Effect of Cistanche Tubulosa Extracts on Male Reproductive Function in Streptozotocin–Nicotinamide-Induced Diabetic Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. In Vitro Analysis
2.2.2. In Vivo Analysis
2.3. Statistical Analysis
3. Results
3.1. In Vitro Analysis
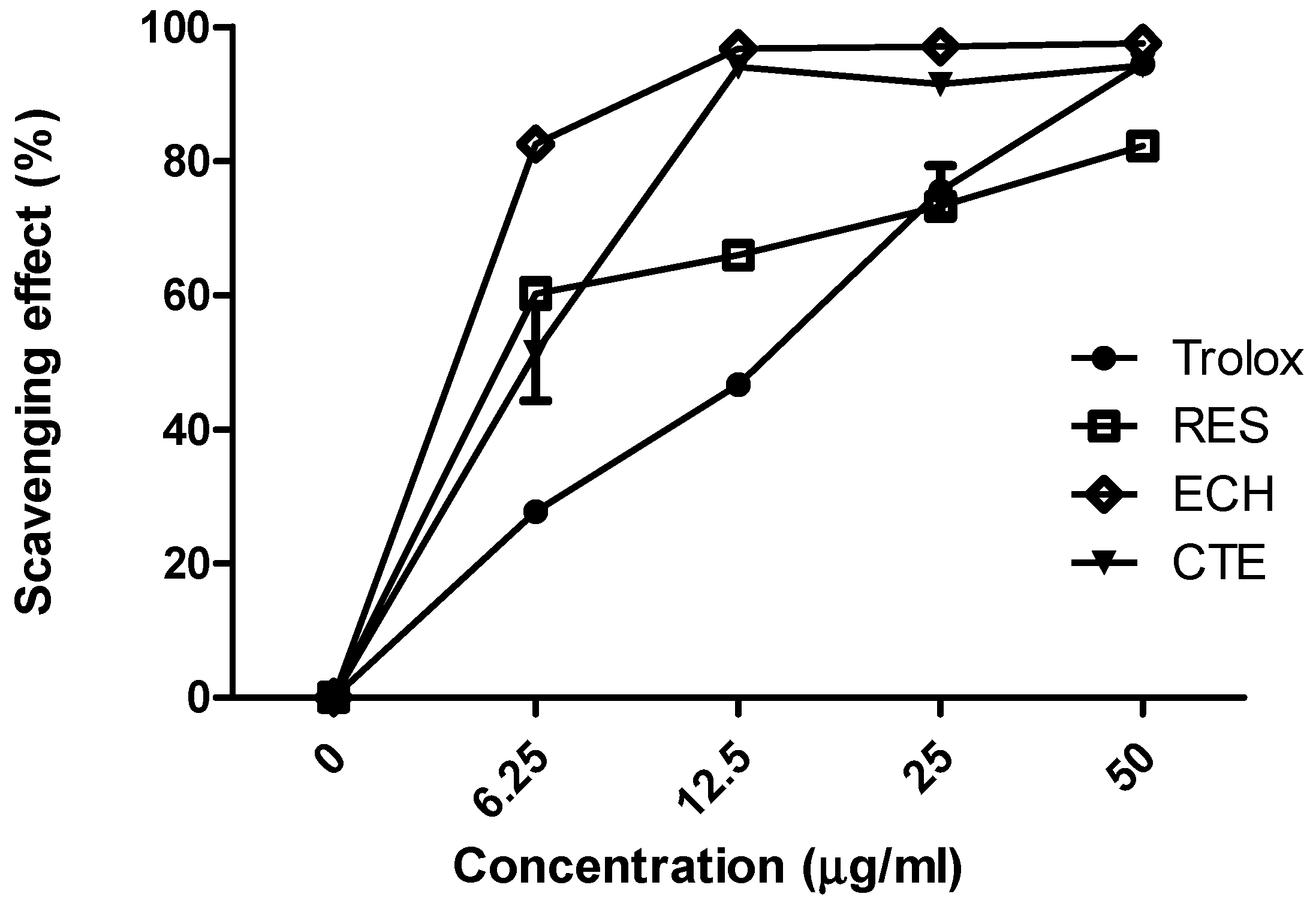
3.1.1. Comparison of Antioxidant Activities of ECH, CTE, and RES
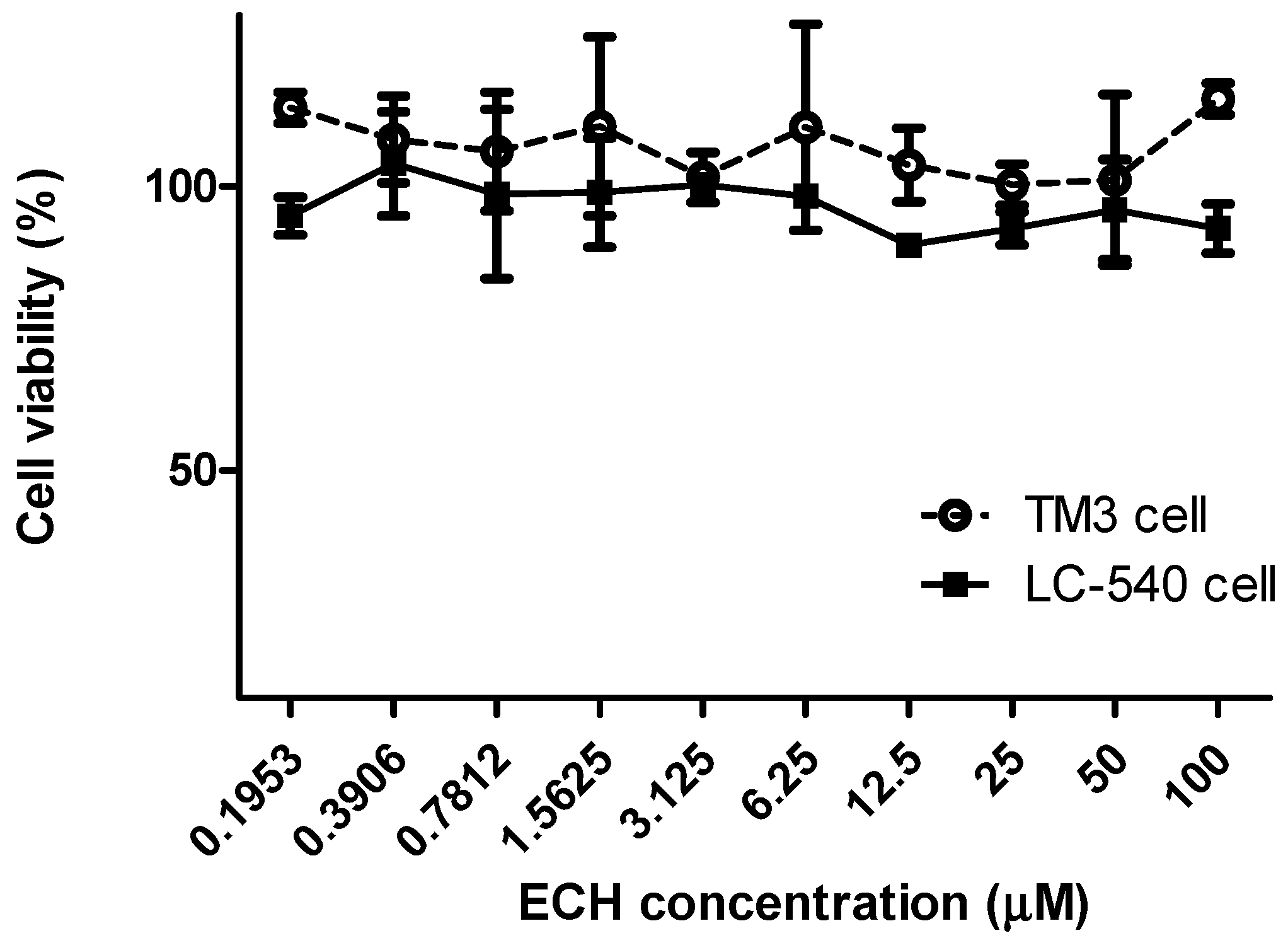
