ESA-MERIS 10-Year Mission Reveals Contrasting Phytoplankton Bloom Dynamics in Two Tropical Regions of Northern Australia
Abstract
:1. Introduction
2. Data and Methods
2.1. Ocean Color Dataset and Algorithms
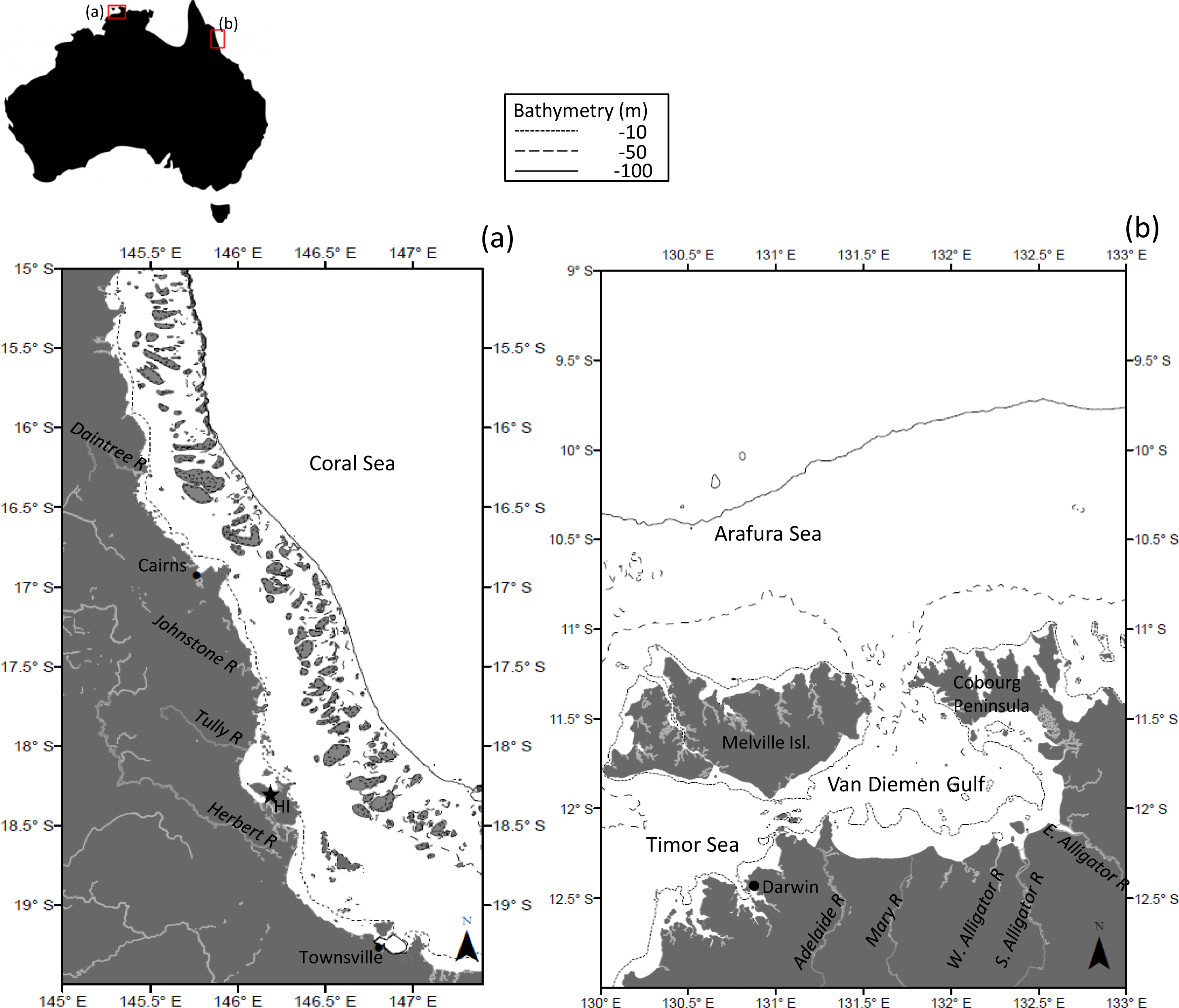
2.2. Regional Characteristics
2.3. Statistics
3. Results
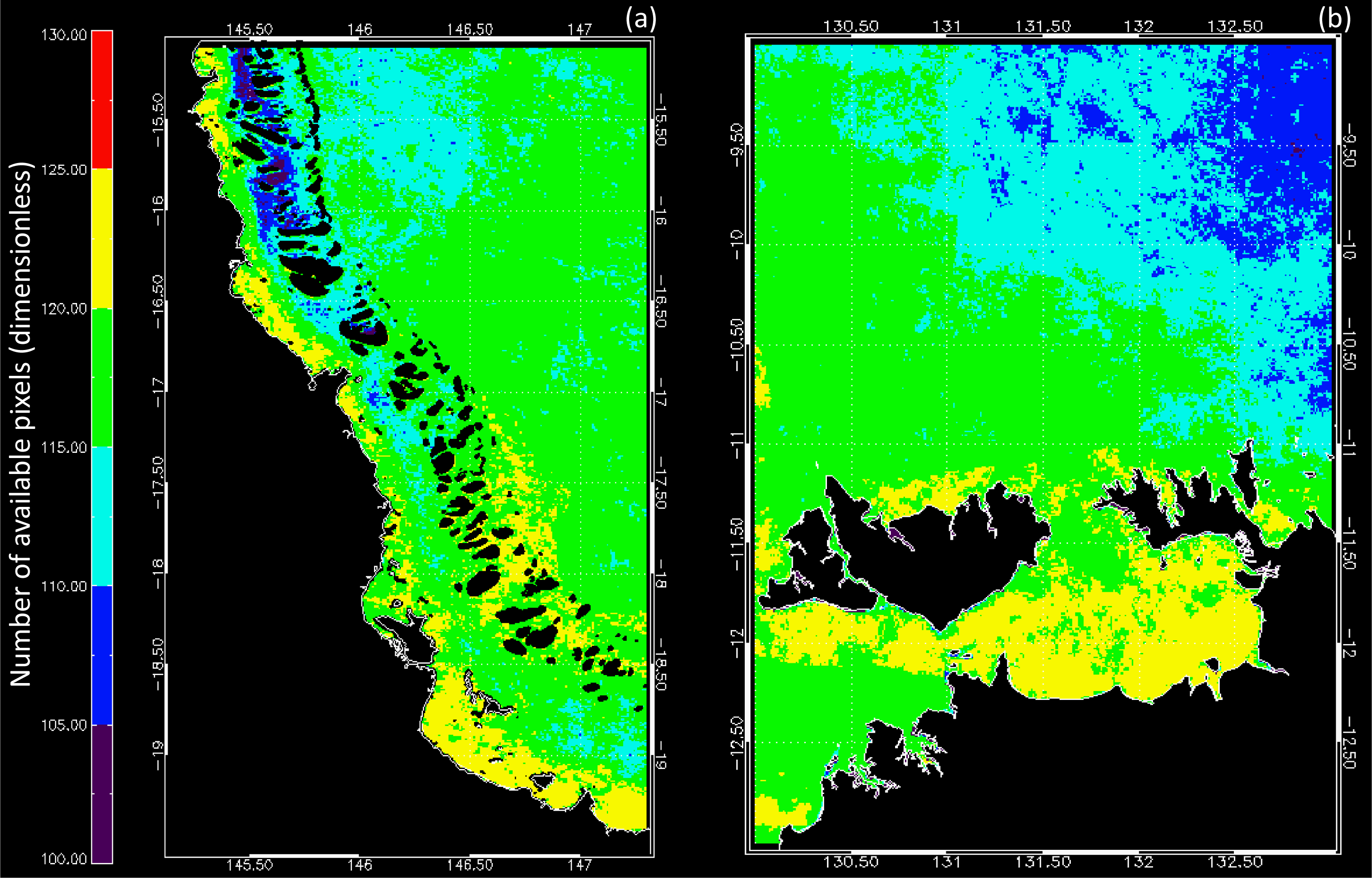
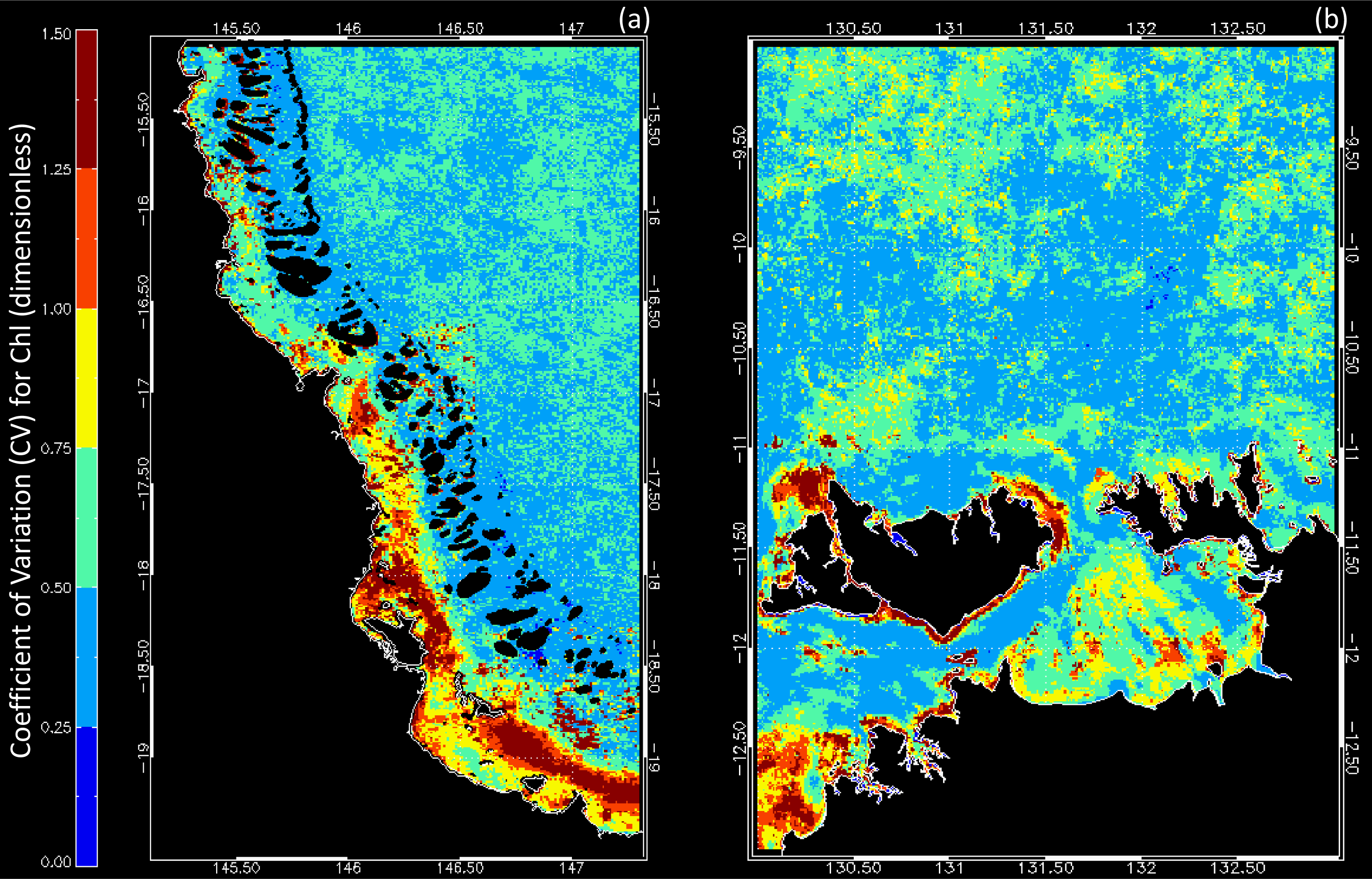
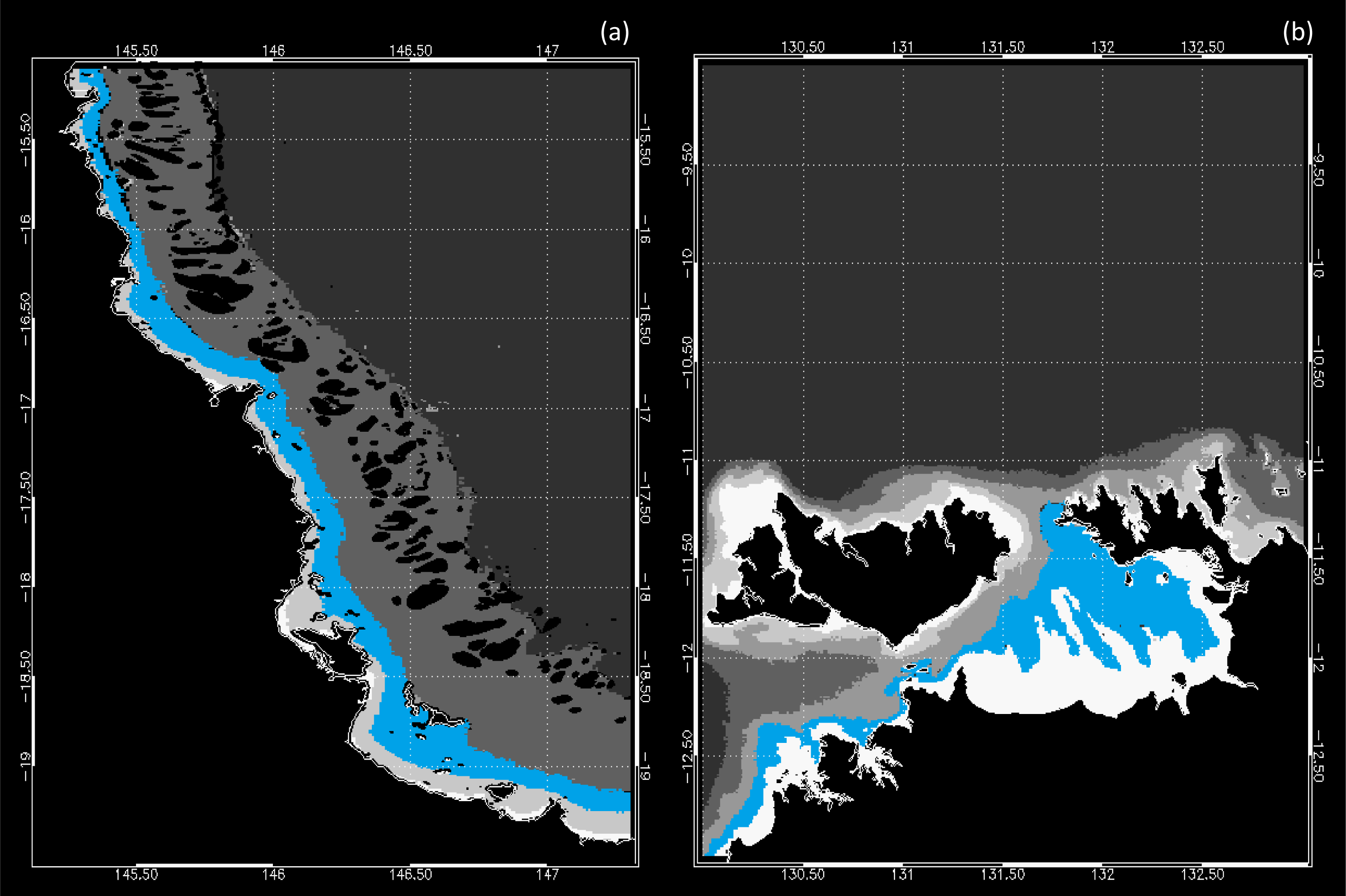
3.1. Data Availability and Chl Variability in the Study Regions
3.2. Cluster Analysis
3.3. Time-Series
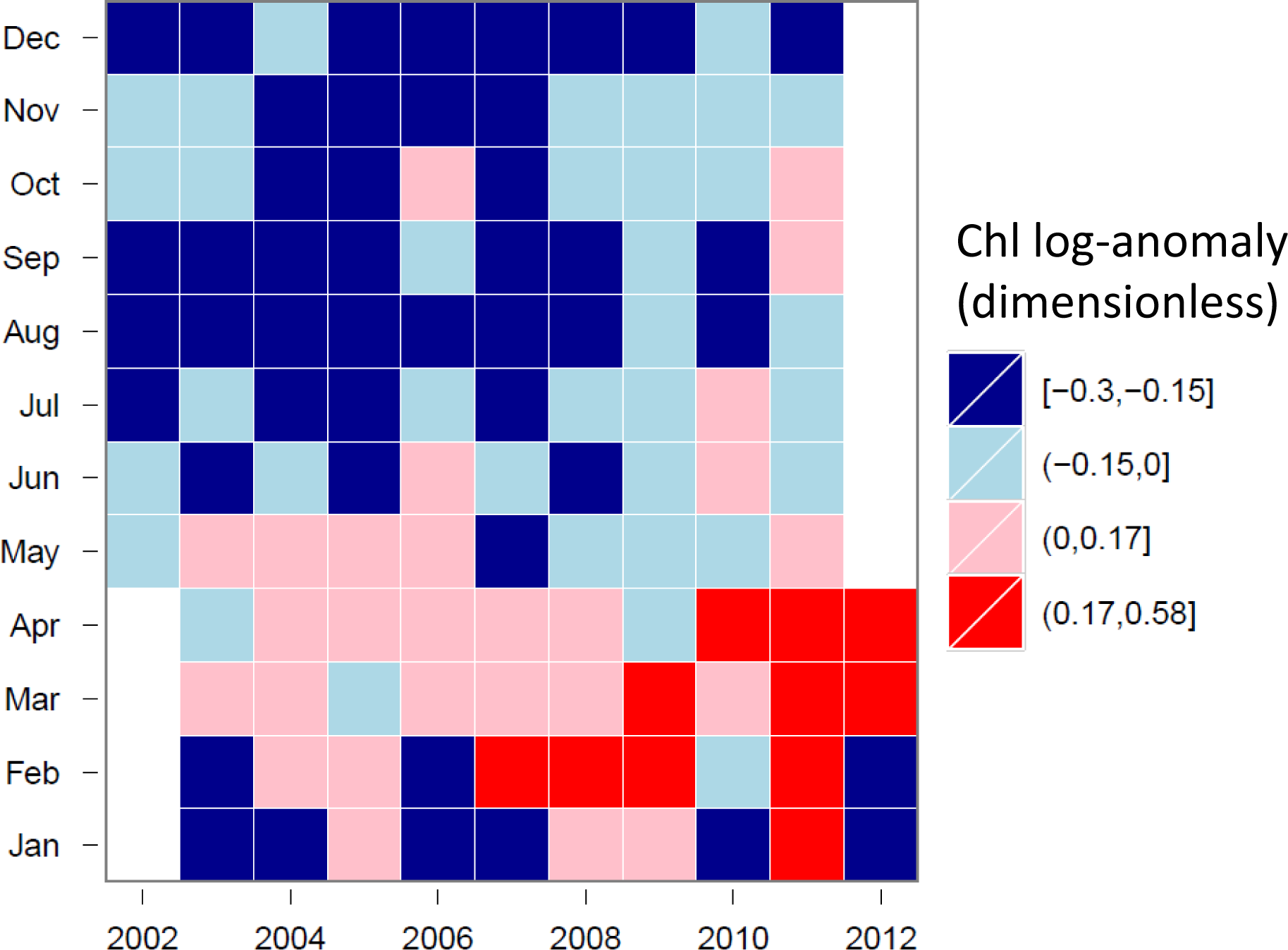
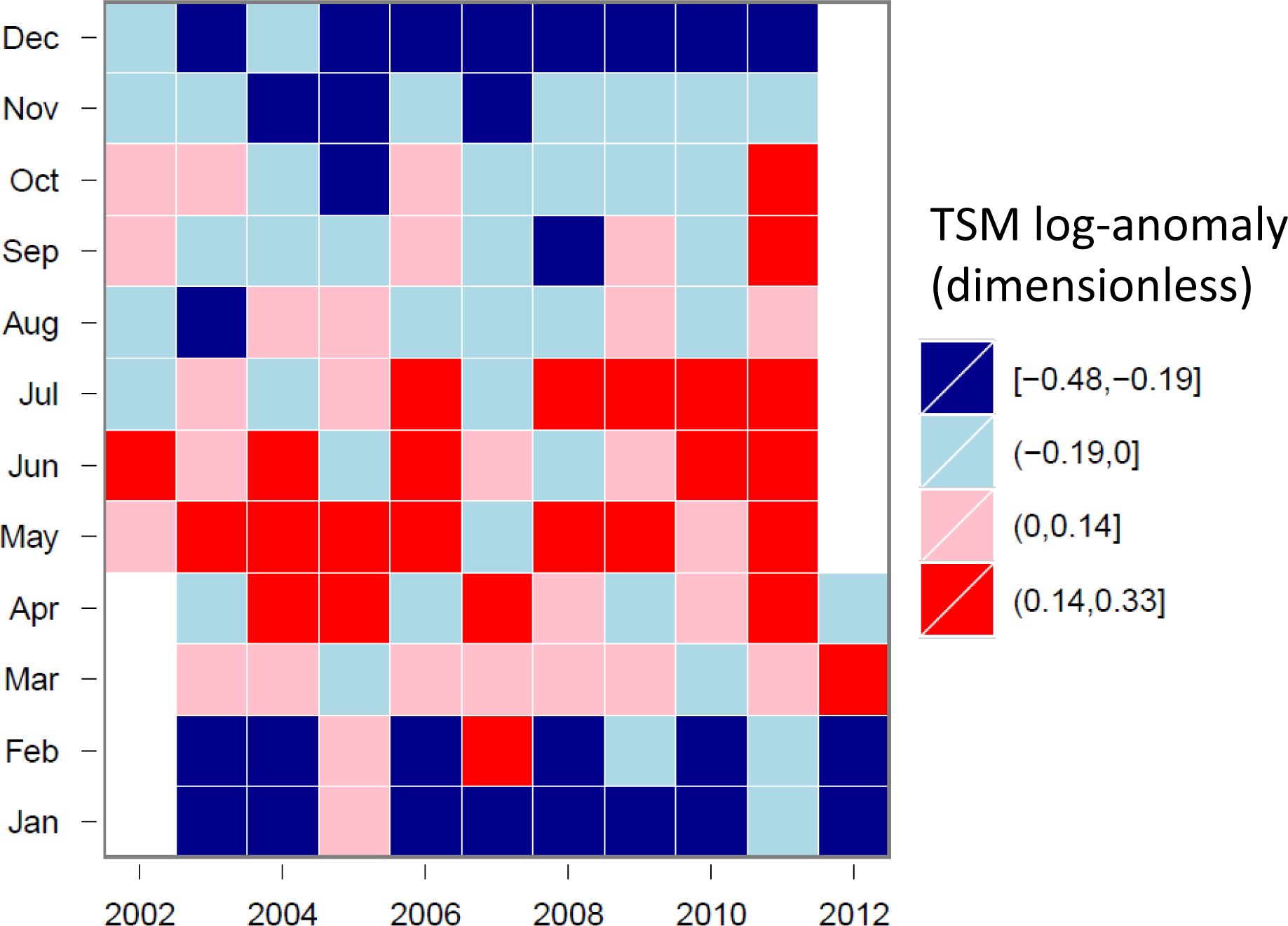
3.4. Hovmöller Diagrams
4. Discussion
4.1. Spatio-Temporal Phytoplankton Dynamics in the GBR Wet Tropics
4.2. Spatio-Temporal Phytoplankton Dynamics in the Van Diemen Gulf
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chiswell, S.M. Annual cycles and spring blooms in phytoplankton: Don’t abandon Sverdrup completely. Mar. Ecol. Prog. Ser 2011, 443, 39–50. [Google Scholar]
- Garcia-Soto, C.; Pingree, R.D. Spring and summer blooms of phytoplankton (SeaWiFS/MODIS) along a ferry line in the Bay of Biscay and Western English Channel. Cont. Shelf Res 2009, 29, 1111–1122. [Google Scholar]
- Furnas, M. Catchments and Corals: Terrestrial Runoff to the Great Barrier Reef; CRC/AIMS: Townsville, Australia, 2003; p. 335. [Google Scholar]
- Condie, S.A. Modeling seasonal circulation, upwelling and tidal mixing in the Arafura and Timor seas. Cont. Shelf Res 2011, 31, 1427–1436. [Google Scholar]
- Henson, S.A.; Sarmiento, J.L.; Dunne, J.P.; Bopp, L.; Lima, I.; Doney, S.C.; John, J.; Beaulieu, C. Detection of anthropogenic climate change in satellite records of ocean chlorophyll and productivity. Biogeosciences 2010, 7, 621–640. [Google Scholar]
- Done, T.; Jones, R. Tropical Coastal Ecosystems and Climate Change Prediction: Global and Local Risks. In Coral Reefs and Climate Change: Science and Management; Phinney, J.T., Strong, A., Skirving, W., Kleypas, J., Hoegh-Guldberg, H., Eds.; American Geophysical Union: Washington, DC, USA, 2006; pp. 5–32. [Google Scholar]
- Burke, L.; Reytar, K.; Spalding, M.; Perry, A. Reefs at Risk Revisited; World Resources Institute: Washington, DC, USA, 2011; p. 114. [Google Scholar]
