From Remote Sensing to Species Distribution Modelling: An Integrated Workflow to Monitor Spreading Species in Key Grassland Habitats
Abstract
1. Introduction
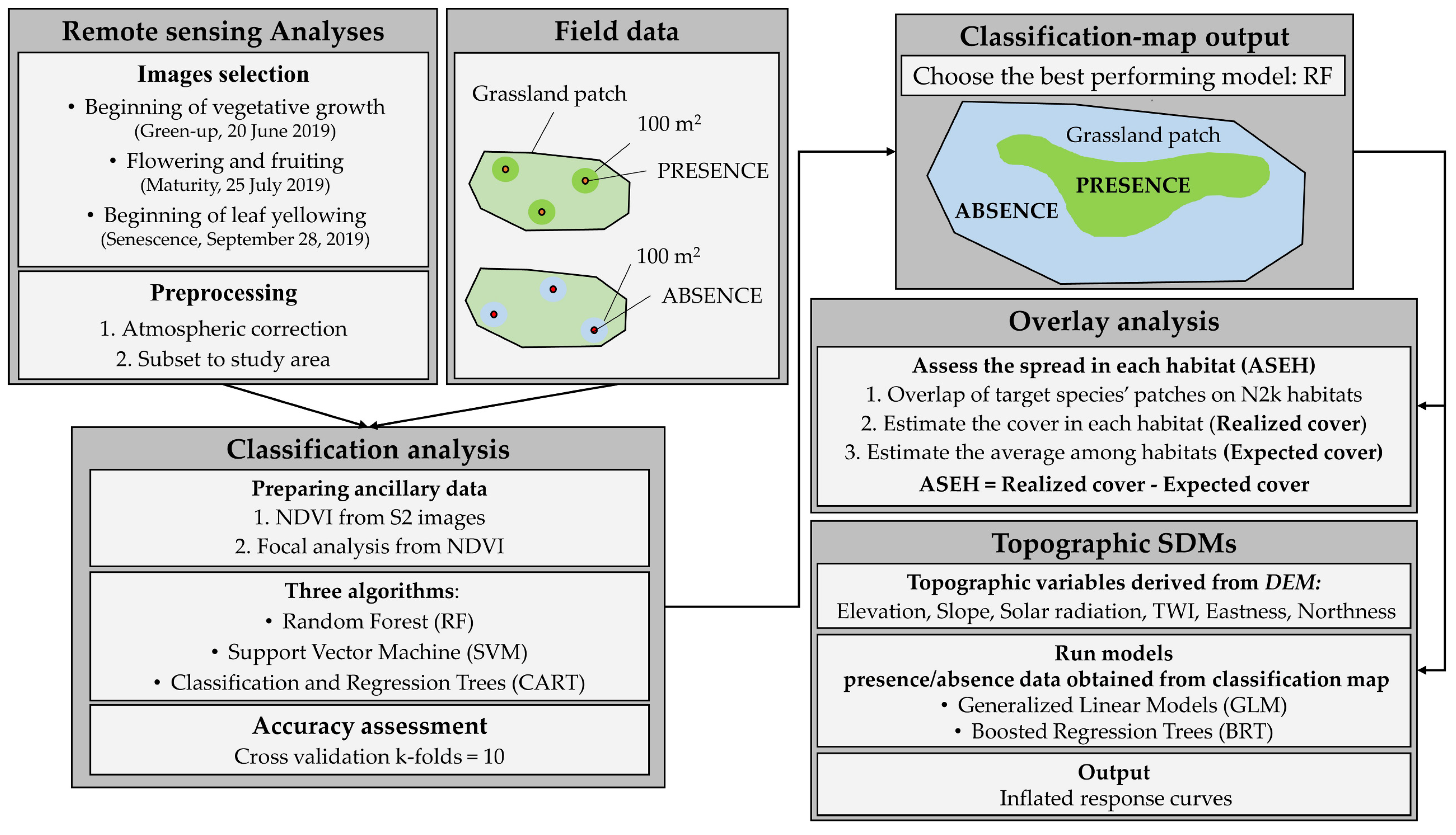
2. Materials and Methods
2.1. Study Area
2.2. Remote Sensing Dataset and Pre-Processing
2.3. Data Collection
2.4. Classification Analysis by Machine Learning
2.5. Accuracy Assessment
2.6. Conversion of Plant Associations into NATURA 2000 Habitats
2.7. GIS Analysis of the Spread of B. genuense on Habitats NATURA 2000
2.8. Topographic SDMs
3. Results
3.1. Classification Output and Accuracy Assessment
3.2. Conversion of Plant Associations into Natura 2000 Habitats
3.3. Overlay Analysis Output
3.4. Topographic Model Output
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Habel, J.C.; Dengler, J.; Janišová, M.; Török, P.; Wellstein, C.; Wiezik, M. European grassland ecosystems: Threatened hotspots of biodiversity. Biodivers. Conserv. 2013, 22, 2131–2138. [Google Scholar] [CrossRef]
- European Commission. Interpretation Manual of European Union Habitats; EC DG Environment: Brussels, Belgium, 2013. [Google Scholar]
- Millennium Ecosystem Assessment. Ecosystems and Human Well-Being; Island Press: Washington, DC, USA, 2005; Volume 5. [Google Scholar]
- Köhler, B.; Gigon, A.; Edwards, P.J.; Krüsi, B.; Langenauer, R.; Lüscher, A.; Ryser, P. Changes in the Species Composition and Conservation Value of Limestone Grasslands in Northern Switzerland after 22 Years of Contrasting Managements. Perspect. Plant Ecol. Evol. Syst. 2005, 7, 51–67. [Google Scholar] [CrossRef]
- Rusina, S.; Kuzemko, A. EDGG Cooperation on Syntaxonomy and Biodiversity of Festuco-Brometea Communities in Transylvania (Romania): Report and Pre-Liminary Results. Bull. Eur. Dry Grassl. Group 2009, 4, 13–19. [Google Scholar]
- Nagendra, H.; Lucas, R.; Honrado, J.P.; Jongman, R.H.; Tarantino, C.; Adamo, M.; Mairota, P. Remote Sensing for Conservation Monitoring: Assessing Protected Areas, Habitat Extent, Habitat Condition, Species Diversity, and Threats. Ecol. Indic. 2013, 33, 45–59. [Google Scholar] [CrossRef]
- Wang, R.; Gamon, J.A. Remote Sensing of Terrestrial Plant Biodiversity. Remote Sens. Environ. 2019, 231, 111218. [Google Scholar] [CrossRef]
- De Simone, W.; Di Musciano, M.; Di Cecco, V.; Ferella, G.; Frattaroli, A.R. The Potentiality of Sentinel-2 to Assess the Effect of Fire Events on Mediterranean Mountain Vegetation. Plant Sociol. 2020, 57, 11–22. [Google Scholar] [CrossRef]
- Shahabi, H.; Shirzadi, A.; Ghaderi, K.; Omidvar, E.; Al-Ansari, N.; Clague, J.J.; Geertsema, M.; Khosravi, K.; Amini, A.; Bahrami, S. Flood Detection and Susceptibility Mapping Using Sentinel-1 Remote Sensing Data and a Machine Learning Approach: Hybrid Intelligence of Bagging Ensemble Based on K-Nearest Neighbor Classifier. Remote Sens. 2020, 12, 266. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, S.; Zhang, H.; Bai, Y.; Zhang, J. Monitoring Drought Using Composite Drought Indices Based on Remote Sensing. Sci. Total Environ. 2020, 711, 134585. [Google Scholar] [CrossRef]
