Carbon Dynamics of Reclaimed Coal Mine Soil under Agricultural Use: A Chronosequence Study in the Dongtan Mining Area, Shandong Province, China
Abstract
:1. Introduction
2. Materials and Methods
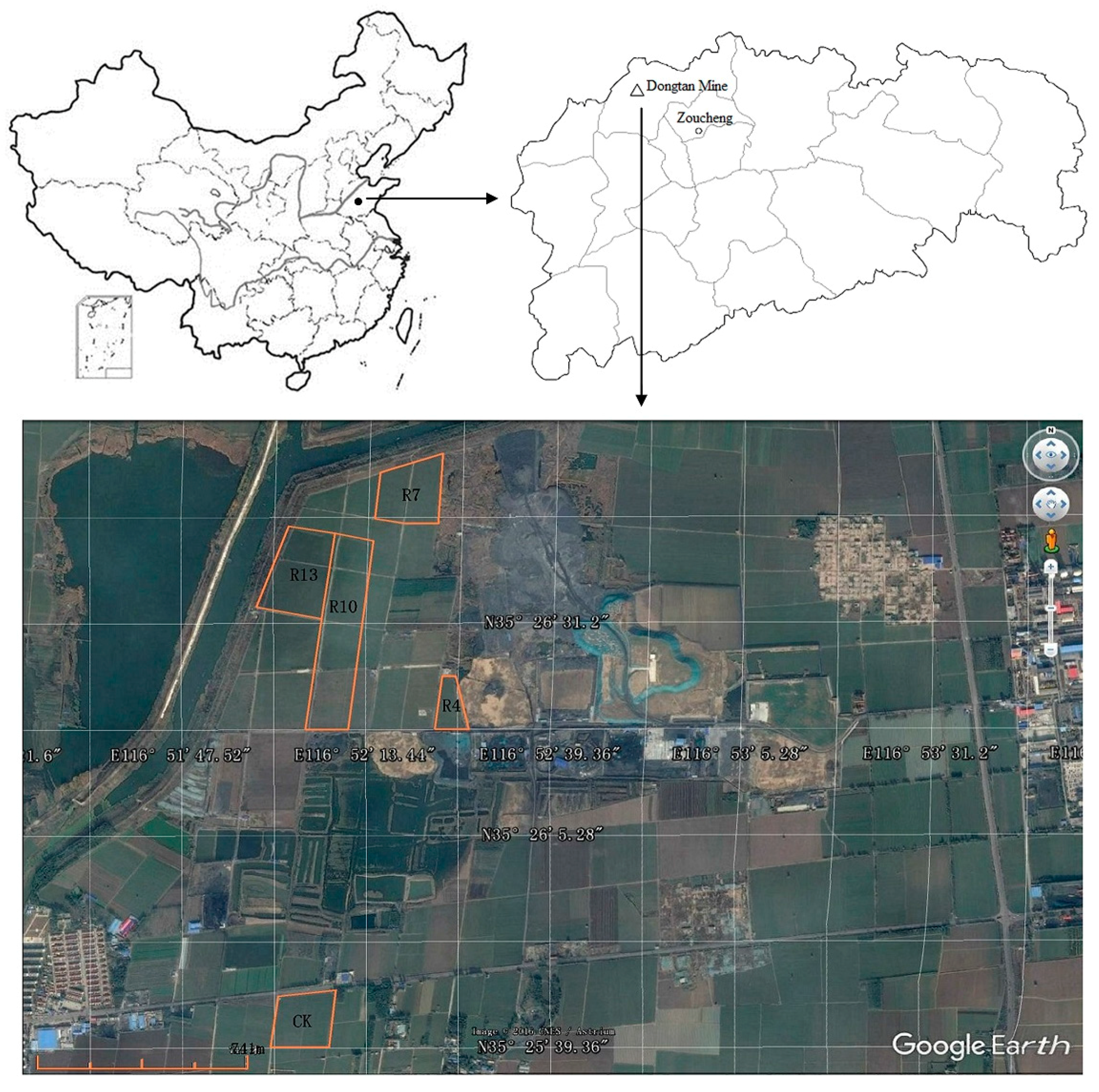
2.1. Study Site
2.2. Collection of Mine Soil Samples
2.3. Soil Analyses
2.4. Statistical Analysis
3. Results
3.1. Basic Physicochemical Properties
3.2. TC, SIC, and SOC
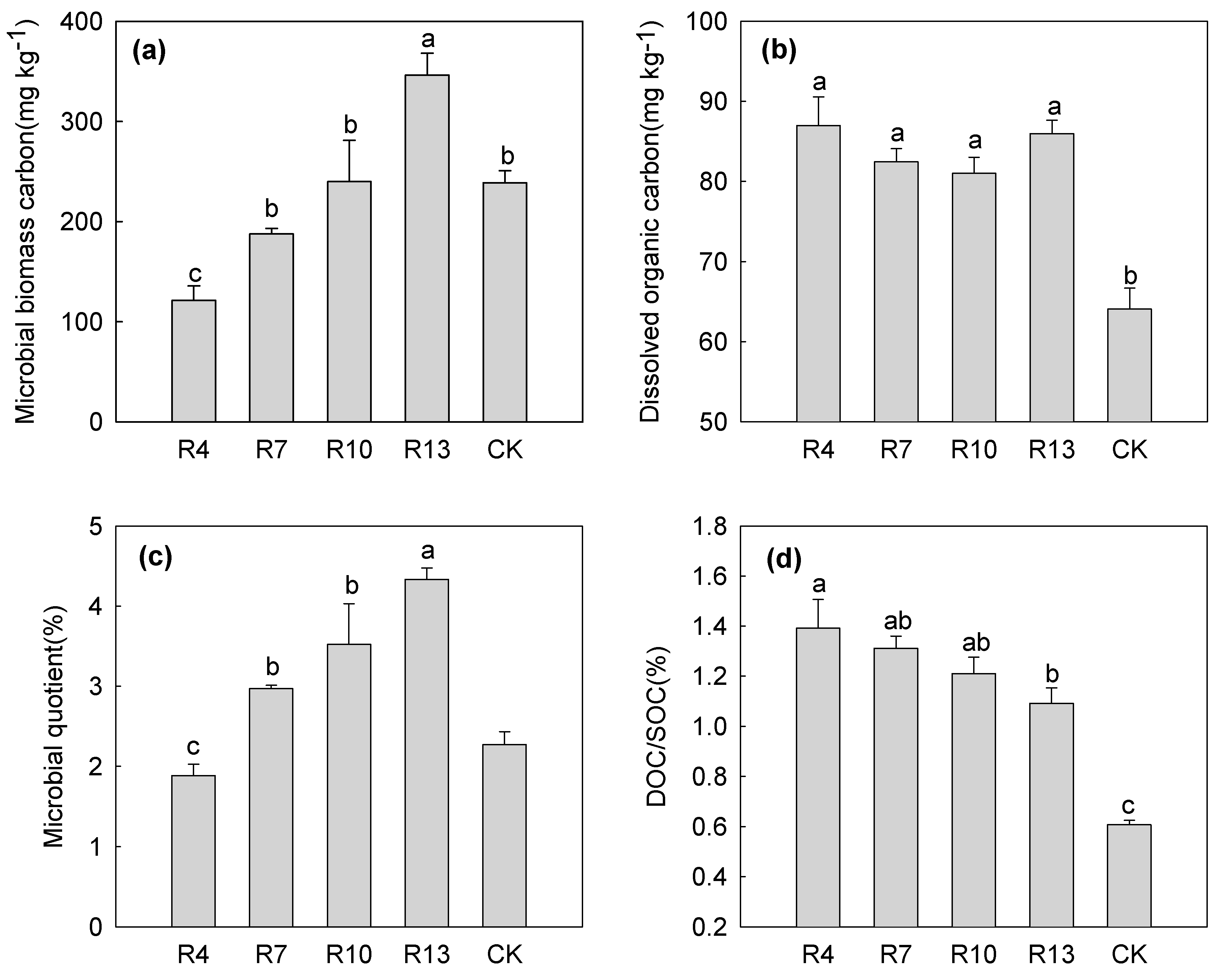
3.3. MBC and DOC
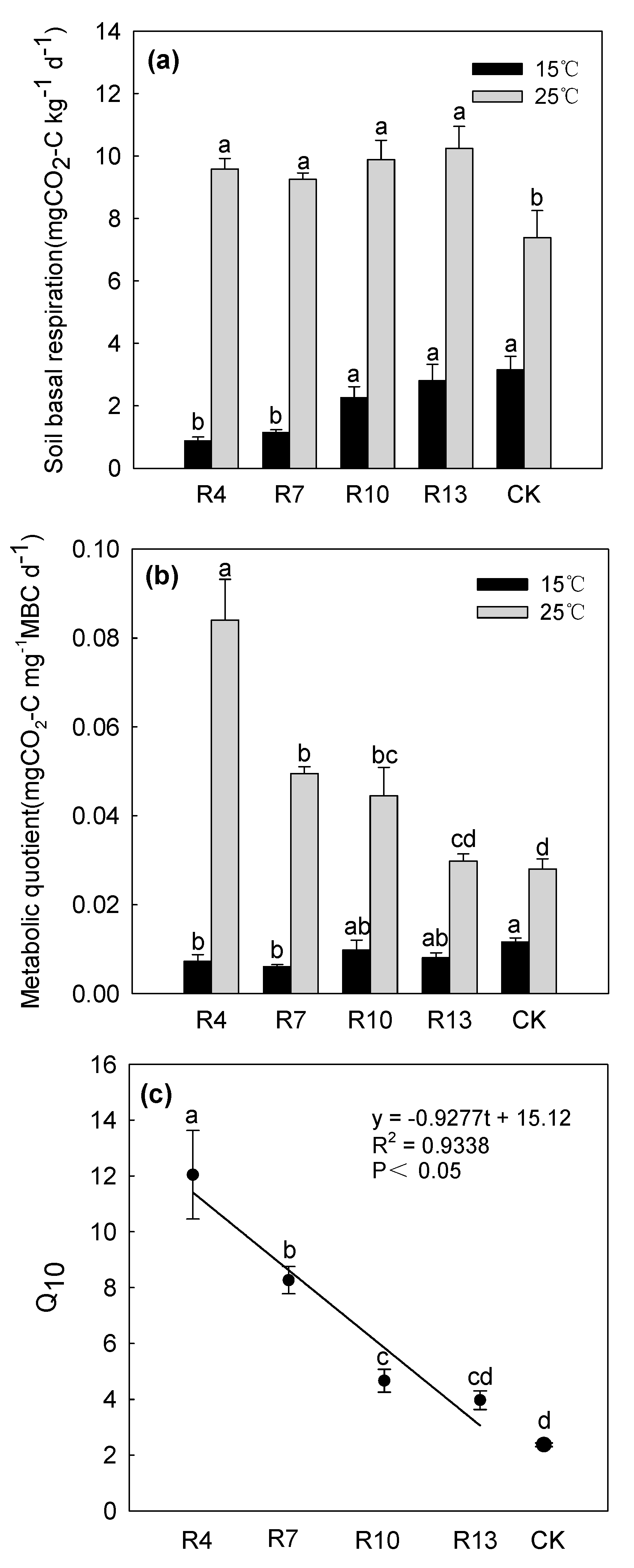
3.4. SBR and qCO2
3.5. Correlations between the Soil Carbon Content, the SBR Rate, and Physicochemical Properties
