The Effect of a High-Dose Vitamin B Multivitamin Supplement on the Relationship between Brain Metabolism and Blood Biomarkers of Oxidative Stress: A Randomized Control Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Procedure
2.3. Treatment
2.4. Blood Biomarker Data Collection
2.5. 1H-MRS Biomarker Data Collection
2.6. 1H-MRS Biomarker Data Analysis
2.7. Statistical Analysis
3. Results
3.1. Blood Biomarker Group Differences
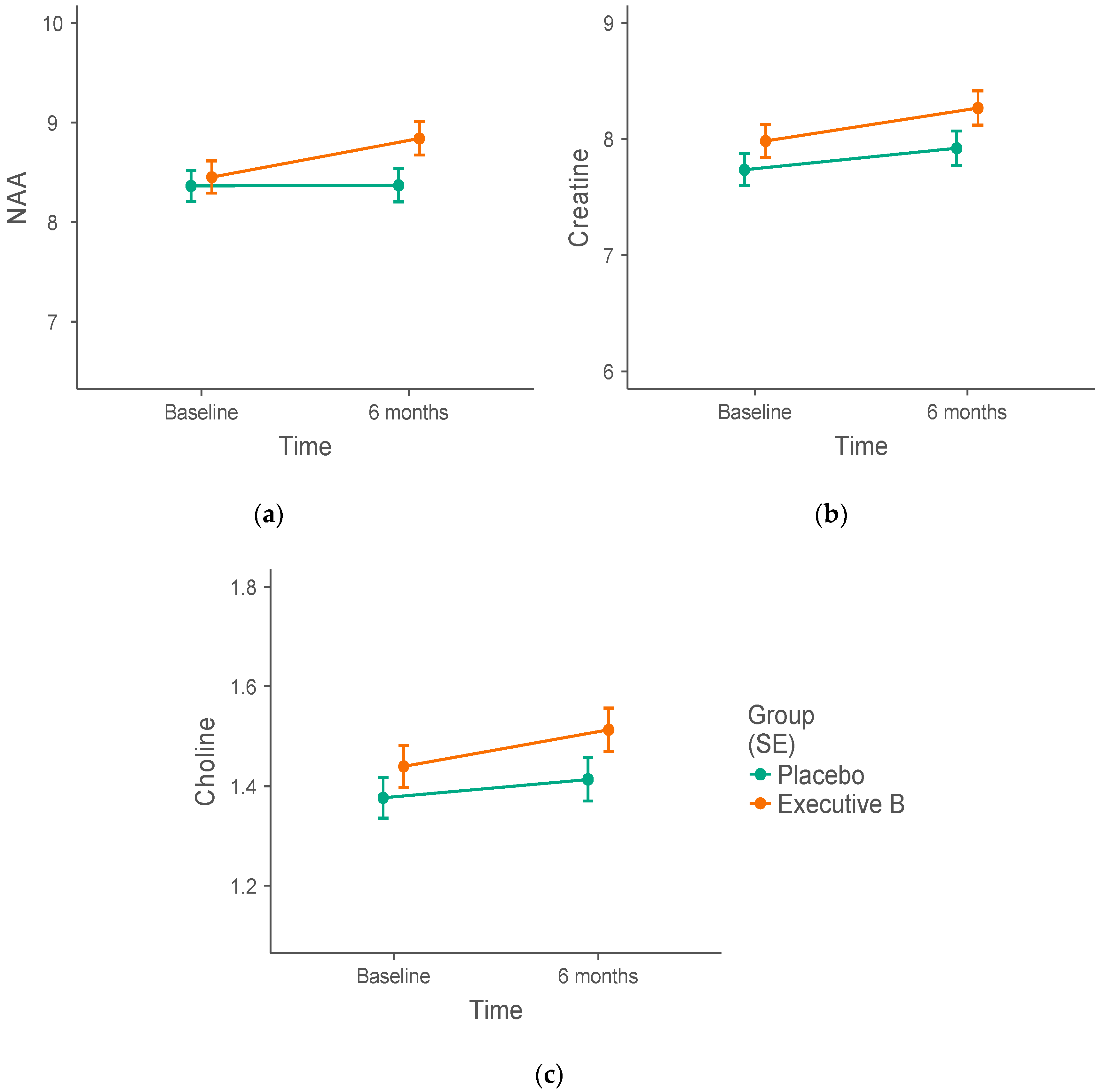
3.2. 1H-MRS Metabolite Biomarker Group Differences
3.3. Blood and Metabolite Biomarker Correlations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Smith, A.D.; Smith, S.M.; De Jager, C.A.; Whitbread, P.; Johnston, C.; Agacinski, G.; Oulhaj, A.; Bradley, K.M.; Jacoby, R.; Refsum, H. Homocysteine-lowering by B vitamins slows the rate of accelerated brain atrophy in mild cognitive impairment: A randomized controlled trial. PLoS ONE 2010, 5, e12244. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, E. Vitamin B12, folic acid, and the nervous system. Lancet Neurol. 2006, 5, 949–960. [Google Scholar] [CrossRef]
- Gröber, U.; Kisters, K.; Schmidt, J. Neuroenhancement with vitamin B12—Underestimated neurological significance. Nutrients 2013, 5, 5031–5045. [Google Scholar] [CrossRef] [PubMed]
- Smithells, R.; Sheppard, S.; Schorah, C. Vitamin dificiencies and neural tube defects. Arch. Dis. Child. 1976, 51, 944–950. [Google Scholar] [CrossRef] [PubMed]
- Haskell, C.F.; Robertson, B.; Jones, E.; Forster, J.; Jones, R.; Wilde, A.; Maggini, S.; Kennedy, D.O. Effects of a multi-vitamin/mineral supplement on cognitive function and fatigue during extended multi-tasking. Hum. Psychopharmacol. Clin. Exp. 2010, 25, 448–461. [Google Scholar] [CrossRef] [PubMed]
- Haskell, C.F.; Scholey, A.B.; Jackson, P.A.; Elliott, J.M.; Defeyter, M.A.; Greer, J.; Robertson, B.C.; Buchanan, T.; Tiplady, B.; Kennedy, D.O. Cognitive and mood effects in healthy children during 12 weeks’ supplementation with multi-vitamin/minerals. Br. J. Nutr. 2008, 100, 1086–1096. [Google Scholar] [CrossRef] [PubMed]
- Stough, C.; Scholey, A.; Lloyd, J.; Spong, J.; Myers, S.; Downey, L.A. The effect of 90 day administration of a high dose vitamin B-complex on work stress. Hum. Psychopharmacol. 2011, 26, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Scholey, A.; Bauer, I.; Neale, C.; Savage, K.; Camfield, D.; White, D.; Maggini, S.; Pipingas, A.; Stough, C.; Hughes, M. Acute effects of different multivitamin mineral preparations with and without guaraná on mood, cognitive performance and functional brain activation. Nutrients 2013, 5, 3589–3604. [Google Scholar] [CrossRef] [PubMed]
