Aerial and Ground Based Sensing of Tolerance to Beet Cyst Nematode in Sugar Beet
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Site
2.2. Experimental Design
2.3. Plant and Nematode Evaluation
2.4. In-Field Measurements
UAV-Based Data Acquisition
2.5. Statistical Data Analysis
3. Results
3.1. Spatial BCN Distribution in the Field
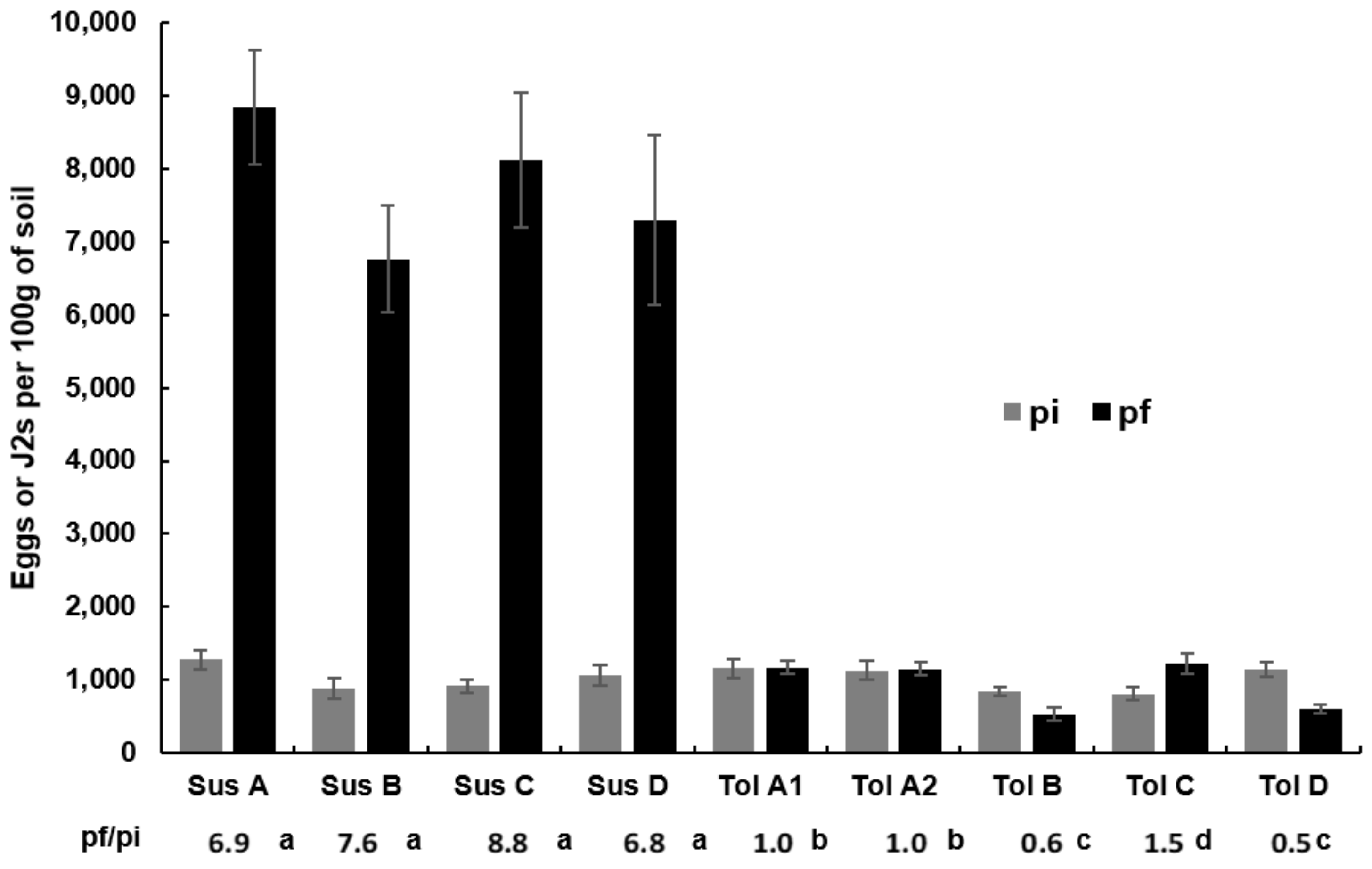
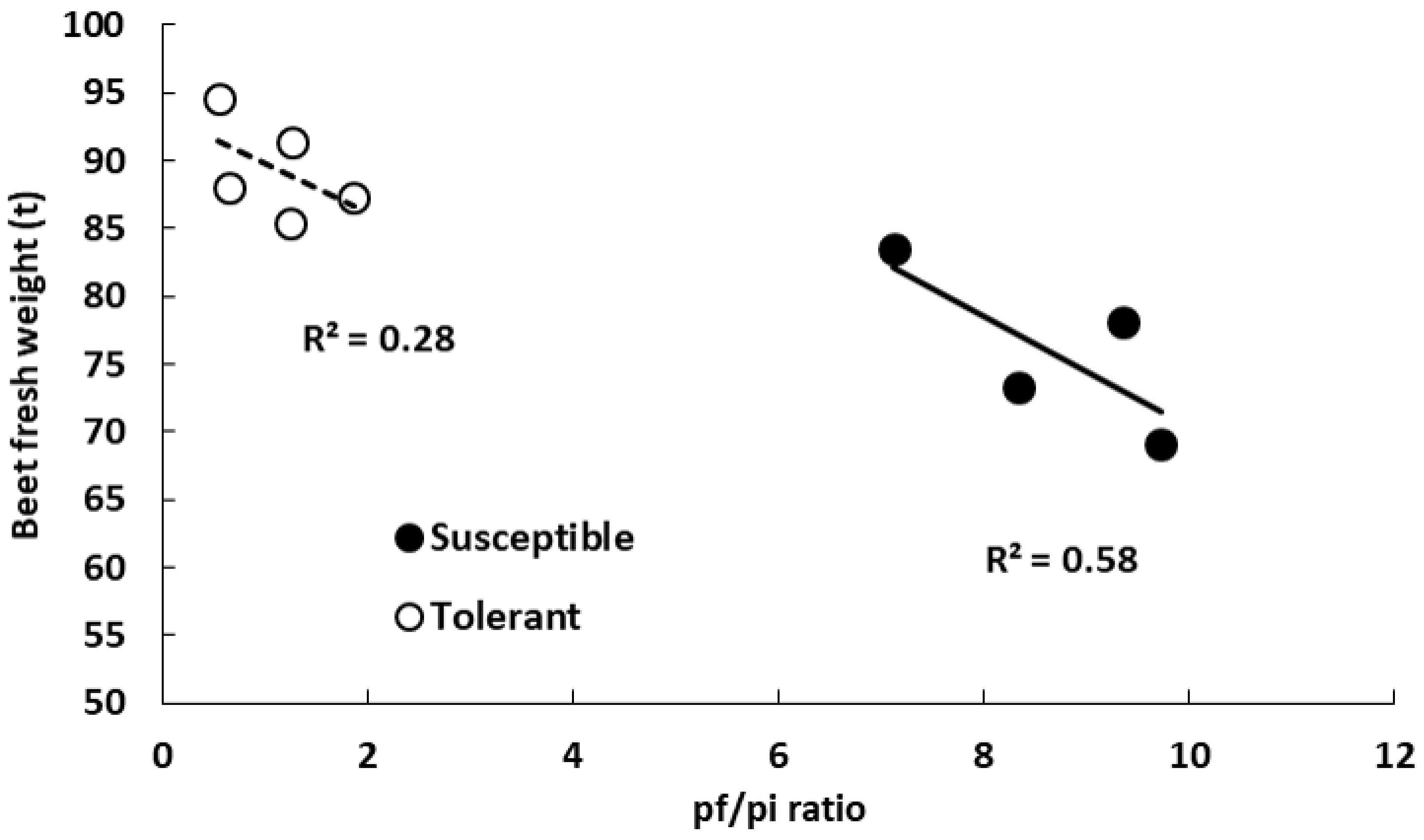
3.2. Beet Fresh Weight and Nematode Population
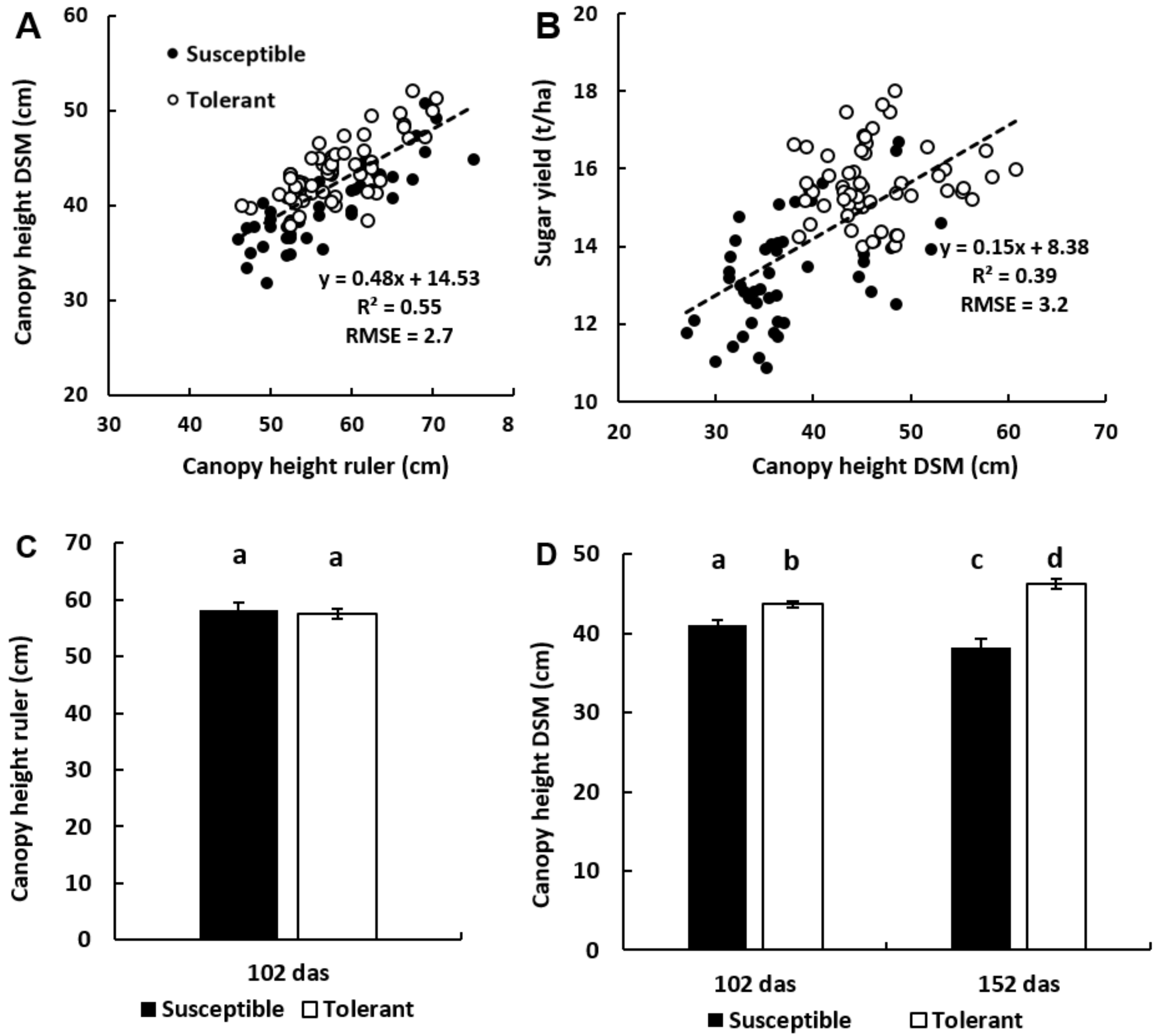
3.3. Canopy Height Measurements
3.4. Thermography
3.5. Spectrometry and UAV Hyperspectral Imaging
3.5.1. Discrimination of Susceptible and Tolerant Cultivars
3.5.2. Correlations with the Yield in Susceptible and Tolerant Cultivars
3.5.3. Field Spectrometer versus UAV Hyperspectral Imager
3.6. Multivariate Analysis
3.7. Decision Trees
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Food and Agriculture Organization (FAO). Global Agriculture towards 2050; Food and Agriculture Organization (FAO): Rome, Itlay, 2009. [Google Scholar]
- Müller, J. The economic importance of Heterodera schachtii in Europe. Helminthologia 1999, 36, 205–213. [Google Scholar]
- Biancardi, E.; McGrath, J.M.; Panella, L.W.; Lewellen, R.T.; Stevanato, P. Sugar beet. In Root and Tuber Crops; Springer: New York, NY, USA, 2010; pp. 173–219. [Google Scholar]
- Steele, A.E.; Arnold, E. The host range of the sugar beet nematode; Heterodera schachtii Schmidt. J. Am. Soc. Sugar Beet Technol. 1965, 13, 573–603. [Google Scholar] [CrossRef]
- Harveson, R.M.; Jackson, T.M. Sugar Beet Cyst Nematode; University of Nebraska–Lincoln Extension: Lincoln, NE, USA, 2008. [Google Scholar]
- Cooke, D.A. Beet cyst nematode (Heterodera schachtii Schmidt) and its control on sugar beet. Agric. Zool. Rev. 1987, 2, 135–183. [Google Scholar]
- Schmitz, A.; Tartachnyk, I.I.; Kiewnick, S.; Sikora, R.A.; Kühbauch, W. Detection of Heterodera schachtii infestation in sugar beet by means of laser-induced and pulse amplitude modulated chlorophyll fluorescence. Nematology 2006, 8, 273–286. [Google Scholar] [CrossRef]
- Heath, W.L.; Haydock, P.P.J.; Wilcox, A.; Evans, K. The potential use of spectral reflectance from the potato crop for remote sensing of infection by potato cyst nematodes. Asp. Appl. Biol. 2000, 60, 185–188. [Google Scholar]
- Nutter, F.W.; Tylka, G.L.; Guan, J.; Moreira, A.J.D.; Marett, C.C.; Rosburg, T.R. Use of remote sensing to detect soybean cyst nematode-induced plant stress. J. Nematol. 2002, 34, 222–231. [Google Scholar] [PubMed]
- Laudien, R. Entwicklung Eines GIS-Gestützten Schlagbezogenen Führungsinformationssystems für die Zuckerwirtschaft. (Development of a Field- and GIS-Based Management Information System for the Sugar Beet Industry). Ph.D. Thesis, University of Hohenheim, Stuttgart, Germany, 2005. [Google Scholar]
- Hillnhütter, C.; Mahlein, A.K.; Sikora, R.A.; Oerke, E.C. Use of imaging spectroscopy to discriminate symptoms caused by Heterodera schachtii and Rhizoctonia solani on sugar beet. Precis. Agric. 2012, 13, 17–32. [Google Scholar] [CrossRef]
- Joalland, S.; Screpanti, C.; Gaume, A.; Walter, A. Belowground biomass accumulation assessed by digital image based leaf area detection. Plant Soil. 2016, 398, 257–266. [Google Scholar] [CrossRef]
- Joalland, S.; Screpanti, C.; Liebisch, F.; Varella, H.V.; Gaume, A.; Walter, A. Comparison of visible imaging; thermography and spectrometry methods to evaluate the effect of Heterodera schachtii inoculation on sugar beets. Plant Methods 2017, 13, 73. [Google Scholar] [CrossRef] [PubMed]
