Exposure of Marine Turtle Nesting Grounds to Named Storms Along the Continental USA
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Region
2.2. Datasets
2.2.1. Marine Turtle Nesting Grounds
2.2.2. Named Storms
2.3. Overlap between Marine Turtles and Named Storms
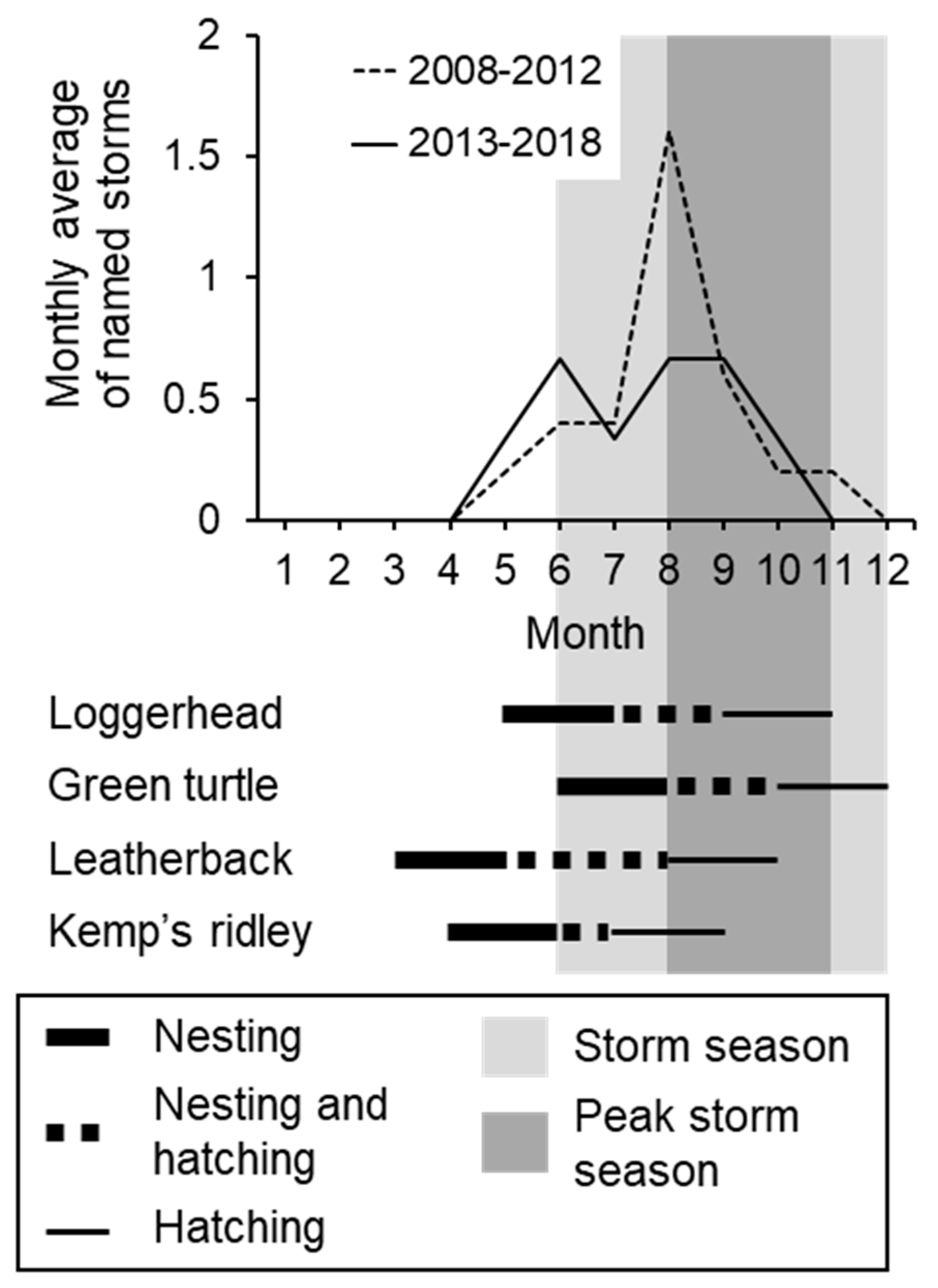
2.3.1. Temporal Overlap
2.3.2. Spatial Overlap
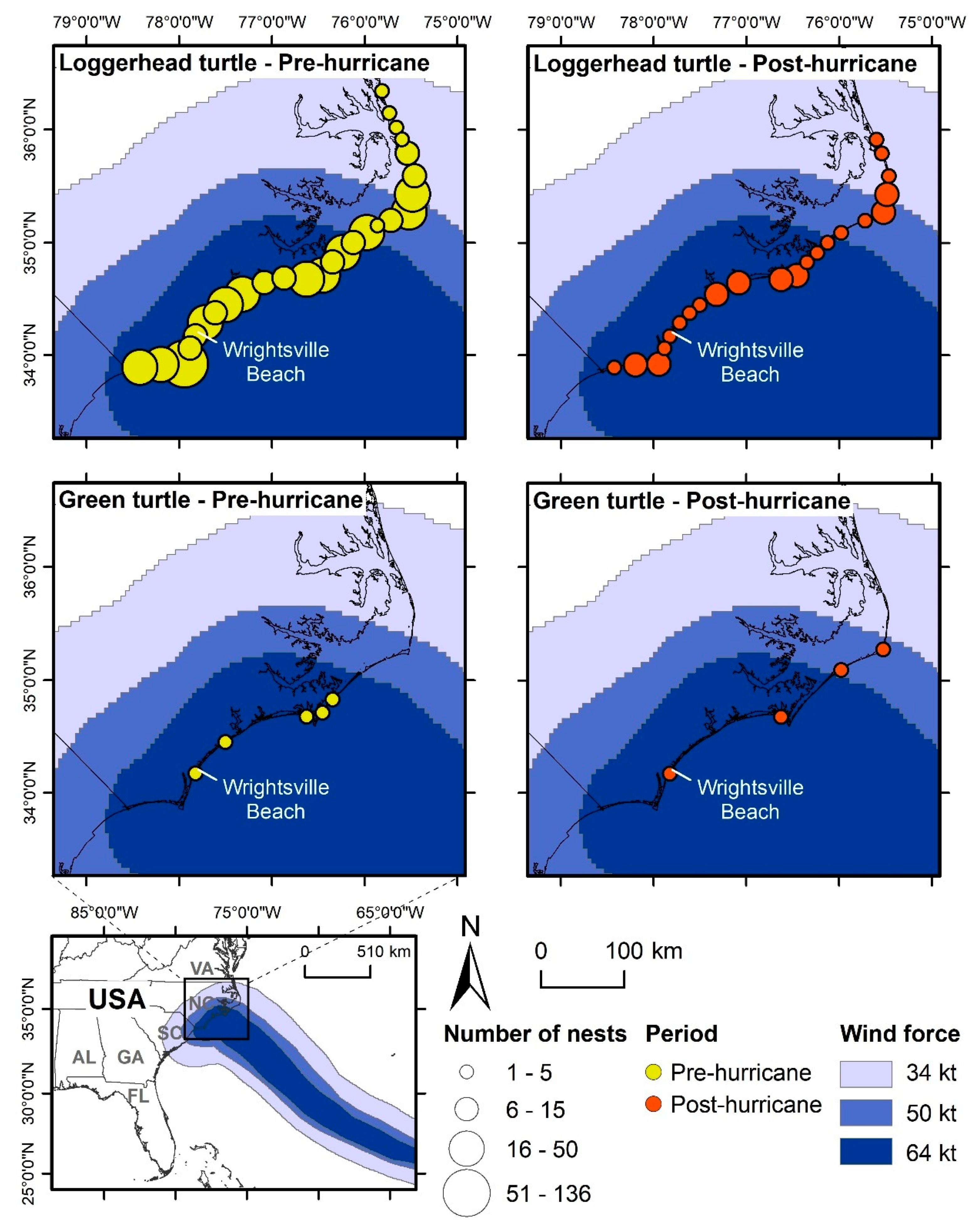
2.3.3. Case Study: Disturbance by Named Storms in North Carolina, USA
3. Results
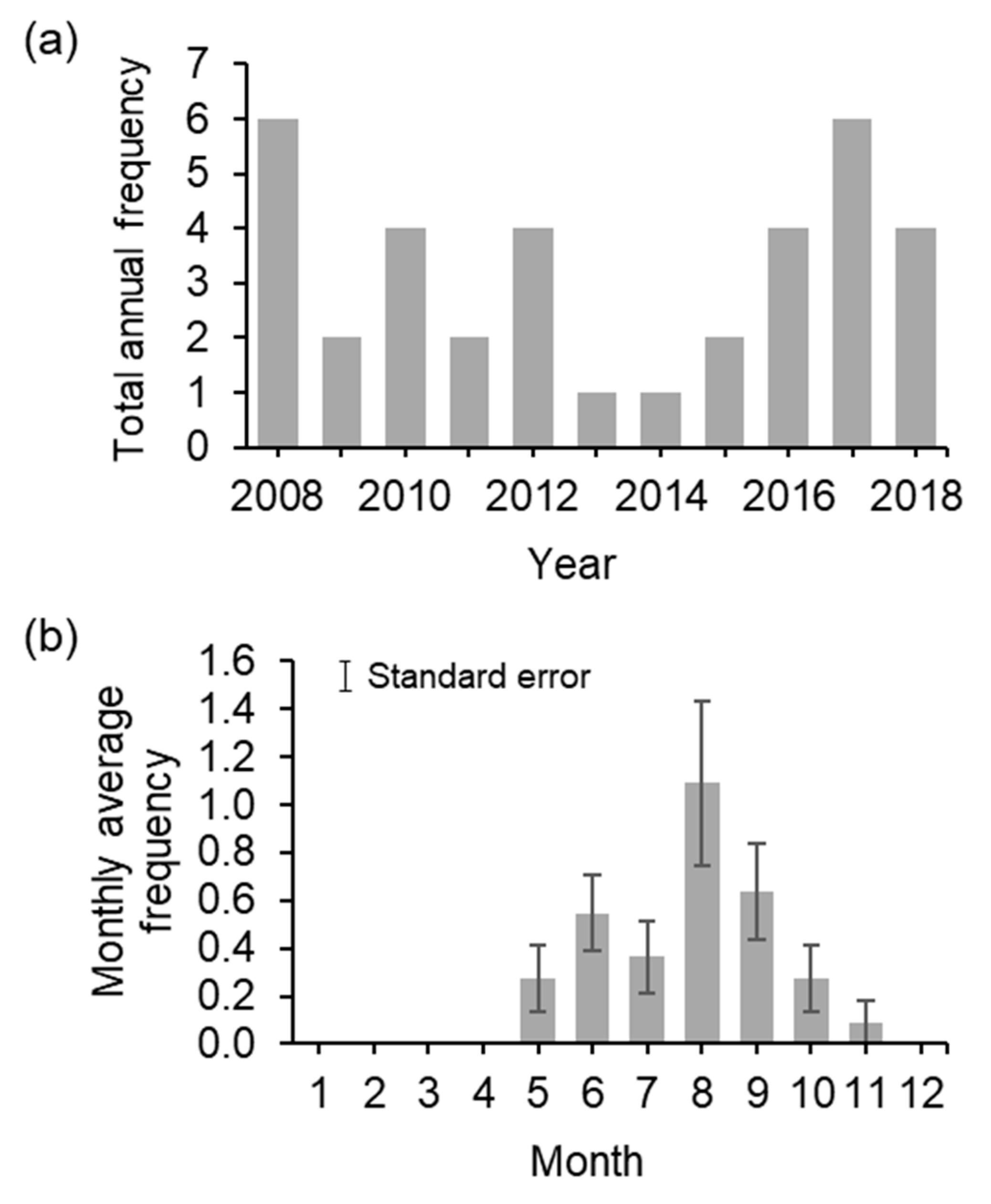
3.1. Temporal Overlap between Nesting Season and Named Storms
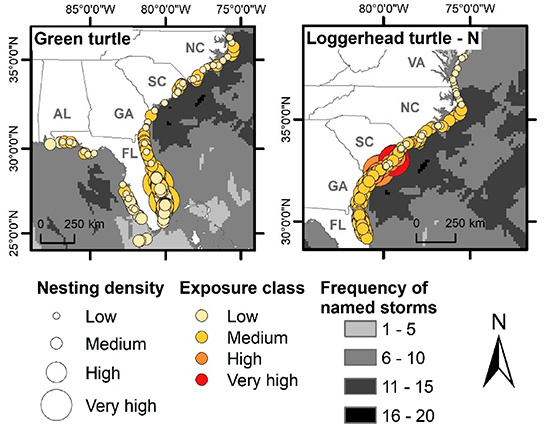
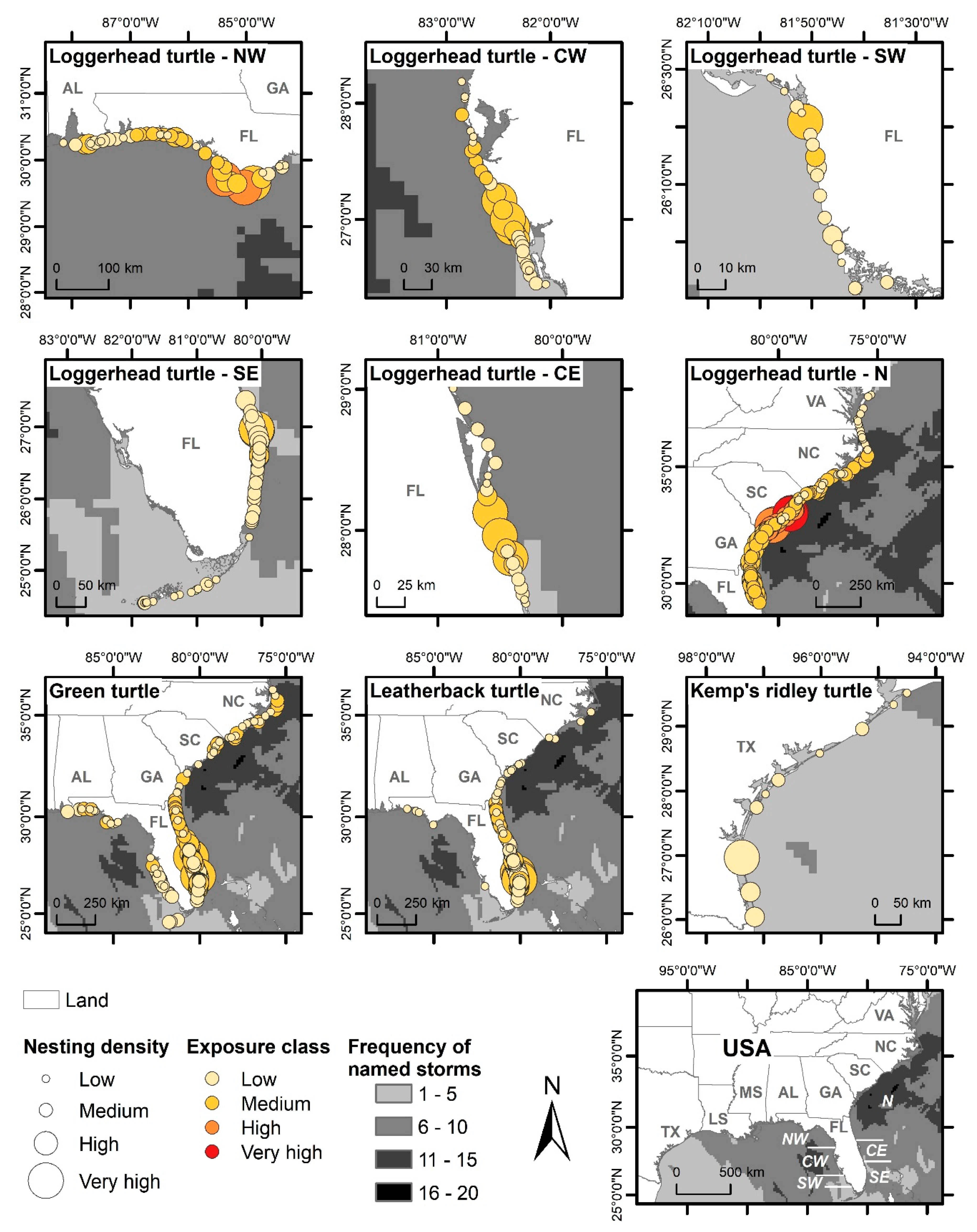
3.2. Spatial Exposure of Nesting Grounds to Named Storms
3.2.1. Loggerhead Turtles
3.2.2. Green Turtles
3.2.3. Leatherback Turtles
3.2.4. Kemp’s Ridley Turtles
3.3. Case Study: Disturbance by Named Storms in North Carolina, USA
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wallace, B.P.; Hutchinson, B.J.; Mast, R.B.; Pilcher, N.J. Putting conservation priority-setting for marine turtles in context. Anim. Conserv. 2011, 14, 14–15. [Google Scholar] [CrossRef]
- Wallace, B.P.; DiMatteo, A.D.; Bolten, A.B.; Chaloupka, M.Y.; Hutchinson, B.J.; Abreu-Grobois, F.A.; Mortimer, J.A.; Seminoff, J.A.; Amorocho, D.; Bjorndal, K.A.; et al. Global conservation priorities for marine turtles. PLoS ONE 2011, 6, e24510. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, M.M.P.B.; Blackwood, J.; Jones, B.; Kim, M.; Leis, B.; Limpus, C.J.; Marsh, H.; Mitchell, J.; Pouzols, F.M.; Pressey, R.L.; et al. A decision framework for prioritizing multiple management actions for threatened marine mega-fauna. Ecol. Appl. 2014, 25, 200–214. [Google Scholar] [CrossRef] [PubMed]