- Waterhouse, J.; Brodie, J.; Lewis, S.; Mitchell, A. Quantifying the sources of pollutants in the Great Barrier Reef catchments and the relative risk to reef ecosystems. Mar. Pollut. Bull 2012, 65, 394–406. [Google Scholar]
- Brodie, J.E.; Kroon, F.J.; Schaffelke, B.; Wolanski, E.C.; Lewis, S.E.; Devlin, M.J.; Bohnet, I.C.; Bainbridge, Z.T.; Waterhouse, J.; Davis, A.M. Terrestrial pollutant runoff to the Great Barrier Reef: An update of issues, priorities and management responses. Mar. Pollut. Bull 2012, 65, 81–100. [Google Scholar]
- Wooldridge, S.A. Water quality and coral bleaching thresholds: Formalising the linkage for the inshore reefs of the Great Barrier Reef, Australia. Mar. Pollut. Bull 2009, 58, 745–751. [Google Scholar]
- Brodie, J.; Schroeder, T.; Rohde, K.; Faithful, J; Masters, B.; Dekker, A.; Brando, V.E.; Maughan, M. Dispersal of suspended sediments and nutrients in the Great Barrier Reef lagoon during river-discharge events: Conclusions from satellite remote sensing and concurrent flood-plume sampling. Mar. Freshw. Res 2010, 61, 651–664. [Google Scholar]
- Brodie, J.E.; Devlin, M.; Haynes, D.; Waterhouse, J. Assessment of the eutrophication status of the Great Barrier Reef Lagoon (Australia). Biogeochemistry 2011, 106, 281–302. [Google Scholar]
- Halpern, B.S.; Walbridge, S.; Selkoe, K.A.; Kappel, C.V.; Micheli, F.; D’Agrosa, C.; Bruno, J.F.; Casey, K.S.; Ebert, C.; Fox, H.E.; et al. A global map of human impact on marine ecosystems. Science 2008, 319, 948–952. [Google Scholar]
- Hallegraeff, G.M.; Jeffrey, S.W. Tropical phytoplankton species and pigments in continental shelf waters of North and North-West Australia. Mar. Ecol. Prog. Ser 1984, 20, 59–74. [Google Scholar]
- IOCCG. Remote Sensing of Ocean Colour in Coastal, and Other Optically-Complex, Waters: 3; Bedford Institude: Dartmouth, NS, Canada, 2000; p. 145. [Google Scholar]
- Odermatt, D.; Gitelson, A.; Brando, V.E.; Schaepman, M. Review of constituent retrieval in optically deep and complex waters from satellite imagery. Remote Sens. Environ 2012, 118, 116–126. [Google Scholar]
- Brando, V.E.; Dekker, A.G.; Park, Y.J.; Schroeder, T. An adaptive semi-analytical inversion of ocean colour radiometry in optically complex waters. Appl. Opt 2012, 51, 2808–2833. [Google Scholar]
