Considerations for Secondary Prevention of Nutritional Deficiencies in High-Risk Groups in High-Income Countries
Abstract
:1. Introduction
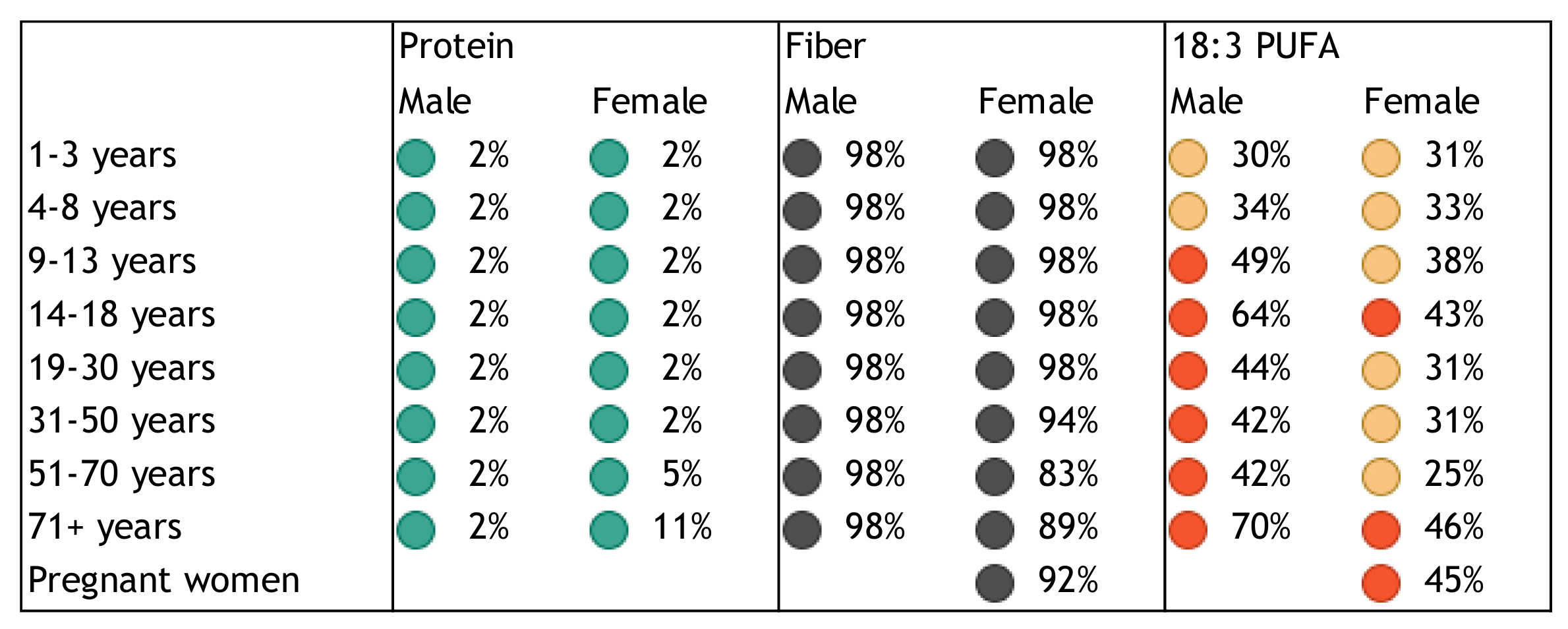
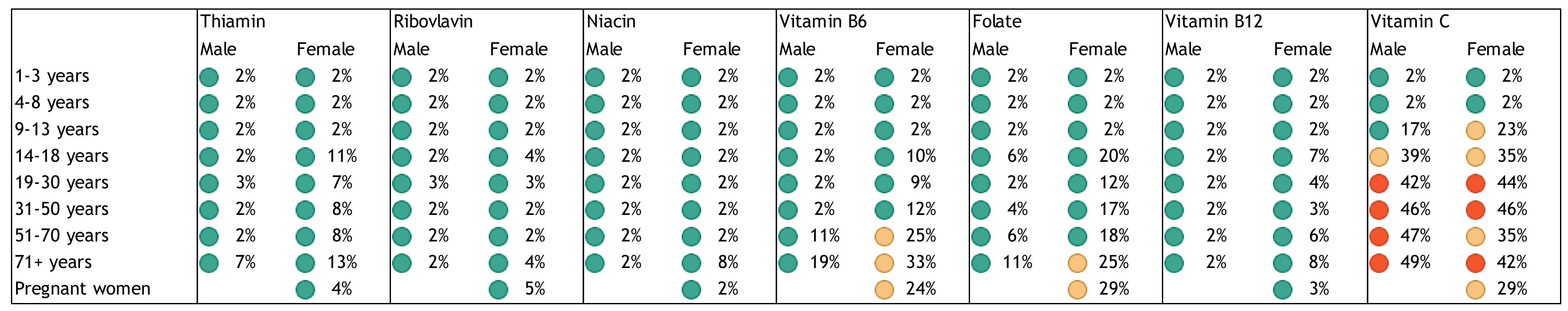
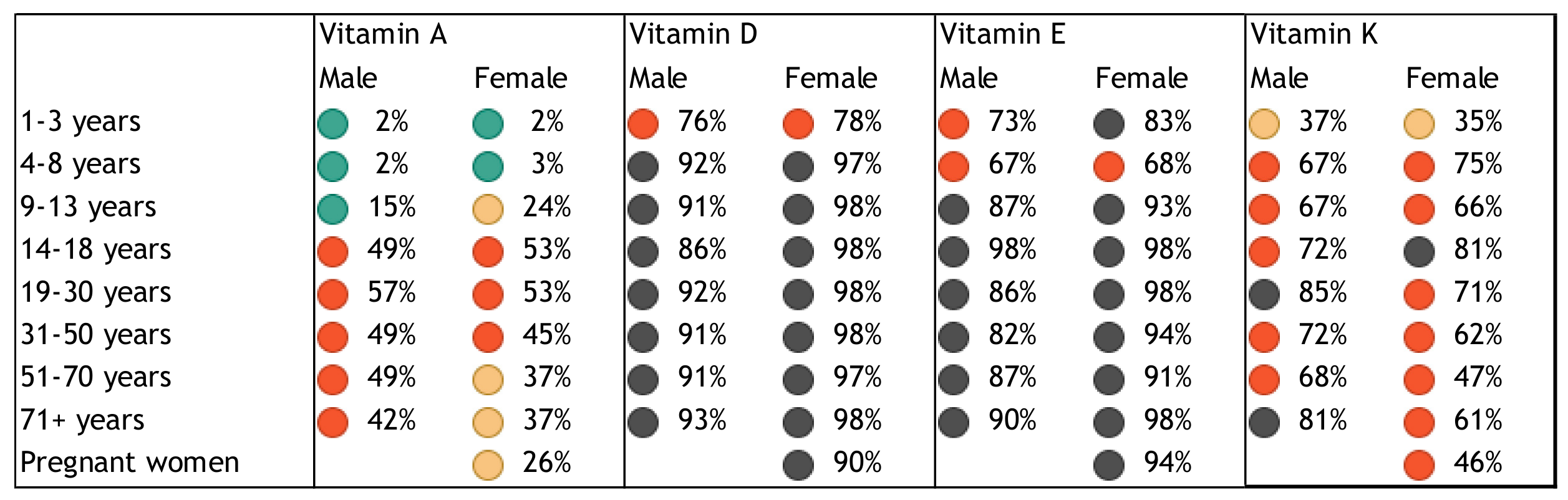
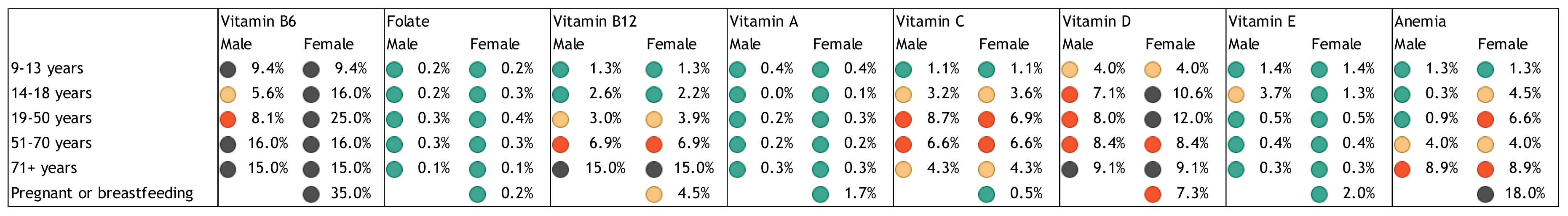
2. Nutrient Inadequacies and Deficiencies
3. Public Health Problem of Inadequate Intakes and Deficiencies of Nutrients
4. Common Approaches to Prevent Nutrient Inadequacies and Deficiencies
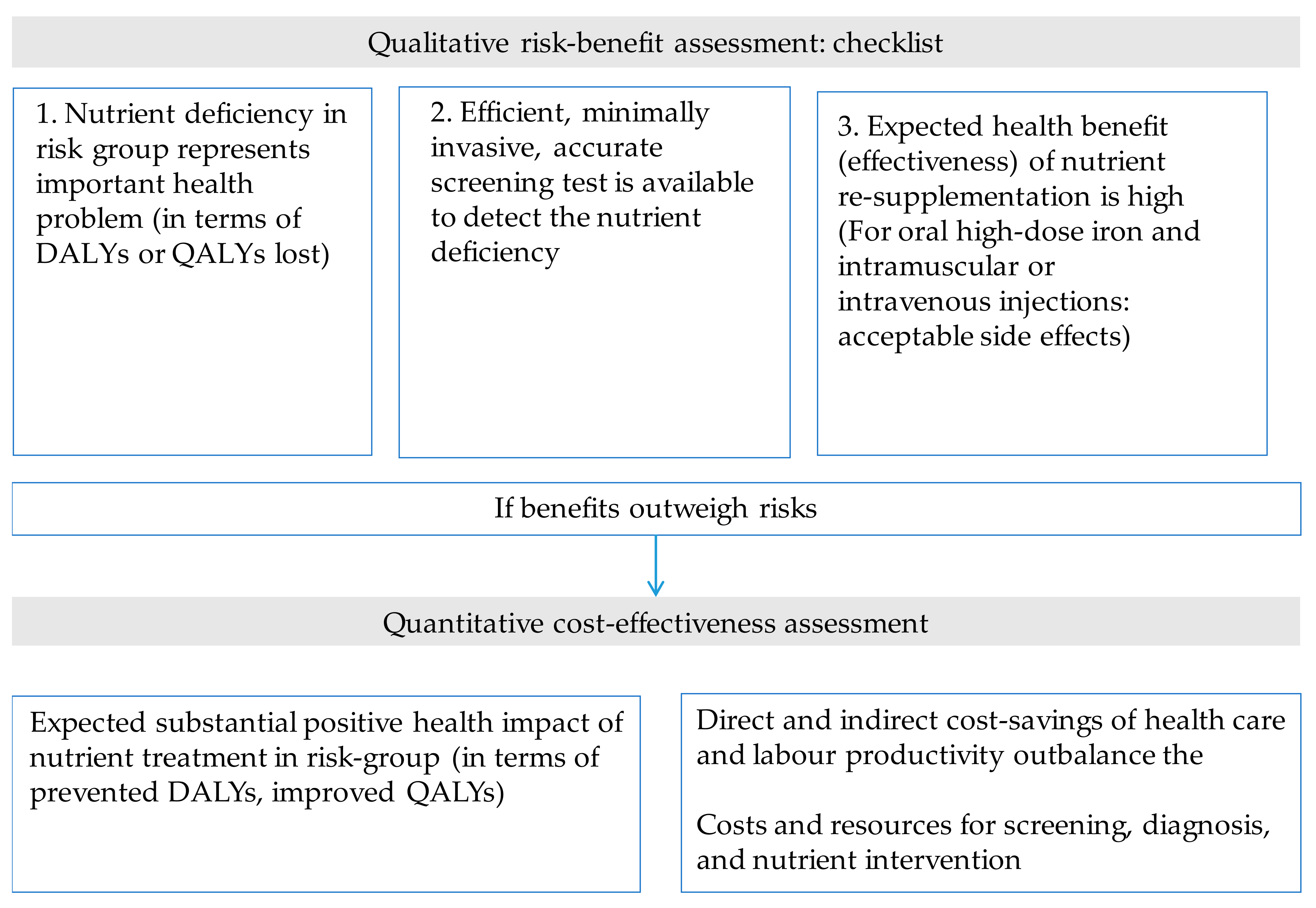
5. Criteria Determining Cost-Effectiveness
5.1. Public Health and Economic Consequences
5.2. Evidence Base Supporting Improved Health Outcomes, Discomfort and Risks
5.3. Availability of an Accurate Test
5.4. Adoption and Adherence
5.5. Costs and Cost Savings
5.6. Evidence Gaps in Evaluating Secondary Nutrition Strategies
6. Discussion
7. Conclusions
Author Contributions
Conflicts of Interest
Funding
References
- Boyle, M.A.; Holben, D.H. Community Nutrition in Action: An Entrepreneurial Approach, 5th ed.; Wadsworth: Belmont, CA, USA, 2010. [Google Scholar]
- Herring, D.; Chang, S.; Bard, S.; Gavey, E. Five years of myplate-looking back and what’s ahead. J. Acad. Nutr. Diet. 2016, 116, 1069–1071. [Google Scholar] [CrossRef] [PubMed]
- Montagnese, C.; Santarpia, L.; Iavarone, F.; Strangio, F.; Caldara, A.R.; Silvestri, E.; Contaldo, F.; Pasanisi, F. North and south american countries food-based dietary guidelines: A comparison. Nutrition 2017, 42, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Montagnese, C.; Santarpia, L.; Buonifacio, M.; Nardelli, A.; Caldara, A.R.; Silvestri, E.; Contaldo, F.; Pasanisi, F. European food-based dietary guidelines: A comparison and update. Nutrition 2015, 31, 908–915. