Dietary Intake of Vitamin D in the Czech Population: A Comparison with Dietary Reference Values, Main Food Sources Identified by a Total Diet Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Data on Food Consumption
2.2. Data on Vitamin D Content in Foods
2.3. Analytical Measurement of Vitamin D in Foods
2.4. Estimation of Vitamin D Usual Dietary Intake in the Czech Population
2.5. Assessment of Vitamin D Dietary Intake
2.6. Limitation of Study Results
3. Results
3.1. Usual Dietary Intake and Its Distribution in Ten Czech Population Groups
3.2. Food Sources of Vitamin D Intake in the Czech Population Groups
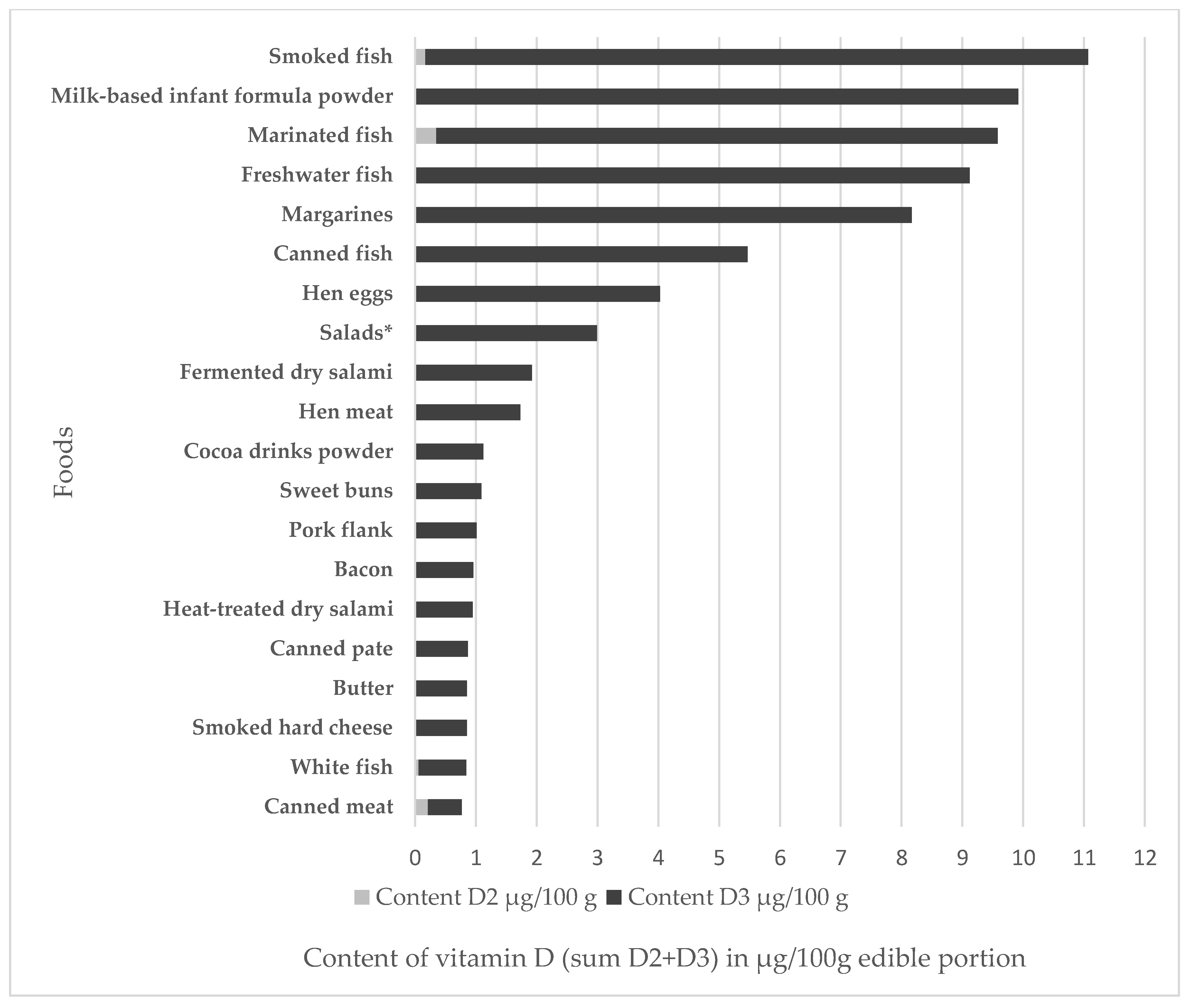
3.3. Vitamin D Content in Foods and Food Groups
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Spiro, A.; Buttriss, L. Vitamin D: An overview of vitamin D status and intake in Europe. Nutr. Bull. 2014, 39, 322–350. [Google Scholar] [CrossRef] [PubMed]
- Pludowski, P.; Holick, M.F.; Pilz, F.; Wagner, C.L.; Hollis, B.W.; Grant, W.B.; Shoenfeld, Y.; Lerchbaum, E.; Llewellyn, D.J.; Kienreich, K.; Soni, M. Vitamin D effects on musculoskeletal health, immunity, autoimmunity, cardiovascular disease, cancer, fertility, pregnancy, dementia and mortality—A review of recent evidence. Autoimmun. Rev. 2013, 12, 976–989. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Scientific opinion on dietary reference values for vitamin D. EFSA J. 2016, 14, 1–145. [Google Scholar] [CrossRef]
- Finglas, P.M.; Roe, M.A.; Pinchen, H.M.; Berry, R.; Church, S.M.; Dodhia, S.K.; Farron-Wilson, M.; Swan, G. McCance and Widdowson’s The Composition of Foods, Seventh Summary Edition; Royal Society of Chemistry: Cambridge, UK, 2015; p. 630, ISBN 978-1-84973-636-7. [Google Scholar]
- Mattila, P.H.; Piironen, V.I.; Uusi-Rauva, E.J.; Koivistoinen, P.E. Vitamin D Contents in Edible Mushrooms. J. Agric. Food Chem. 1994, 42, 2449–2453. [Google Scholar] [CrossRef]
- Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D; The National Academies Press: Washington, DC, USA, 2011; pp. 1–1132. ISBN 978-0-309-16394-1. [Google Scholar]
