Lactose Intolerance, Dairy Avoidance, and Treatment Options
Abstract
1. Introduction
2. Lactose Sources, Synthesis, and Metabolism
3. Causes of Lactose Maldigestion
4. Lactose Intolerance
4.1. Diagnosis of Lactose Maldigestion
4.2. Pathophysiology of Symptoms Related to Lactose
4.3. The Impact of Knowledge about Lactose Intolerance on Dairy Avoidance
4.4. Merging of Lactose Intolerance with Symptoms of Other Disorders
5. Consequences of Dairy Product Consumption
5.1. Bone Formation, Maintenance, and Bone Disease
5.2. Cardiometabolic Syndromes
5.3. Colorectal Cancer and Inflammatory Bowel Diseases (IBD)
5.4. Other Cancers
6. Treatment Options for Lactose Intolerance
6.1. Lactose-Free and Lactose-Reduced Products
6.2. Non-Dairy Substitutes and Comparison of Inherent Nutrients with Dairy Products
6.3. Exogenous Oral Enzymes
6.4. Adaptation and Prebiotics for Treatment of Lactose Intolerance
6.5. Probiotics
7. Other Treatments Which Encompass a More General Food Intolerance Symptoms
8. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grant, W.B.; Garland, C.F. The association of solar ultraviolet B (UVB) with reducing risk of cancer: Multifactorial ecologic analysis of geographic variation in age-adjusted cancer mortality rates. Anticancer Res. 2006, 26, 2687–2699. [Google Scholar] [PubMed]
- Grant, W.B. The role of geographical ecological studies in identifying diseases linked to UVB exposure and/or vitamin D. Dermatoendocrinol 2016, 8, e1137400. [Google Scholar] [CrossRef]
- Shrier, I.; Szilagyi, A.; Correa, J.A. Impact of lactose containing foods and the genetics of lactase on diseases: An analytical review of population data. Nutr. Cancer 2008, 60, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Lember, M.; Torniainen, S.; Kull, M.; Kallikorm, R.; Saadla, P.; Rajasalu, T.; Komu, H.; Järvelä, I. Lactase non-persistence and milk consumption in Estonia. World J. Gastroenterol. 2006, 12, 7329–7331. [Google Scholar] [CrossRef] [PubMed]
- Almon, R.; Sjöström, M.; Nilsson, T.K. Lactase non-persistence as a determinant of milk avoidance and calcium intake in children and adolescents. J. Nutr. Sci. 2013, 2, e26. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Venema, K.; Priebe, M.; Welling, G.; Brummer, R.; Vonk, R. The role of colonic metabolism in lactose intolerance. Eur. J. Clin. Investig. 2008, 38, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Szilagyi, A.; Leighton, H.; Burstein, B.; Shrier, I. Significant positive correlation between sunshine and lactase nonpersistence in Europe May implicate both in similarly altering risks for some diseases. Nutr. Cancer 2011, 63, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Brüssow, H. Nutrition, population growth and disease: A short history of lactose. Environ. Microbiol. 2013, 15, 2154–2161. [Google Scholar] [CrossRef]
- Kuhn, R.; Low, I. The occurrence of lactose in the plant kingdom. Chem. Ber. 1949, 82, 479–481. [Google Scholar] [CrossRef]
- Toba, T.; Nagashima, S.; Adachi, S. Is lactose really present in plants? J. Sci. Food Agric. 1991, 54, 305–308. [Google Scholar] [CrossRef]
- Schaafsma, G. Lactose and lactose derivatives as bioactive ingredients in human nutrition. Int. Dairy J. 2008, 18, 458–465. [Google Scholar] [CrossRef]
- Bode, L. Human milk oligosaccharides: Every baby needs a sugar mama. Glycobiology 2012, 22, 1147–1162. [Google Scholar] [CrossRef] [PubMed]
- Wijesinha-Bettoni, R.; Burlingame, B. Milk and Dairy Product Composition; Food Agriculture Organization (FAO): Rome, Italy, 2013. [Google Scholar]
- Holden, H.M.; Rayment, I.; Thoden, J.B. Structure and function of enzymes of the Leloir pathway for galactose metabolism. J. Biol. Chem. 2003, 278, 43885–43888. [Google Scholar] [CrossRef] [PubMed]
- Demirbas, D.; Coelho, A.I.; Rubio-Gozalbo, M.E.; Berry, G.T. Hereditary galactosemia. Metabolism 2018, 83, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Day, A.J.; Cañada, F.J.; Díaz, J.C.; Kroon, P.A.; Mclauchlan, R.; Faulds, C.B.; Plumb, G.W.; Morgan, M.R.; Williamson, G. Dietary flavonoid and isoflavone glycosides are hydrolysed by the lactase site of lactase phlorizin hydrolase. FEBS Lett. 2000, 468, 166–170. [Google Scholar] [CrossRef]
- Zecca, L.; Mesonero, J.E.; Stutz, A.; Poirée, J.C.; Giudicelli, J.; Cursio, R.; Gloor, S.M.; Semenza, G. Intestinal lactase-phlorizin hydrolase (LPH): The two catalytic sites; the role of the pancreas in pro-LPH maturation. FEBS Lett. 1998, 435, 225–228. [Google Scholar] [CrossRef]
- Lawrence, R.A.; Lawrence, R.M. Breastfeeding: A Guide for the Medical Profession, 8th ed.; Elsevier: Philadelphia, PA, USA, 2016; pp. 56–90. [Google Scholar]
- Gilat, T.; Russo, S.; Gelman-Malachi, E.; Aldor, T.A. Lactase in man: A nonadaptable enzyme. Gastroenterology 1972, 62, 1125–1127. [Google Scholar]
- Swallow, D.M. Genetics of lactase persistence and lactose intolerance. Ann. Hum. Genet. 2003, 37, 197–219. [Google Scholar] [CrossRef]
- Kruse, T.A.; Bolund, L.; Grzeschik, K.H.; Ropers, H.H.; Sjöström, H.; Norén, O.; Mantei, N.; Semenza, G. The human lactase-phlorizin gene is located on chromosome 2. FEBS Lett. 1988, 240, 123–126. [Google Scholar] [CrossRef]
- Enattah, N.S.; Jensen, T.G.; Nielsen, M.; Lewinski, R.; Kuokkanen, M.; Rasinpera, H.; Khalil, I.F. Independent introduction of two lactase-persistence alleles into human populations reflects different history of adaptation to milk culture. Am. J. Hum. Genet. 2008, 82, 57–72. [Google Scholar] [CrossRef]
- Ingram, C.J.; Mulcare, C.A.; Itan, Y.; Thomas, M.G.; Swallow, D.M. Lactose digestion and the evolutionary genetics of lactase persistence. Hum. Genet. 2009, 124, 579–591. [Google Scholar] [CrossRef]
- Ranciaro, A.; Campbell, M.C.; Hirbo, J.B.; Ko, W.-Y.; Froment, A.; Anagnostou, P.; Omar, S.A. Genetic origins of lactase persistence and the spread of pastoralism in Africa. Am. J. Hum. Genet. 2014, 94, 496–510. [Google Scholar] [CrossRef]
- Tishkoff, S.A.; Reed, F.A.; Ranciaro, A.; Voight, B.F.; Babbitt, C.C.; Silverman, J.S.; Powell, K.; Mortensen, H.M.; Hirbo, J.B.; Osman, M.; et al. Convergent adaptation of human lactase persistence in Africa and Europe. Nat. Genet. 2007, 39, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Labrie, V.; Buske, O.J.; Oh, E.; Jeremian, R.; Ptak, C.; Gasiūnas, G.; Maleckas, A.; Petereit, R.; Žvirbliene, A.; Adamonis, K.; et al. Lactase non persistence is directed by DNA-variation-dependent epigenetic aging. Nat. Struct. Mol. Biol. 2016, 23, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Oh, E.; Jeremian, R.; Oh, G.; Groot, D.; Susic, M.; Lee, K.; Foy, K.; Laird, P.W.; Petronis, A.; Labrie, V. Transcriptional heterogeneity in the lactase gene within cell-type is linked to the epigenome. Sci. Rep. 2017, 7, 41843. [Google Scholar] [CrossRef] [PubMed]
- Lewinsky, R.H.; Jensen, T.G.; Møller, J.; Stensballe, A.; Olsen, J.; Troelsen, J.T. T-13910 DNA variant associated with lactase persistence interacts with Oct-1 and stimulates lactase promoter activity in vitro. Hum. Mol. Genet. 2005, 14, 3945–3953. [Google Scholar] [CrossRef]
- Olsen, L.; Bressendorff, S.; Troelsen, J.T.; Olsen, J. Differentiation-dependent activation of the human intestinal alkaline phosphatase promoter by HNF-4 in intestinal cells. Am. J. Physiol. Gastrointest. Liver Physiol. 2005, 289, G220–G226. [Google Scholar] [CrossRef]
- Jensen, T.G.; Liebert, A.; Lewinsky, R.; Swallow, D.M.; Olsen, J.; Troelsen, J.T. The −14010*C variant associated with lactase persistence is located between an Oct-1 and HNF1α binding site and increases lactase promoter activity. Hum. Genet. 2011, 130, 483–493. [Google Scholar] [CrossRef]
- Fumery, M.; Speca, S.; Langlois, A.; Davila, A.M.; Dubuquoy, C.; Grauso, M.; Martin-Mena, A.; Figeac, M.; Metzger, D.; Rousseaux, C.; et al. Peroxisome proliferator-activated receptor gamma (PPARγ) regulates lactase expression and activity in the gut. EMBO Mol. Med. 2017, 9, 1471–1481. [Google Scholar] [CrossRef]
