1. Introduction
Pumpkin (
Cucurbita pepo) is produced on about 1200 ha annually in New Jersey. It is an important cash crop for many roadside markets with varying forms of agrotourism. One of the major challenges of growing pumpkins is protecting the foliage from disease. Powdery mildew (
Podosphaera (sect.
sphaerotheca)
xanthii (castagne) U. Braun & N. shishkoff (also known as
spaerotheca) fusca (Fr) S. Blumer and S.
fuliginea (schlechtend.Fr) Pollacci) is one of the most problematic diseases attacking pumpkin and other cucurbit crops throughout the United States [
1]. Although the pathogen may overwinter on crop debris, in most years, it is wind-dispersed into northern regions from the Southern States during the production season [
1]. The pathogen typically infects older leaves and stems first, causing premature loss of foliage and resulting in a reduction in yield as the size and number of fruit decrease [
2].
This pervasive and difficult-to-manage disease is especially a challenge for organic growers where effective control options are limited compared to conventional growers. All conventional and some organic growers use weekly protectant fungicide applications to control powdery mildew. Costs of fungicides, equipment needed, time, and labor greatly contribute to the cost of production. Furthermore, the frequent use and/or overuse of the same conventional fungicides (
i.e., same mode-of-action) may enable pathogens such as cucurbit powdery mildew to develop resistance [
3]. Organic pumpkin growers, in most cases, rely on the use of powdery mildew-tolerant and/or resistant cultivars, if such seed is available.
Soil fertility management and optimizing plant nutrition can be an effective and a more sustainable approach to preventing or at least suppressing plant disease [
4]. Adding silicon to soil has been shown to effectively reduce the incidence and severity of powdery mildew disease in many crops, including pumpkin [
5,
6,
7,
8,
9]. Silicon is an abundant element in mineral soils but is mostly insoluble and sparingly available for plant uptake [
7]. Though not officially regarded as an essential plant nutrient, silicon is now recognized as a plant beneficial substance [
10].
For the production of cucurbits, enhanced silicon nutrition has been shown to suppress and delay the onset of powdery mildew disease [
8,
11]. According to a review by Datnoff [
10], “the effect of Si on plant resistance to disease is considered to be due to either an accumulation of absorbed Si in the epidermal tissue or an expression of metabolic or pathogenesis-mediated host defense responses”. Besides disease suppression, studies have shown that silicon alleviates many types of abiotic stress, including metal toxicity, salt tolerance, temperature extremes, drought, and solar radiation [
10].
With increasing awareness of the role of Si helping plants tolerate abiotic and biotic stress, organic and sustainable growers are asking about how to supply this beneficial substance to the soil using natural sources approved for organic farming. The best means of supplying plant available Si for crop production needs investigation so organic and conventional growers can take advantage of this sustainable approach for management of important diseases such as powdery mildew.
Soil amendments that may be useful for supplying plant available Si include naturally occurring deposits of minerals that may be mined from the earth, steel mill slags, and plant ash [
12]. Naturally occurring mined materials that are not on the USDA National Organic Program prohibited materials list are generally allowed for use in organic agriculture [
13]. Wollastonite is one such naturally occurring mineral form of calcium silicate that is listed by Organic Materials Review Institute (OMRI) [
14] as approved for use in organic agriculture. An alternative to wollastonite is calcium-magnesium-silicate-slag, a by-product of iron and steel manufacturing. However, it is questionable if this industrially processed material would be permitted for use in organic agriculture. Furthermore, questionable materials should be tested for heavy metal (Pb, Cd, Ni, Cr) content prior to land application. Another commercially available soil amendment, called MontanaGrow™ (MontanaGrow, Inc., Bonner, MT, USA), is promoted as a Si source for crop production and is also listed by OMRI. Wood ash contains the residual minerals, including variable amounts of Si, previously accumulated by growing trees. Ash from plant materials, for which no synthetic substance has been added, is generally allowed in organic agriculture as a soil amendment [
13]. Wood ash, crop residues, and compost are examples of materials that contain some Si. However, once Si is taken up by plants, some of it is converted to crystalline forms or phytoliths of low solubility [
15,
16]. Glacial rock flour, a mineral material deposited by melting glacial waters, consists of fine particles of naturally occurring rock with a high Si content much like the earth’s crust [
17].