- Heaney, R.; Garland, C.; Baggerly, C.; French, C.; Gorham, E. Letter to Veugelers, P.J. and Ekwaru, J.P., A Statistical Error in the Estimation of the Recommended Dietary Allowance for Vitamin D. Nutrients 2014, 6, 4472–4475. [Google Scholar] [CrossRef]
- D-A-CH Referenzwerte für die Nährstoffzufuhr, 2nd ed.; Deutsche Gesellschaft für Ernährung, Österreichische Gesellschaft für Ernährung, Schweizerische Gesellschaft für Ernährung: Bonn, Germany, 2015; ISBN 978-3-86528-148-7.
- European Food Safety Authority/Food and Agriculture Organization of the United Nations/World Health Organisation. Towards a harmonised Total Diet Study approach: A guidance document. EFSA J. 2011, 9, 1–66. [Google Scholar] [CrossRef]
- Brussaard, J.H.; Löwik, M.R.H.; Steingrimsdottir, L.; Møller, A.; Kearney, J.; De Henauw, S.; Becker, W. A European food consumption survey method—Conclusions and recommendations. Eur. J. Clin. Nutr. 2002, 56, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Black, A.E. Critical evaluation of energy intake using the Goldberg cut-off for energy intake: Basal metabolic rate. A practical guide to its calculation, use and limitations. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 1119–1130. [Google Scholar] [CrossRef] [PubMed]
- European Food safety Authority. The EFSA Comprehensive European Food Consumption Database. Available online: https://www.efsa.europa.eu/en/food-consumption/comprehensive-database (accessed on 6 September 2018).
- Merten, C.; Ferrari, P.; Bakker, M.; Boss, A.; Hearty, A.; Leclercq, C.; Lindtner, O.; Tlustos, C.; Verger, P.; Volatier, J.L.; Arcella, D. Methodological characteristics of the national dietary surveys carried out in the European Union as included in the European Food Safety Authority (EFSA) Comprehensive European Food Consumption Database. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2011, 28, 975–995. [Google Scholar] [CrossRef] [PubMed]
- Ruprich, J.; Dofkova, M.; Rehurkova, I.; Slamenikova, E.; Resova, D. Individual food consumption - the national study SISP04. CHFCH NIPH in Prague, 2006. Available online: http://czvp.szu.cz/spotrebapotravin.htm (accessed on 6 June 2018).
