Sentinel-MSI VNIR and SWIR Bands Sensitivity Analysis for Soil Salinity Discrimination in an Arid Landscape
Abstract
1. Introduction
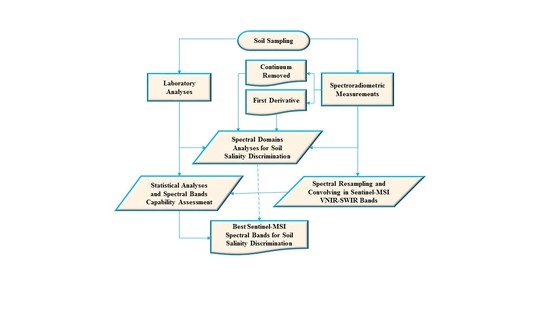
2. Materials and Methods
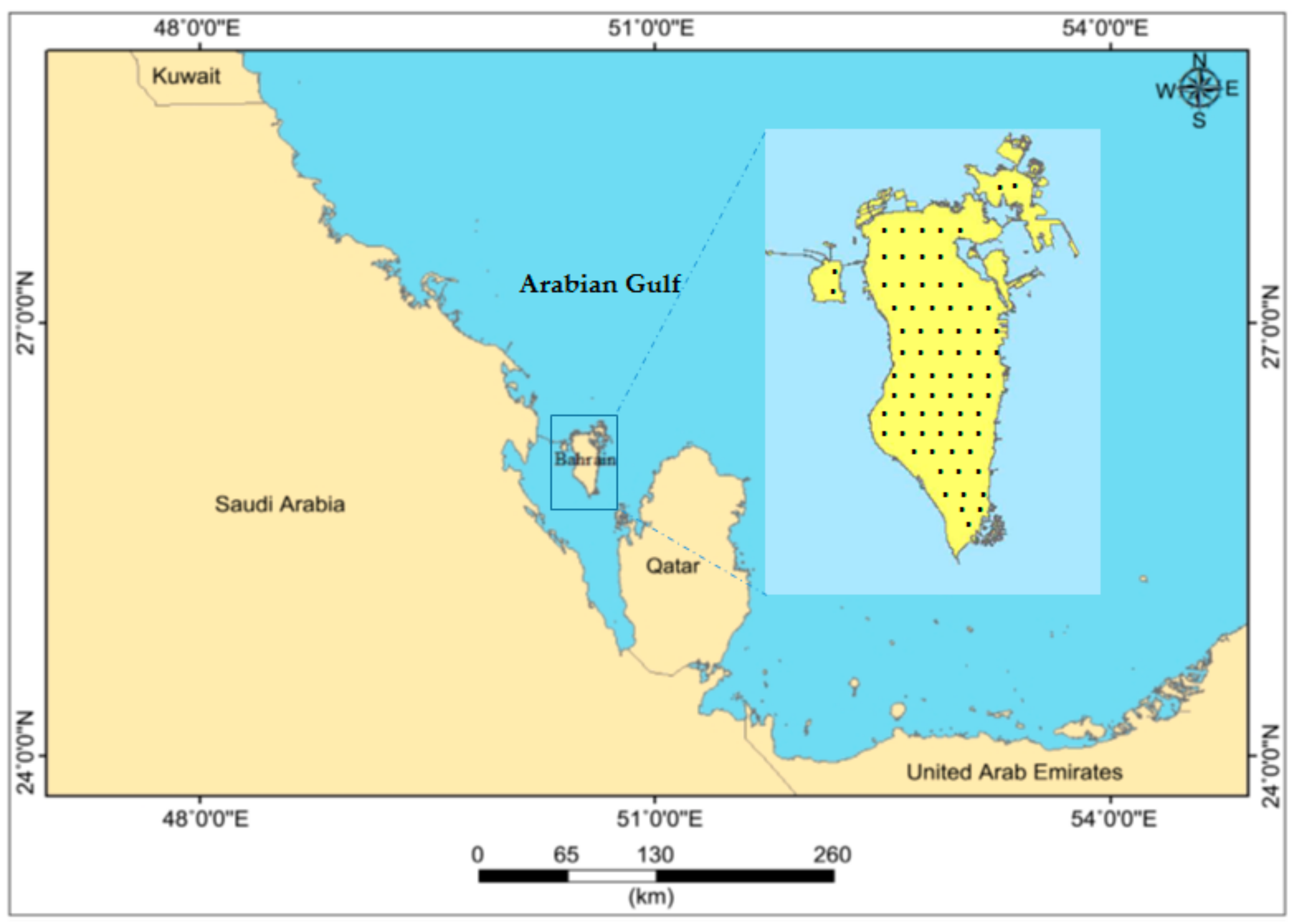
2.1. Study Site
2.2. Soil Sampling and Laboratory Analyses
2.3. Spectroradiometric Measurements
2.4. Continuum-Removed Reflectance Spectrum
2.5. First Derivative
2.6. Sentinel-MSI Simulated Data
3. Results and Discussion
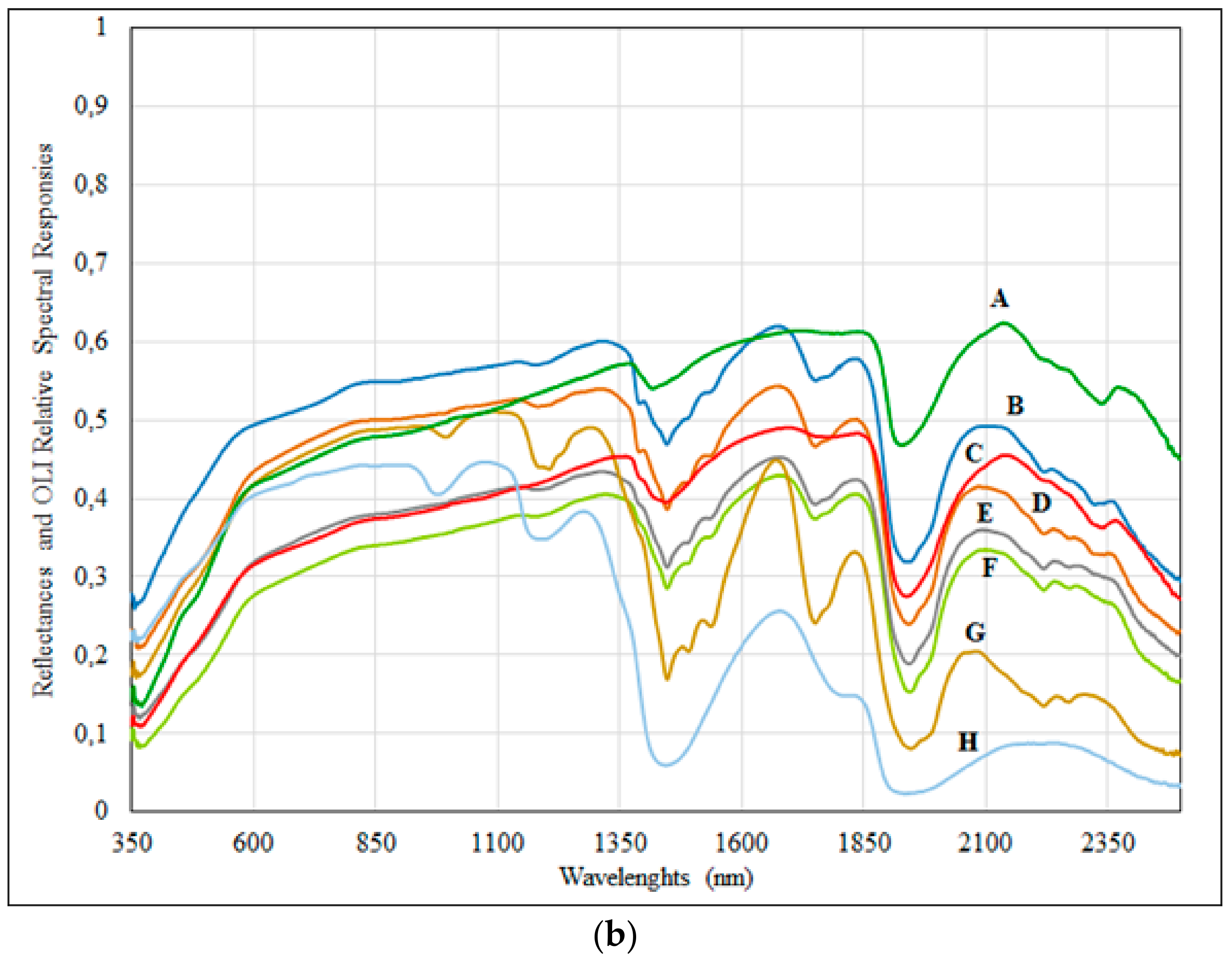
3.1. Spectral and Laboratory Analyses
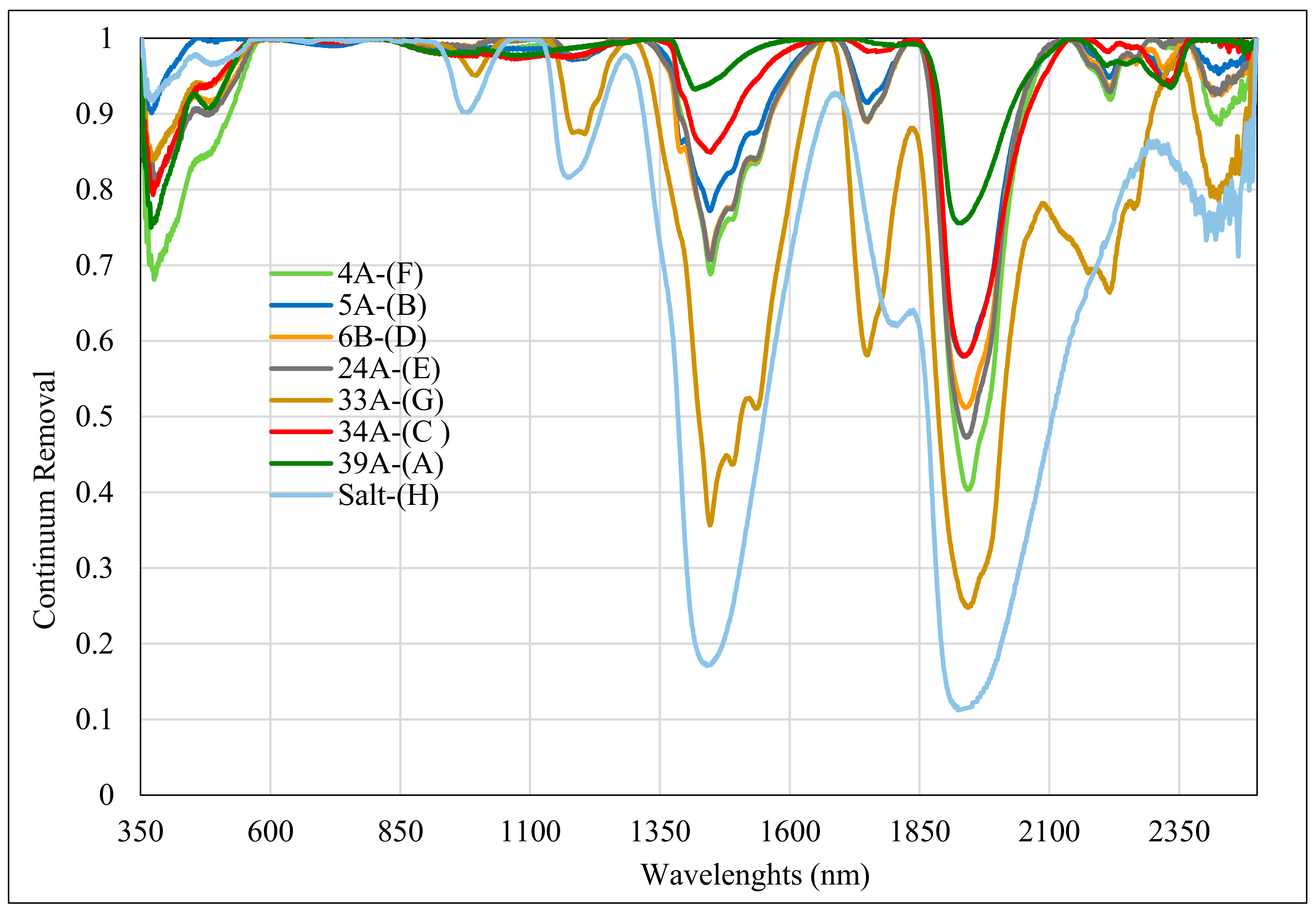
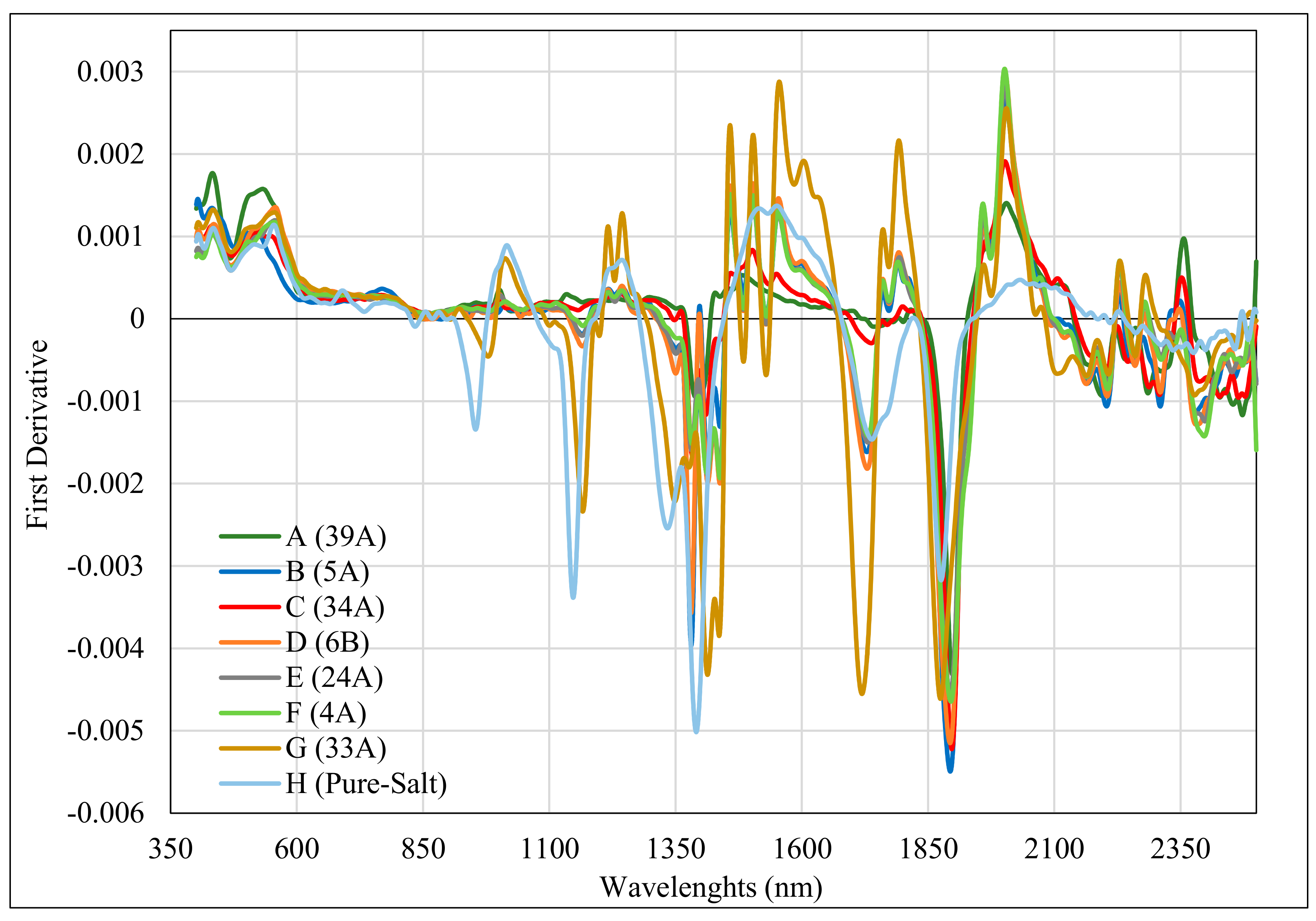
3.2. CRRS and FD Analyses
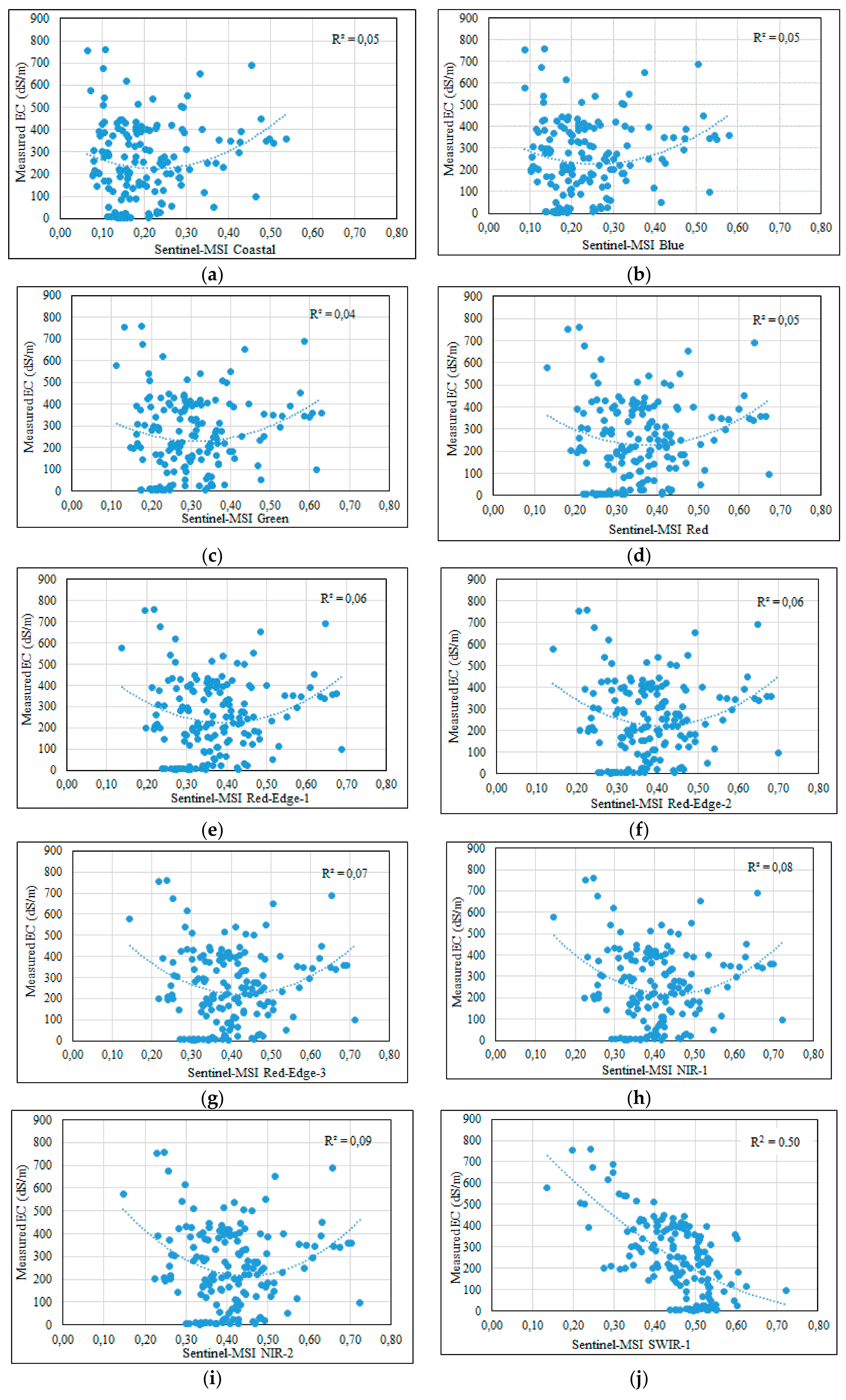
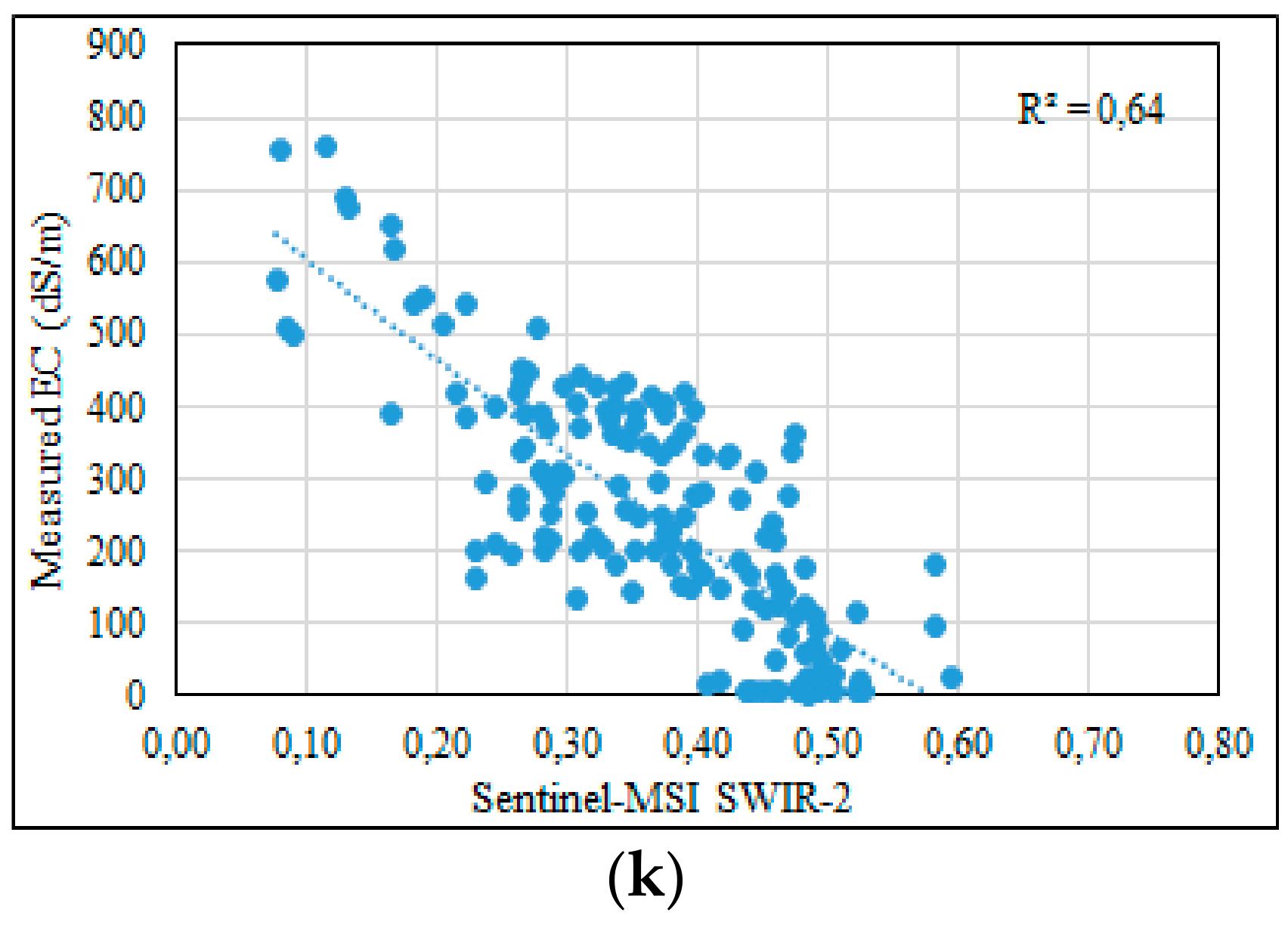
3.3. Statistical Analysis between EC-Lab and Spectral Bands of MSI
3.4. Discussion
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Metternicht, G.; Zinck, J.A. Remote Sensing of Soil Salinization: Impact on Land Management; CRC Press Taylor and Francis Group: Boca Raton, FL, USA, 2009; 374p. [Google Scholar]
- Metternicht, G.I.; Zinck, J.A. Remote sensing of soil salinity: Potentials and constraints. Remote Sens. Environ. 2003, 85, 1–20. [Google Scholar] [CrossRef]
- Smedema, L.K. Salinity control in irrigated land: Use of remote sensing techniques in irrigation and drainage. In Proceedings of the Expert Consultation, Session 3—Drainage and Salinity Monitoring and Control, Montpellier, France, 2–4 November 1993; FAO, Water Reports 4. pp. 141–150. [Google Scholar]
- Zhang, H.; Schroder, J.L.; Pittman, J.J.; Wang, J.J.; Payton, M.E. Soil Salinity Using Saturated Paste and 1:1 Soil to Water Extracts. Soil Sci. Soc. Am. J. 2005, 69, 1146–1151. [Google Scholar] [CrossRef]
