Designing Climate-Resilient Marine Protected Area Networks by Combining Remotely Sensed Coral Reef Habitat with Coastal Multi-Use Maps
Abstract
:1. Introduction and Background
2. Data and Methodology
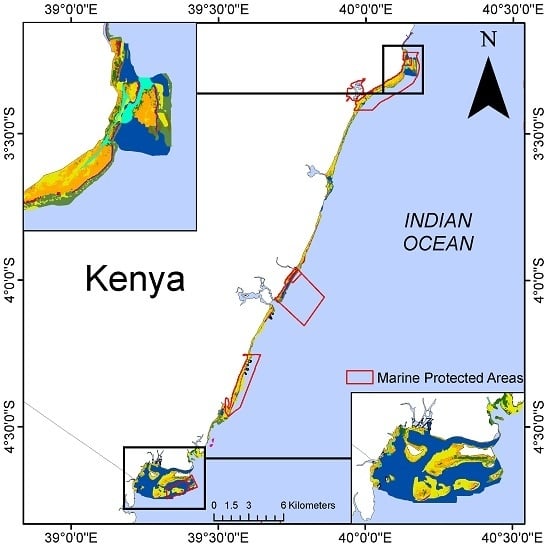
2.1. Kenya Case Study
2.2. Field Sampling for Habitat Mapping
2.3. Satellite Image Analysis
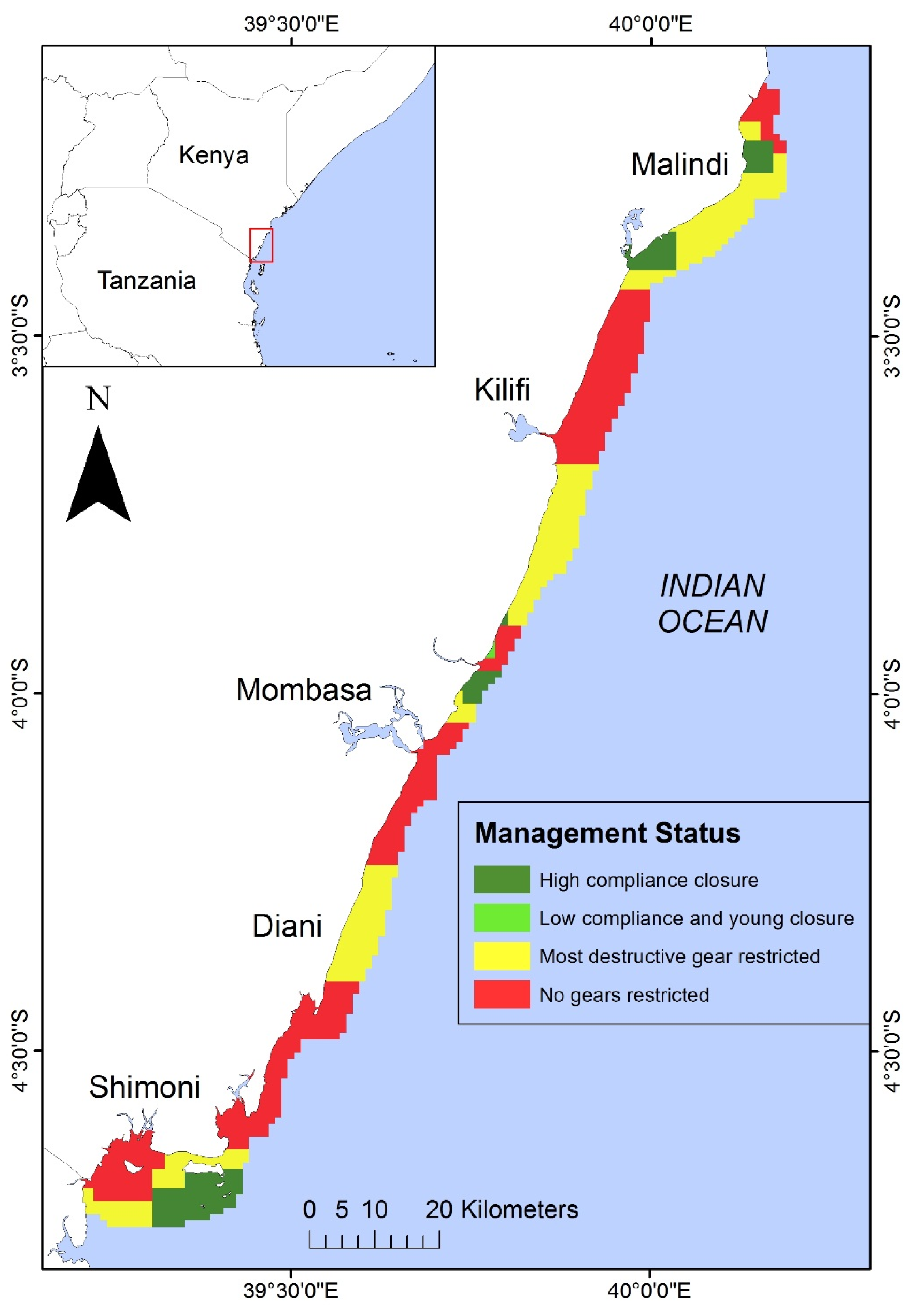
