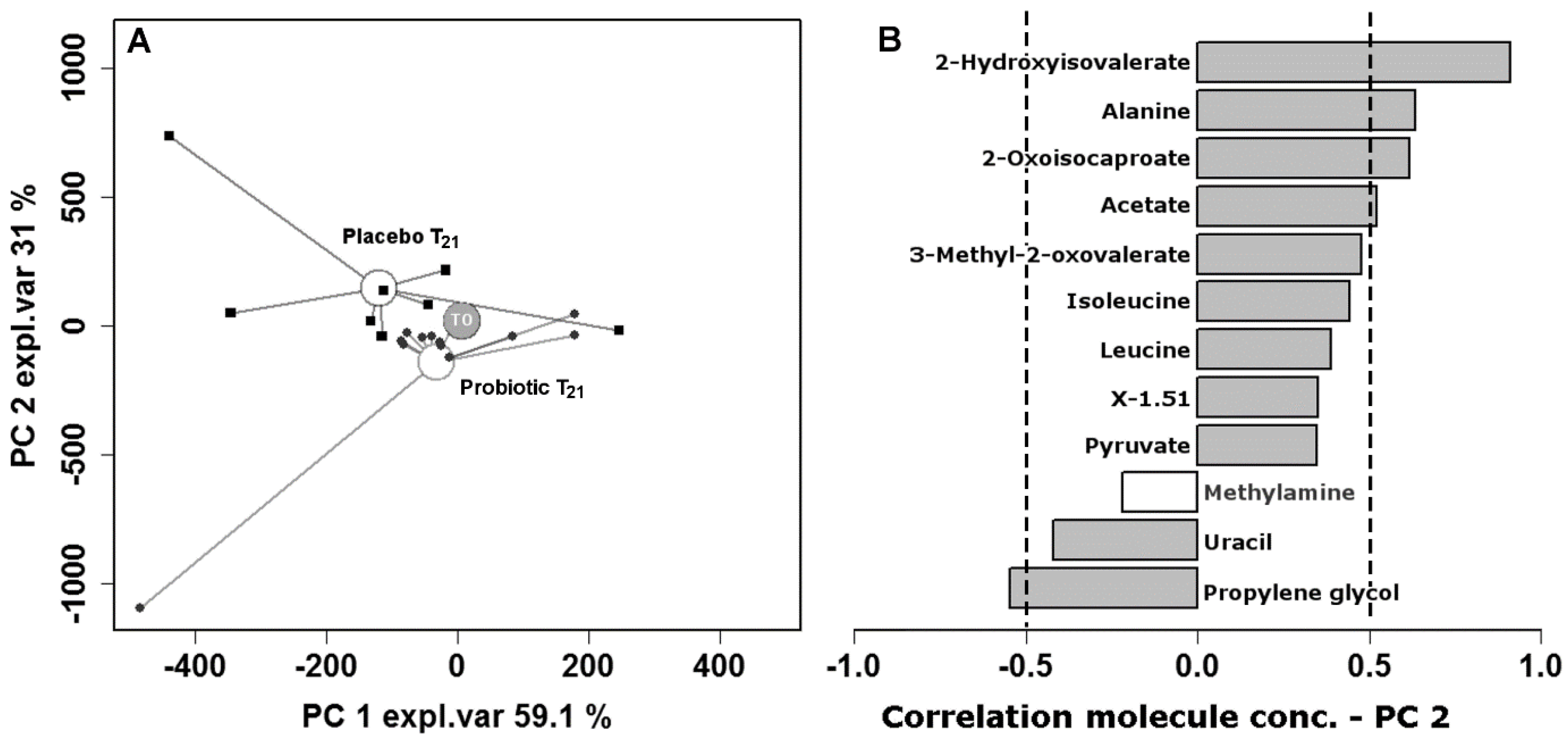
| Placebo (n = 26) | Probiotic (n = 27) | p Value | |
|---|---|---|---|
| Gestational age (week) | 38.5 ± 1.8 | 39.0 ± 1.6 | 0.25 * |
| Male | 13 (50.0) | 18 (66.7) | 0.21 ° |
| Vaginal | 7 (26.9) | 15 (55.6) | 0.03 ° |
| Birth weight (g) | 3156.5 ± 492.1 | 3367.9 ± 478.5 | 0.12 * |
| Age at enrolment (day) | 40.9 ± 21.7 | 37.7 ± 17.2 | 0.56 * |
| Weight at enrolment (g) | 4556.9 ± 985.6 | 4442.0 ± 691.9 | 0.62 * |
| Length at enrolment (cm) | 55.1 ± 4.1 | 54.8 ± 2.6 | 0.75 * |
| Head circumference at enrolment (cm) | 37.8 ± 2.2 | 37.9 ± 1.8 | 0.75 * |
| Family history of gastrointestinal disease | 2 (7.7) | 9 (33.3) | 0.02 ° |
| Family history of atopy | 7 (26.9) | 15 (55.6) | 0.03 ° |
| Duration of crying, min/day at baseline | 211.6 ± 31.6 | 226.2 ± 31.0 | 0.09 † |
| Placebo (n = 26) | Probiotic (n = 27) | p Value | |
|---|---|---|---|
| Responder vs. non responder, n (%) | |||
| Day 7 | 0 (0.0) | 3 (11.1) | 0.24 ∆ |
| Day 14 | 5 (19.2) | 12 (44.4) | 0.04 ° |
| Day 21 | 17 (65.4) | 26 (96.3) | 0.00 ° |
| Placebo (n = 26) | Probiotic (n = 27) | p Value | |
|---|---|---|---|
| Duration of crying, (mean ± SD) min/day | |||
| Baseline | 211.6 ± 31.6 | 226.2 ± 31.0 | 0.09 † |
| Day 7 | 163.7 ± 35.9 | 152.2 ± 32.0 | 0.23 † |
| Day 14 | 128.3 ± 36.6 | 112.3 ± 29.7 | 0.28 † |
| Day 21 | 98.7 ± 39.2 | 68.4 ± 28.2 | 0.00 † |
| Total crying/21 days, min | 391 ± 96.9 | 333 ± 71.2 | 0.00 * |
| Family quality of life (mean ± SD) | |||
| Baseline | 5.8 ± 2.3 | 5.5 ± 2.1 | 0.28 * |
| Day 7 | 7.1 ± 1.5 | 6.5 ± 1.3 | 0.11 * |
| Day 14 | 7.1 ± 1.2 | 7.7 ± 0.9 | 0.02 * |
| Day 21 | 6.7 ± 1.6 | 8.3 ± 1.0 | 0.00 * |
| Bristol Scale Score (n, % score = 7) | |||
| Baseline | 12 (46.2) | 8 (29.6) | 0.32 ° |
| Day 7 | 14 (53.9) | 9 (33.3) | 0.13 ° |
| Day 14 | 12 (46.2) | 11 (40.7) | 0.36 ° |
| Day 21 | 10 (38.5) | 11 (40.7) | 0.86 ° |
| Mean Bowel movement per day (mean ± SD) | |||
| Baseline | 4.3 ± 2.4 | 3.4 ± 2.6 | 0.41 * |
| Day 7 | 3.8 ± 2.5 | 2.9 ± 2.1 | 0.35 * |
| Day 14 | 3.2 ± 2.4 | 2.6 ± 1.8 | 0.48 * |
| Day 21 | 3.3 ± 2.4 | 2.6 ± 1.9 | 0.27 * |
