Effects of Whole Milk Supplementation on Gut Microbiota and Cardiometabolic Biomarkers in Subjects with and without Lactose Malabsorption
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Milk Intervention and Dietary Intake Assessment
2.3. Cardiometabolic Biomarkers Measurement
2.4. Fecal Samples Collection and DNA Extraction
2.5. 16S Ribosomal RNA Gene Sequencing
2.6. Fecal Microbiota Analysis
2.7. Fecal SCFA Analysis
2.8. Quantitative Polymerase Chain Reaction (qPCR)
2.9. Statistical Analysis
3. Results
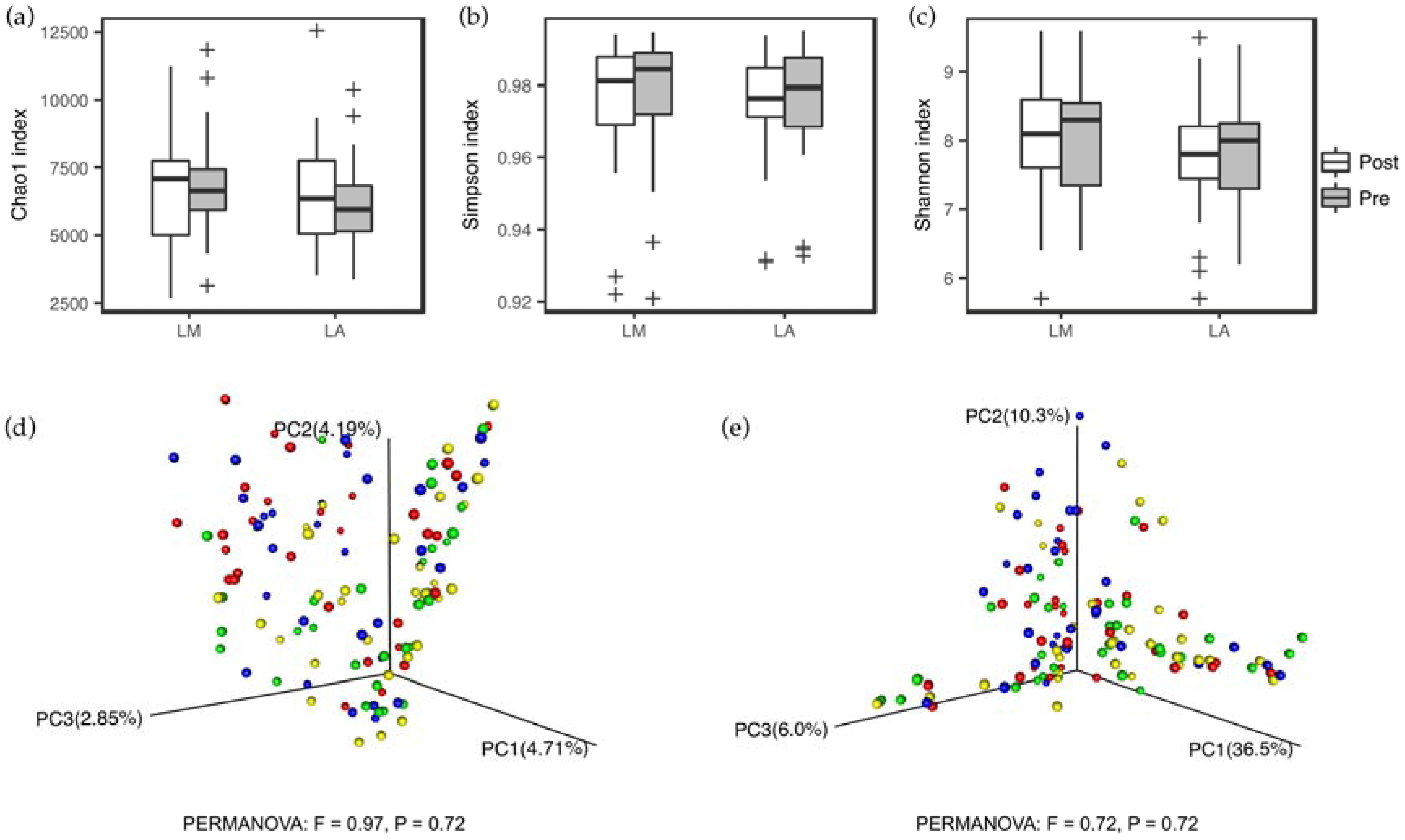
3.1. Fecal Microbiota
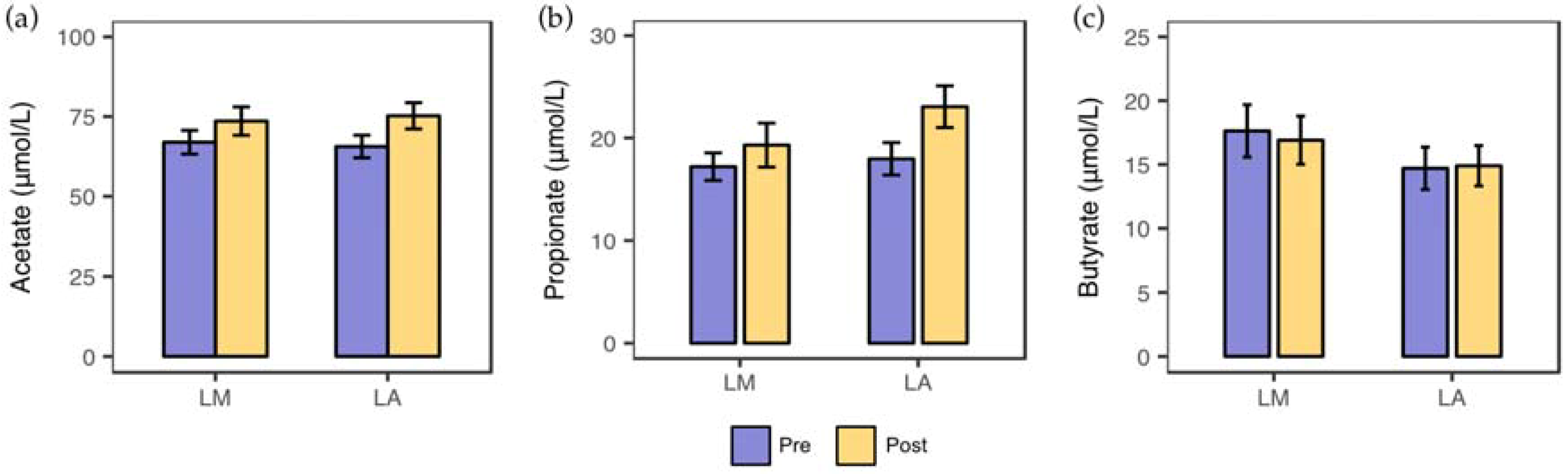
3.2. Fecal Short-Chain Fatty Acids Concentrations
3.3. Body Composition and Cardiometabolic Markers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pfeuffer, M.; Schrezenmeir, J. Milk and the metabolic syndrome. Obes. Rev. 2007, 8, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Rautiainen, S.; Wang, L.; Lee, I.-M.; Manson, J.E.; Buring, J.E.; Sesso, H.D. Dairy consumption in association with weight change and risk of becoming overweight or obese in middle-aged and older women: A prospective cohort study. Am. J. Clin. Nutr. 2016, 103, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Soedamah-Muthu, S.S.; Verberne, L.D.M.; Ding, E.L.; Engberink, M.F.; Geleijnse, J.M. Dairy consumption and incidence of hypertension: A dose-response meta-analysis of prospective cohort studies. Hypertension 2012, 60, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Dairy products and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis of cohort studies. Am. J. Clin. Nutr. 2013, 98, 1066–1083. [Google Scholar] [CrossRef] [PubMed]
- Soedamah-Muthu, S.S.; Ding, E.L.; Al-Delaimy, W.K.; Hu, F.B.; Engberink, M.F.; Willett, W.C.; Geleijnse, J.M. Milk and dairy consumption and incidence of cardiovascular diseases and all-cause mortality: Dose-response meta-analysis of prospective cohort studies. Am. J. Clin. Nutr. 2011, 93, 158–171. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Venema, K.; Priebe, M.G.; Welling, G.W.; Brummer, R.-J.M.; Vonk, R.J. The role of colonic metabolism in lactose intolerance. Eur. J. Clin. Investig. 2008, 38, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Suarez, F.L.; Savaiano, D.A.; Levitt, M.D. A comparison of symptoms after the consumption of milk or lactose-hydrolyzed milk by people with self-reported severe lactose intolerance. N. Engl. J. Med. 1995, 333, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.; Huang, T.; Bergholdt, H.K.; Nordestgaard, B.G.; Ellervik, C.; Qi, L. CHARGE Consortium Dairy consumption, systolic blood pressure, and risk of hypertension: Mendelian randomization study. BMJ 2017, 356, j1000. [Google Scholar] [CrossRef] [PubMed]
