An Integrated Field and Remote Sensing Method for Mapping Seagrass Species, Cover, and Biomass in Southern Thailand
Abstract
:1. Introduction
2. Materials and Methods
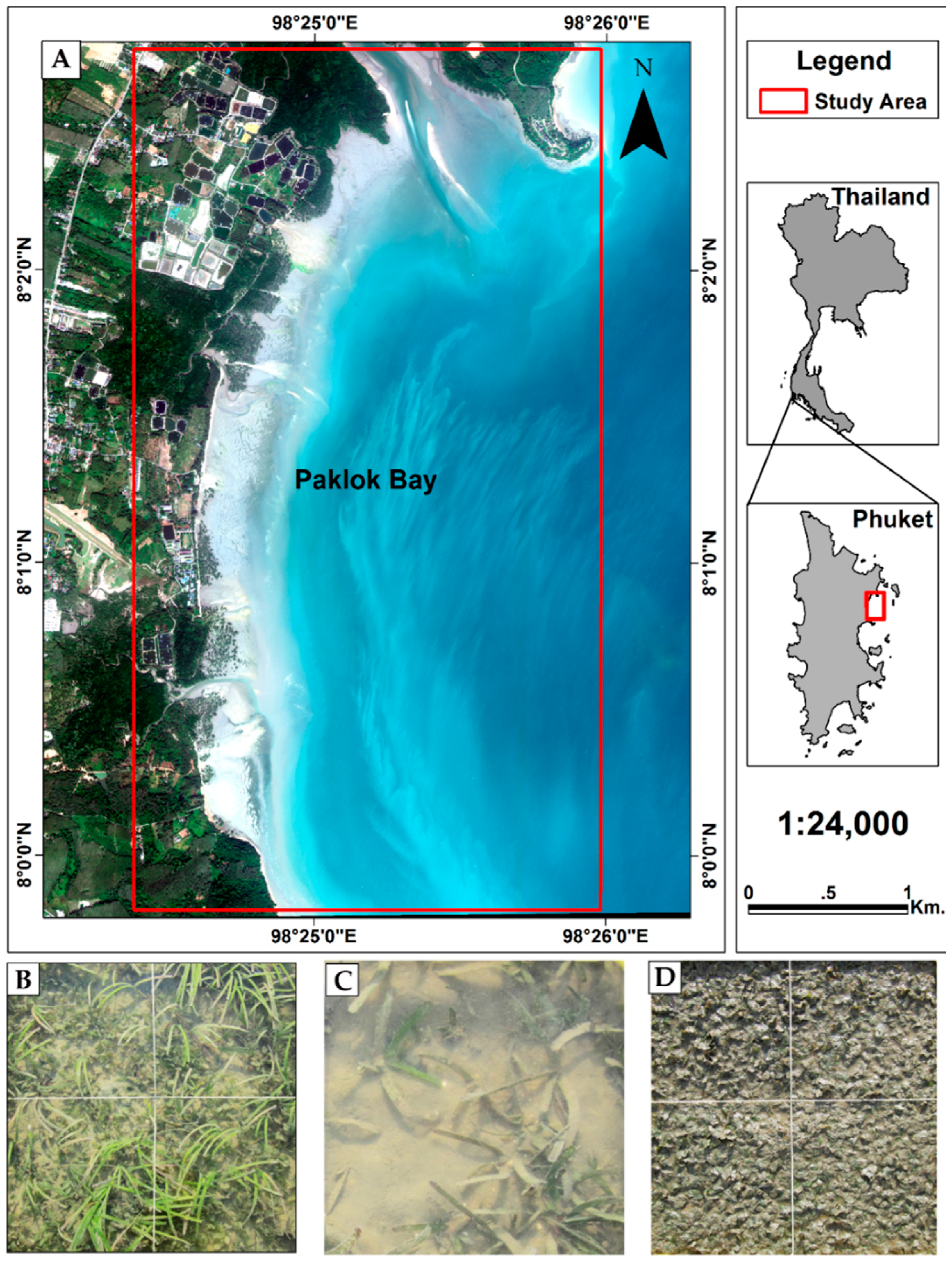
2.1. Study Site
2.2. Field Data Collection
2.3. Remote-Sensing Data and Its Processing
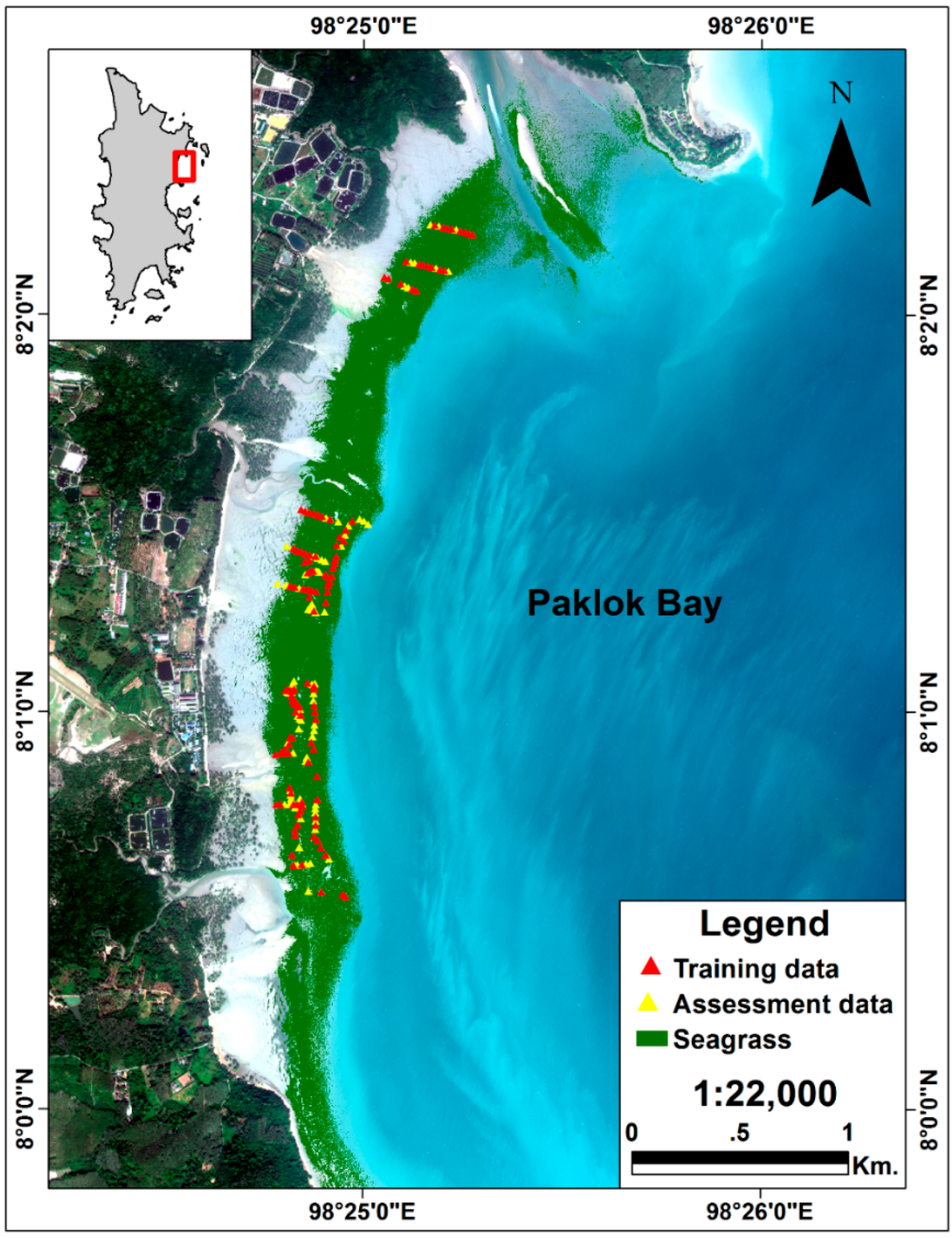
2.4. Spatial Distribution of Seagrass Areas
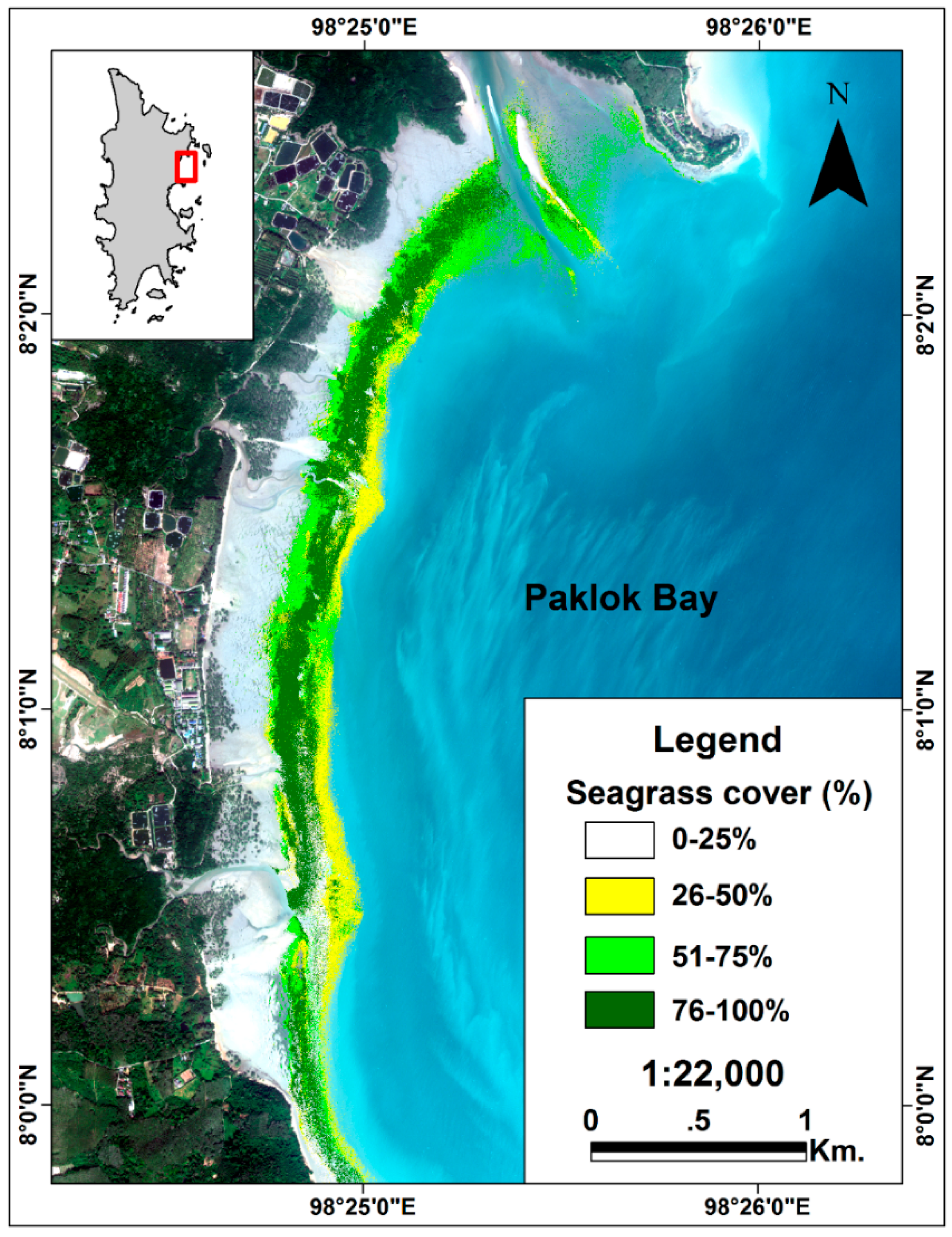
2.5. Percentage Cover and Seagrass Species Mapping
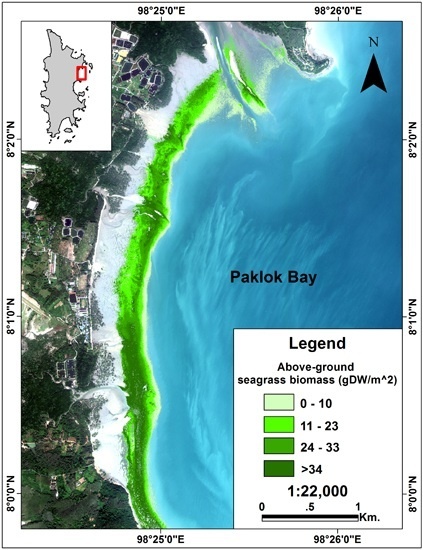
2.6. Above-Ground Biomass Mapping
2.7. Accuracy Assessments
3. Results
3.1. Spatial Distribution of Seagrass Area
3.2. Percentage Cover Mapping
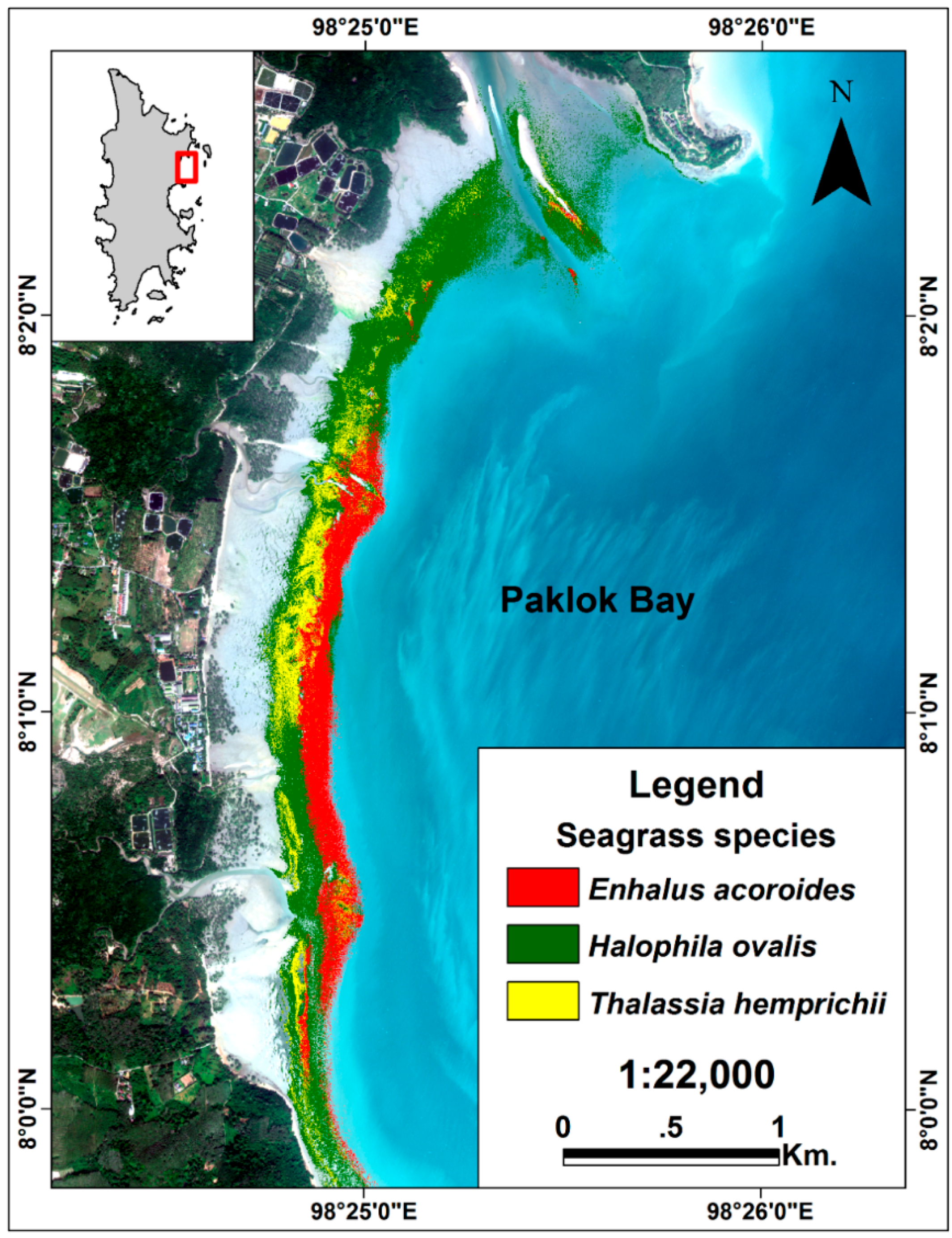
3.3. Seagrass Species Mapping
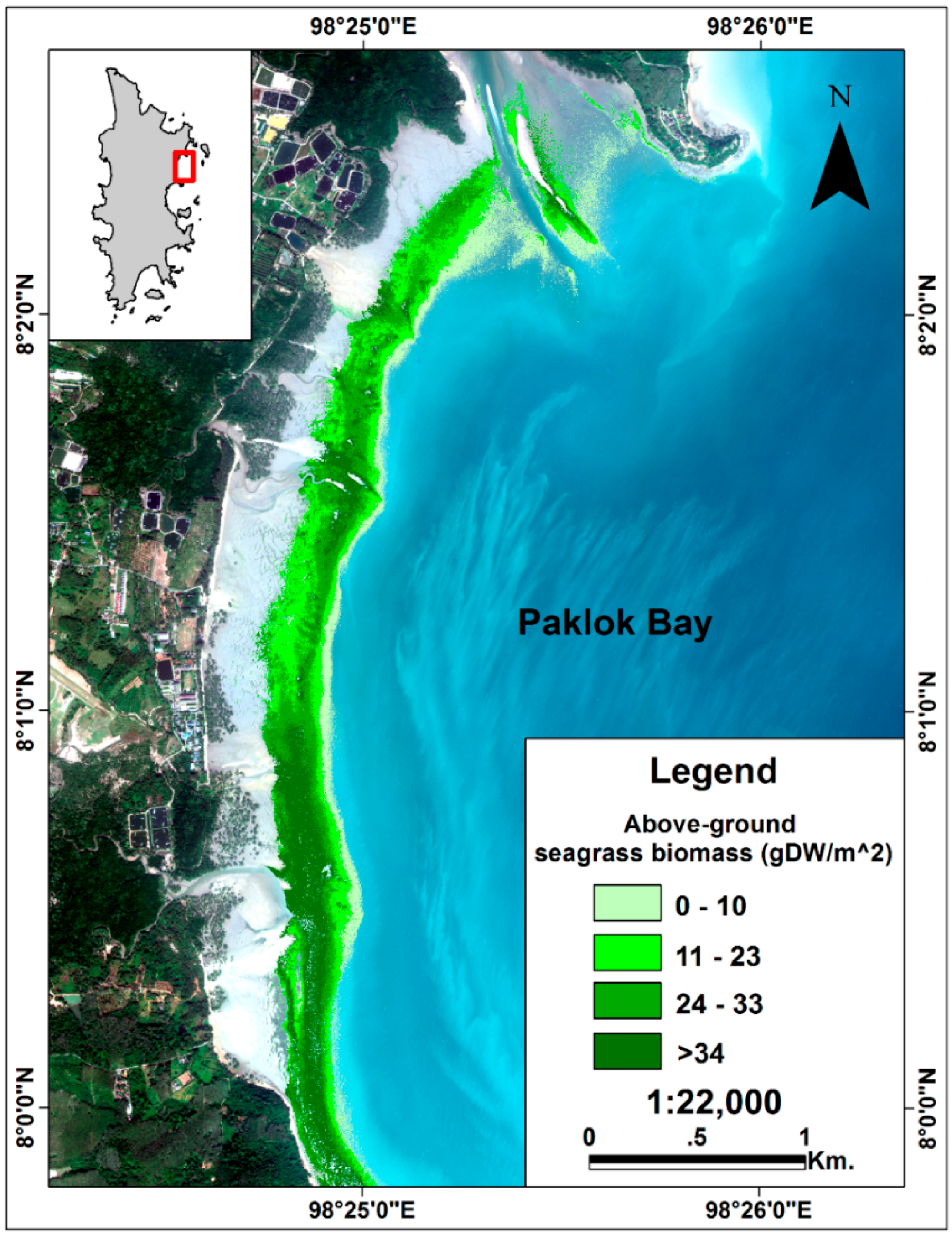
3.4. Above-Ground Biomass Mapping
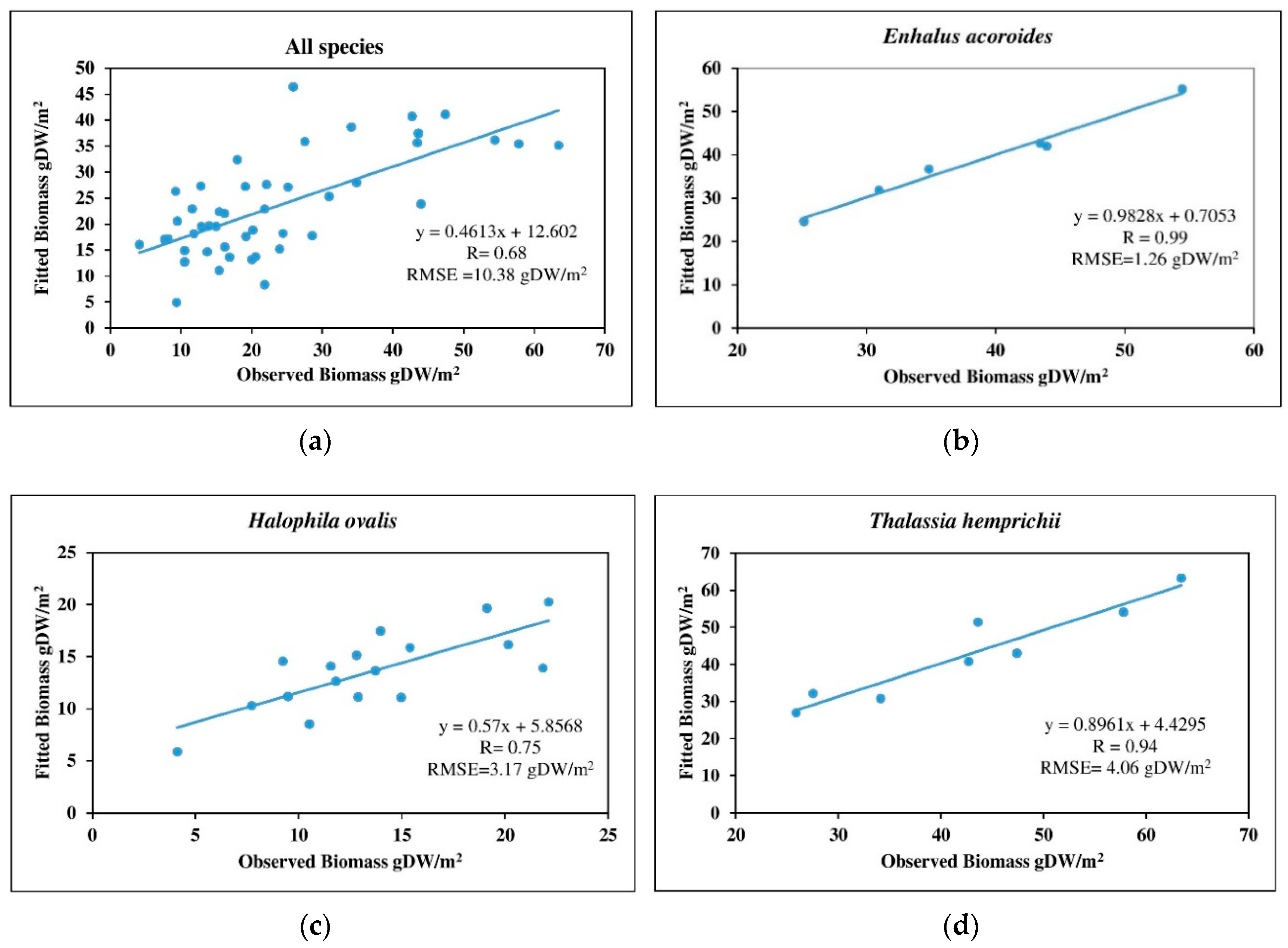
3.4.1. SMLR Models with Spectral Bands as Independent Variables
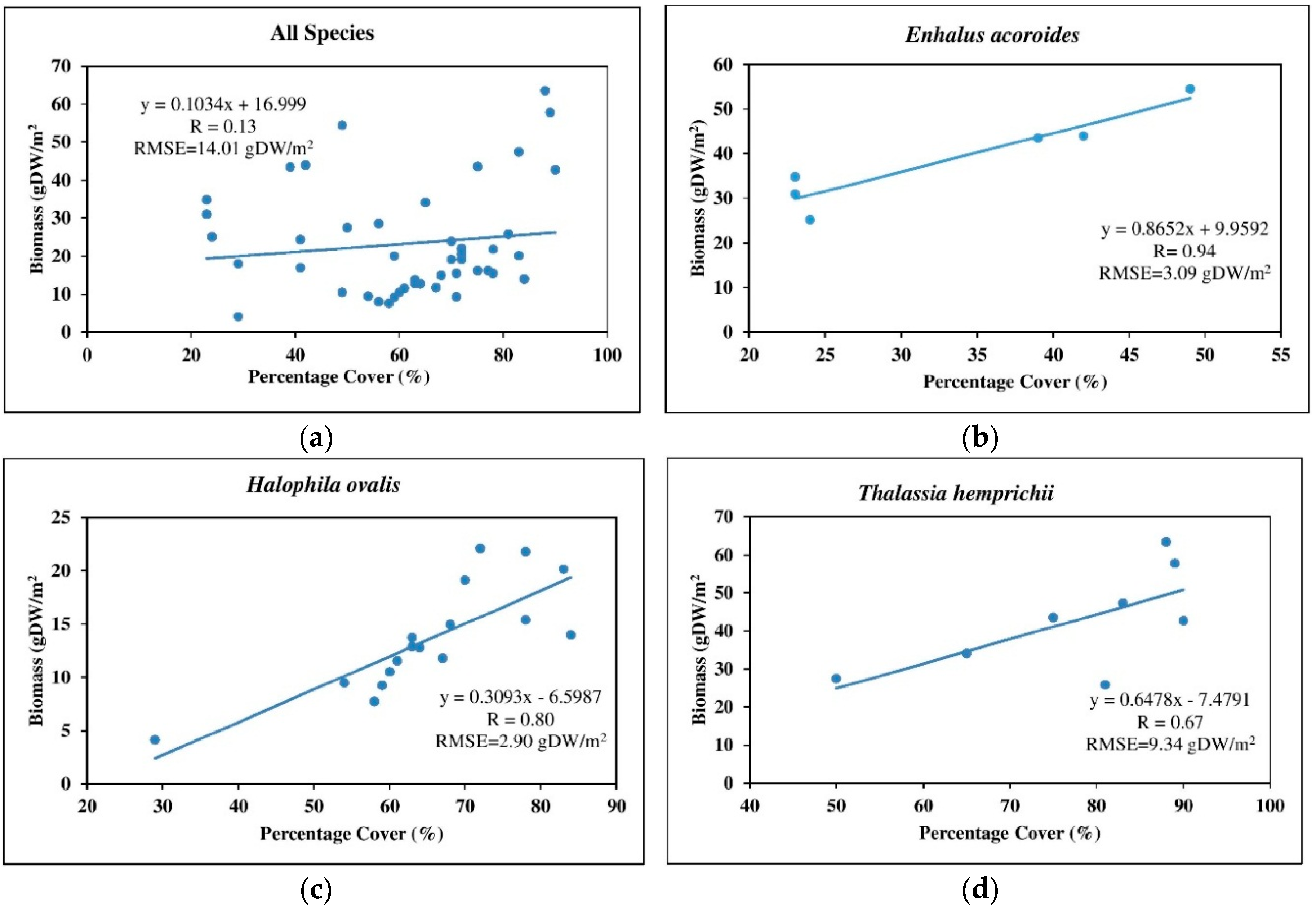
3.4.2. Simple Linear Regression with Percentage Cover as the Independent Variable
4. Discussion
4.1. Spatial Distribution of Seagrass Area
4.2. Percentage Cover Mapping
4.3. Seagrass Species Mapping
4.4. Above-Ground Biomass Mapping
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| RMSE | Root Mean Square Error |
| WV-2 | Worldview-2 |
| RTK | Real Time Kinematic |
| GNSS | Global Navigation Satellite System |
| CPCe | Coral Point Count with Excel extensions |
| UTM | Universal Transverse Mercator |
| GISTDA | Geo-Informatics and Space Technology Development Agency (Public Organization) |
| MLC | Maximum Likelihood Classification |
