Dietary Cholesterol Intake and Risk of Lung Cancer: A Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Study Selection
2.3. Data Extraction
2.4. Quality Assessment
2.5. Statistical Analysis
3. Results
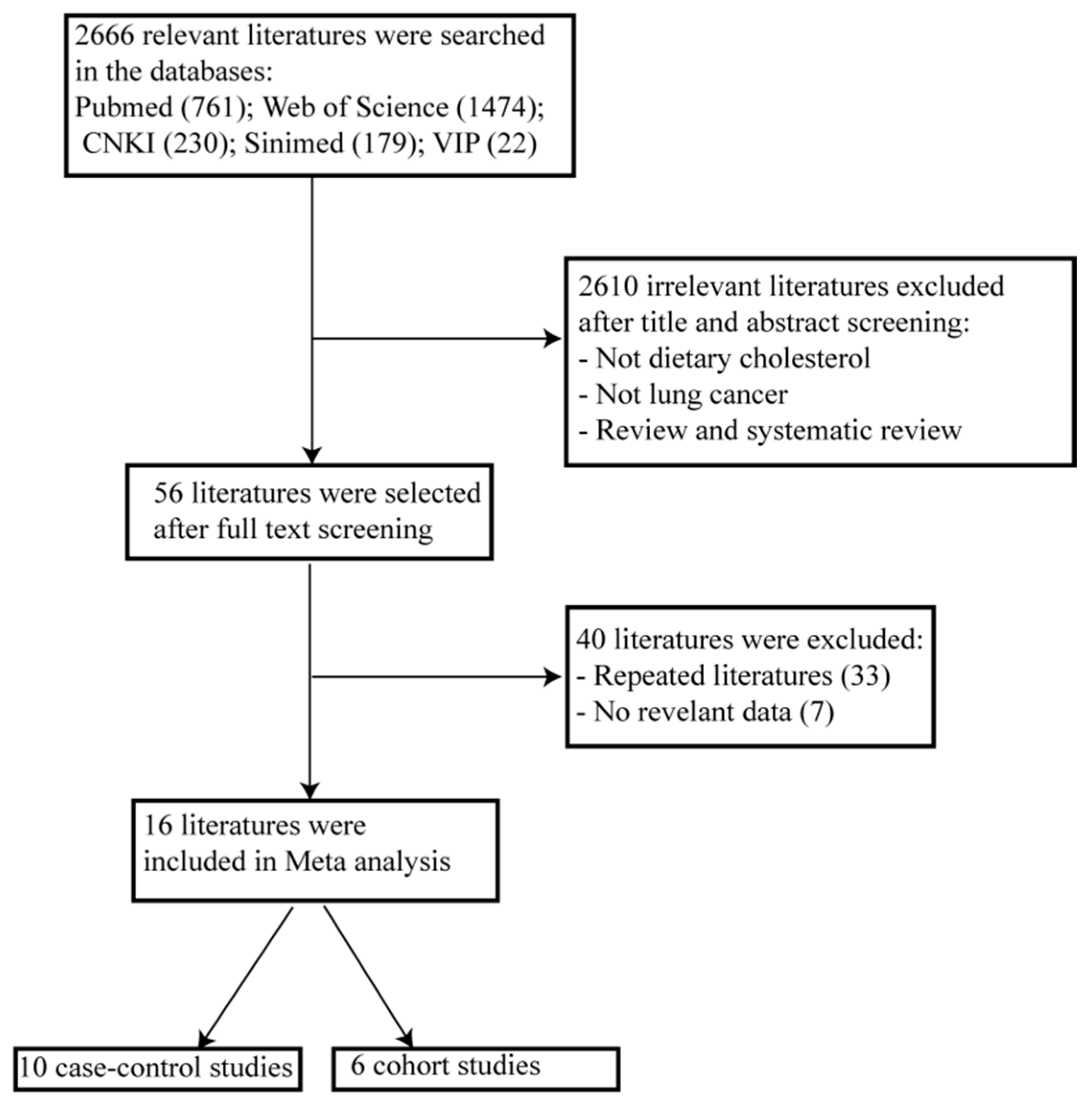
3.1. Literature Search and Study Characteristics
3.2. Data Analysis
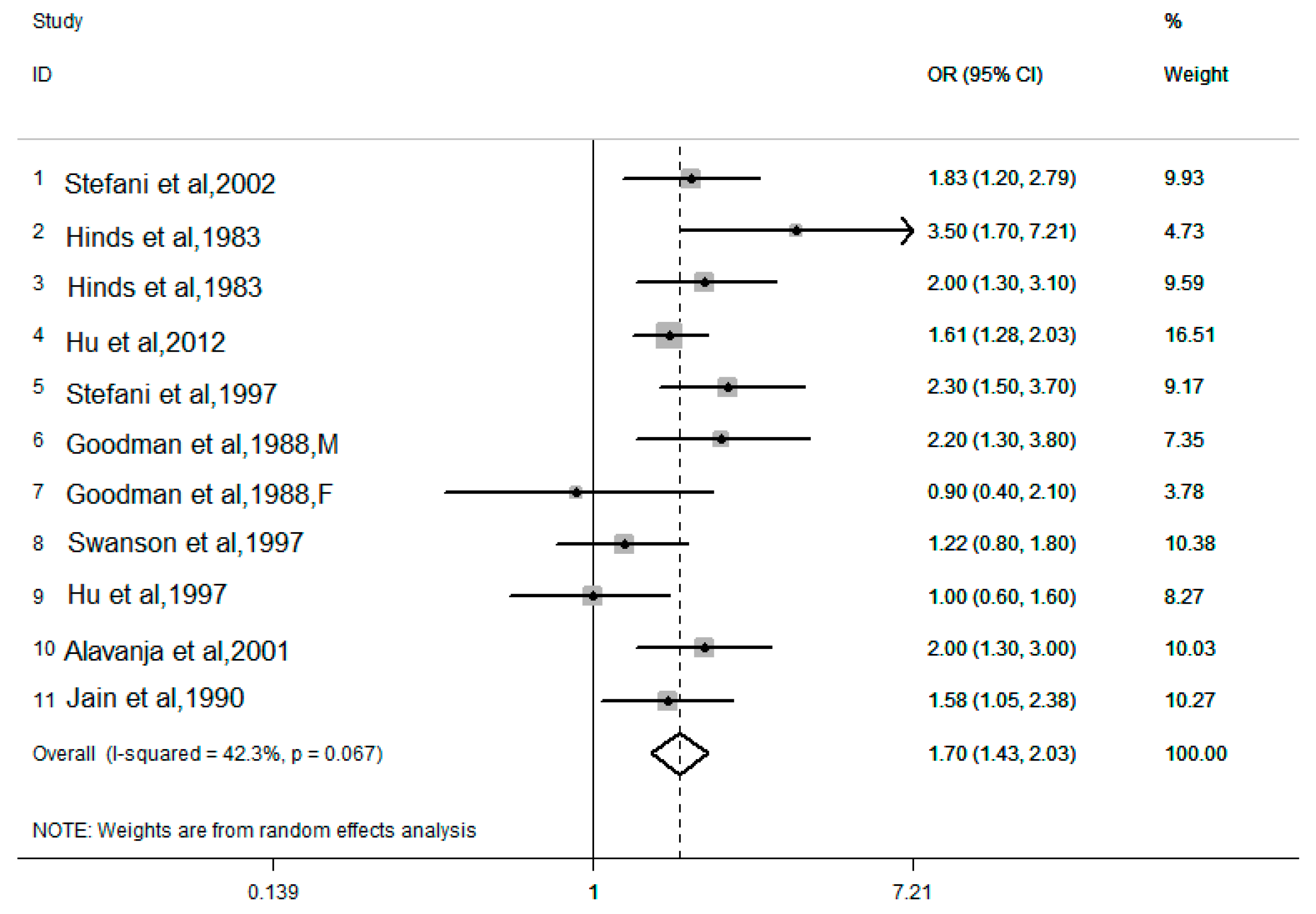
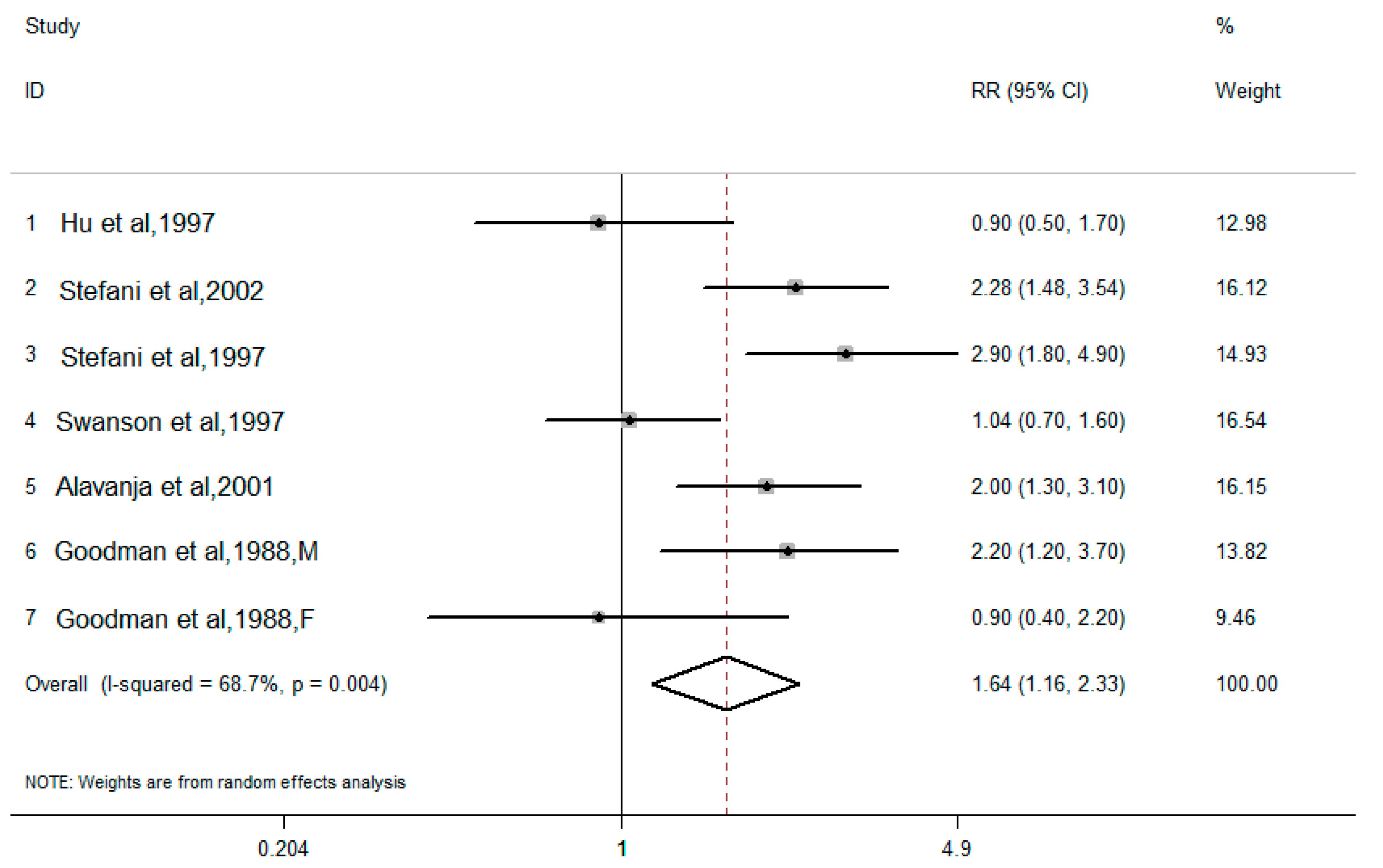
3.2.1. Dietary Cholesterol and Lung Cancer Risk
3.2.2. Dietary Total Fat and Lung Cancer Risk
3.3. Publication Bias
3.4. Sensitivity Analysis
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Schiller, J.H.; Gazdar, A.F. Lung cancer in never smokers—A different disease. Nat. Rev. Cancer 2007, 7, 778–790. [Google Scholar] [CrossRef] [PubMed]
- Baena Ruiz, R.; Salinas Hernández, P. Diet and cancer: Risk factors and epidemiological evidence. Maturitas 2014, 77, 202–208. [Google Scholar] [CrossRef] [PubMed]
- World Cancer Research Fund/American Institute for Cancer Research. Food Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective; American Institute for Cancer Research (AICR): Washington, DC, USA, 2007; pp. 259–264. ISBN 9780972252225.