- Matthews, M.W. A current review of empirical procedures of remote sensing in inland and near-coastal transitional waters. Int. J. Remote Sens 2011, 32, 6855–6899. [Google Scholar]
- Sharples, J.; Ross, O.N.; Scott, B.E.; Greenstreet, S.P.R.; Fraser, H. Inter-annual variability in the timing of stratification and the spring bloom in the North-Western North Sea. Cont. Shelf Res 2006, 26, 733–751. [Google Scholar]
- Gower, J.; King, S. An antarctic ice-related “superbloom” observed with the MERIS satellite imager. Geophys. Res. Lett 2007, 34. [Google Scholar] [CrossRef]
- Kahru, M.; Brotas, V.; Manzano-Sarabia, M.; Mitchell, B.G. Are phytoplankton blooms occurring earlier in the Arctic? Glob. Chang. Biol 2010, 17, 1733–1739. [Google Scholar]
- Brewin, R.J.W.; Hirata, T.; Hardman-Mountford, N.J.; Lavender, S.J.; Sathyendranath, S.; Barlow, R. The influence of the indian ocean dipole on interannual variations in phytoplankton size structure as revealed by Earth Observation. Deep Sea Res. Part II Top. Stud. Oceanogr 2012, 77–80, 117–127. [Google Scholar]
- Ryan, J.P.; Fischer, A.M.; Kudela, R.M.; Gower, J.; King, S.A.; MarinIIIa, R.; Chavez, F.P. Influences of upwelling and downwelling winds on red tide bloom dynamics in Monterey Bay, California. Cont. Shelf Res 2009, 29, 785–795. [Google Scholar]
- Peñaflor, E.L.; Villanoy, C.L.; Liu, C.-T.; David, L.T. Detection of monsoonal phytoplankton blooms in Luzon Strait with MODIS data. Remote Sens. Environ 2007, 109, 443–450. [Google Scholar]
- Tian, T.; Merico, A.; Su, J.; Staneva, J.; Wiltshire, K.; Wirtz, K. Importance of resuspended sediment dynamics for the phytoplankton spring bloom in a coastal marine ecosystem. J. Sea Res 2009, 62, 214–228. [Google Scholar]
- Blondeau-Patissier, D.; Gower, J.F.R.; Dekker, A.G.; Phinn, S.R.; Brando, V.E. A review of ocean color remote sensing methods and statistical techniques for the detection, mapping and analysis of phytoplankton blooms in coastal and open oceans. Prog. Oceanogr 2014, in press.. [Google Scholar]
- Doerffer, R.; Sørensen, K.; Aiken, J. MERIS potential for coastal zone applications. Int. J. Remote Sens 1999, 20, 1809–1818. [Google Scholar]