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brevik, E.C.; Cerdà, A.; Mataix-Solera, J.; Pereg, L.; Quinton, J.N.; Six, J.; van Oost, K. The interdisciplinary nature of SOIL. Soil 2015, 1, 117–129. [Google Scholar] [CrossRef]
- Mol, G.; Keesstra, S. Soil science in a changing world. Curr. Opin. Environ. Sustain. 2012, 4, 473–477. [Google Scholar] [CrossRef]
- Keesstra, S.D.; Geissen, V.; Mosse, K.; Piiranen, S.; Scudiero, E.; Leistra, M.; van Schaik, L. Soil as a filter for groundwater quality. Curr. Opin. Environ. Sustain. 2012, 4, 507–516. [Google Scholar] [CrossRef]
- Keesstra, S.D.; Bouma, J.; Wallinga, J.; Tittonell, P.; Smith, P.; Cerdà, A.; Montanarella, L.; Quinton, J.N.; Pachepsky, Y.; van der Putten, W.H.; et al. The significance of soils and soil science towards realization of the United Nations Sustainable Development Goals. Soil 2016, 2, 111–128. [Google Scholar] [CrossRef]
- Lechner, A.M.; Baumgartl, T.; Matthew, P.; Glenn, V. The Impact of Underground Longwall Mining on Prime Agricultural Land: A Review and Research Agenda. Land Degrad. Dev. 2016, 27, 1650–1663. [Google Scholar] [CrossRef]
- Enerdata Global Energy Statistical Yearbook. Available online: https://yearbook.enerdata.net/#coal-and-lignite-production.html (accessed on 15 August 2016).