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, C.M.; Sternberg, M.R.; Schleicher, R.L.; Haynes, B.M.; Rybak, M.E.; Pirkle, J.L. The CDC’s second national report on biochemical indicators of diet and nutrition in the U.S. Population is a valuable tool for researchers and policy makers. J. Nutr. 2013, 143, 938S–947S. [Google Scholar] [CrossRef] [PubMed]
- Spiro, A.; Buttriss, J.L. Vitamin D: An overview of vitamin D status and intake in Europe. Nutr. Bull. 2014, 39, 322–350. [Google Scholar] [CrossRef] [PubMed]
- Troesch, B.; Hoeft, B.; McBurney, M.; Eggersdorfer, M.; Weber, P. Dietary surveys indicate vitamin intakes below recommendations are common in representative western countries. Br. J. Nutr. 2012, 108, 692–698. [Google Scholar] [CrossRef] [PubMed]
- Diethelm, K.; Huybrechts, I.; Moreno, L.; De Henauw, S.; Manios, Y.; Beghin, L.; Gonzalez-Gross, M.; Le Donne, C.; Cuenca-Garcia, M.; Castillo, M.J.; et al. Nutrient intake of european adolescents: Results of the helena (healthy lifestyle in Europe by nutrition in adolescence) study. Public Health Nutr. 2014, 17, 486–497. [Google Scholar] [CrossRef] [PubMed]
- Herbert, V. The five possible causes of all nutrient deficiency: Illustrated by deficiencies of vitamin B12 and folic acid. Aust. N. Z. J. Med. 1972, 2, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Serra-Majem, L.; Ribas, L.; Perez-Rodrigo, C.; Garcia-Closas, R.; Pena-Quintana, L.; Aranceta, J. Determinants of nutrient intake among children and adolescents: Results from the enkid study. Ann. Nutr. Metab. 