- MERIS Quality Working Group. MERIS 3rd Data Reprocessing—Software and ADF Updates; MERIS Quality Working Group: Nice, France, 2011; p. 84. [Google Scholar]
- Steinmetz, F.; Deschamps, P.Y.; Ramon, D. Atmospheric correction in presence of sun glint: Application to MERIS. Opt. Express 2011, 19, 9783–9800. [Google Scholar]
- Gower, J.; Doerffer, R.; Borstad, G.A. Interpretation of the 685 nm peak in water-leaving radiance spectra in terms of fluorescence, absorption and scattering, and its observation by MERIS. Int. J. Remote Sens 1999, 20, 1771–1786. [Google Scholar]
- Gower, J.; King, S.; Goncalves, P. Global monitoring of plankton blooms using MERIS MCI. Int. J. Remote Sens 2008, 29. [Google Scholar] [CrossRef]
- Schroeder, T.; Behnert, I.; Schaale, M.; Fischer, J.; Doerffer, R. Atmospheric correction algorithm for MERIS above case-2 waters. Int. J. Remote Sens 2007, 28, 1469–1486. [Google Scholar]
- Fomferra, N.; Brockmann, C. Beam—The ENVISAT MERIS and AATSR Toolbox. In Proceedings of the MERIS-(A)ATSR Workshop, Frascati, Italy, 26–30 September 2005; p. 3.
- Schroeder, T.; Schaale, M.; Fisher, J. Retrieval of atmospheric and oceanic properties from meris measurements: A new Case-2 water processor for BEAM. Int. J. Remote Sens 2007, 28, 5627–5632. [Google Scholar]
- Blondeau-Patissier, D. Detection and Quantification of Algal Bloom Dynamics in the Great Barrier Reef Lagoonal Waters Using Remote Sensing and Bio-Optics. Ph.D. Thesis, University Of Queensland, Brisbane, Australia,. 2011. [Google Scholar]
- Binding, C.E.; Greenberg, T.A.; Bukata, R.P. The MERIS maximum chlorophyll index; its merits and limitations for inland water algal bloom monitoring. J. Great Lakes Res 2013, 39, 100–107. [Google Scholar]
- Gower, J.; King, S.A. Distribution of floating Sargassum in the Gulf of Mexico and the Atlantic ocean mapped using MERIS. Int. J. Remote Sens 2011, 32, 1917–1929. [Google Scholar]
- Montes-Hugo, M.A.; Fiorani, L.; Marullo, S.; Roy, S.; Gagné, J.-P.; Borelli, R.; Demers, S.; Palucci, A. A comparison between local and global spaceborne Chlorophyll indices in the St. Lawrence estuary. Remote Sens 2012, 4, 3666–3688. [Google Scholar]