The heterogeneous nature of these potentially useful soil amendments suggest a need to have them evaluated for how well they function for suppression of diseases such as powdery mildew. In addition to adding Si to the soil, these amendments may supply valuable amounts of other nutrients, and some of them may also serve as liming materials [
8].
The objectives of the greenhouse study was to: (i) compare several possible sources of plant available silicon for their ability to supply silicon for uptake by pumpkin plants; (ii) to compare ability of the amendments to neutralize soil acidity and raise soil pH; and (iii) to compare amendments for the ability to suppress powdery mildew development.
2. Materials and Methods
A Sassafras sandy loam (siliceous, mesic, typical Hapludult) soil was collected at the Rutgers University Vegetable Research Farm in North Brunswick, NJ, USA from an area which had no recent history of chemical fertilizer or pesticide inputs. Although not certified organic land, nothing in its recent history prevented this land or soil from being transitioned into organic farming. Prior to soil collection, the field was tilled to a depth of 15 cm using a rotary tiller. The soil collected from the tilled layer was sieved through a 2 mm screen to remove stones and plant litter.
Soil pH was measured using a 1:1 soil volume to water ratio. Soil tests for Si were performed using the acetic acid extraction method of Korndorfer [
18]. All extractions were analyzed by inductively coupled plasma atomic emission spectroscopy (ICP-AES).
The rate of limestone (as calcium carbonate) needed to raise soil pH to the recommended level (pH 6.5) for pumpkin was estimated to be 8960 kg·ha−1 based on initial pH, soil texture class and Rutgers Cooperative Extension Vegetable Production Recommendations for pumpkin production. Pots (Poly-Tainer-Can #2, Nurseries Supplies Inc., Orange, CA, USA) were filled with 11 kg of dry soil. Treatments consisted of untreated soil or soil with one of seven different amendments at the rate of 8960 kg·ha−1 (or 44 g per 11 kg dry soil).
Agricultural liming materials, consisting of calcite and dolomite, with nearly matching calcium carbonate equivalent (CCE 98% vs. CCE 95%, respectively) ratings were applied at the same rates as the silicon fertilizers to the control pots. Treatments also included wood ash, obtained from a local wood stove that burned mixed hardwood tree species; glacial rock flour, a collection of naturally occurring fines deposited by glacial activity; CaMg silicate slag (CrossOverTM , HARSCO, Sarver, PA, USA), a proprietary silicon fertilizer manufactured from slag by-product; wollastonite (R.T. Vanderbilt Company Inc., Norwalk, CT, USA), a naturally occurring mined calcium silicate; and MontanaGrowTM (Drummond, MT, USA), a naturally occurring mined fertilizer. Both wollastonite and MontanaGrowTM are OMRI-listed soil amendments.
The greenhouse experiment was conducted in the presence of powdery mildew infected pumpkin plants. These infected plants were first grown outdoors during the late summer months of 2013 to capture natural inocula of powdery mildew pathogen. On 15 November 2013, pumpkin cv. Howden was seeded and grown inside the greenhouse in the six liter experimental pots containing soil collected from the land area as described previously. The amendments were applied to this soil at the same rates as stated previously. Treatments were replicated four times in a completely randomized design. Before seeding, all pots received Perdue AgriRecycle’s microSTART60 (3N-4P-5K), an OMRI-listed product, at the rate of 40 g·kg−1 of soil.