- Bergholdt, H.K.M.; Larsen, M.K.; Varbo, A.; Nordestgaard, B.G.; Ellervik, C. Lactase persistence, milk intake, hip fracture and bone mineral density: A study of 97,811 Danish individuals and a meta-analysis. J. Int. Med. 2018, 11, 462. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.G.; Yan, Y.S.; Xu, J.J.; Du, R.F.; Flatz, S.D.; Kühnau, W.; Flatz, G. Prevalence of primary adult lactose malabsorption in three populations of northern China. Hum. Genet. 1984, 67, 103–106. [Google Scholar] [PubMed]
- National Health and Family Planning Commission of the People’s Republic of China. Reprot of Chinese Nation Nutrition and Chronic Disease Survey, 1st ed.; People’s Medical Publishing House: Beijing, China, 2015; pp. 13–14. [Google Scholar]
- China Nutrition Society. The Chinese Dietary Guidelines, 1st ed.; People’s Medical Publishing House: Beijing, China, 2016; pp. 42–45. [Google Scholar]
- Szilagyi, A.; Shrier, I.; Heilpern, D.; Je, J.; Park, S.; Chong, G.; Lalonde, C.; Cote, L.-F.; Lee, B. Differential impact of lactose/lactase phenotype on colonic microflora. Can. J. Gastroenterol. 2010, 24, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Flint, H.J.; Duncan, S.H.; Scott, K.P.; Louis, P. Interactions and competition within the microbial community of the human colon: Links between diet and health. Environ. Microbiol. 2007, 9, 1101–1111. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhang, F.; Ding, X.; Wu, G.; Lam, Y.Y.; Wang, X.; Fu, H.; Xue, X.; Lu, C.; Ma, J.; et al. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science 2018, 359, 1151–1156. [Google Scholar] [CrossRef] [PubMed]
- Kellow, N.J.; Coughlan, M.T.; Reid, C.M. Metabolic benefits of dietary prebiotics in human subjects: A systematic review of randomised controlled trials. Br. J. Nutr. 2014, 111, 1147–1161. [Google Scholar] [CrossRef] [PubMed]
- Gasbarrini, A.; Corazza, G.R.; Gasbarrini, G.; Montalto, M.; Di Stefano, M.; Basilisco, G.; Parodi, A.; Usai-Satta, P.; Satta, P.U.; Vernia, P.; et al. Methodology and indications of H2-breath testing in gastrointestinal diseases: The Rome Consensus conference. Aliment. Pharmacol. Ther. 2009, 29, 1–49. [Google Scholar] [PubMed]