- Hu, Z.; Fu, Y.; Xiao, W.; Zhao, Y.; Wei, T. Ecological restoration plan for abandoned underground coal mine site in Eastern China. Int. J. Min. Reclam. Environ. 2015, 29, 316–330. [Google Scholar] [CrossRef]
- Wang, P.; Hu, Z.; Yost, R.S.; Shao, F.; Liu, J.; Li, X. Assessment of chemical properties of reclaimed subsidence land by the integrated technology using Yellow River sediment in Jining, China. Environ. Earth Sci. 2016, 75, 1–15. [Google Scholar] [CrossRef]
- Hu, Z.; Xiao, W. Optimization of concurrent mining and reclamation plans for single coal seam: A case study in northern Anhui, China. Environ. Earth Sci. 2012, 68, 1247–1254. [Google Scholar] [CrossRef]
- Xiao, W.; Hu, Z.; Chugh, Y.P.; Zhao, Y. Dynamic subsidence simulation and topsoil removal strategy in high groundwater table and underground coal mining area: A case study in Shandong Province. Int. J. Min. Reclam. Environ. 2013, 28, 250–263. [Google Scholar] [CrossRef]
- Chodak, M.; Pietrzykowski, M.; Niklińska, M. Development of microbial properties in a chronosequence of sandy mine soils. Appl. Soil Ecol. 2009, 41, 259–268. [Google Scholar] [CrossRef]
- Maiti, S.K. Ecorestoration of the Coalmine Degraded Lands; Springer: New York, NY, USA, 2013. [Google Scholar]
- Ahirwal, J.; Maiti, S.K. Assessment of soil properties of different land uses generated due to surface coal mining activities in tropical Sal (Shorea robusta) forest, India. Catena 2016, 140, 155–163. [Google Scholar] [CrossRef]
- Moreno-De Las Heras, M. Development of soil physical structure and biological functionality in mining spoils affected by soil erosion in a Mediterranean-Continental environment. Geoderma 2009, 149, 249–256. [Google Scholar] [CrossRef]
- Bodlák, L.; Křováková, K.; Kobesová, M.; Brom, J.; Šťastný, J.; Pecharová, E. SOC content—An appropriate tool for evaluating the soil quality in a reclaimed post-mining landscape. Ecol. Eng. 2012, 43, 53–59. [Google Scholar] [CrossRef]
- Pietrzykowski, M.; Daniels, W.L. Estimation of carbon sequestration by pine (Pinus sylvestris L.) ecosystems developed on reforested post-mining sites in Poland on differing mine soil substrates. Ecol. Eng. 2014, 73, 209–218. [Google Scholar] [CrossRef]
- Akala, V.A.; Lal, R. Soil organic carbon pools and sequestration rates in reclaimed minesoils in Ohio. J. Environ. Qual. 2001, 30, 2098–2104. [Google Scholar] [CrossRef] [PubMed]
- Ussiri, D.A.N.; Lal, R. Carbon sequestration in reclaimed minesoils. Crit. Rev. Plant Sci. 2005, 24, 151–165. [Google Scholar] [CrossRef]
- Akala, V.A.; Lal, R. Potential of mine land reclamation for soil organic carbon sequestration in Ohio. Land Degrad. Dev. 2000, 11, 289–297. [Google Scholar] [CrossRef]
- Shrestha, R.K.; Lal, R. Soil carbon and nitrogen in 28-year-old land uses in reclaimed coal mine soils of Ohio. J. Environ. Qual. 2007, 36, 1775–1783. [Google Scholar] [CrossRef] [PubMed]
- Helingerová, M.; Frouz, J.; Šantrůčková, H. Microbial activity in reclaimed and unreclaimed post-mining sites near Sokolov (Czech Republic). Ecol. Eng. 2010, 36, 768–776. [Google Scholar] [CrossRef]
- Šourková, M.; Frouz, J.; Fettweis, U.; Bens, O.; Hüttl, R.F.; Šantrůčková, H. Soil development and properties of microbial biomass succession in reclaimed post mining sites near Sokolov (Czech Republic) and near Cottbus (Germany). Geoderma 2005, 129, 73–80. [Google Scholar] [CrossRef]