- Norman, C.P.; Lyle, C.W.; Heuperman, A.F.; Poulton, D. Tragowel Plains—Challenge of the Plains. In Tragowel Plains Salinity Management Plan, Soil Salinity Survey, Tragowel Plains Subregional Working Group; Victorian Department of Agriculture: Melbourne, Australia, 1989; pp. 49–89. [Google Scholar]
- Mougenot, B.; Pouget, M.; Epema, G. Remote sensing of salt affected soils. Remote Sens. Rev. 1994, 7, 241–259. [Google Scholar] [CrossRef]
- Verma, K.S.; Saxena, R.K.; Barthwal, A.K.; Deshmukh, S.N. Remote sensing technique for mapping salt affected soils. Int. J. Remote Sens. 1994, 15, 1901–1914. [Google Scholar] [CrossRef]
- Metternicht, G.I.; Zinck, J.A. Spatial discrimination of salt- and sodium-affected soil surfaces. Int. J. Remote Sens. 1997, 18, 2571–2586. [Google Scholar] [CrossRef]
- Hashem, M.; El-Khattib, N.; El-Mowelhi, M.; Abd El-Salam, A. Desertification and land degradation using high resolution satellite data in the Nile Delta, Egypt. In Proceedings of the IGARSS-1997, Singapore, 3–8 August 1997; pp. 197–199. [Google Scholar]
- Goosens, R.; El Badawi, M.; Ghabour, T.; De Dapper, M. A simulated model to monitor the soil salinity in irrigated arable land in arid areas based upon remote sensing and GIS. EARSeL Adv. Remote Sens. 1998, 2, 165–171. [Google Scholar]
- Ben-Dor, E.; Metternicht, G.; Goldshleger, N.; Mor, E.; Mirlas, V.; Basson, U. Review of Remote Sensing-Based Methods to Assess Soil Salinity. In Remote Sensing of Soil Salinization: Impact on Land Management; Metternicht, G., Zinck, J.A., Eds.; CRC Press Taylor and Francis Group: Boca Raton, FL, USA, 2009; Chapter 13; pp. 39–60. [Google Scholar]
