The Influence of Spectral Pretreatment on the Selection of Representative Calibration Samples for Soil Organic Matter Estimation Using Vis-NIR Reflectance Spectroscopy
Abstract
1. Introduction
2. Materials and Methods
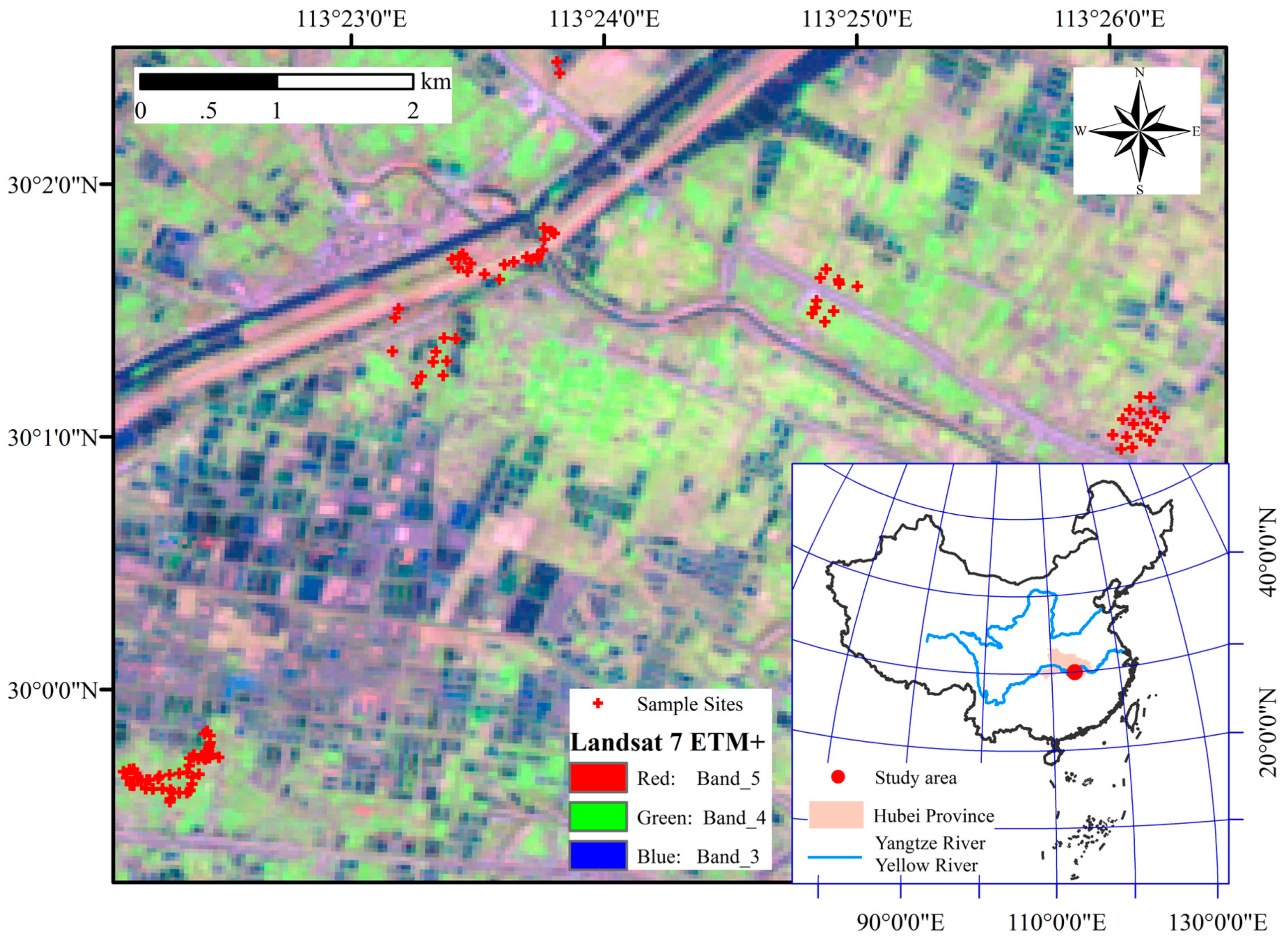
2.1. Study Area
2.2. Sample Collection
2.3. Spectral Measurement and Chemical Analysis
2.4. Spectral Pretreatment
2.5. Model Calibration
2.5.1. Sample Selection Method
2.5.2. Inclusion of Pretreatment in Sample Selection
2.5.3. PLSR Models
2.6. Performance of Models
3. Results
3.1. Descriptive Statistics of Soil Samples
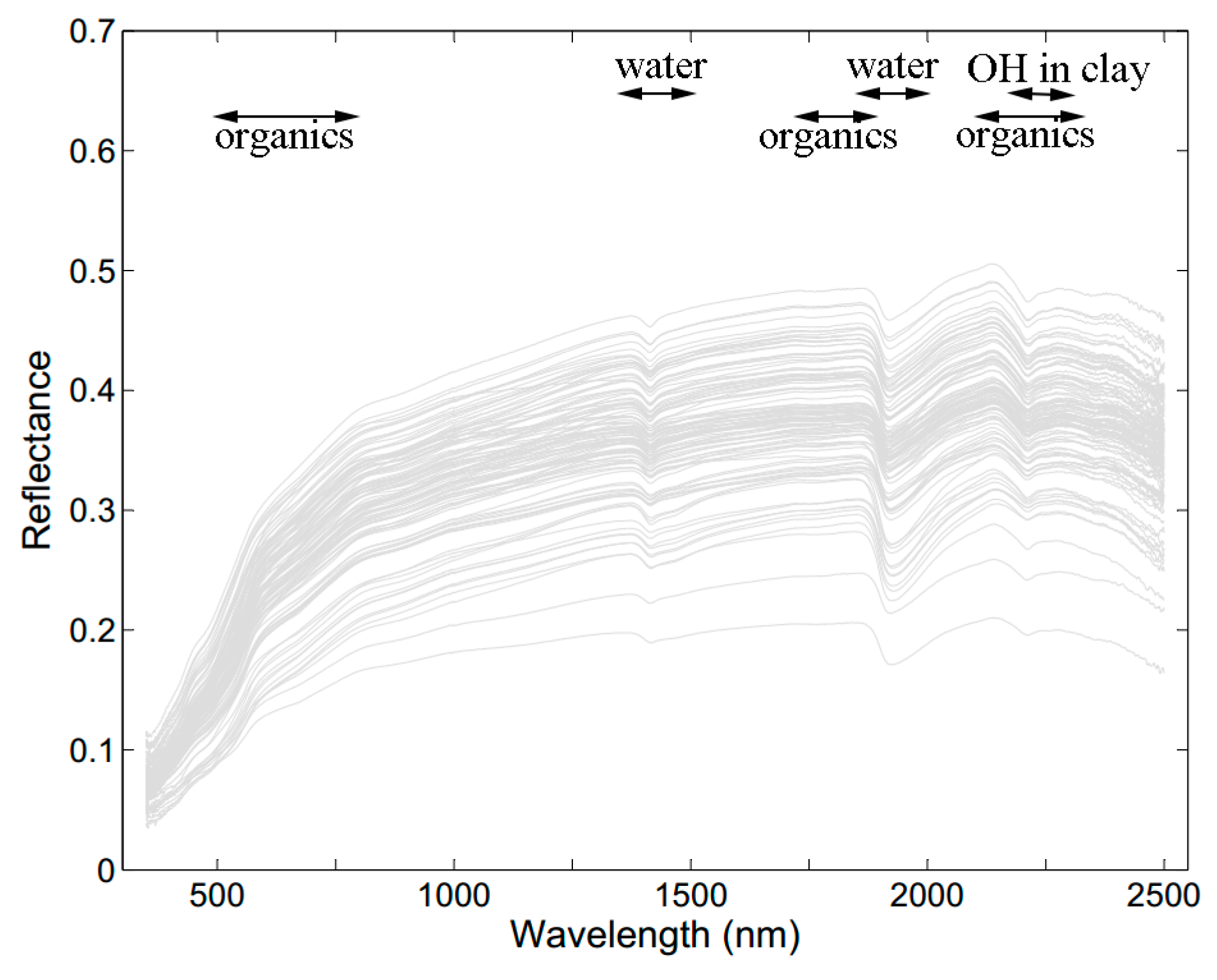
3.2. Soil Spectral Characteristics
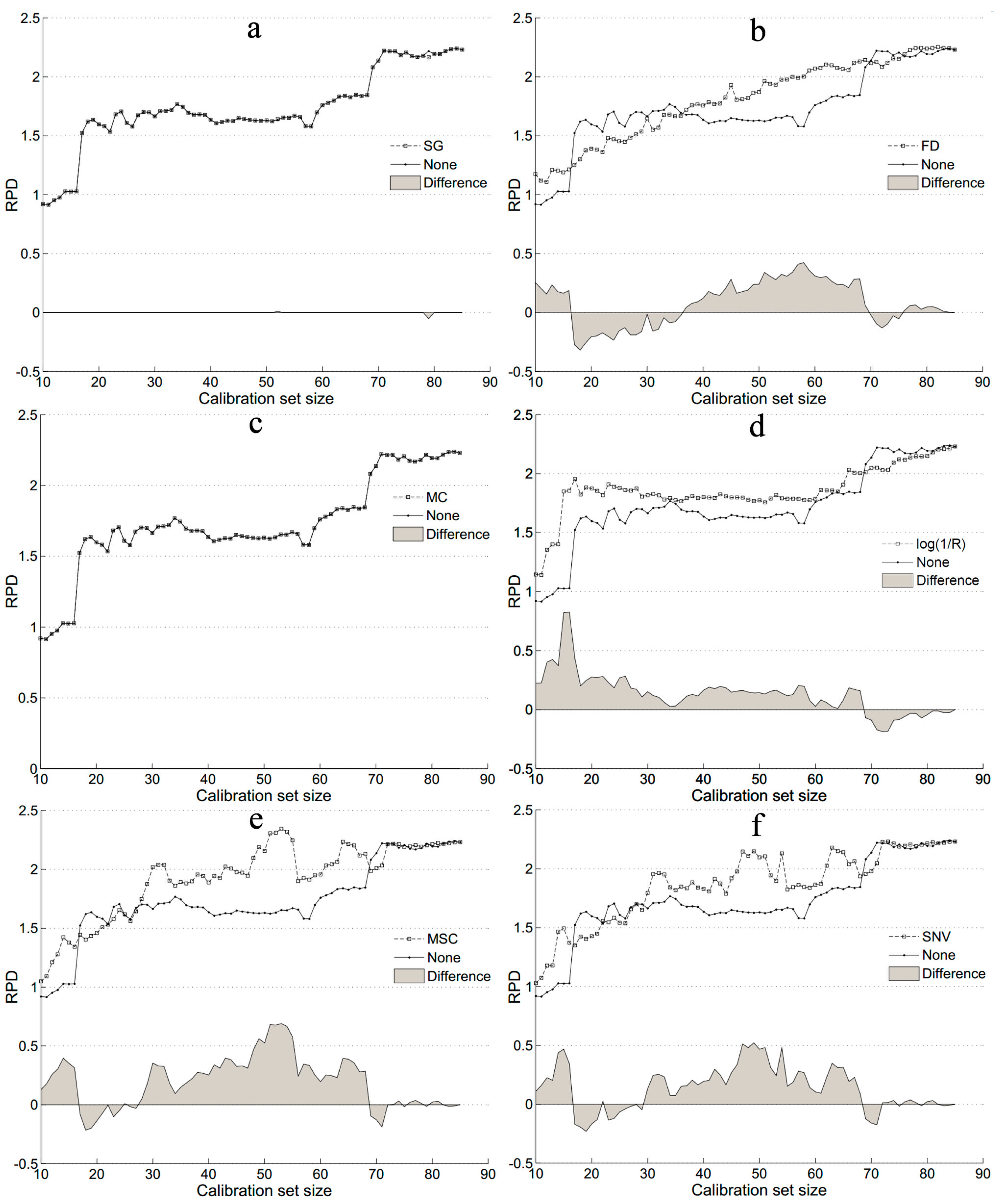
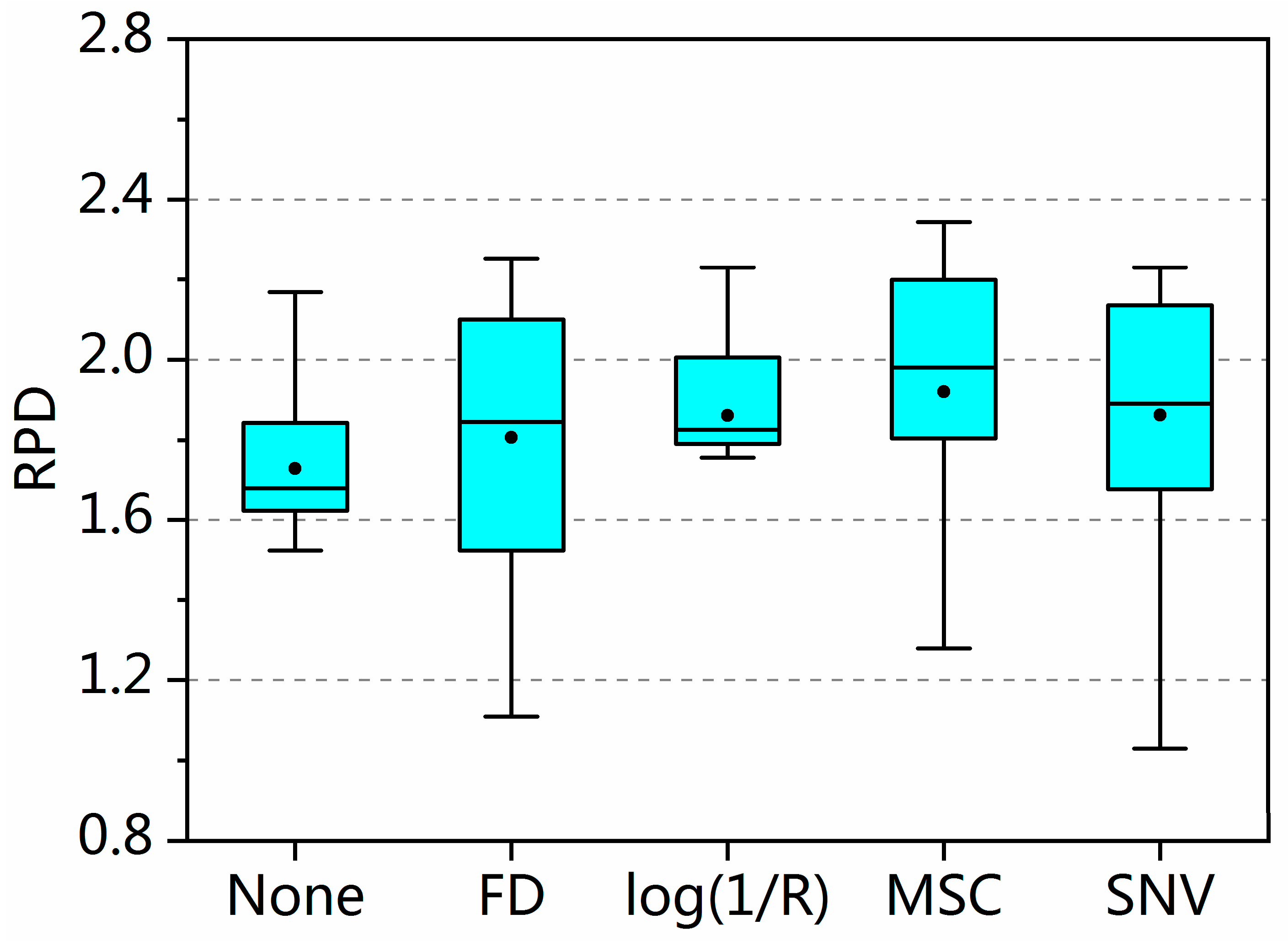
3.3. Accuracy of SOM Prediction after Including Pretreatment in Sample Selection
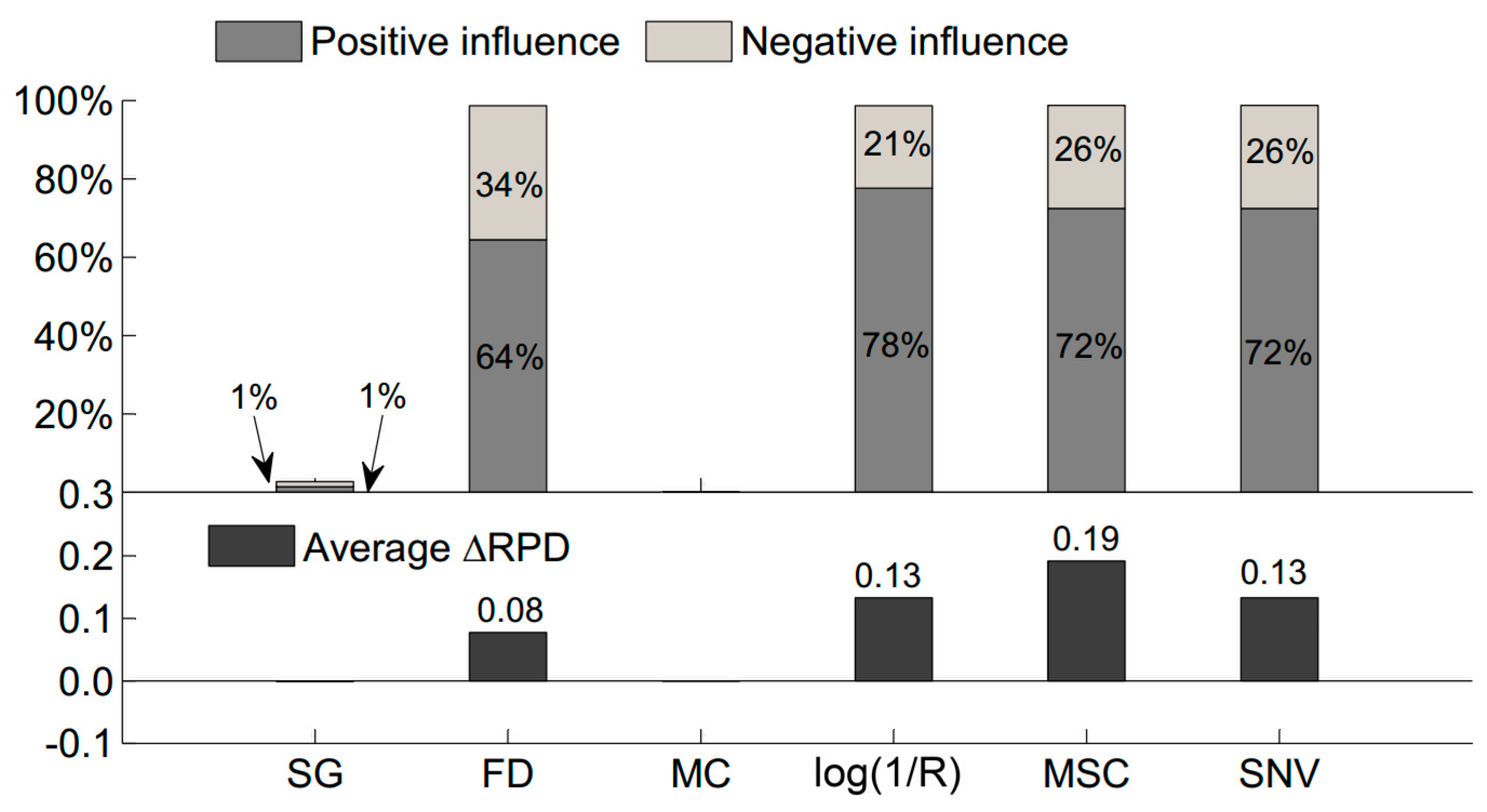
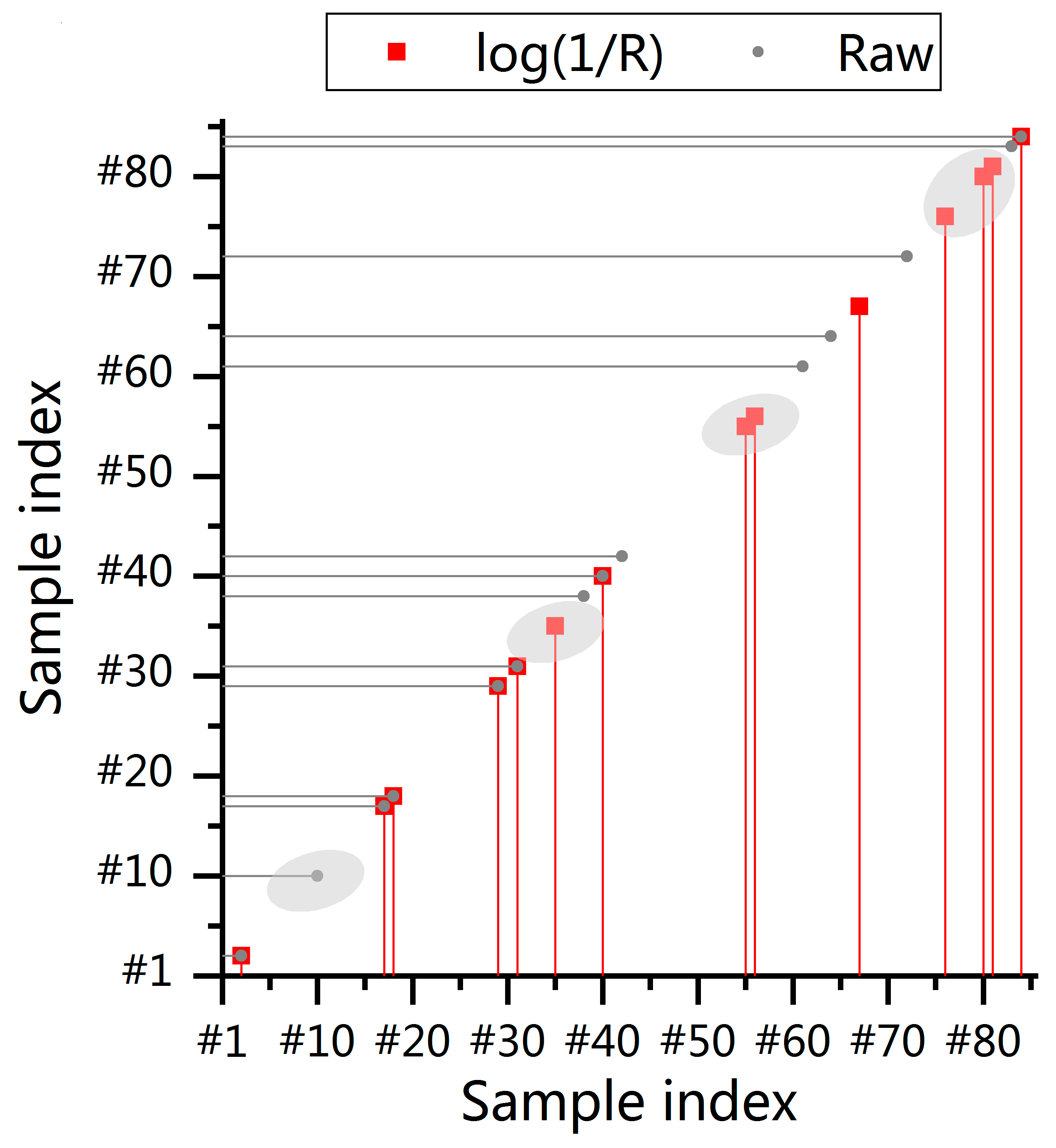
3.4. Proportion of Pretreatment’s Positive or Negative Influence on Sample Selection
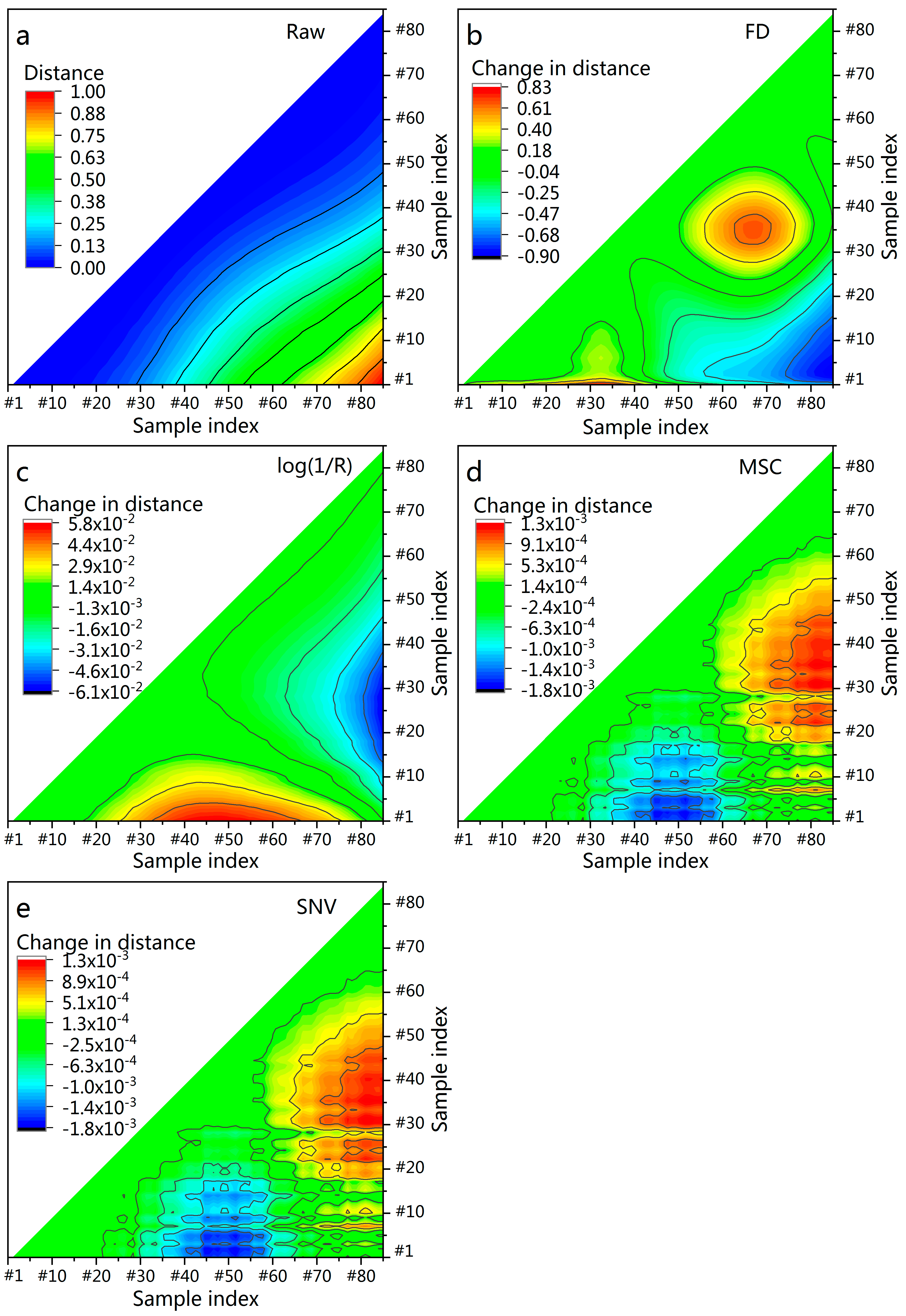
3.5. Euclidean Distance between Samples after Pretreatment
4. Discussion
4.1. Influence of Pretreatment on Sample Selection
4.2. How Pretreatment Affects Sample Selection
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lal, R. Soil carbon sequestration impacts on global climate change and food security. Science 2004, 304, 1623–1627. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.W.; Torn, M.S.; Abiven, S.; Dittmar, T.; Guggenberger, G.; Janssens, I.A.; Kleber, M.; Kögel-Knabner, I.; Lehmann, J.; Manning, D.A. Persistence of soil organic matter as an ecosystem property. Nature 2011, 478, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, J.; Kleber, M. The contentious nature of soil organic matter. Nature 2015, 528, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Walkley, A.; Black, I.A. An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci. 1934, 37, 29–38. [Google Scholar] [CrossRef]
- Nelson, D.; Sommers, L.E. Total carbon, organic carbon, and organic matter. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; American Society of Agronomy Inc.: Madison, WI, USA, 1982; pp. 539–579. [Google Scholar]
- Shi, Z.; Ji, W.; Viscarra Rossel, R.A.; Chen, S.; Zhou, Y. Prediction of soil organic matter using a spatially constrained local partial least squares regression and the Chinese vis–NIR spectral library. Eur. J. Soil Sci. 2015, 66, 679–687. [Google Scholar] [CrossRef]
