Calcium Intake in Bone Health: A Focus on Calcium-Rich Mineral Waters
Abstract
1. Introduction
2. Background
3. Calcium-Rich Mineral Waters and Bone Metabolism
4. Case Report
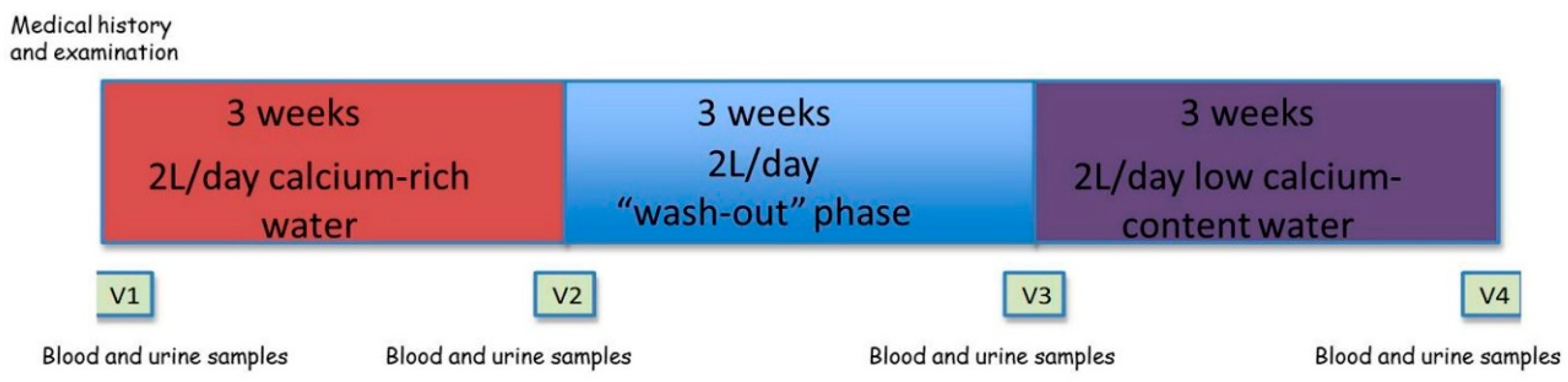
4.1. Subjects and Methods
4.2. Statistical Analysis
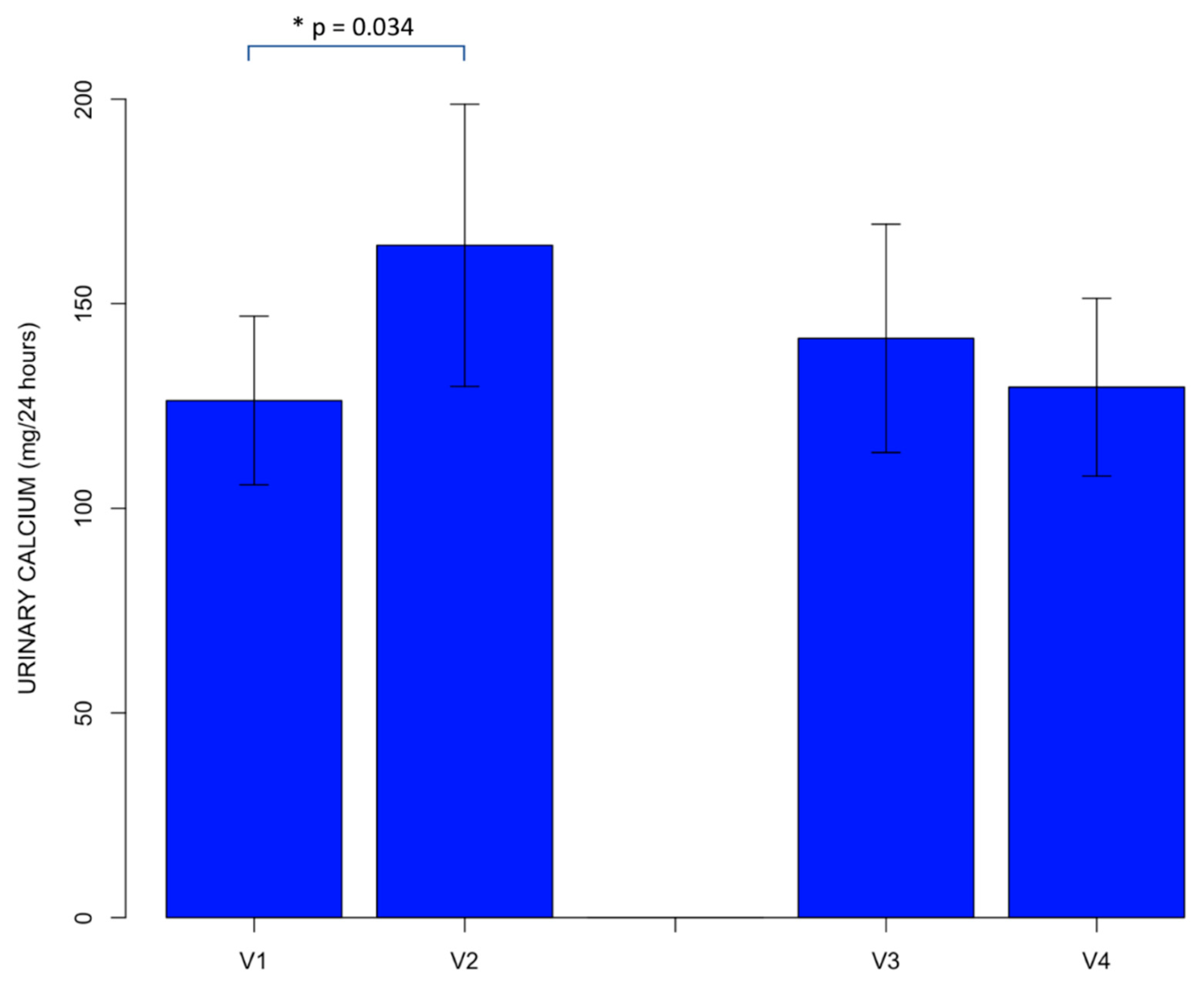
4.3. Results
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PTH | parathyroid hormone |
| 1,25(OH)2D | 1,25-dihydroxyvitamin D |
| FGF23 | Fibroblast Growth Factor 23 |
| VDR | vitamin D receptor |
| CTX | type 1 collagen cross-linked C-telopeptide |
| BALP | bone alkaline phosphatase |
References
- Bronner, F. Extracellular and intracellular regulation of calcium homeostasis. Sci. World J. 2001, 1, 919–925. [Google Scholar] [CrossRef] [PubMed]
- Bootman, M.D.; Collins, T.J.; Peppiatt, C.M.; Prothero, L.S.; MacKenzie, L.; de Smet, P.; Travers, M.; Tovey, S.C.; Seo, J.T.; Berridge, M.J.; et al. Calcium signalling—An overview. Semin. Cell. Dev. Biol. 2001, 12, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Khundmiri, S.J.; Murray, R.D.; Lederer, E. PTH and Vitamin D. Compr. Physiol. 2016, 6, 561–601. [Google Scholar] [CrossRef] [PubMed]
- Coulston, A.M.; Boushey, C.J.; Ferruzzi, M.G. Nutrition in the Prevention and Treatment of Disease, 3rd ed.; Academic Press: Cambridge, MA, USA, 2013; ISBN 978-0-12-391884-0. [Google Scholar]
- Wehrli, F.W. Structural and functional assessment of trabecular and cortical bone by micro magnetic resonance imaging. J. Magn. Reson. Imaging 2007, 25, 390–409. [Google Scholar] [CrossRef] [PubMed]
- Datta, H.K.; Ng, W.F.; Walker, J.A.; Tuck, S.P.; Varanasi, S.S. The cell biology of bone metabolism. J. Clin. Pathol. 2008, 61, 577–587. [Google Scholar] [CrossRef] [PubMed]
- Florencio-Silva, R.; Sasso, G.R.; Sasso-Cerri, E.; Simões, M.J.; Cerri, P.S. Biology of Bone Tissue: Structure, Function, and Factors That Influence Bone Cells. BioMed Res. Int. 2015, 2015, 421746. [Google Scholar] [CrossRef] [PubMed]
- Khosla, S.; Oursler, M.J.; Monroe, D.G. Estrogen and the skeleton. Trends Endocrinol. Metab. 2012, 23, 576–581. [Google Scholar] [CrossRef]
- Adams, D.J.; Rowe, D.W.; Ackert-Bicknell, C.L. Genetics of aging bone. Mamm. Genome 2016, 27, 367–380. [Google Scholar] [CrossRef]
- Sobacchi, C.; Schulz, A.; Coxon, F.P.; Villa, A.; Helfrich, M.H. Osteopetrosis: Genetics, treatment and new insights into osteoclast function. Nat. Rev. Endocrinol. 2013, 9, 522–536. [Google Scholar] [CrossRef]