- Klein, C.J.; Beher, J.; Chaloupka, M.Y.; Hamann, M.; Limpus, C.J.; Possingham, H.P. Prioritization of marine turtle management projects: A protocol that accounts for threats to different life history stages. Conserv. Lett. 2016, 10, 547–554. [Google Scholar] [CrossRef]
- Bolten, A.B.; Crowder, L.B.; Dodd, M.G.; MacPherson, S.L.; Musick, J.A.; Schroeder, B.A.; Witherington, B.E.; Long, K.J.; Snover, M.L. Quantifying multiple threats to endangered species: An example from loggerhead sea turtles. Front. Ecol. Environ. 2011, 9, 295–301. [Google Scholar] [CrossRef]
- Wallace, B.P.; Lewison, R.L.; McDonald, S.L.; McDonald, R.K.; Kot, C.Y.; Kelez, S.; Bjorkland, R.K.; Finkbeiner, E.M.; Helmbrecht, S.; Crowder, L.B. Global patterns of marine turtle bycatch. Conserv. Lett. 2010, 3, 131–142. [Google Scholar] [CrossRef]
- Lewison, R.L.; Crowder, L.B.; Wallace, B.P.; Moore, J.E.; Cox, T.; Zydelis, R.; McDonald, S.; DiMatteo, A.; Dunn, D.C.; Kot, C.Y.; et al. Global patterns of marine mammal, seabird, and sea turtle bycatch reveal taxa-specific and cumulative megafauna hotspots. Proc. Natl. Acad. Sci. USA 2014, 111, 5271–5276. [Google Scholar] [CrossRef]
- Nelson Sella, K.A.; Fuentes, M.M.P.B. Exposure of marine turtle nesting grounds to coastal modifications: Implications for management. Ocean Coast. Manag. 2019, 169, 182–190. [Google Scholar] [CrossRef]
- Fuentes, M.M.P.B.; Gredzens, C.; Bateman, B.L.; Boettcher, R.; Ceriani, S.A.; Godfrey, M.H.; Helmers, D.; Ingram, D.K.; Kamrowski, R.L.; Pate, M.; et al. Conservation hotspots for marine turtle nesting in the United States based on coastal development. Ecol. Appl. 2016, 26, 2706–2717. [Google Scholar] [CrossRef]
- Nelson Sella, K.; Sicius, L.; Fuentes, M.M.P.B. Using expert elicitation to determine the relative impact of coastal modifications on marine turtle nesting grounds. Coast. Manag. 2019, 47, 492–506. [Google Scholar] [CrossRef]