- Iannella, M.; De Simone, W.; D’Alessandro, P.; Console, G.; Biondi, M. Investigating the Current and Future Co-Occurrence of Ambrosia Artemisiifolia and Ophraella Communa in Europe through Ecological Modelling and Remote Sensing Data Analysis. Int. J. Environ. Res. Public. Health 2019, 16, 3416. [Google Scholar] [CrossRef]
- Iannella, M.; D’Alessandro, P.; Longo, S.; Biondi, M. New Records and Potential Distribution by Ecological Niche Modelling of the Adventive Leaf Beetle Monoxia Obesula Blake in the Mediterranean Area (Coleoptera, Chrysomelidae, Galerucinae). Bull. Insectology 2019, 72, 135–142. [Google Scholar]
- Geldmann, J.; Joppa, L.N.; Burgess, N.D. Mapping Change in Human Pressure Globally on Land and within Protected Areas. Conserv. Biol. 2014, 28, 1604–1616. [Google Scholar] [CrossRef]
- Iannella, M.; Liberatore, L.; Biondi, M. The Effects of a Sudden Urbanization on Micromammal Communities: A Case Study of Post-Earthquake L’Aquila (Abruzzi Region, Italy). Ital. J. Zool. 2016, 83, 255–262. [Google Scholar] [CrossRef][Green Version]
- De Simone, W.; Iannella, M.; D’Alessandro, P.; Biondi, M. Assessing Influence in Biofuel Production and Ecosystem Services When Environmental Changes Affect Plant–Pest Relationships. GCB Bioenergy 2020, 12, 864–877. [Google Scholar] [CrossRef]
- Knick, S.T.; Rotenberry, J.T.; Zarriello, T.J. Supervised Classification of Landsat Thematic Mapper Imagery in a Semi-Arid Rangeland by Nonparametric Discriminant Analysis. Photogramm. Eng. Remote Sens. 1997, 63, 79–86. [Google Scholar]
- Zha, Y.; Gao, J.; Ni, S.; Liu, Y.; Jiang, J.; Wei, Y. A Spectral Reflectance-Based Approach to Quantification of Grassland Cover from Landsat TM Imagery. Remote Sens. Environ. 2003, 87, 371–375. [Google Scholar] [CrossRef]
- Lehnert, L.W.; Meyer, H.; Wang, Y.; Miehe, G.; Thies, B.; Reudenbach, C.; Bendix, J. Retrieval of Grassland Plant Coverage on the Tibetan Plateau Based on a Multi-Scale, Multi-Sensor and Multi-Method Approach. Remote Sens. Environ. 2015, 164, 197–207. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, H.; Shen, Q. Spectral–Spatial Classification of Hyperspectral Imagery with 3D Convolutional Neural Network. Remote Sens. 2017, 9, 67. [Google Scholar] [CrossRef]
- Griffiths, P.; Nendel, C.; Pickert, J.; Hostert, P. Towards National-Scale Characterization of Grassland Use Intensity from Integrated Sentinel-2 and Landsat Time Series. Remote Sens. Environ. 2020, 238, 111124. [Google Scholar] [CrossRef]
- Rapinel, S.; Mony, C.; Lecoq, L.; Clement, B.; Thomas, A.; Hubert-Moy, L. Evaluation of Sentinel-2 Time-Series for Mapping Floodplain Grassland Plant Communities. Remote Sens. Environ. 2019, 223, 115–129. [Google Scholar] [CrossRef]
- Odindi, J.; Mutanga, O.; Rajah, P. Assessing the Synergistic Potential of Sentinel-2 Spectral Reflectance Bands and Derived Vegetation Indices for Detecting and Mapping Invasive Alien Plant Species. S. Afr. J. Geomat. 2020, 9, 75–88. [Google Scholar]
- Guisan, A.; Thuiller, W.; Zimmermann, N.E. Habitat Suitability and Distribution Models: With Applications in R; Cambridge University Press: Cambridge, UK, 2017; ISBN 0-521-76513-7. [Google Scholar]