2002, 46 (Suppl. 1), 31–38. [Google Scholar] [CrossRef] [PubMed]
- Cordain, L.; Eaton, S.B.; Sebastian, A.; Mann, N.; Lindeberg, S.; Watkins, B.A.; O’Keefe, J.H.; Brand-Miller, J. Origins and evolution of the western diet: Health implications for the 21st century. Am. J. Clin. Nutr. 2005, 81, 341–354. [Google Scholar] [PubMed]
- Marangoni, F.; Cetin, I.; Verduci, E.; Canzone, G.; Giovannini, M.; Scollo, P.; Corsello, G.; Poli, A. Maternal diet and nutrient requirements in pregnancy and breastfeeding. An Italian consensus document. Nutrients 2016, 8, 629. [Google Scholar] [CrossRef] [PubMed]
- Dewey, K.G. The challenge of meeting nutrient needs of infants and young children during the period of complementary feeding: An evolutionary perspective. J. Nutr. 2013, 143, 2050–2054. [Google Scholar] [CrossRef] [PubMed]
- Story, M.; Hermanson, J. Nutrient needs during adolescence and pregnancy: A practical reference guide. In Nutrient Needs during Adolescence and Pregnancy; Story, M., Stang, J., Eds.; Center for Leadership, Education, and Training in Maternal and Child Nutrition, University of Minnesota: Minneapolis, MN, USA, 2000; Chapter 5. [Google Scholar]
- Leslie, W.; Hankey, C. Aging, nutritional status and health. Healthcare 2015, 3, 648–658. [Google Scholar] [CrossRef] [PubMed]