- Schuyler, Q.A.; Wilcox, C.; Townsend, K.A.; Wedemeyer-Strombel, K.R.; Balazs, G.H.; van Sebille, E.; Hardesty, B.D. Risk analysis reveals global hotspots for marine debris ingestion by sea turtles. Glob. Chang. Biol. 2016, 22, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Kamrowski, R.L.; Limpus, C.J.; Moloney, J.; Hamann, M. Coastal light pollution and marine turtles: Assessing the magnitude of the problem. Endanger. Species Res. 2012, 19, 85–98. [Google Scholar] [CrossRef]
- Fuentes, M.M.P.B.; Bateman, B.L.; Hamann, M. Relationship between tropical cyclones and the distribution of sea turtle nesting grounds. J. Biogeogr. 2011, 38, 1886–1896. [Google Scholar] [CrossRef]
- Dewald, J.R.; Pike, D.A. Geographical variation in hurricane impacts among sea turtle populations. J. Biogeogr. 2014, 41, 307–316. [Google Scholar] [CrossRef]
- Morton, R.A.; Barras, J.A. Hurricane impacts on coastal wetlands: A half-century record of storm-generated features from southern Louisiana. J. Coast. Res. 2011, 27, 27–43. [Google Scholar] [CrossRef]
- Sherman, D.J.; Hales, B.U.; Potts, M.K.; Ellis, J.T.; Liu, H.; Houser, C. Impacts of Hurricane Ike on the beaches of the Bolivar Peninsula, TX, USA. Geomorphology 2013, 199, 62–81. [Google Scholar] [CrossRef]
- Van Houtan, K.S.; Bass, O.L. Stormy oceans are associated with declines in sea turtle hatching. Curr. Biol. 2007, 17, R590–R591. [Google Scholar] [CrossRef]
- Milton, S.L.; Leone-Kabler, S.L.; Schulman, A.A.; Lutz, P.L. Effects of Hurricane Andrew on the sea turtle nesting beaches of south Florida. Bull. Mar. Sci. 1994, 54, 974–981. [Google Scholar]
