Physiological and Hyperspectral Responses of Individual European Beech Trees to Drought Stress: A Pilot Study During a Compound Drought and Heatwave Event
Highlights
- Water-stressed beech exhibited up to 70% reductions in photosynthesis and 35% reductions in chlorophyll content under severe drought conditions.
- Red-edge hyperspectral indices successfully detected individual-tree stress; the traditional NDVI failed.
- Early drought stress impairs photosynthesis before visible symptoms appear, underscoring the need for proactive monitoring systems for forest management.
- Airborne remote sensing enables stress detection in beeches facing increasing climate extremes.
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site and Experimental Setup
2.2. Climate Data and Weather Conditions During the Field Campaign
2.3. In Situ and In-Laboratory Physiological, Biochemical and Spectral Measurements at the Leaf Level
2.3.1. Gas Exchange Measurements
2.3.2. Chlorophyll Content Measurements
2.4. Remotely Sensed Data
2.4.1. LiDAR Data Acquisition and Processing
2.4.2. Hyperspectral Data Acquisition and Processing
2.5. Data Analysis
3. Results
3.1. Effect of Water Stress on European Beech Physiological and Biochemical Parameters
3.2. Effect of Water Stress on European Beech Crown Reflectance and Spectral Vegetation Indices
4. Discussion
4.1. Effect of the CDHW Event and the Manipulation-Induced Water Stress on European Beech Physiological and Biochemical Parameters
4.2. Traceability of Drought Stress Through Hyperspectral Indices Measured at the Tree-Crown Level
4.3. Study Limitations and Future Research Priorities
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Amax | maximum photosynthesis rate |
| An | net photosynthesis |
| Asat | net photosynthesis at a saturating PAR |
| CDHW | compound drought and heatwave |
| CHI | chlorophyll index |
| CIRE | chlorophyll red-edge index |
| ETR | electron transport rate |
| Fm | maximum fluorescence yield in the dark-adapted state |
| Fm’ | maximum fluorescence yield in the light-adapted state |
| Fo | minimum fluorescence yield in dark-adapted state |
| Fs | steady-state fluorescence level in the light-adapted state |
| Fv/Fm | maximum potential quantum efficiency of photosystem II |
| gs | stomatal conductance |
| IRGA | infrared gas analyser |
| LAI | leaf area index |
| NDVI | normalised difference vegetation index |
| NDWI | normalised difference water index |
| NIR | near-infrared |
| NPQ | non-photochemical quenching |
| NWI | normalised water index |
| PAM | pulse-amplitude-modulated |
| PAR | photosynthetically active radiation |
| PRI | photochemical reflectance index |
| PSII | photosystem II |
| Rd | dark respiration rate |
| RENDVI | red-edge normalised difference vegetation index |
| ROI | region of interest |
| SPRI | scaled photochemical reflectance index |
| SVI | spectral vegetation index |
| SWIR | shortwave infrared |
| Tr | transpiration rate |
| VIS | visible |
| VNIR | visible and near-infrared |
References
- Beniston, M. Extreme climatic events: Examples from the alpine region. J. De Phys. IV 2004, 121, 139–149. [Google Scholar] [CrossRef]
- Reichstein, M.; Bahn, M.; Ciais, P.; Frank, D.; Mahecha, M.D.; Seneviratne, S.I.; Zscheischler, J.; Beer, C.; Buchmann, N.; Frank, D.C.; et al. Climate extremes and the carbon cycle. Nature 2013, 500, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Zwiers, F.W.; Alexander, L.V.; Hegerl, G.C.; Knutson, T.R.; Kossin, J.P.; Naveau, P.; Nicholls, N.; Schär, C.; Seneviratne, S.I.; Zhang, X. Climate Extremes: Challenges in Estimating and Understanding Recent Changes in the Frequency and Intensity of Extreme Climate and Weather Events. In Climate Science for Serving Society; Asrar, G., Hurrell, J., Eds.; Springer: Dordrecht, The Netherlands, 2013. [Google Scholar] [CrossRef]