- Allbed, A.; Kumar, L.; Sinha, P. Mapping and Modelling Spatial Variation in Soil Salinity in the Al Hassa Oasis Based on Remote Sensing Indicators and Regression Techniques. Remote Sens. 2014, 6, 1137–1157. [Google Scholar] [CrossRef]
- Nawar, S.; Buddenbaum, H.; Hill, J.; Kozak, J. Modeling and Mapping of Soil Salinity with Reflectance Spectroscopy and Landsat Data Using Two Quantitative Methods (PLSR and MARS). Remote Sens. 2014, 6, 10813–10834. [Google Scholar] [CrossRef]
- Bannari, A.; Guedon, A.M.; El-Harti, A.; Cherkaoui, F.Z.; El-Ghmari, A. Characterization of Slight and Moderate Saline and Sodic Soils in Irrigated Agricultural Land Using Simulated Data of ALI (EO-1) Sensor. Commun. Soil Sci. Plant Anal. 2008, 39, 2795–2811. [Google Scholar] [CrossRef]
- Bannari, A.; Guedon, A.M.; El-Ghmari, A. Mapping Slight and Moderate Saline Soils in Irrigated Agricultural Land Using Advanced Land Imager Sensor (EO-1) Data and Semi-Empirical Models. Commun. Soil Sci. Plant Anal. 2016, 47, 1883–1906. [Google Scholar] [CrossRef]
- Scudiero, E.; Skaggs, T.H.; Corwin, D.L. Comparative regional-scale soil salinity assessment with near-ground apparent electrical conductivity and remote sensing canopy reflectance. Ecol. Indic. 2016, 70, 276–284. [Google Scholar] [CrossRef]
- Nijat, K.; Tashpolat, T.; Abdugheni, A.; Ilyas, N.; Rukeya, S.; Balati, M. Mapping and Modeling of Soil Salinity Using WorldView-2 Data and EM38-KM2 in an Arid Region of the Keriya River, China. Photogramm. Eng. Remote Sens. 2018, 84, 43–52. [Google Scholar] [CrossRef]