- Elith, J.; Leathwick, J.R. Species Distribution Models: Ecological Explanation and Prediction across Space and Time. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 677–697. [Google Scholar] [CrossRef]
- Di Musciano, M.; Di Cecco, V.; Bartolucci, F.; Conti, F.; Frattaroli, A.R.; Di Martino, L. Dispersal Ability of Threatened Species Affects Future Distributions. Plant Ecol. 2020, 221, 265–281. [Google Scholar] [CrossRef]
- Reddy, S.; Dávalos, L.M. Geographical Sampling Bias and Its Implications for Conservation Priorities in Africa. J. Biogeogr. 2003, 30, 1719–1727. [Google Scholar] [CrossRef]
- Iannella, M.; D’Alessandro, P.; Biondi, M. Entomological Knowledge in Madagascar by GBIF Datasets: Estimates on the Coverage and Possible Biases (Insecta). Fragm. Entomol. 2019, 51, 1–10. [Google Scholar] [CrossRef]
- Graham, C.H.; Elith, J.; Hijmans, R.J.; Guisan, A.; Peterson, A.T.; Loiselle, B.A. The Influence of Spatial Errors in Species Occurrence Data Used in Distribution Models. J. Appl. Ecol. 2008, 45, 239–247. [Google Scholar] [CrossRef]
- He, Y.; Chen, G.; Potter, C.; Meentemeyer, R.K. Integrating Multi-Sensor Remote Sensing and Species Distribution Modeling to Map the Spread of Emerging Forest Disease and Tree Mortality. Remote Sens. Environ. 2019, 231, 111238. [Google Scholar] [CrossRef]
- Bonanomi, G.; Caporaso, S.; Allegrezza, M. Short-Term Effects of Nitrogen Enrichment, Litter Removal and Cutting on a Mediterranean Grassland. Acta Oecol. 2006, 30, 419–425. [Google Scholar] [CrossRef]
- Bonanomi, G.; Caporaso, S.; Allegrezza, M. Effects of Nitrogen Enrichment, Plant Litter Removal and Cutting on a Species-rich Mediterranean Calcareous Grassland. Plant Biosyst. 2009, 143, 443–455. [Google Scholar] [CrossRef]
- Allegrezza, M.; Ballelli, S.; Ciucci, V.; Mentoni, M.; Pesaresi, S. The Vegetation and the Plant Landscape of Monte Sassotetto (Sibillini Mountains, Central Apennines). Plant Sociol. 2014, 51, 59–87. [Google Scholar]
- Catorci, A.; Cesaretti, S.; Gatti, R.; Ottaviani, G. Abiotic and Biotic Changes due to Spread of Brachypodium Genuense (DC.) Roem. & Schult. in Sub-Mediterranean Meadows. Community Ecol. 2011, 12, 117–125. [Google Scholar]
- Tardella, F.M.; Bricca, A.; Piermarteri, K.; Postiglione, N.; Catorci, A. Context-Dependent Variation of SLA and Plant Height of a Dominant, Invasive Tall Grass (Brachypodium Genuense) in Sub-Mediterranean Grasslands. Flora 2017, 229, 116–123. [Google Scholar] [CrossRef]
- Buckland, S.M.; Thompson, K.; Hodgson, J.G.; Grime, J.P. Grassland Invasions: Effects of Manipulations of Climate and Management. J. Appl. Ecol. 2001, 38, 301–309. [Google Scholar] [CrossRef]
- Kosić, I.V.; Tardella, F.M.; Grbeša, D.; Škvorc, Ž.; Catorci, A. Effects of Abandonment on the Functional Composition and Forage Nutritive Value of a North Adriatic Dry Grassland Community (Ćićarija, Croatia). Appl. Ecol. Environ. Res. 2014, 12, 285. [Google Scholar] [CrossRef]
- Natura 2000 Network. Available online: https://ec.europa.eu/environment/nature/natura2000/index_en.htm (accessed on 24 January 2021).