- Perkins, S.E.; Alexander, L.V. On the measurement of heat waves. J. Clim. 2013, 26, 4500–4517. [Google Scholar] [CrossRef]
- Van Loon, A.F. Hydrological drought explained. Wiley Interdiscip. Rev. Water 2015, 2, 359–392. [Google Scholar] [CrossRef]
- Zhang, Q.; She, D.; Zhang, L.; Wang, G.; Chen, J.; Hao, Z. High Sensitivity of Compound Drought and Heatwave Events to Global Warming in the Future. Earth’s Future 2022, 10, e2022EF002833. [Google Scholar] [CrossRef]
- Peel, M.C.; Finlayson, B.L.; Mcmahon, T.A. Hydrology and Earth System Sciences Updated world map of the Köppen-Geiger climate classification. Hydrol. Earth Syst. Sci. 2007, 11, 1633–1644. [Google Scholar] [CrossRef]
- Stephan, R.; Erfurt, M.; Terzi, S.; Zun, M.; Kristan, B.; Haslinger, K.; Stahl, K. An inventory of Alpine drought impact reports to explore past droughts in a mountain region. Nat. Hazards Earth Syst. Sci. 2021, 21, 2485–2501. [Google Scholar] [CrossRef]
- Calanca, P. Climate change and drought occurrence in the Alpine region: How severe are becoming the extremes? Glob. Planet. Change 2007, 57, 151–160. [Google Scholar] [CrossRef]
- Beniston, M.; Stephenson, D.B. Extreme climatic events and their evolution under changing climatic conditions. Glob. Planet. Change 2004, 44, 1–9. [Google Scholar] [CrossRef]
- Lhotka, O.; Kyselý, J. The 2021 European Heat Wave in the Context of Past Major Heat Waves. Earth Space Sci. 2022, 9, e2022EA002567. [Google Scholar] [CrossRef]
- EEA. European forest types. In Categories and Types for Sustainable Forest Management Reporting and Policy; EEA Technical Report No 9/2006; Office for Official Publications of the European Communities: Luxembourg, 2006; ISSN 1725-2237. Available online: https://foresteurope.org/wp-content/uploads/2022/01/WFC_4_eea_technical_report_92006.pdf (accessed on 5 December 2025).
- Oddi, L.; Migliavacca, M.; Cremonese, E.; Filippa, G.; Vacchiano, G.; Siniscalco, C.; Morra Di Cella, U.; Galvagno, M. Contrasting responses of forest growth and carbon sequestration to heat and drought in the Alps. Environ. Res. Lett. 2022, 17, 045015. [Google Scholar] [CrossRef]
- Páscoa, P.; Gouveia, C.M.; Russo, A.C.; Bojariu, R.; Vicente-Serrano, S.M.; Trigo, R.M. Vegetation vulnerability to drought on southeastern Europe. Hydrol. Earth Syst. Sci. Discuss. 2018, 2018, 1–29. [Google Scholar] [CrossRef]
- Bussotti, F.; Papitto, G.; di Martino, D.; Cocciufa, C.; Cindolo, C.; Cenni, E.; Bettini, D.; Iacopetti, G.; Ghelardini, L.; Moricca, S.; et al. Extreme climatic events, biotic interactions and species-specific responses drive tree crown defoliation and mortality in Italian forests. IForest 2024, 17, 300–308. [Google Scholar] [CrossRef]
- Laoué, J.; Havaux, M.; Ksas, B.; Orts, J.P.; Reiter, I.M.; Fernandez, C.; Ormeno, E. A decade of rain exclusion in a Mediterranean forest reveals trade-offs of leaf chemical defenses and drought legacy effects. Sci. Rep. 2024, 14, 24119. [Google Scholar] [CrossRef]
- Iacopetti, G.; Selvi, F.; Bussotti, F.; Pollastrini, M.; Jucker, T.; Bouriaud, O. Tree diversity and identity modulate the growth response of thermophilous deciduous forests to climate warming. Oikos 2023, 2023, e08875. [Google Scholar] [CrossRef]