- Vieira, A.R.; Abar, L.; Vingeliene, S.; Chan, D.S.; Aune, D.; Navarro-Rosenblatt, D.; Stevens, C.; Greenwood, D.; Norat, T. Fruits, vegetables and lung cancer risk: A systematic review and meta-analysis. Ann. Oncol. 2016, 27, 81–96. [Google Scholar] [CrossRef] [PubMed]
- Gowdy, K.M.; Fessler, M.B. Emerging roles for cholesterol and lipoproteins in lung disease. Pulm. Pharmacol. Ther. 2013, 26, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Kuzu, O.F.; Noory, M.A.; Robertson, G.P. The role of cholesterol in cancer. Cancer Res. 2016, 76, 2063–2070. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, W.J.; Zhai, L.; Zhang, D.F. Association of cholesterol with risk of pancreatic cancer: A meta-analysis. World J. Gastroenterol. 2015, 21, 3711–3719. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Yang, L.; Zhang, D.; Jiang, W. Systematic review and meta-analysis suggest that dietary cholesterol intake increases risk of breast cancer. Nutr. Res. 2016, 36, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Shea, B.; O’ Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality If Nonrandomized Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.htm (accessed on 20 October 2017).
- Hinds, M.W.; Kolonel, L.N.; Lee, J.; Hankin, J.H. Dietary cholesterol and lung cancer risk among men in Hawaii. Am. J. Clin. Nutr. 1983, 37, 192–193. [Google Scholar] [CrossRef] [PubMed]
- Hinds, M.W.; Kolonel, L.N.; Hankin, J.H.; Lee, J. Dietary cholesterol and lung cancer risk in a multiethnic population in Hawaii. Int. J. Cancer 1983, 32, 727–732. [Google Scholar] [CrossRef] [PubMed]
- Goodman, M.T.; Kolonel, L.N.; Yoshizawa, C.N.; Hankin, J.H. The effect of dietary cholesterol and fat on the risk of lung cancer in Hawaii. Am. J. Epidemiol. 1988, 128, 1241–1255. [Google Scholar] [CrossRef] [PubMed]
- Jain, M.; Burch, J.D.; Howe, G.R.; Risch, H.A.; Miller, A.B. Dietary factors and risk of lung cancer: Results from a case-control study, Toronto, 1981–1985. Int. J. Cancer 1990, 45, 287–293. [Google Scholar] [CrossRef] [PubMed]
- De Stefani, E.; Deneo-Pellegrini, H.; Mendilaharsu, M.; Carzoglio, J.C.; Ronco, A. Dietary fat and lung cancer: A case-control study in Uruguay. Cancer Causes Control 1997, 8, 913–921. [Google Scholar] [CrossRef] [PubMed]
- Swanson, C.A.; Brown, C.C.; Sinha, R.; Kulldorff, M.; Brownson, R.C.; Alavanja, M.C. Dietary fats and lung cancer risk among women: The Missouri Women’s Health Study (United States). Cancer Causes Control 1997, 8, 883–893. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Johnson, K.C.; Mao, Y.; Xu, T.; Lin, Q.; Wang, C.; Zhao, F.; Wang, G.; Chen, Y.; Yang, Y. A case-control study of diet and lung cancer in northeast China. Int. J. Cancer 1997, 71, 924–931. [Google Scholar] [CrossRef]
- Alavanja, M.C.; Field, R.W.; Sinha, R.; Brus, C.P.; Shavers, V.L.; Fisher, E.L.; Curtain, J.; Lynch, C.F. Lung cancer risk and red meat consumption among Iowa women. Lung Cancer 2001, 34, 37–46. [Google Scholar] [CrossRef]