- White, D.J.; Cox, K.H.; Peters, R.; Pipingas, A.; Scholey, A.B. Effects of four-week supplementation with a multi-vitamin/mineral preparation on mood and blood biomarkers in young adults: A randomised, double-blind, placebo-controlled trial. Nutrients 2015, 7, 9005–9017. [Google Scholar] [CrossRef] [PubMed]
- Grima, N.A.; Pase, M.P.; Macpherson, H.; Pipingas, A. The effects of multivitamins on cognitive performance: A systematic review and meta-analysis. J. Alzheimer’s Dis. 2012, 29, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Scavuzzo, C.J.; Moulton, C.J.; Larsen, R.J. The use of magnetic resonance spectroscopy for assessing the effect of diet on cognition. Nutr. Neurosci. 2018, 21, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Scholey, A. Nutrients for neurocognition in health and disease: Measures, methodologies and mechanisms. Proc. Nutr. Soc. 2018, 77, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Blom, H.J.; Smulders, Y. Overview of homocysteine and folate metabolism. With special references to cardiovascular disease and neural tube defects. J. Inherit. Metab. Dis. 2011, 34, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Kruman, I.I.; Culmsee, C.; Chan, S.L.; Kruman, Y.; Guo, Z.; Penix, L.; Mattson, M.P. Homocysteine elicits a DNA damage response in neurons that promotes apoptosis and hypersensitivity to excitotoxicity. J. Neurosci. 2000, 20, 6920–6926. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.L. The methionine-homocysteine cycle and its effects on cognitive diseases.(Homocysteine & Cognitive). Altern. Med. Rev. 2003, 8, 7–20. [Google Scholar] [PubMed]
- Bryan, J. The role of nutritional factors in cognitive ageing. In Ageing Brain; CRC Press: Boca Raton, FL, USA, 2003; pp. 205–222. [Google Scholar]
- Carroll, D.; Ring, C.; Suter, M.; Willemsen, G. The effects of an oral multivitamin combination with calcium, magnesium, and zinc on psychological well-being in healthy young male volunteers: A double-blind placebo-controlled trial. Psychopharmacology 2000, 150, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Pipingas, A.; Camfield, D.A.; Stough, C.; Scholey, A.B.; Cox, K.H.; White, D.; Sarris, J.; Sali, A.; Macpherson, H. Effects of multivitamin, mineral and herbal supplement on cognition in younger adults and the contribution of B group vitamins. Hum. Psychopharmacol. Clin. Exp. 2014, 29, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Schoenthaler, S.J.; Bier, I.D.; Young, K.; Nichols, D.; Jansenns, S. The effect of vitamin-mineral supplementation on the intelligence of American schoolchildren: A randomized, double-blind placebo-controlled trial. J. Altern. Complement. Med. 2000, 6, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Mitra-Ganguli, T.; Kalita, S.; Bhushan, S.; Stough, C.; Kean, J.; Wang, N.; Sethi, V.; Khadilkar, A. A Randomized, Double-Blind Study Assessing Changes in Cognitive Function in Indian School Children Receiving a Combination of Bacopa monnieri and Micronutrient Supplementation vs. Placebo. Front. Pharmacol. 2017, 8, 678. [Google Scholar] [CrossRef] [PubMed]