- Brantley, S.L. Understanding soil time. Science 2008, 321, 1454–1455. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, S.; McDonald, L.M.; Pena-Yewtukhiw, E.M.; Skousen, J.; Roy, M. Chemically stabilized soil organic carbon fractions in a reclaimed minesoil chronosequence: Implications for soil carbon sequestration. Environ. Earth Sci. 2013, 70, 1689–1698. [Google Scholar] [CrossRef]
- Chaudhuri, S.; Pena-Yewtukhiw, E.M.; McDonald, L.M.; Skousen, J.; Sperow, M. Early C sequestration rate changes for reclaimed minesoils. Soil Sci. 2012, 177, 443–450. [Google Scholar] [CrossRef]
- Adeli, A.; McLaughlin, M.R.; Brooks, J.P.; Read, J.J.; Willers, J.L.; Lang, D.J.; McGrew, R. Age chronosequence effects on restoration quality of reclaimed coal mine soils in Mississippi agroecosystems. Soil Sci. 2013, 178, 335–343. [Google Scholar] [CrossRef]
- Gasch, C.K.; Huzurbazar, S.V.; Wick, A.F.; Stahl, P.D. Assessing impacts of crested wheatgrass and native species establishment on soil characteristics in reclaimed land using Bayesian posterior predictive distributions. Land Degrad. Dev. 2016, 27, 521–531. [Google Scholar] [CrossRef]
- Kumar, S.; Maiti, S.K.; Chaudhuri, S. Soil development in 2–21 years old coalmine reclaimed spoil with trees: A case study from Sonepur-Bazari opencast project, Raniganj Coalfield, India. Ecol. Eng. 2015, 84, 311–324. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.; Maiti, S.K.; Masto, R.E. Development of mine soil quality index (MSQI) for evaluation of reclamation success: A chronosequence study. Ecol. Eng. 2014, 71, 10–20. [Google Scholar] [CrossRef]
- Dangi, S.R.; Stahl, P.D.; Wick, A.F.; Ingram, L.J.; Buyer, J.S. Soil microbial community recovery in reclaimed soils on a surface coal mine site. Soil Sci. Soc. Am. J. 2012, 76, 915–924. [Google Scholar] [CrossRef]
- Yeomans, J.; Bremmer, J.M. A rapid and precise method four routine determination of organic carbon in soil. Commun. Soil Sci. Plant 1989, 19, 1467–1476. [Google Scholar] [CrossRef]
- Olsen, S.R.; Sommers, L.E. Phosphorus. In Methods of Soil Analysis; Miller, A.L., Keeney, D.R., Eds.; American Society of Agronomy: Madison, WI, USA, 1982; pp. 403–427. [Google Scholar]
- Vance, E.D.; Brookes, P.C.; Jenkinson, D.S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Hochachka, P.W.; Somero, G.N. Biochemical Adaptations; Princeton University Press: Princeton, NJ, USA, 1984. [Google Scholar]
- Zhao, Z.; Shahrour, I.; Bai, Z.; Fan, W.; Feng, L.; Li, H. Soils development in opencast coal mine spoils reclaimed for 1–13 years in the West-Northern Loess Plateau of China. Eur. J. Soil Biol. 2013, 55, 40–46. [Google Scholar] [CrossRef]
- Shrestha, R.K.; Lal, R. Ecosystem carbon budgeting and soil carbon sequestration in reclaimed mine soil. Environ. Int. 2006, 32, 781–796. [Google Scholar] [CrossRef] [PubMed]
- Jacinthe, P.-A.; Lal, R. Tillage effects on carbon sequestration and microbial biomass in reclaimed farmland soils of southwestern Indiana. Soil Sci. Soc. Am. J. 2009, 73, 605–613. [Google Scholar] [CrossRef]
- Wick, A.F.; Ingram, L.J.; Stahl, P.D. Aggregate and organic matter dynamics in reclaimed soils as indicated by stable carbon isotopes. Soil Biol. Biochem. 2009, 41, 201–209. [Google Scholar] [CrossRef]
- Insam, H.; Domsch, K.H. Relationship between soil organic carbon and microbial biomass on chronosequences of reclamation sites. Microb. Ecol. 1988, 15, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, R.K.; Lal, R. Carbon and nitrogen pools in reclaimed land under forest and pasture ecosystems in Ohio, USA. Geoderma 2010, 157, 196–205. [Google Scholar] [CrossRef]