| SMLR | Stepwise Multiple Linear Regression |
| SLR | Simple Linear Regression |
| NIR | Near infrared |
| OA | Overall Accuracy |
| UA | User’s Accuracy |
| PA | Producer’s Accuracy Multidisciplinary Digital Publishing Institute |
References
- Larkum, A.; Orth, R.J.; Duarte, C. Seagrasses: Biology, Ecology and Conservation; Springer: Dordrecht, The Netherlands, 2006. [Google Scholar]
- Hossain, M.S.; Bujang, J.S.; Zakaria, M.H.; Hashim, M. The application of remote sensing to seagrass ecosystems: An overview and future research prospects. Int. J. Remote Sens. 2015, 36, 61–114. [Google Scholar] [CrossRef]
- Knudby, A.; Nordlund, L. Remote sensing of seagrasses in a patchy multi-species environment. Int. J. Remote Sens. 2011, 32, 2227–2244. [Google Scholar] [CrossRef]
- Orth, R.J.; Heck, K.L.; van Montfrans, J. Faunal communities in seagrass beds: A review of the influence of plant structure and prey characteristics on predator-prey relationships. Estuaries 1984, 7, 339–350. [Google Scholar] [CrossRef]
- Roelfsema, C.M.; Phinn, S.R.; Udy, N.; Maxwell, P. An integrated field and remote sensing approach for mapping seagrass cover, Moreton Bay, Australia. J. Spat. Sci. 2009, 54, 45–62. [Google Scholar] [CrossRef]
- Van der Heide, T.; van Nes, E.H.; Geerling, G.W.; Smolders, A.J.; Bouma, T.J.; van Katwijk, M.M. Positive feedbacks in seagrass ecosystems: Implications for success in conservation and restoration. Ecosystems 2007, 10, 1311–1322. [Google Scholar] [CrossRef]
- Green, E.P.; Short, F.T. World Atlas of Seagrasses; University of California Press: Berkeley, CA, USA, 2003. [Google Scholar]