- Dodd, K.W.; Guenther, P.M.; Freedman, L.S.; Subar, A.F.; Kipnis, V.; Midthune, D.; Tooze, J.A.; Krebs-Smith, S.M. Statistical methods for estimating the usual intake of nutrients and foods: A review of the theory. J. Am. Diet. Assoc. 2006, 106, 1640–1650. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Guidance on the EU Menu methodology. EFSA J. 2014, 12, 1–77. [Google Scholar] [CrossRef]
- Environmental Health Monitoring System in the Czech Republic. Subsystem IV. Contaminants in Food Chains and Their Impact on Human Health in 2015. Expert Report, 2015. National Institute of Public Health: Prague: 2016. Available online: http://www.szu.cz/uploads/documents/chzp/odborne_zpravy/OZ_15/Odborna_dieta_2015.pdf (accessed on 6 June 2018).
- Dofkova, M.; Karpisková, R.; Ostry, V.; Ruprich, J.; Rehurkova, I. Contaminants in food chains and their impact on human health, dietary exposure. In Environmental Health Monitoring System in the Czech Republic; Summary report 2016; Puklová, V., Ed.; National Institute of Public Health: Prague, Czech Republic, 2017; pp. 49–60. ISBN 978-80-7071-365-5. [Google Scholar]
- Phillips, K.M.; Byrdwell, W.C.; Exler, J.; Harnly, J.M.; Holde, J.M.; Holick, M.F.; Hollis, B.W.; Horst, R.L.; Lemar, L.E.; Patterson, K.Y.; Tarrago-Trani, M.T.; Wolf, W.R. Development and validation of control materials for the measurement of vitamin D3 in selected US foods. J. Food Comp. 2008, 21, 527–534. [Google Scholar] [CrossRef]
- MCRA (2016). MCRA 8.2 Reference Manual. Report Dec 2016. WUR/Biometris, FERA and RIVM. Available online: https://mcra.rivm.nl (accessed on 6 June 2018).
- Van Rossum, C.T.M.; Fransen, H.P.; Verkaik-Kloosterman, J.; Buurma-Rethans, E.J.M.; Ocké, M.C. Dutch National Food Consumption Survey 2007-2010: Diet of Children and Adults Aged 7 to 69 Years. RIVM National Institute for Public Health and the Environment 2011, 148 pp. Available online: https://www.rivm.nl/dsresource?objectid=3f528864-36cb-400b-8411-a47db4f99df4&type=org&disposition=inline (accessed on 6 June 2018).
- Czech Statistical Office. Food Consumption—2014. Available online: https://www.czso.cz/csu/czso/food-consumption-2014 (accessed on 14 September 2018).
- European Food Safety Authority. The Food Classification and Description System FoodEx2 (Revision 2). Available online: https://efsa.onlinelibrary.wiley.com/doi/epdf/10.2903/sp.efsa.2015.EN-804 (accessed on 12 September 2018).
- European Food Safety Authority. Scientific Opinion on the Tolerable Upper Intake Level of vitamin D. EFSA J. 2012, 10, 1–45. [Google Scholar] [CrossRef]
- Freisling, H.; Fahey, M.T.; Moskal, A.; Ocké, M.C.; Ferrari, P.; Jenab, M.; Norat, T.; Naska, A.; Welch, A.A.; Navarro, C.; et al. N. Region-specific nutrient intake patterns exhibit a geographical gradient within and between European countries. J. Nutr. 2010, 140, 1280–1286. [Google Scholar] [CrossRef] [PubMed]
- Viñas, B.R.; Barba, R.L.; Ngo, J.; Gurinovic, M.; Novakovic, R.; Cavelaars, A.; de Groot, L.C.P.G.M.; van’t Veer, P.; Matthys, C.; et al. Projected Prevalence of Inadequate Nutrient Intakes in Europe. Ann. Nutr. Metab. 2011, 59, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Mensink, G.B.; Fletcher, R.; Gurinovic, M.; Huybrechts, I.; Lafay, L.; Serra-Majem, L.; Szponar, L.; Tetens, I.; Verkaik-Kloosterman, J.; Baka, A.; Stephen, A.M. Mapping low intake of micronutrients across Europe. Br. J. Nutr. 2013, 110, 755–773. [Google Scholar] [CrossRef] [PubMed]
- Olza, J.; Aranceta-Bartrina, J.; González-Gross, M.; Ortega, R.M.; Serra-Majem, L.; Varela-Moreiras, G.; Gil, Á. Reported Dietary Intake, Disparity between the Reported Consumption and the Level Needed for Adequacy and Food Sources of Calcium, Phosphorus, Magnesium and Vitamin D in the Spanish Population: Findings from the ANIBES Study. Nutrients 2017, 9, 168. [Google Scholar] [CrossRef] [PubMed]
- Moyersoen, I.; Devleesschauwer, B.; Dekkers, A.; de Ridder, K.; Tafforeau, J.; van Camp, J.; van Oyen, H.; Lachat, C. Intake of Fat-Soluble Vitamins in the Belgian Population: Adequacy and Contribution of Foods, Fortified Foods and Supplements. Nutrients 2017, 9, 860. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Public Health. Jsou české děti dostatečně zásobeny vitaminem D? Prague, 2017. Available online: http://www.szu.cz/tema/zivotni-prostredi/vitamin-d-u-deti (accessed on 6 June 2018).