- Yang, Y.; Wang, G.; Pan, X. China Food Composition Tables 2002, 2nd ed.; Beijing University Medical Press: Beijing, China, 2009; pp. 1–512. [Google Scholar]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Kuczynski, J.; Stombaugh, J.; Walters, W.A.; Gonzalez, A.; Caporaso, J.G.; Knight, R. Using QIIME to analyze 16S rRNA gene sequences from microbial communities. Curr. Protoc. Microbiol. 2012, 27, 1E.5.1–1E.5.20. [Google Scholar]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.R.; Wang, Q.; Cardenas, E.; Fish, J.; Chai, B.; Farris, R.J.; Kulam-Syed-Mohideen, A.S.; McGarrell, D.M.; Marsh, T.; Garrity, G.M.; et al. The Ribosomal Database Project: Improved alignments and new tools for rRNA analysis. Nucleic Acids Res. 2009, 37, D141–D145. [Google Scholar] [CrossRef] [PubMed]
- Matsuki, T.; Watanabe, K.; Fujimoto, J.; Takada, T.; Tanaka, R. Use of 16S rRNA gene-targeted group-specific primers for real-time PCR analysis of predominant bacteria in human feces. Appl. Environ. Microbiol. 2004, 70, 7220–7228. [Google Scholar] [CrossRef] [PubMed]
- Bartosch, S.; Fite, A.; Macfarlane, G.T.; McMurdo, M.E.T. Characterization of bacterial communities in feces from healthy elderly volunteers and hospitalized elderly patients by using real-time PCR and effects of antibiotic treatment on the fecal microbiota. Appl. Environ. Microbiol. 2004, 70, 3575–3581. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, M.; Raes, J.; Pelletier, E.; Le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; Batto, J.-M.; et al. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Nicolucci, A.C.; Hume, M.P.; Martinez, I.; Mayengbam, S.; Walter, J.; Reimer, R.A. Prebiotics reduce body fat and alter intestinal microbiota in children who are overweight or with obesity. Gastroenterology 2017, 153, 711–722. [Google Scholar] [CrossRef] [PubMed]
- Parche, S.; Beleut, M.; Rezzonico, E.; Jacobs, D.; Arigoni, F.; Titgemeyer, F.; Jankovic, I. Lactose-over-glucose preference in Bifidobacterium longum NCC2705: GlCP, encoding a glucose transporter, is subject to lactose repression. J. Bacteriol. 2006, 188, 1260–1265. [Google Scholar] [CrossRef] [PubMed]
- Bennet, S.M.P.; Böhn, L.; Störsrud, S.; Liljebo, T.; Collin, L.; Lindfors, P.; Törnblom, H.; Öhman, L.; Simrén, M. Multivariate modelling of faecal bacterial profiles of patients with IBS predicts responsiveness to a diet low in FODMAPs. Gut 2018, 67, 872–881. [Google Scholar] [CrossRef] [PubMed]