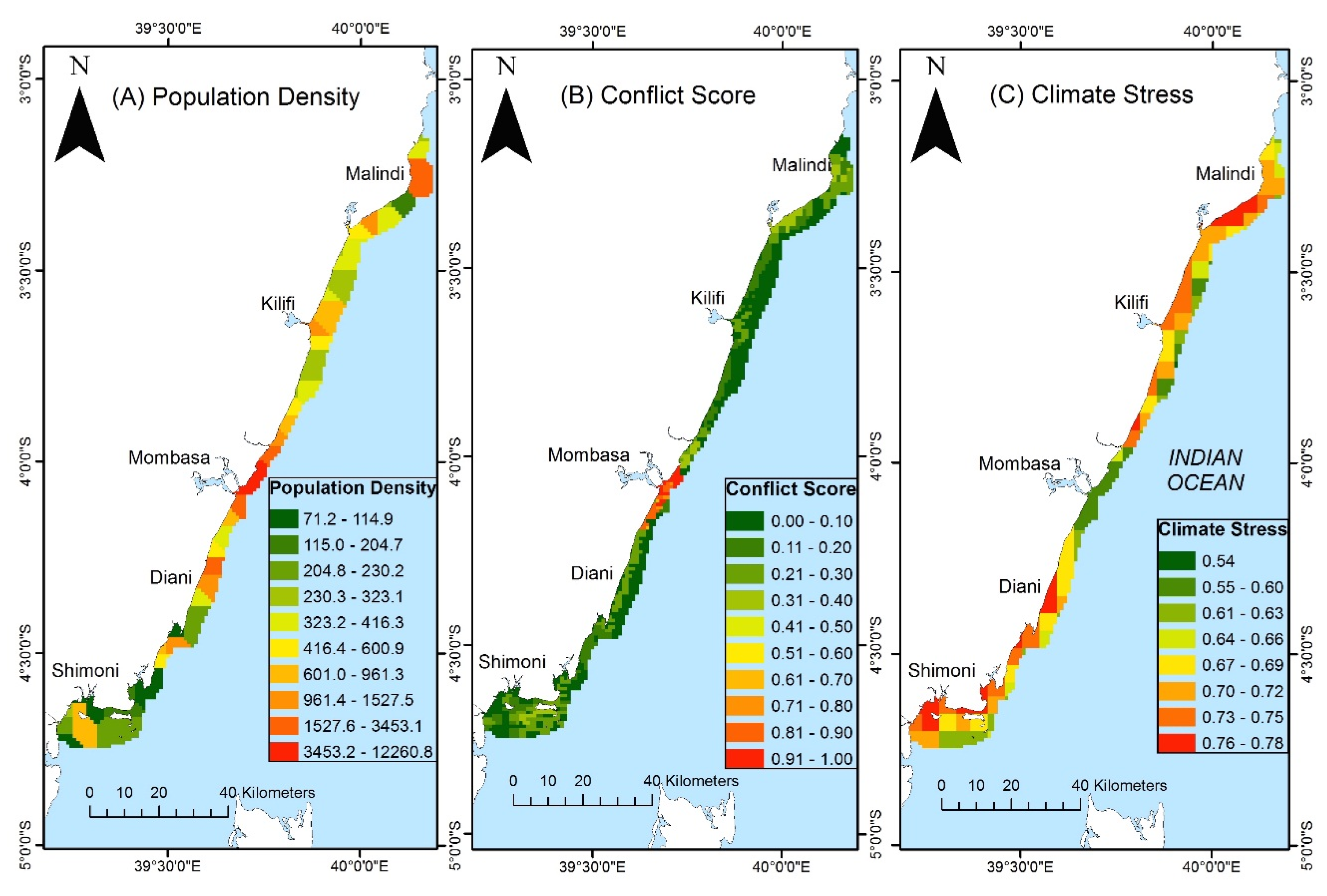
2.4. Mapping Potential Resource Use Conflicts
2.5. Mapping Conservation Priorities
3. Results
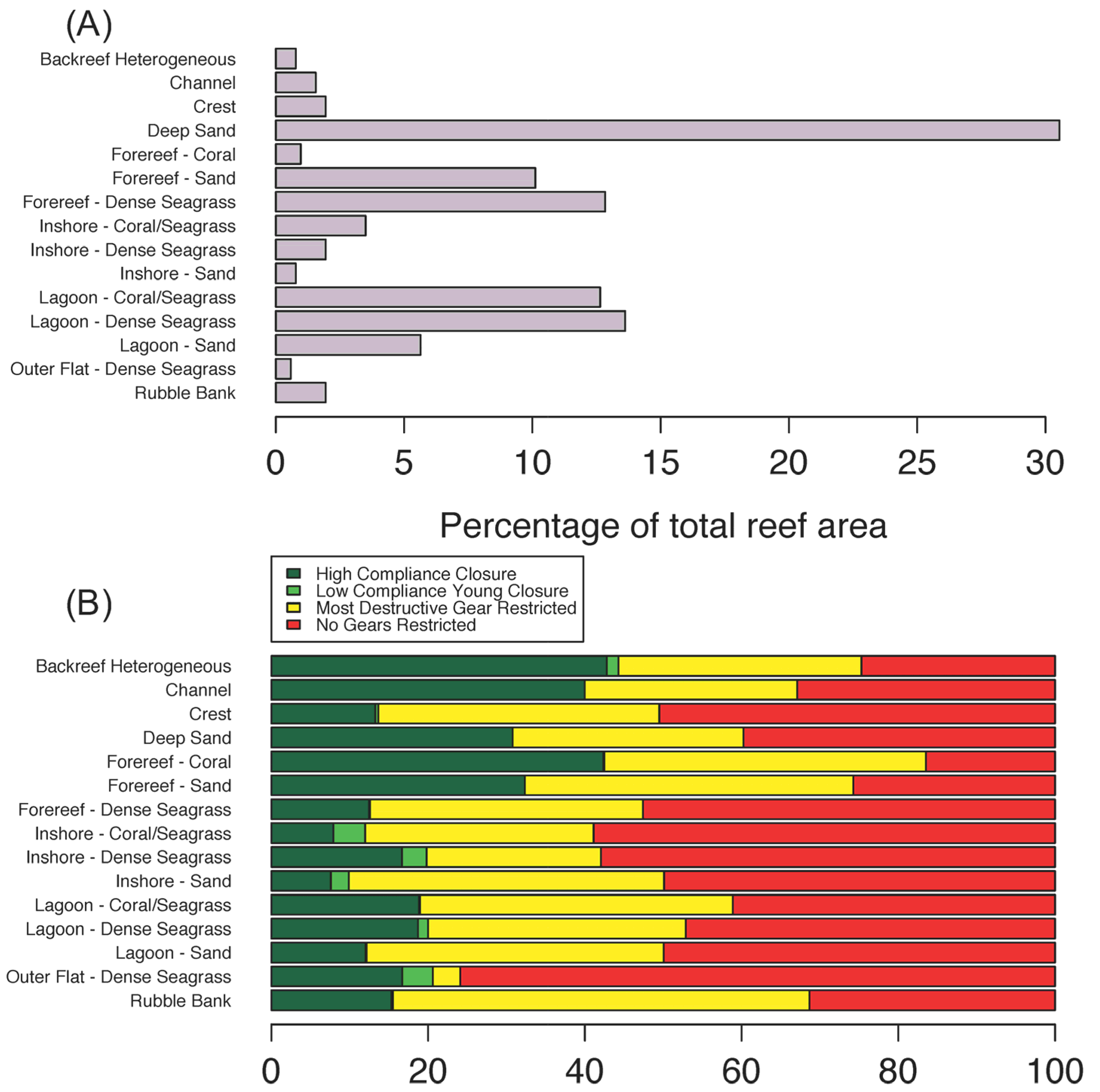
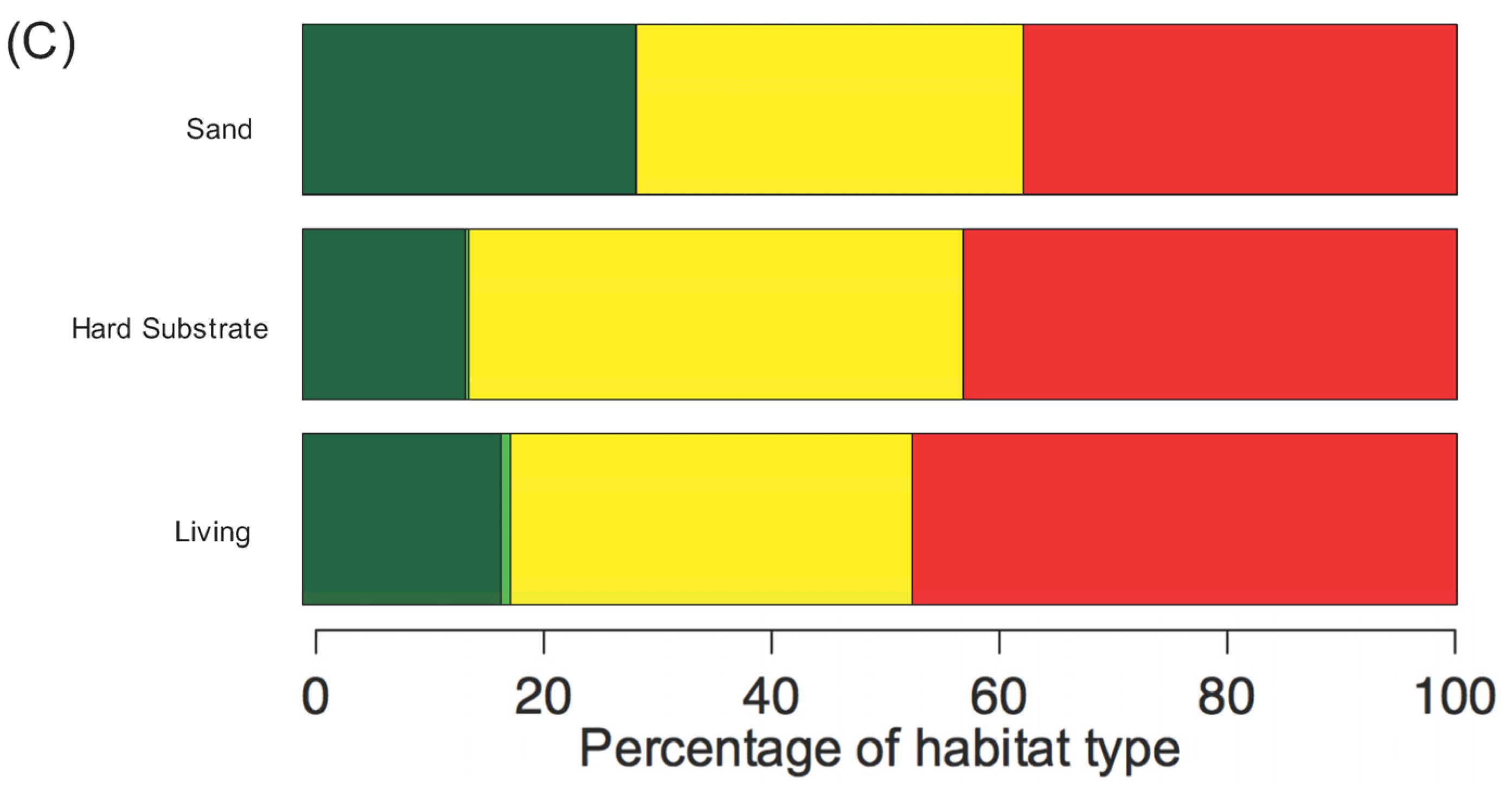
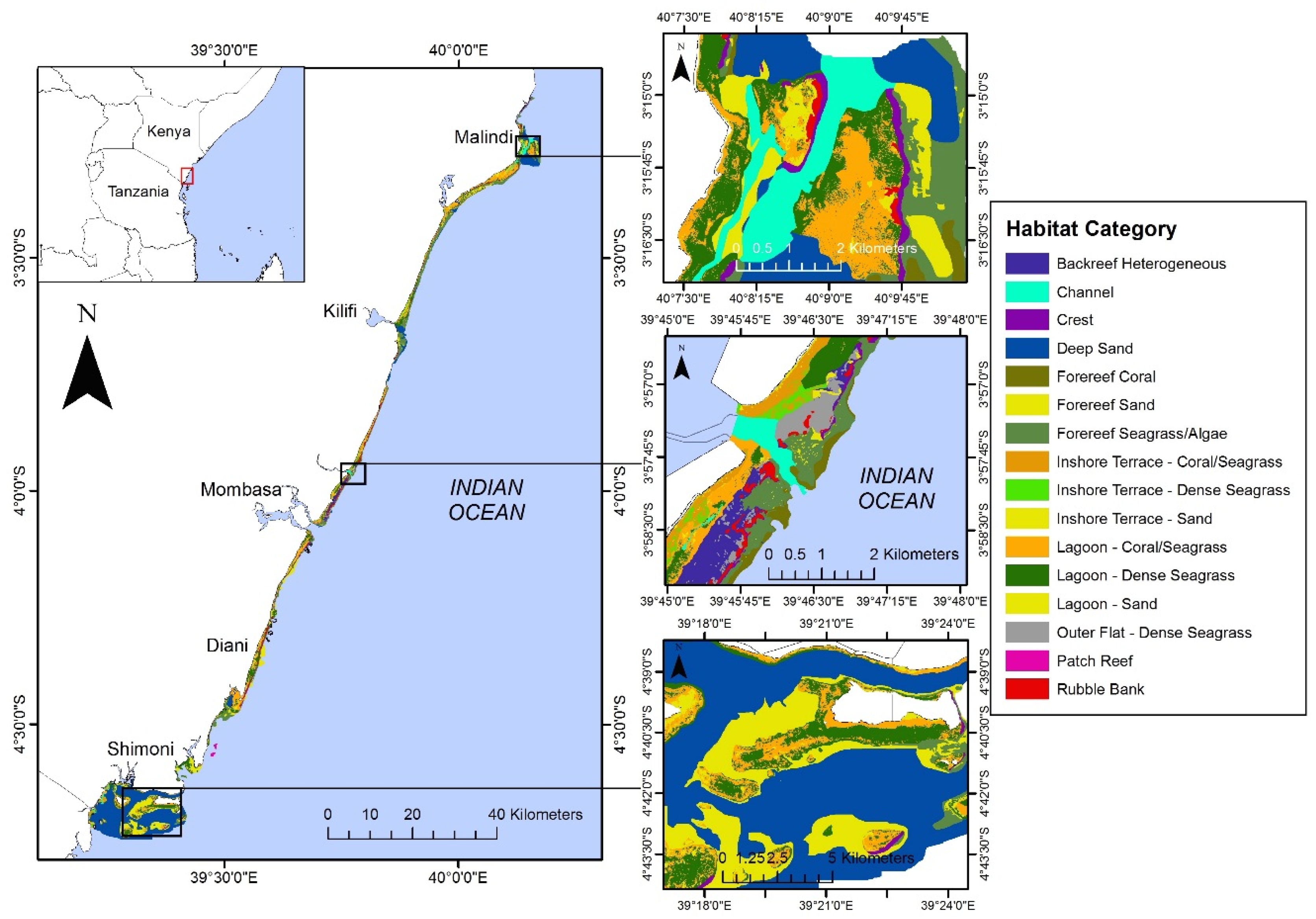
3.1. Current State of the Nearshore Habitats
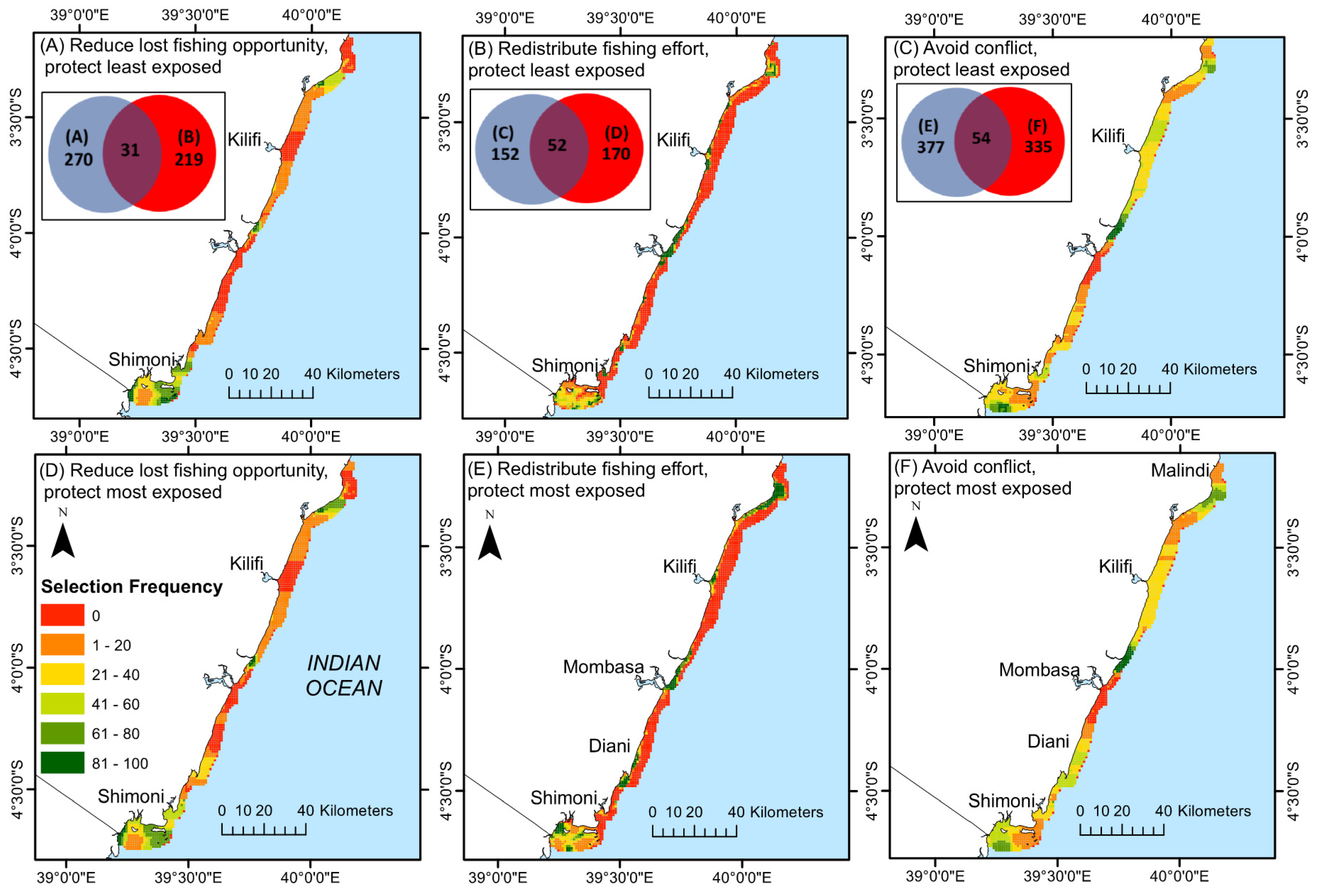