- Carmeliet, G.; Dermauw, V.; Bouillon, R. Vitamin D signaling in calcium and bone homeostasis: A delicate balance. Best Pract. Res. Clin. Endocrinol. Metable. 2015, 29, 621–631. [Google Scholar] [CrossRef]
- Felsenfeld, A.; Rodriguez, M.; Levine, B. New insights in regulation of calcium homeostasis. Curr. Opin. Nephrol. Hypertens. 2013, 22, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, R. Nutritional aspects of bone health. Best Pract. Res. Clin. Endocrinol. Metab. 2014, 28, 795–808. [Google Scholar] [CrossRef] [PubMed]
- Viljakainen, H.T. Factors influencing bone mass accrual: Focus on nutritional aspects. Proc. Nutr. Soc. 2016, 75, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Higgs, J.; Derbyshire, E.; Styles, K. Nutrition and osteoporosis prevention for the orthopaedic surgeon: A wholefoods approach. EFORT Open Rev. 2017, 2, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Dietary Reference Intakes (DRIs): Elements Food and Nutrition Board, Institute of Medicine, National Academies. Available online: https://ods.od.nih.gov/Health_Information/Dietary_Reference_Intakes.aspx (accessed on 4 December 2018).
- Società Italiana di Nutrizione Umana (SINU). Livelli di Assunzione di Riferimento di Nutrienti ed Energia per la Popolazione Italiana (LARN))—IV Revision; SICS Ed.: Milan, Italy, 2014. [Google Scholar]
- Consiglio per la Ricerca in Agricoltura e l’Analisi dell’Economia Agraria (CREA). Linee Guida per una Sana Alimentazione Italiana—Linea Guida n 5: Bevi Ogni Giorno Acqua in Abbondanza; Istituto Nazionale di Ricerca per gli Alimenti e la Nutrizione (INRAN): Rome, Italy, rev. 2003.
- Quattrini, S.; Pampaloni, B.; Brandi, M.L. Natural mineral waters: Chemical characteristics and health effects. Clin. Cases Miner. Bone Metab. 2016, 13, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Dietary Reference Intakes (DRIs): Recommended Dietary Allowances and Adequate Intakes, Total Water and Macronutrients Food and Nutrition Board; Institute of Medicine, National Academies: Washington, DC, USA, 2005.
- European Parliament, Council of the European Union. Directive 2009/54/EC of the European Parliament and of the Council of 18 June 2009 on the Exploitation and Marketing of Natural Mineral Waters; Official Journal of the European Union: Bruxelles, Belgium, 2009. [Google Scholar]