| Weight gain g/day (mean ± SD) | |||
| Day 7 | 38.2 ± 27.0 | 55.8 ± 43.7 | 0.20 † |
| Day 14 | 32.2 ± 10.5 | 21.3 ± 32.1 | 0.10 † |
| Day 21 | 33.7 ± 26.2 | 31.7 ± 49.5 | 0.96 † |
| Length gain cm/w (mean ± SD) | |||
| Day 7 | 0.8 ± 1.1 | 1.2 ± 2.9 | 0.58 † |
| Day 14 | 1.1 ± 1.3 | 1.7 ± 2.8 | 0.83 † |
| Day 21 | 2.2 ± 1.1 | 2.6 ± 2.6 | 0.61 † |
| Cranial Circumference gain cm/w (mean ± SD) | |||
| Day 7 | 0.3 ± 0.6 | 0.5 ± 0.8 | 0.76 † |
| Day 14 | 0.8 ± 0.8 | 0.9 ± 0.9 | 0.69 † |
| Day 21 | 1.3 ± 0.6 | 1.1 ± 0.8 | 0.47 † |
| T0 | T21–T0 | ||||
|---|---|---|---|---|---|
| Placebo | Probiotic | Placebo | Probiotic | p Value | |
| Organic acids | |||||
| 2-Oxoiso-caproate | 4.29 × 10−6 ± 5.42 × 10−6 | 1.94 × 10−5 ± 1.94 × 10−5 | 1.80 × 10−5 ± 1.68 × 10−5 | −4.11 × 10−6 ± 1.90 × 10−5 | 2.54 × 10−3 |
| Pyruvate | 2.86 × 10−4 ± 3.50 × 10−4 | 5.32 × 10−4 ± 5.32 × 10−4 | 8.57 × 10−4 ± 7.08 × 10−4 | −2.60 × 10−6 ± 6.41 × 10−4 | 1.57 × 10−2 |
| Acetate | 2.80 × 10−2 ± 2.00 × 10−2 | 2.83 × 10−2 ± 2.83 × 10−2 | 2.33 × 10−2 ± 2.16 × 10−2 | 2.31 × 10−3 ± 2.91 × 10−2 | 9.08 × 10−2 |
| 2-Hydroxyiso-valerate | 3.60 × 10−5 ± 3.67 × 10−5 | 2.18 × 10−4 ± 2.18 × 10−4 | 5.97 × 10−6 ± 4.16 × 10−5 | −6.81 × 10−5 ± 1.57 × 10−4 | 9.08 × 10−2 |
| Amino acids | |||||
| Alanine | 2.56 × 10−3 ± 1.97 × 10−3 | 2.19 × 10−3 ± 2.19 × 10−3 | 1.52 × 10−3 ± 2.38 × 10−3 | −2.07 × 10−4 ± 1.35 × 10−3 | 3.28 × 10−2 |
| Leucine | 8.53 × 10−4 ± 4.31 × 10−4 | 4.73 × 10−4 ± 4.73 × 10−4 | 3.91 × 10−4 ± 1.11 × 10−3 | −2.54 × 10−4 ± 4.54 × 10−4 | 7.54 × 10−2 |
| Isoleucine | 3.87 × 10−4 ± 1.94 × 10−4 | 2.28 × 10−4 ± 2.28 × 10−4 | 1.08 × 10−4 ± 3.45 × 10−4 | −1.55 × 10−4 ± 2.37 × 10−4 | 6.20 × 10−2 |
| Alcohols | |||||
| Propylene glycol | 9.79 × 10−2 ± 2.44 × 10−1 | 2.84 × 10−3 ± 2.84 × 10−3 | −8.92 × 10−2 ± 2.42 × 10−1 | 2.54 × 10−3 ± 9.53 × 10−3 | 5.06 × 10−2 |
| Others | |||||
| Uracil | 2.15 × 10−4 ± 2.29 × 10−4 | 6.79 × 10−5 ± 6.79 × 10−5 | −1.20 × 10−4 ± 2.23 × 10−4 | 1.93 × 10−5 ± 7.10 × 10−5 | 1.57 × 10−2 |
| Methylamine | 3.00 × 10−5 ± 5.46 × 10−5 | 7.52 × 10−6 ± 7.52 × 10−6 | −2.19 × 10−5 ± 5.44 × 10−5 | 5.89 × 10−6 ± 1.26 × 10−5 | 6.20 × 10−2 |