- Wynne, T.T.; Stumpf, R.P.; Tomlinson, M.C.; Warner, R.A.; Tester, P.A.; Dyble, J.; Fahnenstiel, G.L. Relating spectral shape to Cyanobacterial blooms in the Laurentian Great Lakes. Int. J. Remote Sens 2008, 29, 3665–3672. [Google Scholar]
- Wynne, T.T.; Stumpf, R.P.; Briggs, T.O. Comparing MODIS and MERIS spectral shapes for Cyanobacterial bloom detection. Int. J. Remote Sens 2013, 34, 6668–6678. [Google Scholar]
- Schroeder, T.; Devlin, M.J.; Brando, V.E.; Dekker, A.G.; Brodie, J.E.; Clementson, L.A.; McKinna, L. Inter-annual variability of wet season freshwater plume extent into the Great Barrier Reef Lagoon based on satellite coastal ocean colour observations. Mar. Pollut. Bull 2012, 65, 210–223. [Google Scholar]
- Susanto, R.D.; Moore, T.S.; Marra, J. Ocean color variability in the Indonesian Seas during the SeaWiFS era. Geochem. Geophys. Geosyst 2006, 7. [Google Scholar] [CrossRef]
- Andrews, J.C.; Clegg, S. Coral sea circulation and transport deduced from modal information models. Deep Sea Res. Part A Oceanogr. Res. Pap 1989, 36, 957–974. [Google Scholar]
- Schaffelke, B.; Carleton, J.; Skuza, M.; Zagorskis, I.; Furnas, M.J. Water quality in the inshore Great Barrier Reef lagoon: Implications for long-term monitoring and management. Mar. Pollut. Bull 2012, 65, 249–260. [Google Scholar]
- Neil, D.T.; Orpin, A.; Vid, P.; Yu, B. Sediment yield and impacts from river catchments to the Great Barrier Reef lagoon. Mar. Freshw. Res 2002, 53, 733–752. [Google Scholar]
- King, B.; Wolanski, E. Bottom Friction Reduction in Turbid Estuaries. In Mixing in Estuaries and Coastal Seas; Pattiaratchi, C., Ed.; AGU: Washington, DC, USA, 1996; Volume 50, pp. 325–337. [Google Scholar]
- Furnas, M.J.; Mitchell, A.W. Biological Oceanography of the Great Barrier Reef. In The Great Barrier Reef: Science, Use and Management; Great Barrier Reef Marine Park Authority: Townsville, Queensland, Australia, 1997; pp. 75–81. [Google Scholar]
- Furnas, M. Net in situ growth rates of phytoplankton in an oligotrophic, tropical shelf ecosystem. Limnol. Oceanogr 1991, 36, 13–29. [Google Scholar]
- Blondeau-Patissier, D.; Brando, V.E.; Oubelkheir, K.; Dekker, A.G.; Clementson, L.A.; Daniel, P. Bio-optical variability of the absorption and scattering properties of the Queensland inshore and reef waters, Australia. J. Geophys. Res 2009, 114. [Google Scholar] [CrossRef]