- Cervellini, M.; Zannini, P.; Di Musciano, M.; Fattorini, S.; Jiménez-Alfaro, B.; Rocchini, D.; Field, R.; Vetaas, O.R.; Irl, S.D.H.; Beierkuhnlein, C.; et al. A grid-based map for the Biogeographical Regions of Europe. Biodivers. Data J. 2020, 8, e53720. [Google Scholar] [CrossRef]
- Calandra, R. I Suoli Di “Campo Imperatore” (Gran Sasso d’Italia). Braun-Blanquetia 1999, 16, 21–32. [Google Scholar]
- Conti, F.; Bartolucci, F. The Vascular Flora of Gran Sasso and Monti Della Laga National Park (Central Italy). Phytotaxa 2016, 256, 1–119. [Google Scholar] [CrossRef]
- Biondi, E.; Ballelli, S.; Allegrezza, M.; Taffetani, F.; Frattaroli, A.; Guitian, J.; Zuccarello, V. La Vegetazione Di Campo Imperatore (Gran Sasso d’Italia). In “Ricerche Di Geobotanica Ed Ecologia Vegetale Di Campo Imperatore (Gran Sasso d’Italia). Braun-Blanquetia 1999, 16, 53–119. [Google Scholar]
- Council Directive 2009/147/EEC. Available online: https://ec.europa.eu/environment/nature/legislation/birdsdirective/index_en.htm (accessed on 24 January 2021).
- Council Directive 92/43/EEC. Available online: https://ec.europa.eu/environment/nature/legislation/habitatsdirective/index_en.htm (accessed on 23 January 2021).
- Drusch, M.; Del Bello, U.; Carlier, S.; Colin, O.; Fernandez, V.; Gascon, F.; Hoersch, B.; Isola, C.; Laberinti, P.; Martimort, P.; et al. Sentinel-2: ESA’s Optical High-Resolution Mission for GMES Operational Services. Remote Sens. Environ. 2012, 120, 25–36. [Google Scholar] [CrossRef]
- Lim, C.H.; An, J.H.; Jung, S.H.; Nam, G.B.; Cho, Y.C.; Kim, N.S.; Lee, C.S. Ecological Consideration for Several Methodologies to Diagnose Vegetation Phenology. Ecol. Res. 2018, 33, 363–377. [Google Scholar] [CrossRef]
- Szantoi, Z.; Strobl, P. Copernicus Sentinel-2 Calibration and Validation. Eur. J. Remote Sens. 2019, 52, 253–255. [Google Scholar] [CrossRef]
- Louis, J.; Debaecker, V.; Pflug, B.; Main-Knorn, M.; Bieniarz, J.; Mueller-Wilm, U.; Cadau, E.; Gascon, F. Sentinel-2 Sen2cor: L2a Processor for Users. Proceedings Living Planet Symposium. 2016. Available online: https://elib.dlr.de/107381/1/LPS2016_sm10_3louis.pdf (accessed on 15 January 2021).
- Gascon, F.; Ramoino, F. Sentinel-2 Data Exploitation with ESA’s Sentinel-2 Toolbox. In Proceedings of the 19th European Geosciences Union (EGU) General Assembly, Vienna, Austria, 23–28 April 2017. [Google Scholar]
- Rouse, J.W.; Haas, R.W.; Deering, D.W.; Schell, J.A.; Harlan, J.C. Monitoring the vernal advancement and retrogradation (Greenwave effect) of natural vegetation. In NASA/GSFCT Type III Final Report; NASA: Washington, DC, USA, 1974. [Google Scholar]
- Viana-Soto, A.; Aguado, I.; Martínez, S. Assessment of Post-Fire Vegetation Recovery Using Fire Severity and Geographical Data in the Mediterranean Region (Spain). Environments 2017, 4, 90. [Google Scholar] [CrossRef]
- Wegmann, M.; Leutner, B.; Dech, S. Remote Sensing and GIS for Ecologists: Using Open Source Software; Pelagic Publishing Ltd.: Exeter, UK, 2016; ISBN 1-78427-024-5. [Google Scholar]
- Wood, E.M.; Pidgeon, A.M.; Radeloff, V.C.; Keuler, N.S. Image Texture as a Remotely Sensed Measure of Vegetation Structure. Remote Sens. Environ. 2012, 121, 516–526. [Google Scholar] [CrossRef]
- Catonica, C.; Tinti, D.; De Bonis, L.; Di Santo, D.; Calzolaio, A.; De Paulis, S. Carta Della Natura per La Zonazione Del Piano Del Parco Nazionale Del Gran Sasso e Monti Della Laga; Ente Parco Nazionale del Gran Sasso e Monti della Laga: Rome, Italy, 2015. Available online: https://www.isprambiente.gov.it/it/servizi/sistema-carta-della-natura/la-carta-della-natura-di-interesse-locale-1/la-carta-della-natura-del-parco-nazionale-del-gran-sasso-e-dei-monti-della-laga (accessed on 18 January 2021).