- Pike, D.A.; Stiner, J.C. Sea turtle species vary in their susceptibility to tropical cyclones. Oecologia 2007, 153, 471–478. [Google Scholar] [CrossRef]
- Martin, R.E. Storm impacts on loggerhead turtle reproductive sucess. Mar. Turt. Newsl. 1996, 73, 10–12. [Google Scholar]
- Long, T.M.; Angelo, J.; Weishampel, J.F. LiDAR-derived measures of hurricane- and restoration-generated beach morphodynamics in relation to sea turtle nesting behaviour. Int. J. Remote Sens. 2011, 32, 231–241. [Google Scholar] [CrossRef]
- Reed, P. The Sex Ratios of Hatchling Loggerhead Turtles—The Progeny of Two Nesting Adult Females. Ph.D. Thesis, James Cook University, Townsville, Australia, 1980. [Google Scholar]
- Godfrey, M.H.; Barreto, R.; Mrosovsky, N. Estimating past and present sex ratios of sea turtles in Suriname. Can. J. Zool. 1996, 74, 267–277. [Google Scholar] [CrossRef]
- Houghton, J.D.R.; Myers, A.E.; Lloyd, C.; King, R.S.; Isaacs, C.; Hays, G.C. Protracted rainfall decreases temperature within leatherback turtle (Dermochelys coriacea) clutches in Grenada, West Indies: Ecological implications for a species displaying temperature dependent sex determination. J. Exp. Mar. Bio. Ecol. 2007, 345, 71–77. [Google Scholar] [CrossRef]
- Miller, J.D.; Limpus, C.J. Incubation period and sexual differentiation in the green turtle Chelonia mydas L. In Proceedings of the Melbourne Herpetological Symposium; Banks, C.B., Martin, A.A., Eds.; The Zoological Board of Victoria: Melbourne, Australia, 1981; pp. 66–73. [Google Scholar]
- Morreale, S.J.; Ruiz, G.J.; Spotila, J.R.; Standora, E.A. Temperature-dependent sex determination: Current practices threaten conservation of sea turtles. Science 1982, 216, 1245–1247. [Google Scholar] [CrossRef] [PubMed]