- Bacciottini, L.; Tanini, A.; Falchetti, A.; Masi, L.; Franceschelli, F.; Pampaloni, B.; Giorgi, G.; Brandi, M.L. Calcium bioavailability from a calcium-rich mineral water, with some observations on method. J. Clin. Gastroenterol. 2004, 38, 761–766. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P.; Dowell, M.S. Absorbability of the calcium in a high-calcium mineral water. Osteoporos. Int. 1994, 4, 323–324. [Google Scholar] [CrossRef] [PubMed]
- Halpern, G.M.; van de Water, J.; Delabroise, A.M.; Keen, C.L.; Gershwin, M.E. Comparative uptake of calcium from milk and a calcium-rich mineral water in lactose intolerant adults: Implications for treatment of osteoporosis. Am. J. Prev. Med. 1991, 7, 379–383. [Google Scholar] [CrossRef]
- Couzy, F.; Kastenmayer, P.; Vigo, M.; Clough, J.; Munoz-Box, R.; Barclay, D.V. Calcium bioavailability from a calcium- and sulphate-rich mineral water, compared with milk, in young adult women. Am. J. Clin. Nutr. 1995, 62, 1239–1244. [Google Scholar] [CrossRef] [PubMed]
- Van Dokkum, W.; de la Gueronniere, V.; Schaafsma, G.; Bouley, C.; Luten, J.; Latgé, C. Bioavailability of calcium of fresh cheeses, enteral food and mineral water: A study with stable calcium isotopes in young adult women. Br. J. Nutr. 1996, 75, 893–903. [Google Scholar] [CrossRef] [PubMed]
- Wynckel, A.; Hanrotel, C.; Wuillai, A.; Charnard, J. Intestinal calcium absorption from mineral water. Miner. Electrolyte Metab. 1997, 23, 88–92. [Google Scholar] [PubMed]
- Greupner, T.; Schneider, I.; Hahn, A. Calcium bioavailability from mineral waters with different mineralization in comparison to milk and a supplement. J. Am. Coll. Nutr. 2017, 36, 386–390. [Google Scholar] [CrossRef]
- Brandolini, M.; Guéguen, L.; Boirie, Y.; Rousset, P.; Bertière, M.C.; Beaufrère, B. Higher calcium urinary loss induced by a calcium sulphate-rich mineral water intake than by milk in young women. Br. J. Nutr. 2005, 93, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Guillemant, J.; Le, H.T.; Guillemant, S.; Delabroise, A.M.; Arnaud, M.J. Acute effects induced by a calcium-rich mineral water on calcium metabolism and on parathyroid function. Osteoporos. Int. 1997, 7, 85–86. [Google Scholar] [CrossRef]
- Bohmer, H.; Müller, H.; Resch, K.L. Calcium supplementation with calcium-rich mineral waters: A systematic review and meta-analysis of its bioavailability. Osteoporos. Int. 2000, 11, 938–943. [Google Scholar] [CrossRef]