- Bates, B.; Lennox, A.; Prentice, A.; Bates, C.; Page, P.; Nicholson, S.; Swan, G. National Diet and Nutrition Survey Results from Years 1, 2, 3 and 4 (Combined) of the Rolling Programme (2008/2009–2011/2012). A survey carried out on behalf of Public Health England and the Food Standards Agency 2014. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/594361/NDNS_Y1_to_4_UK_report_full_text_revised_February_2017.pdf (accessed on 7 June 2018).
- Irish Universities Nutrition Alliance (IUNA). National Adult Nutrition Survey. Summary Report March 2011. Available online: https://irp-cdn.multiscreensite.com/46a7ad27/files/uploaded/The%20National%20Adult%20Nutrition%20Survey%20Summary%20Report%20March%202011.pdf (accessed on 7 June 2018).
- ANSES—French Agency for Food, Environmental and Occupational Health & Safety. 2016. Vitamin D Presentation, Food Sources and Nutritional Needs. Available online: https://www.anses.fr/en/content/vitamin-d (accessed on 7 June 2018).
- AESAN (Agencia Española de Seguridad Alimentaria y Nutrición). Evaluación nutricional de la dieta española. II Micronutrientes. Sobredatos de la Encuesta Nacional de Ingesta Dietética (ENIDE) 2011. Available online: http://www.laboratoriolcn.com/f/docs/Valoracion_nutricional_ENIDE_micronutrientes.pdf (accessed on 7 June 2018).
- Pietinen, P.; Paturi, M.; Reinivuo, H.; Tapanainen, H.; Valsta, L.M. FINDIET 2007 Survey: Energy and nutrient intakes. Public Health Nutr. 2010, 13, 920–924. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, L.; Stepien, M.; Gibney, M.J.; Nugent, A.P.; Brennan, L. The potential role of vitamin D enhanced foods in improving vitamin D status. Nutrients 2011, 3, 1023–1041. [Google Scholar] [CrossRef]
- Lamberg-Allardt, C.; Brustad, M.; Meyer, H.E.; Steingrimsdottir, L. Vitamin D—A systematic literature review for the 5th edition of the Nordic Nutrition Recommendations. Food. Nutr. Res 2013, 57, 1–31. [Google Scholar] [CrossRef] [PubMed]
- The Czech Society for Nutrition. Nutritional recommendations for the population of the Czech Republic—2012. Available online: http://www.vyzivaspol.cz/vyzivova-doporuceni-pro-obyvatelstvo-ceske-republiky/ (accessed on 23 June 2018).
- Czech Statistical Office. Food Consumption—2016. Available online: https://www.czso.cz/csu/czso/spotreba-potravin-2016 (accessed on 7 June 2018).