- Congedo, L.; Sallustio, L.; Munafò, M.; Ottaviano, M.; Tonti, D.; Marchetti, M. Copernicus High-Resolution Layers for Land Cover Classification in Italy. J. Maps 2016, 12, 1195–1205. [Google Scholar] [CrossRef]
- European Environmental Agency. CORINE Land Cover—Copernicus Land Monitoring Service. Available online: https://land.copernicus.eu/pan-european/corine-land-cover (accessed on 19 January 2020).
- Tilahun, A. Accuracy Assessment of Land Use Land Cover Classification Using Google Earth. Am. J. Environ. Prot. 2015, 4, 193. [Google Scholar] [CrossRef]
- Muñoz-Marí, J.; Bruzzone, L.; Camps-Valls, G. A Support Vector Domain Description Approach to Supervised Classification of Remote Sensing Images. IEEE Trans. Geosci. Remote Sens. 2007, 45, 2683–2692. [Google Scholar] [CrossRef]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Noble, W.S. What Is a Support Vector Machine? Nat. Biotechnol. 2006, 24, 1565–1567. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Kumar, V. The Top Ten Algorithms in Data Mining; CRC Press: Boca Raton, FL, USA, 2009; ISBN 978-1-4200-8965-3. [Google Scholar]
- Leutner, B.; Horning, N.; Schwalb-Willmann, J.; Hijmans, R.J. RStoolbox: Tools for Remote Sensing Data Analysis, 0.2.6. 2020. Available online: https://github.com/bleutner/RStoolbox (accessed on 10 December 2020).
- R Core Team. R: A Language and Environment for Statistical Computing, 3.6.3; R Core Team: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 7 December 2020).
- Li, C.; Wang, J.; Wang, L.; Hu, L.; Gong, P. Comparison of Classification Algorithms and Training Sample Sizes in Urban Land Classification with Landsat Thematic Mapper Imagery. Remote Sens. 2014, 6, 964–983. [Google Scholar] [CrossRef]
- Meyer, H.; Pebesma, E. Predicting into Unknown Space? Estimating the Area of Applicability of Spatial Prediction Models. arXiv 2020, arXiv:200507939. [Google Scholar]
- Meyer, H.; Reudenbach, C.; Wöllauer, S.; Nauss, T. Importance of Spatial Predictor Variable Selection in Machine Learning Applications–Moving from Data Reproduction to Spatial Prediction. Ecol. Model. 2019, 411, 108815. [Google Scholar] [CrossRef]
- Meyer, H. CAST: ‘caret’ Applications for Spatial-Temporal Models. R Package Version 0.4.2. Available online: https://CRAN.R-project.org/package=CAST (accessed on 11 December 2020).
- Anselin, L. Local Indicators of Spatial Association—LISA. Geogr. Anal. 1995, 27, 93–115. [Google Scholar] [CrossRef]
- Biondi, E.; Blasi, C.; Burrascano, S.; Casavecchia, S.; Copiz, R.; Del Vico, E.; Galdenzi, D.; Gigante, D.; Lasen, C.; Spampinato, G.; et al. Italian Interpretation Manual of the 92/43/EEC Directive Habitats. Ministero dell’Ambiente e della Tutela del Territorio e del Mare, 2009; Available online: http://vnr.unipg.it/habitat/ (accessed on 21 January 2021).