- El-Battay, A.; Bannari, A.; Hameid, N.A.; Abahussain, A.A. Comparative Study among Different Semi-Empirical Models for Soil Salinity Prediction in an Arid Environment Using OLI Landsat-8 Data. Adv. Remote Sens. 2017, 6, 23–39. [Google Scholar] [CrossRef][Green Version]
- Rahmati, M.; Hamzehpour, N. Quantitative remote sensing of soil electrical conductivity using ETM+ and ground measured data. Int. J. Remote Sens. 2017, 38, 123–140. [Google Scholar] [CrossRef]
- Zinck, J.A. Monitoring soil salinity from remote sensing data. In Proceedings of the 1st Workshop EARSel Special Interest Group on Remote Sensing for Developing Countries, Gent, Belgium, 13–15 September 2000; pp. 359–368. [Google Scholar]
- Mandanici, E.; Bitelli, G. Preliminary Comparison of Sentinel-2 and Landsat 8 Imagery for a Combined Use. Remote Sens. 2016, 8, 1014. [Google Scholar] [CrossRef]
- Van-derWerff, H.; Van-derMeer, F. Sentinel-2A MSI and Landsat 8 OLI provide data continuity for geological remote sensing. Remote Sens. 2016, 8, 883. [Google Scholar] [CrossRef]
- Analytical Spectral Devices, ASD Inc., 1999. Available online: http://www.asdi.com/products-spectroradiometers.asp (accessed on 18 March 2017).
- Clark, R.N.; King, T.V.V.; Gorelick, N.S. Automatic continuum analysis of reflectance spectra. In Proceedings of the JPL 3rd Airborne Imaging Spectrometer Data Analysis Workshop; 1987; pp. 138–142. Available online: https://ntrs.nasa.gov/archive/nasa/casi.ntrs.nasa.gov/19880004388.pdf (accessed on 18 March 2017).