- Mehta, N.M.; Duggan, C.P. Nutritional deficiencies during critical illness. Pediatr. Clin. N. Am. 2009, 56, 1143–1160. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.M.; Chiolero, R.L. Key vitamins and trace elements in the critically ill. Nestle Nutr. Workshop Ser. Clin. Perform. Programme 2003, 8, 99–111, discussion 111–117. [Google Scholar] [PubMed]
- Wong, C.W. Vitamin B12 deficiency in the elderly: Is it worth screening? Hong Kong Med. J. 2015, 21, 155–164. [Google Scholar] [PubMed]
- Hurrell, R.; Egli, I. Iron bioavailability and dietary reference values. Am. J. Clin. Nutr. 2010, 91, 1461S–1467S. [Google Scholar] [CrossRef] [PubMed]
- West, C.E.; Eilander, A.; van Lieshout, M. Consequences of revised estimates of carotenoid bioefficacy for dietary control of vitamin A deficiency in developing countries. J. Nutr. 2002, 132, 2920S–2926S. [Google Scholar] [PubMed]
- Reilly, W.; Ilich, J.Z. Prescription drugs and nutrient depletion: How much is known? Adv. Nutr. 2017, 8, 23. [Google Scholar]
- Stover, P.J. Influence of human genetic variation on nutritional requirements. Am. J. Clin. Nutr. 2006, 83, 436S–442S. [Google Scholar] [PubMed]
- Suskind, D.L. Nutritional deficiencies during normal growth. Pediatr. Clin. N. Am. 2009, 56, 1035–1053. [Google Scholar] [CrossRef] [PubMed]
- Weininger, J. Nutritional Disease: Nutrient Deficiencies. Available online: https://www.britannica.com/science/nutritional-disease (accessed on 2 January 2018).
- National Institutes of Health (NIH), Office of Dietary Supplement. Dietary Supplement Fact Sheets. Available online: https://ods.od.nih.gov/factsheets/ (accessed on 2 January 2018).