- Gerbault, P.; Moret, C.; Currat, M.; Sanchez-Mazas, A. Impact of selection and demography on the diffusion of lactase persistence. PLoS ONE 2000, 4, e6369. [Google Scholar] [CrossRef]
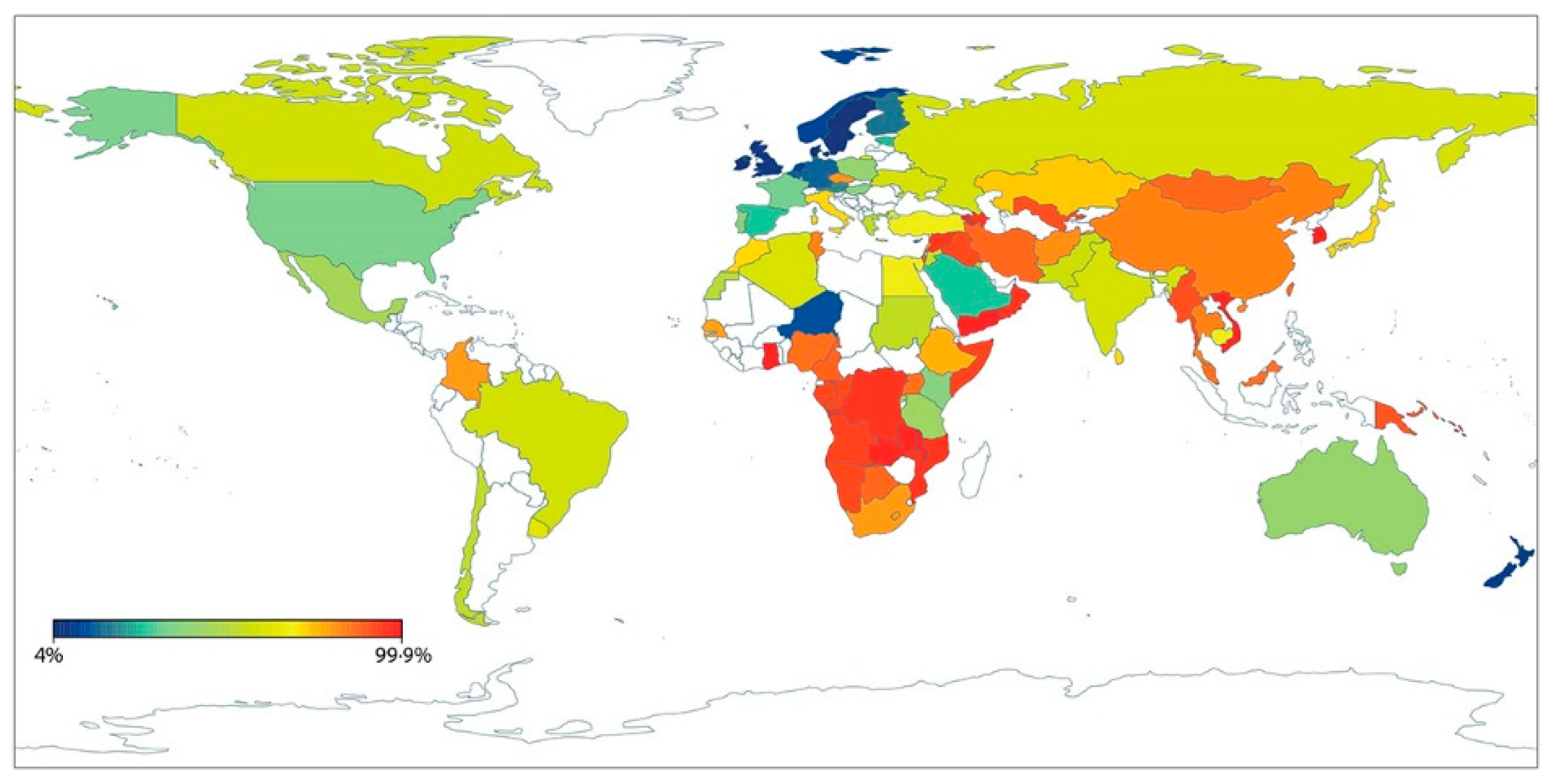
- Storhaug, C.L.; Fosse, S.K.; Fadnes, L.T. Country, regional, and global estimates for lactose malabsorption in adults: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2017, 2, 738–746. [Google Scholar] [CrossRef]
- Fazeli, W.; Kaczmarek, S.; Kirschstein, M.; Santer, R. A novel mutation within the lactase gene (LCT): The first report of congenital lactase deficiency diagnosed in Central Europe. BMC Gastroenterol. 2015, 15. [Google Scholar] [CrossRef] [PubMed]
- Uchida, N.; Sakamoto, O.; Irie, M.; Abukawa, D.; Takeyama, J.; Kure, S.; Tsuchiya, S. Two novel mutations in the lactase gene in a Japanese infant with congenital lactase deficiency. Tohoku J. Exp. Med. 2012, 227, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Robayo-Torres, C.C.; Nichols, B.L. Molecular differentiation of congenital lactase deficiency from adult-type hypolactasia. Nutr. Rev. 2007, 65, 95. [Google Scholar] [CrossRef] [PubMed]
- Torniainen, S.; Freddara, R.; Routi, T.; Gijsbers, C.; Catassi, C.; Höglund, P.; Savilahti, E.; Järvelä, I. Four novel mutations in the lactase gene (LCT) underlying congenital lactase deficiency (CLD). BMC Gastroenterol. 2009, 9, 8. [Google Scholar] [CrossRef] [PubMed]
- Saarela, T.; Similä, S.; Koivisto, M. Hypercalcemia and nephrocalcinosis in patients with congenital lactase deficiency. J. Pediatr. 1995, 6, 920–923. [Google Scholar] [CrossRef]
- Weaver, L.T.; Laker, M.F.; Nelson, R. Neonatal intestinal lactase activity. Arch. Dis. Child 1986, 61, 896–899. [Google Scholar] [CrossRef] [PubMed]
- Francavilla, R.; Calasso, M.; Calace, L.; Siragusa, S.; Ndagijimana, M.; Vernocchi, P.; Guerzoni, E. Effect of lactose on gut microbiota and metabolome of infants with cow’s milk allergy. Pediatr. Allergy Immunol. 2012, 23, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.L.N.; Muhardi, L.; Osatakul, S.; Hegar, B.; Vandenplas, Y.; Ludwig, T.; Bindels, J.; Van der Beek, E.M.; Quak, S.H. An electronic questionnaire survey evaluating the perceived prevalence and practices of lactose intolerance in 1 to 5 year old children in South East Asia. Pediatr. Gastroenterol. Hepatol. Nutr. 2018, 21, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Sahi, T. Genetics and epidemiology of adult-type hypolactasia. Scand. J. Gastroenterol. 1994, 29, 7–20. [Google Scholar] [CrossRef]
- Seppo, L.; Tuure, T.; Korpela, R.; Järvelä, I.; Rasinperä, H.; Sahi, T. Can primary hypolactasia manifest itself after the age of 20 years? A two-decade follow-up study. Scand. J. Gastroenterol. 2008, 43, 1082–1087. [Google Scholar] [CrossRef] [PubMed]
- Bayless, T.M.; Christopher, N.L. Disaccharidase deficiency. Am. J. Clin. Nutr. 1969, 22, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Rezaie, A.; Buresi, M.; Lembo, A.; Lin, H.; McCallum, R.; Rao, S.; Schmulson, M.; Valdovinos, M.; Zakko, S.; Pimentel, M. Hydrogen and methane-based breath testing in gastrointestinal disorders: The North American Consensus. Am. J. Gastroenterol. 2017, 112, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Dzialanski, Z.; Barany, M.; Engfeldt, P.; Magnuson, A.; Olsson, L.A.; Nilsson, T.K. Lactase persistence versus lactose intolerance: Is there an intermediate phenotype? Clin. Biochem. 2016, 49, 248–252. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, C.D.; Di Palma, J.A. Carbohydrate challenge tests: Do you need to measure methane? South Med. J. 2012, 105, 251–253. [Google Scholar] [CrossRef] [PubMed]
- Romagnuolo, J.; Schiller, D.; Bailey, R.J. Using breath tests wisely in a gastroenterology practice: An evidence-based review of indications and pitfalls in interpretation. Am. J. Gastroenterol. 2002, 97, 1113–1126. [Google Scholar] [CrossRef]
- Marton, A.; Xue, X.; Szilagyi, A. Meta-analysis: The diagnostic accuracy of lactose breath hydrogen or lactose tolerance tests for predicting the North European lactase polymorphism C/T-13910. Aliment. Pharmacol. Ther. 2012, 4, 429–440. [Google Scholar] [CrossRef]
- Furnari, M.; Bonfanti, D.; Parodi, A.; Franzè, J.; Savarino, E.; Bruzzone, L.; Savarino, V. A comparison between lactose breath test and quick test on duodenal biopsies for diagnosing lactase deficiency in patients with self-reported lactose intolerance. J. Gastroenterol. 2013, 47, 148–152. [Google Scholar] [CrossRef]
- Grant, J.D.; Bezerra, J.A.; Thompson, S.H.; Lemen, R.J.; Koldovsky, O.; Udall, J.N. Assessment of lactose absorption by measurement of urinary galactose. Gastroenterology 1989, 97, 895–899. [Google Scholar] [CrossRef]
- Bond, J.H.; Levitt, M.D. Quantitative measurement of lactose absorption. Gastroenterology 1976, 70, 1058–1062. [Google Scholar]
- Hertzler, S.R.; Huynh, B.-C.L.; Savaiano, D.A. How much lactose is low lactose? J. Am. Diet. Assoc. 1996, 96, 243–246. [Google Scholar] [CrossRef]
- Vesa, T.H.; Korpela, R.A.; Sahi, T. Tolerance to small amounts of lactose in lactose maldigesters. Am. J. Clin. Nutr. 1996, 64, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Suarez, F.L.; Savaiano, D.A.; Levitt, M.D. A comparison of symptoms after the consumption of milk or lactose-hydrolyzed milk by people with self-reported severe lactose intolerance. N. Engl. J. Med. 1995, 333, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Suarez, F.L.; Savaiano, D.; Arbisi, P.; Levitt, M.D. Tolerance to the daily ingestion of two cups of milk by individuals claiming lactose intolerance. Am. J. Clin. Nutr. 1997, 65, 1502–1506. [Google Scholar] [CrossRef] [PubMed]
- Oku, T.; Nakamura, S.; Ichinose, M. Maximum permissive dosage of lactose and lactitol for transitory diarrhea, and utilizable capacity for lactose in Japanese female adults. J. Nutr. Sci. Vitam. 2005, 51, 51–57. [Google Scholar] [CrossRef]
- Eadala, P.; Waud, J.; Matthews, S.; Green, J.; Campbell, A. Quantifying the ‘hidden’lactose in drugs used for the treatment of gastrointestinal conditions. Aliment. Pharmacol. Ther. 2009, 29, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Montalto, M.; Gallo, A.; Santoro, L.; D’onofrio, F.; Curigliano, V.; Covino, M.; Gasbarrini, G. Low-dose lactose in drugs neither increases breath hydrogen excretion nor causes gastrointestinal symptoms. Aliment. Pharmacol. Ther. 2008, 28, 1003–1012. [Google Scholar] [CrossRef]
- Dehkordi, N.; Rao, D.; Warren, A.; Chawan, C. Lactose malabsorption as influenced by chocolate milk, skim milk, sucrose, whole milk, and lactic cultures. J. Am. Diet. Assoc. 1995, 95, 484–486. [Google Scholar] [CrossRef]
- Laxminarayan, S.; Reifman, J.; Edwards, S.S.; Wolpert, H.; Steil, G.M. Bolus estimation—Rethinking the effect of meal fat content. Diabates Technol. Ther. 2015, 17, 860–866. [Google Scholar] [CrossRef]
- Villar, J.; Kestler, E.; Castillo, P.; Juarez, A.; Menendez, R.; Solomons, N.W. Improved lactose digestion during pregnancy: A case of physiologic adaptation? Obstet. Gynecol. 1988, 71, 697–700. [Google Scholar]
- Szilagyi, A.; Salomon, R.; Seidman, E. Influence of loperamide on lactose handling and oral-caecal transit time. Aliment. Pharmacol. Ther. 1996, 10, 765–770. [Google Scholar] [CrossRef] [PubMed]
- Kelle, J.; Bassotti, G.; Clarke, J.; Dinning, P.; Fox, M.; Grover, M.; Hellström, P.M.; Ke, M.; Layer, P.; Malagelada, C.; et al. International working group for disorders of gastrointestinal motility and function.expert consensus document: Advances in the diagnosis and classification of gastric and intestinal motility disorders. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 291–308. [Google Scholar] [CrossRef] [PubMed]