3.2. Mapping Conservation Priorities
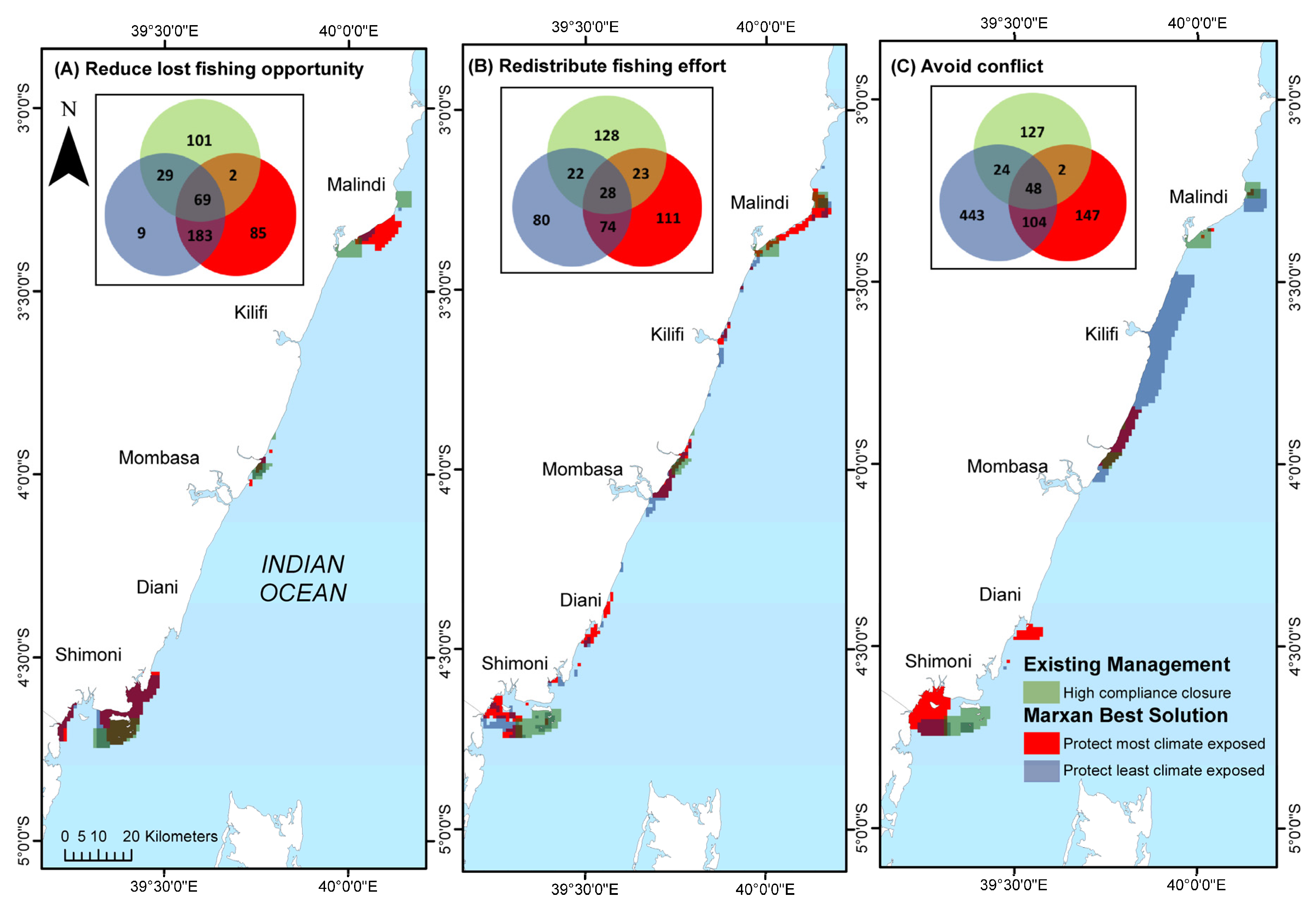
4. Discussion
4.1. Current Status of Kenyan Fringing Reef Habitats
4.2. Selecting Priority Management Areas
4.3. Comparing Selection Scenarios
4.4. Caveats
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Capolsini, P.; Andréfouët, S.; Rion, C.; Payri, C. A comparison of Landsat ETM+, SPOT HRV, IKONOS, ASTER, and airborne master data for coral reef habitat mapping in South Pacific Islands. Can. J. Remote Sens. 2003, 29, 187–200. [Google Scholar] [CrossRef]
- Walker, B.K.; Riegl, B.; Dodge, R.E. Mapping coral reef habitats in Southeast Florida using a combined technique approach. J. Coast. Res. 2008, 24, 1138–1150. [Google Scholar] [CrossRef]
- Andréfouët, S.; Muller-Karger, F.; Robinson, J.; Kranenburg, C.; Torres-Pulliza, D.; Spraggins, S.; Murch, B. Global assessment of modern coral reef extent and diversity for regional science and management applications: A view from space. In Proceedings of 10th International Coral Reef Symposium, Okinawa, Japan, 28 June–2 July 2004.