- Tinker, S.C.; Hamner, H.C.; Qi, Y.P.; Crider, K.S. U.S. Women of childbearing age who are at possible increased risk of a neural tube defect-affected pregnancy due to suboptimal red blood cell folate concentrations, national health and nutrition examination survey 2007 to 2012. Birth Defects Res. Part A Clin. Mol. Teratol. 2015, 103, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Peter, S.; Eggersdorfer, M.; van Asselt, D.; Buskens, E.; Detzel, P.; Freijer, K.; Koletzko, B.; Kraemer, K.; Kuipers, F.; Neufeld, L.; et al. Selected nutrients and their implications for health and disease across the lifespan: A roadmap. Nutrients 2014, 6, 6076–6094. [Google Scholar] [CrossRef] [PubMed]
- Darnton-Hill, I.; Webb, P.; Harvey, P.W.; Hunt, J.M.; Dalmiya, N.; Chopra, M.; Ball, M.J.; Bloem, M.W.; de Benoist, B. Micronutrient deficiencies and gender: Social and economic costs. Am. J. Clin. Nutr. 2005, 81, 1198S–1205S. [Google Scholar] [PubMed]
- Departments of Agriculture and Health and Human Services. Part D. Chapter 1: Food and nutrient intakes, and health: Current status and trends. In Scientific Report of the 2015 Dietary Guidelines Advisory Committee; Departments of Agriculture and Health and Human Services: Washington, DC, USA, 2015. [Google Scholar]
- Marvin-Dowle, K.; Burley, V.J.; Soltani, H. Nutrient intakes and nutritional biomarkers in pregnant adolescents: A systematic review of studies in developed countries. BMC Pregnancy Childbirth 2016, 16, 268. [Google Scholar] [CrossRef] [PubMed]
- Blumfield, M.L.; Hure, A.J.; Macdonald-Wicks, L.; Smith, R.; Collins, C.E. A systematic review and meta-analysis of micronutrient intakes during pregnancy in developed countries. Nutr. Rev. 2013, 71, 118–132. [Google Scholar] [CrossRef] [PubMed]
- Gernand, A.D.; Schulze, K.J.; Stewart, C.P.; West, K.P., Jr.; Christian, P. Micronutrient deficiencies in pregnancy worldwide: Health effects and prevention. Nat. Rev. Endocrinol. 2016, 12, 274–289. [Google Scholar] [CrossRef] [PubMed]
- Bird, J.K.; Murphy, R.A.; Ciappio, E.D.; McBurney, M.I. Risk of deficiency in multiple concurrent micronutrients in children and adults in the united states. Nutrients 2017, 9, 655. [Google Scholar] [CrossRef] [PubMed]
- Hilger, J.; Goerig, T.; Weber, P.; Hoeft, B.; Eggersdorfer, M.; Carvalho, N.C.; Goldberger, U.; Hoffmann, K. Micronutrient intake in healthy toddlers: A multinational perspective. Nutrients 2015, 7, 6938–6955. [Google Scholar] [CrossRef] [PubMed]
- Akkermans, M.D.; Eussen, S.R.; van der Horst-Graat, J.M.; van Elburg, R.M.; van Goudoever, J.B.; Brus, F. A micronutrient-fortified young-child formula improves the iron and vitamin D status of healthy young European children: A randomized, double-blind controlled trial. Am. J. Clin. Nutr. 2017, 105, 391. [Google Scholar] [CrossRef] [PubMed]
- Kaganov, B.; Caroli, M.; Mazur, A.; Singhal, A.; Vania, A. Suboptimal micronutrient intake among children in Europe. Nutrients 2015, 7, 3524–3535. [Google Scholar] [CrossRef] [PubMed]
- Forrest, K.Y.; Stuhldreher, W.L. Prevalence and correlates of vitamin D deficiency in US adults. Nutr. Res. 2011, 31, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Tussing-Humphreys, L.; Van Nguyen, T.Q. Adipose tissue and adipokines in health and disease, nutrition and health. In Obesity and Micronutrient Deficiencies; Fantuzzi, G., Mazzone, T., Eds.; Springer: New York, NY, USA, 2014. [Google Scholar]
- Food Surveys Research Group; Beltsville Human Nutrition Research Center; Agricultural Research Service, U.S. Department of Agriculture (USDA ARS). What We Eat in America, NHANES 2007–2010, Individuals 1 Year and over (Excluding Breast-Fed Children and Pregnant or Lactating Females), Dietary Intake Data. Available online: https://www.cdc.gov/nchs/nhanes/wweia.htm (accessed on 2 January 2018).