- Wooldridge, S.; Brodie, J.; Furnas, M. Exposure of inner-shelf reefs to nutrient enriched runoff entering the Great Barrier Reef Lagoon: Post-european changes and the design of water quality targets. Mar. Pollut. Bull 2006, 52, 1467–1479. [Google Scholar]
- Sorokin, Y.I.; Sorokin, P. Plankton of the central Great Barrier Reef: Abundance, production and trophodynamic roles. J. Mar. Biol. Assoc. UK 2010, 90, 1173–1187. [Google Scholar]
- Crosbie, N.D.; Furnas, M.J. Abundance, distribution and flow-cytometric characterization of picophytoprokaryote populations in central (17°S) and southern (20°S) shelf waters of the Great Barrier Reef. J. Plankton Res 2001, 23, 809–828. [Google Scholar]
- McKinna, L.I.W.; Furnas, M.J.; Ridd, P.V. A simple, binary classification algorithm for the detection of Trichodesmium spp. within the Great Barrier Reef using MODIS imagery. Limnol. Oceanogr. Methods 2011, 9, 50–66. [Google Scholar]
- Drewry, J.; Dostine, P.L.; Fortune, J.; Majid, M.; Schult, J.; Lamche, G. Darwin Harbour Region Report Cards 2010; 25/2010D; Department of Natural Resources, Environment, The Arts and Sport: Palmerston, Northern Territory, Australia, 2010; p. 74. [Google Scholar]
- Blondeau-Patissier, D.; Dekker, A.G.; Schroeder, T.; Brando, V.E. Thematic Findings: Surface Phytoplankton Blooms and Phytoplankton Biomass in Coastal Waters. In Australia State of the Environment 2011. Independent Report to the Australian Government Minister for Sustainability, Environment, Water, Population and Communities; DSEWPaC: Canberra, Australia, 2011; pp. 410–411. [Google Scholar]
- Ferreira, A.; Garcia, C.A.E.; Dogliotti, A.I.; Garcia, V.M.T. Bio-optical characteristics of the Patagonia shelf break waters: Implications for ocean color algorithms. Remote Sens. Environ 2013, 136, 416–432. [Google Scholar]
- IOCCG. Partition of the Ocean into Ecological Provinces: Role of Ocean-Colour Radiometry; Report number 9; Bedford Institude: Dartmouth, NS, Canada, 2009. [Google Scholar]
- Jassby, A.D.; Cloern, J.E. Package “wq”—Some Tools for Exploring Water Quality Monitoring Data, Version 0.3–4; R-CRAN, R Foundation for Statistical Computing: Vienna, Austria, 2011. [Google Scholar]