- Bellon-Maurel, V.; McBratney, A. Near-infrared (NIR) and mid-infrared (MIR) spectroscopic techniques for assessing the amount of carbon stock in soils—Critical review and research perspectives. Soil Biol. Biochem. 2011, 43, 1398–1410. [Google Scholar] [CrossRef]
- Ben-Dor, E.; Banin, A. Near infrared analysis (NIRA) as a method to simultaneously evaluate spectral featureless constituents in soils. Soil Sci. 1995, 159, 259–270. [Google Scholar] [CrossRef]
- Guerrero, C.; Zornoza, R.; Gómez, I.; Mataix-Beneyto, J. Spiking of NIR regional models using samples from target sites: Effect of model size on prediction accuracy. Geoderma 2010, 158, 66–77. [Google Scholar] [CrossRef]
- Fidencio, P.H.; Poppi, R.J.; de Andrade, J.C. Determination of organic matter in soils using radial basis function networks and near infrared spectroscopy. Anal. Chim. Acta 2002, 453, 125–134. [Google Scholar] [CrossRef]
- Ge, Y.; Morgan, C.; Thomasson, J.; Waiser, T. A new perspective to near-infrared reflectance spectroscopy: A wavelet approach. Trans. ASABE 2007, 50, 303–311. [Google Scholar] [CrossRef]
- Gholizadeh, A.; Borůvka, L.; Saberioon, M.; Vašát, R. Visible, near-infrared, and mid-infrared spectroscopy applications for soil assessment with emphasis on soil organic matter content and quality: State-of-the-art and key issues. Appl. Spectrosc. 2013, 67, 1349–1362. [Google Scholar] [CrossRef] [PubMed]
- Shetty, N.; Rinnan, Å.; Gislum, R. Selection of representative calibration sample sets for near-infrared reflectance spectroscopy to predict nitrogen concentration in grasses. Chemom. Intell. Lab. Syst. 2012, 111, 59–65. [Google Scholar] [CrossRef]
- Ramirez-Lopez, L.; Schmidt, K.; Behrens, T.; van Wesemael, B.; Dematte, J.A.; Scholten, T. Sampling optimal calibration sets in soil infrared spectroscopy. Geoderma 2014, 226, 140–150. [Google Scholar] [CrossRef]
- Kuang, B.; Mouazen, A.M. Influence of the number of samples on prediction error of visible and near infrared spectroscopy of selected soil properties at the farm scale. Eur. J. Soil Sci. 2012, 63, 421–429. [Google Scholar] [CrossRef]
- Kennard, R.W.; Stone, L.A. Computer aided design of experiments. Technometrics 1969, 11, 137–148. [Google Scholar] [CrossRef]
- Massart, D.L.; Vandeginste, B.G.; Buydens, L.; Lewi, P.; Smeyers-Verbeke, J.; Jong, S.D. Handbook of Chemometrics and Qualimetrics: Part A; Elsevier Science Inc.: Amsterdam, The Netherlands, 1997. [Google Scholar]