- Schmulson, M.J.; Drossman, D.A. What Is New in Rome IV. J. Neurogastroenterol. Motil. 2017, 23, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Savaiano, D.A.; Boushey, C.J.; McCabe, G.P. Lactose intolerance symptoms assessed by meta-analysis: A grain of truth that leads to exaggeration. J. Nutr. 2006, 136, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Jellema, P.; Schellevis, F.G.; van der Windt, D.A.; Kneepkens, C.M.; van der Horst, H.E. Lactose malabsorption and intolerance: A systematic review on the diagnostic value of gastrointestinal symptoms and self-reported milk intolerance. QJM 2010, 103, 555–572. [Google Scholar] [CrossRef] [PubMed]
- Matthews, S.B.; Campbell, A.K. When sugar is not so sweet. Lancet 2000, 15, 1330. [Google Scholar] [CrossRef]
- Matthews, S.B.; Waud, J.P.; Roberts, A.G.; Campbell, A.K. Systemic lactose intolerance: A new perspective on an old problem. Postgrad. Med. J. 2005, 81, 167–173. [Google Scholar] [CrossRef]
- Eadala, P.; Matthews, S.B.; Waud, J.P.; Geen, J.T.; Campbell, A.K. Association of lactose sensitivity with inflammatory bowel disease–demonstrated by analysis of genetic polymorphism, breath gases and symptoms. Aliment. Pharmacol. Ther. 2011, 34, 735–746. [Google Scholar] [CrossRef]
- Ledochowski, M.; Sperner-Unterweger, B.; Fuchs, D. Lactose malabsorption is associated with early signs of mental depression in females (A Preliminary Report). Digest. Dis. Sci. 1998, 43, 2513–2517. [Google Scholar] [CrossRef]
- Flaten, M.A.; Simonsen, T.; Olsen, H. Drug-related information generates placebo and nocebo responses that modify the drug response. Psychosom. Med. 1999, 61, 250–255. [Google Scholar] [CrossRef]
- Briet, F.; Pochart, P.; Marteau, P.; Flourie, B.; Arrigoni, E.; Rambaud, J.C. Improved clinical tolerance to chronic lactose ingestion in subjects with lactose intolerance: A placebo effect? Gut 1997, 41, 632–635. [Google Scholar] [CrossRef] [PubMed]
- Vernia, P.; Di Camillo, M.; Foglietta, T.; Avallone, V.E.; De Carolis, A. Diagnosis of lactose intolerance and the “nocebo” effect: The role of negative expectations. Dig. Liver Dis. 2010, 42, 616–619. [Google Scholar] [CrossRef] [PubMed]
- Tack, J.; Talley, N.J.; Camilleri, M.; Holtmann, G.; Hu, P.; Malagelada, J.R.; Stanghellini, V. Functional gastroduodenal disorders. Gastroenterology 2006, 130, 1466–1479. [Google Scholar] [CrossRef] [PubMed]
- Talley, N.J. Decade in review-FGIDs: ‘Functional’ gastrointestinal disorders—A paradigm shift. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 649–650. [Google Scholar] [CrossRef] [PubMed]
- Ford, A.C.; Lacy, B.E.; Talley, N.J. Irritable bowel syndrome. N. Engl. J. Med. 2017, 2017 29, 2566–2578. [Google Scholar] [CrossRef]
- Martin, C.R.; Osadchiy, V.; Kalani, A.; Mayer, E.A. The Brain-Gut-Microbiome Axis. Cell. Mol. Gastroenterol. Hepatol. 2018, 6, 133–148. [Google Scholar] [CrossRef]
- Barr, S.I. Perceived lactose intolerance in adult Canadians: A national survey. Appl. Physiol. Nutr. Metab. 2013, 38, 830–835. [Google Scholar] [CrossRef]
- Farup, P.; Monsbakken, K.; Vandvik, P. Lactose malabsorption in a population with irritable bowel syndrome. Scand. J. Gastroenterol. 2004, 39, 645–649. [Google Scholar] [CrossRef]
- Yang, J.; Deng, Y.; Chu, H.; Cong, Y.; Zhao, J.; Pohl, D.; Fox, M. Prevalence and presentation of lactose intolerance and effects on dairy product intake in healthy subjects and patients with irritable bowel syndrome. Clin. Gastroenterol. Hepatol. 2013, 3, 262–268. [Google Scholar] [CrossRef]
- Carabotti, M.; Scirocco, A.; Maselli, M.A.; Severi, C. The gut-brain axis: Interactions between enteric microbiota, central and enteric nervous systems. Ann. Gastroenterol. 2015, 28, 203–209. [Google Scholar]
- Lisker, R.; Solomons, N.W.; Briceno, R.P.; Mata, M.R. Lactase and placebo in the management of the irritable bowel syndrome: A double-blind, cross-over study. Am. J. Gastroenterol. 1989, 84, 756–762. [Google Scholar] [PubMed]
- Vesa, T.H.; Seppo, L.M.; Marteau, P.R.; Sahi, T.; Korpela, R. Role of irritable bowel syndrome in subjective lactose intolerance. Am. J. Clin. Nutr. 1998, 67, 710–715. [Google Scholar] [CrossRef] [PubMed]
- Lovell, R.M.; Ford, A.C. Global prevalence of and risk factors for irritable bowel syndrome. Clin. Gastroenterol. Hepatol. 2012, 10, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Szilagyi, A.; Xue, X. Comparison of geographic distributions of irritable bowel syndrome with inflammatory bowel disease fail to support common evolutionary roots. Irritable bowel syndrome and inflammatory bowel diseases are not related by evolution. Med. Hypotheses 2018, 110, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Fox, M.; Cong, Y.; Chu, H.; Zheng, X.; Long, Y.; Fried, M.; Dai, N. Lactose intolerance in irritable bowel syndrome patients with diarrhoea: The roles of anxiety, activation of the innate mucosal immune system and visceral sensitivity. Aliment. Pharmacol. Ther. 2014, 39, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Mishkin, S. Dairy sensitivity, lactose malabsorption, and elimination diets in inflammatory bowel disease. Am. J. Clin. Nutr. 1997, 65, 564–567. [Google Scholar] [CrossRef] [PubMed]
- Nolan-Clark, D.; Tapsell, L.C.; Hu, R.; Han, D.Y.; Ferguson, L.R. Effects of dairy products on Crohn’s disease symptoms are influenced by fat content and disease location but not lactose content or disease activity status in a New Zealand population. J. Am. Diet. Assoc. 2011, 1111, 1165–1172. [Google Scholar] [CrossRef]
- Jianqin, S.; Leiming, X.; Lu, X.; Yelland, G.W.; Ni, J.; Clarke, A.J. Effects of milk containing only A2 beta casein versus milk containing both A1 and A2 beta casein proteins on gastrointestinal physiology, symptoms of discomfort, and cognitive behavior of people with self-reported intolerance to traditional cows’ milk. Nutr. J. 2016, 15, 35. [Google Scholar] [CrossRef]
- Pal, S.; Woodford, K.; Kukuljan, S.; Ho, S. Milk intolerance, beta-casein and lactose. Nutrients 2015, 7, 7285–7297. [Google Scholar] [CrossRef]
- Høst, A.; Jacobsen, H.; Halken, S.; Holmenlund, D. The natural history of cow’s milk protein allergy/intolerance. Eur. J. Clin. Nutr. 1995, 49 (Suppl. 1), S13–S18. [Google Scholar]
- Wal, J.M. Bovine milk allergenicity. Ann. Allergy Asthma Immunol. 2004, 93, S2–S11. [Google Scholar] [CrossRef]
- Virta, L.J.; Kautiainen, H.; Kolho, K.L. Symptoms suggestive of cow’s milk allergy in infancy and pediatric inflammatory bowel disease. Pediatr. Allergy Immunol. 2016, 27, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Lam, H.Y.; Van Hoffen, E.; Michelsen, A.; Guikers, K.; Van Der Tas, C.; Bruijnzeel-Koomen, C.; Knulst, A. Cow’s milk allergy in adults is rare but severe: Both casein and whey proteins are involved. Clin. Exp. Allergy 2008, 38, 995–1002. [Google Scholar] [CrossRef] [PubMed]
- Paajanen, L.; Korpela, R.; Tuuren, T.; Honkanen, J.; Järvelä, I.; Ilonen, J.; Kokkonen, J. Cow milk is not responsible for most gastrointestinal immune-like syndromes-evidence from a population-based study. Am. J. Clin. Nutr. 2005, 82, 1327–1335. [Google Scholar] [CrossRef] [PubMed]
- Suchy, F.; Brannon, P.; Carpenter, T.; Fernandez, J.; Gilsanz, V.; Gould, J.; Mennella, J. National Institutes of Health Consensus Development Conference: Lactose intolerance and health. Ann. Intern. Med. 2010, 152, 792–796. [Google Scholar] [CrossRef] [PubMed]
- Shetty, S.; Kapoor, N.; Bondu, J.D.; Thomas, N.; Paul, T.V. Bone turnover markers: Emerging tool in the management of osteoporosis. Indian J. Endocrinol. Metab. 2016, 20, 846–852. [Google Scholar] [PubMed]
- Heaney, R.P. Dairy and bone health. J. Am. Coll. Nutr. 2009, 28 (Suppl. 1), 82S–90S. [Google Scholar] [CrossRef]
- Heaney, R.P. Calcium, dairy products and osteoporosis. J. Am. Coll. Nutr. 2000, 19 (Suppl. 2), 83S–99S. [Google Scholar] [CrossRef]