- Tsai, F.; Philpot, W.D. Derivative analysis of hyperspectral data. Remote Sens. Environ. 1998, 66, 41–51. [Google Scholar] [CrossRef]
- Teillet, P.; Santer, R. Terrain Elevation and Sensor Altitude Dependence in a Semi-Analytical Atmospheric Code. Can. J. Remote Sens. 1991, 17, 36–44. [Google Scholar]
- USDA-NRCS. Soil Survey Laboratory Methods Manual; Soil Survey Investigations Report, No. 42 Version 4; Burt, R., Ed.; USDA-NRCS: Washington, DC, USA, 2004; 736p. [Google Scholar]
- Elagib, N.A.; Abdu, S.A.A. Climate variability and aridity in Bahrain. J. Arid Environ. 1997, 36, 405–419. [Google Scholar] [CrossRef]
- FAO Bahrain: Geography, Climate and Population. 2015. Available online: http://www.fao.org/nr/water/aquastat/countries_regions/bahrain/index.stm (accessed on 18 March 2017).
- Boonthaiiwai, C.; Saenjan, P. Food Security and Socio-economic Impacts of Soil Salinization in Northeast Thailand. Int. J. Environ. Rural Dev. 2013, 4, 76–81. [Google Scholar]
- Doomkamp, J.C.; Brunsden, D.; Jones, D.K.C. Geology, Geomorphology and Pedology of Bahrain; Geo-Abstracts Ltd., University of East Anglia: Norwich, UK, 1980; 443p. [Google Scholar]
- Jackson, R.D.; Pinter, P.J.; Paul, J.; Reginato, R.J.; Robert, J.; Idso, S.B. Hand-Held Radiometry; Agricultural Reviews and Manuals, ARM-W-19; U.S. Department of Agriculture Science and Education Administration: Phoenix, AZ, USA, 1980.
- Fabri, A.; Giezeman, G.-J.; Kettner, L.; Schirra, S.; Sven, S. On the Design of CGAL, the Computational Geometry Algorithms Library. RR-3407. 1998. Available online: https://hal.inria.fr/inria-00073283/document (accessed on 18 March 2017).