- Sher-Kaul, S.; Oertli, B.; Castella, E.; Lachavanne, J.B. Relationship between biomass and surface area of six submerged aquatic plant species. Aquat. Bot. 1995, 51, 147–154. [Google Scholar] [CrossRef]
- Smith, S.M.; Garrett, P.B.; Leeds, J.A.; McCormick, P.V. Evaluation of digital photography for estimating live and dead aboveground biomass in monospecific macrophyte stands. Aquat. Bot. 2000, 67, 69–77. [Google Scholar] [CrossRef]
- Mizoue, N.; Masutani, T. Image analysis measure of crown condition; foliage biomass and stem growth relationships of Chamaecyparis obtusa. For. Ecol. Manag. 2003, 172, 79–88. [Google Scholar] [CrossRef]
- Tackenberg, O. A new method for non-destructive measurement of biomass; growth rates; vertical biomass distribution and dry matter content based on digital image analysis. Ann. Bot. Lond. 2007, 99, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Baret, F.; Guyot, G. Potentials and limits of vegetation indices for LAI and APAR assessment. Remote Sens. Environ. 1991, 35, 161–173. [Google Scholar] [CrossRef]
- Hillnhütter, C.; Mahlein, A.K.; Sikora, R.A.; Oerke, E.C. Remote sensing to detect plant stress induced by Heterodera schachtii and Rhizoctonia solani in sugar beet fields. Field Crop Res. 2011, 122, 70–77. [Google Scholar] [CrossRef]
- Schmitz, A.; Kiewnick, S.; Schlang, J.; Sikora, R.A. Use of high resolution digital thermography to detect Heterodera schachtii infestation in sugar beets. Commun. Agric. Appl. Biol. Sci. 2004, 69, 359–363. [Google Scholar] [PubMed]
- Colomina, I.; Molina, P. Unmanned aerial systems for photogrammetry and remote sensing: A review. ISPRS J. Photogramm. Remote Sens. 2014, 92, 79–97. [Google Scholar] [CrossRef]
- Araus, J.L.; Cairns, J. Field high-throughput phenotyping—The new crop breeding frontier. Trends Plant Sci. 2014, 19, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Walter, A.; Liebisch, F.; Hund, A. Plant phenotyping: From bean weighing to image analysis. Plant Methods 2015, 11, 14. [Google Scholar] [CrossRef] [PubMed]
- Bendig, J.; Bolten, A.; Bareth, G. Introducing a low-cost mini-UAV for thermal- and multispectral-imaging. In Proceedings of the XXII ISPRS Congress, Melbourne, Australia, 25 August–1 September 2012; pp. 345–349. [Google Scholar]
- Guo, T.; Kujirai, T.; Watanabe, T. Mapping crop status from an unmanned aerial vehicle for precision agriculture applications. In Proceedings of the XXII ISPRS Congress, Melbourne, Australia, 25 August–1 September 2012; pp. 485–490. [Google Scholar]
- Primicerio, J.; Di Gennaro, S.F.; Fiorillo, E.; Genesio, L.; Lugato, E.; Matese, A.; Vaccari, F.P.A. Flexible unmanned aerial vehicle for precision agriculture. Precis. Agric. 2012, 13, 517–523. [Google Scholar] [CrossRef]
- Tattaris, M.; Reynolds, M.P.; Chapman, S.C. A Direct Comparison of Remote Sensing Approaches for High-Throughput Phenotyping in Plant Breeding. Front. Plant Sci. 2016, 7, 1131. [Google Scholar] [CrossRef] [PubMed]
- Akhtman, Y.; Golubeva, E.; Tutubalina, O.; Zimin, M. Application of hyperspectral images and ground data for precision farming. Geogr. Environ. Sustain. 2017, 10, 117–128. [Google Scholar] [CrossRef]
- Constantin, D.; Rehak, M.; Akhtman, Y.; Liebisch, F. Detection of crop properties by means of hyperspectral remote sensing from a micro UAV. In Bornimer Agrartechnische Berichte; Leibniz-Institut für Agrartechnik Potsdam-Bornim eV: Potsdam, Germany, 2015; pp. 129–137. [Google Scholar]