- Pouwels, P.J.W.; Frahm, J. Regional Metabolite Concentration in Human Brains as Determined by Qulaitative Localized Proton MRS. Magn. Resonance Med. 1998, 39, 53–60. [Google Scholar] [CrossRef]
- Moffett, J.R.; Ross, B.; Arun, P.; Madhavarao, C.N.; Namboodiri, A.M. N-Acetylaspartate in the CNS: From neurodiagnostics to neurobiology. Progr. Neurobiol. 2007, 81, 89–131. [Google Scholar] [CrossRef] [PubMed]
- Pfefferbaum, A.; Adalsteinsson, E.; Spielman, D.; Sullivan, E.V.; Lim, K.O. In vivo spectroscopic quantification of the N-acetyl moiety, creatine, and choline from large volumes of brain gray and white matter: Effects of normal aging. Magn. Resonance Med. 1999, 41, 276–284. [Google Scholar] [CrossRef]
- Rae, C.D. A guide to the metabolic pathways and function of metabolites observed in human brain 1 H magnetic resonance spectra. Neurochem. Res. 2014, 39, 1–36. [Google Scholar] [CrossRef] [PubMed]
- Bisschops, R.H.; van der Graaf, Y.; Willem, P.T.M.; van der Grond, J. Elevated levels of plasma homocysteine are associated with neurotoxicity. Atherosclerosis 2004, 174, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-S.; Kuo, Y.-T.; Tsai, H.-Y.; Li, C.-W.; Lee, C.-C.; Yen, C.-F.; Lin, H.-F.; Ko, C.-H.; Juo, S.-H.H.; Yeh, Y.-C. Brain biochemical correlates of the plasma homocysteine level: A proton magnetic resonance spectroscopy study in the elderly subjects. Am. J. Geriatr. Psychiatry 2011, 19, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Leech, R.; Sharp, D.J. The role of the posterior cingulate cortex in cognition and disease. Brain 2013, 137, 12–32. [Google Scholar] [CrossRef] [PubMed]
- Raichle, M.E.; MacLeod, A.M.; Snyder, A.Z.; Powers, W.J.; Gusnard, D.A.; Shulman, G.L. A default mode of brain function. Proc. Natl. Acad. Sci. 2001, 98, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Elliott, R. Executive functions and their disordersImaging in clinical neuroscience. Br. Med. Bull. 2003, 65, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Stough, C.; Simpson, T.; Lomas, J.; McPhee, G.; Billings, C.; Myers, S.; Oliver, C.; Downey, L.A. Reducing occupational stress with a B-vitamin focussed intervention: A randomized clinical trial: Study protocol. Nutr. J. 2014, 13, 122. [Google Scholar] [CrossRef] [PubMed]
- Nutrient Reference Values for Australia and New Zealand Including Recommended Dietary Intakes. Available online: https://www.google.com.tw/url?sa=t&rct=j&q=&esrc=s&source=web&cd=2&ved=2ahUKEwj6k-vho-TeAhXLwrwKHQIkCeQQFjABegQIARAC&url=https%3A%2F%2Fnhmrc.gov.au%2Fsites%2Fdefault%2Ffiles%2Fimages%2Fnutrient-refererence-dietary-intakes.pdf&usg=AOvVaw3KsbZ99JYFGIamzVSkKIC6 (accessed on 18 October 2018).