- Ferwerda, J.G.; de Leeuw, J.; Atzberger, C.; Vekerdy, Z. Satellite-based monitoring of tropical seagrass vegetation: Current techniques and future developments. Hydrobiologia 2007, 591, 59–71. [Google Scholar] [CrossRef]
- Waycott, M.; Duarte, C.M.; Carruthers, T.J.; Orth, R.J.; Dennison, W.C.; Olyarnik, S.; Calladine, A.; Fourqurean, J.W.; Heck, K.L.; Hughes, A.R. Accelerating loss of seagrasses across the globe threatens coastal ecosystems. Proc. Natl. Acad. Sci. USA 2009, 106, 12377–12381. [Google Scholar] [CrossRef] [PubMed]
- Roelfsema, C.M.; Lyons, M.; Kovacs, E.M.; Maxwell, P.; Saunders, M.I.; Samper-Villarreal, J.; Phinn, S.R. Multi-temporal mapping of seagrass cover, species and biomass: A semi-automated object based image analysis approach. Remote Sens. Environ. 2014, 150, 172–187. [Google Scholar] [CrossRef]
- Armstrong, R.A. Remote sensing of submerged vegetation canopies for biomass estimation. Int. J. Remote Sens. 1993, 14, 621–627. [Google Scholar] [CrossRef]
- Lyons, M.; Phinn, S.; Roelfsema, C. Integrating Quickbird multi-spectral satellite and field data: Mapping bathymetry, seagrass cover, seagrass species and change in Moreton Bay, Australia in 2004 and 2007. Remote Sens. 2011, 3, 42–64. [Google Scholar] [CrossRef]
- Howari, F.M.; Jordan, B.R.; Bouhouche, N.; Wyllie-Echeverria, S. Field and remote-sensing assessment of mangrove forests and seagrass beds in the Northwestern part of the United Arab Emirates. J. Coast. Res. 2009, 25, 48–56. [Google Scholar] [CrossRef]
- Ferguson, R.L.; Wood, L.L.; Graham, D.B. Monitoring spatial change in seagrass habitat with aerial photography. Photogramm. Eng. Remote Sens. 1993, 59, 1033–1038. [Google Scholar]