- Khanna, R.; Möller, M.; Pfeifer, J.; Liebisch, F.; Walter, A.; Siegwart, R. Beyond point clouds-3d mapping and field parameter measurements using UAVs. In Proceedings of the IEEE 20th Conference on Emerging Technologies and Factory Automation (ETFA), Luxembourg, 8–11 September 2015; pp. 1–4. [Google Scholar]
- Liebisch, F.; Kirchgessner, N.; Schneider, D.; Walter, A.; Hund, A. Remote, aerial phenotyping of maize traits with a mobile multi-sensor approach. Plant Methods 2015, 11, 9. [Google Scholar] [CrossRef] [PubMed]
- Burkart, A.; Hecht, V.L.; Kraska, T.; Rascher, U. Phenological analysis of unmanned aerial vehicle based time series of barley imagery with high temporal resolution. Precis. Agric. 2018, 19, 134–146. [Google Scholar] [CrossRef]
- Jimenez-Bello, M.A.; Royuela, A.; Manzano, J.; Zarco-Tejada, P.J.; Intrigliolo, D. Assessment of drip irrigation sub-units using airborne thermal imagery acquired with an Unmanned Aerial Vehicle (UAV). In Precision Agriculture 13; Wageningen Academic Publishers: Wageningen, The Netherlands, 2013; pp. 705–711. [Google Scholar]
- Diaz-Varela, R.A.; de la Rosa, R.; Leon, L.; Zarco-Tejada, P.J. High-resolution airborne UAV imagery to assess olive tree crown parameters using 3D photo reconstruction: Application in breeding trials. Remote Sens. 2015, 7, 4213–4232. [Google Scholar] [CrossRef]
- Roth, L.; Streit, B. Predicting cover crop biomass by lightweight UAS-based RGB and NIR photography: An applied photogrammetric approach. Precis. Agric. 2017, 1–22. [Google Scholar] [CrossRef]
- Sankaran, S.; Khot, L.R.; Espinoza, C.Z.; Jarolmasjed, S.; Sathuvalli, V.R.; Vandemark, G.J.; Miklas, P.N.; Carter, A.H.; Pumphrey, M.O.; Knowles, N.R.; et al. Low-altitude; high-resolution aerial imaging systems for row and field crop phenotyping: A review. Eur. J. Agron. 2015, 70, 112–123. [Google Scholar] [CrossRef]
- Yang, G.; Liu, J.; Zhao, C.; Li, Z.; Huang, Y.; Yu, H.; Xu, B.; Yang, X.; Zhu, D.; Zhang, X.; Zhang, R.; et al. Unmanned Aerial Vehicle Remote Sensing for Field-Based Crop Phenotyping: Current Status and Perspectives. Front. Plant Sci. 2017, 8, 1111. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.; Lenz-Wiedemann, V.; Chen, X.; Bareth, G. Estimating leaf chlorophyll of barley at different growth stages using spectral indices to reduce soil background and canopy structure effects. ISPRS J. Photogramm. 2014, 97, 58–77. [Google Scholar] [CrossRef]
- Sa, I.; Chen, Z.; Popovic, M.; Khanna, R.; Liebisch, F.; Nieto, J.; Siegwart, R. weedNet: Dense Semantic Weed Classification Using Multispectral Images and MAV for Smart Farming. IEEE Robot. Autom. Lett. 2018, 3, 588–595. [Google Scholar] [CrossRef]
- Reuther, M.; Lang, C.; Grundler, F.M.W. Nematode-tolerant sugar beet varieties—Resistant or susceptible to the Beet Cyst Nematode Heterodera schachtii? Sugar Ind. 2017, 142, 277–284. [Google Scholar]
- Grosse, E.; Banasiak, L.; Lyr, H.; Jock, M. Neuer Labortest zum Nachweis des Rübennematoden (Heterodera schachtii). Nachr.-Bl. Pflanzenschutz DDR 1985, 39, 111–112. [Google Scholar]
- Grosse, E.; Decker, H. Untersuchungen zur Eignung von Biotest und Schlupftest für den quantitativen Nachweis des Rübenzystenälchen (Heterodera schachtii) in Bodenproben. Nachr.-Bl. Pflanzenschutz DDR 1989, 43, 227–230. [Google Scholar]