- Haas, A. Localization of Unaffected Spins in NMR Imaging and Spectroscopy (LOCUS Spectroscopy). Magn. Reson. Med. 1986, 3, 963–969. [Google Scholar] [CrossRef]
- Wilson, M.; Reynolds, G.; Kauppinen, R.A.; Arvanitis, T.N.; Peet, A.C. A constrained least-squares approach to the automated quantitation of in vivo (1)H magnetic resonance spectroscopy data. Magn. Reson. Med. 2011, 65, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Egerton, A.; Stone, J.M.; Chaddock, C.A.; Barker, G.J.; Bonoldi, I.; Howard, R.M.; Merritt, K.; Allen, P.; Howes, O.D.; Murray, R.M. Relationship between brain glutamate levels and clinical outcome in individuals at ultra high risk of psychosis. Neuropsychopharmacology 2014, 39, 2891. [Google Scholar] [CrossRef] [PubMed]
- Berger, G.E.; Wood, S.J.; Wellard, R.M.; Proffitt, T.M.; McConchie, M.; Amminger, G.P.; Jackson, G.D.; Velakoulis, D.; Pantelis, C.; McGorry, P.D. Ethyl-eicosapentaenoic acid in first-episode psychosis. A 1H-MRS study. Neuropsychopharmacology 2008, 33, 2467. [Google Scholar] [CrossRef] [PubMed]
- Rousselet, G.A.; Pernet, C.R. Improving standards in brain-behavior correlation analyses. Front. Hum. Neurosci. 2012, 6, 119. [Google Scholar] [CrossRef] [PubMed]
- Strimmer, K. fdrtool: A versatile R package for estimating local and tail area-based false discovery rates. Bioinformatics 2008, 24, 1461–1462. [Google Scholar] [CrossRef] [PubMed]
- Jamovi Project Jamovi (Version 0.9) [Computer Software]. Available online: https://www.jamovi.org/ (accessed on 1 March 2018).
- Mattson, M.P.; Shea, T.B. Folate and homocysteine metabolism in neural plasticity and neurodegenerative disorders. Trends Neurosci. 2003, 26, 137–146. [Google Scholar] [CrossRef]
| Baseline | 6 Months | ||||
|---|---|---|---|---|---|
| Active | Placebo | Active | Placebo | ||
| Sex | Female | 9 | 11 | 8 | 10 |
| Male | 6 | 6 | 6 | 4 | |
| Total | 15 | 17 | 14 | 14 | |
| Age | 30–38 | 5 | 6 | 5 | 5 |
| 39–47 | 6 | 4 | 5 | 3 | |
| 48–56 | 2 | 4 | 2 | 3 | |
| 57–65 | 2 | 3 | 2 | 3 | |
| Total | 15 | 17 | 14 | 14 | |
| Active Ingredients (per tablet) | Dosage | RDI/AI | |
|---|---|---|---|
| Male | Female | ||
| Vitamin B1 (thiamine) | 75 mg | 6250% | 6818% |
| Vitamin B2 (riboflavin) | 10 mg | 769% | 909% |
| Vitamin B3 (nicotinamide/niacin) | 100 mg | 625% | 714% |
| Vitamin B5 | 68.7 mg | 1145% | 1718% |
| Vitamin B6 (pyridoxine) | 25 mg | 1923% * | 1923% * |
| 1470% # | 1667% # | ||
| Vitamin B7 (biotin) | 20 μg | 66.7% | 80% |
| Vitamin B9 (folic acid) | 150 μg | 37.5% | 37.5% |
| Vitamin B12 (cyanocobalamin) | 30 μg | 1250% | 1250% |
| Calcium phosphate | 100 mg | 10% | 10% |
| Calcium ascorbate | 145 mg | 14.5% | 14.5% * |
| 11.2% # | |||
| Ascorbic acid | 130 mg | 289% | 289% |
| Total vitamin C | 250 mg | 556% | 556% |
| d-alpha-tocopheryl acid succinate (vitamin E) | 41.3 mg | 413% | 590% |
| Magnesium phosphate | 140 mg | 33.3% | 43.8% |
| Potassium phosphate monobasic | 117.3 mg | 3.1% | 4.1% |