- Vanhellemont, M.; Sousa-Silva, R.; Maes, S.L.; van den Bulcke, J.; Hertzog, L.; de Groote, S.R.E.; van Acker, J.; Bonte, D.; Martel, A.; Lens, L.; et al. Distinct growth responses to drought for oak and beech in temperate mixed forests. Sci. Total Environ. 2019, 650, 3017–3026. [Google Scholar] [CrossRef]
- Rennenberg, H.; Loreto, F.; Polle, A.; Brilli, F.; Fares, S.; Beniwal, R.S.; Gessler, A. Physiological responses of forest trees to heat and drought. Plant Biol. 2006, 8, 556–571. [Google Scholar] [CrossRef]
- Teskey, R.; Wertin, T.; Bauweraerts, I.; Ameye, M.; McGuire, M.A.; Steppe, K. Responses of tree species to heat waves and extreme heat events. Plant Cell Environ. 2015, 38, 1699–1712. [Google Scholar] [CrossRef]
- Putzenlechner, B.; Koal, P.; Kappas, M.; Löw, M.; Mundhenk, P.; Tischer, A.; Wernicke, J.; Koukal, T. Towards precision forestry: Drought response from remote sensing-based disturbance monitoring and fine-scale soil information in Central Europe. Sci. Total Environ. 2023, 880, 163114. [Google Scholar] [CrossRef]
- Xu, C.; Liu, H.; Ciais, P.; Hartmann, H.; Camarero, J.J.; Wu, X.; Hammond, W.M.; Allen, C.D.; Chen, F. Enhanced Drought Exposure Increasingly Threatens More Forests Than Observed. Earth’s Future 2024, 12, e2023EF003705. [Google Scholar] [CrossRef]
- Zeng, Y.; Hao, D.; Huete, A.; Dechant, B.; Berry, J.; Chen, J.M.; Joiner, J.; Frankenberg, C.; Bond-Lamberty, B.; Ryu, Y.; et al. Optical vegetation indices for monitoring terrestrial ecosystems globally. Nat. Rev. Earth Environ. 2022, 3, 477–493. [Google Scholar] [CrossRef]
- Rouse, J.W., Jr.; Haas, R.H.; Schell, J.A.; Deering, D.W. Monitoring the Vernal Advancement and Retrogradation (Green Wave Effect) of Natural Vegetation (No. NASA-CR-132982). 1973. Available online: https://ntrs.nasa.gov/api/citations/19730017588/downloads/19730017588.pdf (accessed on 5 December 2025).
- Imran, H.A.; Gianelle, D.; Rocchini, D.; Dalponte, M.; Martín, M.P.; Sakowska, K.; Wohlfahrt, G.; Vescovo, L. VIS-NIR, Red-Edge and NIR-Shoulder Based Normalised Vegetation Indices Response to Co-Varying Leaf and Canopy Structural Traits in Heterogeneous Grasslands. Remote Sens. 2020, 12, 2254. [Google Scholar] [CrossRef]
- Castro, S.; Sanchez-Azofeifa, A. Testing of automated photochemical reflectance index sensors as proxy measurements of light use efficiency in an aspen forest. Sensors 2018, 18, 3302. [Google Scholar] [CrossRef]
- Gamon, J.A.; Wang, R.; Russo, S.E. Contrasting photoprotective responses of forest trees revealed using PRI light responses sampled with airborne imaging spectrometry. New Phytol. 2023, 238, 1318–1332. [Google Scholar] [CrossRef]
- Nakamura, Y.; Tsujimoto, K.; Ogawa, T.; Noda, H.M.; Hikosaka, K. Correction of photochemical reflectance index (PRI) by optical indices to predict non-photochemical quenching (NPQ) across various species. Remote Sens. Environ. 2024, 305, 114062. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, A.; Li, J.; Wu, J. Water content estimation of conifer needles using leaf-level hyperspectral data. Front. Plant Sci. 2024, 15, 1428212. [Google Scholar] [CrossRef]
- Liu, Q.; Jia, W.; Li, F. Determination of the most effective design for the measurement of photosynthetic light-response curves for planted Larix olgensis trees. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Roy, J.; Tardieu, F.; Tixier-Boichard, M.; Schurr, U. European infrastructures for sustainable agriculture. Nat. Plants 2017, 3, 756–758. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Xu, K.; Chen, R.; Chen, W.; Li, F.; Lv, C. Heat waves in summer 2022 and increasing concern regarding heat waves in general. Atmos. Ocean. Sci. Lett. 2023, 16, 100290. [Google Scholar] [CrossRef]