| Sex/Age Category | Number of Subjects | Characteristics of Age Groups According to Percentiles 25th–Mean–75th | Mean Body Weight (kg) | Mean Energy Intake (kcal·day−1) (Data Including Misreporters) |
|---|---|---|---|---|
| Children, 4–6 years | 196 | 4.0–5.1–5.4 | 21.3 | 1903 |
| Children, 7–10 years | 311 | 7.4–8.8–9.2 | 32.6 | 2192 |
| Males, 11–14 years | 49 | 11.2–12.6–13.0 | 47.8 | 2891 |
| Females, 11–14 years | 51 | 11.0–12.4–13.0 | 46.6 | 2340 |
| Males, 15–17 years | 56 | 15.1–16.2–16.4 | 66.2 | 3474 |
| Females, 15–17 years | 58 | 15.0–16.1–16.4 | 55.8 | 2098 |
| Males, 18–64 years | 793 | 30.3–41.9–53.0 | 82.6 | 3148 |
| Females, 18–64 years | 873 | 31.5–44.0–55.5 | 69.1 | 2018 |
| Males, ≥65 years | 80 | 65.7–70.9–74.3 | 82.9 | 2660 |
| Females, ≥65 years | 123 | 66.8–71.8–75.3 | 73.3 | 1980 |
| Sex/Age Category | n | Percentiles of Usual Intakes (LB-UB) (1) | Requirement AI (2) (μg/day) | % Inadequacy (3) | Requirement EAR | % of Individuals Exceeding the EAR (LB-UB Intakes) (4) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 5 | 25 | 50 | 75 | 95 | |||||||
| (μg/person/day) | EFSA * 2016 | DACH * 2015 | IoM US * 2011 | ||||||||
| Children, both sexes, 4–6 years | 196 | 1.6–2.2 | 2.1–2.8 | 2.5–3.4 | 3.1–4.0 | 4.0–5.1 | 15 | 20 | ns | 10 | 0–0 |
| Children, both sexes, 7–10 years | 311 | 1.8–2.4 | 2.3–3.1 | 2.7–3.7 | 3.3–4.3 | 4.2–5.5 | 15 | 20 | ns | 10 | 0–0 |
| Males, 11–14 years | 49 | 2.0–2.7 | 3.0–3.8 | 3.8–4.9 | 4.9–6.2 | 7.1–8.9 | 15 | 20 | ns | 10 | 0.6–2.5 |
| Females, 11–14 years | 51 | 1.2–1.7 | 1.8–2.6 | 2.5–3.5 | 3.5–4.6 | 5.6–7.0 | 15 | 20 | ns | 10 | 0.3–0.7 |
| Males, 15–17 years | 56 | 1.9–2.5 | 2.8–3.7 | 3.8–4.9 | 5.0–6.5 | 7.6–9.8 | 15 | 20 | ns | 10 | 1.1–4.5 |
| Females, 15–17 years | 58 | 1.0–1.2 | 1.7–2.1 | 2.5–3.1 | 3.6–4.7 | 6.1–8.3 | 15 | 20 | ns | 10 | 0.5–2.5 |
| Males, 18–64 years | 793 | 2.0–2.6 | 3.0–3.9 | 3.9–5.1 | 5.2–6.7 | 7.6–9.9 | 15 | 20 | ns | 10 | 1.0–4.8 |
| Females, 18–64 years | 873 | 1.4–1.9 | 2.1–2.7 | 2.8–3.5 | 3.7–4.6 | 5.6–6.7 | 15 | 20 | ns | 10 | 0.1–0.3 |
| Males, ≥65 years | 80 | 1.6–2.2 | 2.5–3.4 | 3.5–4.6 | 4.7–6.2 | 7.3–9.5 | 15 | 20 | ns | 10 | 1.0–3.9 |
| Females, ≥65 years | 123 | 2.1–2.6 | 2.7–3.3 | 3.1–3.9 | 3.6–4.5 | 4.4–5.7 | 15 | 20 | ns | 10 | 0–0 |
| Sex/Age Category | Food | % Contribution to Usual Dietary Intake of Vitamin D | Sex/Age Category | Food | % Contribution to Usual Dietary Intake of Vitamin D |
|---|---|---|---|---|---|
| Children, 4–6 years | Hen eggs | 20.9 | Children, 7–10 years | Hen eggs | 21.4 |
| Fine bakery wares (excluding biscuits) | 15.3 | Fine bakery wares (excluding biscuits) | 19.2 | ||
| Cow’s milk | 12.5 | Margarines | 10.4 | ||
| Margarines | 12.4 | Cow’s milk | 9.8 | ||
| Cocoa drinks powder | 5.5 | Butter | 5.1 | ||
| ∑ | 66.6 | ∑ | 65.9 | ||
| Males, 11–14 years | Hen eggs | 25.7 | Females, 11–14 years | Hen eggs | 22.7 |
| Margarines | 15.8 | Fine bakery wares (excluding biscuits) | 17.5 | ||
| Fine bakery wares (excluding biscuits) | 14.4 | Margarines | 13.1 | ||
| Cow’s milk | 7.7 | Cow’s milk | 8.9 | ||
| Butter | 5.1 | Freshwater fish | 5.8 | ||
| ∑ | 68.7 | ∑ | 68.0 | ||
| Males, 15–17 years | Hen eggs | 23.8 | Females, 15–17 years | Hen eggs | 27.9 |