| Choline birartrate | 25 mg | 4.5% | 5.9% |
| Lecithin | 50 mg | NR | NR |
| Inositol | 25 mg | NR | NR |
| Avena sativa (oats) | 100 mg | NR | NR |
| Passifloraincarnata (passion flower) | 250 mg | NR | NR |
| Baseline | 6 Months | |||
|---|---|---|---|---|
| Active | Placebo | Active | Placebo | |
| N | 15 | 15 | 13 | 13 |
| SNR | 22.96 (3.68) | 26.38 (3.56) | 24.91 (2.37) | 26.92 (4.29) |
| FWHMwater | 8.15 (1.38) | 8.38 (1.35) | 7.84 (0.88) | 7.91 (1.05) |
| GM (%) | 73.27 (3.59) | 72.39 (4.35) | 72.36 (3.73) | 71.81 (3.80) |
| WM (%) | 22.17 (3.48) | 23.55 (4.89) | 22.83 (3.43) | 23.57 (4.58) |
| CSF (%) | 4.56 (1.58) | 4.05 (1.63) | 4.82 (1.82) | 4.63 (1.79) |
| Baseline | 6 Months | ||||
|---|---|---|---|---|---|
| Active | Placebo | Active | Placebo | ||
| Blood | N | 15 | 15 | 14 | 13 |
| HCy (μmol/L) | 9.57 ± 0.49 | 8.95 ± 0.51 | 7.98 ± 0.46 * | 10.05 ± 0.54 | |
| Vit B6 (nmol/L) | 191.07 ± 68.2 | 124.13 ± 34.6 | 619.79 ± 76.4 * | 82.25 ± 5.6 | |
| Vit B12 (pmol/L) | 285.53 ± 21.5 | 296.27 ± 28.4 | 443.50 ± 43.3 * | 275.85 ± 25.8 | |
| Folate (nmol/L) | 1265.60 ± 58.8 | 1141.27 ± 57.9 | 1317.07 ± 45.7 | 1242.08 ± 42.9 | |
| 1H-MRS | N | 14 | 15 | 13 | 13 |
| PCC | NAA (IU) | 8.46 ± 0.17 | 8.36 ± 0.20 | 8.85 ± 0.13 | 8.38 ± 0.14 |
| Choline (IU) | 1.44 ± 0.05 | 1.38 ± 0.04 | 1.52 ± 0.03 | 1.42 ± 0.04 | |
| Creatine (IU) | 7.97 ± 0.19 | 7.74 ± 0.15 | 8.23 ± 0.12 | 7.96 ± 0.13 | |
| mI (IU) | 4.95 ± 0.21 | 4.53 ± 0.15 | 4.99 ± 0.13 | 4.73 ± 0.15 | |
| Glx (IU) | 13.56 ± 0.70 | 11.44 ± 0.47 | 12.05 ± 0.37 | 11.82 ± 0.64 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ford, T.C.; Downey, L.A.; Simpson, T.; McPhee, G.; Oliver, C.; Stough, C. The Effect of a High-Dose Vitamin B Multivitamin Supplement on the Relationship between Brain Metabolism and Blood Biomarkers of Oxidative Stress: A Randomized Control Trial. Nutrients 2018, 10, 1860. https://doi.org/10.3390/nu10121860
Ford TC, Downey LA, Simpson T, McPhee G, Oliver C, Stough C. The Effect of a High-Dose Vitamin B Multivitamin Supplement on the Relationship between Brain Metabolism and Blood Biomarkers of Oxidative Stress: A Randomized Control Trial. Nutrients. 2018; 10(12):1860. https://doi.org/10.3390/nu10121860
Chicago/Turabian StyleFord, Talitha C., Luke A. Downey, Tamara Simpson, Grace McPhee, Chris Oliver, and Con Stough. 2018. "The Effect of a High-Dose Vitamin B Multivitamin Supplement on the Relationship between Brain Metabolism and Blood Biomarkers of Oxidative Stress: A Randomized Control Trial" Nutrients 10, no. 12: 1860. https://doi.org/10.3390/nu10121860
APA StyleFord, T. C., Downey, L. A., Simpson, T., McPhee, G., Oliver, C., & Stough, C. (2018). The Effect of a High-Dose Vitamin B Multivitamin Supplement on the Relationship between Brain Metabolism and Blood Biomarkers of Oxidative Stress: A Randomized Control Trial. Nutrients, 10(12), 1860. https://doi.org/10.3390/nu10121860