- Galvao, R.K.H.; Araujo, M.C.U.; Jose, G.E.; Pontes, M.J.C.; Silva, E.C.; Saldanha, T.C.B. A method for calibration and validation subset partitioning. Talanta 2005, 67, 736–740. [Google Scholar] [CrossRef] [PubMed]
- Udelhoven, T.; Emmerling, C.; Jarmer, T. Quantitative analysis of soil chemical properties with diffuse reflectance spectrometry and partial least-square regression: A feasibility study. Plant Soil 2003, 251, 319–329. [Google Scholar] [CrossRef]
- Stevens, A.; Udelhoven, T.; Denis, A.; Tychon, B.; Lioy, R.; Hoffmann, L.; Van Wesemael, B. Measuring soil organic carbon in croplands at regional scale using airborne imaging spectroscopy. Geoderma 2010, 158, 32–45. [Google Scholar] [CrossRef]
- Nocita, M.; Stevens, A.; Toth, G.; Panagos, P.; van Wesemael, B.; Montanarella, L. Prediction of soil organic carbon content by diffuse reflectance spectroscopy using a local partial least square regression approach. Soil Biol. Biochem. 2014, 68, 337–347. [Google Scholar] [CrossRef]
- Peng, Y.; Knadel, M.; Gislum, R.; Deng, F.; Norgaard, T.; de Jonge, L.W.; Moldrup, P.; Greve, M.H. Predicting soil organic carbon at field scale using a national soil spectral library. J. Near Infrared Spectrosc. 2013, 21, 213–222. [Google Scholar] [CrossRef]
- Vasques, G.M.; Grunwald, S.; Harris, W.G. Spectroscopic models of soil organic carbon in Florida, USA. J. Environ. Q. 2010, 39, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Wienhold, B.J.; Power, J.F.; Doran, J.W. Agricultural accomplishments and impending concerns. Soil Sci. 2000, 165, 13–30. [Google Scholar] [CrossRef]
- Ji, W.J.; Li, S.; Chen, S.C.; Shi, Z.; Viscarra Rossel, R.A.; Mouazen, A.M. Prediction of soil attributes using the Chinese soil spectral library and standardized spectra recorded at field conditions. Soil Tillage Res. 2016, 155, 492–500. [Google Scholar] [CrossRef]
- De Gruijter, J.; Brus, D.J.; Bierkens, M.F.; Knotters, M. Sampling for Natural Resource Monitoring; Springer Science & Business Media: Berlin, Germany, 2006. [Google Scholar]
- Liu, Y.; Shi, Z.; Zhang, G.; Chen, Y.; Li, S.; Hong, Y.; Shi, T.; Wang, J.; Liu, Y. Application of Spectrally Derived Soil Type as Ancillary Data to Improve the Estimation of Soil Organic Carbon by Using the Chinese Soil Vis-NIR Spectral Library. Remote Sens. 2018, 10, 1747. [Google Scholar] [CrossRef]