- Webster, P.J.; Holland, G.J.; Curry, J.A.; Chang, H.-R. Changes in tropical cyclone number, duration, and intensity in a warming environment. Science 2005, 309, 1844–1846. [Google Scholar] [CrossRef] [PubMed]
- Goldenberg, S.B.; Landsea, C.W.; Mestas-Nuñez, A.M.; Gray, W.M. The recent increase in Atlantic hurricane activity: Causes and implications. Science 2001, 293, 474–479. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Duan, X.; Raga, G.B.; Sun, F. Potential large-scale forcing mechanisms driving enhanced North Atlantic tropical cyclone activity since the mid-1990s. J. Clim. 2018, 31, 1377–1397. [Google Scholar] [CrossRef]
- Fraza, E.; Elsner, J.B.; Jagger, T.H. A space–time statistical climate model for hurricane intensification in the North Atlantic basin. Adv. Stat. Climatol. Meteorol. Oceanogr. 2016, 2, 105–114. [Google Scholar] [CrossRef][Green Version]
- Shamblin, B.M.; Dodd, M.G.; Bagley, D.A.; Ehrhart, L.M.; Tucker, A.D.; Johnson, C.; Carthy, R.R.; Scarpino, R.A.; McMichael, E.; Addison, D.S.; et al. Genetic structure of the southeastern United States loggerhead turtle nesting aggregation: Evidence of additional structure within the peninsular Florida recovery unit. Mar. Biol. 2011, 158, 571–587. [Google Scholar] [CrossRef]
- Shamblin, B.M.; Bolten, A.B.; Bjorndal, K.A.; Dutton, P.H.; Nielsen, J.T.; Abreu-Grobois, F.A.; Reich, K.J.; Witherington, B.E.; Bagley, D.A.; Ehrhart, L.M.; et al. Expanded mitochondrial control region sequences increase resolution of stock structure among North Atlantic loggerhead turtle rookeries. Mar. Ecol. Prog. Ser. 2012, 469, 145–160. [Google Scholar] [CrossRef]
- U.S. Fish and Wildlife Service; U.S. National Marine Fisheries Service. Recovery Plan for U.S. Population of Atlantic Green Turtle (Chelonia Mydas); NMFS: Washington, DC, USA, 1991.