3.1.2. Cell Viability of ECH on LC-540 and TM3 Leydig Cells
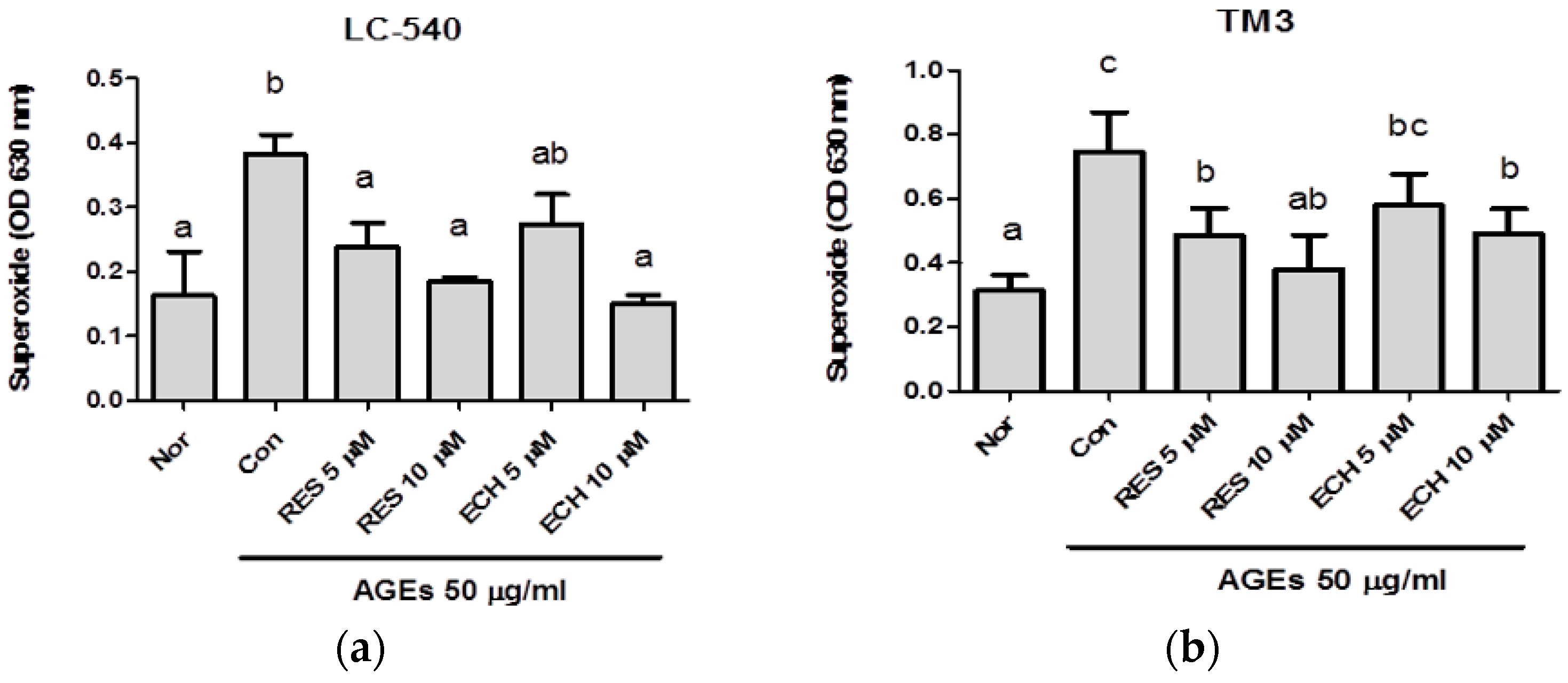
3.1.3. Effect of ECH on AGE-Induced Superoxide Production by NBT Assay in LC-540 and TM3 Leydig Cells
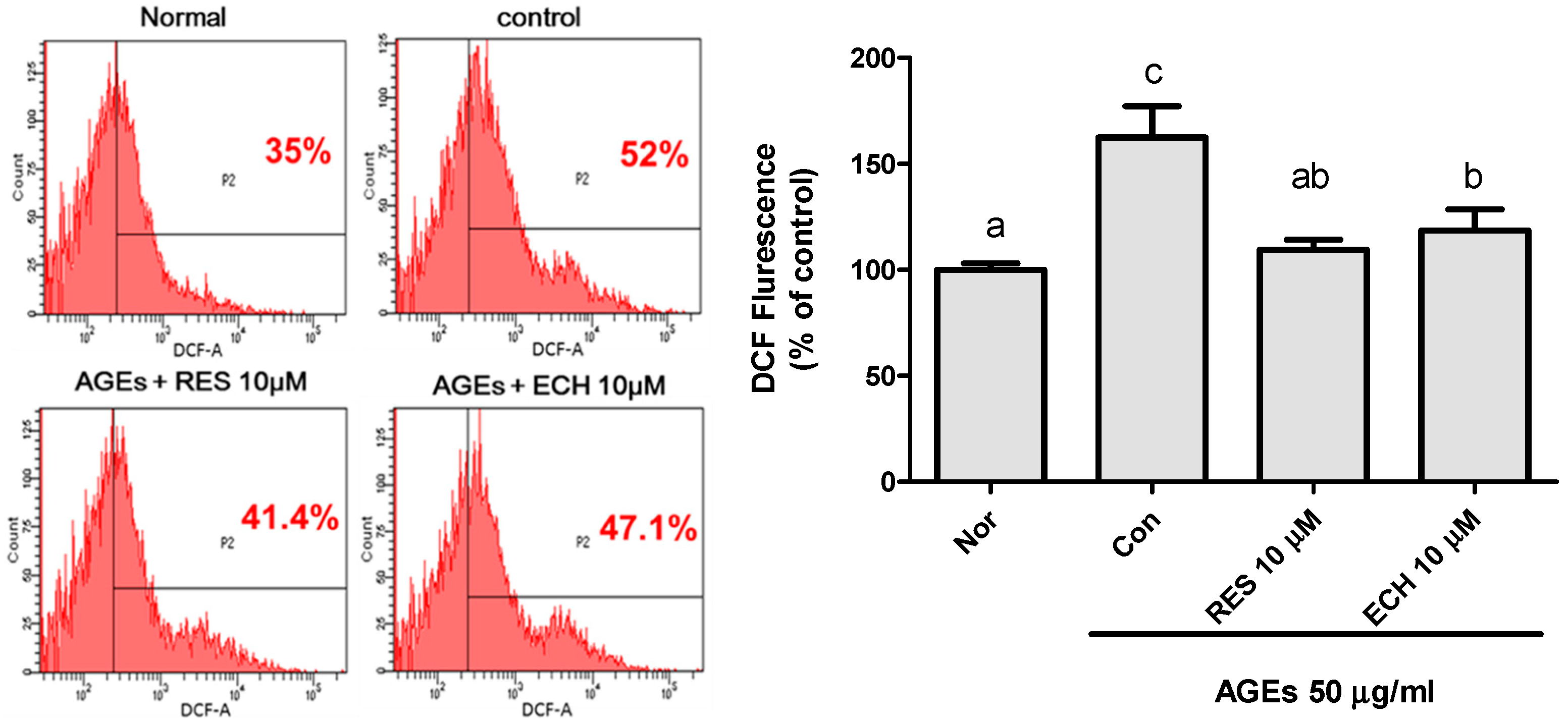
3.1.4. Effect of ECH on H2O2 Production in AGE-Stimulated LC-540 Leydig Cells
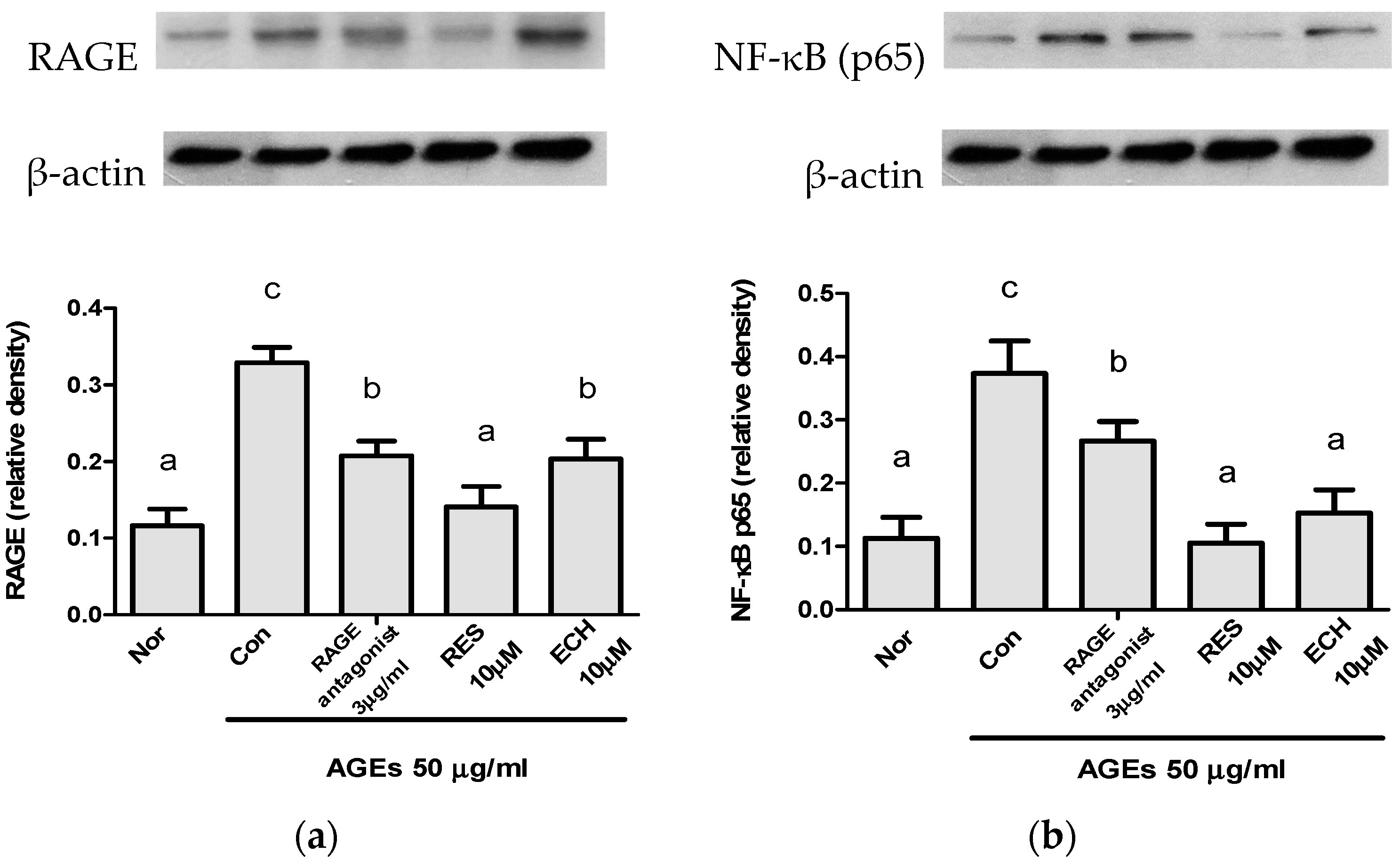
3.1.5. Effect of ECH on RAGE and NF-κB Protein Expression Levels in AGE-Stimulated LC-540 Leydig Cells