- QGIS Development Team. QGIS Geographic Information System; Version 3.14; Open Source Geospatial Foundation: Beaverton, OR, USA, 2020. [Google Scholar]
- Erdős, L.; Bátori, Z. The Moving Split Window (MSW) Analysis in Vegetation Science–An Overview. Appl. Ecol. Environ. Res. 2014, 12, 787–805. [Google Scholar] [CrossRef]
- Chang, Y.; Bu, R.; Hu, Y.; Xu, C.; Wang, Q. Detecting Forest Landscape Boundary between Mountain Birch and Evergreen Coniferous Forest in the Northern Slope of Changbai Mountain. J. Environ. Sci. 2003, 15, 182–186. [Google Scholar]
- Jacquez, G.M.; Maruca, S.; Fortin, M.-J. From Fields to Objects: A Review of Geographic Boundary Analysis. J. Geogr. Syst. 2000, 2, 221–241. [Google Scholar] [CrossRef]
- Elith, J.; Graham, C.H.; Anderson, R.P.; Dudík, M.; Ferrier, S.; Guisan, A.; Hijmans, R.J.; Huettmann, F.; Leathwick, J.R.; Lehmann, A. Novel Methods Improve Prediction of Species’ Distributions from Occurrence Data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Dormann, C.F.; Elith, J.; Bacher, S.; Buchmann, C.; Carl, G.; Carré, G.; Marquéz, J.R.G.; Gruber, B.; Lafourcade, B.; Leitão, P.J. Collinearity: A Review of Methods to Deal with It and a Simulation Study Evaluating Their Performance. Ecography 2013, 36, 27–46. [Google Scholar] [CrossRef]
- Naimi, B.; Hamm, N.A.; Groen, T.A.; Skidmore, A.K.; Toxopeus, A.G. Where Is Positional Uncertainty a Problem for Species Distribution Modelling? Ecography 2014, 37, 191–203. [Google Scholar] [CrossRef]
- Iannella, M.; Cerasoli, F.; D’Alessandro, P.; Console, G.; Biondi, M. Coupling GIS Spatial Analysis and Ensemble Niche Modelling to Investigate Climate Change-Related Threats to the Sicilian Pond Turtle Emys Trinacris, an Endangered Species from the Mediterranean. PeerJ 2018, 6, e4969. [Google Scholar] [CrossRef] [PubMed]
- Leathwick, J.R.; Rowe, D.; Richardson, J.; Elith, J.; Hastie, T. Using Multivariate Adaptive Regression Splines to Predict the Distributions of New Zealand’s Freshwater Diadromous Fish. Freshw. Biol. 2005, 50, 2034–2052. [Google Scholar] [CrossRef]
- Elith, J.; Leathwick, J.R.; Hastie, T. A Working Guide to Boosted Regression Trees. J. Anim. Ecol. 2008, 77, 802–813. [Google Scholar] [CrossRef] [PubMed]
- Thuiller, W.; Georges, D.; Engler, R.; Breiner, F.; Georges, M.D.; Thuiller, C.W. Package ‘Biomod2’: Ensemble Platform for Species Distribution Modeling. Available online: https://cran.r-project.org/web/packages/biomod2/biomod2.pdf (accessed on 9 December 2020).
- Di Cola, V.; Broennimann, O.; Petitpierre, B.; Breiner, F.T.; D’Amen, M.; Randin, C.; Engler, R.; Pottier, J.; Pio, D.; Dubuis, A. Ecospat: An R Package to Support Spatial Analyses and Modeling of Species Niches and Distributions. Ecography 2017, 40, 774–787. [Google Scholar] [CrossRef]
- Mattivi, P.; Franci, F.; Lambertini, A.; Bitelli, G. TWI Computation: A Comparison of Different Open Source GISs. Open Geospat. Data Softw. Stand. 2019, 4, 1–12. [Google Scholar] [CrossRef]
- Peterson, E.B. Mapping Percent-Cover of the Invasive Species Bromus Tectorum (Cheatgrass) over a Large Portion of Nevada from Satellite Imagery; Nevada Natural Heritage Program: Carson City, NV, USA, 2003. [Google Scholar]
- Wang, A.; Chen, J.; Jing, C.; Ye, G.; Wu, J.; Huang, Z.; Zhou, C. Monitoring the Invasion of Spartina Alterniflora from 1993 to 2014 with Landsat TM and SPOT 6 Satellite Data in Yueqing Bay, China. PLoS ONE 2015, 10, e0135538. [Google Scholar] [CrossRef] [PubMed]
- Kganyago, M.; Odindi, J.; Adjorlolo, C.; Mhangara, P. Evaluating the Capability of Landsat 8 OLI and SPOT 6 for Discriminating Invasive Alien Species in the African Savanna Landscape. Int. J. Appl. Earth Obs. Geoinf. 2018, 67, 10–19. [Google Scholar] [CrossRef]
- Tian, Y.; Jia, M.; Wang, Z.; Mao, D.; Du, B.; Wang, C. Monitoring Invasion Process of Spartina Alterniflora by Seasonal Sentinel-2 Imagery and an Object-Based Random Forest Classification. Remote Sens. 2020, 12, 1383. [Google Scholar] [CrossRef]