- Barillé, L.; Robin, M.; Harin, N.; Bargain, A.; Launeau, P. Increase in seagrass distribution at Bourgneuf Bay (France) detected by spatial remote sensing. Aquat. Bot. 2010, 92, 185–194. [Google Scholar] [CrossRef]
- Kaewsrikhaw, R.; Ritchie, R.J.; Prathep, A. Variations of tidal exposures and seasons on growth, morphology, anatomy and physiology of the seagrass Halophila ovalis (R.Br.) Hook. f. in a seagrass bed in Trang Province, Southern Thailand. Aquat. Bot. 2016, 130, 11–20. [Google Scholar] [CrossRef]
- Gullström, M.; de la Torre Castro, M.; Bandeira, S.O.; Björk, M.; Dahlberg, M.; Kautsky, N.; Rönnbäck, P.; Öhman, M.C. Seagrass ecosystems in the western Indian Ocean. AMBIO J. Hum. Environ. 2002, 31, 588–596. [Google Scholar] [CrossRef]
- Phinn, S.; Roelfsema, C.; Dekker, A.; Brando, V.; Anstee, J. Mapping seagrass species, cover and biomass in shallow waters: An assessment of satellite multi-spectral and airborne hyper-spectral imaging systems in Moreton Bay (Australia). Remote Sens. Environ. 2008, 112, 3413–3425. [Google Scholar] [CrossRef]
- Wolter, P.T.; Johnston, C.A.; Niemi, G.J. Mapping submergent aquatic vegetation in the US Great Lakes using Quickbird satellite data. Int. J. Remote Sens. 2005, 26, 5255–5274. [Google Scholar] [CrossRef]
- Adulyanukosol, K.; Poovachiranon, S. Dugong (Dugong dugon) and seagrass in Thailand: Present status and future challenges. In Proceedings of the 3rd International Symposium SEASTAR and Asian Biologging Science, Bangkok, Thailand, 13–14 December 2006; pp. 41–50.