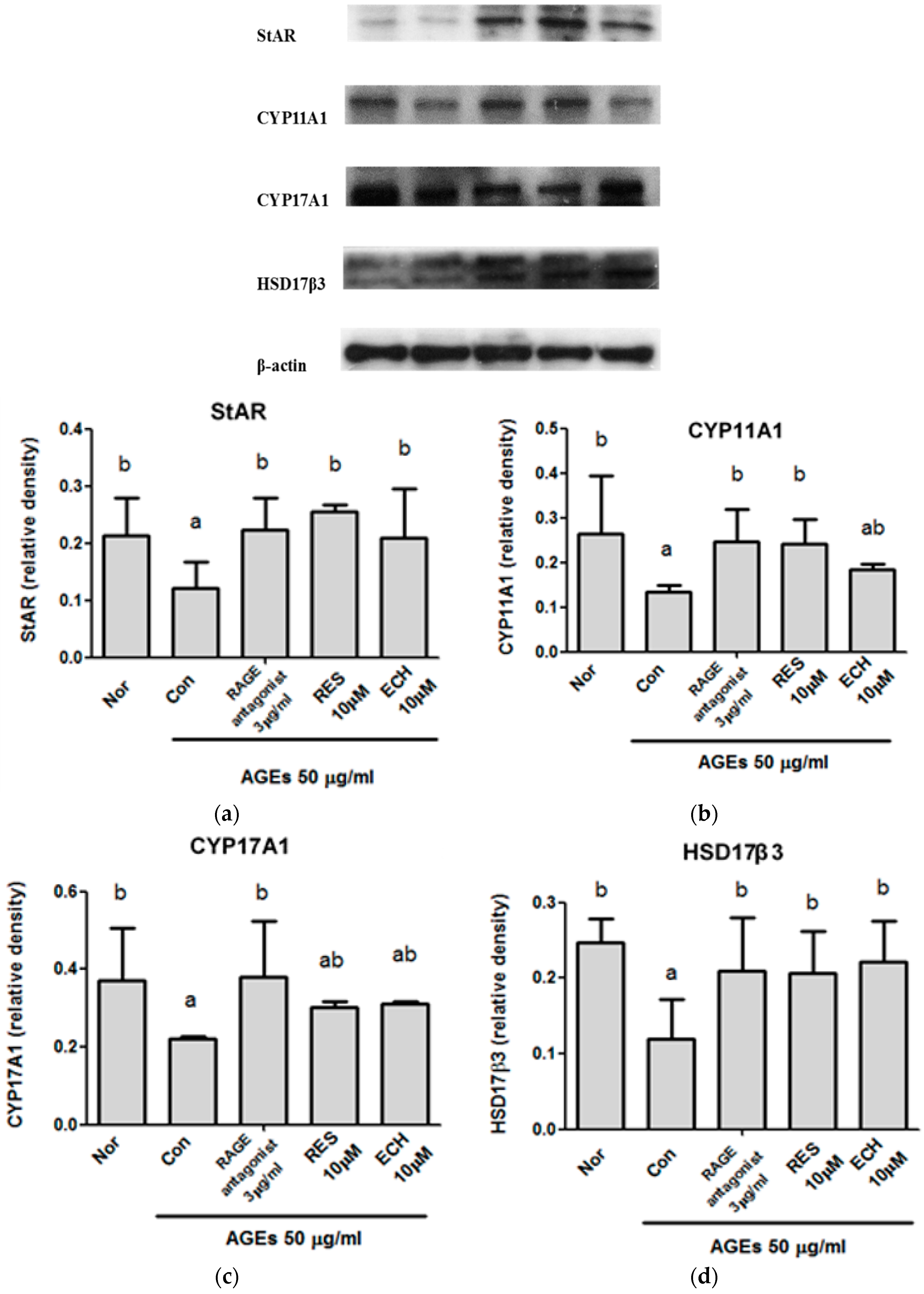
3.1.6. Effect of ECH on the Testosterone Synthesis pathway in AGE-Stimulated LC-540 Leydig cells.
3.2. In Vivo Analysis
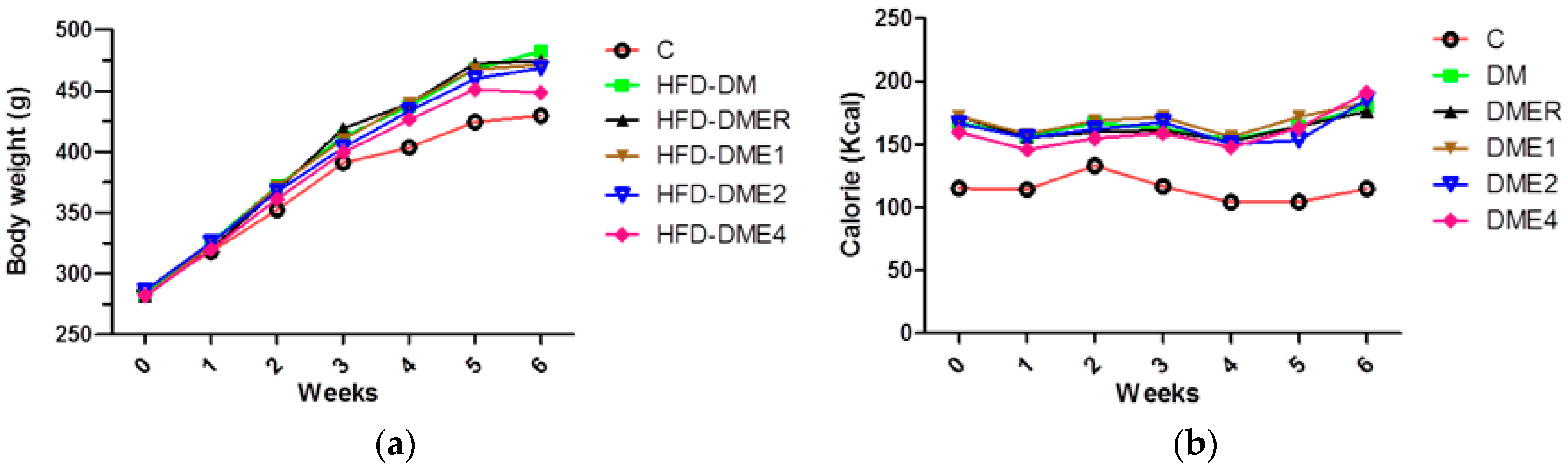
3.2.1. Effects of CTE on Body Weight and Calorie Intake
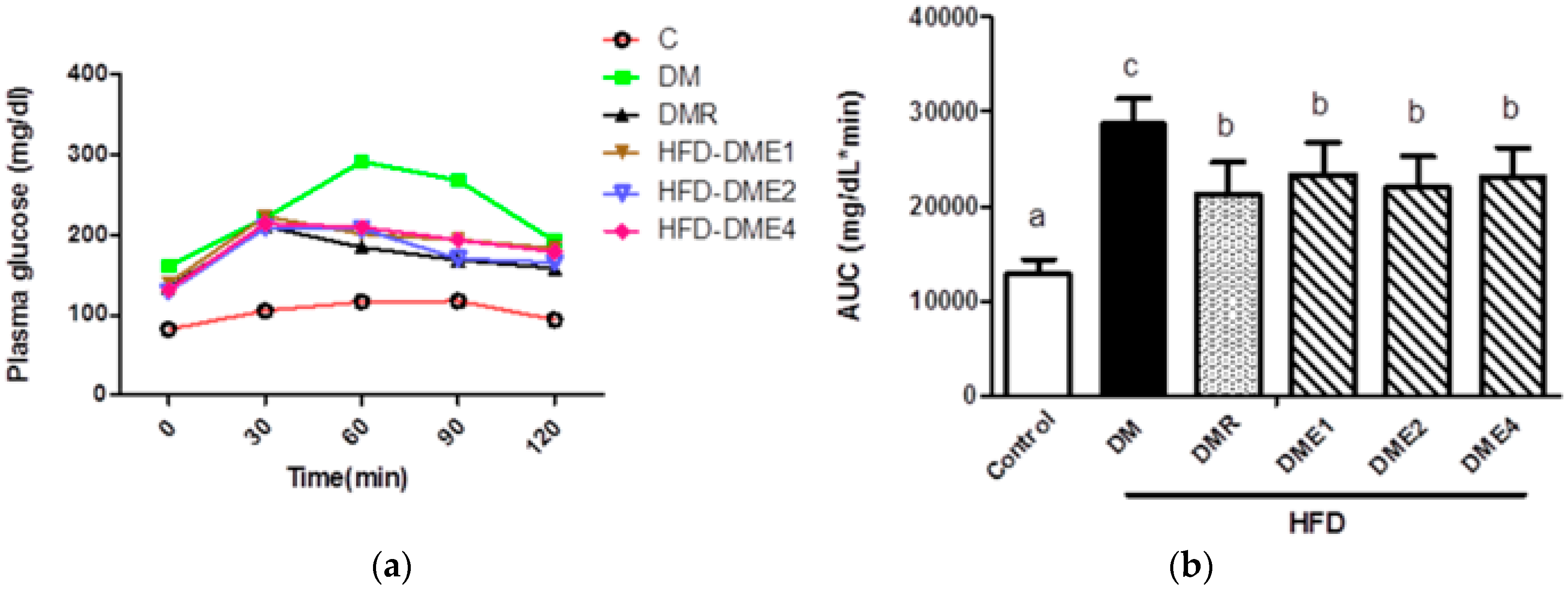
3.2.2. Oral Glucose Tolerance Test (OGTT) to Determine the Successful Induction of Diabetes
3.2.3. Total Plasma Glucose, Cholesterol, and Triglyceride Contents