- Heaney, R.P. Absorbability and utility of calcium in mineral waters. Am. J. Clin. Nutr. 2006, 84, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Wynn, E.; Raetz, E.; Burckhardt, P. The composition of mineral waters sourced from Europe and North America in respect to bone health: Composition of mineral water optimal for bone. Br. J. Nutr. 2009, 101, 1195–1199. [Google Scholar] [CrossRef] [PubMed]
- Tucker, K.L.; Hannan, M.T.; Kiel, D.P. The acid-base hypothesis: Diet and bone in the Framingham Osteoporosis Study. Eur. J. Nutr. 2001, 40, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Meunier, P.J.; Jenvrin, C.; Munoz, F.; de la Gueronnière, V.; Garnero, P.; Menz, M. Consumption of a high calcium mineral water lowers biochemical indices of bone remodeling in postmenopausal women with low calcium intake. Osteoporos. Int. 2005, 16, 1203–1209. [Google Scholar] [CrossRef]
- Wynn, E.; Krieg, M.A.; Aeschlimann, J.M.; Burckhardt, P. Alkaline mineral water lowers bone resorption even in calcium sufficiency: Alkaline mineral water and bone metabolism. Bone 2009, 44, 120–124. [Google Scholar] [CrossRef]
- Cepollaro, C.; Orlandi, G.; Gonnelli, S.; Ferrucci, G.; Arditti, J.C.; Borracelli, D.; Toti, E.; Gennari, C. Effect of calcium supplementation as a high-calcium mineral water on bone loss in early postmenopausal women. Calcif. Tissue Int. 1996, 59, 238–239. [Google Scholar] [CrossRef] [PubMed]
- Costi, D.; Calcaterra, P.G.; Iori, N.; Vourna, S.; Nappi, G.; Passeri, M. Importance of bioavailable calcium drinking water for the maintenance of bone mass in post-menopausal women. J. Endocrinol. Investig. 1999, 22, 852–856. [Google Scholar] [CrossRef] [PubMed]
- Aptel, I.; Cance-Rouzaud, A.; Grandjean, H. Association between calcium ingested from drinking water and femoral bone density in elderly women: Evidence from the EPIDOS cohort. J. Bone Miner. Res. 1999, 14, 829–833. [Google Scholar] [CrossRef] [PubMed]
- Guillemant, J.; Le, H.T.; Accarie, C.; du Montcel, S.T.; Delabroise, A.M.; Arnaud, M.J.; Guillemant, S. Mineral water as a source of dietary calcium: Acute effects on parathyroid function and bone resorption in young men. Am. J. Clin. Nutr. 2000, 71, 999–1002. [Google Scholar] [CrossRef] [PubMed]
- Calcium Calculator. Available online: www.iofbonehealth.org/calcium-calculator (accessed on 26 January 2017).