- Bruckner, A.; Rowlands, G.; Riegl, B.; Purkis, S.J.; Williams, A.; Renaud, P. Khaled bin Sultan Living Oceans Foundation Atlas of Saudi Arabian Red Sea Marine Habitats; Panoramic Press: Phoenix, AZ, USA, 2012. [Google Scholar]
- Rowlands, G.; Purkis, S.; Riegl, B.; Metsamaa, L.; Bruckner, A.; Renaud, P. Satellite imaging coral reef resilience at regional scale. A case-study from Saudi Arabia. Mar. Pollut. Bull. 2012, 64, 1222–1237. [Google Scholar] [CrossRef] [PubMed]
- Andréfouët, S. Coral reef habitat mapping using remote sensing: A user vs. producer perspective. Implications for research, management and capacity building. J. Spat. Sci. 2008, 53, 113–129. [Google Scholar] [CrossRef]
- Dalleau, M.; Andréfouët, S.; Wabnitz, C.C.; Payri, C.; Wantiez, L.; Pichon, M.; Friedman, K.; Vigliola, L.; Benzoni, F. Use of habitats as surrogates of biodiversity for efficient coral reef conservation planning in Pacific Ocean islands. Conserv. Biol. 2010, 24, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Wabnitz, C.C.; Andréfouët, S.; Torres-Pulliza, D.; Müller-Karger, F.E.; Kramer, P.A. Regional-scale seagrass habitat mapping in the wider caribbean region using landsat sensors: Applications to conservation and ecology. Remote Sens. Environ. 2008, 112, 3455–3467. [Google Scholar] [CrossRef]
- Wilkinson, C. Status of Coral Reefs of the World: 2008; Global Coral Reef Monitoring Network and Reef and Rainforest Research Centre: Townsville, QLD, Australia, 2008; p. 296. [Google Scholar]
- Cinner, J.E.; McClanahan, T.R. A sea change on the African coast? Preliminary social and ecological outcomes of a governance transformation in Kenyan fisheries. Glob. Environ. Chang. 2015, 30, 133–139. [Google Scholar] [CrossRef]
- McClanahan, T.R.; Abunge, C.A. Perceptions of fishing access restrictions and the disparity of benefits among stakeholder communities and nations of South-Eastern Africa. Fish Fish. 2015. [Google Scholar] [CrossRef]
- Almany, G.; Connolly, S.; Heath, D.; Hogan, J.; Jones, G.; McCook, L.; Mills, M.; Pressey, R.; Williamson, D. Connectivity, biodiversity conservation and the design of marine reserve networks for coral reefs. Coral Reefs 2009, 28, 339–351. [Google Scholar] [CrossRef]
- McCook, L.J.; Ayling, T.; Cappo, M.; Choat, J.H.; Evans, R.D.; de Freitas, D.M.; Heupel, M.; Hughes, T.P.; Jones, G.P.; Mapstone, B. Adaptive management of the great barrier reef: A globally significant demonstration of the benefits of networks of marine reserves. Proc. Natl. Acad. Sci. USA 2010, 107, 18278–18285. [Google Scholar] [CrossRef] [PubMed]
- Roff, J.C.; Taylor, M.E. National frameworks for marine conservation—A hierarchical geophysical approach. Aquat. Conserv. Mar. Freshw. Ecosyst. 2000, 10, 209–223. [Google Scholar] [CrossRef]
- Pressey, R.L.; Watts, M.E.; Barrett, T.W. Is maximizing protection the same as minimizing loss? Efficiency and retention as alternative measures of the effectiveness of proposed reserves. Ecol. Lett. 2004, 7, 1035–1046. [Google Scholar] [CrossRef]
- Ward, T.; Vanderklift, M.; Nicholls, A.; Kenchington, R. Selecting marine reserves using habitats and species assemblages as surrogates for biological diversity. Ecol. Appl. 1999, 9, 691–698. [Google Scholar] [CrossRef]
- Ecoregion, WWF Eastern African Marine. Towards a Western Indian Ocean Dugong Conservation Strategy: The Status of Dugongs in the Western Indian Ocean Region and Priority Conservation Actions; World Wide Fund: Dar es Salaam, Tanzania, 2004. [Google Scholar]
- Duda, A.M. Strengthening global governance of large marine ecosystems by incorporating coastal management and marine protected areas. Environ. Dev. 2015, in press. [Google Scholar] [CrossRef]
- Aswani, S.; Hamilton, R.J. Integrating indigenous ecological knowledge and customary sea tenure with marine and social science for conservation of bumphead parrotfish (Bolbometopon muricatum) in the Roviana Lagoon, Solomon Islands. Environ. Conserv. 2004, 31, 69–83. [Google Scholar] [CrossRef]
- Game, E.T.; McDonald-Madden, E.; Puotinen, M.L.; Possingham, H.P. Should we protect the strong or the weak? Risk, resilience, and the selection of marine protected areas. Conserv. Biol. 2008, 22, 1619–1629. [Google Scholar] [CrossRef] [PubMed]