- Shi, T.; Chen, Y.; Liu, Y.; Wu, G. Visible and near-infrared reflectance spectroscopy-An alternative for monitoring soil contamination by heavy metals. J. Hazard. Mater. 2014, 265, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Gholizadeh, A.; Borůvka, L.; Saberioon, M.M.; Kozak, J.; Vasat, R.; Nemecek, K. Comparing different data preprocessing methods for monitoring soil heavy metals based on soil spectral features. Soil Water Res. 2015, 10, 218–227. [Google Scholar] [CrossRef]
- Gholizadeh, A.; Saberioon, M.; Carmon, N.; Borůvka, L.; Ben-Dor, E. Examining the Performance of PARACUDA-II Data-Mining Engine versus Selected Techniques to Model Soil Carbon from Reflectance Spectra. Remote Sens. 2018, 10, 1172. [Google Scholar] [CrossRef]
- Gholizadeh, A.; Carmon, N.; Klement, A.; Ben-Dor, E.; Borůvka, L. Agricultural Soil Spectral Response and Properties Assessment: Effects of Measurement Protocol and Data Mining Technique. Remote Sens. 2017, 9, 1078. [Google Scholar] [CrossRef]
- Debaene, G.; Niedźwiecki, J.; Pecio, A.; Żurek, A. Effect of the number of calibration samples on the prediction of several soil properties at the farm-scale. Geoderma 2014, 214, 114–125. [Google Scholar] [CrossRef]
- Li, Z.; Liu, J.; Shan, P.; Peng, S.; Lv, J.; Ma, Z. Strategy for constructing calibration sets based on a derivative spectra information space consensus. Chemom. Intell. Lab. Syst. 2016, 156, 7–13. [Google Scholar] [CrossRef]
- Stevens, A.; Nocita, M.; Tóth, G.; Montanarella, L.; van Wesemael, B. Prediction of soil organic carbon at the European scale by visible and near infrared reflectance spectroscopy. PLoS ONE 2013, 8, e66409. [Google Scholar] [CrossRef] [PubMed]
- Cao, N. Calibration Optimization and Efficiency in Near Infrared Spectroscopy. Ph.D. Thesis, Iowa State University, Ames, IA, USA, 2013. [Google Scholar]
- Liu, Y.; Guo, L.; Jiang, Q.; Zhang, H.T.; Chen, Y. Comparing geospatial techniques to predict SOC stocks. Soil Tillage Res. 2015, 148, 46–58. [Google Scholar] [CrossRef]
- Knotters, M.; Brus, D. Purposive versus random sampling for map validation: A case study on ecotope maps of floodplains in the Netherlands. Ecohydrology 2013, 6, 425–434. [Google Scholar] [CrossRef]
- Liu, Y.; Jiang, Q.; Fei, T.; Wang, J.; Shi, T.; Guo, K.; Li, X.; Chen, Y. Transferability of a Visible and Near-Infrared Model for Soil Organic Matter Estimation in Riparian Landscapes. Remote Sens. 2014, 6, 4305–4322. [Google Scholar] [CrossRef]
- Liu, Y.L.; Jiang, Q.H.; Shi, T.Z.; Fei, T.; Wang, J.J.; Liu, G.L.; Chen, Y.Y. Prediction of total nitrogen in cropland soil at different levels of soil moisture with Vis/NIR spectroscopy. Acta Agric. Scand. Sect. B Soil Plant Sci. 2014, 64, 267–281. [Google Scholar] [CrossRef]