- Massey, L.K.; Whiting, S.J. Dietary salt, urinary calcium, and bone loss. J. Bone Miner. Res. 1996, 11, 731–736. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, K.O.; Abrams, S.A.; Stuff, J.E.; Liang, L.K.; Welch, T.R. Variables related to urinary calcium excretion in young girls. J. Pediatr. Gastroenterol. Nutr. 1996, 23, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Burckhardt, P. The effect of the alkali load of mineral water on bone metabolism: Interventional studies. J. Nutr. 2008, 138, 435S–437S. [Google Scholar] [CrossRef] [PubMed]
| Life Stage Group | RDA/AI * (mg/day) |
|---|---|
| Infants | |
| 0 to 6 months | * 200 |
| 6 to 12 months | * 260 |
| Children | |
| 1–3 years | 700 |
| 4–8 years | 1000 |
| Males/Females | |
| 9–13 years | 1300 |
| 14–18 years | 1300 |
| 19–30 years | 1000 |
| 31–50 years | 1000 |
| 51–70 years (males) | 1000 |
| 51–70 years (females) | 1200 |
| >70 years | 1200 |
| Pregnancy/Breastfeeding | |
| 14–18 years | 1300 |
| 19–50 years | 1000 |
| Scientific Article | Year of Publication | Number of Recruited Subjects | Aim of the Study | Endpoint |
|---|---|---|---|---|
| Halpern et al. [24] | 1991 | 15 adult lactose-intolerant men | Bioavailability of calcium: calcium-rich water vs. milk | Tracer technique with calcium isotope |
| Heaney and Dowell [23] | 1994 | 18 healthy women | Bioavailability of calcium: calcium-rich water vs. milk | Tracer technique with calcium isotope |
| Couzy et al. [25] | 1995 | 9 healthy young adult women | Bioavailability of calcium: calcium- and sulphate-rich water vs. milk | Tracer technique with calcium isotope |
| Van Dokkum et al. [26] | 1996 | 12 healthy young adult women | Bioavailability of calcium: calcium-rich water vs. dairy products | Tracer technique with calcium isotope |
| Cepollaro et al. [37] | 1996 | 45 early postmenopausal women | Effects of calcium from calcium-rich water on densitometric parameters | Distal radius DXA |
| Wynckel et al. [27] | 1997 | 12 students (8 males and 4 females) | Intestinal absorption of calcium from mineral waters with different calcium content | Tracer technique with calcium isotope |
| Costi et al. [38] | 1999 | 255 pre- and post-menopausal women | Effects of calcium from calcium-rich water on densitometric parameters | Lumbar DXA |
| Aptel et al. [39] | 1999 | Data collection of 4434 women over 75 years from EPIDOS trial | Effects of calcium from drinking water on densitometric parameters | Femoral DXA |
| Guillemant et al. [40] | 2000 | 12 healthy young male students | Acute effects (within 4 h) of calcium from calcium-rich water on biochemical parameters of bone metabolism | PTH, serum and urinary CTX |
| Bacciottini et al. [22] | 2004 | 9 adult men + 9 pre-menopausal women + 9 post-menopausal women | Bioavailability of calcium: calcium-rich water vs. milk | Tracer technique with calcium isotope |
| Brandolini et al. [29] | 2005 | 37 healthy young women | Bioavailability of calcium: calcium- and sulphate-rich water vs. milk | Urinary calcium |
| Meunier et al. [35] | 2005 | 152 postmenopausal women with low dietary calcium intake | Effects of calcium from calcium-rich water on biochemical parameters of bone metabolism during a 6-month study period | PTH and biochemical markers of bone remodeling |