- U.S. National Marine Fisheries Service; U.S. Fish and Wildlife Service. Recovery Plan for the Northwest Atlantic Population of the Loggerhead Sea Turtle (Caretta Caretta), 2nd ed.; NMFS: Silver Spring, MD, USA, 2008.
- U.S. National Marine Fisheries Service; U.S. Fish and Wildlife Service; Mexican Secretariat of Environmental and Natural Resources. Bi-National Recovery Plan for the Kemp’s Ridley Sea Turtle (Lepidochelys Kempii), 2nd ed.; NMFS: Silver Spring, MD, USA, 2011.
- U.S. National Marine Fisheries Service; U.S. Fish and Wildlife Service. Recovery Plan for Leatherback Turtles in the U.S. Caribbean, Atlantic and Gulf of Mexico; NMFS: Washington, DC, USA, 1992.
- Shaver, D.J.; Rubio, C.; Walker, J.S.; George, J.; Amos, A.F.; Reich, K.J.; Jones, C.; Shearer, T. Kemp’s ridley sea turtle (Lepidochelys kempii) nesting on the Texas coast: Geographic, temporal, and demographic trends through 2014. Gulf Mex. Sci. 2016, 2, 158–178. [Google Scholar] [CrossRef]
- South Carolina DNR Loggerhead Sea Turtle (Caretta caretta). Available online: http://www.dnr.sc.gov/marine/turtles/cc.htm (accessed on 15 January 2019).
- Georgia DNR Loggerhead Sea Turtle Fact Sheet. Available online: https://georgiawildlife.com/conservation/seaturtles (accessed on 15 January 2019).
- Talbert, O.R., Jr.; Stancyk, S.E.; Dean, J.M.; Will, J.M. Nesting activity of the loggerhead turtle (Caretta caretta) in South Carolina I: A rookery in transition. Copeia 1980, 1980, 709–719. [Google Scholar] [CrossRef]
- Halls, J.N.; Randall, A.L. Nesting patterns of loggerhead sea turtles (Caretta caretta): Development of a multiple regression model tested in North Carolina, USA. Int. J. Geo-Inf. 2018, 7, 348. [Google Scholar] [CrossRef]
- Davis, G.E.; Whiting, M.C. Loggerhead sea turtle nesting Everglades National Park, Florida, USA. Herpetologica 1977, 33, 18–28. [Google Scholar]
- Dodd, M.G. Synopsis of the Biological Data on the Loggerhead Sea Turtle Caretta Caretta (Linnaeus 1758); Biological Report, Fish and Wildlife Service; U.S. Department of the Interior: Washington, DC, USA, 1988.
- Witherington, B.E.; Bresette, M.; Herren, R.M. Chelonia mydas—Green turtle. In Biology and Conservation of Florida Turtles; Meylan, P.A., Ed.; Chelonian Research Foundation: Phnom Penh, Cambodia, 2006; pp. 90–104. [Google Scholar]
- Brost, B.; Witherington, B.E.; Meylan, A.B.; Leone, E.; Ehrhart, L.M.; Bagley, D.A. Sea turtle hatchling production from Florida (USA) beaches, 2002–2012, with recommendations for analyzing hatching success. Endanger. Species Res. 2015, 27, 53–68. [Google Scholar] [CrossRef]
- Caldwell, D.K. The loggerhead turtles of Cape Romain, South Carolina. Bull. Fla. State Mus. 1959, 4, 319–348. [Google Scholar]
- U.S. Fish and Wildlife Service Green Sea Turtle (Chelonia mydas). Available online: https://www.fws.gov/northflorida/seaturtles/turtle factsheets/green-sea-turtle.htm (accessed on 15 January 2019).
- U.S. Fish and Wildlife Service Leatherback Sea Turtle (Dermochelys coriacea). Available online: https://www.fws.gov/northflorida/seaturtles/turtle factsheets/leatherback-sea-turtle.htm (accessed on 15 January 2019).
- Stewart, K.R.; Johnson, C. Dermochelys coriacea—Leatherback sea turtle. In Biology and Conservation of Florida Turtles; Meylan, P.A., Ed.; Chelonian Research Foundation: Phnom Penh, Cambodia, 2006; pp. 144–157. [Google Scholar]
- National Hurricane Center National Hurricane Center. Available online: https://www.nhc.noaa.gov/ (accessed on 20 September 2002).