- Team, R.C. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Hirsch, R.M.; Slack, J.R.; Smith, R.A. Techniques of trend analysis formonthly water quality data. Water Resour. Res 1982, 18, 107–121. [Google Scholar]
- Brodie, J.; Wolanski, E.; Lewis, S.; Bainbridge, Z. An assessment of residence times of land-sourced contaminants in the Great Barrier Reef Lagoon and the implications for management and reef recovery. Mar. Pollut. Bull 2012, 65, 267–279. [Google Scholar]
- Weeks, S.; Werdell, P.J.; Schaffelke, B.; Canto, M.; Lee, Z.; Wilding, J.G.; Feldman, G.C. Satellite-derived photic depth on the great barrier reef: Spatio-temporal patterns of water clarity. Remote Sens 2012, 4, 3781–3795. [Google Scholar]
- Brodie, J.; De’ath, G.; Devlin, M.; Furnas, M.; Wright, M. Spatial and temporal patterns of near-surface chlorophyll a in the Great Barrier Reef lagoon. Mar. Freshw. Res 2007, 58, 342–353. [Google Scholar]
- Brinkman, R.; Wolanski, E.; Deleersnijderb, E.; McAllistera, F.; Skirvinga, W. Oceanic inflow from the coral sea into the Great Barrier Reef. Estuar. Coast. Shelf Sci 2001, 54, 655–668. [Google Scholar]
- Hancock, G.J.; Webster, I.T.; Stieglitz, T.C. Horizontal mixing of Great Barrier Reef waters: Offshore diffusivity determined from radium isotope distribution. J. Geophys. Res 2006, 111. [Google Scholar] [CrossRef]
- Gower, J.; King, S.; Borstad, G.; Brown, L. The importance of a band at 709 nm for interpreting water-leaving spectral radiance. Can. J. Remote Sens 2008, 34, 287–295. [Google Scholar]
- Devlin, M.J.; Brodie, J.E.; Bainbridge, Z.T.; Lewis, S.E. Flood Plumes in the Great Barrier Reef–the Burdekin and Fitzroy Flood Plumes (2007/08): Case Studies for Marine Monitoring Program; James Cook University: Townsville, Australia, 2008; p. 63. [Google Scholar]
- Lyon, P.; Acker, J.; Hoge, F.; Shen, S.; Roffer, M.; Gawlikowski, G. Interaction of Hurricane Katrina with Optically Complex Water in the Gulf of Mexico: Interpretation Using Satellite-Derived Inherent Optical Properties and Chlorophyll Concentration. In Proceedings of the 2008 Fall Meeting of the American Geophysical Union, San Francisco, CA, USA, 15–19 December 2008.