- Rodriguez-Galiano, V.F.; Ghimire, B.; Rogan, J.; Chica-Olmo, M.; Rigol-Sanchez, J.P. An Assessment of the Effectiveness of a Random Forest Classifier for Land-Cover Classification. ISPRS J. Photogramm. Remote Sens. 2012, 67, 93–104. [Google Scholar] [CrossRef]
- Barrett, B.; Raab, C.; Cawkwell, F.; Green, S. Upland Vegetation Mapping Using Random Forests with Optical and Radar Satellite Data. Remote Sens. Ecol. Conserv. 2016, 2, 212–231. [Google Scholar] [CrossRef]
- Belgiu, M.; Drăguţ, L. Random Forest in Remote Sensing: A Review of Applications and Future Directions. ISPRS J. Photogramm. Remote Sens. 2016, 114, 24–31. [Google Scholar] [CrossRef]
- Stenzel, S.; Fassnacht, F.E.; Mack, B.; Schmidtlein, S. Identification of High Nature Value Grassland with Remote Sensing and Minimal Field Data. Ecol. Indic. 2017, 74, 28–38. [Google Scholar] [CrossRef]
- Grabska, E.; Hawryło, P.; Socha, J. Continuous Detection of Small-Scale Changes in Scots Pine Dominated Stands Using Dense Sentinel-2 Time Series. Remote Sens. 2020, 12, 1298. [Google Scholar] [CrossRef]
- Elkind, K.; Sankey, T.T.; Munson, S.M.; Aslan, C.E. Invasive Buffelgrass Detection Using High-resolution Satellite and UAV Imagery on Google Earth Engine. Remote Sens. Ecol. Conserv. 2019, 5, 318–331. [Google Scholar] [CrossRef]
- Corazza, M.; Tardella, F.M.; Ferrari, C.; Catorci, A. Tall Grass Invasion After Grassland Abandonment Influences the Availability of Palatable Plants for Wild Herbivores: Insight into the Conservation of the Apennine Chamois Rupicapra Pyrenaicaornata. Environ. Manag. 2016, 57, 1247–1261. [Google Scholar] [CrossRef]
- Catorci, A.; Cesaretti, S.; Tardella, F.M. Effect of Tall-Grass Invasion on the Flowering-Related Functional Pattern of Submediterranean Hay-Meadows. Plant Biosyst. Int. J. Deal. Asp. Plant Biol. 2014, 148, 1127–1137. [Google Scholar] [CrossRef]
- Catorci, A.; Ottaviani, G.; Kosić, I.V.; Cesaretti, S. Effect of Spatial and Temporal Patterns of Stress and Disturbance Intensities in a Sub-Mediterranean Grassland. Plant Biosyst. Int. J. Deal. Asp. Plant Biol. 2012, 146, 352–367. [Google Scholar] [CrossRef]
- Nagendra, H. Using Remote Sensing to Assess Biodiversity. Int. J. Remote Sens. 2001, 22, 2377–2400. [Google Scholar] [CrossRef]
- Feilhauer, H.; Dahlke, C.; Doktor, D.; Lausch, A.; Schmidtlein, S.; Schulz, G.; Stenzel, S. Mapping the Local Variability of Natura 2000 Habitats with Remote Sensing. Appl. Veg. Sci. 2014, 17, 765–779. [Google Scholar] [CrossRef]
- Cerrejón, C.; Valeria, O.; Mansuy, N.; Barbé, M.; Fenton, N.J. Predictive mapping of bryophyte richness patterns in boreal forests using species distribution models and remote sensing data. Ecol. Indic. 2020, 119, 106826. [Google Scholar] [CrossRef]
- Ahmed, N.; Atzberger, C.; Zewdie, W. Species Distribution Modelling performance and its implication for Sentinel-2-based prediction of invasive Prosopis juliflora in lower Awash River basin, Ethiopia. Ecol. Process. 2021, 10, 1–16. [Google Scholar] [CrossRef]
- Lassueur, T.; Joost, S.; Randin, C.F. Very High Resolution Digital Elevation Models: Do They Improve Models of Plant Species Distribution? Ecol. Model. 2006, 198, 139–153. [Google Scholar] [CrossRef]
- Zuccarello, V.; Allegrezza, M.; Biondi, E.; Calandra, R. Valenza ecologica di specie e di associazioni prative e modelli di distribuzione lungo gradienti sulla base della teoria degli insiemi sfocati (Fuzzy Set Theory). Braun Blanquetia 1999, 16, 121–225. [Google Scholar]
- Moeslund, J.E.; Arge, L.; Bøcher, P.K.; Dalgaard, T.; Ejrnæs, R.; Odgaard, M.V.; Svenning, J.-C. Topographically Controlled Soil Moisture Drives Plant Diversity Patterns within Grasslands. Biodivers. Conserv. 2013, 22, 2151–2166. [Google Scholar] [CrossRef]
- Pickett, S.T.A.; Bazzaz, F.A. Organization of an Assemblage of Early Successional Species on a Soil Moisture Gradient. Ecology 1978, 59, 1248–1255. [Google Scholar] [CrossRef]