- Matlik, L.; Savaiano, D.; McCabe, G.; VanLoan, M.; Blue, C.L.; Boushey, C.J. Perceived milk intolerance is related to bone mineral content in 10- to 13-year-old female adolescents. Pediatrics 2007, 120, e669–e677. [Google Scholar] [CrossRef]
- Morales, E.; Azocar, L.; Maul, X.; Perez, C.; Chianale, J.; Miquel, J.F. The European lactase persistence genotype determines the lactase persistence state and correlates with gastrointestinal symptoms in the Hispanic and Amerindian Chilean population: A case-control and population-based study. BMJ Open 2011, 1, e000125. [Google Scholar] [CrossRef]
- Windey, K.; Houben, E.; Deroover, L.; Verbeke, K. Contribution of colonic fermentation and fecal water toxicity to the pathophysiology of lactose-intolerance. Nutrients 2015, 7, 7505–7522. [Google Scholar] [CrossRef] [PubMed]
- Lukito, W.; Malik, S.G.; Surono, I.S.; Wahlqvist, M.L. From lactose intolerance to lactose nutrition. Asia Pac. J. Clin. Nutr. 2015, 24, S1–S8. [Google Scholar] [PubMed]
- Baldan, A.; Tagliati, S.; Saccomandi, D.; Brusaferro, A.; Busoli, L.; Scala, A.; Malaventura, C.; Maggiore, G.; Borgna-Pignatti, C. Assessment of Lactose-Free Diet on the Phalangeal Bone Mineral Status in Italian Adolescents Affected by Adult-Type Hypolactasia. Nutrients 2018, 10, 558. [Google Scholar] [CrossRef] [PubMed]
- Balk, E.M.; Adam, G.P.; Langberg, V.N.; Earley, A.; Clark, P.; Ebeling, P.R.; Mithal, A.; Rizzoli, R.; Zerbini, C.A.F.; Pierroz, D.D.; et al. International Osteoporosis Foundation Calcium Steering Committee. Global dietary calcium intake among adults: A systematic review. Osteoporos. Int. 2017, 28, 3315–3324. [Google Scholar] [CrossRef] [PubMed]
- Honkanen, R.; Pulkkinen, P.; Järvinen, R.; Kröger, H.; Lindstedt, K.; Tuppurainen, M.; Uusitupa, M. Does lactose intolerance predispose to low bone density? A population-based study of perimenopausal Finnish women. Bone 1996, 19, 23–28. [Google Scholar] [CrossRef]
- Obermayer-Pietsch, B.M.; Bonelli, C.M.; Walter, D.E.; Kuhn, R.J.; Fahrleitner-Pammer, A.; Berghold, A.; Goessler, W.; Stepan, V.; Dobnig, H.; Leb, G.; et al. Genetic predisposition for adult lactose intolerance and relation to diet, bone density, and bone fractures. J. Bone Miner. Res. 2004, 19, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Enattah, N.; Välimäki, V.V.; Välimäki, M.J.; Löyttyniemi, E.; Sahi, T.; Järvelä, I. Molecularly defined lactose malabsorption, peak bone mass and bone turnover rate in young finnish men. Calcif. Tissue Int. 2004, 75, 488–493. [Google Scholar] [CrossRef]
- Goulding, A.; Taylor, R.W.; Keil, D.; Gold, E.; Lewis-Barned, N.J.; Williams, S.M. Lactose malabsorption and rate of bone loss in older women. Age Ageing. 1999, 28, 175–180. [Google Scholar] [CrossRef]
- Di Stefano, M.; Veneto, G.; Malservisi, S.; Cecchetti, L.; Minguzzi, L.; Strocchi, A.; Corazza, G.R. Lactose malabsorption and intolerance and peak bone mass. Gastroenterology 2002, 122, 1793–1799. [Google Scholar] [CrossRef]
- Wade, S.W.; Strader, C.; Fitzpatrick, L.A.; Anthony, M.S.; O’Malley, C.D. Estimating prevalence of osteoporosis: Examples from industrialized countries. Arch. Osteoporos. 2014, 9, 182. [Google Scholar] [CrossRef]
- Hernlund, E.; Svedb, A.; Ivergård, M.; Compston, J.; Cooper, C.; Stenmark, J.; McCloskey, E.V.; Jönsson, B.; Kanis, J.A. Osteoporosis in the European Union: Medical management, epidemiology and economic burden. A report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch. Osteoporos. 2013, 8, 136. [Google Scholar] [CrossRef] [PubMed]
- Thorning, T.K.; Bertram, H.C.; Bonjour, J.P.; de Groot, L.; Dupont, D.; Feeney, E.; Ipsen, R.; Lecerf, J.M.; Mackie, A.; McKinley, M.C.; et al. Whole dairy matrix or single nutrients in assessment of health effects: Current evidence and knowledge gaps. Am. J. Clin. Nutr. 2017, 105, 1033–1045. [Google Scholar] [CrossRef] [PubMed]
- Brouwer-Brolsma, E.M.; Sluik, D.; Singh-Povel, C.M.; Feskens, E.J.M. Dairy shows different associations with abdominal and BMI-defined overweight: Cross-sectional analyses exploring a variety of dairy products. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 451–460. [Google Scholar] [CrossRef]
- Drouin-Chartier, J.P.; Brassard, D.; Tessier-Grenier, M.; Côté, J.A.; Labonté, M.È.; Desroches, S.; Couture, P.; Lamarche, B. Systematic Review of the Association between Dairy Product Consumption and Risk of Cardiovascular-Related Clinical Outcomes. Adv. Nutr. 2016, 7, 1026–1040. [Google Scholar] [CrossRef] [PubMed]
- Wolf-Maier, K.; Cooper, R.S.; Kramer, H.; Banegas, J.R.; Giampaoli, S.; Joffres, M.R.; Poulter, N.; Primatesta, P.; Stegmayr, B.; Thamm, M. Hypertension treatment and control in five European countries, Canada, and the United States. Hypertension 2004, 43, 10–17. [Google Scholar] [CrossRef]
- Jayedi, A.; Zargar, M.S. Dietary calcium intake and hypertension risk: A dose-response meta-analysis of prospective cohort studies. Eur. J. Clin. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Schwedhelm, C.; Hoffmann, G.; Knüppel, S.; Iqbal, K.; Andriolo, V.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food Groups and Risk of Hypertension: A Systematic Review and Dose-Response Meta-Analysis of Prospective Studies. Adv. Nutr. 2017, 8, 793–803. [Google Scholar] [CrossRef] [PubMed]
- Ralston, R.A.; Lee, J.H.; Truby, H.; Palermo, C.E.; Walker, K.Z. A systematic review and meta-analysis of elevated blood pressure and consumption of dairy foods. J. Hum. Hypertens. 2012, 26, 3–13. [Google Scholar] [CrossRef]
- Soedamah-Muthu, S.S.; Verberne, L.D.; Ding, E.L.; Engberink, M.F.; Geleijnse, J.M. Dairy consumption and incidence of hypertension: A dose-response meta-analysis of prospective cohort studies. Hypertension. 2012, 60, 1131–1137. [Google Scholar] [CrossRef]
- Xu, J.Y.; Qin, L.Q.; Wang, P.Y.; Li, W.; Chang, C. Effect of milk tripeptides on blood pressure: A meta-analysis of randomized controlled trials. Nutrition 2008, 24, 933–940. [Google Scholar] [CrossRef]
- Cicero, A.F.; Aubin, F.; Azais-Braesco, V.; Borghi, C. Do the lactotripeptides isoleucine-proline-proline and valine-proline-proline reduce systolic blood pressure in European subjects? A meta-analysis of randomized controlled trials. Am. J. Hypertens. 2013, 26, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Timpson, N.J.; Greenwood, C.M.T.; Soranzo, N.; Lawson, D.J.; Richards, J.B. Genetic architecture: The shape of the genetic contribution to human traits and disease. Nat. Rev. Genet. 2018, 19, 110–124. [Google Scholar] [CrossRef] [PubMed]
- Hartwig, F.P.; Horta, B.L.; Smith, G.D.; de Mola, C.L.; Victora, C.G. Association of lactase persistence genotype with milk consumption, obesity and blood pressure: A Mendelian randomization study in the 1982 Pelotas (Brazil) Birth Cohort, with a systematic review and meta-analysis. Int. J. Epidemiol. 2016, 45, 1573–1587. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.; Huang, T.; Bergholdt, H.K.; Nordestgaard, B.G.; Ellervik, C.; Qi, L.; CHARGE Consortium. Dairy consumption, systolic blood pressure, and risk of hypertension: Mendelian randomization study. BMJ 2017, 356, j1000. [Google Scholar] [CrossRef] [PubMed]
- Bergholdt, H.K.; Nordestgaard, B.G.; Varbo, A.; Ellervik, C. Milk intake is not associated with ischaemic heart disease in observational or Mendelian randomization analyses in 98,529 Danish adults. Int. J. Epidemiol. 2015, 44, 587–603. [Google Scholar] [CrossRef] [PubMed]
- Turner, K.M.; Keogh, J.B.; Clifton, P.M. Dairy consumption and insulin sensitivity: A systematic review of short- and long-term intervention studies. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 3–8. [Google Scholar] [CrossRef]