- Poovachiranon, S.; Chansang, H. Community structure and biomass of seagrass beds in the Andaman Sea. I. Mangrove-associated seagrass beds. Phuket Mar. Biol. Cent. Res. Bull. 1994, 59, 53–64. [Google Scholar]
- Department of Marine and Coastal Resources. The Survey and Assessment of the Status and Potential of Marine and Coastal Resources: Coral and Seagrass 2014; Department of Marine and Coastal Resources: Bangkok, Thailand, 2014. [Google Scholar]
- El-Rabbany, A. Introduction to GPS: The Global Positioning System; Artech House: Norwood, MA, USA, 2002. [Google Scholar]
- Wongkamhaeng, K.; Paphavasit, N.; Bussarawit, S.; Nabhitabhata, J. Seagrass gammarid amphipods of Libong Island, Trang Province, Thailand. Nat. Hist. J. Chulalongkorn Univ. 2009, 9, 69–83. [Google Scholar]
- English, S.A.; Baker, V.J.; Wilkinson, C.R. Survey Manual for Tropical Marine Resources; Australian Institute of Marine Science: Townsville, QLD, Austrilia, 1997. [Google Scholar]
- Kohler, K.E.; Gill, S.M. Coral point count with excel extensions (CPCe): A visual basic program for the determination of coral and substrate coverage using random point count methodology. Comput. Geosci. 2006, 32, 1259–1269. [Google Scholar] [CrossRef]
- Foody, G.M.; Boyd, D.S.; Cutler, M.E. Predictive relations of tropical forest biomass from Landsat TM data and their transferability between regions. Remote Sens. Environ. 2003, 85, 463–474. [Google Scholar] [CrossRef]
- ERDAS. ERDAS Field Guide; Erdas Inc.: Atlanta, GA, USA, 1999. [Google Scholar]