- McClanahan, T.; Cinner, J.; Maina, J.; Graham, N.; Daw, T.; Stead, S.; Wamukota, A.; Brown, K.; Ateweberhan, M.; Venus, V. Conservation action in a changing climate. Conserv. Lett. 2008, 1, 53–59. [Google Scholar] [CrossRef]
- Allnutt, T.F.; McClanahan, T.R.; Andréfouët, S.; Baker, M.; Lagabrielle, E.; McClennen, C.; Rakotomanjaka, A.J.; Tianarisoa, T.F.; Watson, R.; Kremen, C. Comparison of marine spatial planning methods in madagascar demonstrates value of alternative approaches. PLoS ONE 2012, 7, e28969. [Google Scholar] [CrossRef] [PubMed]
- Ban, N.C.; Mills, M.; Tam, J.; Hicks, C.C.; Klain, S.; Stoeckl, N.; Bottrill, M.C.; Levine, J.; Pressey, R.L.; Satterfield, T. A social-ecological approach to conservation planning: Embedding social considerations. Front. Ecol. Environ. 2013, 11, 194–202. [Google Scholar] [CrossRef]
- Tuda, A.O.; Stevens, T.F.; Rodwell, L.D. Resolving coastal conflicts using marine spatial planning. J. Environ. Manag. 2014, 133, 59–68. [Google Scholar] [CrossRef] [PubMed]
- West, J.M.; Salm, R.V. Resistance and resilience to coral bleaching: Implications for coral reef conservation and management. Conserv. Biol. 2003, 17, 956–967. [Google Scholar] [CrossRef]
- Obura, D.O. Resilience and climate change: Lessons from coral reefs and bleaching in the western Indian Ocean. Estuar. Coast. Shelf Sci. 2005, 63, 353–372. [Google Scholar] [CrossRef]
- Scheffer, M.; Barrett, S.; Carpenter, S.; Folke, C.; Green, A.J.; Holmgren, M.; Hughes, T.; Kosten, S.; van de Leemput, I.; Nepstad, D. Creating a safe operating space for iconic ecosystems. Science 2015, 347, 1317–1319. [Google Scholar] [CrossRef] [PubMed]
- Watts, M.E.; Ball, I.R.; Stewart, R.S.; Klein, C.J.; Wilson, K.; Steinback, C.; Lourival, R.; Kircher, L.; Possingham, H.P. Marxan with zones: Software for optimal conservation based land-and sea-use zoning. Environ. Model. Softw. 2009, 24, 1513–1521. [Google Scholar] [CrossRef]
- Maina, J.; McClanahan, T.R.; Venus, V.; Ateweberhan, M.; Madin, J. Global gradients of coral exposure to environmental stresses and implications for local management. PLoS ONE 2011, 6, e23064. [Google Scholar] [CrossRef] [PubMed]
- Obura, D.O. Participatory monitoring of shallow tropical marine fisheries by artisanal fishers in Diani, Kenya. Bull. Mar. Sci. 2001, 69, 777–791. [Google Scholar]
- Andrefouet, S.; Rilwan, Y.; Hamel, M.A. Habitat mapping for conservation planning in baa atoll, republic of maldives. Atoll Res. Bull. 2012, 590, 207–222. [Google Scholar]
- Phinn, S.R.; Roelfsema, C.M.; Mumby, P.J. Multi-scale, object-based image analysis for mapping geomorphic and ecological zones on coral reefs. Int. J. Remote Sens. 2012, 33, 3768–3797. [Google Scholar] [CrossRef]
- Lyzenga, D.R. Remote sensing of bottom reflectance and water attenuation parameters in shallow water using aircraft and landsat data. Int. J. Remote Sens. 1981, 2, 71–82. [Google Scholar] [CrossRef]
- Malczewski, J. GIS and Multicriteria Decision Analysis; John Wiley & Sons: New York, NY, USA, 1999. [Google Scholar]
- Saaty, R.W. The analytic hierarchy process—What it is and how it is used. Math. Model. 1987, 9, 161–176. [Google Scholar] [CrossRef]
- Game, E.T.; Watts, M.E.; Wooldridge, S.; Possingham, H.P. Planning for persistence in marine reserves: A question of catastrophic importance. Ecol. Appl. 2008, 18, 670–680. [Google Scholar] [CrossRef] [PubMed]
- Cinner, J.E.; Graham, N.A.; Huchery, C.; Macneil, M.A. Global effects of local human population density and distance to markets on the condition of coral reef fisheries. Conserv. Biol. 2013, 27, 453–458. [Google Scholar] [CrossRef] [PubMed]
- CIESIN, C. Gridded Population of the World Version 3 (GPWV3): Population Density Grids; Socioeconomic Data and Applications Center (SEDAC), Columbia University: Palisades, NY, USA, 2005. [Google Scholar]
- Symes, D.; Phillipson, J. Inshore Fisheries Management; Springer Science & Business Media: Dordrecht, The Netherlands, 2013; Volume 2. [Google Scholar]
- Wabnitz, C.C.; Andréfouët, S.; Muller-Karger, F.E. Measuring progress toward global marine conservation targets. Front. Ecol. Environ. 2009, 8, 124–129. [Google Scholar] [CrossRef]
- McClanahan, T.; Mwaguni, S.; Muthiga, N. Management of the Kenyan coast. Ocean Coast. Manag. 2005, 48, 901–931. [Google Scholar] [CrossRef]
- McClanahan, T.R. Is there a future for coral reef parks in poor tropical countries? Coral reefs 1999, 18, 321–325. [Google Scholar] [CrossRef]