- Hwalla, N.; Al Dhaheri, A.S.; Radwan, H.; Alfawaz, H.A.; Fouda, M.A.; Al-Daghri, N.M.; Zaghloul, S.; Blumberg, J.B. The prevalence of micronutrient deficiencies and inadequacies in the middle east and approaches to interventions. Nutrients 2017, 9, 229. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, E.M.A. Multiple sclerosis is prominent in the gulf states: Review. Pathogenesis 2016, 3, 19–38. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations (FAO). Regional Overview of Food Insecurity: Europe and Central Asia; FAO: Budapest, Hungary, 2017. [Google Scholar]
- Global Burden of Disease (GBD) 2013 Risk Factors Collaborators. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2015: A systematic analysis for the global burden of disease study 2015. Lancet 2016, 388, 1659–1724. [Google Scholar]
- World Health Organization (WHO). Iron Deficiency Anaemia Assessment, Prevention and Control: A Guide for Programme Managers; WHO: Geneva, Switzerland, 2001. [Google Scholar]
- Imamura, F.; Micha, R.; Khatibzadeh, S.; Fahimi, S.; Shi, P.; Powles, J.; Mozaffarian, D.; Global Burden of Diseases Nutrition and Chronic Diseases Expert Group (NutriCoDE). Dietary quality among men and women in 187 countries in 1990 and 2010: A systematic assessment. Lancet Glob. Health 2015, 3, e132–e142. [Google Scholar] [CrossRef]
- United States Department of Agriculture (USDA), Food and Nutrition Service. In Women, Infants and Children (WIC). About WIC—How WIC Helps. Available online: https://www.fns.usda.gov/wic/about-wic-how-wic-helps (accessed on 2 January 2018).
- Organisation for Economic Co-operation and Development (OECD). Health at a Glance 2015: OECD Indicators; OECD: Paris, France, 2015. [Google Scholar]
- The Royal Australian College of General Practitioners (RACGP). Guidelines for Preventive Activities in General Practice, 9th ed.; RACGP: Melbourne, Australia, 2016. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Available online: https://www.cdc.gov/ (accessed on 2 January 2018).
- Segal, L.; Opie, R.S. A nutrition strategy to reduce the burden of diet related disease: Access to dietician services must complement population health approaches. Front. Pharmacol. 2015, 6, 160. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Capewell, S. Proportion of the decline in cardiovascular mortality disease due to prevention versus treatment: Public health versus clinical care. Annu. Rev. Public Health 2011, 32, 5–22. [Google Scholar] [CrossRef] [PubMed]
- Langan, R.C.; Zawistoski, K.J. Update on vitamin B12 deficiency. Am. Fam. Physician 2011, 83, 1425–1430. [Google Scholar] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Endocrine, S. Evaluation, treatment, and prevention of vitamin D deficiency: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Short, M.W.; Domagalski, J.E. Iron deficiency anemia: Evaluation and management. Am. Fam. Physician 2013, 87, 98–104. [Google Scholar] [PubMed]
- Eck, L.M. Should family physicians routinely screen for vitamin D deficiency? Yes: Targeted screening in at-risk populations is prudent. Am. Fam. Physician 2013, 87, od1. [Google Scholar] [PubMed]
- Lachner, C.; Martin, C.; John, D.; Nekkalapu, S.; Sasan, A.; Steinle, N.; Regenold, W.T. Older adult psychiatric inpatients with non-cognitive disorders should be screened for vitamin B12 deficiency. J. Nutr. Health Aging 2014, 18, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Prüss-Üstün, A.; Campbell-Lendrum, D.; Corvalán, C.; Woodward, A. The Global Burden of Disease Concept; WHO Environmental Burden of Disease Series; World Health Organization (WHO): Geneva, Switzerland, 2003; Volume 1. [Google Scholar]
- The Royal Children’s Hospital Melbourne, Centre for Community Child Health. Early Childhood and the Lifecourse. Policy brief No 1. 2006. Available online: https://rch.org.au/ccch/policybrief (accessed on 2 January 2018).