- Morio, B.; Fardet, A.; Legrand, P.; Lecerf, J.M. Involvement of dietary saturated fats, from all sources or of dairy origin only, in insulin resistance and type 2 diabetes. Nutr. Rev. 2016, 74, 33–47. [Google Scholar] [CrossRef]
- Bergholdt, H.K.; Nordestgaard, B.G.; Ellervik, C. Milk intake is not associated with low risk of diabetes or overweight-obesity: A Mendelian randomization study in 97,811 Danish individuals. Am. J. Clin. Nutr. 2015, 102, 487–496. [Google Scholar] [CrossRef]
- Szilagyi, A. Complex interactions of obesity, dairy food intake and genetics of lactase. J. Obs. Chronic Dis. 2018, 2, 44–54. Available online: http://jobesitydiseases.com/2018/08/02/complex-interactions-of-obesity-dairy-food-intake-and-genetics-of-lactase/ (accessed on 11 December 2018). [CrossRef]
- Mendelian Randomization of Dairy Consumption Working Group. Dairy Consumption and Body Mass Index among Adults. Mendelian Randomization Analysis of 184802 Individuals from 25 Studies. Clin. Chem. 2018, 64, 183–191. [Google Scholar] [CrossRef]
- Cho, E.; Smith-Warner, S.A.; Spiegelman, D.; Beeson, W.L.; van den Brandt, P.A.; Colditz, G.A.; Folsom, A.R.; Fraser, G.E.; Freudenheim, J.L.; Giovannucci, E.; et al. Dairy foods, calcium, and colorectal cancer: A pooled analysis of 10 cohort studies. J. Natl. Cancer Inst. 2004, 96, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Lau, R.; Chan, D.S.; Vieira, R.; Greenwood, D.C.; Kampman, E.; Norat, T. Dairy products and colorectal cancer risk: A systematic review and meta-analysis of cohort studies. Ann. Oncol. 2012, 23, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Szilagyi, A.; Nathwani, U.; Vinokuroff, C.; Correa, J.A.; Shrier, I. The effect of lactose maldigestion on the relationship between dairy food intake and colorectal cancer: A systematic review. Nutr. Cancer 2006, 55, 141–150. [Google Scholar] [CrossRef]
- Hartman, T.J.; Albert, P.S.; Snyder, K.; Slattery, M.L.; Caan, B.; Paskett, E.; Iber, F.; Kikendall, J.W.; Marshall, J.; Shike, M.; et al. The association of calcium and vitamin D with risk of colorectal adenomas. J. Nutr. 2005, 135, 252–259. [Google Scholar] [PubMed]
- Park, S.-Y.; Murphy, S.P.; Wilkens, L.R.; Nomura, A.M.; Henderson, B.E.; Kolonel, L.N. Calcium and vitamin D intake and risk of colorectal cancer: The multiethnic cohort study. Am. J. Epidemiol. 2007, 165, 784–793. [Google Scholar] [CrossRef] [PubMed]
- Wallace, K.; Baron, J.A.; Cole, B.F.; Sandler, R.S.; Karagas, M.R.; Beach, M.A.; Haile, R.W.; Burke, C.A.; Pearson, L.H.; Mandel, J.S.; et al. Effect of calcium supplementation on the risk of large bowel polyps. J. Natl. Cancer Inst. 2004, 96, 921–925. [Google Scholar] [CrossRef] [PubMed]
- Weingarten, M.A.; Zalmanovici, A.; Yaphe, J. Dietary calcium supplementation for preventing colorectal cancer and adenomatous polyps. Cochrane Database Syst. Rev. 2008. [Google Scholar] [CrossRef] [PubMed]
- Baron, J.A.; Beach, M.; Mandel, S.; van Stolk, R.U.; Haile, R.W.; Sandler, R.S.; Rothstein, R.; Summers, R.W.; Snover, D.C.; Beck, G.J.; et al. Calcium supplements for the prevention of colorectal adenomas. N. Engl. J. Med. 1999, 340, 101–107. [Google Scholar] [CrossRef]
- Baron, J.A.; Barry, E.L.; Mott, L.A.; Rees, J.R.; Sandler, R.S.; Snover, D.C.; Bostick, R.M.; Ivanova, A.; Cole, B.F.; Ahnen, D.J.; et al. A Trial of Calcium and Vitamin D for the Prevention of Colorectal Adenomas. N. Engl. J. Med. 2015, 373, 1519–1530. [Google Scholar] [CrossRef]
- Crockett, S.D.; Barry, E.L.; Mott, L.A.; Ahnen, D.J.; Robertson, D.J.; Anderson, J.C.; Wallace, K.; Burke, C.A.; Bresalier, R.S.; Figueiredo, J.C.; et al. Calcium and vitamin D supplementation and increased risk of serrated polyps: Results from a randomized clinical trial. Gut 2018. [Google Scholar] [CrossRef]
- Zanabria, R.; Tellez, A.M.; Griffiths, M.; Corredig, M. Milk fat globule membrane isolate induces apoptosis in HT-29 human colon cancer cells. Food Funct. 2013, 4, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, A.; Baskaran, S.A.; Amalaradjou, M.A.; Venkitanarayanan, K. Anticarcinogenic properties of medium chain fatty acids on human colorectal, skin and breast cancer cells in vitro. Int. J. Mol. Sci. 2015, 16, 5014–5027. [Google Scholar] [CrossRef]
- Mohammadzadeh, M.; Faramarzi, E.; Mahdavi, R.; Nasirimotlagh, B.; Asghari Jafarabadi, M. Effect of conjugated linoleic acid supplementation on inflammatory factors and matrix metalloproteinase enzymes in rectal cancer patients undergoing chemoradiotherapy. Integr. Cancer Ther. 2013, 12, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Xiao, R.; Badger, T.M.; Simme, F.A. Dietary exposure to soy or whey proteins alters colonic global gene expression profiles during rat colon tumorigenesis. Mol. Cancer 2005, 4, 1. [Google Scholar] [CrossRef]
- Octoratou, M.; Merikas, E.; Malgarinos, G.; Stanciu, C.; Triantafillidis, J.K.A. Prospective study of pre-illness diet in newly diagnosed patients with Crohn’s disease. Rev. Med. Chir. Soc. Med. Nat. Iasi. 2012, 116, 40–49. [Google Scholar] [PubMed]
- Abubakar, I.; Myhill, D.J.; Hart, A.R.; Lake, I.R.; Harvey, I.; Rhodes, J.M.; Robinson, R.; Lobo, A.J.; Probert, C.S.; Hunter, P.R. A case-control study of drinking water and dairy products in Crohn’s Disease–further investigation of the possible role of Mycobacterium avium paratuberculosis. Am. J. Epidemiol. 2007, 165, 776–783. [Google Scholar] [CrossRef] [PubMed]
- Opstelten, J.L.; Leenders, M.; Dik, V.K.; Chan, S.S.; van Schaik, F.D.; Khaw, K.T.; Luben, R.; Hallmans, G.; Karling, P.; Lindgren, S.; et al. Dairy Products, Dietary Calcium, and Risk of Inflammatory Bowel Disease: Results from a European Prospective Cohort Investigation. Inflamm. Bowel Dis. 2016, 22, 1403–1411. [Google Scholar] [CrossRef] [PubMed]
- Cramer, D.W.; Harlow, B.L.; Willett, W.C.; Welch, W.R.; Bell, D.A.; Scully, R.E.; Ng, W.G.; Knapp, R.C. Galactose consumption and metabolism in relation to the risk of ovarian cancer. Lancet 1989, 2, 66–71. [Google Scholar] [CrossRef]
- Cramer, D.W. Epidemiologic aspects of early menopause and ovarian cancer. Ann. N. Y. Acad. Sci. 1990, 592, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Webb, P.M.; Bain, C.J.; Purdie, D.M.; Harvey, P.W.; Green, A. Milk consumption, galactose metabolism and ovarian cancer (Australia). Cancer Causes Control 1998, 9, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.; Moorman, P.G.; Alberg, A.J.; Barnholtz-Sloan, J.S.; Bondy, M.; Cote, M.L.; Funkhouser, E.; Peters, E.S.; Schwartz, A.G.; Terry, P.; et al. Dairy, calcium, vitamin D and ovarian cancer risk in African-American women. Br. J. Cancer 2016, 115, 1122–1130. [Google Scholar] [CrossRef] [PubMed]
- Faber, M.T.; Jensen, A.; Søgaard, M.; Høgdall, E.; Høgdall, C.; Blaakaer, J.; Kjaer, S.K. Use of dairy products, lactose, and calcium and risk of ovarian cancer—Results from a Danish case-control study. Acta Oncol. 2012, 51, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Orsini, N.; Wolk, A. Milk, milk products and lactose intake and ovarian cancer risk: A meta-analysis of epidemiological studies. Int. J. Cancer 2006, 118, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Genkinger, J.M.; Hunter, D.J.; Spiegelman, D.; Anderson, K.E.; Arslan, A.; Beeson, W.L.; Buring, J.E.; Fraser, G.E.; Freudenheim, J.L.; Goldbohm, R.A.; et al. Dairy products and ovarian cancer: A pooled analysis of 12 cohort studies. Cancer Epidemiol. Prev. Biomark. 2006, 15, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Merritt, M.A.; Poole, E.M.; Hankinson, S.E.; Willett, W.C.; Tworoger, S.S. Dairy food and nutrient intake in different life periods in relation to risk of ovarian cancer. Cancer Causes Control 2014, 25, 795–808. [Google Scholar] [CrossRef]
- Koralek, D.O.; Bertone-Johnson, E.R.; Leitzmann, M.F.; Sturgeon, S.R.; Lacey, J.V., Jr.; Schairer, C.; Schatzkin, A. Relationship between calcium, lactose, vitamin D, and dairy products and ovarian cancer. Nutr. Cancer 2006, 56, 22–30. [Google Scholar] [CrossRef]