- Tripathy, K.P.; Mishra, A.K. How Unusual Is the 2022 European Compound Drought and Heatwave Event? Geophys. Res. Lett. 2023, 50, e2023GL105453. [Google Scholar] [CrossRef]
- Feser, F.; van Garderen, L.; Hansen, F. The Summer Heatwave 2022 over Western Europe: An Attribution to Anthropogenic Climate Change. Bull. Am. Meteorol. Soc. 2024, 105, E2175–E2179. [Google Scholar] [CrossRef]
- Faranda, D.; Pascale, S.; Bulut, B. Persistent anticyclonic conditions and climate change exacerbated the exceptional 2022 European-Mediterranean drought. Environ. Res. Lett. 2023, 18, 034030. [Google Scholar] [CrossRef]
- Choler, P. Above-treeline ecosystems facing drought: Lessons from the 2022 European summer heat wave. Biogeosciences 2023, 20, 4259–4272. [Google Scholar] [CrossRef]
- Kellerer-Pirklbauer, A.; Eulenstein, J. The summer heatwave in 2022 and its role in changing permafrost and periglacial conditions at a historic mountain pass in the Eastern Alps (Hochtor, Hohe Tauern Range, Austria). Permafr. Periglac. Process. 2023, 34, 547–565. [Google Scholar] [CrossRef] [PubMed]
- Lichtenthaler, H.K.; Buschmann, C. Chlorophylls and Carotenoids: Measurement and Characterisation by UV-VIS Spectroscopy. Curr. Protoc. Food Anal. Chem. 2001, 1, F4.3.1–F4.3.8. [Google Scholar] [CrossRef]
- Moncholi-Estornell, A.; Cendrero-Mateo, M.P.; Antala, M.; Cogliati, S.; Moreno, J.; van Wittenberghe, S. Enhancing Solar-Induced Fluorescence Interpretation: Quantifying Fractional Sunlit Vegetation Cover Using Linear Spectral Unmixing. Remote Sens. 2023, 15, 4274. [Google Scholar] [CrossRef]
- Evangelides, C.; Nobajas, A. Red-Edge Normalised Difference Vegetation Index (NDVI705) from Sentinel-2 imagery to assess post-fire regeneration. Remote Sens. Appl. Soc. Environ. 2020, 17, 100283. [Google Scholar] [CrossRef]
- Zhang, H.; Li, J.; Liu, Q.; Lin, S.; Huete, A.; Liu, L.; Croft, H.; Clevers, J.G.P.W.; Zeng, Y.; Wang, X.; et al. A novel red-edge spectral index for retrieving the leaf chlorophyll content. Methods Ecol. Evol. 2022, 13, 2771–2787. [Google Scholar] [CrossRef]
- Drolet, G.G.; Huemmrich, K.F.; Hall, F.G.; Middleton, E.M.; Black, T.A.; Barr, A.G.; Margolis, H.A. A MODIS-derived photochemical reflectance index to detect inter-annual variations in the photosynthetic light-use efficiency of a boreal deciduous forest. Remote Sens. Environ. 2005, 98, 212–224. [Google Scholar] [CrossRef]
- Sukhova, E.; Sukhov, V. Connection of the photochemical reflectance index (PRI) with the photosystem II quantum yield and non-photochemical quenching can be dependent on variations of photosynthetic parameters among investigated plants: A meta-analysis. Remote Sens. 2018, 10, 771. [Google Scholar] [CrossRef]
- Gao, B.-C. NDWI—A normalised difference water index for remote sensing of vegetation liquid water from space. Remote Sens. Environ. 1996, 58, 257–266. [Google Scholar] [CrossRef]
- Babar, M.A.; Reynolds, M.P.; van Ginkel, M.; Klatt, A.R.; Raun, W.R.; Stone, M.L. Spectral reflectance indices as a potential indirect selection criteria for wheat yield under irrigation. Crop Sci. 2006, 46, 578–588. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2025; Available online: https://www.R-project.org/ (accessed on 5 December 2025).