- Oerke, E.C.; Steiner, U. Potential of digital thermography for disease control. In Precision Crop Protection—The Challenge and Use of Heterogeneity; Oerke, E.C., Gerhards, R., Menz, G., Sikora, R.A., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 167–182. [Google Scholar]
- Rouse, J.W.; Haas, R.H.; Schell, J.A.; Deering, D.W. Monitoring Vegetation Systems in the Great Plains with ERTS; NASA: Goddard, MD, USA, 1974; Volume 351, p. 309. [Google Scholar]
- Mistele, B.; Gutser, R.; Schmidhalter, U.; Mulla, D.J. Validation of field-scaled spectral measurements of the nitrogen status in winter wheat. In Proceedings of the 7th International Conference on Precision Agriculture and Other Precision Resources Management, Hyatt Regency, Minneapolis, MN, USA, 25–28 July 2004; Precision Agriculture Center, Department of Soil, Water and Climate, University of Minnesota: Minneapolis, MN, USA, 2004; pp. 25–28, 1187–1195. [Google Scholar]
- Haboudane, D.; Miller, J.R.; Tremblay, N.; Zarco-Tejada, P.J.; Dextraze, L. Integrated narrow-band vegetation indices for prediction of crop chlorophyll content for application to precision agriculture. Remote Sens. Environ. 2002, 81, 416–426. [Google Scholar] [CrossRef]
- Hunt, E.R.; Daughtry, C.S.T.; Eitel, J.U.; Long, D.S. Remote sensing leaf chlorophyll content using a visible band index. Agron. J. 2011, 103, 1090–1099. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Keydan, G.P.; Merzlyak, M.N. Three-band model for noninvasive estimation of chlorophyll; carotenoids; and anthocyanin contents in higher plant leaves. Geophys. Res. Lett. 2006, 33. [Google Scholar] [CrossRef]
- Gamon, J.A.; Penuelas, J.; Field, C.B. A narrow-waveband spectral index that tracks diurnal changes in photosynthetic efficiency. Remote Sens. Environ. 1992, 41, 35–44. [Google Scholar] [CrossRef]
- Gao, B.C. NDWI—A normalized difference water index for remote sensing of vegetation liquid water from space. Remote Sens. Environ. 1996, 58, 257–266. [Google Scholar] [CrossRef]
- Clay, D.E.; Kim, K.I.; Chang, J.; Clay, S.A.; Dalsted, K. Characterizing water and nitrogen stress in corn using remote sensing. Agron. J. 2006, 98, 579–587. [Google Scholar] [CrossRef]
- Penuelas, J.; Pinol, J.; Ogaya, R.; Filella, I. Estimation of plant water concentration by the reflectance water index WI (R900/R970). Int. J. Remote Sens. 1997, 18, 2869–2875. [Google Scholar] [CrossRef]
- Mahlein, A.K.; Rumpf, T.; Welke, P.; Dehne, H.W.; Plümer, L.; Steiner, U.; Oerke, E.C. Development of spectral indices for detecting and identifying plant diseases. Remote Sens. Environ. 2013, 128, 21–30. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2008. Available online: http://www.R-project.org (accessed on 1 July 2017).
- Witten, I.H.; Frank, E.; Hall, M.A.; Pal, C.J. Data Mining: Practical Machine Learning Tools and Techniques; Morgan Kaufmann: Burlington, MA, USA, 2016. [Google Scholar]
- Weiss, S.M.; Kulikowski, C.A. Computer Systems that Learn; Kaufmann Publishers: San Mateo, CA, USA, 1991. [Google Scholar]
- Breiman, L.; Friedman, J.; Olshen, R.; Stone, C. Classification and Regression Trees; Wadsworth International Group: Belmont, CA, USA, 1984. [Google Scholar]
- Oostenbrink, M. Major characteristics of the relations between nematodes and plants. In Proceedings of the 8th International Symposium of nematology, Antibes, France, 8–14 September 1966. [Google Scholar]
- Sherrod, P.H. DTREG Predictive Modeling Software. Users Manual. Available online: www.dtreg. com/DTREG.pdf (accessed on 1 February 2018).