- Paola, J.D.; Schowengerdt, R.A. A detailed comparison of backpropagation neural network and maximum-likelihood classifiers for urban land use classification. IEEE Trans. Geosci. Remote Sens. 1995, 33, 981–996. [Google Scholar] [CrossRef]
- Congalton, R.G.; Green, K. Assessing the Accuracy of Remotely Sensed Data: Principles and Practices; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Nakaoka, M.; Supanwanid, C. Quantitative estimation of the distribution and biomass of seagrass at Haad Chao Mai National Park, Trang Province, Thailand. Kasetsart Univ. Fish. Res. Bull. 2000, 22, 10–22. [Google Scholar]
- Short, F.T.; Koch, E.W.; Creed, J.C.; Magalhaes, K.M.; Fernandez, E.; Gaeckle, J.L. SeagrassNet monitoring across the Americas: Case studies of seagrass decline. Mar. Ecol. 2006, 27, 277–289. [Google Scholar] [CrossRef]
- Richards, D.R.; Friess, D.A. Rates and drivers of mangrove deforestation in Southeast Asia, 2000–2012. Proc. Natl. Acad. Sci. USA 2016, 113, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Burkholder, J.M.; Tomasko, D.A.; Touchette, B.W. Seagrasses and eutrophication. J. Exp. Mar. Biol. Ecol. 2007, 350, 46–72. [Google Scholar] [CrossRef]
| Worldview-2 | Details |
|---|---|
| Date Acquired | 27 December 2013 |
| Time Acquired (Local) | 10:24 |
| Source | Digital Globe |
| Tidal level | −1.5 m |
| Spatial Resolution | 2 m |
| Band ranges | Coastal: 400–450 nm |
| Blue: 450–510 nm | |
| Green: 510–580 nm | |
| Yellow: 585–625 nm | |
| Red: 630–690 nm | |
| Red Edge: 705–745 nm | |
| NIR-1: 770–895 nm | |
| NIR-2: 860–1040 nm |
| Bands Combination | Classification Accuracy | |
|---|---|---|
| OA (%) | Kappa | |