- Martini, D.; Sakowska, K.; Wohlfahrt, G.; Pacheco-Labrador, J.; van der Tol, C.; Porcar-Castell, A.; Magney, T.S.; Carrara, A.; Colombo, R.; El-Madany, T.S.; et al. Heatwave breaks down the linearity between sun-induced fluorescence and gross primary production. New Phytol. 2022, 233, 2415–2428. [Google Scholar] [CrossRef]
- Jia, Q.; Liu, Z.; Guo, C.; Wang, Y.; Yang, J.; Yu, Q.; Wang, J.; Zheng, F.; Lu, X. Relationship between Photosynthetic CO2 Assimilation and Chlorophyll Fluorescence for Winter Wheat under Water Stress. Plants 2023, 12, 3365. [Google Scholar] [CrossRef]
- Nour, M.M.; Aljabi, H.R.; AL-Huqail, A.A.; Hornebur, B.; Mohammed, A.E.; Alotaibi, M.O. Drought responses andadaptation in plants differing in life-form. Front. Ecol. Evol. 2024, 12, 1452427. [Google Scholar] [CrossRef]
- Tognetti, R.; Johnson, J.D.; Michelozzi, M. The response of European beech (Fagus sylvatica L.) seedlings from two Italian populations to drought and recovery. Trees 1995, 9, 348–354. [Google Scholar] [CrossRef]
- Sellers, P.J. Canopy reflectance, photosynthesis and transpiration. Int. J. Remote Sens. 1985, 6, 1335–1372. [Google Scholar] [CrossRef]
- Gao, X.; Huete, A.R.; Ni, W.; Miura, T. Optical–Biophysical Relationships of Vegetation Spectra without Background Contamination. Remote Sens. Environ. 2000, 74, 609–620. [Google Scholar] [CrossRef]
- Leuschner, C. Drought response of European beech (Fagus sylvatica L.)—A review. Perspect. Plant Ecol. Evol. Syst. 2020, 47, 125576. [Google Scholar] [CrossRef]
- Buddenbaum, H.; Stern, O.; Paschmionka, B.; Hass, E.; Gattung, T.; Stoffels, J.; Hill, J.; Werner, W. Using VNIR and SWIR field imaging spectroscopy for drought stress monitoring of beech seedlings. Int. J. Remote Sens. 2015, 36, 4590–4605. [Google Scholar] [CrossRef]