- Landis, J.R.; Koch, G.G. The measurement of observer agreement for categorical data. Biometrics 1977, 33, 159–174. [Google Scholar] [CrossRef] [PubMed]
- Seinhorst, J.W. The relation between nematode density and damage to plants. Nematologica 1965, 11, 137–154. [Google Scholar] [CrossRef]
- Cooke, D.A.; Thomason, I.J. The relationship between population density of Heterodera schachtii; soil temperature; and sugarbeet yields. J. Nematol. 1979, 11, 124. [Google Scholar] [PubMed]
- Hauer, M.; Koch, H.J.; Märländer, B. Water use efficiency of sugar beet cultivars (Beta vulgaris L.) susceptible; tolerant or resistant to Heterodera schachtii (Schmidt) in environments with contrasting infestation levels. Field Crops Res. 2015, 183, 356–364. [Google Scholar] [CrossRef]
- Trudgill, D.L. Resistance to and tolerance of plant parasitic nematodes in plants. Annu. Rev. Phytopathol. 1991, 29, 167–192. [Google Scholar] [CrossRef]
- Inoue, Y.; Kimball, B.A.; Jackson, R.D.; Pinter, P.J.; Reginato, R.J. Remote estimation of leaf transpiration rate and stomatal resistance based on infrared thermometry. Agric. For. Meteorol. 1990, 51, 21–33. [Google Scholar] [CrossRef]
- Jones, H.G.; Schofield, P. Thermal and other remote sensing of plant stress. Gen. Appl. Plant Physiol. 2008, 34, 19–32. [Google Scholar]
- Trudgill, D.L. Effects of Globodera rostochiensis and fertilisers on the mineral nutrient content and yield of potato plants. Nematologica 1980, 26, 243–254. [Google Scholar] [CrossRef]
- Haverkort, A.J.; Fasan, T.; Van de Waart, M. The influence of cyst nematodes and drought on potato growth. 2. Effects on plant water relations under semi-controlled conditions. Eur. J. Plant Pathol. 1991, 97, 162–170. [Google Scholar] [CrossRef]
- Jones, H.G. Application of thermal imaging and infrared sensing in plant physiology and ecophysiology. Adv. Bot. Res. 2004, 41, 107–163. [Google Scholar]
- Evans, K.; Franco, J. Tolerance to cyst-nematode attack in commercial potato cultivars and some possible mechanisms for its operation. Nematologica 1979, 25, 153–162. [Google Scholar] [CrossRef]