- Zhang, J.; Xu, Z. Dye tracer infiltration technique to investigate macropore flow paths in Maka Mountain, Yunnan Province, China. J. Cent. South Univ. 2016, 23, 2101–2109. [Google Scholar] [CrossRef]
- Rinnan, A.; van den Berg, F.; Engelsen, S.B. Review of the most common pre-processing techniques for near-infrared spectra. Trac-Trends Anal. Chem. 2009, 28, 1201–1222. [Google Scholar] [CrossRef]
- Steinier, J.; Termonia, Y.; Deltour, J. Smoothing and differentiation of data by simplified least square procedure. Anal. Chem. 1972, 44, 1906–1909. [Google Scholar] [CrossRef]
- Naes, T.; Martens, H. Multivariate Calibration; Norwegian Food Research Institute: Oslovegen, Norway, 1989. [Google Scholar]
- Stenberg, B.; Viscarra Rossel, R.A.; Mouazen, A.M.; Wetterlind, J. Chapter five-visible and near infrared spectroscopy in soil science. Adv. Agron. 2010, 107, 163–215. [Google Scholar]
- Rossel Viscarra, R.A. ParLeS: Software for chemometric analysis of spectroscopic data. Chemom. Intell. Lab. Syst. 2008, 90, 72–83. [Google Scholar] [CrossRef]
- Barnes, R.; Dhanoa, M.; Lister, S.J. Standard normal variate transformation and de-trending of near-infrared diffuse reflectance spectra. Appl. Spectrosc. 1989, 43, 772–777. [Google Scholar] [CrossRef]
- Geladi, P.; Macdougall, D.; Martens, H. Linearization and Scatter-Correction for Near-Infrared Reflectance Spectra of Meat. ApSpe 1985, 39, 491–500. [Google Scholar] [CrossRef]
- Askari, M.S.; Cui, J.; O’Rourke, S.M.; Holden, N.M. Evaluation of soil structural quality using VIS–NIR spectra. Soil Tillage Res. 2015, 146, 108–117. [Google Scholar] [CrossRef]
- Hook, J. Smoothing non-smooth systems with low-pass filters. Phys. D Nonlinear Phenom. 2014, 269, 76–85. [Google Scholar] [CrossRef]
- Delwiche, S.R. A graphical method to evaluate spectral preprocessing in multivariate regression calibrations: Example with Savitzky-Golay filters and partial least squares regression. Appl. Spectrosc. 2010, 64, 73–82. [Google Scholar] [CrossRef]
- Shetty, N.; Gislum, R. Quantification of fructan concentration in grasses using NIR spectroscopy and PLSR. Field Crops Res. 2011, 120, 31–37. [Google Scholar] [CrossRef]
- West, J.B.; Bowen, G.J.; Dawson, T.E.; Tu, K.P. Isoscapes: Understanding Movement, Pattern, and Process on Earth through Isotope Mapping; Springer: Heidelberg, Germany, 2009; Volume 3, p. 76. [Google Scholar]