3.2.4. Plasma Insulin Levels, Plasma Leptin Level, and Homeostasis Model Assessment–Insulin Resistance (HOMA-IR) Values
3.2.5. Effect of CTE on Plasma LH and Testosterone Levels in Diabetic Rats
3.2.6. Effect of CTE on the Sperm Parameters of Diabetic Rats
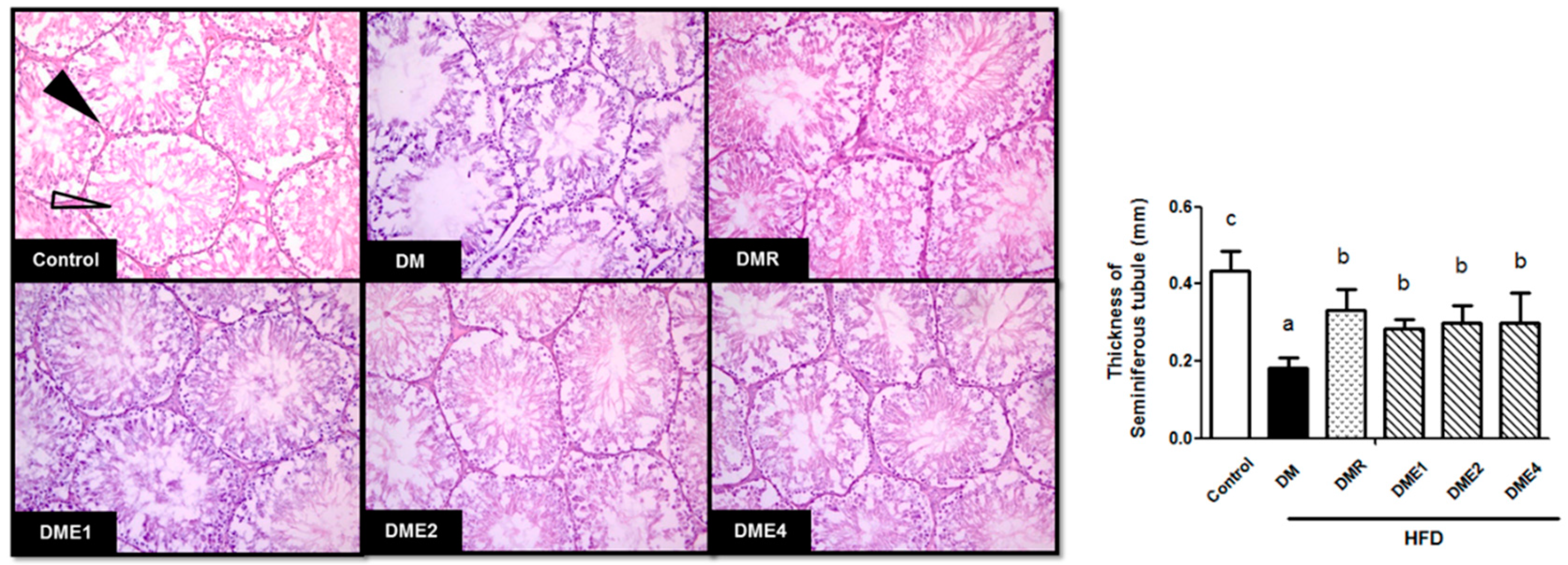
3.2.7. Effect of CTE on the Morphology of Seminiferous Tubules in Diabetic Rats
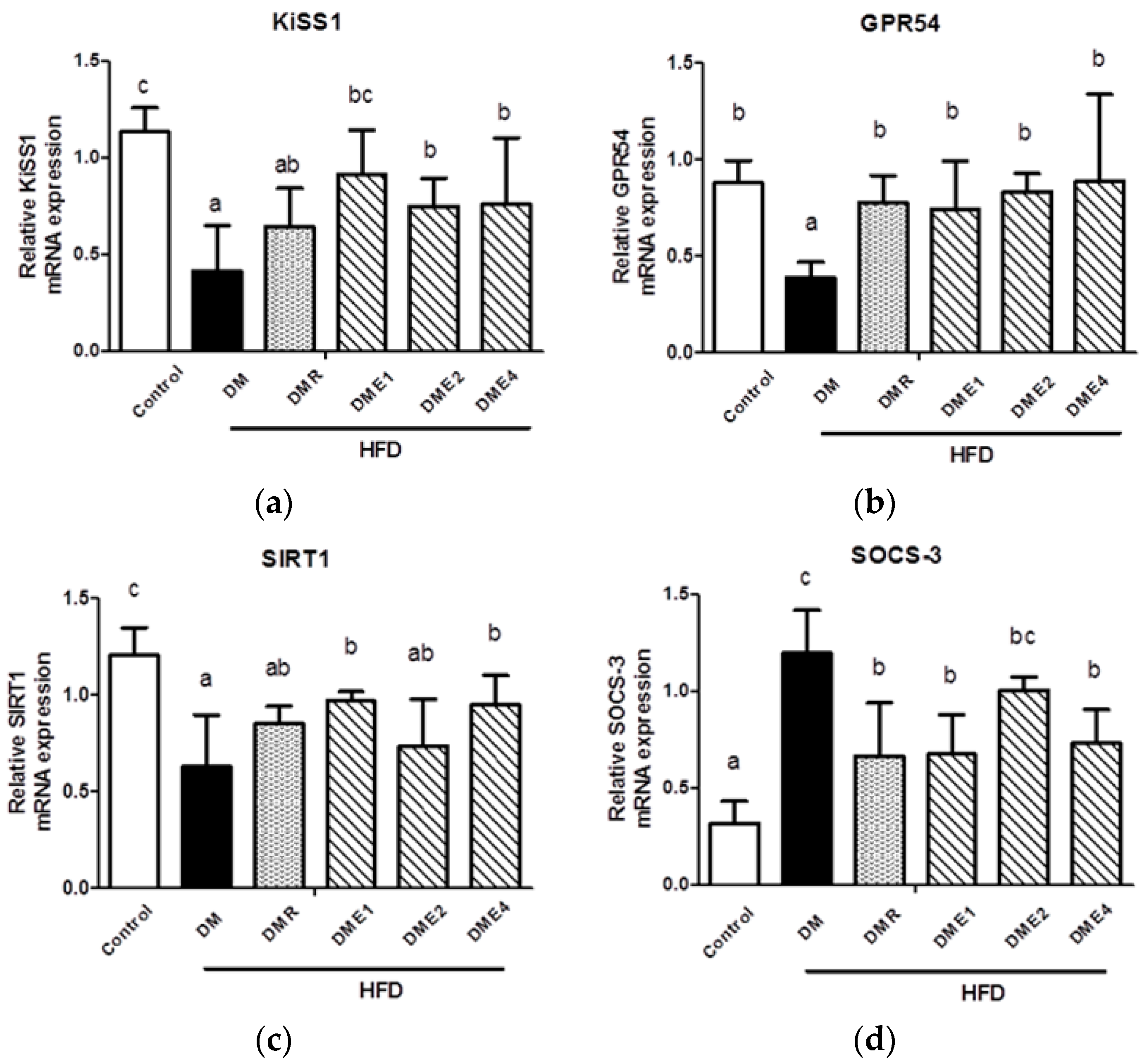