- Mommers, M.; Schouten, L.J.; Goldbohm, R.A.; van den Brandt, P.A. Dairy consumption and ovarian cancer risk in the Netherlands Cohort Study on Diet and Cancer. Br. J. Cancer 2006, 94, 165–170. [Google Scholar] [CrossRef]
- Song, X.; Li, Z.; Ji, X.; Zhang, D. Calcium Intake and the Risk of Ovarian Cancer: A Meta-Analysis. Nutrients 2017, 9, 679. [Google Scholar] [CrossRef]
- Białek, A.; Tokarz, A. Conjugated linoleic acid as a potential protective factor in prevention of breast cancer. Postepy Hig. Med. Dosw. 2013, 67, 6–14. [Google Scholar] [CrossRef]
- Davoodi, H.; Esmaeili, S.; Mortazavian, A.M. Effects of milk and milk products consumption on cancer: A review. Compr. Rev. Food Sci. Food Saf. 2013, 12, 249–264. [Google Scholar] [CrossRef]
- Zang, J.; Shen, M.; Du, S.; Chen, T.; Zou, S. The Association between Dairy Intake and Breast Cancer in Western and Asian Populations: A Systematic Review and Meta-Analysis. J. Breast Cancer 2015, 18, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.Y.; Zhang, L.; He, K.; Qin, L.Q. Dairy consumption and risk of breast cancer: A meta-analysis of prospective cohort studies. Breast Cancer Res. Treat. 2011, 127, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.B.; Yu, J.C.; Kang, W.M.; Ma, Z.Q.; Ye, X.; Cao, Z.J. Association between dairy intake and gastric cancer: A meta-analysis of observational studies. PLoS ONE 2014, 9, e101728. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Shan, Z.; Ren, H.; Chen, W. Dairy consumption and gastric cancer risk: A meta-analysis of epidemiological studies. Nutr. Cancer 2015, 67, 555–568. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wang, X.; Yao, Q.; Qin, L.; Xu, C. Dairy Product, Calcium Intake and Lung Cancer Risk: A Systematic Review with Meta-Analysis. Sci. Rep. 2016, 6, 20624. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.Q.; Xu, J.Y.; Wang, P.Y.; Tong, J.; Hoshi, K. Milk consumption is a risk factor for prostate cancer in Western countries: Evidence from cohort studies. Asia Pac. J. Clin. Nutr. 2007, 16, 467–476. [Google Scholar] [PubMed]
- Lu, W.; Chen, H.; Niu, Y.; Wu, H.; Xia, D.; Wu, Y. Dairy products intake and cancer mortality risk: A meta-analysis of 11 population-based cohort studies. Nutr. J. 2016, 15, 91. [Google Scholar] [CrossRef]
- Travis, R.C.; Appleby, P.N.; Siddiq, A.; Allen, N.E.; Kaaks, R.; Canzian, F.; Feller, S.; Tjønneland, A.; Føns-Johnsen, N.; Overvad, K.; et al. Genetic variation in the lactase gene, dairy product intake and risk for prostate cancer in the European prospective investigation into cancer and nutrition. Int. J. Cancer 2013, 132, 1901–1910. [Google Scholar] [CrossRef]
- Harrison, S.; Lennon, R.; Holly, J.; Higgins, J.P.T.; Gardner, M.; Perks, C.; Gaunt, T.; Tan, V.; Borwick, C.; Emmet, P.; et al. Does milk intake promote prostate cancer initiation or progression via effects on insulin-like growth factors (IGFs)? A systematic review and meta-analysis. Cancer Causes Control 2017, 28, 497–528. [Google Scholar] [CrossRef]
- Belfiore, A.; Malaguarnera, R.; Vella, V.; Lawrence, M.C.; Sciacca, L.; Frasca, F.; Morrione, A.; Vigneri, R. Insulin Receptor Isoforms in Physiology and Disease: An Updated View. Endocr. Rev. 2017, 38, 379–431. [Google Scholar] [CrossRef]
- Bergström, R.; Adami, H.O.; Möhner, M.; Zatonski, W.; Storm, H.; Ekbom, A.; Tretli, S.; Teppo, L.; Akre, O.; Hakulinen, T. Increase in testicular cancer incidence in six European countries: A birth cohort phenomenon. J. Natl. Cancer Inst. 1996, 88, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Kim, J.; Elghiaty, A.; Ham, W.S. Recent global trends in testicular cancer incidence and mortality. Med. (Baltim.) 2018, 97, e12390. [Google Scholar] [CrossRef] [PubMed]
- Davies, T.W.; Palmer, C.R.; Ruja, E.; Lipscombe, J.M. Adolescent milk, dairy product and fruit consumption and testicular cancer. Br. J. Cancer 1996, 74, 657–660. [Google Scholar] [CrossRef]
- Stang, A.; Ahrens, W.; Baumgardt-Elms, C.; Stegmaier, C.; Merzenich, H.; de Vrese, M.; Schrezenmeir, J.; Jöckel, K.H. Adolescent milk fat and galactose consumption and testicular germ cell cancer. Cancer Epidemiol. Prev. Biomark. 2006, 15, 2189–2195. [Google Scholar] [CrossRef] [PubMed]
- McGlynn, K.A.; Sakoda, L.C.; Rubertone, M.V.; Sesterhenn, I.A.; Lyu, C.; Graubard, B.I.; Erickson, R.L. Body size, dairy consumption, puberty, and risk of testicular germ cell tumors. Am. J. Epidemiol. 2007, 165, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, X.; Zhang, D. Dairy Product Consumption and Risk of Non-Hodgkin Lymphoma: A Meta-Analysis. Nutrients 2016, 8, 120. [Google Scholar] [CrossRef]
- Thorning, T.K.; Raben, A.; Tholstrup, T.; Soedamah-Muthu, S.S.; Givens, I.; Astrup, A. Milk and dairy products: Good or bad for human health? An assessment of the totality of scientific evidence. Food Nutr. Res. 2016, 60, 32527. [Google Scholar] [CrossRef]
- Heine, R.G.; AlRefaee, F.; Bachina, P.; Deleon, J.C.; Geng, L.; Gong, S.; Madrazo, J.A.; Ngamphaiboon, J.; Ong, C.; Rogacion, J.M. Lactose intolerance and gastrointestinal cow’s milk allergy in infants and children-common misconceptions revisited. World Allergy Organ. J. 2017, 10, 1–8. [Google Scholar] [CrossRef]
- Silanikove, N.; Leitner, G.; Merin, U. The Interrelationships between lactose intolerance and the Modern Dairy Industry: Global Perspectives in Evolutional and Historical Backgrounds. Nutrients 2015, 7, 7312–7331. [Google Scholar] [CrossRef]
- Rozenberg, S.; Body, J.J.; Bruyère, O.; Bergmann, P.; Brandi, M.L.; Cooper, C.; Devogelaer, J.P.; Gielen, E.; Goemaere, S.; Kaufman, J.M.; et al. Effects of Dairy Products Consumption on Health: Benefits and Beliefs—A Commentary from the Belgian Bone Club and the European Society for clinical and Economic Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases. Calcif. Tissue Int. 2016, 98, 1–17. [Google Scholar] [CrossRef]
- Health Canada. Canadian Nutrient File, version 2015. 2015. Available online: https://www.canada.ca/en/health-canada/services/food-nutrition/healthy-eating/nutrient-data/canadian-nutrient-file-2015-download-files.html (accessed on 11 December 2018).
- Singhal, S.; Baker, R.D.; Baker, S.S. A Comparison of the Nutritional Value of Cow’s Milk and Nondairy beverages. J. Ped. Gastroenterol. Nutr. 2017, 64, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Mathai, J.K.; Liu, Y.; Stein, H.H. Values for digestible indispensable amino acid scores (DIAAS) for some dairy and plant proteins may better describe protein quality than values calculated using the concept for protein digestibility-corrected amino acid scores (PDCAAS). Br. J. Nutr. 2017, 117, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Isaac, K. Navigating the Milk Aisle. Dairy Farmers of Canada. Nourish Move Thrive. Available online: http://www.nourishmovethrive.ca/blog/2018/08/01/navigating-the-milk-aisle/ (accessed on 11 December 2018).
- Lehmann, U.; Hirche, F.; Stangl, G.I.; Hinz, K.; Westphal, S.; Dierkes, J. Bioavailability of vitamin D(2) and D(3) in healthy volunteers, a randomized placebo-controlled trial. J. Clin. Endocrinol. Metab. 2013, 98, 4339–4345. [Google Scholar] [CrossRef] [PubMed]
- Morency, M.E.; Birken, C.S.; Lebovic, G.; Chen, Y.; L’Abbé, M.; Lee, G.J.; Maguire, J.L.; TARGet Kids! Collaboration. Association between noncow milk beverage consumption and childhood height. Am. J. Clin. Nutr. 2017, 106, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Fenton, T. Plant Based Beverages—Are They Really Healthier for Young Children. November 2017. Available online: http://healthaction.ca/media-centre/members-in-the-news/312-dietitians-paediatricians-advise-parents-to-exercise-caution-with-plant-based-beverages.html (accessed on 11 December 2018).