- Turroni, F.; Milani, C.; Duranti, S.; Mahony, J.; van Sinderen, D.; Ventura, M. Glycan utilization and cross-feeding activities by bifidobacteria. Trends Microbiol. 2018, 26, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Arboleya, S.; Stanton, C.; Ryan, C.A.; Dempsey, E.; Ross, P.R. Bosom Buddies: The symbiotic relationship between infants and Bifidobacterium longum ssp. longum and ssp. infantis. genetic and probiotic features. Annu. Rev. Food Sci. Technol. 2016, 7, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Falony, G.; Vlachou, A.; Verbrugghe, K.; De Vuyst, L. Cross-feeding between Bifidobacterium longum BB536 and acetate-converting, butyrate-producing colon bacteria during growth on oligofructose. Appl. Environ. Microbiol. 2006, 72, 7835–7841. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, N.; Lin, H.C.; McSweeney, C.S.; Mackie, R.I.; Gaskins, H.R. Mechanisms of microbial hydrogen disposal in the human colon and implications for health and disease. Annu. Rev. Food Sci. Technol. 2010, 1, 363–395. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.; Zhang, Y.; Zhu, X.; Liu, K.; Wang, X.; Chen, M.; Wang, J.; Chen, H.; Hui, S.; Huang, L.; et al. Healthy subjects differentially respond to dietary capsaicin correlating with specific gut enterotypes. J. Clin. Endocrinol. Metab. 2016, 101, 4681–4689. [Google Scholar] [CrossRef] [PubMed]
- Roager, H.M.; Licht, T.R.; Poulsen, S.K.; Larsen, T.M.; Bahl, M.I. Microbial enterotypes, inferred by the prevotella-to-bacteroides ratio, remained stable during a 6-month randomized controlled diet intervention with the new nordic diet. Appl. Environ. Microbiol. 2014, 80, 1142–1149. [Google Scholar] [CrossRef] [PubMed]
- Boets, E.; Gomand, S.V.; Deroover, L.; Preston, T.; Vermeulen, K.; De Preter, V.; Hamer, H.M.; Van den Mooter, G.; De Vuyst, L.; Courtin, C.M.; et al. Systemic availability and metabolism of colonic-derived short-chain fatty acids in healthy subjects: A stable isotope study. J. Physiol. 2016, 595, 541–555. [Google Scholar] [CrossRef] [PubMed]
- Canfora, E.E.; Jocken, J.W.; Blaak, E.E. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat. Rev. Endocrinol. 2015, 11, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Pan, A.; Malik, V.S.; Hu, F.B. Effects of dairy intake on body weight and fat: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2012, 96, 735–747. [Google Scholar] [CrossRef] [PubMed]
- Crichton, G.E.; Bryan, J.; Buckley, J.; Murphy, K.J. Dairy consumption and metabolic syndrome: A systematic review of findings and methodological issues. Obes. Rev. 2011, 12, e190–e201. [Google Scholar] [CrossRef] [PubMed]