- Stahl, P.D.; Wick, A.F.; Ganjegunte, G.; Norton, U.; Ingram, L.J. Redevelopment of soil carbon pools on reclaimed surface mine lands. In Revitalizing the Environment: Proven Solutions and Innovative Approaches; Barnhisel, R.I., Ed.; ASMR: Billings, MT, USA, 2009; pp. 1348–1370. [Google Scholar]
- Das, R.; Maiti, S.K. Importance of carbon fractionation for the estimation of carbon sequestration in reclaimed coalmine soils—A case study from Jharia coalfields, Jharkhand, India. Ecol. Eng. 2016, 90, 135–140. [Google Scholar] [CrossRef]
- Rodrigue, J.A.; Burger, J.A. Forest soil productivity of mined land in the Midwestern and eastern coalfield regions. Soil Sci. Soc. Am. J. 2004, 68, 833–844. [Google Scholar] [CrossRef]
- Chatterjee, A.; Lal, R.; Shrestha, R.K.; Ussiri, D.A.N. Soil carbon pools of reclaimed minesoils under grass and forest landuses. Land Degrad. Dev. 2009, 20, 300–307. [Google Scholar] [CrossRef]
- Keskin, T.; Makineci, E. Some soil properties on coal mine spoils reclaimed with black locust (Robinia pceudoacacia L.) and umbrella pine (Pinus pinea L.) in Agacli-Istanbul. Environ. Monit. Assess. 2009, 159, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Boldt-Burisch, K.; Anne Naeth, M.; Schneider, B.U.; Huttl, R.F. Linkage between root systems of three pioneer plant species and soil nitrogen during early reclamation of a mine site in Lusatia, Germany. Restor. Ecol. 2015, 23, 357–365. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.; Masto, R.E.; Yadav, A.; George, J.; Ram, L.C.; Shukla, S.P. Soil quality index for evaluation of reclaimed coal mine spoil. Sci. Total Environ. 2016, 542, 540–550. [Google Scholar] [CrossRef] [PubMed]
- Banning, N.C.; Grant, C.D.; Jones, D.L.; Murphy, D.V. Recovery of soil organic matter, organic matter turnover and nitrogen cycling in a post-mining forest rehabilitation chronosequence. Soil Biol. Biochem. 2008, 40, 2021–2031. [Google Scholar] [CrossRef]
- Anderson, J.D.; Ingram, L.J.; Stahl, P.D. Influence of reclamation management practices on microbial biomass carbon and soil organic carbon accumulation in semiarid mined lands of Wyoming. Appl. Soil Ecol. 2008, 40, 387–397. [Google Scholar] [CrossRef]
- Ingram, L.J.; Schuman, G.E.; Stahl, P.D.; Spackman, L.K. Microbial respiration and organic carbon indicate nutrient cycling recovery in reclaimed soils. Soil Sci. Soc. Am. J. 2005, 69, 1737–1745. [Google Scholar] [CrossRef]
- Asensio, V.; Vega, F.A.; Covelo, E.F. Effect of soil reclamation process on soil C fractions. Chemosphere 2014, 95, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Andreasson, F.; Bergkvist, B.; Bååth, E. Bioavailability of DOC in leachates, soil matrix solutions and soil water extracts from beech forest floors. Soil Biol. Biochem. 2009, 41, 1652–1658. [Google Scholar] [CrossRef]
- Bu, X.; Ding, J.; Wang, L.; Yu, X.; Huang, W.; Ruan, H. Biodegradation and chemical characteristics of hot-water extractable organic matter from soils under four different vegetation types in the Wuyi Mountains, southeastern China. Eur. J. Soil Biol. 2011, 47, 102–107. [Google Scholar] [CrossRef]
- Sanderman, J.; Amundson, R. A comparative study of dissolved organic carbon transport and stabilization in California forest and grassland soils. Biogeochemistry 2008, 89, 309–327. [Google Scholar] [CrossRef]
- Cookson, W.R.; Osman, M.; Marschner, P.; Abaye, D.A.; Clark, I.; Murphy, D.V.; Stockdale, E.A.; Watson, C.A. Controls on soil nitrogen cycling and microbial community composition across land use and incubation temperature. Soil Biol. Biochem. 2007, 39, 744–756. [Google Scholar] [CrossRef]