- Trudgill, D.L. Concepts of resistance; tolerance and susceptibility in relation to cyst nematodes. In Cyst Nematodes; Springer: Boston, MA, USA, 1986; pp. 179–189. [Google Scholar]
- Radcliffe, D.E.; Hussey, R.S.; McClendon, R.W. Cyst nematode vs. tolerant and intolerant soybean cultivars. Agron. J. 1990, 82, 855–860. [Google Scholar] [CrossRef]
- Aasen, H.; Bolten, A. Multi-temporal high-resolution imaging spectroscopy with hyperspectral 2D imagers–From theory to application. Remote Sens. Environ. 2018, 205, 374–389. [Google Scholar] [CrossRef]
- Shakoor, N.; Lee, S.; Mockler, T.C. High throughput phenotyping to accelerate crop breeding and monitoring of diseases in the field. Curr. Opin. Plant Biol. 2017, 38, 184–192. [Google Scholar] [CrossRef] [PubMed]
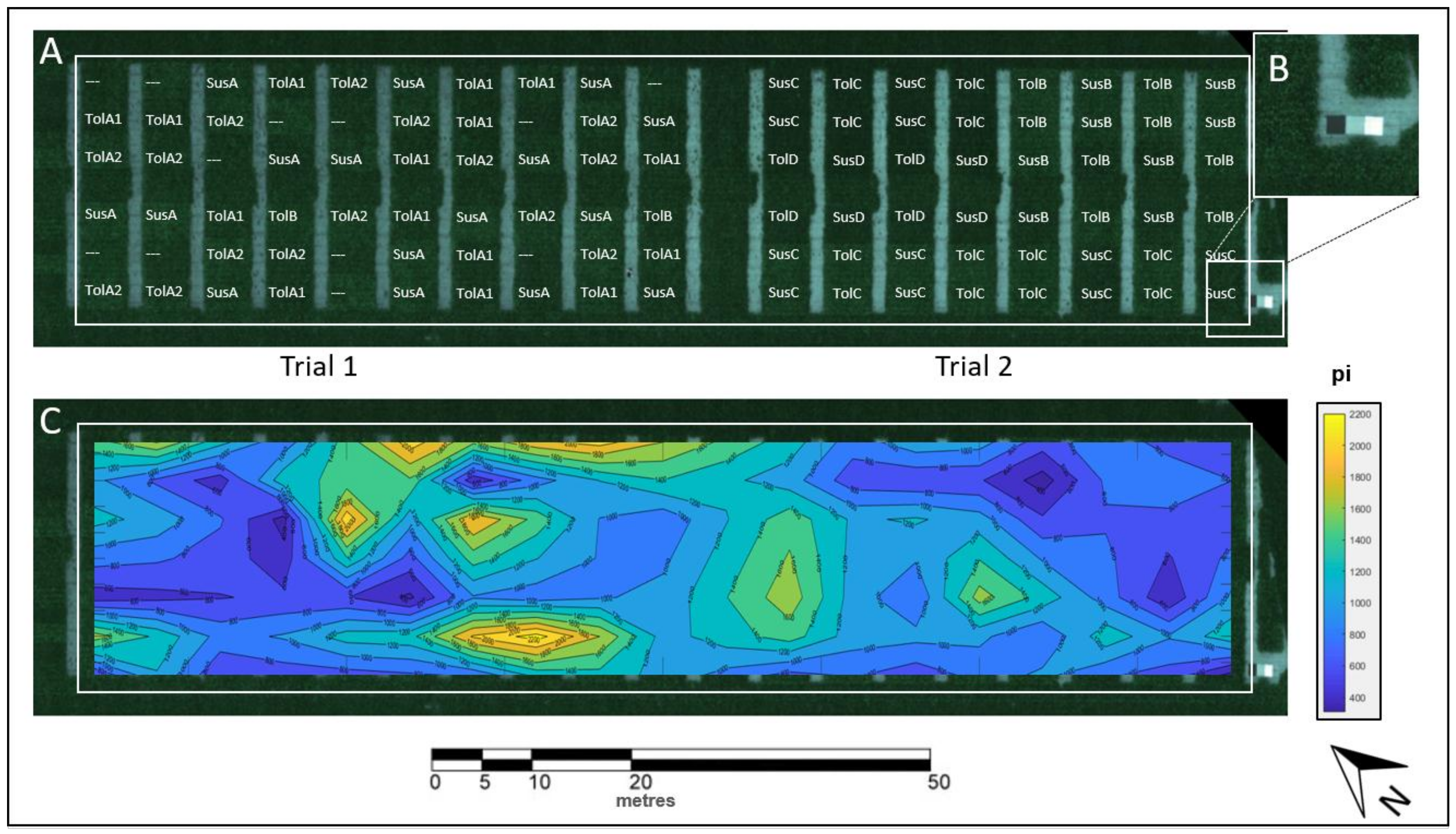
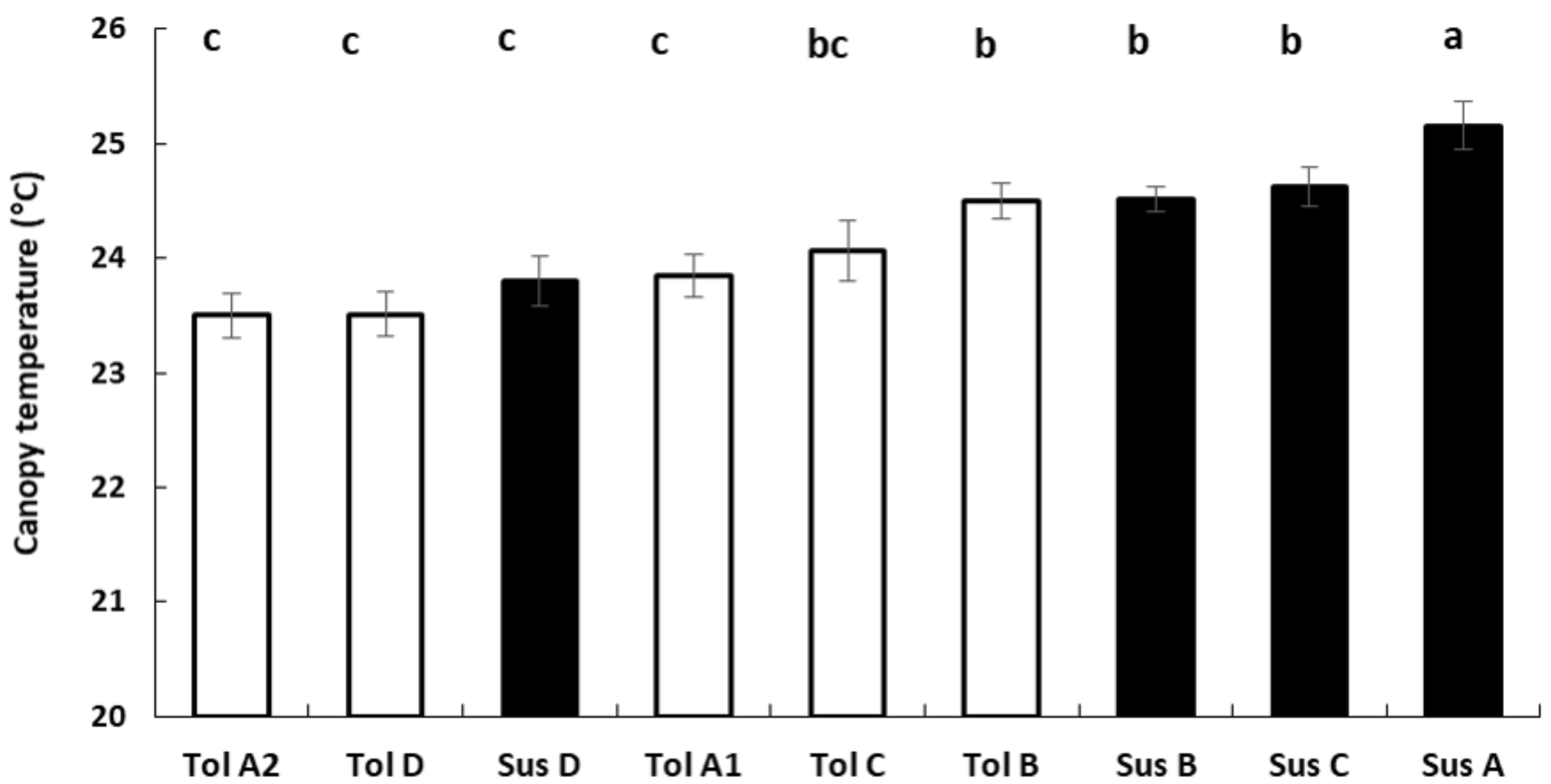
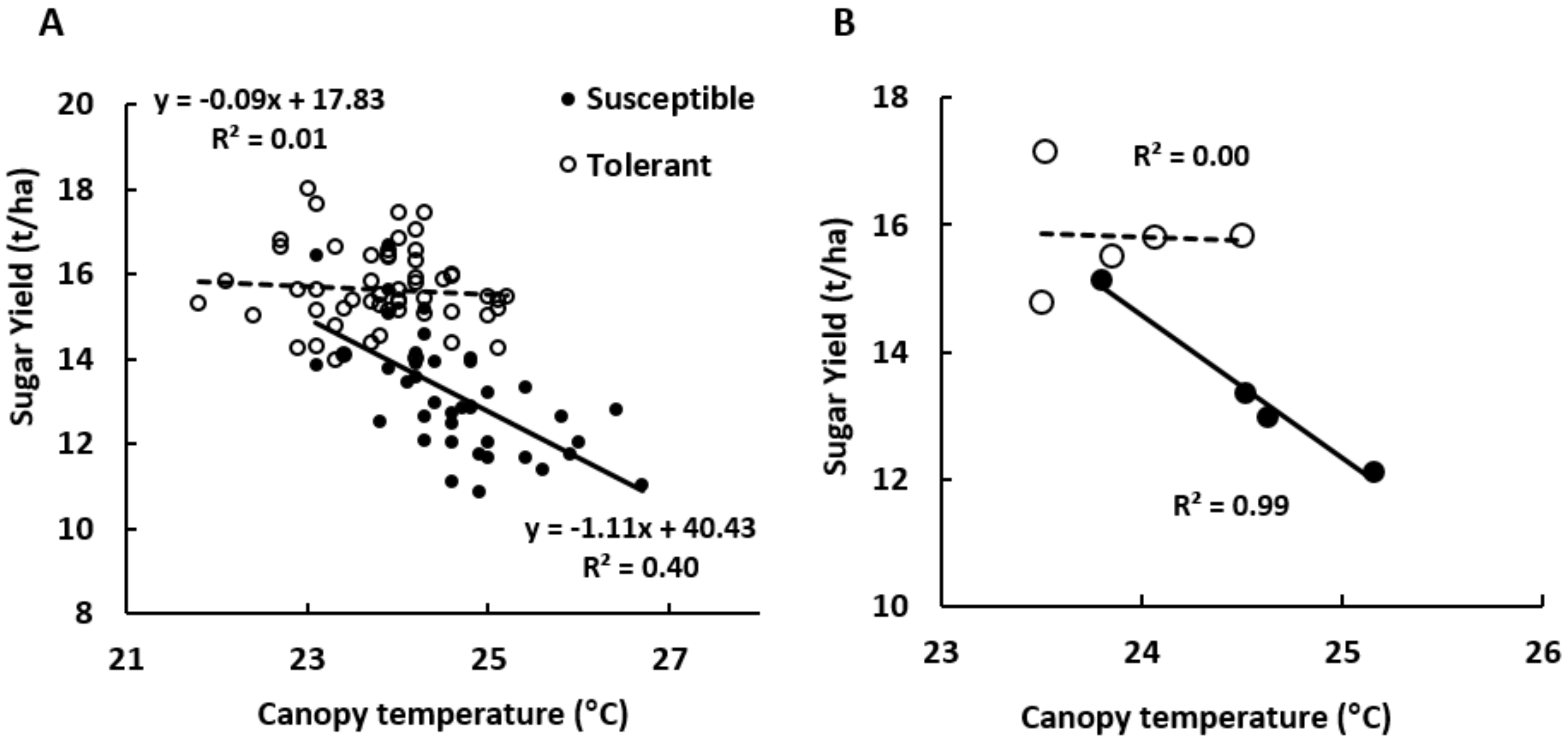
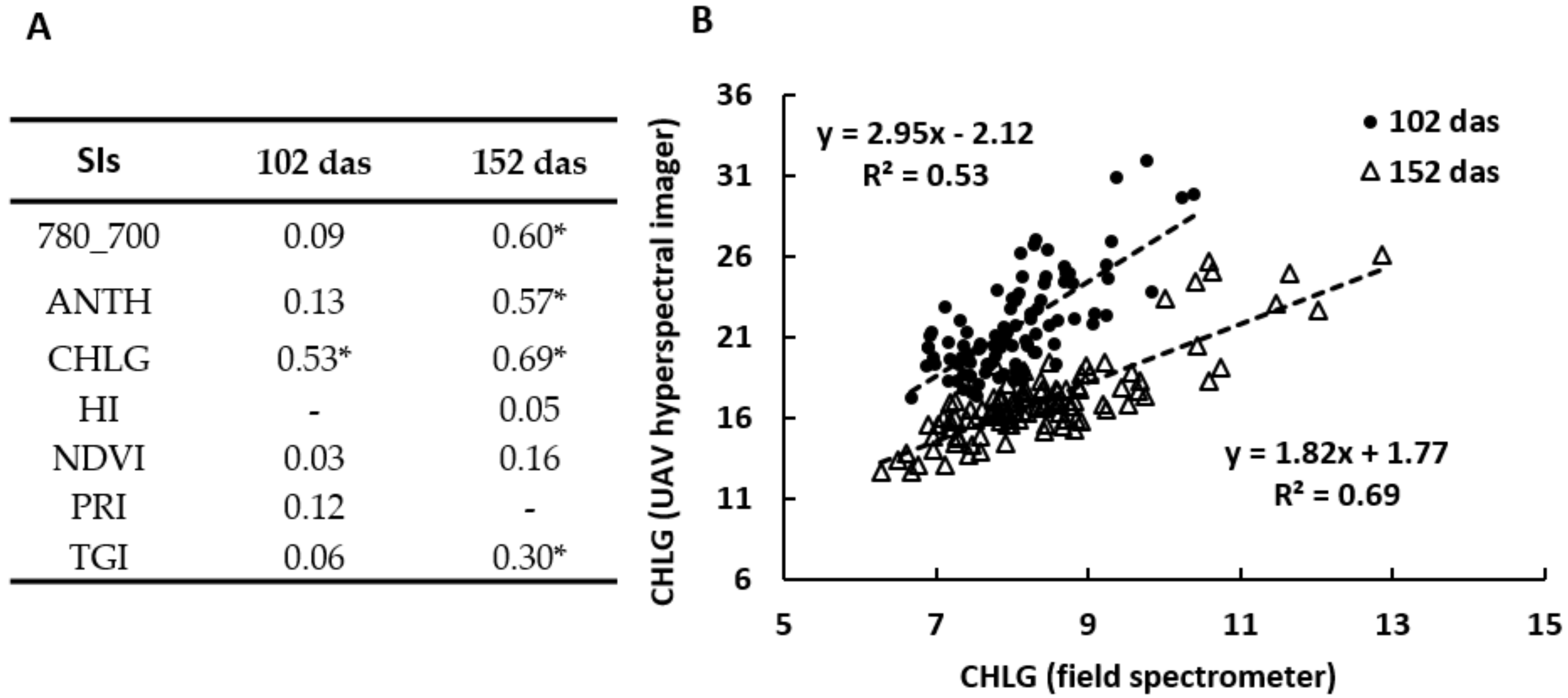
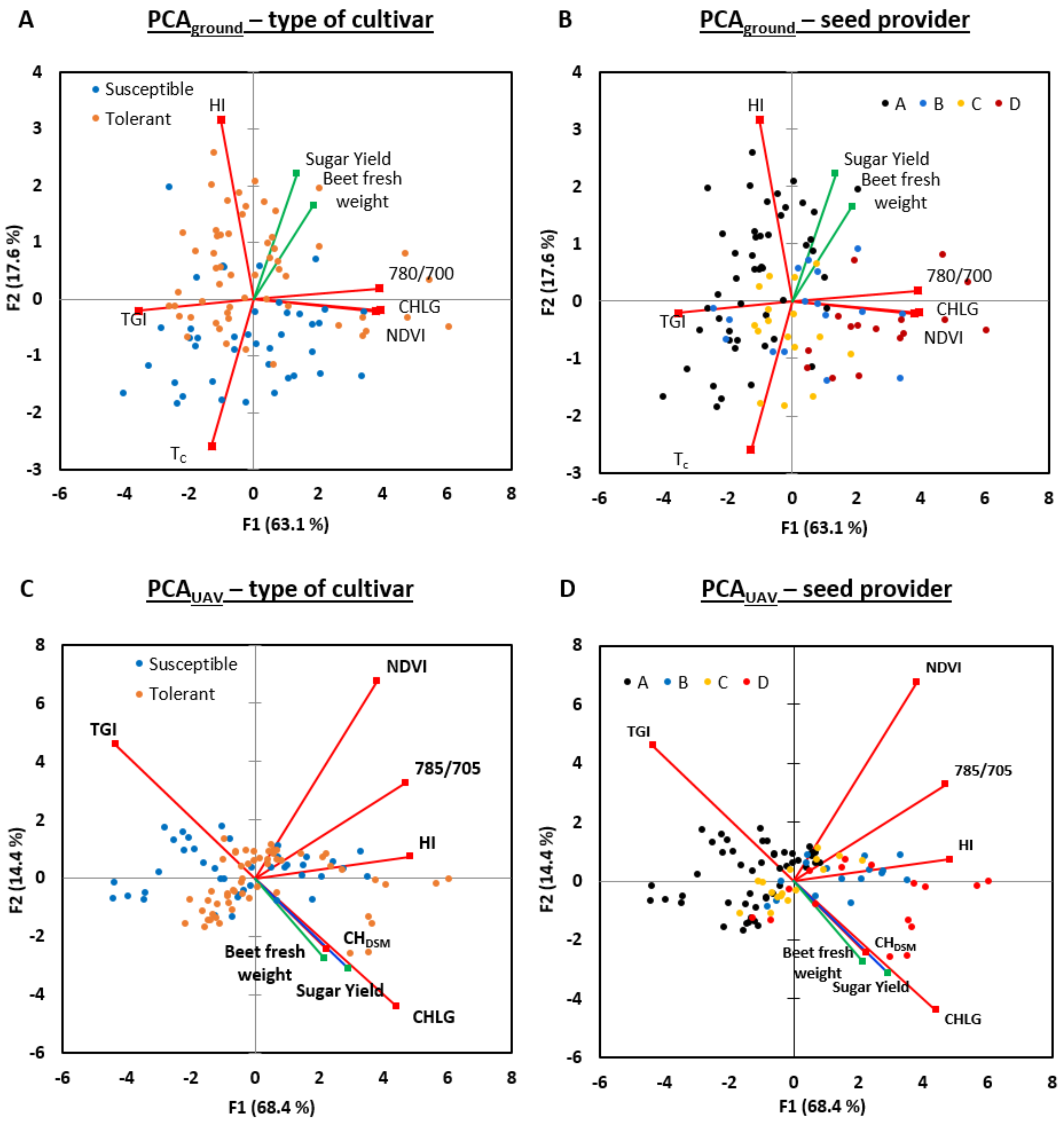
| Sugar beet cultivars | Susceptible | Tolerant | |
| Sus A | Tol A1 | ||
| Tol A2 | |||
| Sus B | Tol B | ||
| Sus C | Tol C | ||
| Sus D | Tol D | ||
| Sowing | 24 March 2016 | ||
| Fertilizer application | Mid-March | ||
| Herbicide applications | 18 April | ||
| 2 May | |||
| 7 May | |||
| 17 May | |||
| Fungicide application | 19 July | ||
| 18 August | |||
| Ground measurements | Spectrometry | 20 June (88 das *) | |
| 4 July (102 das) | |||
| 23 August (152 das) | |||
| Canopy height | 4 July (102 das) | ||
| Thermography | 23 August (152 das) | ||
| UAV Hyperspectral images acquisition | 4 July (102 das) | ||
| 23 August (152 das) | |||
| Harvest and sampling | 6 October | ||
| SIs | Equation | Traits | Reference |
|---|---|---|---|
| NDVI | (R800 − R680)/(R800 + R680) | Biomass, coverage | [44] |
| 780/740 | R780/R740 | Nitrogen content | [45] |
| 780/700 | R780/R700 | Nitrogen content | [45] |
| TCARI | 3 × [(R700 − R670) − 0.2 × (R700 − R550) × (R700/R670)] | Chlorophyll content | [46] |
| TGI | −0.5×[(W670 − W480)×(R670 − R550) − (W670 − W550)×(R670 − R480)] | Chlorophyll content | [47] |