- Machin, D.R.; Park, W.; Alkatan, M.; Mouton, M.; Tanaka, H. Hypotensive effects of solitary addition of conventional nonfat dairy products to the routine diet: A randomized controlled trial. Am. J. Clin. Nutr. 2014, 100, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Stancliffe, R.A.; Thorpe, T.; Zemel, M.B. Dairy attentuates oxidative and inflammatory stress in metabolic syndrome. Am. J. Clin. Nutr. 2011, 94, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Sun, Q.; Giovannucci, E.; Mozaffarian, D.; Manson, J.E.; Willett, W.C.; Hu, F.B. Dairy consumption and risk of type 2 diabetes: 3 cohorts of US adults and an updated meta-analysis. BMC Med. 2014, 12, 215. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Neyrinck, A.M.; Fava, F.; Knauf, C.; Burcelin, R.G.; Tuohy, K.M.; Gibson, G.R.; Delzenne, N.M. Selective increases of bifidobacteria in gut microflora improve high-fat-diet-induced diabetes in mice through a mechanism associated with endotoxaemia. Diabetologia 2007, 50, 2374–2383. [Google Scholar] [CrossRef] [PubMed]
- Nishijima, S.; Suda, W.; Oshima, K.; Kim, S.-W.; Hirose, Y.; Morita, H.; Hattori, M. The gut microbiome of healthy Japanese and its microbial and functional uniqueness. DNA Res. 2016, 23, 125–133. [Google Scholar] [CrossRef] [PubMed]


| Parameters | LM (n = 31) | LA (n = 31) | P |
|---|---|---|---|
| Gender (Male/Female) | 22/9 | 22/9 | 1.00 |
| Age (years) | 24.7 ± 0.4 | 24.8 ± 0.4 | 0.86 |
| Height (cm) | 170.0 ± 1.4 | 169.6 ± 1.4 | 0.86 |
| Weight (kg) | 62.8 ± 2.2 | 64.5 ± 2.1 | 0.58 |
| BMI (kg/m2) | 21.6 ± 0.6 | 22.3 ± 0.6 | 0.42 |
| Waist-hip ratio | 0.81 ± 0.01 | 0.82 ± 0.01 | 0.57 |
| DBP (mmHg) | 75.6 ± 1.1 | 75.7 ± 1.7 | 0.98 |
| SBP (mmHg) | 115.9 ± 1.9 | 116.2 ± 2.4 | 0.92 |
| FPG (mmol/L) | 5.14 ± 0.06 | 5.15 ± 0.07 | 0.89 |
| FPI (mU/L) | 5.72 ± 0.63 | 5.86 ± 0.64 | 0.91 |
| HOMA-IR | 1.30 ± 0.15 | 1.36 ± 0.16 | 0.89 |
| TG (mmol/L) | 0.74 ± 0.07 | 0.86 ± 0.06 | 0.09 |
| TC (mmol/L) | 4.22 ± 0.15 | 4.19 ± 0.22 | 0.93 |
| LDL-C (mmol/L) | 2.52 ± 0.14 | 2.39 ± 0.14 | 0.51 |
| HDL-C (mmol/L) | 1.32 ± 0.06 | 1.40 ± 0.08 | 0.40 |
| Dairy intake (servings/day) | 0.51 ± 0.12 | 0.58 ± 0.13 | 0.34 |
| ΔH2 (ppm) | 73.8 ± 7.3 | 11.7 ± 0.8 | <0.01 |
| Parameters | LM (n = 31) | LA (n = 31) | P | ||
|---|---|---|---|---|---|
| Pre | Post | Pre | Post | ||
| Weight (kg) | 62.8 ± 2.2 | 62.5 ± 2.2 | 64.5 ± 2.1 | 64.0 ± 2.2 | 0.55 |
| BMI (kg/m2) | 21.6 ± 0.6 | 21.5 ± 0.6 | 22.3 ± 0.6 | 22.2 ± 0.6 | 0.60 |