- Dvorak, V.F. A Technique for the Analysis and Forecasting of Tropical Cyclone Intensities from Satellite Pictures; NOAA Tech(National Technical Information Service): Washington, DC, USA, 1973. [Google Scholar]
- National Hurricane Center. National Hurricane Center Product Description Document: A User’s Guide to Hurricane Products; National Hurricane Center: Miami, FL, USA, 2019.
- Knapp, K.R.; Kruk, M.C.; Levinson, D.H.; Diamond, H.J.; Neumann, C.J. The international best track archive for climate stewardship (IBTrACS). Bull. Am. Meteorol. Soc. 2010, 91, 363–376. [Google Scholar] [CrossRef]
- Wildermann, N. Flatbacks at Sea: Understanding Ecology in Foraging Populations. Ph.D. Thesis, James Cook University, Townsville, Australia, 2017. [Google Scholar]
- Shamblin, B.M.; Godfrey, M.H.; Pate, S.M.; Thompson, W.P.; Sutton, H.; Altman, J.; Fair, K.; McClary, J.; Wilson, A.M.N.; Milligan, B.; et al. Green turtles nesting at their northern range limit in the United States represent a distinct subpopulation. Chelonian Conserv. Biol. 2018, 17, 314–319. [Google Scholar] [CrossRef]
- Shamblin, B.M.; Bagley, D.A.; Ehrhart, L.M.; Desjardin, N.A.; Martin, E.; Hart, K.M.; Naro-Maciel, E.; Rusenko, K.C.; Stiner, J.; Sobel, D.; et al. Genetic structure of Florida green turtle rookeries as indicated by mitochondrial DNA control region sequences. Conserv. Genet. 2014, 16, 673–685. [Google Scholar] [CrossRef]
- Team, R.C. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Miller, J.D. Determining clutch size and hatching success. In Research and Management Techniques for the Conservation of Sea Turtles; Eckert, K.L., Bjorndal, K.A., Abreu-Grobois, F.A., Donnelly, M., Eds.; IUCN/SSC Marine Turtle Specialist Group: Gland, Switzerland, 1999; pp. 130–135. [Google Scholar]
- Ross, J.P. Hurricane effects on nesting Caretta caretta. Mar. Turt. Newsl. 2005, 108, 13–14. [Google Scholar]
- Richards, P.M.; Epperly, S.P.; Heppell, S.S.; King, R.T.; Sasso, C.R.; Moncada, F.; Nodarse, G.; Shaver, D.J.; Medina, Y.; Zurita, J.C. Sea turtle population estimates incorporating uncertainty: A new approach applied to western North Atlantic loggerheads Caretta caretta. Endanger. Species Res. 2011, 15, 151–158. [Google Scholar] [CrossRef]
- Fuentes, M.M.P.B.; Limpus, C.J.; Hamann, M.; Dawson, J. Potential impacts of projected sea-level rise on sea turtle rookeries. Aquat. Conserv. Mar. Freshw. Ecosyst. 2010, 20, 132–139. [Google Scholar] [CrossRef]
- Hays, G.C.; Speakman, J.R. Nest placement by loggerhead turtles, Caretta caretta. Anim. Behav. 1993, 45, 47–53. [Google Scholar] [CrossRef]
- Wetterer, J.K.; Wood, L.D.; Johnson, C.; Krahe, H.; Fitchett, S. Predaceous ants, beach replenishment, and nest placement by sea turtles. Environ. Entomol. 2007, 36, 655–656. [Google Scholar] [CrossRef]
- Miller, J.D. Reproduction in sea turtles. In The Biology of Sea Turtles; Lutz, P.L., Musick, J.A., Eds.; CRC Press: Boca Raton, FL, USA, 1997; Volume 1, pp. 51–81. [Google Scholar]
- Heppell, S.S.; Limpus, C.J.; Crouse, D.T.; Frazer, N.B.; Crowder, L.B. Population model analysis for the loggerhead sea turtle, Caretta caretta, in Queensland. Wildl. Res. 1996, 23, 143–161. [Google Scholar] [CrossRef]
- Zimmermann, N.E.; Yoccoz, N.G.; Edwards, T.C.; Meier, E.S.; Thuiller, W.; Guisan, A.; Schmatz, D.R.; Pearman, P.B. Climatic extremes improve predictions of spatial patterns of tree species. Proc. Natl. Acad. Sci. USA 2009, 106, 19723–19728. [Google Scholar] [CrossRef]
- Wootton, J.T. Effects of disturbance on species diversity: A multitrophic perspective. Am. Nat. 1998, 152, 803–825. [Google Scholar] [CrossRef]
- Knutson, T.; Camargo, S.J.; Chan, J.C.L.; Emanuel, K.; Ho, C.-H.; Kossin, J.; Mohapatra, M.; Satoh, M.; Sugi, M.; Walsh, K.; et al. Tropical cyclones and climate change assessment: Part II. Projected response to anthropogenic warming. Bull. Am. Meteorol. Soc. 2019. [Google Scholar] [CrossRef]