3.2.8. Effect of CTE on KiSS1, GPR54, SOCS-3, and SIRT1 mRNAs in the Hypothalamus of Diabetic Rats
3.2.9. Effect of CTE on Antioxidant Enzymes in Plasma and Testis of Diabetic Rats
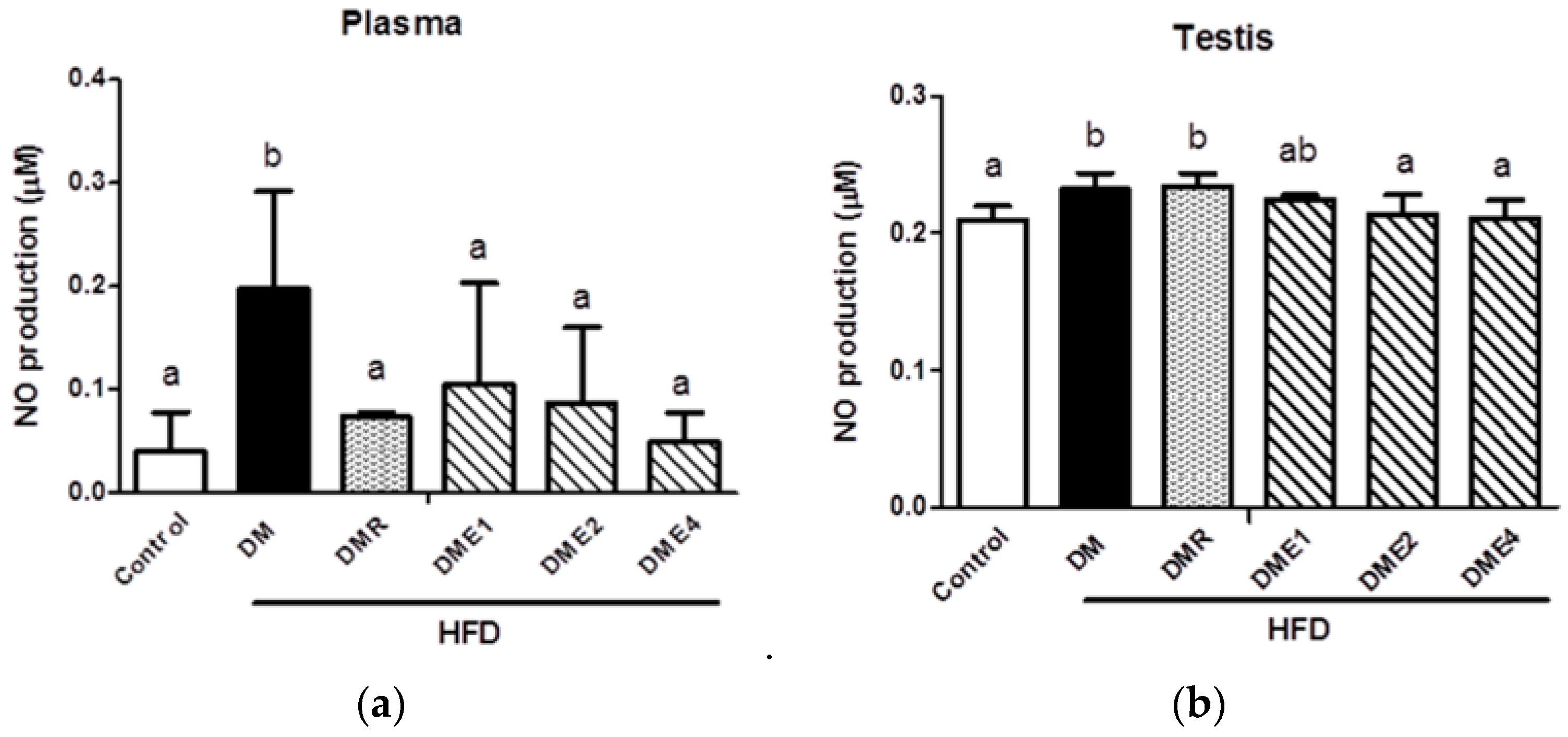
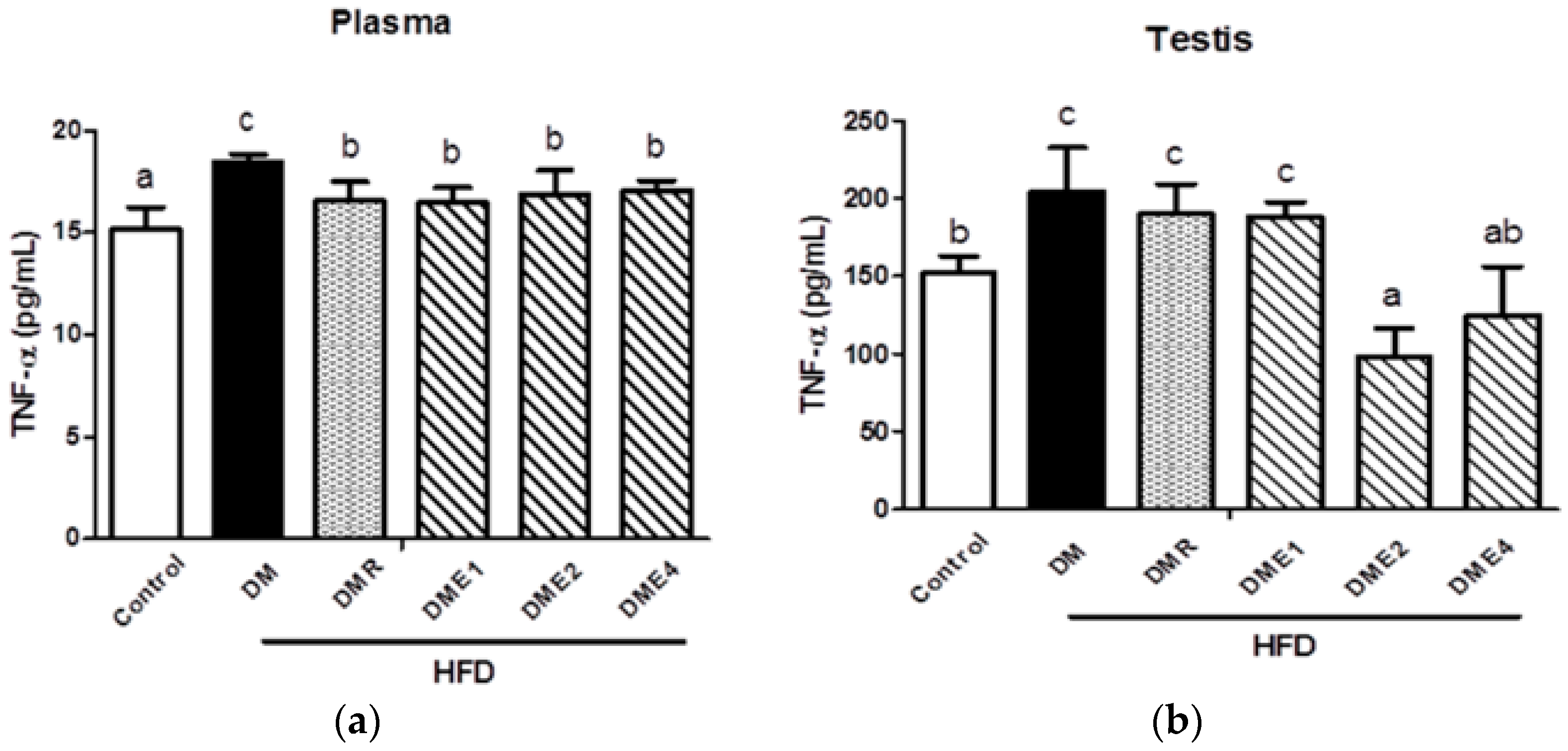
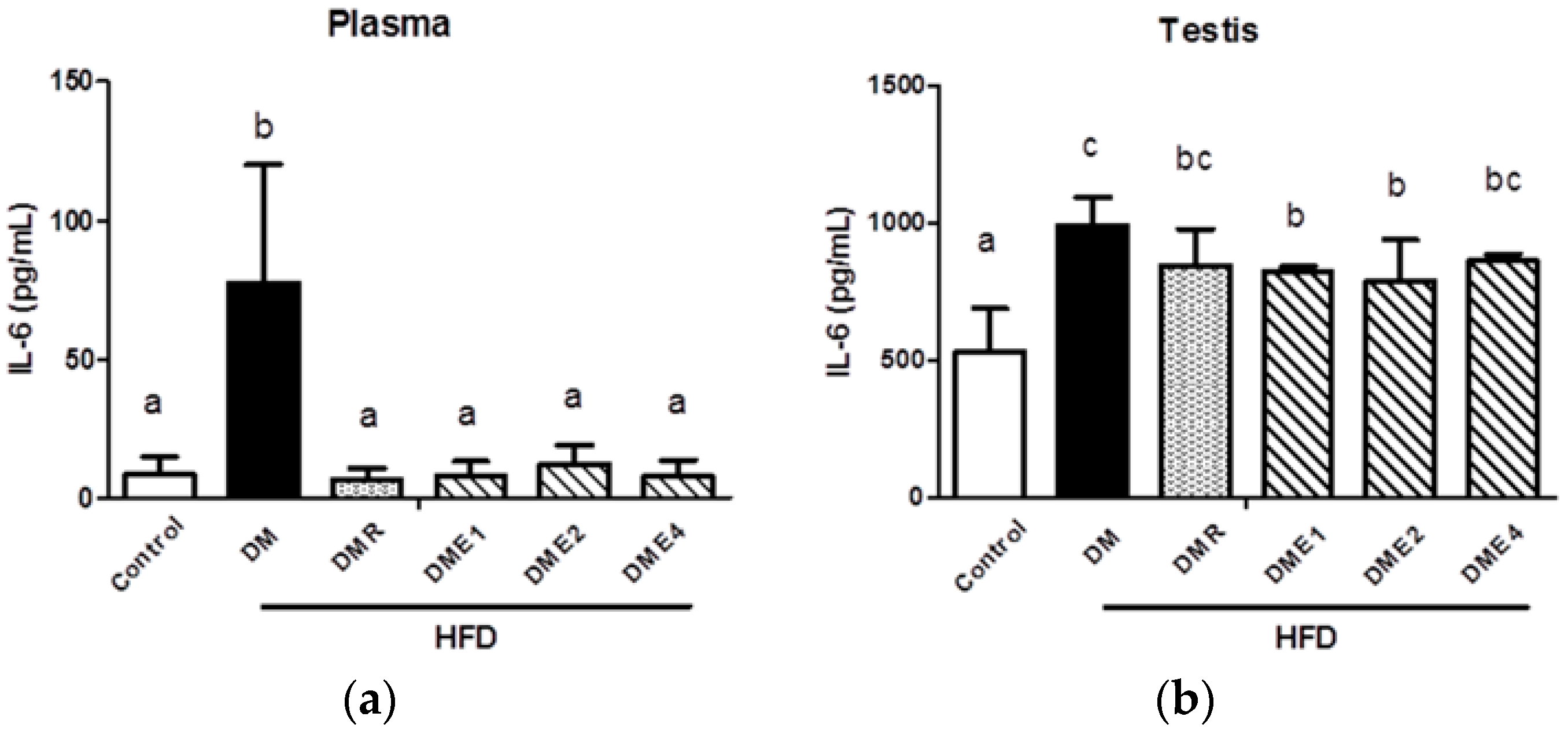
3.2.10. Effects of CTE on Oxidative Stress and Inflammation in the Plasma and Testis of Diabetic Rats