- Dalponte, M.; Andreatta, D.; Coomes, D.A.; Marchesini, L.B.; Marinelli, D.; Vescovo, L.; Gianelle, D. Canopy spectral responses of temperate forests to late spring frost and hot drought events assessed with Sentinel-2 NDVI time series. Remote Sens. Appl. Soc. Environ. 2025, 40, 101737. [Google Scholar] [CrossRef]
- Wang, R.; Gamon, J.A.; Russo, S.E.; Nishimwe, A.V.; Ellerman, H.; Wardlow, B. Multitemporal airborne imaging spectrometry and fluorometry reveal contrasting photoprotective responses of trees. Remote Sens. Environ. 2024, 311, 114295. [Google Scholar] [CrossRef]
- Michel, A.; Haggenmüller, K.; Kirchner, T.; Prescher, A.K.; Schwärzel, K. (Eds.) Forest Condition in Europe: The 2025 Assessment. In ICP Forests Technical Report Under the UNECE Convention on Long-Range Transboundary Air Pollution (Air Convention); Thünen Institute: Eberswalde, Germany, 2025; 100p. [Google Scholar] [CrossRef]
- Thomas, F.M.; Blank, R.; Hartmann, G. Abiotic and biotic factors and their interactions as causes of oak decline in Central Europe. For. Pathol. 2002, 32, 277–307. [Google Scholar] [CrossRef]
- Bigler, C.; Bräker, O.U.; Bugmann, H.; Dobbertin, M.; Rigling, A. Drought as an Inciting Mortality Factor in Scots Pine Stands of the Valais, Switzerland. Ecosystems 2006, 9, 330–343. [Google Scholar] [CrossRef]
- Liepiņš, K.; Bleive, A. The Potential of European Beech (Fagus sylvatica L.) in the Hemiboreal Baltic Region: A Review. Forests 2025, 16, 109. [Google Scholar] [CrossRef]
- Machar, I.; Vlckova, V.; Bucek, A.; Vozenilek, V.; Salek, L.; Jerabkova, L. Modelling of Climate Conditions in Forest Vegetation Zones as a Support Tool for Forest Management Strategy in European Beech Dominated Forests. Forests 2017, 8, 82. [Google Scholar] [CrossRef]
- Mathes, T.; Seidel, D.; Klemmt, H.J.; Thom, T.; Annighöfer, P. The effect of forest structure on drought stress in beech forests (Fagus sylvatica L.). For. Ecol. Manag. 2024, 554, 121667. [Google Scholar] [CrossRef]