- Van-Der-Meera, F. Analysis of spectral absorption features in hyperspectral imagery. Int. J. Appl. Earth Obs. Geoinf. 2004, 5, 55–68. [Google Scholar] [CrossRef]
- Crowley, J.K.; Brickey, D.W.; Rowan, L.C. Airborne imaging spectrometer data of the Ruby Mountains, Montana: Mineral discrimination using relative absorption-band-depth images. Remote Sens. Environ. 1989, 29, 121–134. [Google Scholar] [CrossRef]
- Clark, R.N.; Gallagher, A.J.; Swayze, G.A. Material absorption-band depth mapping of imaging spectrometer data using the complete band shape least-squares algorithm simultaneously fit to multiple spectral features from multiple materials. In Proceedings of the Third Airborne Visible/Infrared Imaging Spectrometer (AVIRIS) Workshop, Jet Propulsion Laboratory, Pasadena, CA, USA, 20–21 May 1991. [Google Scholar]
- Clark, R.N.; Swayze, G.A. Mapping minerals, amorphous materials, environmental materials, vegetation, water, ice, and other materials: The USGS Tricorder Algorithm. In Proceedings of the Summaries of the Fifth Annual JPL Airborne Earth Science Workshop, Pasadena, CA, USA, 23–26 January 1995; JPL Publication 95-1. Volume 2, pp. 39–40. [Google Scholar]
- Clark, R.N.; Swayze, G.A.; Livo, K.E.; Kokaly, R.F.; Sutley, S.J.; Dalton, J.B.; McDougal, R.R.; Gent, C.A. Imaging spectroscopy: Earth and planetary remote sensing with the USGS Tetracorder and expert systems. J. Geophys. Res. 2003, 108, 5131. [Google Scholar] [CrossRef]
- Clark, R.N.; Swayze, G.A.; Carlson, R.; Grundy, W.; Noll, K. Spectroscopy from Space. Rev. Mineral. Geochem. 2014, 78, 399–446. [Google Scholar] [CrossRef]
- ENVI. Exelis Visual Information Solutions (ENVI) Tutorials; ENVI: Boulder, CO, USA, 2012; Available online: http://www.exelisvis.com/docs/Tutorials.html (accessed on 26 May 2017).
- Tsai, F.; Philpot, W.D. A Derivative-Aided Hyperspectral Image Analysis System for Land-Cover Classification. IEEE Trans. Geosci. Remote Sens. 2002, 40, 416–425. [Google Scholar] [CrossRef]
- Demetriades-Shah, T.H.; Evapotranspiration Laboratory, Department of Agronomy, Kansas State University, Manhattan USA; Steven, M.D.; Clark, J.A. High-resolution derivative spectra in remote sensing. Remote Sens. Environ. 1990, 33, 55–64. [Google Scholar] [CrossRef]
- Morrey, J.R. On Determining Spectral Peak Positions from Composite Spectra with a Digital Computer. Anal. Chem. 1968, 40, 905–914. [Google Scholar] [CrossRef]
- ASD. What Is a Derivative Spectrum? 2017. Available online: https://www.asdi.com/learn/faqs/what-is-a-derivative-spectrum (accessed on 2 February 2017).
- Owen, A.J. Uses of Derivative Spectroscopy. Application Note, Agilent Technologies Innovating the HP Way. 1995. Available online: http://www.whoi.edu/cms/files/derivative_spectroscopy_59633940_175744.pdf (accessed on 2 February 2017).
- MATLAB. MathWorks: MATLAB V-8.0; The MathWorks Inc.: Natick, MA, USA, 2012; Available online: http://www.mathworks.com/products/matlab/whatsnew.html (accessed on 6 November 2016).
- Hedley, J.; Roelfsema, C.; Koetz, B.; Phinn, S. Capability of the Sentinel 2 mission for tropical coral reef mapping and coral bleaching detection. Remote Sens. Environ. 2012, 120, 145–155. [Google Scholar] [CrossRef]
- Mulders, M. Remote Sensing in Soil Science. In Development in Soil Science; Elsevier: Amsterdam, The Netherlands, 1987; 379p. [Google Scholar]
- Csillag, F.; Pasztor, L.; Biehl, L. Spectral band selection for the characterization of salinity statues of soils. Remote Sens. Environ. 1993, 43, 231–242. [Google Scholar] [CrossRef]
- Hawari, F. Spectroscopy of evaporates. Per. Miner. 2002, 71, 191–200. [Google Scholar]
- Farifteh, J. Imaging Spectroscopy of Salt-Affected Soils: Model-Based Integrated Method. Ph.D. Thesis, International Institute for Geo-information Science and Earth Observation, Utrecht University, Utrecht, Enschede, The Netherlands, 2007. Dissertation No. 143, ITC. 235p. [Google Scholar]
- Hunt, G.R.; Salisbury, J.W.; Lenhhoff, C.J. Visible and near infrared spectra of minerals and rocks: III. Oxides and Hydroxides. Mod. Geol. 1971, 2, 193–205. [Google Scholar]
- Drake, N.A. Reflectance spectra of evaporite minerals (400–2500 nm): Applications for remote sensing. Int. J. Remote Sens. 1995, 16, 55–71. [Google Scholar] [CrossRef]
- Goldshleger, N.; Ben-Dor, E.; Benyamini, Y.; Agassi, M.; Blumber, D. Characterization of soil’s structural crust by spectral reflectance in the SWIR region (1.2–2.5 μm). Terra Nova 2001, 13, 12–17. [Google Scholar] [CrossRef]
- Amos, B.J.; Greenbaum, D. Alteration detection using TM imagery: The effects of supergene weathering in an arid climate. Int. J. Remote Sens. 1989, 10, 515–527. [Google Scholar] [CrossRef]