- Acker, J.; Lyon, P.; Hoge, F.; Shen, S.; Roffer, M.; Gawlikowski, G. Interaction of Hurricane Katrina with optically complex water in the Gulf of Mexico: Interpretation using satellite-derived inherent optical properties and chlorophyll concentration. IEEE Geosci. Remote Sens. Lett 2009, 6, 209–213. [Google Scholar]
- Neveux, J.; Tenório, M.M.B.; Jacquet, S.; Torreton, J.P.; Douillet, P.; Ouillon, S.; Dupouy, C. Chlorophylls and phycoerythrins as markers of environmental forcings including cyclone Erica effect (March 2003) on phytoplankton in the south-west Lagoon of New Caledonia and oceanic adjacent area. Int. J. Oceanogr 2009, 2009, 1–19. [Google Scholar]
- Shi, W.; Wang, M. Observations of a Hurricane Katrina-induced phytoplankton bloom in the Gulf of Mexico. Geophys. Res. Lett 2007, 34. [Google Scholar] [CrossRef]
- Campbell, D.; Hurry, V.; Clarke, A.K.; Gustafsson, P.; Oquist, G. Chlorophyll fluorescence analysis of Cyanobacterial photosynthesis and acclimation. Microbiol. Mol. Biol. Rev 1998, 62, 667–683. [Google Scholar]
- Qu, T.; Du, Y.; Strachan, J.; Meyers, G.; Slingo, J. Sea surface temperature and its variability in the Indonesian region. Oceanography 2005, 18, 50–61. [Google Scholar]
- Huisman, J.; Hulot, F.D. Population Dynamics of Harmful Cyanobacteria. In Harmful Cyanobacteria; Huisman, J., Matthijs, H.C.P., Visser, M., Eds.; Springer: Dordrecht, the Netherlands, 2005; pp. 143–176. [Google Scholar]
- Fu, F.-X.; Bell, P.R.F. Effect of salinity on growth, pigmentation, N2 fixation and alkaline phosphatase activity of cultured Trichodesmium sp. Mar. Ecol. Prog. Ser 2003, 257, 69–76. [Google Scholar]
- Gazeau, F.; Smith, S.V.; Gentili, B.; Frankignoulle, M.; Gattuso, J.-P. The European coastal zone: Characterization and first assessment of ecosystem metabolism. Estuar. Coast. Shelf Sci 2004, 60, 673–694. [Google Scholar]
- Siegel, D.A.; Antoine, D.; Behrenfeld, M.J.; Fanton d’Andon, O.H.; Fields, E.; Franz, B.A.; Goryl, P.; Maritorena, S.; McClain, C.R.; Wang, M.; et al. Global Ocean Phytoplankton. In State of the Climate in 2010, Bulletin of the American Meteorological Society; AMS: Boston, MA, USA, 2011; Volume 93, pp. S107–S110. [Google Scholar]
















© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Blondeau-Patissier, D.; Schroeder, T.; Brando, V.E.; Maier, S.W.; Dekker, A.G.; Phinn, S. ESA-MERIS 10-Year Mission Reveals Contrasting Phytoplankton Bloom Dynamics in Two Tropical Regions of Northern Australia. Remote Sens. 2014, 6, 2963-2988. https://doi.org/10.3390/rs6042963
Blondeau-Patissier D, Schroeder T, Brando VE, Maier SW, Dekker AG, Phinn S. ESA-MERIS 10-Year Mission Reveals Contrasting Phytoplankton Bloom Dynamics in Two Tropical Regions of Northern Australia. Remote Sensing. 2014; 6(4):2963-2988. https://doi.org/10.3390/rs6042963
Chicago/Turabian StyleBlondeau-Patissier, David, Thomas Schroeder, Vittorio E. Brando, Stefan W. Maier, Arnold G. Dekker, and Stuart Phinn. 2014. "ESA-MERIS 10-Year Mission Reveals Contrasting Phytoplankton Bloom Dynamics in Two Tropical Regions of Northern Australia" Remote Sensing 6, no. 4: 2963-2988. https://doi.org/10.3390/rs6042963
APA StyleBlondeau-Patissier, D., Schroeder, T., Brando, V. E., Maier, S. W., Dekker, A. G., & Phinn, S. (2014). ESA-MERIS 10-Year Mission Reveals Contrasting Phytoplankton Bloom Dynamics in Two Tropical Regions of Northern Australia. Remote Sensing, 6(4), 2963-2988. https://doi.org/10.3390/rs6042963