| coastal, green, red edge, NIR-2 | 88.00 | 0.79 |
| coastal, green, yellow, red edge, NIR-2 | 88.67 | 0.80 |
| coastal, green, red, red edge, NIR-1, NIR-2 | 90.67 | 0.84 |
| Classes | Seagrass | Sand | Deep Water | Total | UA (%) |
|---|---|---|---|---|---|
| Seagrass | 78 | 11 | 0 | 89 | 87.64 |
| Sand | 3 | 38 | 0 | 41 | 92.68 |
| Deep Water | 0 | 0 | 20 | 20 | 100.00 |
| Total | 81 | 49 | 20 | 150 | |
| PA (%) | 96.30 | 77.50 | 100.00 |
| Band Combination | Classification Accuracy | |
|---|---|---|
| OA (%) | Kappa | |
| coastal blue, blue, green, red, red-edge | 68.69 | 0.57 |
| coastal blue, green, red, red-edge, NIR-1 | 73.74 | 0.64 |
| coastal blue, blue, green, red, red-edge, NIR-1 | 72.73 | 0.63 |
| Classes | 0%–25% | 26%–50% | 51%–75% | 76%–100% | Total | UA (%) |
|---|---|---|---|---|---|---|
| 0%–25% | 30 | 2 | 0 | 0 | 32 | 93.75 |
| 26%–50% | 0 | 14 | 7 | 2 | 23 | 60.87 |
| 51%–75% | 0 | 1 | 4 | 1 | 6 | 66.67 |
| 76%–100% | 0 | 3 | 10 | 25 | 38 | 65.79 |
| Total | 30 | 20 | 21 | 28 | 99 | |
| PA (%) | 100.00 | 70.00 | 19.05 | 89.29 |
| Bands Combination | Classification Accuracy | |
|---|---|---|
| OA (%) | Kappa | |
| coastal blue, blue, green, red-edge, NIR-2 | 71.15 | 0.56 |
| coastal blue, green, yellow, red-edge, NIR-2 | 75.00 | 0.61 |
| coastal, green, red, red-edge, NIR-1, NIR-2 | 73.10 | 0.59 |
| Classes | Enhalus acoroides | Halophila ovalis | Thalassia hemprichii | Total | UA (%) |
|---|---|---|---|---|---|
| Enhalus acoroides | 19 | 5 | 6 | 30 | 63.33 |
| Halophila ovalis | 0 | 11 | 0 | 11 | 100.00 |
| Thalassia hemprichii | 1 | 1 | 9 | 11 | 81.82 |