- Kuhn, M.; Johnson, K. Data pre-processing. In Applied Predictive Modeling; Springer: New York, NY, USA, 2013; pp. 59. [Google Scholar]
- Echambadi, R.; Hess, J.D. Mean-centering does not alleviate collinearity problems in moderated multiple regression models. Mark. Sci. 2007, 26, 438–445. [Google Scholar] [CrossRef]
- Martens, H.; Jensen, S.; Geladi, P. Multivariate linearity transformation for near-infrared reflectance spectrometry. In Proceedings of the Nordic Symposium on Applied Statistics; Stokkand Forlag Publishers: Stavanger, Norway, 1983; pp. 205–234. [Google Scholar]
- Rozenstein, O.; Paz-Kagan, T.; Salbach, C.; Karnieli, A. Comparing the Effect of Preprocessing Transformations on Methods of Land-Use Classification Derived From Spectral Soil Measurements. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 2015, 8, 2393–2404. [Google Scholar] [CrossRef]
- Dhanoa, M.; Lister, S.; Sanderson, R.; Barnes, R. The link between multiplicative scatter correction (MSC) and standard normal variate (SNV) transformations of NIR spectra. J. Near Infrared Spectrosc. 1994, 2, 43–47. [Google Scholar] [CrossRef]
- Candolfi, A.; De Maesschalck, R.; Jouan-Rimbaud, D.; Hailey, P.; Massart, D. The influence of data pre-processing in the pattern recognition of excipients near-infrared spectra. J. Pharm. Biomed. Anal. 1999, 21, 115–132. [Google Scholar] [CrossRef]
- Brus, D.J.; Kempen, B.; Heuvelink, G.B.M. Sampling for validation of digital soil maps. Eur. J. Soil Sci. 2011, 62, 394–407. [Google Scholar] [CrossRef]
- Rossiter, D. Assessing the thematic accuracy of area-class soil maps; Soil Science Division, ITC: Enschede, The Netherlands, 2001; waiting publication. [Google Scholar]
- Li, S.; Shi, Z.; Chen, S.; Ji, W.; Zhou, L.; Yu, W.; Webster, R. In situ measurements of organic carbon in soil profiles using vis-NIR spectroscopy on the Qinghai–Tibet plateau. Environ. Sci. Technol. 2015, 49, 4980–4987. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Yu, L.; Chen, Y.; Liu, Y.; Liu, Y.; Liu, Y.; Cheng, H. Prediction of Soil Organic Matter by VIS–NIR Spectroscopy Using Normalized Soil Moisture Index as a Proxy of Soil Moisture. Remote Sens. 2017, 10, 28. [Google Scholar] [CrossRef]
- Goodarzi, M.; Sharma, S.; Ramon, H.; Saeys, W. Multivariate calibration of NIR spectroscopic sensors for continuous glucose monitoring. TrAC Trends Anal. Chem. 2015, 67, 147–158. [Google Scholar] [CrossRef]