- Bartley, K.A.; Underwood, B.A.; Deckelbaum, R.J. A life cycle micronutrient perspective for women’s health. Am. J. Clin. Nutr. 2005, 81, 1188S–1193S. [Google Scholar] [PubMed]
- Ferguson, L.R.; Eck, P.; Simopoulos, A.P.; Gillies, P.J.; Vanden Heuvel, J.P. Section 1: Examples of some key nutrient-gene interactions. In Nutrigenomics and Nutrigenetics in Functional Foods and Personalized Nutrition; Ferguson, L.R., Ed.; CRC Press: Boca Raton, FL, USA, 2016; pp. 3–118. [Google Scholar]
- Murphy, M.M.; Fernandez-Ballart, J.D. Homocysteine in pregnancy. Adv. Clin. Chem. 2011, 53, 105–137. [Google Scholar]
- Teucher, B.; Olivares, M.; Cori, H. Enhancers of iron absorption: Ascorbic acid and other organic acids. Int. J. Vitam. Nutr. Res. 2004, 74, 403–419. [Google Scholar] [CrossRef] [PubMed]
- Anadón, A.; Martínez-Larrañaga, M.R.; Ares, I.; Aránzazu Martínez, M. Interactions between nutraceuticals/nutrients and therapeutic drugs. In Nutraceuticals, Efficacy, Safety and Toxicity; Gupta, R.C., Ed.; Academic Press: Hopkinsville, KY, USA, 2016; pp. 855–874. [Google Scholar]
- Sultan, S.; Jahangir, A. Drug–nutrient interactions in the elderly. In Molecular Basis of Nutrition and Aging, 2nd ed.; Malavolta, M., Mocchegiani, E., Eds.; Academic Press: San Diego, CA, USA, 2016; pp. 73–107. [Google Scholar]
- European Food Safety Authority (EFSA). Vitamin D and risk of falling. EFSA J. 2011, 9, 2382. [Google Scholar]
- Field, M.S.; Stover, P.J. Safety of folic acid. Ann. N. Y. Acad. Sci. 2017. [Google Scholar] [CrossRef] [PubMed]
- Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. N. Engl. J. Med. 1994, 330, 1029–1035. [Google Scholar]
- Omenn, G.S.; Goodman, G.E.; Thornquist, M.D.; Balmes, J.; Cullen, M.R.; Glass, A.; Keogh, J.P.; Meyskens, F.L., Jr.; Valanis, B.; Williams, J.H., Jr.; et al. Risk factors for lung cancer and for intervention effects in caret, the beta-carotene and retinol efficacy trial. J. Natl. Cancer Inst. 1996, 88, 1550–1559. [Google Scholar] [CrossRef] [PubMed]
- Hennekens, C.H.; Buring, J.E.; Manson, J.E.; Stampfer, M.; Rosner, B.; Cook, N.R.; Belanger, C.; LaMotte, F.; Gaziano, J.M.; Ridker, P.M.; et al. Lack of effect of long-term supplementation with beta carotene on the incidence of malignant neoplasms and cardiovascular disease. N. Engl. J. Med. 1996, 334, 1145–1149. [Google Scholar] [CrossRef] [PubMed]
- Bushra, R.; Aslam, N.; Khan, A.Y. Food-drug interactions. Oman Med. J. 2011, 26, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Lonnerdal, B. Calcium and iron absorption–mechanisms and public health relevance. Int. J. Vitam. Nutr. Res. 2010, 80, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Santiago, P. Ferrous versus ferric oral iron formulations for the treatment of iron deficiency: A clinical overview. Sci. World J. 2012, 2012, 846824. [Google Scholar] [CrossRef] [PubMed]
- Wong, L.; Smith, S.; Gilstrop, M.; Derman, R.; Auerbach, S.; London, N.; Lenowitz, S.; Bahrain, H.; McClintock, J.; Auerbach, M. Safety and efficacy of rapid (1000 mg in 1 h) intravenous iron dextran for treatment of maternal iron deficient anemia of pregnancy. Am. J. Hematol. 2016, 91, 590–593. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Kelsberg, G.; Safranek, S. Clinical inquiry: Is high-dose oral B12 a safe and effective alternative to a B12 injection? J. Fam. Pract. 2012, 61, 162–163. [Google Scholar] [PubMed]
- Masucci, L.; Goeree, R. Vitamin B12 intramuscular injections versus oral supplements: A budget impact analysis. Ont. Health Technol. Assess. Ser. 2013, 13, 1–24. [Google Scholar] [PubMed]
- Lee, R.D.; Nieman, D.C. Nutritional Assessment, 6th ed.; McGraw Hill Higher Education: New York, NY, USA, 2010. [Google Scholar]
- Klingler, M.; Koletzko, B. Novel methodologies for assessing omega-3 fatty acid status—A systematic review. Br. J. Nutr. 2012, 107 (Suppl. 2), S53–S63. [Google Scholar] [CrossRef] [PubMed]
- Goddard, A.F.; James, M.W.; McIntyre, A.S.; Scott, B.B.; British Society of Gastroenterology. Guidelines for the management of iron deficiency anaemia. Gut 2011, 60, 1309–1316. [Google Scholar] [CrossRef] [PubMed]
- Vemulapati, S.; Rey, E.; O’Dell, D.; Mehta, S.; Erickson, D. A quantitative point-of-need assay for the assessment of vitamin D3 deficiency. Sci. Rep. 2017, 7, 14142. [Google Scholar] [CrossRef] [PubMed]
- McDonagh, M.; Blazina, I.; Dana, T.; Cantor, A.; Bougatsos, C. Routine Iron Supplementation and Screening for Iron Deficiency Anemia in Children Ages 6 to 24 Months: A Systematic Review to Update the U.S. Preventive Services Task Force Recommendation; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2015. [Google Scholar]
- Zhang, Z.; Pereira, S.L.; Luo, M.; Matheson, E.M. Evaluation of blood biomarkers associated with risk of malnutrition in older adults: A systematic review and meta-analysis. Nutrients 2017, 9, 829. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute (NIH). Epidemiology and Genomics Research Program: Register of Validated Short Dietary Assessment Instruments. Available online: https://epi.grants.cancer.gov/diet/shortreg/register.php (accessed on 2 January 2018).