- Ben-Dor, E.; Banin, A. Near-infrared reflectance analysis of carbonate concentration in soils. Appl. Spectrosc. 1990, 44, 1064–1069. [Google Scholar] [CrossRef]
- Lobell, D.B.; Asner, G.P. Moisture effects on soil reflectance. Soil Sci. Soc. Am. J. 2002, 66, 722–727. [Google Scholar] [CrossRef]
- Whiting, M.L.; Li, L.; Ustin, S.L. Predicting water content using Gaussian model on soil spectra. Remote Sens. Environ. 2004, 89, 535–552. [Google Scholar] [CrossRef]
- Mashimbye, Z.E. Remote Sensing of Salt-Affected Soil. Ph.D. Thesis, Faculty of Agri-Sciences, Stellenbosch University, Stellenbosch, South Africa, 2013; 151p. [Google Scholar]
- Nawar, S.; Buddenbaum, H.; Hill, J. Digital Mapping of Soil Properties Using Multivariate Statistical Analysis and ASTER Data in an Arid Region. Remote Sens. 2015, 7, 1181–1205. [Google Scholar] [CrossRef]
- Bannari, A.; El-Battay, A.; Hameid, N.; Tashtoush, F. Salt-Affected Soil Mapping in an Arid Environment using Semi-Empirical Model and Landsat-OLI Data. Adv. Remote Sens. 2017, 6, 260–291. [Google Scholar] [CrossRef][Green Version]
- Bannari, A.; Shahid, S.A.; El-Battay, A.; Alshankiti, A.; Hameid, N.A.; Tashtoush, F. Potential of WorldView-3 data for Soil Salinity Modeling and Mapping in an Arid Environment. In Proceedings of the International Geoscience and Remote Sensing Symposium (IGARSS-2017), Fort Worth, TX, USA, 23–28 July 2017; pp. 1585–1588. [Google Scholar]
- Farifteh, J.; van der Meer, F.; van der Meijde, M.; Atzberger, C. Spectral characteristics of salt-affected soils: A laboratory experiment. Geoderma 2007, 145, 196–206. [Google Scholar] [CrossRef]
- Weng, Y.; Gong, P.; Zhu, Z. Soil salt content estimation in the Yellow River delta with satellite hyperspectral data. Can. J. Remote Sens. 2008, 34, 259–270. [Google Scholar]
- Metternicht, G.I. Detecting and Monitoring Land Degradation Features and Processes in the Cochamba Valleys, Bolivia: A Synergistic Approach. Ph.D. Thesis, International Institute for Geo-information Science and Earth Observation, Utrecht University, Utrecht, Enschede, The Netherlands, 1996. Dissertation No. 36, ITC. 422p. [Google Scholar]
- Chapman, J.E.; Rothery, D.A.; Francis, P.W.; Pontual, A. Remote sensing of evaporite mineral zonation in salt flats (salars). Int. J. Remote Sens. 1989, 10, 245–255. [Google Scholar] [CrossRef]
- Taylor, G.; Deehan, R. Mapping soil salinity with hyperspectral imagery. In Proceedings of the 14th International Conference Applied Geologic Remote Sensing, Las Vegas, NV, USA, 6–8 November 2000; pp. 512–520. [Google Scholar]
- Katawatin, R.; Kotrapat, W. Use of LANDSAT-7 ETM+ with ancillary data for soil salinity mapping in Northeast Thailand. In Proceedings of the Third International Conference on Experimental Mechanics and Third Conference of the Asian Committee on Experimental Mechanics, Singapore, 29 November–1 December 2004; Volume 5852, pp. 708–716. [Google Scholar]
- Shrestha, R.P. Relating soil electrical conductivity to remote sensing and other soil properties for assessing soil salinity in northeast Thailand. Land Degrad. Dev. 2006, 17, 677–689. [Google Scholar] [CrossRef]
- Leone, A.P.; Menenti, M.; Buondonno, A.; Letizia, A.; Maffei, C.; Sorrentino, G. A field experiment on spectrometry of crop response to soil salinity. Agric. Water Manag. 2007, 89, 39–48. [Google Scholar] [CrossRef]
- Odeh, I.O.A.; Onus, A. Spatial analysis of soil salinity and soil structural stability in a semiarid region of New South Wales, Australia. Environ. Manag. 2008, 42, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.T.; Zeng, S.L.; Gao, Y.; Ouyang, Z.T.; Li, B.; Fang, C.M.; Zhao, B. Using hyperspectral vegetation indices as a roxy to monitor soil salinity. Ecol. Indic. 2011, 11, 1552–1562. [Google Scholar] [CrossRef]
- Fan, X.W.; Liu, Y.B.; Tao, J.M.; Weng, Y.L. Soil salinity retrieval from advanced multi-spectral sensor with partial least square regression. Remote Sens. 2015, 7, 488–511. [Google Scholar] [CrossRef]
- Madani, A.A. Soil salinity detection and monitoring using Landsat data: A case study from Siwa Oasis, Egypt. GISci. Remote Sens. 2005, 42, 171–181. [Google Scholar] [CrossRef]
- El-Harti, A.; Lhissoua, R.; Chokmani, K.; Ouzemou, J.; Hassouna, M.; Bachaouia, E.M.; El-Ghmari, A. Spatiotemporal Monitoring of Soil Salinization in Irrigated Tadla Plain (Morocco) using Satellite Spectral Indices. Int. J. Appl. Earth Obs. Geoinf. 2016, 50, 64–73. [Google Scholar] [CrossRef]
- Bai, L.; Wang, C.; Zang, S.; Zhang, Y.; Hao, Q.; Wu, Y. Remote Sensing of Soil Alkalinity and Salinity in the Wuyu’er-Shuangyang River Basin, Northeast China. Remote Sens. 2016, 8, 163. [Google Scholar] [CrossRef]
- NASA Landsat Science. 2013. Available online: http://landsat.gsfc.nasa.gov/landsat-8/landsat-8-bands/ (accessed on 29 January 2017).