- Harley, C.D.G.; Randall Hughes, A.; Hultgren, K.M.; Miner, B.G.; Sorte, C.J.B.; Thornber, C.S.; Rodriguez, L.F.; Tomanek, L.; Williams, S.L. The impacts of climate change in coastal marine systems. Ecol. Lett. 2006, 9, 228–241. [Google Scholar] [CrossRef]
- Darling, E.S.; Côté, I.M. Quantifying the evidence for ecological synergies. Ecol. Lett. 2008, 11, 1278–1286. [Google Scholar] [CrossRef]
- Witherington, B.E.; Hirama, S.; Mosier, A. Sea turtle responses to barriers on their nesting beach. J. Exp. Mar. Bio. Ecol. 2011, 401, 1–6. [Google Scholar] [CrossRef]
- Witherington, B.E.; Hirama, S.; Mosier, A.E. Barriers to sea turtle nesting on Florida (United States) beaches: Linear extent and changes following storms. J. Coast. Res. 2011, 27, 450–458. [Google Scholar] [CrossRef]
- Klemas, V.V. The role of remote sensing in predicting and determining coastal storm impacts. J. Coast. Res. 2009, 1264–1275. [Google Scholar] [CrossRef]
| Average Number of Named Storms from 2008–2018 (Range) | Average Named Storm Exposure (Range) | Average Named Storm Exposure Category | |
|---|---|---|---|
| Loggerhead turtles | 7.6 (2–14) | 0.27 (0.05–0.92) | Medium |
| Green turtles | 7.2 (4–13) | 0.24 (0.07–0.42) | Low |
| Leatherback turtles | 6.8 (4–12) | 0.22 (0.08–0.32) | Low |
| Kemp’s ridley turtles | 3.7 (2–6) | 0.12 (0.05–0.21) | Low |
| Loggerhead management unit | |||
| North–western Florida (NW), | 6.7 (2–9) | 0.27 (0.10–0.57) | Medium |
| Central–western Florida (CW) | 6.2 (4–8) | 0.23 (0.07–0.50) | Low |
| South–western Florida (SW) | 4.8 (4–5) | 0.16 (0.07–0.35) | Low |
| South–eastern Florida (SE) | 4.9 (3–7) | 0.17 (0.05–0.32) | Low |
| Central–eastern Florida (CE) | 5.5 (4–8) | 0.21 (0.07–0.42) | Low |
| Northern (N) | 10.0 (6–14) | 0.36 (0.10–0.92) | Medium |
| HS for Pre-Hurricane Nests (SD) | Number of Nests (% of Nests for Each Species) | HS for Post-Hurricane Nests (SD) | Number of Nests (% of Nests for Each Species) | p Value | |
|---|---|---|---|---|---|
| Loggerhead turtles | 73.1 (32.1) | 635 (84%) | 18.7 (33.9) | 122 (16%) | <0.0001 |
| Green turtles | 36.7 (35.9) | 7 (35%) | 29.4 (43.0) | 13 (65%) | >0.05 |
| Kemp’s ridley turtles | 64.5 (34.8) | 12 (100%) | None laid | None laid | n/a |
| Leatherback turtles | 1.5 (2.0) | 2 (100%) | None laid | None laid | n/a |
| HS <35° Latitude (SD) | Number of Nests (% of Nests for Each Species) | HS >°35 Latitude (SD) | Number of Nests (% of Nests for Each Species) | p Value (Dunn’s Multiple Comparison Test) | |
|---|---|---|---|---|---|
| Loggerhead turtles | 16.5 (33.9) | 79 (64.7%) | 22.9 (33.9) | 43 (35.3%) | >0.05 |
| Green turtles | 2.3 (6.5) | 8 (61.5%) | 72.7 (40.8) | 5 (38.5%) | <0.01 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fuentes, M.M.P.B.; Godfrey, M.H.; Shaver, D.; Ceriani, S.; Gredzens, C.; Boettcher, R.; Ingram, D.; Ware, M.; Wildermann, N. Exposure of Marine Turtle Nesting Grounds to Named Storms Along the Continental USA. Remote Sens. 2019, 11, 2996. https://doi.org/10.3390/rs11242996
Fuentes MMPB, Godfrey MH, Shaver D, Ceriani S, Gredzens C, Boettcher R, Ingram D, Ware M, Wildermann N. Exposure of Marine Turtle Nesting Grounds to Named Storms Along the Continental USA. Remote Sensing. 2019; 11(24):2996. https://doi.org/10.3390/rs11242996
Chicago/Turabian StyleFuentes, Mariana M. P. B., Matthew H. Godfrey, Donna Shaver, Simona Ceriani, Christian Gredzens, Ruth Boettcher, Dianne Ingram, Matthew Ware, and Natalie Wildermann. 2019. "Exposure of Marine Turtle Nesting Grounds to Named Storms Along the Continental USA" Remote Sensing 11, no. 24: 2996. https://doi.org/10.3390/rs11242996
APA StyleFuentes, M. M. P. B., Godfrey, M. H., Shaver, D., Ceriani, S., Gredzens, C., Boettcher, R., Ingram, D., Ware, M., & Wildermann, N. (2019). Exposure of Marine Turtle Nesting Grounds to Named Storms Along the Continental USA. Remote Sensing, 11(24), 2996. https://doi.org/10.3390/rs11242996