The pumpkin plants were thinned to two plants per pot at one week after germination. The number of powdery mildew lesions per plant was counted on 15, 17, 19, and 21 December 2013. A visual percentage estimate of leaf area infected with powdery mildew was determined on 23, 25, 27, 28, 30, and 31 December 2013 and 2 January 2014. The area under disease progress curve (AUDPC) values for the development of powdery mildew in each treatment were calculated using the trapezoid method [
19]. Above ground biomass was obtained by collecting the plant vine tissue above the third node. A soil sample representing each treatment was collected by taking a soil core (2 cm diameter × 15 cm deep) from each pot immediately following biomass harvest. Soils tests were performed as described previously.
All plant samples were dried at 70C for 48 h and weighed. The samples were ground in a Wiley mill (Thomas Scientific, Swedesboro, NJ, USA) to pass a 1 mm sieve. The samples were analyzed for mineral nutrients using ICP-AES after samples were digested with nitric acid and hydrogen peroxide [
20]. The silicon concentration was determined using the acetic acid extract method [
21].
Experimental data was subjected to analysis of variance using the t test (least significant difference, LSD) and REGWQ procedure of SAS (SAS Institute, 1999–2000, Cary, NC, USA). Correlation analysis was used to examine relationships between soil tests Si, plant tissue Si and disease or biomass yield measurements.
3. Results
At the conclusion of the greenhouse experiment, or six weeks after the soils were amended, it was visually apparent that some of the amendments were effective as liming materials and others had little impact on soil pH. Soil pH levels associated with each amendment were as follows: pH 6.3 calcite, pH 6.0 dolomite, pH 5.9 CaMg silicate, pH 6.2 wollastonite, pH 6.5 wood ash, pH 4.3 MontanaGrow™, pH 4.6 glacial rock floor. These materials, with the exceptions of MontanaGrow™ and glacial rock floor, increased soil pH near to the desired pH level of 6.5 for pumpkin.
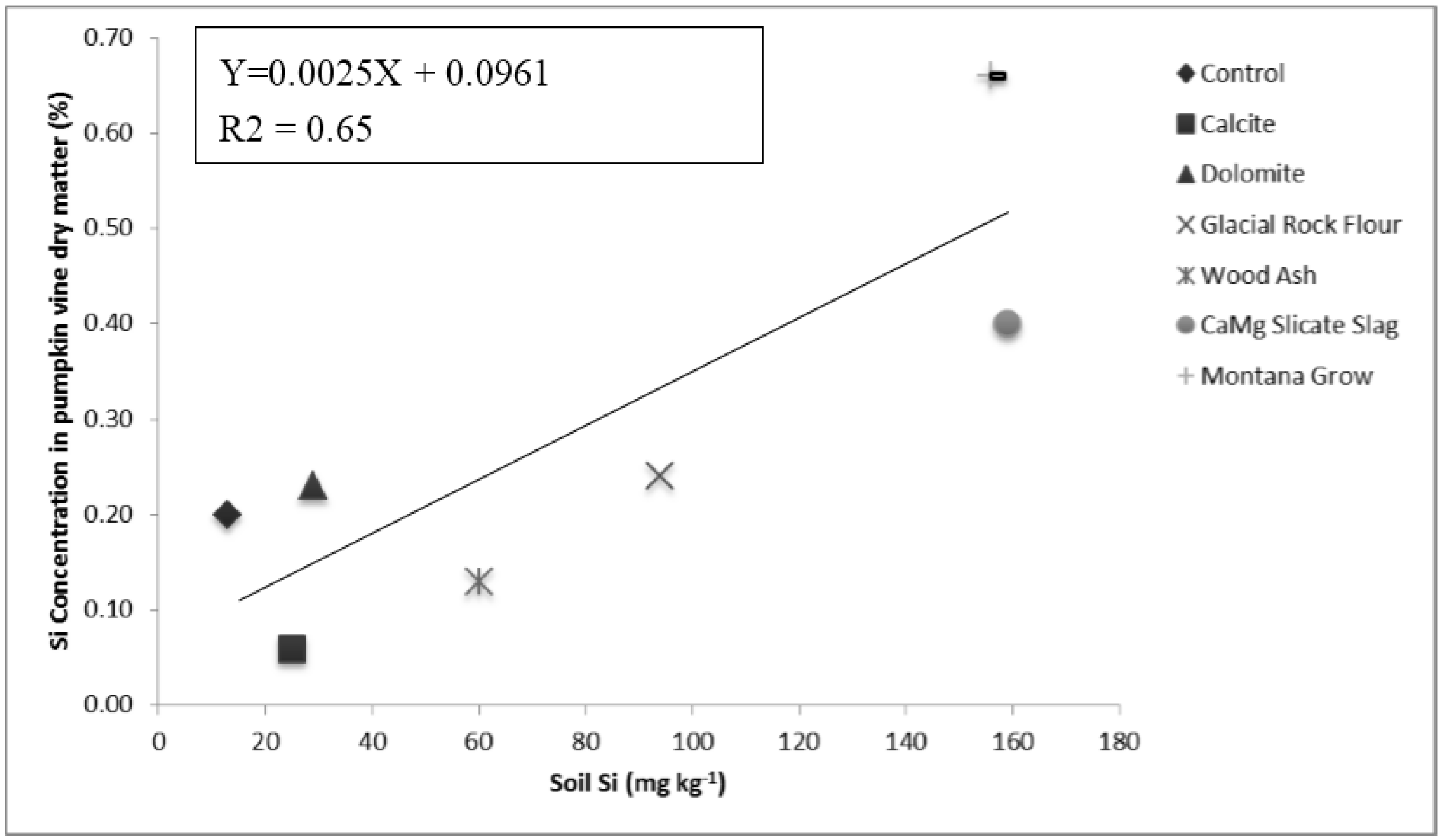