- Mölder, I.; Leuschner, C. European beech grows better and is less drought sensitive in mixed than in pure stands: Tree neighbourhood effects on radial increment. Trees 2014, 28, 777–792. [Google Scholar] [CrossRef]
- Schmied, G.; Pretzsch, H.; Ambs, D.; Uhl, E.; Schmucker, J.; Fäth, J.; Biber, P.; Hoffmann, Y.-D.; Šeho, M.; Mellert, K.H.; et al. Rapid beech decline under recurrent drought stress: Individual neighborhood structure and soil properties matter. For. Ecol. Manag. 2023, 545, 121305. [Google Scholar] [CrossRef]

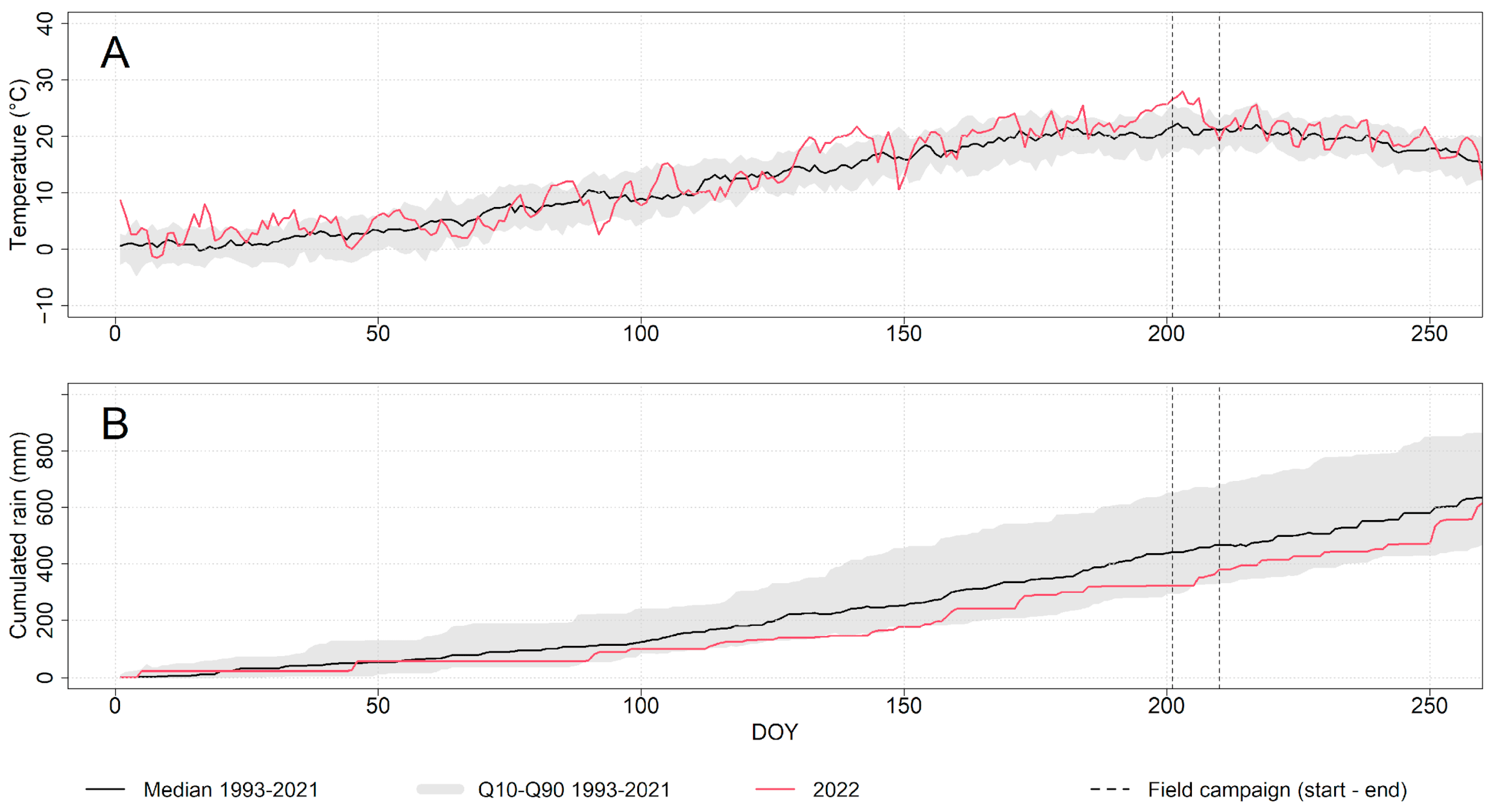


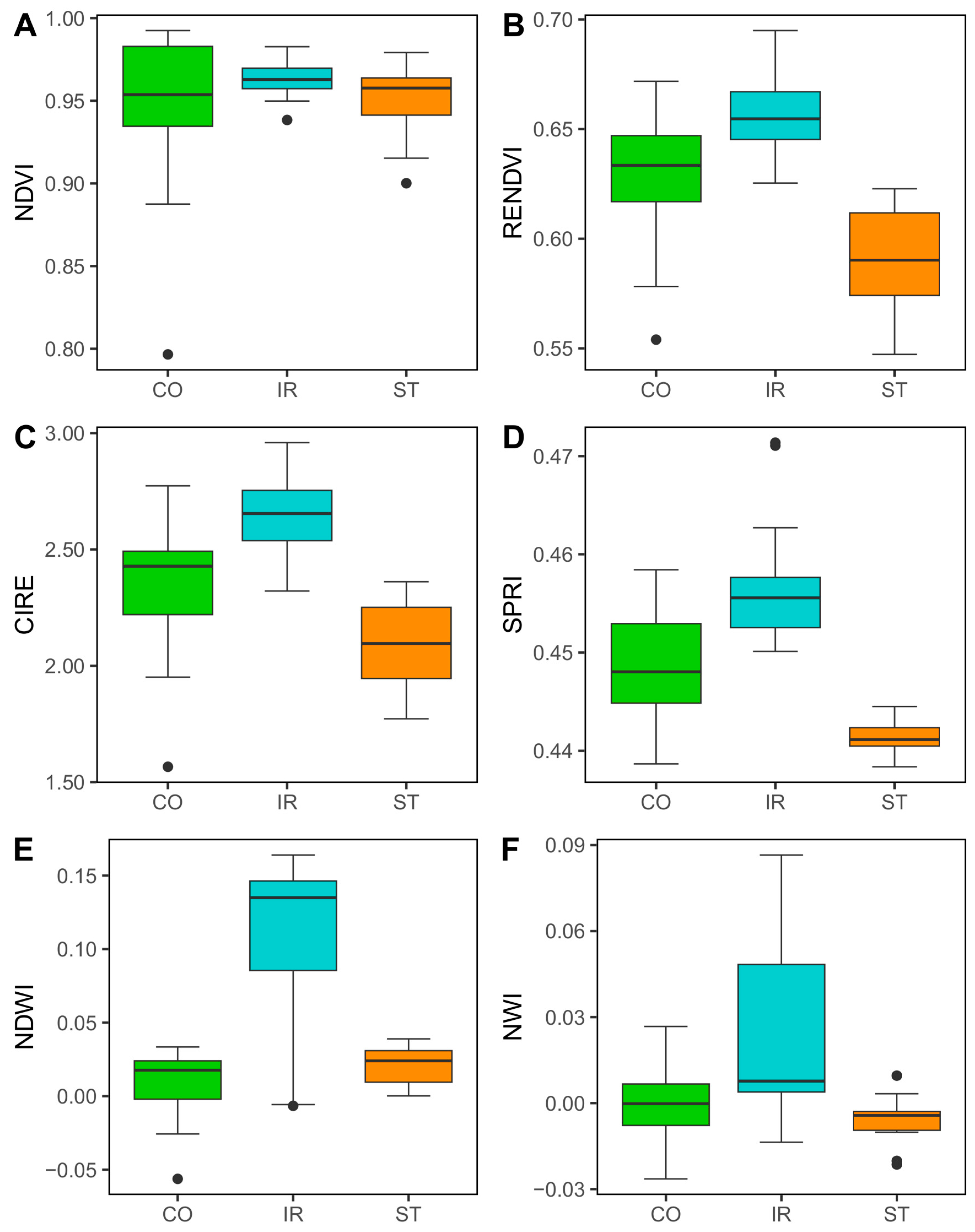
| Treatment (Tree Type) | Treatment Acronym | Tree Code | DBH (cm) | Tree Height (m) | Mean Tree Crown Diameter (m) |
|---|---|---|---|---|---|
| Control (control trees, untreated and facing the CDHW event of 2022) | CO | CO1 | 23.0 | 20.0 | 7.0 |
| CO2 | 30.5 | 19.5 | 6.5 | ||
| Irrigation (irrigated tree, facing the CDHW event of 2022 and supplemented with water) | IR | IR1 | 29.5 | 19.2 | 5.2 |
| Water stress (water-stressed tree, facing the CDHW event of 2022 in addition to water stress imposed through a longer-term manipulation experiment) | ST | ST1 | 27.3 | 20.1 | 5.2 |
| Vegetation Index | Equation | Reference |
|---|---|---|
| Normalised Difference Vegetation Index | [39] | |
| Red-Edge Normalised Difference Vegetation Index | [40] | |
| Chlorophyll Index—Red-Edge | [41] | |
| Scaled Photochemical Reflectance Index | where | [42,43] |
| Normalised Difference Water Index | [44] | |
| Normalised Water Index | [45] |
| Treatment | |||
|---|---|---|---|
| CO | IR | ST | |
| Tr (mmol H2O m−2 s−1) | 4.79 ± 1.01 | 8.19 ± 0.693 | 2.99 ± 0.63 |
| Asat (µmol CO2 m−2 s−1) | 12.24 ± 3.39 | 18.14 ± 3.07 | 4.99 ± 2.34 |
| gs (mmol m−2 s−1) | 250.54 ± 63.39 | 532.63 ± 71.30 | 132.0 ± 33.63 |