- Wu, J.M.; Xi, M.; Kong, F.L.; Li, Y.; Zhang, Q.L. Review of researches on the factors influencing the dynamics of dissolved organic carbon in soils. Geol. Rev. 2013, 59, 953–961. [Google Scholar]
- Singh, J.S.; Raghubanshi, R.S.; Singh, R.S.; Srivastava, S.C. Microbial biomass acts as a source of plant nutrient in dry tropical forest and savanna. Nature 1989, 338, 499–500. [Google Scholar] [CrossRef]
- Chodak, M.; Niklińska, M. Effect of texture and tree species on microbial properties of mine soils. Appl. Soil Ecol. 2010, 46, 268–275. [Google Scholar] [CrossRef]
- Wiseman, P.E.; Seiler, J.R. Soil CO2 efflux across four age classes of plantation loblolly pine (Pinus taeda L.) on the Virginia Piedmont. For. Ecol. Manag. 2004, 192, 297–311. [Google Scholar] [CrossRef]
- Subke, J.A.; Bahn, M. On the ‘temperature sensitivity’ of soil respiration: Can we use the immeasurable to predict the unknown? Soil Biol. Biochem. 2010, 42, 1653–1656. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Bölter, M.; Chang, Q.; Duttmann, R.; Scheltz, A.; Petersen, J.F.; Wang, Z. Driving factors of temporal variation in agricultural soil respiration. Acta. Agric. Scand. Sect. B Soil Plant Sci. 2015, 65, 589–604. [Google Scholar] [CrossRef]
- Gershenson, A.; Bader, N.E.; Cheng, W. Effects of substrate availability on the temperature sensitivity of soil organic matter decomposition. Glob. Chang. Biol. 2009, 15, 176–183. [Google Scholar] [CrossRef]
- Liu, H. Thermal response of soil microbial respiration is positively associated with labile carbon content and soil microbial activity. Geoderma 2013, 193, 275–281. [Google Scholar] [CrossRef]
- Bujalský, L.; Kaneda, S.; Dvorščík, P.; Frouz, J. In situ soil respiration at reclaimed and unreclaimed post-mining sites: Responses to temperature and reclamation treatment. Ecol. Eng. 2014, 68, 53–59. [Google Scholar] [CrossRef]
- Fan, W.; Bai, Z.; Li, H.; Qiao, J.; Xu, J. Effects of different vegetation restoration patterns and reclamation years on microbes in reclaimed soil. Trans. Chin. Soc. Agric. Eng. 2011, 27, 330–336. [Google Scholar]
- Biasi, C.; Rusalimova, O.; Meyer, H.; Kaiser, C.; Wanek, W.; Barsukov, P.; Junger, H.; Richter, A. Temperature-dependent shift from labile to recalcitrant carbon sources of arctic heterotrophs. Rapid Commun. Mass Spectrom. RCM 2005, 19, 1401–1408. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Y.; Wang, F.; Yan, J.P.; Liu, Y.; Tan, H.Y.; Zhou, Y.Z. Effect of coversoil thickness on diurnal variation characteristics of reclaimed soil respiration in coal mining areas. J. Chin. Univ. Min. Technol. 2016, 45, 163–169. [Google Scholar]
- Šantr, H.; SiraŠicraba, M. On the relationship between specific respiration activity and microbial biomass in soils. Soil Biol. Biochem. 1991, 23, 525–532. [Google Scholar] [CrossRef]
- Anderson, T.-H. Microbial eco-physiological indicators to asses soil quality. Agric. Ecosyst. Environ. 2003, 98, 285–293. [Google Scholar] [CrossRef]
- Mastrogianni, A.; Papatheodorou, E.M.; Monokrousos, N.; Menkissoglu-Spiroudi, U.; Stamou, G.P. Reclamation of lignite mine areas with Triticum aestivum: The dynamics of soil functions and microbial communities. Appl. Soil Ecol. 2014, 80, 51–59. [Google Scholar] [CrossRef]
- Tripathi, S.; Kumari, S.; Chakraborty, A.; Gupta, A.; Chakrabarti, K.; Bandyapadhyay, B.K. Microbial biomass and its activities in salt-affected coastal soils. Biol. Fertil. Soils 2005, 42, 273–277. [Google Scholar] [CrossRef]