- Lin, M.Y.; Dipalma, J.A.; Martini, M.C.; Gross, C.J.; Harlander, S.K.; Savaiano, D.A. Comparative effects of exogenous lactase (beta-galactosidase) preparations on in vivo lactose digestion. Dig. Dis. Sci. 1993, 38, 2022–2027. [Google Scholar] [CrossRef] [PubMed]
- Ibba, I.; Gilli, A.; Boi, M.F.; Usai, P. Effects of exogenous lactase administration on hydrogen breath excretion and intestinal symptoms in patients presenting lactose malabsorption and intolerance. Biomed. Res. Int. 2014, 2014, 680196. [Google Scholar] [CrossRef] [PubMed]
- De Vrese, M.; Laue, C.; Offick, B.; Soeth, E.; Repenning, F.; Thoß, A.; Schrezenmeir, J. A combination of acid lactase from Aspergillus oryzae and yogurt bacteria improves lactose digestion in lactose maldigesters synergistically: A randomized, controlled, double-blind cross-over trial. Clin. Nutr. 2015, 34, 394–399. [Google Scholar] [CrossRef]
- Habte, D.; Sterby, G.; Jijalmassen, B. Lactose malabsorption in Ethiopian children. Acta Paediatr. Scand. 1973, 62, 649–654. [Google Scholar] [CrossRef]
- Sadre, M.; Karbasi, K. Lactose intolerance in Iran. Am. J. Clin. Nutr. 1979, 32, 1948–1954. [Google Scholar] [CrossRef]
- Hertzler, S.R.; Savaiano, D.A. Colonic adaptation to daily lactose feeding in lactose maldigesters reduces lactose intolerance. Am. J. Clin. Nutr. 1996, 64, 232–236. [Google Scholar] [CrossRef]
- Szilagyi, A.; Malolepszy, P.; Yesovitch, S.; Nathwani, U.; Vinokuroff, C.; Cohen, A.; Xue, X. Inverse Dose effect of pretest dietary lactose intake on breath hydrogen results and symptoms in lactase non persistent subjects. Dig. Dis. Sci. 2005, 50, 2178–2182. [Google Scholar] [CrossRef] [PubMed]
- Szilagyi, A.; Rivard, J.; Fokeeff, K. Improved parameters of lactose maldigestion using lactulose. Dig. Dis. Sci. 2001, 46, 1509–1519. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Savaiano, D.A. In vitro lactose fermentation by human colonic bacteria is modified by Lactobacillus acidophilus supplementation. J. Nutr. 1997, 127, 1489–1495. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Savaiano, D.A. Modification of colonic fermentation by bifidobacteria and pH in vitro. Impact on lactose metabolism, short-chain fatty acid, and lactate production. Dig. Dis. Sci. 1997, 42, 2370–2377. [Google Scholar] [CrossRef] [PubMed]
- Szilagyi, A.; Shrier, I.; Heilpern, D.; Je, J.; Park, S.; Chong, G.; Lalonde, C.; Cote, L.F.; Lee, B. Differential impact of lactose/lactase phenotype on colonic microflora. Can. J. Gastroenterol. Hepatol. 2010, 24, 373–379. [Google Scholar] [CrossRef]
- Li, X.; Yin, J.; Zhu, Y.; Wang, X.; Hu, X.; Bao, W.; Huang, Y.; Chen, L.; Chen, S.; Yang, W.; et al. Effects of whole milk supplementation on gut microbiota and cardiometabolic biomarkers in subjects with and without lactose malabsorption. Nutrients 2018, 10, 1403. [Google Scholar] [CrossRef] [PubMed]
- Savaiano, D.A.; Ritter, A.J.; Klaenhammer, T.R.; James, G.M.; Longcore, A.T.; Chandler, J.R.; Walker, W.A.; Foyt, H.L. Improving lactose digestion and symptoms of lactose intolerance with a novel galacto-oligosaccharide (RP-G28): A randomized, double-blind clinical trial. Nutr. J. 2013, 12, 160. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Savaiano, D.A. Lactose digestion from yogurt: Mechanism and relevance. Am. J. Clin. Nutr. 2014, 99 (Suppl. 5), 1251S–1255S. [Google Scholar] [CrossRef]
- He, T.; Priebe, M.G.; Vonk, R.J.; Welling, G.W. Identification of bacteria with beta-galactosidase activity in faeces from lactase non-persistent subjects. FEMS Microbiol. Ecol. 2005, 54, 463–469. [Google Scholar] [CrossRef]
- Riboulet-Bisson, E.; Sturme, M.H.; Jeffery, I.B.; O’Donnell, M.M.; Neville, B.A.; Forde, B.M.; Claesson, M.J.; Harris, H.; Gardiner, G.E.; Casey, P.G.; et al. Effect of Lactobacillus salivarius bacteriocin Abp118 on the mouse and pig intestinal microbiota. PLoS ONE 2012, 7, e31113. [Google Scholar] [CrossRef] [PubMed]
- Ojetti, V.; Gigante, G.; Gabrielli, M.; Ainora, M.E.; Mannocci, A.; Lauritano, E.C.; Gasbarrini, G.; Gasbarrini, A. The effect of oral supplementation with Lactobacillus reuteri or tilactase in lactose intolerant patients: Randomized trial. Eur. Rev. Med. Pharmacol. Sci. 2010, 14, 163–170. [Google Scholar] [PubMed]
- Malolepszy, P.; Shrier, I.; Szilagyi, A. Adaptation to lactose may not be achieved by long term ingetsion of a multi-species containing probiotic: An extended study. Int. J. Probiotics Prebiotics 2006, 1, 113–120. [Google Scholar]
- Almeida, C.C.; Lorena, S.L.; Pavan, C.R.; Akasaka, H.M.; Mesquita, M.A. Beneficial effects of long-term consumption of a probiotic combination of Lactobacillus casei Shirota and Bifidobacterium breve Yakult may persist after suspension of therapy in lactose-intolerant patients. Nutr. Clin. Pract. 2012, 27, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Levri, K.M.; Ketvertis, K.; Deramo, M.; Merenstein, J.H.; D’Amico, F. Do probiotics reduce adult lactose intolerance? A systematic review. J. Fam. Pract. 2005, 54, 613–620. [Google Scholar] [PubMed]
- Oak, S.J.; Jha, R. The effects of probiotics in lactose intolerance: A systematic review. Crit. Rev. Food Sci. Nutr. 2018, 57, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.; Shepherd, S. Personal view: Food for thought–western lifestyle and susceptibility to Crohn’s disease. The FODMAP hypothesis. Aliment. Pharmacol. Therap. 2005, 21, 1399–1409. [Google Scholar] [CrossRef] [PubMed]
- Tuck, C.J.; Vanner, S.J. Dietary therapies for functional bowel symptoms: Recent advances, challenges, and future directions. Neurogastroenterol. Motil. 2018, 30. [Google Scholar] [CrossRef]
- O’Keeffe, M.; Jansen, C.; Martin, L.; Williams, M.; Seamark, L.; Staudacher, H.; Lomer, M. Long-term impact of the low-FODMAP diet on gastrointestinal symptoms, dietary intake, patient acceptability, and healthcare utilization in irritable bowel syndrome. Neurogastroenterol. Motil. 2017, 30, e13154. [Google Scholar] [CrossRef]
- Wilder-Smith, C.H.; Olesen, S.S.; Materna, A.; Drewes, A.M. Predictors of response to a low-FODMAP diet in patients with functional gastrointestinal disorders and lactose or fructose intolerance. Aliment. Pharmacol. Ther. 2017, 45, 1094–1106. [Google Scholar] [CrossRef]
- Aziz, I.; Trott, N.; Briggs, R.; North, J.R.; Hadjivassiliou, M.; Sanders, D.S. Efficacy of a gluten-free diet in subjects with irritable bowel syndrome-diarrhea unaware of their HLA-DQ2/8 genotype. Clin. Gastroenterol. Hepatol. 2016, 14, 696–703. [Google Scholar] [CrossRef] [PubMed]
- El-Salhy, M.; Gunnar Hatlebakk, J.; Helge Gilja, O.; Hausken, T. The relation between celiac disease, nonceliac gluten sensitivity and irritable bowel syndrome. Nutr. J. 2015, 69, 519–524. [Google Scholar] [CrossRef]
- Skodje, G.I.; Sarna, V.K.; Minelle, I.H.; Rolfsen, K.L.; Muir, J.G.; Gibson, P.R.; Veierød, M.B.; Henriksen, C.; Lundin, K.E.A. Fructan, Rather Than Gluten, Induces Symptoms in Patients With Self-Reported Non-Celiac Gluten Sensitivity. Gastroenterology 2018, 154, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Krogsgaard, L.; Lyngesen, M.; Bytzer, P. Systematic review: Quality of trials on the symptomatic effects of the low FODMAP diet for irritable bowel syndrome. Aliment. Pharmacol. Therap. 2017, 45, 1506–1513. [Google Scholar] [CrossRef]
- Dionne, J.; Yuan, Y.; Chey, W.D.; Lacy, B.E.; Saito, Y.A.; Quigley, E.M.M.; Moayyedi, P.A. Systematic Review and Meta-Analysis Evaluating the Efficacy of a Gluten-Free Diet and a Low FODMAPs Diet in Treating Symptoms of Irritable Bowel Syndrome. Am. J. Gastroenterol. 2018, 113, 1290–1300. [Google Scholar] [CrossRef] [PubMed]