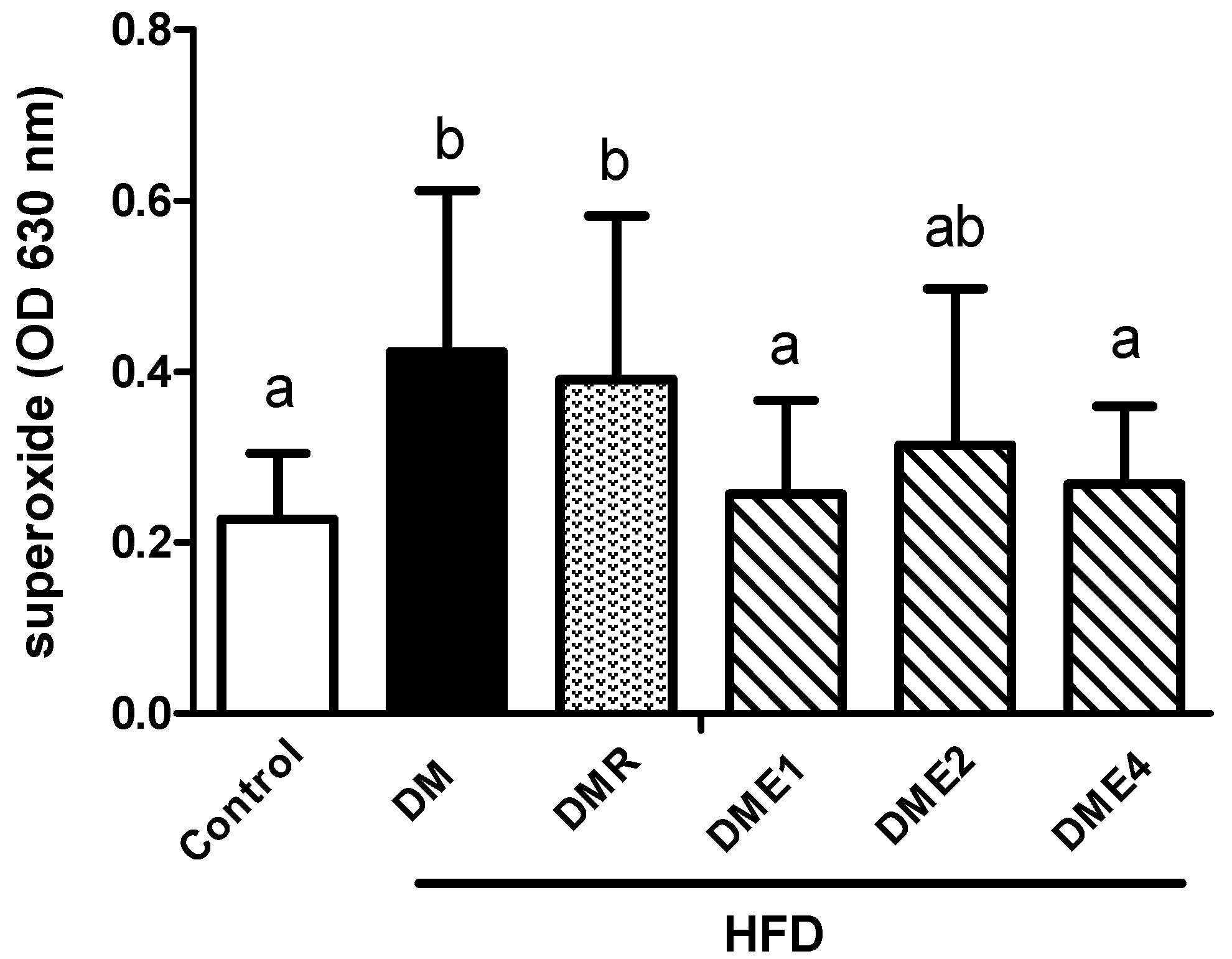
3.2.11. Effects of CTE on Oxidative Stress and Inflammation in Spermatozoa of Diabetic Rats Induced by High-Fat Diet
3.2.12. Effects of CTE on Lipid Peroxidation in Spermatozoa of Diabetic Rats Induced by High-Fat Diet
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Global Reports on Diabetes. 2017. Available online: http://www.who.int/news-room/fact-sheets/detail/diabetes (accessed on 25 August 2018).