Soil test Si levels, as measured using acetic acid extract, were increased by more than tenfold over unamended soil by the CaMg silicate slag and wollastonite amendments. Glacial rock flour and wood ash increased extractable Si levels to a lesser extent. Other amendments exhibited little impact on soil test extractable Si. MontanaGrow™, which is marketed as a Si soil amendment, had little influence on Si soil test level (
Figure 1).
Within the harvested plant tissue, concentrations of N, P, Ca, S, B, Fe, and Cu were not influenced by soil amendment, but concentrations of Si, K, Mg, Mn, and Zn were sometimes changed (
Table 1).
The major cations in plants are known to compete for uptake. Thus, when any one of these cations is supplied in abundance, the uptake of those in relatively lower supply may be depressed [
22]. This antagonism among cations tended to be exhibited but in most cases it did not result in statistically significant differences. Calcite and wollastonite, which are especially rich in Ca and low in Mg content, tended to suppress Mg uptake (
Table 1).
Micronutrient availability is often influenced by liming and elevations in soil pH. The soil amendments that were effective liming materials reduced tissue concentrations of Mn and Zn. In the case of Mn, the soil amendments which were shown to be effective as liming materials reduced tissue concentrations from what might be considered excessive levels to normal levels [
23].
Silicon in the plant tissue varied among treatments from the lowest concentration (0.06% Si for calcite) to the highest (0.66% ppm Si for wollastonite) by a factor of more than ten (
Table 1). Wollastonite was the only treatment to increase the concentration Si in the plant tissue over the control soil. CaMg silicate slag also tended to increase the concentrations of Si in plant tissue but not significantly In general, plant tissue Si exhibited a positive relationship (R
2 = 0.65) with increasing acetic acid soil test levels of Si (
Figure 1).
Along with calcite and dolomite limestone, other soil amendments that did not increase plant Si uptake included MontanaGrow™, wood ash, and glacial rock flour (
Table 1). Pumpkin plant vein dry matter yield exhibited only weak positive relationships with Si concentration in the plant tissue, or with soil test levels of Si. This short term greenhouse study was not designed to measure pumpkin fruit yield responses to treatments.