- De Stefani, E.; Brennan, P.; Boffetta, P.; Mendilaharsu, M.; Deneo-Pellegrini, H.; Ronco, A.; Olivera, L.; Kasdorf, H. Diet and adenocarcinoma of the lung: A case-control study in Uruguay. Lung Cancer 2002, 35, 43–51. [Google Scholar] [CrossRef]
- Hu, J.; La Vecchia, C.; de Groh, M.; Negri, E.; Morrison, H.; Mery, L.; Canadian Cancer Registries Epidemiology Research Group. Dietary cholesterol intake and cancer. Ann. Oncol. 2012, 23, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Heilbrun, L.K.; Nomura, A.M.; Stemmermann, G.N. Dietary cholesterol and lung cancer risk among Japanese men in Hawaii. Am. J. Clin. Nutr. 1984, 39, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Knekt, P.; Seppänen, R.; Järvinen, R.; Virtamo, J.; Hyvönen, L.; Pukkala, E.; Teppo, L. Dietary cholesterol, fatty acids, and the risk of lung cancer among men. Nutr. Cancer 1991, 16, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zheng, W.; Sellers, T.A.; Kushi, L.H.; Bostick, R.M.; Potter, J.D. Dietary cholesterol, fat, and lung cancer incidence among older women: The Iowa Women’s Health Study (United States). Cancer Causes Control 1994, 5, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Bandera, E.V.; Freudenheim, J.L.; Marshall, J.R.; Zielezny, M.; Priore, R.L.; Brasure, J.; Baptiste, M.; Graham, S. Diet and alcohol consumption and lung cancer risk in the New York State Cohort (United States). Cancer Causes Control 1997, 8, 828–840. [Google Scholar] [CrossRef] [PubMed]
- Veierød, M.B.; Laake, P.; Thelle, D.S. Dietary fat intake and risk of lung cancer: A prospective study of 51,452 Norwegian men and women. Eur. J. Cancer Prev. 1997, 6, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Speizer, F.E.; Colditz, G.A.; Hunter, D.J.; Rosner, B.; Hennekens, C. Prospective study of smoking, antioxidant intake, and lung cancer in middle-aged women (USA). Cancer Causes Control 1999, 10, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Smith-Warner, S.A.; Ritz, J.; Hunter, D.J.; Albanes, D.; Beeson, W.L.; van den Brandt, P.A.; Colditz, G.; Folsom, A.R.; Fraser, G.E.; Freudenheim, J.L.; et al. Dietary fat and risk of lung cancer in a pooled analysis of prospective studies. Cancer Epidemiol. Biomark. Prev. 2002, 11, 987–992. [Google Scholar]
- Yang, J.J.; Yu, D.; Takata, Y.; Smith-Warner, S.A.; Blot, W.; White, E.; Robien, K.; Park, Y.; Xiang, Y.B.; Sinha, R.; et al. Dietary fat intake and lung cancer risk: A pooled analysis. J. Clin. Oncol. 2017, 35, 3055–3064. [Google Scholar] [CrossRef] [PubMed]
- Li, L.M. Epidemiology, 4th ed.; People’s Health Publishing House: Beijing, China, 2008; pp. 76–99. ISBN 9787117034982. [Google Scholar]
- Shen, H.; Qi, X. Epidemiology, 8th ed.; People’s Medical Publishing House: Beijing, China, 2013; p. 54. ISBN 9787117170727. [Google Scholar]