| Total | 20 | 17 | 15 | 52 | |
| PA (%) | 95.00 | 64.71 | 60.00 |
| Species | Estimated by Spectral Bands | Estimated by Percentage Cover | |||
|---|---|---|---|---|---|
| Bands | R | RMSE (g·DW/m2) | R | RMSE (g·DW/m2) | |
| All species | green, yellow, and NIR-1 | 0.68 | 10.38 | 0.13 | 14.01 |
| Enhalus acoroides | coastal blue, blue, and yellow | 0.99 | 1.26 | 0.94 | 3.09 |
| Halophila ovalis | coastal blue, blue, yellow, and red-edge | 0.75 | 3.17 | 0.80 | 2.90 |
| Thalassia hemprichii | blue, green, yellow, and red-edge | 0.94 | 4.06 | 0.67 | 9.34 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koedsin, W.; Intararuang, W.; Ritchie, R.J.; Huete, A. An Integrated Field and Remote Sensing Method for Mapping Seagrass Species, Cover, and Biomass in Southern Thailand. Remote Sens. 2016, 8, 292. https://doi.org/10.3390/rs8040292
Koedsin W, Intararuang W, Ritchie RJ, Huete A. An Integrated Field and Remote Sensing Method for Mapping Seagrass Species, Cover, and Biomass in Southern Thailand. Remote Sensing. 2016; 8(4):292. https://doi.org/10.3390/rs8040292
Chicago/Turabian StyleKoedsin, Werapong, Wissarut Intararuang, Raymond J. Ritchie, and Alfredo Huete. 2016. "An Integrated Field and Remote Sensing Method for Mapping Seagrass Species, Cover, and Biomass in Southern Thailand" Remote Sensing 8, no. 4: 292. https://doi.org/10.3390/rs8040292
APA StyleKoedsin, W., Intararuang, W., Ritchie, R. J., & Huete, A. (2016). An Integrated Field and Remote Sensing Method for Mapping Seagrass Species, Cover, and Biomass in Southern Thailand. Remote Sensing, 8(4), 292. https://doi.org/10.3390/rs8040292