- Haynes, N.E.; Corbett, W.L.; Bizzarro, F.T.; Guertin, K.R.; Hilliard, D.W.; Holland, G.W.; Kester, R.F.; Mahaney, P.E.; Qi, L.; Spence, C.L.; et al. Discovery, structure-activity relationships, pharmacokinetics, and efficacy of glucokinase activator (2R)-3-cyclopentyl-2-(4-methanesulfonylphenyl)-N-thiazol-2-yl-propionamide (RO0281675). J. Med. Chem. 2010, 53, 3618–3625. [Google Scholar] [CrossRef] [PubMed]
- Agbaje, I.M.; Rogers, D.A.; McVicar, C.M.; McClure, N.; Atkinson, A.B.; Mallidis, C.; Lewis, S.E. Insulin dependant diabetes mellitus: Implications for male reproductive function. Hum. Reprod. 2007, 22, 1871–1877. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fouchécourt, S.; Metayer, S.; Locatelli, A.; Dacheux, F.; Dacheux, J.L. Stallion epididymal fluid proteome: Qualitative and quantitative characterization; Secretion and dynamic changes of major proteins1. Biol. Reprod. 2000, 62, 1790–1803. [Google Scholar] [CrossRef] [PubMed]
- Vernet, P.; Aitken, R.J.; Drevet, J.R. Antioxidant strategies in the epididymis. Mol. Cell. Endocrinol. 2004, 216, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Szkudelski, T. Streptozotocin–nicotinamide-induced diabetes in the rat. Characteristics of the experimental model. Exp. Biol. Med. 2012, 237, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.; Jiang, X.; Wu, L.; Wang, Z.; Huang, L. Chemical and genetic discrimination of Cistanches herba based on UPLC-QTOF/MS and DNA barcoding. PLoS ONE 2014, 9, e98061. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, J.; Aipire, A.; Gao, L.; Huo, S.; Luo, J.; Zhang, F. Phenylethanoid glycosides from Cistanche tubulosa inhibits the growth of B16-F10 Cells both in vitro and in vivo by induction of apoptosis via mitochondria-dependent Pathway. J. Cancer 2016, 7, 1877–1887. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Gao, J.; Cai, D. Neurotrophic and neurorescue effects of echinacoside in the subacute MPTP mouse model of parkinson’s disease. Brain Res. 2010, 1346, 224–236. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Matsuda, H.; Morikawa, T.; Xie, H.; Nakamura, S.; Muraoka, O. Phenylethanoid oligoglycosides and acylated oligosugars with vasorelaxant activity from Cistanche tubulosa. Bioorg. Med. Chem. 2006, 14, 7468–7475. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Wang, J.; Li, X.; Zhang, X. Echinacoside and Cistanche tubulosa (Schenk) R. wight ameliorate bisphenol A-induced testicular and sperm damage in rats through gonad axis regulated steroidogenic enzymes. J. Ethnopharmacol. 2016, 193, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Placer, Z.A.; Cushman, L.L.; Johnon, B.C. Estimation of product of lipid peroxidation (malonyl dialdehyde) in biochemical systems. Anal. Biochem. 1966, 16, 359–364. [Google Scholar] [CrossRef]
- Younglai, E.V.; Holt, D.; Brown, P.; Jurisicova, A.; Casper, R.F. Sperm swim-up techniques and DNA fragmentation. Hum. Reprod. 2001, 16, 1950–1953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yokoi, K.; Uthus, E.O.; Nielsen, F.H. Nickel deficiency diminishes sperm quantity and movement in rats. Biol. Trace Elem. Res. 2003, 93, 141–153. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813. [Google Scholar] [CrossRef] [PubMed]
- Shimoda, H.; Tanaka, J.; Takahara, Y.; Takemoto, K.; Shan, S.J.; Su, M.H. The Hypocholesterolemic Effects of Cistanche tubulosa Extract, a Chinese Traditional Crude Medicine, in Mice. Am. J. Chin. Med. 2009, 37, 1125–1138. [Google Scholar] [CrossRef] [PubMed]
- Baynes, J.W.; Thorpe, S.R. Role of oxidative stress in diabetic complications: A new perspective on an old paradigm. Diabetes 1999, 48, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Akkus, I.; Kalak, S.; Vural, H.; Caglayan, O.; Menekse, E.; Can, G.; Durmus, B. Leukocyte lipid peroxidation, superoxide dismutase, glutathione peroxidase and serum and leukocyte vitamin C levels of patients with type II diabetes mellitus. Clin. Chim. Acta 1996, 244, 221–227. [Google Scholar] [CrossRef]
- Matough, F.A.; Budin, S.B.; Hamid, Z.A.; Alwahaibi, N.; Mohamed, J. The role of oxidative stress and antioxidants in diabetic complications. Sultan. Qaboos Univ. Med. J. 2012, 12, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.; Yang, X.; Huang, L. Cistanches herba: A neuropharmacology review. Front. Pharmacol. 2016, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Xiong, W.T.; Gu, L.; Wang, C.; Sun, H.X.; Liu, X. Anti-hyperglycemic and hypolipidemic effects of Cistanche tubulosa in type 2 diabetic db/db mice. J. Ethnopharmacol. 2013, 150, 935–945. [Google Scholar] [CrossRef] [PubMed]
- Chodavarapu, H.; Grobe, N.; Somineni, H.K.; Salem, E.S.B.; Madhu, M.; Elased, K.M. Rosiglitazone Treatment of Type 2 Diabetic db/db Mice Attenuates Urinary Albumin and Angiotensin Converting Enzyme 2 Excretion. PLoS ONE 2013, 8, e62833. [Google Scholar] [CrossRef] [PubMed]
- Bhat, K.P.L.; Kosmeder, J.W.; Pezzuto, J.M. Biological effects of resveratrol. Antioxid. Redox Signal. 2001, 3, 1041–1064. [Google Scholar] [CrossRef] [PubMed]
- Aziz, M.H.; Kumar, R.; Ahmad, N. Cancer chemoprevention by resveratrol: In vitro and in vivo studies and the underlying mechanisms (review). Int. J. Oncol. 2003, 23, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Vlassara, H.; Uribarri, J. Advanced glycation end products (AGE) and diabetes: Cause, effect, or both? Curr. Diab. Rep. 2014, 14, 453. [Google Scholar] [CrossRef] [PubMed]
- Nowotny, K.; Jung, T.; Höhn, A.; Weber, D.; Grune, T. Advanced glycation end products and oxidative stress in type 2 diabetes mellitus. Biomolecules 2015, 5, 194–222. [Google Scholar] [CrossRef] [PubMed]