- Della Riccia Giacomo, D.Z.S. A multivariate regression model for detection of fumonisins content in maize from near infrared spectra. Food Chem. 2013, 141, 4289–4294. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.C. Variable affecting near infrared reflectance spectroscopic analysis. Near-Infrared Technol. Agric. Food Ind. 1987, 143–167. [Google Scholar]
- Yang, Q.Y.; Jiang, Z.C.; Li, W.J.; Li, H. Prediction of soil organic matter in peak-cluster depression region using kriging and terrain indices. Soil Tillage Res. 2014, 144, 126–132. [Google Scholar] [CrossRef]
- Summers, D.; Lewis, M.; Ostendorf, B.; Chittleborough, D. Visible near-infrared reflectance spectroscopy as a predictive indicator of soil properties. Ecol. Indic. 2011, 11, 123–131. [Google Scholar] [CrossRef]
- Li, D.; Chen, X.; Peng, Z.; Chen, S.; Chen, W.; Han, L.; Li, Y. Prediction of soil organic matter content in a litchi orchard of South China using spectral indices. Soil Tillage Res. 2012, 123, 78–86. [Google Scholar] [CrossRef]
- Isaksson, T.; Næs, T. Selection of samples for calibration in near-infrared spectroscopy. Part II: Selection based on spectral measurements. Appl. Spectrosc. 1990, 44, 1152–1158. [Google Scholar] [CrossRef]
- Walczak, B.; Massart, D.L. Application of Radial Basis Functions—Partial Least Squares to non-linear pattern recognition problems: Diagnosis of process faults. Anal. Chim. Acta 1996, 331, 187–193. [Google Scholar] [CrossRef]
- Bakeev, K.A. Process Analytical technology: Spectroscopic Tools and Implementation Strategies for the Chemical and Pharmaceutical Industries; John Wiley & Sons: New York, NY, USA, 2010. [Google Scholar]
- Gras, J.-P.; Barthès, B.G.; Mahaut, B.; Trupin, S. Best practices for obtaining and processing field visible and near infrared (VNIR) spectra of topsoils. Geoderma 2014, 214, 126–134. [Google Scholar] [CrossRef]
- Schwartz, G.; Ben-Dor, E.; Eshel, G. Quantitative assessment of hydrocarbon contamination in soil using reflectance spectroscopy: A "multipath" approach. Appl. Spectrosc. 2013, 67, 1323. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.; Rossel Viscarra, R.A.; Shi, Z. Accounting for the effects of water and the environment on proximally sensed vis-NIR soil spectra and their calibrations. Eur. J. Soil Sci. 2015, 66, 555–565. [Google Scholar] [CrossRef]
- Ge, Y.F.; Morgan, C.L.S.; Ackerson, J.P. VisNIR spectra of dried ground soils predict properties of soils scanned moist and intact. Geoderma 2014, 221, 61–69. [Google Scholar] [CrossRef]