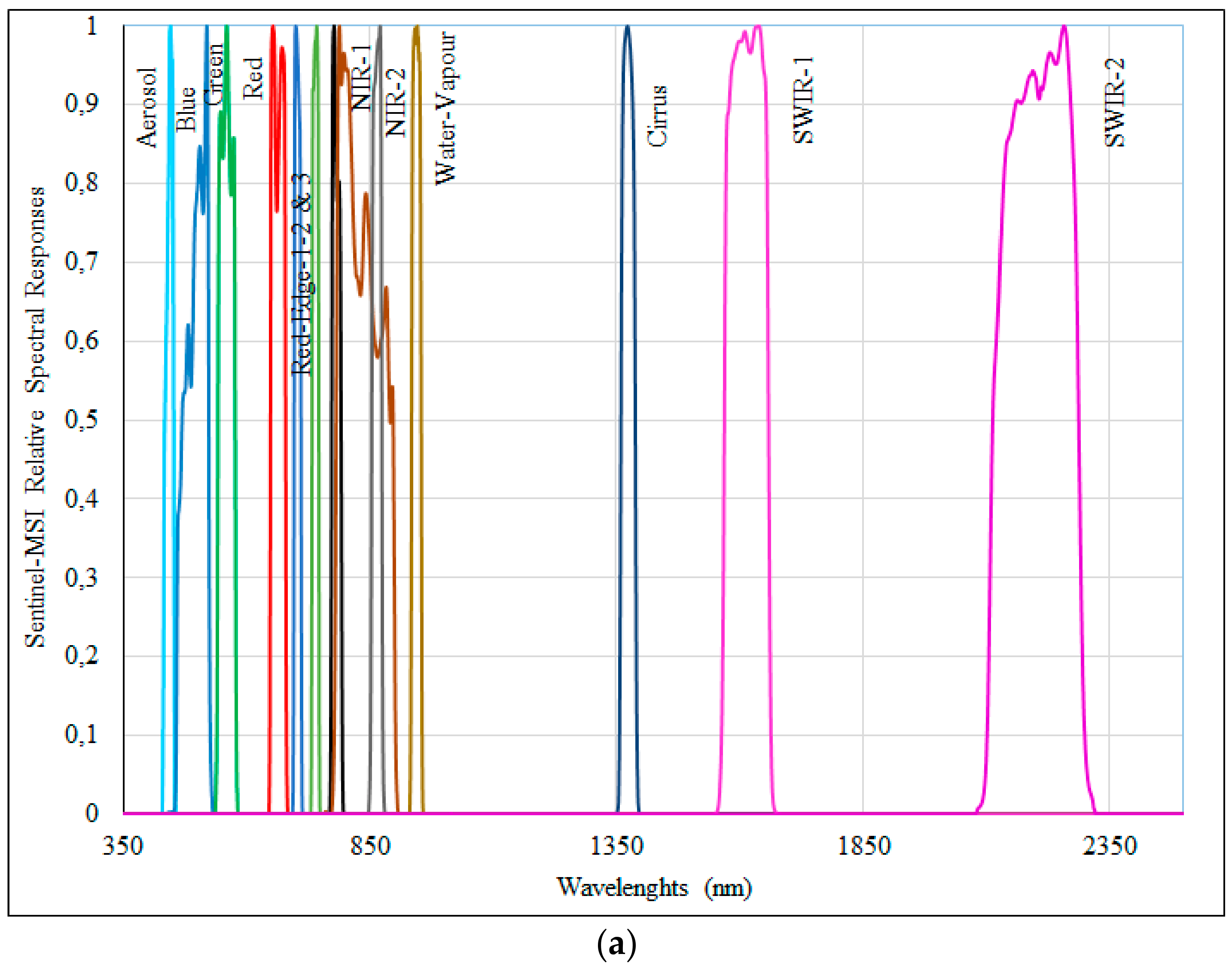

| Spectral Bands | Sentinel-MSI | ||
|---|---|---|---|
| λ Centre (nm) | ∆λ (nm) | Pixel Size (m) | |
| Coastal-Aerosol | 443 | 20 | 60 |
| Blue | 490 | 65 | 10 |
| Green | 560 | 35 | 10 |
| Red | 655 | 30 | 10 |
| Red-Edge-1 | 705 | 15 | 20 |
| Red-Edge-2 | 740 | 15 | 20 |
| Red-Edge-3 | 783 | 20 | 20 |
| NIR-1 | 842 | 115 | 10 |
| NIR-2 | 865 | 20 | 20 |
| Water-vapor * | 945 | 20 | 60 |
| Cirrus * | 1375 | 30 | 60 |
| SWIR-1 | 1609 | 85 | 20 |
| SWIR-2 | 2201 | 187 | 20 |
| Sample | Munsell Color | Standard Color | Texture | Remarks |
|---|---|---|---|---|
| A | 10YR 7/6 | Yellow | Sandy | Sandy soil without gypsum and shells |
| B | 10YR 8/1 | White | Sandy-Clay-Loam | With small amount of gypsum crystals and shells |
| C | 10YR 7/2 | Light-gray | Loamy-Sandy | Sandy soil with small amount of gypsum crystals and shells |
| D | 10YR 7/2 | Light-gray | Sandy-Loam | Beginning of salt crust formation. Small amount of gypsum crystals and shells |
| E | 10YR 7/2 | Light-gray | Sandy-Clay-Loam | Beginning of salt crust formation. Small amount of gypsum crystals and shells |
| F | 10YR 7/2 | Light-gray | Sandy-Clay-Loam | Crust of salt with gypsum, calcium carbonate, and small amount of shells |
| G | 5Y 8/1 | White | Sandy | Pure gypsum crystal deposited by wind erosion |
| H | 10YR 8/1 | White | Pure salt (halite) | Sabkha |
| Sample | pH | EC-Lab | Cl− | HCO3− | SO4−2 | Ca2+ | K+ | Mg2+ | Na+ | SAR (mmoles/L)0.5 |
|---|---|---|---|---|---|---|---|---|---|---|
| (mg/L) | (mg/L) | |||||||||
| A | 8.33 | 26.0 | 9567.5 | 305.1 | 4624.7 | 563.6 | 367.6 | 558.2 | 6599.4 | 46.9 |
| B | 8.10 | 55.6 | 23,209.9 | 305.1 | 6771.7 | 392.5 | 697.2 | 1247.9 | 15,164.8 | 84.5 |
| C | 7.71 | 119.6 | 56,341.7 | 305.1 | 30,495.2 | 1787.8 | 1132.9 | 2327.9 | 44,057.0 | 162.0 |
| D | 7.47 | 195.3 | 120,833.4 | 305.1 | 27,527.6 | 2056.0 | 1840.0 | 4484.8 | 79,607.6 | 225.9 |
| E | 7.57 | 333.0 | 142,094.4 | 305.1 | 6696.6 | 1342.3 | 1236.1 | 2487.4 | 86,990.4 | 325.2 |
| F | 7.35 | 406.5 | 185,325.1 | 305.1 | 68,488.8 | 1580.3 | 3105.0 | 4643.2 | 140,500.0 | 403.6 |
| G | 7.60 | 445.5 | 135,716.1 | 610.2 | 1700.0 | 1128.1 | 843.4 | 1239.7 | 84,795.9 | 415.2 |
| H | 7.60 | 507.0 | 142,803.1 | 610.2 | 17,107.4 | 1227.2 | 1644.9 | 1399.0 | 95,860.0 | 444.7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bannari, A.; El-Battay, A.; Bannari, R.; Rhinane, H. Sentinel-MSI VNIR and SWIR Bands Sensitivity Analysis for Soil Salinity Discrimination in an Arid Landscape. Remote Sens. 2018, 10, 855. https://doi.org/10.3390/rs10060855
Bannari A, El-Battay A, Bannari R, Rhinane H. Sentinel-MSI VNIR and SWIR Bands Sensitivity Analysis for Soil Salinity Discrimination in an Arid Landscape. Remote Sensing. 2018; 10(6):855. https://doi.org/10.3390/rs10060855
Chicago/Turabian StyleBannari, Abderrazak, Ali El-Battay, Rachid Bannari, and Hassan Rhinane. 2018. "Sentinel-MSI VNIR and SWIR Bands Sensitivity Analysis for Soil Salinity Discrimination in an Arid Landscape" Remote Sensing 10, no. 6: 855. https://doi.org/10.3390/rs10060855
APA StyleBannari, A., El-Battay, A., Bannari, R., & Rhinane, H. (2018). Sentinel-MSI VNIR and SWIR Bands Sensitivity Analysis for Soil Salinity Discrimination in an Arid Landscape. Remote Sensing, 10(6), 855. https://doi.org/10.3390/rs10060855