| Body fat mass (kg) | 13.6 ± 1.1 | 12.3 ± 1.1 * | 13.9 ± 1.2 | 12.9 ± 1.1 ** | 0.54 |
| Lean mass (kg) | 27.4 ± 1.0 | 28.2 ± 1.1 | 28.5 ± 1.1 | 28.6 ± 1.1 | 0.25 |
| Body fat (%) | 21.5 ± 1.2 | 19.5 ± 1.3 * | 21.4 ± 1.4 | 19.9 ± 1.3 ** | 0.56 |
| DBP (mmHg) | 75.6 ± 1.1 | 74.5 ± 1.2 | 75.7 ± 1.7 | 78.6 ± 1.6 | 0.15 |
| SBP (mmHg) | 115.9 ± 1.9 | 114.4 ± 1.7 | 116.2 ± 2.4 | 112.9 ± 2.0 | 0.47 |
| FPG (mmol/L) | 5.14 ± 0.06 | 5.20 ± 0.06 | 5.15 ± 0.07 | 5.28 ± 0.07 | 0.45 |
| FPI (mU/L) | 5.72 ± 0.63 | 6.22 ± 0.59 | 5.86 ± 0.64 | 5.48 ± 0.53 | 0.78 |
| HOMA-IR | 1.30 ± 0.15 | 1.45 ± 0.15 | 1.36 ± 0.16 | 1.22 ± 0.14 | 0.39 |
| C-peptide (nmol/L) | 0.45 ± 0.03 | 0.44 ± 0.03 | 0.45 ± 0.04 | 0.43 ± 0.03 | 0.84 |
| TG (mmol/L) | 0.74 ± 0.07 | 0.77 ± 0.07 | 0.86 ± 0.06 | 0.92 ± 0.09 | 0.85 |
| TC (mmol/L) | 4.22 ± 0.15 | 4.08 ± 0.14 | 4.19 ± 0.22 | 3.88 ± 0.16 | 0.93 |
| LDL-C (mmol/L) | 2.52 ± 0.14 | 2.52 ± 0.13 | 2.39 ± 0.14 | 2.50 ± 0.13 | 0.42 |
| HDL-C (mmol/L) | 1.32 ± 0.06 | 1.33 ± 0.07 | 1.40 ± 0.08 | 1.36 ± 0.07 | 0.52 |
| CRP (μg/mL) | 0.68 ± 0.22 | 0.81 ± 0.22 | 0.80 ± 0.31 | 0.66 ± 0.20 | 0.71 |
| MDA (nmol/mL) | 4.95 ± 0.19 | 4.87 ± 0.19 | 4.92 ± 0.19 | 4.84 ± 0.15 | 0.98 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Yin, J.; Zhu, Y.; Wang, X.; Hu, X.; Bao, W.; Huang, Y.; Chen, L.; Chen, S.; Yang, W.; et al. Effects of Whole Milk Supplementation on Gut Microbiota and Cardiometabolic Biomarkers in Subjects with and without Lactose Malabsorption. Nutrients 2018, 10, 1403. https://doi.org/10.3390/nu10101403
Li X, Yin J, Zhu Y, Wang X, Hu X, Bao W, Huang Y, Chen L, Chen S, Yang W, et al. Effects of Whole Milk Supplementation on Gut Microbiota and Cardiometabolic Biomarkers in Subjects with and without Lactose Malabsorption. Nutrients. 2018; 10(10):1403. https://doi.org/10.3390/nu10101403
Chicago/Turabian StyleLi, Xiaoqin, Jiawei Yin, Yalun Zhu, Xiaoqian Wang, Xiaoli Hu, Wei Bao, Yue Huang, Liangkai Chen, Sijing Chen, Wei Yang, and et al. 2018. "Effects of Whole Milk Supplementation on Gut Microbiota and Cardiometabolic Biomarkers in Subjects with and without Lactose Malabsorption" Nutrients 10, no. 10: 1403. https://doi.org/10.3390/nu10101403
APA StyleLi, X., Yin, J., Zhu, Y., Wang, X., Hu, X., Bao, W., Huang, Y., Chen, L., Chen, S., Yang, W., Shan, Z., & Liu, L. (2018). Effects of Whole Milk Supplementation on Gut Microbiota and Cardiometabolic Biomarkers in Subjects with and without Lactose Malabsorption. Nutrients, 10(10), 1403. https://doi.org/10.3390/nu10101403