- Buri, A.; Cianfrani, C.; Pradervand, J.N.; Guisan, A. Predicting Plant Distribution in an Heterogeneous Alpine Landscape: Does Soil Matter? In Proceedings of the EGU General Assembly Conference, Vienna, Austria, 17–22 April 2016. [Google Scholar]
- Bennie, J.; Huntley, B.; Wiltshire, A.; Hill, M.O.; Baxter, R. Slope, Aspect and Climate: Spatially Explicit and Implicit Models of Topographic Microclimate in Chalk Grassland. Ecol. Model. 2008, 216, 47–59. [Google Scholar] [CrossRef]
- Potter, K.A.; Arthur Woods, H.; Pincebourde, S. Microclimatic Challenges in Global Change Biology. Glob. Chang. Biol. 2013, 19, 2932–2939. [Google Scholar] [CrossRef]
- Dengler, J. Zwischen Estland Und Portugal–Gemeinsamkeiten Und Unterschiede Der Phytodiversitätsmuster Europäischer Trockenrasen. Tuexenia 2005, 25, 387–405. [Google Scholar]
- Galiè, M.; Casavecchia, S.; Galdenzi, D.; Gasparri, R.; Soriano, P.; Estrelles, E.; Biondi, E. Seed Germination Behavior of Two Brachypodium Species with a Key Role in the Improvement of Marginal Areas. Plant Sociol. 2013, 50, 91–107. [Google Scholar]
- Bricca, A.; Tardella, F.M.; Tolu, F.; Goia, I.; Ferrara, A.; Catorci, A. Disentangling the Effects of Disturbance from Those of Dominant Tall Grass Features in Driving the Functional Variation of Restored Grassland in a Sub-Mediterranean Context. Diversity 2020, 12, 11. [Google Scholar] [CrossRef]
- Allegrezza, M.; Biondi, E.; Ballelli, S.; Tesei, G.; Ottaviani, C.; Zitti, S. Brachypodium Rupestre (Host) Roem. & Schult. Herbaceous Communities of Heliophilous Edge in the Trifolio Medii-Geranietea Sanguinei Müller 1962 Class. Plant Sociol. 2016, 53, 59–76. [Google Scholar]




| Model Name | Validation Type | Overall Accuracy (%) | Kappa Index (%) |
|---|---|---|---|
| rf | cv (kfold = 10) | 94.79 | 88.00 |
| svmRadialCost | cv (kfold = 10) | 91.66 | 82.09 |
| rpart2 | cv (kfold = 10) | 90.62 | 79.07 |
| Habitat Code | Habitat Cover (ha) | Cover (ha) B. genuense | Cover (%) B. genuense | ASEH (ha) | Cumulative Contribution of B. genuense % |
|---|---|---|---|---|---|
| 4060 | 197.62 | 30.61 | 15.49 | 14.28 | 3.93 |
| 6170 | 3254.98 | 112.34 | 3.45 | −156.52 | 14.45 |
| 8120 | 721.11 | 3.72 | 0.51 | −55.84 | 0.48 |
| *6210 | 3874.06 | 575.30 | 14.85 | 255.30 | 74.04 |
| *6230 | 291.39 | 40.21 | 13.80 | 16.14 | 5.17 |
| Mosaic | 1063.67 | 14.79 | 1.39 | −73.07 | 1.90 |
| TOTAL | 9402.84 | 776.98 | 8.26 |
| Sensitivity | Specificity | |
|---|---|---|
| KAPPA | 97.54 | 85.05 |
| TSS | 97.60 | 85.00 |
| ROC | 97.60 | 85.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Simone, W.; Allegrezza, M.; Frattaroli, A.R.; Montecchiari, S.; Tesei, G.; Zuccarello, V.; Di Musciano, M. From Remote Sensing to Species Distribution Modelling: An Integrated Workflow to Monitor Spreading Species in Key Grassland Habitats. Remote Sens. 2021, 13, 1904. https://doi.org/10.3390/rs13101904
De Simone W, Allegrezza M, Frattaroli AR, Montecchiari S, Tesei G, Zuccarello V, Di Musciano M. From Remote Sensing to Species Distribution Modelling: An Integrated Workflow to Monitor Spreading Species in Key Grassland Habitats. Remote Sensing. 2021; 13(10):1904. https://doi.org/10.3390/rs13101904
Chicago/Turabian StyleDe Simone, Walter, Marina Allegrezza, Anna Rita Frattaroli, Silvia Montecchiari, Giulio Tesei, Vincenzo Zuccarello, and Michele Di Musciano. 2021. "From Remote Sensing to Species Distribution Modelling: An Integrated Workflow to Monitor Spreading Species in Key Grassland Habitats" Remote Sensing 13, no. 10: 1904. https://doi.org/10.3390/rs13101904
APA StyleDe Simone, W., Allegrezza, M., Frattaroli, A. R., Montecchiari, S., Tesei, G., Zuccarello, V., & Di Musciano, M. (2021). From Remote Sensing to Species Distribution Modelling: An Integrated Workflow to Monitor Spreading Species in Key Grassland Habitats. Remote Sensing, 13(10), 1904. https://doi.org/10.3390/rs13101904