- Dai, Y.B.; Miao, Y.F.; Wu, W.F.; Li, Y.; D’Errico, F.; Su, W.; Burns, A.R.; Huang, B.; Maneix, L.; Warner, M.; et al. Ablation of Liver X receptors α and β leads to spontaneous peripheral squamous cell lung cancer in mice. Proc. Natl. Acad. Sci. USA 2016, 113, 7614–7619. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Lu, L.; Xu, C.; Lin, X.; Leung, Y.K.; Ho, S.M.; Ruan, X.Z.; Lian, X. Inhibition role of atherogenic diet on ethyl carbamate induced lung tumorigenesis in C57BL/6J mice. Sci. Rep. 2017, 7, 4723. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Lu, L.; Liu, L.; Wei, S.; He, Y.; Chang, J.; Lian, X. Blood lipids profile and lung cancer risk in a meta-analysis of prospective cohort studies. J. Clin. Lipidol. 2017, 11, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Howell, W.H.; McNamara, D.J.; Tosca, M.A.; Smith, B.T.; Gaines, J.A. Plasma lipid and lipoprotein responses to dietary fat and cholesterol: A meta-analysis. Am. J. Clin. Nutr. 1997, 65, 1747–1764. [Google Scholar] [CrossRef] [PubMed]
- Trumbo, P.R.; Shimakawa, T. Tolerable upper intake levels for trans fat, saturated fat, and cholesterol. Nutr. Rev. 2011, 69, 270–278. [Google Scholar] [CrossRef] [PubMed]

| Author | Year | Location | Cases | Controls | Gender | Adjusted OR (95% CI) | Dietary Cholesterol (Tool) | Quality | Adjustments |
|---|---|---|---|---|---|---|---|---|---|
| Hinds et al. [11] | 1983 | USA | 188 | 294 | Male Female | 3.50 (1.70–7.21) | >3500 mg/week vs. <999 mg/week (FFQ) | 6 | Ethnicity, age, pack-years of cigarettes smoked, and occupational exposure to carcinogens |
| Hinds et al. [12] | 1983 | USA | 364 | 627 | Male Female | 2.0 (1.3–3.1) | >2071 mg/week vs. <750 mg/week (FFQ) | 7 | Vitamin A intake, sex, age, pack-years of smoking, ethnicity, occupational status |
| Goodman et al. [13] | 1988 | USA | 326 | 865 | Male | 2.2 (1.3–3.8) | Q4 vs. Q1 (FFQ) | 7 | Age, ethnicity, and pack-years of cigarette smoking |
| Female | 0.9 (0.4–2.1) | ||||||||
| Jain et al. [14] | 1990 | Canada | 839 | 772 | Male Female | 1.58 (1.05–2.38) | >468 mg/day vs. <234 mg/day (FFQ) | 7 | Age and cumulative cigarette smoking |
| Stefani et al. [15] | 1997 | Uruguay | 426 | 419 | Male | 2.3 (1.5–3.7) | >610.7 mg/day vs. ≤354.6 mg/day (FFQ) | 7 | Age, residence, urban/rural status, tobacco smoking, total energy intake, a term for all vegetables and fruits and α-carotene intake |
| Swanson et al. [16] | 1997 | USA | 624 | 587 | Female | 1.22 (0.8–1.8) | >177 mg/1000 Kcal vs. <102 mg/1000 Kcal (FFQ) | 6 | Age and total calories, education, pack-years of smoking, body mass index, consumption of vegetables and fruit |
| Hu et al. [17] | 1997 | China | 227 | 227 | Male Female | 1.0 (0.6–1.6) | >108.22 mg/day vs. <19.02 mg/day ( FFQ) | 8 | Cigarettes per day, duration, and family income |