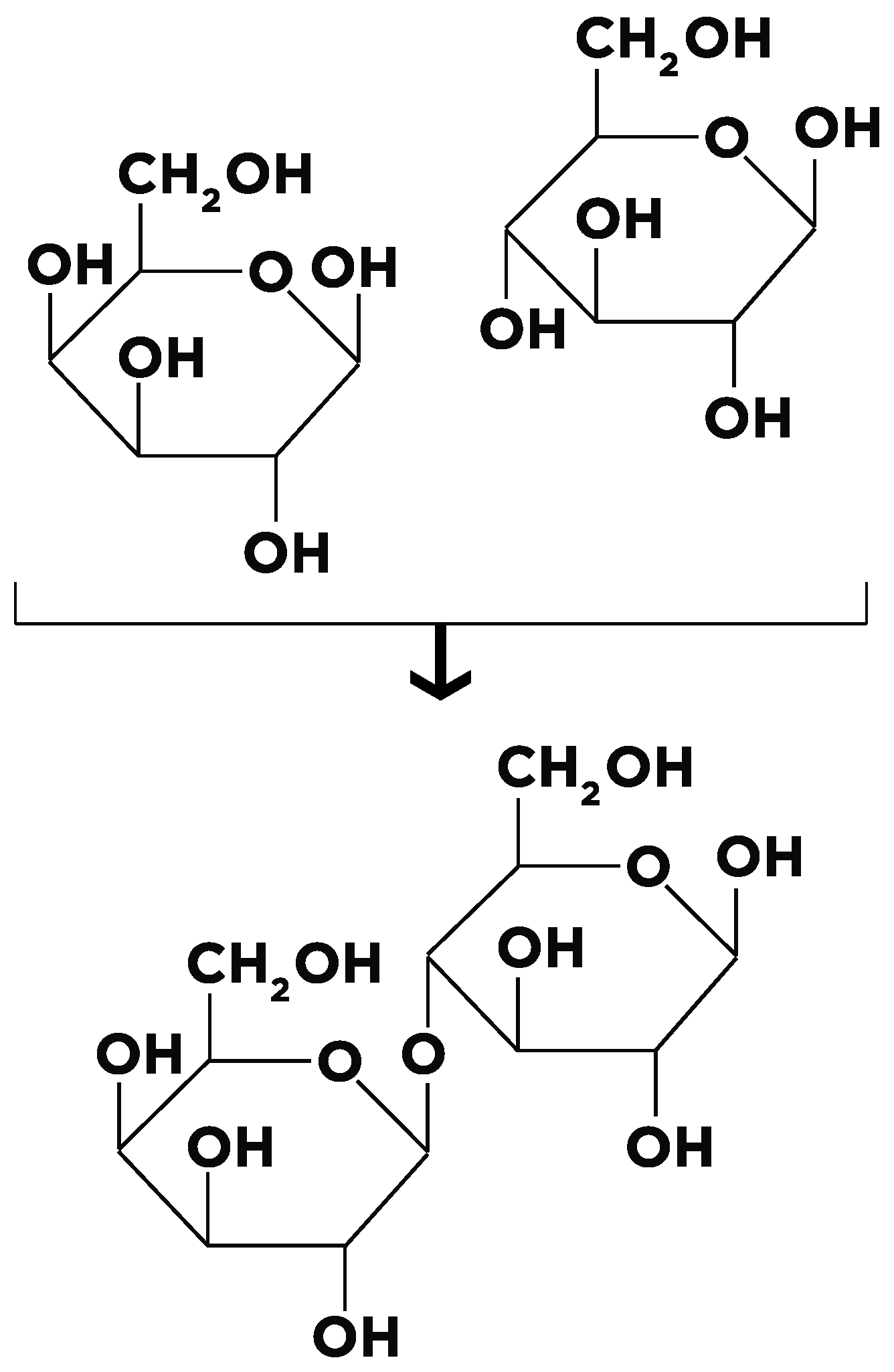
| Congenital Lactase deficiency [34,35,36,37] | Rare mutation at the lactase gene site described initially in Finland, now a few cases elsewhere also. It is associated with severe diarrhea acidosis and hypercalcemia. |
| Developmental Lactase deficiency [39,40] | Premature neonates usually have insufficient intestinal lactase Some term neonates may also have incomplete development of the enzyme |
| Primary Adult Lactase deficiency [42] | The most common cause of lactase deficiency. This is due to several polymorphisms in the transcription promoter region of the lactase gene. The ability to digest lactose into adulthood is a dominant trait and affects about 1/4–1/3 of the world’s population. |
| Secondary Lactase deficiency [44] | Diseases or toxins which affect the proximal small intestine can lead to loss of intestinal surface area with resulting lactase deficiency. After recovery the surface can improve, and unless there is also genetic predisposition lactose digestion may improve. Some examples of diseases are viral illness, e.g., Rota virus infection in children, unicellular parasites, e.g., Giardia, celiac disease, malnutrition, radiation exposure, upper gastrointestinal surgery, and some medications, e.g., olmesartan. |
| Term | Definition |
|---|---|
| Lactase Persistence (LP) | A dominant genetic trait usually associated with continued high levels of lactase production into adulthood. |
| Lactase Non-Persistence (LNP) | A recessive and ancestral genetic trait associated with a decline in intestinal lactase to < 10 u/g of tissue sometime between the end of weaning and adulthood. |
| Lactase Deficiency (LD) | Reduction of intestinal lactase enzyme from any cause, either genetic (LNP) or any secondary causes, like diseases or injury of the proximal small bowel mucosa. |
| Lactose Maldigestion (LM) | Inability to digest lactose for any reason, primary LNP, but also secondary causes. Most common tests for lactase deficiency are actually for LM. |
| Lactose Intolerance (LI) | Adverse symptoms resulting from the ingestion of lactose, including flatus, gas, bloating, cramps, diarrhea and, rarely, vomiting. LI may occur without LM. |
| Self-Reported LI (SRLI) | Persons believing themselves to be lactose intolerant without testing for LM. Nocebo and psychological characteristics may play a role in milk avoidance. |
| Lactose Sensitivity (LS) | Adverse symptoms with or without symptoms of LI and may also include depression, headache, and fatigue, with or without LM. LS symptoms may overlap with Irritable Bowel Syndrome. |
| Disease | TDF | Milk | Lfat | Hfat | Lactose | Cheese | Yogurt | Ferment | Calcium |
|---|---|---|---|---|---|---|---|---|---|
| Bone | - | - | - | - | ? - | - | - | - | - |
| MetS | - | ||||||||
| CAD | n | n | n | n | n | n | - | ? - | |
| CVD | n | n | n | ||||||
| HTN * | - | n | - | n | - | n | - | ||
| Stroke | - | - | - | ||||||
| T2Dm * | - | n | - | n | - | - | ? n/+ | ||
| Obes * | ?-/? n | ?-/?n | |||||||
| CRC | - | - | ? - | ? - | |||||
| IBD | - | - | - | ||||||
| Brst ca | ? - | - | ? - | - | - | ||||
| Ovar ca | ? + | n | n | ? n | ? n | ? - | |||
| Sto ca | ? - | n | |||||||
| Pros ca | + | + | + | + | |||||
| Test ca | ? + | ||||||||
| Lun ca | n | ||||||||
| Panc ca | n | ||||||||
| Blad ca | ? - | ||||||||
| NHL | ? + | + | + | n |
| 1. Gradual introduction of cow milk | Start with 30–60 mL per day and gradually increase to a maximum of 250 mL per day. Consume with meals rather than on an empty stomach to slow release of lactose in small intestine [181]. Higher fat milk may be better tolerated due to slower transit time. Consistency of consumption on a daily basis is key to building tolerance |
| 2. Inclusion of aged cheeses | Generally well tolerated due to their low lactose content (0.1–0.9 g of lactose in 30 g of hard cheese) [180] |
| 3. Inclusion of lactose reduced milk products | These are nutritionally identical to regular milk products [181] |
| 4. Use of lactose tablets and drops | Can be taken prior to consuming dairy foods or simultaneously with dairy meal |
| 5. Inclusion of other food sources of calcium such as dark green leafy vegetables, dried beans and legumes | This can help boost the intake of this mineral. Green leafy vegetables have the added benefit of contributing Vitamin K which plays an important role in calcium regulation and bone formation. Calcium bioavailability from these foods is lower than that from dairy due to the fibers, phytic, and oxalic acids [182]. Amounts of non-dairy foods evaluated to provide same amount of calcium from one serving of dairy (250 mL milk) are as follows: 1.1 servings fortified soy beverage 1.2 servings of bony fish 2.2 servings of green leafy vegetables It is important to note that these foods do not provide the equivalent profile of other nutrients and amounts needed can be unrealistic to consume in some cases [180] |
| 6. Consumption of fermented products like yogurt | These are produced by bacterial fermentation of milk lactose into lactic acid. Yogurts are also a source of probiotics and prebiotics, and both exert beneficial effects on gastrointestinal microflora [181]. Cultured bio yogurts and cultured milk blends which contain additional bacterial strains have become more available in recent years. However, results from an Israeli survey of leading brands showed a high lactose content not suitable for lactose intolerant people [181]. The rationale on the use of yogurt is further discussed below in conjunction with probiotics |
| NUTRIENT CONTENT per 250 mL | unit | Cow’s Milk Whole * | Soy Beverage ** | Plant Milk Beverage ** | Almond Beverage ** | Coconut Beverage ** | Cashew Beverage ** | 0at Beverage ** | Rice Beverage ** | Hemp Beverage ** |
|---|---|---|---|---|---|---|---|---|---|---|
| energy | k/cal | 157 | 90 | 110 | 30 | 50 | 25 | 130 | 130 | 60 |
| protein | g | 8 | 8 | 10 | 1 | 0.2 | 1 | 4 | 1 | 3 |
| Total fat | g | 8 | 4.5 | 5 | 2.5 | 4.5 | 2 | 2 | 2 | 4.5 |
| carbohydrate | g | 12 | 4 | 6 | 1 | 1 | 1 | 25 | 27 | 0 |
| calcium | mg | 291 | 300 | 450 | 300 | 300 | 300 | 121 | 360 | 282 |
| sodium | mg | 111 | 90 | 140 | 160 | 35 | 160 | 105 | N/A | 110 |
| potassium | mg | 340 | 360 | 330 | 35 | 30 | N/A | 133 | N/A | 100 |
| zinc | mg | 0.95 | 1.1 | 1.6 | 1.1 | 1.1 | 1.1 | N/A | N/A | 1 |
| iron | mg | 0.08 | 1.44 | N/A | 0.36 | 0.36 | 0.72 | 1 | 1 | 2 |
| Vitamin A | IU | 396 | 300 | 366 | 300 | 300 | 300 | N/A | 300 | N/A |
| Vitamin D | IU | 104 | 270 | 160 | 270 | 270 | 270 | 60 | 150 | 6µ0 |
| Vitamin B12 | µg | 1.16 | 1.2 | 2.7 | 1.2 | 1.2 | 1.2 | N/A | N/A | N/A |
| riboflavin | mg | 0.4 | 0.325 | N/A | 0.325 | 0.325 | 0.325 | N/A | N/A | N/A |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szilagyi, A.; Ishayek, N. Lactose Intolerance, Dairy Avoidance, and Treatment Options. Nutrients 2018, 10, 1994. https://doi.org/10.3390/nu10121994
Szilagyi A, Ishayek N. Lactose Intolerance, Dairy Avoidance, and Treatment Options. Nutrients. 2018; 10(12):1994. https://doi.org/10.3390/nu10121994
Chicago/Turabian StyleSzilagyi, Andrew, and Norma Ishayek. 2018. "Lactose Intolerance, Dairy Avoidance, and Treatment Options" Nutrients 10, no. 12: 1994. https://doi.org/10.3390/nu10121994
APA StyleSzilagyi, A., & Ishayek, N. (2018). Lactose Intolerance, Dairy Avoidance, and Treatment Options. Nutrients, 10(12), 1994. https://doi.org/10.3390/nu10121994