| Tleaf (°C) | 28.45 ± 0.88 | 29.04 ± 1.18 | 28.54 ± 0.97 |
| Treatment | |||
|---|---|---|---|
| CO | IR | ST | |
| CHI | 37.48 ± 5.87 | 39.05 ± 2.73 | 27.79 ± 4.28 |
| Total Chl (mg/g) | 5.98 ± 1.06 | 5.84 ± 0.87 | 3.86 ± 0.94 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sakowska, K.; Belelli Marchesini, L.; Dalponte, M.; Elfahl, M.; Rodeghiero, M.; Ugolini, F.; Pilati, S.; Vescovo, L.; Alonso Chorda, L.; Torresan, C. Physiological and Hyperspectral Responses of Individual European Beech Trees to Drought Stress: A Pilot Study During a Compound Drought and Heatwave Event. Remote Sens. 2026, 18, 488. https://doi.org/10.3390/rs18030488
Sakowska K, Belelli Marchesini L, Dalponte M, Elfahl M, Rodeghiero M, Ugolini F, Pilati S, Vescovo L, Alonso Chorda L, Torresan C. Physiological and Hyperspectral Responses of Individual European Beech Trees to Drought Stress: A Pilot Study During a Compound Drought and Heatwave Event. Remote Sensing. 2026; 18(3):488. https://doi.org/10.3390/rs18030488
Chicago/Turabian StyleSakowska, Karolina, Luca Belelli Marchesini, Michele Dalponte, Mustafa Elfahl, Mirco Rodeghiero, Francesca Ugolini, Stefania Pilati, Loris Vescovo, Luis Alonso Chorda, and Chiara Torresan. 2026. "Physiological and Hyperspectral Responses of Individual European Beech Trees to Drought Stress: A Pilot Study During a Compound Drought and Heatwave Event" Remote Sensing 18, no. 3: 488. https://doi.org/10.3390/rs18030488
APA StyleSakowska, K., Belelli Marchesini, L., Dalponte, M., Elfahl, M., Rodeghiero, M., Ugolini, F., Pilati, S., Vescovo, L., Alonso Chorda, L., & Torresan, C. (2026). Physiological and Hyperspectral Responses of Individual European Beech Trees to Drought Stress: A Pilot Study During a Compound Drought and Heatwave Event. Remote Sensing, 18(3), 488. https://doi.org/10.3390/rs18030488