- Tornatore, L.; Thotakura, A.K.; Bennett, J.; Moretti, M.; Franzoso, G. The nuclear factor kappa B signaling pathway: Integrating metabolism with inflammation. Trends Cell Biol. 2012, 22, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Olokoba, A.B.; Obateru, O.A.; Olokoba, L.B. Type 2 diabetes mellitus: A review of current trends. Oman Med. J. 2012, 27, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Abrams, J.J.; Ginsberg, H.; Grundy, S.M. Metabolism of cholesterol and plasma triglycerides in nonketotic diabetes mellitus. Diabetes 1982, 31, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Al Sheikh, M.H. The determinants of leptin levels in diabetic and nondiabetic saudi males. Int. J. Endocrinol. 2017, 2017, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Jain, G.; Jangir, R. Modulation of diabetes-mellitus-induced male reproductive dysfunctions in experimental animal models with medicinal plants. Pharmacogn. Rev. 2014, 8, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Dudek, M.; Kołodziejski, P.A.; Pruszyńska-Oszmałek, E.; Sassek, M.; Ziarniak, K.; Nowak, K.W.; Sliwowska, J.H. Effects of high-fat diet-induced obesity and diabetes on Kiss1 and GPR54 expression in the hypothalamic-pituitary-gonadal (HPG) axis and peripheral organs (fat, pancreas and liver) in male rats. Neuropeptides 2016, 56, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Torisu, T.; Sato, N.; Yoshiga, D.; Kobayashi, T.; Yoshioka, T.; Mori, H.; Iida, M.; Yoshimura, A. The dual function of hepatic SOCS3 in insulin resistance in vivo. Genes Cells 2007, 12, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Sindhu, R.K.; Koo, J.R.; Roberts, C.K.; Vaziri, N.D. Dysregulation of hepatic superoxide dismutase, catalase and glutathione peroxidase in diabetes: Response to insulin and antioxidant therapies. Clin. Exp. Hypertens. 2004, 26, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Noberasco, G.; Odetti, P.; Boeri, D.; Maiello, M.; Adezati, L. Malondialdehyde (MDA) level in diabetic subjects. Relationship with blood glucose and glycosylated hemoglobin. Biomed. Pharmacother. 1991, 45, 193–196. [Google Scholar] [CrossRef]
| Gene Bank Accession Number | Expected Size (bp) |
|---|---|
| Kiss1 sense (5′-ATGATCTCGCTGGCTTCTTG-3′) Kiss1 antisense (5′-AGGCTTGCTCTCTGCATACC-3′) | 174 |
| Gpr54 sense (5′-CTGGGAGACTTCATGTGCAA-3′) Gpr54 antisense (5′-GAACCCACCCAGATGCTAAG-3′) | 182 |
| Socs-3 sense (5′-GTC GGGGACCAAGAACCTAC-3′) Socs-3 antisense (5′-GGCTGCTCCTGAACCTCAAA-3′) | 192 |
| Sirt1 sense (5′-ATTTATGCTCGCCTTGCTGT-3′) Sirt1 antisense (5′-GATCCTTTGGATTCCTGCAA-3′) | 243 |
| Internal Control Gene | |
| Rp-s11 sense (5′-GGCGGACATTCAGACGGAG-3′) Rp-s11 antisense (5′-CGTCACAACACCAGACAGGA -3′) | 232 |
| mg/dL | HFD | |||||
|---|---|---|---|---|---|---|
| C | DM | DMR | DME1 | DME2 | DME4 | |
| Fasting glucose | 82.2 ± 13.68 a | 160.82 ± 19.71 c | 134.37 ± 17.49 b | 141.68 ± 16.41 b | 129.23 ± 18.74 b | 131.04 ± 15.68 b |
| Total cholesterol | 101.30 ± 27.92 b | 104.26 ± 20.92 b | 106.90 ± 19.26 b | 101.85 ± 19.64 b | 104.51 ± 19.78 b | 81.08 ± 11.58 a |
| Triglyceride | 60.82 ± 15.88 a | 87.07 ± 22.42 b | 62.77 ± 16.05 a | 78.46 ± 29.85 ab | 73.44 ± 17.88 ab | 56.29 ± 14.74 a |
| ng/mL | HFD | |||||
|---|---|---|---|---|---|---|
| C | DM | DMR | DME1 | DME2 | DME4 | |
| Insulin | 3.33 ± 1.83 a | 5.41 ± 2.28 b | 3.57 ± 2.15 a | 3.17 ± 1.74 a | 2.34 ± 2.03 a | 2.28 ± 1.43 a |
| Leptin | 2.97 ± 1.51 a | 7.36 ± 1.76 b | 6.38 ± 2.05 b | 6.26 ± 2.68 b | 3.66 ± 3.05 a | 3.57 ± 1.71 a |
| HOMA-IR (µg.mmol/L2) | 1.38 ± 0.70 a | 4.43 ± 1.87 b | 2.37 ± 1.21 a | 2.45 ± 1.79 a | 1.62 ± 1.57 a | 1.59 ± 1.04 a |
| HFD | ||||||
|---|---|---|---|---|---|---|
| C | DM | DMR | DME1 | DME2 | DME4 | |
| LH (IU/L) | 6.20 ± 0.09 b | 6.09 ± 0.01 a | 6.17 ± 0.06 b | 6.18 ± 0.02 b | 6.16 ± 0.02 b | 6.22 ± 0.09 b |
| Testosterone (ng/dL) | 3.84 ± 1.04 d | 1.30 ± 0.33 a | 1.95 ± 0.69 b | 3.51 ± 0.43 c | 2.43 ± 0.24 bc | 2.64 ± 0.68 bc |
| Sperm | HFD | |||||
|---|---|---|---|---|---|---|
| C | DM | DMR | DME1 | DME2 | DME4 | |
| Total count (*105) | 21.50 ± 4.34 b | 16.60 ± 1.50 a | 31.30 ± 4.96 c | 32.50 ± 8.02 c | 38.80 ± 7.85 d | 23.10 ± 7.69 b |
| Motility (% total) | 21.70 ± 12.43 b | 11.74 ± 12.03 a | 15.83 ± 7.09 ab | 20.54 ± 7.83 b | 14.22 ± 1.87 b | 49.60 ± 3.88 c |
| Abnormal morphology (% total) | 5.00 ± 4.28 a | 16.57 ± 18.59 b | 3.28 ± 3.90 a | 4.37 ± 3.21 a | 3.09 ± 3.91 a | 5.76 ± 7.23 a |
| (Units/mg Protein) | ||||||
|---|---|---|---|---|---|---|
| HFD | ||||||
| C | DM | DMR | DME1 | DME2 | DME4 | |
| SOD | 0.60 ± 0.13 b | 0.40 ± 0.05 a | 0.65 ± 0.15 b | 0.64 ± 0.24 b | 0.63 ± 0.25 b | 0.55 ± 0.03 ab |
| Catalase | 81.58 ± 32.37 b | 44.07 ± 6.79 a | 60.05 ± 9.92 ab | 68.24 ± 27.80 b | 70.53 ± 33.79 b | 58.29 ± 8.50 ab |
| GPx | 1974.36 ± 609.87 c | 606.41 ± 498.49 a | 811.08 ± 359.72 ab | 918.66 ± 228.38 ab | 948.06 ± 501.99 ab | 1065.95 ± 168.89 b |
| (Units/mg Protein) | ||||||
|---|---|---|---|---|---|---|
| HFD | ||||||
| C | DM | DMR | DME1 | DME2 | DME4 | |
| SOD | 17.29 ± 5.13 b | 13.74 ± 2.01 a | 13.55 ± 3.08 a | 18.26 ± 4.27 b | 16.30 ± 1.18 ab | 16.16 ± 2.12 ab |
| Catalase | 416.69 ± 80.41 b | 213.39 ± 126.59 a | 205.06 ± 114.58 a | 393.95 ± 202.83 ab | 426.55 ± 76.79 b | 421.61 ±144.60 b |
| nmol/mL | HFD | |||||
|---|---|---|---|---|---|---|
| C | DM | DMR | DME1 | DME2 | DME4 | |
| Plasma MDA | 10.15 ± 1.20 a | 16.87 ± 2.42 c | 13.38 ± 2.12 b | 9.90 ± 2.09 a | 10.66 ± 1.94 a | 9.95 ± 1.55 a |
| Testis MDA | 10.16 ± 0.79 a | 13.31 ± 0.71 b | 11.64 ± 2.70 a | 11.75 ± 0.50 a | 11.54 ± 1.71 a | 11.30 ± 2.00 a |
| Sperm MDA | 1.39 ± 0.73 a | 2.12 ± 0.28 b | 1.75 ± 0.41 ab | 1.39 ± 0.78 a | 1.47 ± 0.50 a | 1.42 ± 0.82 a |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kong, Z.-L.; Johnson, A.; Ko, F.-C.; He, J.-L.; Cheng, S.-C. Effect of Cistanche Tubulosa Extracts on Male Reproductive Function in Streptozotocin–Nicotinamide-Induced Diabetic Rats. Nutrients 2018, 10, 1562. https://doi.org/10.3390/nu10101562
Kong Z-L, Johnson A, Ko F-C, He J-L, Cheng S-C. Effect of Cistanche Tubulosa Extracts on Male Reproductive Function in Streptozotocin–Nicotinamide-Induced Diabetic Rats. Nutrients. 2018; 10(10):1562. https://doi.org/10.3390/nu10101562
Chicago/Turabian StyleKong, Zwe-Ling, Athira Johnson, Fan-Chi Ko, Jia-Ling He, and Shu-Chun Cheng. 2018. "Effect of Cistanche Tubulosa Extracts on Male Reproductive Function in Streptozotocin–Nicotinamide-Induced Diabetic Rats" Nutrients 10, no. 10: 1562. https://doi.org/10.3390/nu10101562
APA StyleKong, Z.-L., Johnson, A., Ko, F.-C., He, J.-L., & Cheng, S.-C. (2018). Effect of Cistanche Tubulosa Extracts on Male Reproductive Function in Streptozotocin–Nicotinamide-Induced Diabetic Rats. Nutrients, 10(10), 1562. https://doi.org/10.3390/nu10101562