| ANTH | R760 − R800 × (1/R540 − R560 − 1/R690 − R710) | Anthocyanins | [48] |
| CHLG | (R760 − R800)/(R540 − R560) | Chlorophyll content | [48] |
| PRI | (R531 − R570)/(R531 + R570) | Stress | [49] |
| NDWI | (R860 − R1240)/(R860 + R1240) | Plant water status | [50] |
| NDWI1650 | (R840 − R1650)/(R840 + R1650) | Plant water status | [51] |
| WI | (R900/R970) | Plant water status | [52] |
| HI | (R534 − R698)/(R534 + R698) − R704/2 | Plant health | [53] |
| Cultivar Type | Genotype | Beet Fresh Weight (t) | White Sugar Yield (t) | Initial BCN Population (Number of J2s per 100 g soil) |
|---|---|---|---|---|
| Susceptible | Susceptible A | 73.27 ± 1.39 a | 12.13 ± 0.23 a | 1283 ± 131 a |
| Susceptible B | 78.08 ± 2.30 a | 13.63 ± 0.44 b | 886 ± 144 a | |
| Susceptible C | 69.10 ± 1.18 c | 13.00 ± 0.22 b | 919 ± 94 a | |
| Susceptible D | 83.56 ± 2.05 d | 15.14 ± 0.38 cd | 1073 ± 140 a | |
| Average | 76.00 ± 1.09 | 13.48 ± 0.22 | 1031 | |
| Tolerant | Tolerant A1 | 91.41 ± 1.01 bf | 15.52 ± 0.17 c | 1160 ± 126 a |
| Tolerant A2 | 85.48 ± 0.97 d | 14.80 ± 0.14 d | 1134 ± 128 a | |
| Tolerant B | 87.99 ±1.53 ef | 15.60 ± 0.30 c | 1512 ± 62 a | |
| Tolerant C | 87.30 ± 0.80 de | 15.81 ± 0.22 c | 814 ± 90 a | |
| Tolerant D | 94.66 ± 1.13 b | 17.17 ± 0.19 e | 1154 ± 103 a | |
| Average | 89.37 ± 0.63 | 15.78 ± 0.13 | 1026 |
| (A) | Experiment 1 | Experiment 2 | ||
| Sus A/Tol A1–Tol A2 | Sus B/Tol B | Sus C/Tol C | Sus D/Tol D | |
| 88 das | ||||
| 102 das | 780/700 HI CHLG PRI NDVI | 780/700 CHLG HI PRI TGI | ANTH HI | NDWI1650 NDWI HI |
| 152 das | 780/700 HI TGI PRI CHLG TCARI NDWI1650 | CHLG NDWI NDWI1650 PRI TCARI TGI | ANTH HI NDWI1650 | 780/700 ANTH CHLG NDVI NDWI NDWI1650 |
| (B) | Experiment 1 | Experiment 2 | ||
| Sus A/Tol A1–Tol A2 | Sus B/Tol B | Sus C/Tol C | Sus D/Tol D | |
| 102 das | CHLG HI ANTH PRI | CHLG ANTH PRI | CHLG HI ANTH PRI | |
| 152 das | CHLG ANTH TGI HI 785/705 NDVI | CHLG ANTH HI | CHLG ANTH TGI HI 785/705 | |
| Spectral Vegetation Index | 88 Das | 102 Das | 152 Das | |||
|---|---|---|---|---|---|---|
| Susceptible | Tolerant | Susceptible | Tolerant | Susceptible | Tolerant | |