- Asaad, G.; Sadegian, M.; Lau, R.; Xu, Y.; Soria-Contreras, D.C.; Bell, R.C.; Chan, C.B. The reliability and validity of the perceived dietary adherence questionnaire for people with type 2 diabetes. Nutrients 2015, 7, 5484–5496. [Google Scholar] [CrossRef] [PubMed]
- Hamirudin, A.H.; Charlton, K.; Walton, K.; Bonney, A.; Albert, G.; Hodgkins, A.; Potter, J.; Milosavljevic, M.; Dalley, A. ‘We are all time poor’. Is routine nutrition screening of older patients feasible? Aust. Fam. Physician 2013, 42, 321–326. [Google Scholar] [PubMed]
- Lubloy, A. Factors affecting the uptake of new medicines: A systematic literature review. BMC Health Serv. Res. 2014, 14, 469. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence (NICE). An Economic Evaluation of Interventions to Improve the Uptake of Vitamin D Supplements in England and Wales. Available online: https://www.nice.org.uk/guidance/ph56/documents/economic-evaluation-report2 (accessed on 2 January 2018).
- Berg, R.L.; Shaw, G.R. Laboratory evaluation for vitamin B12 deficiency: The case for cascade testing. Clin Med. Res. 2013, 11, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.; Lindemann, M.; Colligs, A.; Snowball, C. Economic burden of neural tube defects and impact of prevention with folic acid: A literature review. Eur. J. Pediatr. 2011, 170, 1391–1400. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, G.; Moreira, P.; Rosáriode, R.; Araújoa, A.; Teixeira, V.H.; Lopes, O.; Moreirag, A.; Padrãoa, P. Adherence to the mediterranean diet in children: Is it associated with economic cost? Porto Biomed. J. 2017, 2, 115–119. [Google Scholar] [CrossRef]
- Lammers, M.; Kok, L. Cost-Benefit Analysis of Dietary Treatment; Dutch Association of Dietitians: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Dalziel, K.; Segal, L. Time to give nutrition interventions a higher profile: Cost-effectiveness of 10 nutrition interventions. Health Promot. Int. 2007, 22, 271–283. [Google Scholar] [CrossRef] [PubMed]
- Rukuni, R.; Knight, M.; Murphy, M.F.; Roberts, D.; Stanworth, S.J. Screening for iron deficiency and iron deficiency anaemia in pregnancy: A structured review and gap analysis against UK national screening criteria. BMC Pregnancy Childbirth 2015, 15, 269. [Google Scholar] [CrossRef] [PubMed]
- Gubbins, P.O.; Klepser, M.E.; Dering-Anderson, A.M.; Bauer, K.A.; Darin, K.M.; Klepser, S.; Matthias, K.R.; Scarsi, K. Point-of-care testing for infectious diseases: Opportunities, barriers, and considerations in community pharmacy. J. Am. Pharm. Assoc. (2003) 2014, 54, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Tapley, A.; Magin, P.; Morgan, S.; Henderson, K.; Scott, J.; Thomson, A.; Spike, N.; McArthur, L.; van Driel, M.; McElduff, P.; et al. Test ordering in an evidence free zone: Rates and associations of australian general practice trainees’ vitamin D test ordering. J. Eval. Clin. Pract. 2015, 21, 1151–1156. [Google Scholar] [CrossRef] [PubMed]
- Hynes, V. The trend toward self-diagnosis. Can. Med. Assoc. J. CMAJ 2013, 185, E149–E150. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bruins, M.J.; Bird, J.K.; Aebischer, C.P.; Eggersdorfer, M. Considerations for Secondary Prevention of Nutritional Deficiencies in High-Risk Groups in High-Income Countries. Nutrients 2018, 10, 47. https://doi.org/10.3390/nu10010047
Bruins MJ, Bird JK, Aebischer CP, Eggersdorfer M. Considerations for Secondary Prevention of Nutritional Deficiencies in High-Risk Groups in High-Income Countries. Nutrients. 2018; 10(1):47. https://doi.org/10.3390/nu10010047
Chicago/Turabian StyleBruins, Maaike J., Julia K. Bird, Claude P. Aebischer, and Manfred Eggersdorfer. 2018. "Considerations for Secondary Prevention of Nutritional Deficiencies in High-Risk Groups in High-Income Countries" Nutrients 10, no. 1: 47. https://doi.org/10.3390/nu10010047
APA StyleBruins, M. J., Bird, J. K., Aebischer, C. P., & Eggersdorfer, M. (2018). Considerations for Secondary Prevention of Nutritional Deficiencies in High-Risk Groups in High-Income Countries. Nutrients, 10(1), 47. https://doi.org/10.3390/nu10010047