| Wynn et al. [36] | 2009 | 30 healthy premenopausal women with sufficient dietary calcium intake | Effects of calcium-rich water on biochemical parameters of bone metabolism during a 4-week study period: calcium-rich alkaline water vs. calcium-rich acid water | PTH and serum CTX |
| Greupner et al. [28] | 2017 | 21 healthy men and women | Bioavailability of calcium: 3 calcium-rich waters with different mineral content vs. milk vs. a calcium supplement | Serum and urinary calcium |
| Uliveto Water | B Water | |
|---|---|---|
| Fixed Residue at 180 °C (mg/L) | 745 | 174.1 |
| pH | 5.8 | 7.56 |
| Bicarbonate (mg/L) | 570 | 182.1 |
| Chloride (mg/L) | 80 | 6.78 |
| Litium (mg/L) | 0.18 | - |
| Nitrate (mg/L) | 7.1 | 1.10 |
| Sodium (mg/L) | 67 | 4.13 |
| Silicon (mg/L) | 8.9 | 4.17 |
| Calcium (mg/L) | 173 | 57.63 |
| Fluoride (mg/l) | 1 | 0.13 |
| Magnesium (mg/l) | 25 | 3.23 |
| Potassium (mg/L) | 7.3 | 3.23 |
| Sulphate (mg/L) | 100 | 6.75 |
| Carbon Dioxide (CO2) (mg/L) | 1485 | 8.08 |
| Strontium (mg/L) | - | 0.23 |
| V1 | V2 | p | V3 | V4 | p | |
|---|---|---|---|---|---|---|
| Serum Calcium (mg/dL) | 8.97 ± 0.35 | 8.94 ± 0.34 | 0.496 | 8.79 ± 0.36 | 8.93 ± 0.38 | 0.128 |
| Serum Phosphate (mg/dL) | 3.45 ± 0.48 | 3.51 ± 0.39 | 0.434 | 3.54 ± 0.91 | 3.47 ± 0.50 | 0.669 |
| Serum Magnesium (mg/dL) | 2.21 ± 0.13 | 2.17 ± 0.11 | 0.280 | 2.20 ± 0.20 | 2.19 ± 0.13 | 0.875 |
| Serum Total Proteins (g/dL) | 7.40 ± 0.32 | 7.31 ± 0.35 | 0.212 | 7.27 ± 0.29 | 7.31 ± 0.27 | 0.323 |
| Serum PTH (pmol/L) | 5.71 ± 3.04 | 5.67 ± 1.98 | 0.956 | 5.54 ± 2.09 | 5.41 ± 1.83 | 0.720 |
| Serum 25(OH)vitaminD (ng/mL) | 21.74 ± 7.98 | 21.95 ± 6.92 | 0.838 | 19.84 ± 6.32 | 20.97 ± 4.94 | 0.299 |
| Serum BALP (mcg/L) | 15.31 ± 4.20 | 15.60 ± 4.16 | 0.149 | 14.93 ± 4.14 | 14.75 ± 4.53 | 0.697 |
| Serum Beta-CTX (ng/mL) | 0.310 ± 0.121 | 0.319 ± 0.120 | 0.630 | 0.335 ± 0.132 | 0.337 ± 0.127 | 0.912 |
| Urinary Phosphate (mg/24 h) | 690.31 ± 427.30 | 600.18 ± 485.04 | 0.580 | 565.78 ± 307.40 | 531.14 ± 321.65 | 0.538 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vannucci, L.; Fossi, C.; Quattrini, S.; Guasti, L.; Pampaloni, B.; Gronchi, G.; Giusti, F.; Romagnoli, C.; Cianferotti, L.; Marcucci, G.; et al. Calcium Intake in Bone Health: A Focus on Calcium-Rich Mineral Waters. Nutrients 2018, 10, 1930. https://doi.org/10.3390/nu10121930
Vannucci L, Fossi C, Quattrini S, Guasti L, Pampaloni B, Gronchi G, Giusti F, Romagnoli C, Cianferotti L, Marcucci G, et al. Calcium Intake in Bone Health: A Focus on Calcium-Rich Mineral Waters. Nutrients. 2018; 10(12):1930. https://doi.org/10.3390/nu10121930
Chicago/Turabian StyleVannucci, Letizia, Caterina Fossi, Sara Quattrini, Leonardo Guasti, Barbara Pampaloni, Giorgio Gronchi, Francesca Giusti, Cecilia Romagnoli, Luisella Cianferotti, Gemma Marcucci, and et al. 2018. "Calcium Intake in Bone Health: A Focus on Calcium-Rich Mineral Waters" Nutrients 10, no. 12: 1930. https://doi.org/10.3390/nu10121930
APA StyleVannucci, L., Fossi, C., Quattrini, S., Guasti, L., Pampaloni, B., Gronchi, G., Giusti, F., Romagnoli, C., Cianferotti, L., Marcucci, G., & Brandi, M. L. (2018). Calcium Intake in Bone Health: A Focus on Calcium-Rich Mineral Waters. Nutrients, 10(12), 1930. https://doi.org/10.3390/nu10121930