- Hamel, M.A.; Andréfouët, S.; Pressey, R.L. Compromises between international habitat conservation guidelines and small-scale fisheries in Pacific Island countries. Conserv. Lett. 2013, 6, 46–57. [Google Scholar] [CrossRef]
- McClanahan, T.R.; Graham, N.A.; MacNeil, M.A.; Muthiga, N.A.; Cinner, J.E.; Bruggemann, J.H.; Wilson, S.K. Critical thresholds and tangible targets for ecosystem-based management of coral reef fisheries. Proc. Natl. Acad. Sci. USA 2011, 108, 17230–17233. [Google Scholar] [CrossRef] [PubMed]
- MacNeil, M.A.; Graham, N.A.; Cinner, J.E.; Wilson, S.K.; Williams, I.D.; Maina, J.; Newman, S.; Friedlander, A.M.; Jupiter, S.; Polunin, N.V. Recovery potential of the world’s coral reef fishes. Nature 2015, 520, 341–344. [Google Scholar] [CrossRef] [PubMed]
- McClanahan, T.R.; Davies, J.; Maina, J. Factors influencing resource users and managers’ perceptions towards marine protected area management in Kenya. Environ. Conserv. 2005, 32, 42–49. [Google Scholar] [CrossRef]
- Cinner, J.; Daw, T.; McClanahan, T.; Muthiga, N.; Abunge, C.; Hamed, S.; Mwaka, B.; Rabearisoa, A.; Wamukota, A.; Fisher, E. Transitions toward co-management: The process of marine resource management devolution in three East African countries. Glob. Environ. Chang. 2012, 22, 651–658. [Google Scholar] [CrossRef]
- McClanahan, T.; Abunge, C. Catch rates and income are associated with fisheries management restrictions and not an environmental disturbance, in a heavily exploited tropical fishery. Mar. Ecol. Prog. Ser. 2014, 513, 201–210. [Google Scholar] [CrossRef]
- McClanahan, T.R.; Graham, N.A.; Darling, E.S. Coral reefs in a crystal ball: Predicting the future from the vulnerability of corals and reef fishes to multiple stressors. Curr. Opin. Environ. Sustain. 2014, 7, 59–64. [Google Scholar] [CrossRef]
- Andréfouët, S.; Kramer, P.; Torres-Pulliza, D.; Joyce, K.E.; Hochberg, E.J.; Garza-Pérez, R.; Mumby, P.J.; Riegl, B.; Yamano, H.; White, W.H. Multi-site evaluation of ikonos data for classification of tropical coral reef environments. Remote Sens. Environ. 2003, 88, 128–143. [Google Scholar] [CrossRef]
- Masalu, D.C. Coastal and marine resource use conflicts and sustainable development in tanzania. Ocean Coast. Manag. 2000, 43, 475–494. [Google Scholar] [CrossRef]
- Cicin-Sain, B.; Belfiore, S. Linking marine protected areas to integrated coastal and ocean management: A review of theory and practice. Ocean Coast. Manag. 2005, 48, 847–868. [Google Scholar] [CrossRef]
- Van Wynsberge, S.; Andréfouët, S.; Gaertner-Mazouni, N.; Remoissenet, G. Conservation and resource management in small tropical islands: Trade-offs between planning unit size, data redundancy and data loss. Ocean Coast. Manag. 2015, 116, 37–43. [Google Scholar] [CrossRef]
- Knudby, A.; Jupiter, S.; Roelfsema, C.; Lyons, M.; Phinn, S. Mapping coral reef resilience indicators using field and remotely sensed data. Remote Sens. 2013, 5, 1311–1334. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maina, J.M.; Jones, K.R.; Hicks, C.C.; McClanahan, T.R.; Watson, J.E.M.; Tuda, A.O.; Andréfouët, S. Designing Climate-Resilient Marine Protected Area Networks by Combining Remotely Sensed Coral Reef Habitat with Coastal Multi-Use Maps. Remote Sens. 2015, 7, 16571-16587. https://doi.org/10.3390/rs71215849
Maina JM, Jones KR, Hicks CC, McClanahan TR, Watson JEM, Tuda AO, Andréfouët S. Designing Climate-Resilient Marine Protected Area Networks by Combining Remotely Sensed Coral Reef Habitat with Coastal Multi-Use Maps. Remote Sensing. 2015; 7(12):16571-16587. https://doi.org/10.3390/rs71215849
Chicago/Turabian StyleMaina, Joseph M., Kendall R. Jones, Christina C. Hicks, Tim R. McClanahan, James E. M. Watson, Arthur O. Tuda, and Serge Andréfouët. 2015. "Designing Climate-Resilient Marine Protected Area Networks by Combining Remotely Sensed Coral Reef Habitat with Coastal Multi-Use Maps" Remote Sensing 7, no. 12: 16571-16587. https://doi.org/10.3390/rs71215849
APA StyleMaina, J. M., Jones, K. R., Hicks, C. C., McClanahan, T. R., Watson, J. E. M., Tuda, A. O., & Andréfouët, S. (2015). Designing Climate-Resilient Marine Protected Area Networks by Combining Remotely Sensed Coral Reef Habitat with Coastal Multi-Use Maps. Remote Sensing, 7(12), 16571-16587. https://doi.org/10.3390/rs71215849