| Alavanja et al. [18] | 2001 | USA | 360 | 574 | Female | 2.0 (1.3–3.0) | Q5 vs. Q1 (FFQ) | 7 | Age and nutrient density calories |
| Stefani et al. [19] | 2002 | Uruguay | 200 | 600 | Male | 1.83 (1.20–2.79) | T3 vs. T1 (FFQ) | 7 | Age, residence, urban/rural status, education, body mass index, smoking status, smoking duration, and total energy intake |
| Hu et al. [20] | 2012 | Canada | 3341 | 24,771 | Male Female | 1.61 (1.28–2.03) | ≥1880.266 mg/week vs. ≤966.261 mg/week (FFQ) | 7 | Sex, age group, province, education, body mass index, alcohol drinking, pack-years of smoking, total of vegetable and fruit intake, saturated fat and total energy intake |
| Author | Year | Location | Follow-Up | No. of Cases/Participants | Gender | Adjusted RR (95% CI) | Dietary Cholesterol (Tool) | Quality | Adjustments |
|---|---|---|---|---|---|---|---|---|---|
| Heilbrun et al. [21] | 1984 | USA | 1968–1983 | 113/7539 | Male | 0.98 (0.58–1.66) | >750 mg/day vs. <299 mg/day (24h recall ) | 9 | Age and pack-years of smoking |
| Knekt et al. [22] | 1991 | Finland | 1967–1986 | 117/4538 | Male | 1.03 (0.58–1.85) | >609 mg/day vs. <441 mg/day (FFQ) | 8 | Age, smoking, and energy intake |
| Wu et al. [23] | 1994 | USA | 1985–1991 | 272/41,837 | Female | 0.9 (0.6–1.3) | >365.5 mg/day vs. <200.5 mg/day (FFQ) | 8 | Age, smoking status, pack-years of cigarettes, occupation, physical activity, and total energy intake |
| Bandera et al. [24] | 1997 | USA | 1980–1987 | 525/48,000 | Male | 1.09 (0.85–1.39) | T3 vs. T1 (FFQ) | 9 | Age, education, cigarettes/day, years smoking, and total energy intake (except calories) |
| Female | 0.97 (0.64–1.47) | ||||||||
| Veierød et al. [25] | 1997 | Norway | 1980–1991 | 149/50,712 | Male Female | 1.2 (0.8–1.9) | ≥240.6 mg/day vs. ≤154.9 mg/day (FFQ) | 9 | Smoking status, gender, age at inclusion and attained age |
| Speizer et al. [26] | 1999 | USA | 1980–1992 | 593/89,294 | Female | 1.3 (0.9–1.7) | Q5 vs. Q1 (FFQ) | 8 | Age, total energy intake, smoking, and age of starting to smoke |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, X.; Liu, L.; Fu, Y.; Gao, J.; He, Y.; Wu, Y.; Lian, X. Dietary Cholesterol Intake and Risk of Lung Cancer: A Meta-Analysis. Nutrients 2018, 10, 185. https://doi.org/10.3390/nu10020185
Lin X, Liu L, Fu Y, Gao J, He Y, Wu Y, Lian X. Dietary Cholesterol Intake and Risk of Lung Cancer: A Meta-Analysis. Nutrients. 2018; 10(2):185. https://doi.org/10.3390/nu10020185
Chicago/Turabian StyleLin, Xiaojing, Lingli Liu, Youyun Fu, Jing Gao, Yunyun He, Yang Wu, and Xuemei Lian. 2018. "Dietary Cholesterol Intake and Risk of Lung Cancer: A Meta-Analysis" Nutrients 10, no. 2: 185. https://doi.org/10.3390/nu10020185
APA StyleLin, X., Liu, L., Fu, Y., Gao, J., He, Y., Wu, Y., & Lian, X. (2018). Dietary Cholesterol Intake and Risk of Lung Cancer: A Meta-Analysis. Nutrients, 10(2), 185. https://doi.org/10.3390/nu10020185