| Fine bakery wares (excluding biscuits) | 14.6 | Fine bakery wares excluding biscuits | 12.4 | ||
| Margarines | 8.6 | Margarines | 7.1 | ||
| Cow’s milk | 5.4 | Cow’s milk | 6.4 | ||
| Marinated fish | 5.3 | Smoked fish | 5.6 | ||
| ∑ | 57.7 | ∑ | 59.4 | ||
| Males, 18–64 years | Hen eggs | 21.4 | Females, 18–64 years | Hen eggs | 23.3 |
| Fine bakery wares (excluding biscuits) | 11.4 | Fine bakery wares (excluding biscuits) | 12.3 | ||
| Margarines | 9.4 | Margarines | 12.3 | ||
| Salads * | 6.2 | Freshwater fish | 8,0 | ||
| Pork meat | 4.8 | Salads * | 4.2 | ||
| ∑ | 53.2 | ∑ | 60.1 | ||
| Males, ≥65 years | Hen eggs | 22.6 | Females, ≥65 years | Hen eggs | 21.7 |
| Margarines | 12.7 | Margarines | 17.6 | ||
| Fine bakery wares (excluding biscuits) | 12.3 | Fine bakery wares (excluding biscuits) | 14.2 | ||
| Butter | 5.3 | Freshwater fish | 9.8 | ||
| Marinated fish | 5.2 | Cow’s milk | 5.4 | ||
| ∑ | 58.1 | ∑ | 68.7 |
| Food Group | Number (n) of Kinds of Foods in a Group | Range of Vitamin D Content (Sum D2 + D3) μg/100 g Edible Portion |
|---|---|---|
| Fish and products | 5 | 0.83–11.11 |
| Milk-based infant formula powder | 1 | 9.92 |
| Cocoa drink powder | 1 | 1.12 |
| Chocolate and chocolate products | 2 | <LOQ * |
| Eggs | 1 | 4.03 |
| Milk and cream | 4 | 0.16–0.53 |
| Fermented milk products | 5 | <LOQ *–0.62 |
| Dairy desserts | 4 | 0.07–0.71 |
| Cheese | 7 | 0.05–0.85 |
| Vegetable fats and oils | 4 | <LOQ *–8.17 |
| Animal fats | 2 | <LOQ *–0.85 |
| Meat and offal | 10 | <LOQ *–1.73 |
| Meat products | 16 | <LOQ *–1.93 |
| Cereals and flours | 4 | <LOQ * |
| Bread and similar products | 5 | <LOQ * |
| Fine bakery products | 6 | <LOQ *–1.09 |
| Pasta and doughs | 2 | <LOQ * |
| Fruit, vegetables and cultivated mushrooms | 5 | <LOQ * |
| Miscellaneous foods | 2 | 0.25–2.99 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bischofova, S.; Dofkova, M.; Blahova, J.; Kavrik, R.; Nevrla, J.; Rehurkova, I.; Ruprich, J. Dietary Intake of Vitamin D in the Czech Population: A Comparison with Dietary Reference Values, Main Food Sources Identified by a Total Diet Study. Nutrients 2018, 10, 1452. https://doi.org/10.3390/nu10101452
Bischofova S, Dofkova M, Blahova J, Kavrik R, Nevrla J, Rehurkova I, Ruprich J. Dietary Intake of Vitamin D in the Czech Population: A Comparison with Dietary Reference Values, Main Food Sources Identified by a Total Diet Study. Nutrients. 2018; 10(10):1452. https://doi.org/10.3390/nu10101452
Chicago/Turabian StyleBischofova, Svatava, Marcela Dofkova, Jitka Blahova, Radek Kavrik, Jana Nevrla, Irena Rehurkova, and Jiri Ruprich. 2018. "Dietary Intake of Vitamin D in the Czech Population: A Comparison with Dietary Reference Values, Main Food Sources Identified by a Total Diet Study" Nutrients 10, no. 10: 1452. https://doi.org/10.3390/nu10101452
APA StyleBischofova, S., Dofkova, M., Blahova, J., Kavrik, R., Nevrla, J., Rehurkova, I., & Ruprich, J. (2018). Dietary Intake of Vitamin D in the Czech Population: A Comparison with Dietary Reference Values, Main Food Sources Identified by a Total Diet Study. Nutrients, 10(10), 1452. https://doi.org/10.3390/nu10101452