- Wong, V.N.L.; Dalal, R.C.; Greene, R.S.B. Salinity and sodicity effects on respiration and microbial biomass of soil. Biol. Fertil. Soils 2008, 44, 943–953. [Google Scholar] [CrossRef]
- Blagodatskaya, E.V.; Anderson, T.-H. Interactive effects of pH and substrate quality on the fungal-to-bacterial ratio and qCO2 of microbial communities in forest soils. Soil Biol. Biochem. 1998, 30, 1269–1274. [Google Scholar] [CrossRef]

| R4 1 | R7 | R10 | R13 | CK | |
|---|---|---|---|---|---|
| pH | 8.33 ± 0.07a 2 | 8.65 ± 0.03a | 8.67 ± 0.07a | 8.39 ± 0.09a | 6.64 ± 0.23b |
| EC (μS·cm−1) | 156.83 ± 15.67bc | 222.63 ± 31.63ab | 282.17 ± 30.97a | 230.17 ± 19.45a | 111.28 ± 6.58c |
| TN (g·kg−1) | 0.79 ± 0.06b | 0.78 ± 0.02b | 0.86 ± 0.02b | 1.06 ± 0.03a | 1.10 ± 0.03a |
| AN (mg·kg−1) | 40.97 ± 6.01c | 51.40 ± 1.47c | 64.45 ± 4.89b | 63.22 ± 2.80b | 90.43 ± 3.25a |
| AP (mg·kg−1) | 4.49 ± 0.84b | 8.38 ± 1.08b | 8.80 ± 1.22b | 12.44 ± 4.86b | 23.31 ± 3.30a |
| SOC | MBC | DOC | MQ | DOC/SOC | SBR (15 °C) | SBR (25 °C) | qCO2 (15 °C) | qCO2 (25 °C) | |
|---|---|---|---|---|---|---|---|---|---|
| SOC | 1 | ||||||||
| MBC | 0.737 ** | 1 | |||||||
| DOC | 0.242 | 0.020 | 1 | ||||||
| MQ | 0.502 * | 0.948 ** | −0.076 | 1 | |||||
| DOC/SOC | −0.889 ** | −0.680 ** | 0.153 | −0.504 * | 1 | ||||
| SBR (15 °C) | 0.482 * | 0.687 ** | 0.011 | 0.629 ** | −0.425 | 1 | |||
| SBR (25 °C) | 0.384 | 0.305 | 0.175 | 0.211 | −0.317 | 0.773 ** | 1 | ||
| qCO2 (15 °C) | −0.016 | −0.030 | 0.068 | −0.040 | 0.053 | 0.650 ** | 0.675 ** | 1 | |
| qCO2 (25 °C) | −0.568 ** | −0.849 ** | 0.050 | −0.871 ** | 0.601 ** | −0.442 * | −0.019 | 0.254 | 1 |
| pH | −0.412 * | −0.146 | −0.449 * | 0.054 | 0.131 | −0.089 | −0.213 | 0.011 | −0.155 |
| EC | 0.261 | 0.512 * | 0.053 | 0.570 ** | −0.302 | 0.564 ** | 0.440 * | 0.312 | −0.443 * |
| TN | 0.904 ** | 0.846 ** | 0.332 | 0.689 ** | −0.771 ** | 0.705 ** | 0.480 * | 0.301 | −0.661 ** |
| TC | −0.259 | −0.452 * | 0.294 | −0.524 * | 0.477 * | −0.356 | −0.099 | −0.176 | 0.657 ** |
| AN | 0.525 ** | 0.599 ** | 0.051 | 0.590 ** | −0.561 ** | 0.670 ** | 0.393 | 0.433 | −0.641 ** |
| AP | 0.467 * | 0.510 * | 0.147 | 0.449 * | −0.412 | 0.658 ** | 0.537 * | 0.161 | −0.368 |
| SIC | −0.468 * | −0.591 ** | 0.208 | −0.600 ** | 0.644 ** | −0.470 * | −0.184 | −0.177 | 0.733 ** |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qu, J.-F.; Hou, Y.-L.; Ge, M.-Y.; Wang, K.; Liu, S.; Zhang, S.-L.; Li, G.; Chen, F. Carbon Dynamics of Reclaimed Coal Mine Soil under Agricultural Use: A Chronosequence Study in the Dongtan Mining Area, Shandong Province, China. Sustainability 2017, 9, 629. https://doi.org/10.3390/su9040629
Qu J-F, Hou Y-L, Ge M-Y, Wang K, Liu S, Zhang S-L, Li G, Chen F. Carbon Dynamics of Reclaimed Coal Mine Soil under Agricultural Use: A Chronosequence Study in the Dongtan Mining Area, Shandong Province, China. Sustainability. 2017; 9(4):629. https://doi.org/10.3390/su9040629
Chicago/Turabian StyleQu, Jun-Feng, Yu-Le Hou, Meng-Yu Ge, Kun Wang, Shu Liu, Shao-Liang Zhang, Gang Li, and Fu Chen. 2017. "Carbon Dynamics of Reclaimed Coal Mine Soil under Agricultural Use: A Chronosequence Study in the Dongtan Mining Area, Shandong Province, China" Sustainability 9, no. 4: 629. https://doi.org/10.3390/su9040629
APA StyleQu, J.-F., Hou, Y.-L., Ge, M.-Y., Wang, K., Liu, S., Zhang, S.-L., Li, G., & Chen, F. (2017). Carbon Dynamics of Reclaimed Coal Mine Soil under Agricultural Use: A Chronosequence Study in the Dongtan Mining Area, Shandong Province, China. Sustainability, 9(4), 629. https://doi.org/10.3390/su9040629