| (A) | ||||||
| 780/740 | 0.47 * | 0.12 | 0.73 * | 0.40 * | 0.70 * | 0.62 * |
| 780/700 | 0.62 * | 0.42 * | 0.67 * | 0.39 * | 0.63 * | 0.62 * |
| CHLG | 0.46 * | 0.38 * | 0.66* | 0.37 * | 0.64 * | 0.62 * |
| HI | 0.22 | 0.61 * | 0.33 | 0.32 | 0.20 | |
| NDVI | 0.57 * | 0.42 * | 0.59 * | 0.49 * | 0.61 * | 0.49 * |
| NDWI1650 | 0.71 * | 0.27 | 0.56 * | 0.52 * | ||
| WI | 0.71 * | 0.28 | 0.57 * | 0.56 * | ||
| (B) | ||||||
| 785/555 | 0.62 * | 0.34 * | 0.21 | 0.21 | ||
| ANTH | 0.64 * | 0.34 * | 0.20 | 0.21 | ||
| CHLG | 0.61 * | 0.34 * | 0.20 | 0.21 | ||
| HI | 0.36 * | 0.23 | 0.13 | |||
| Trait Detection Level | Das | Classification Accuracy “Type of Cultivars” (Kappa) | Classification Accuracy “Genetic Background” (Kappa) |
|---|---|---|---|
| Field parameters (123 SIs + Tc + CHruler) | 102 | 0.74 (0.46) | 0.76 (0.61) |
| 152 | 0.78 (0.55) | 0.76 (0.63) | |
| UAV based imager (77 SIs + CHDSM) | 102 | 0.79 (0.57) | 0.72 (0.56) |
| 152 | 0.88 (0.74) | 0.68 (0.53) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joalland, S.; Screpanti, C.; Varella, H.V.; Reuther, M.; Schwind, M.; Lang, C.; Walter, A.; Liebisch, F. Aerial and Ground Based Sensing of Tolerance to Beet Cyst Nematode in Sugar Beet. Remote Sens. 2018, 10, 787. https://doi.org/10.3390/rs10050787
Joalland S, Screpanti C, Varella HV, Reuther M, Schwind M, Lang C, Walter A, Liebisch F. Aerial and Ground Based Sensing of Tolerance to Beet Cyst Nematode in Sugar Beet. Remote Sensing. 2018; 10(5):787. https://doi.org/10.3390/rs10050787
Chicago/Turabian StyleJoalland, Samuel, Claudio Screpanti, Hubert Vincent Varella, Marie Reuther, Mareike Schwind, Christian Lang, Achim Walter, and Frank Liebisch. 2018. "Aerial and Ground Based Sensing of Tolerance to Beet Cyst Nematode in Sugar Beet" Remote Sensing 10, no. 5: 787. https://doi.org/10.3390/rs10050787
APA StyleJoalland, S., Screpanti, C., Varella, H. V., Reuther, M., Schwind, M., Lang, C., Walter, A., & Liebisch, F. (2018). Aerial and Ground Based Sensing of Tolerance to Beet Cyst Nematode in Sugar Beet. Remote Sensing, 10(5), 787. https://doi.org/10.3390/rs10050787