| Sample | Number | SOM (g kg−1) | CV 3 | Skewness | Kurtosis | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Range 1 | Min | Max | Median | Mean | SD 2 | |||||
| Total | 106 | 43.28 | 4.06 | 47.34 | 28.64 | 27.43 | 10.59 | 0.39 | −0.19 | −1.06 |
| Calibration | 85 | 43.28 | 4.06 | 47.34 | 28.69 | 27.45 | 10.67 | 0.39 | −0.20 | −1.03 |
| Validation | 21 | 35.75 | 8.37 | 44.12 | 28.59 | 27.36 | 10.54 | 0.39 | −0.17 | −1.20 |
| Type of Pretreatment | ||||||
|---|---|---|---|---|---|---|
| Variable | N | None | FD | log(1/R) | MSC | SNV |
| RPD (Mean ± Std. Deviation) | 76 | 1.73 ± 0.33 | 1.81 ± 0.34 (p = 0.62) | 1.86 ± 0.21 * (p = 0.03) | 1.92 ± 0.32 * (p = 0.00) | 1.86 ± 0.31 (p = 0.08) |
| Pretreatment | RPD | |||
|---|---|---|---|---|
| None | 0.72 | 5.71 | 1.87 | - |
| SG | 0.72 | 5.71 | 1.87 | −0.00 |
| FD | 0.80 | 4.74 | 2.25 | 0.38 |
| MC | 0.67 | 6.09 | 1.75 | −0.12 |
| log(1/R) | 0.59 | 6.82 | 1.56 | −0.31 |
| MSC | 0.66 | 6.20 | 1.72 | −0.15 |
| SNV | 0.74 | 5.47 | 1.95 | 0.08 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Liu, Y.; Chen, Y.; Zhang, Y.; Shi, T.; Wang, J.; Hong, Y.; Fei, T.; Zhang, Y. The Influence of Spectral Pretreatment on the Selection of Representative Calibration Samples for Soil Organic Matter Estimation Using Vis-NIR Reflectance Spectroscopy. Remote Sens. 2019, 11, 450. https://doi.org/10.3390/rs11040450
Liu Y, Liu Y, Chen Y, Zhang Y, Shi T, Wang J, Hong Y, Fei T, Zhang Y. The Influence of Spectral Pretreatment on the Selection of Representative Calibration Samples for Soil Organic Matter Estimation Using Vis-NIR Reflectance Spectroscopy. Remote Sensing. 2019; 11(4):450. https://doi.org/10.3390/rs11040450
Chicago/Turabian StyleLiu, Yi, Yaolin Liu, Yiyun Chen, Yang Zhang, Tiezhu Shi, Junjie Wang, Yongsheng Hong, Teng Fei, and Yang Zhang. 2019. "The Influence of Spectral Pretreatment on the Selection of Representative Calibration Samples for Soil Organic Matter Estimation Using Vis-NIR Reflectance Spectroscopy" Remote Sensing 11, no. 4: 450. https://doi.org/10.3390/rs11040450
APA StyleLiu, Y., Liu, Y., Chen, Y., Zhang, Y., Shi, T., Wang, J., Hong, Y., Fei, T., & Zhang, Y. (2019). The Influence of Spectral Pretreatment on the Selection of Representative Calibration Samples for Soil Organic Matter Estimation Using Vis-NIR Reflectance Spectroscopy. Remote Sensing, 11(4), 450. https://doi.org/10.3390/rs11040450