| 3-Methyl-2-oxo-valerate | 1.36 × 10−5 ± 1.25 × 10−5 | 2.53 × 10−5 ± 2.53 × 10−5 | 3.63 × 10−5 ± 4.16 × 10−5 | 7.76 × 10−6 ± 2.10 × 10−5 | 9.08 × 10−2 |
| x-1.51 | 1.80 × 10−4 ± 3.98 × 10−4 | 1.81 × 10−4 ± 1.81 × 10−4 | 1.20 × 10−4 ± 4.57 × 10−4 | −2.89 × 10−5 ± 1.25 × 10−4 | 1.57 × 10−2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baldassarre, M.E.; Di Mauro, A.; Tafuri, S.; Rizzo, V.; Gallone, M.S.; Mastromarino, P.; Capobianco, D.; Laghi, L.; Zhu, C.; Capozza, M.; et al. Effectiveness and Safety of a Probiotic-Mixture for the Treatment of Infantile Colic: A Double-Blind, Randomized, Placebo-Controlled Clinical Trial with Fecal Real-Time PCR and NMR-Based Metabolomics Analysis. Nutrients 2018, 10, 195. https://doi.org/10.3390/nu10020195
Baldassarre ME, Di Mauro A, Tafuri S, Rizzo V, Gallone MS, Mastromarino P, Capobianco D, Laghi L, Zhu C, Capozza M, et al. Effectiveness and Safety of a Probiotic-Mixture for the Treatment of Infantile Colic: A Double-Blind, Randomized, Placebo-Controlled Clinical Trial with Fecal Real-Time PCR and NMR-Based Metabolomics Analysis. Nutrients. 2018; 10(2):195. https://doi.org/10.3390/nu10020195
Chicago/Turabian StyleBaldassarre, Maria Elisabetta, Antonio Di Mauro, Silvio Tafuri, Valentina Rizzo, Maria Serena Gallone, Paola Mastromarino, Daniela Capobianco, Luca Laghi, Chenglin Zhu, Manuela Capozza, and et al. 2018. "Effectiveness and Safety of a Probiotic-Mixture for the Treatment of Infantile Colic: A Double-Blind, Randomized, Placebo-Controlled Clinical Trial with Fecal Real-Time PCR and NMR-Based Metabolomics Analysis" Nutrients 10, no. 2: 195. https://doi.org/10.3390/nu10020195
APA StyleBaldassarre, M. E., Di Mauro, A., Tafuri, S., Rizzo, V., Gallone, M. S., Mastromarino, P., Capobianco, D., Laghi, L., Zhu, C., Capozza, M., & Laforgia, N. (2018). Effectiveness and Safety of a Probiotic-Mixture for the Treatment of Infantile Colic: A Double-Blind, Randomized, Placebo-Controlled Clinical Trial with Fecal Real-Time PCR and NMR-Based Metabolomics Analysis. Nutrients, 10(2), 195. https://doi.org/10.3390/nu10020195