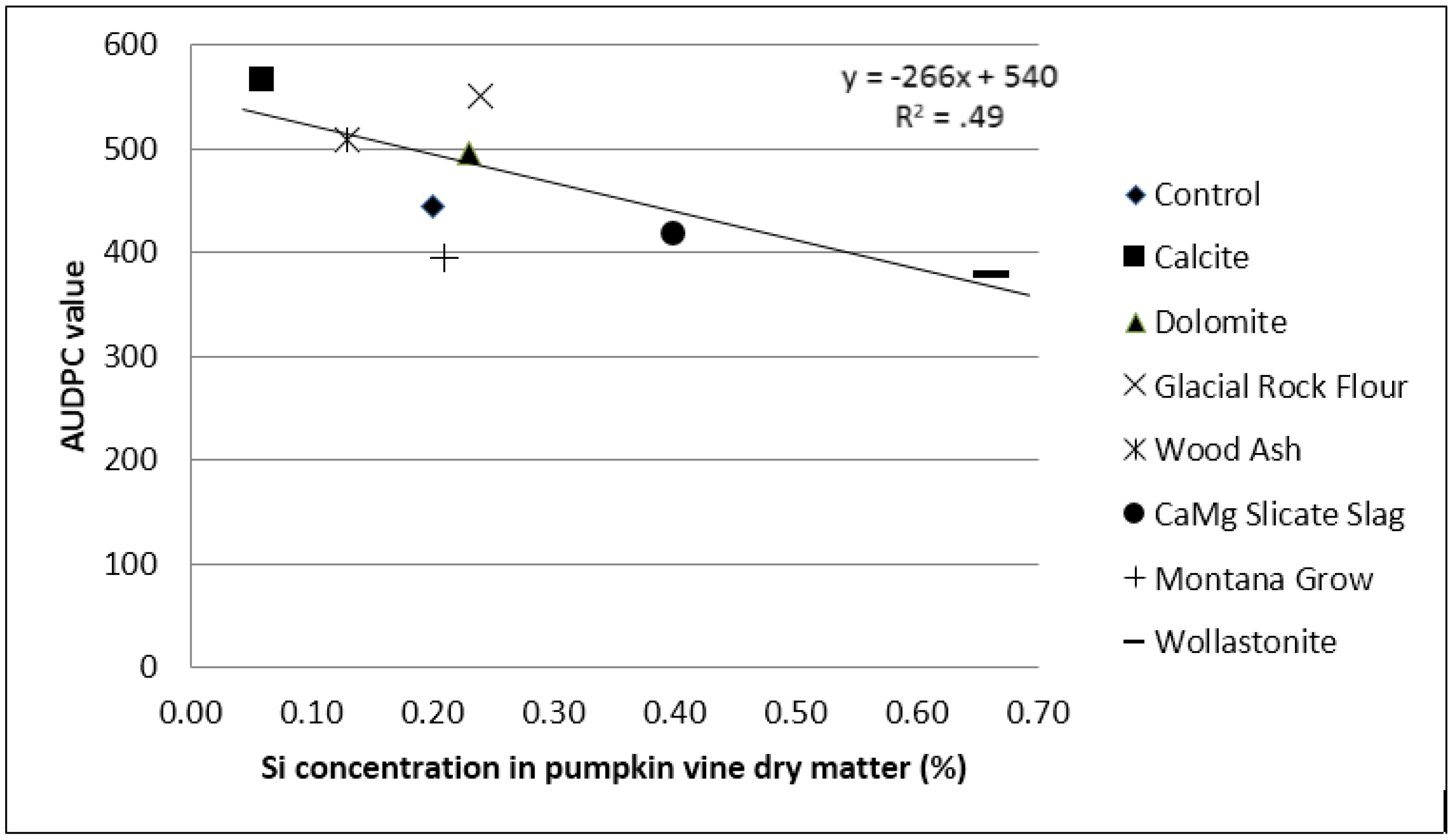
In the first week, during the onset of the disease, the wollastonite treatment consistently ranked as having the fewest number of powdery mildew lesions on pumpkin leaves (
Table 2 and
Table 3). The percent leaf area covered by powdery mildew tended to be lowest for wollastonite. As Si concentration increased in the plant, powdery mildew development as measured by AUDPC value decreased, suggesting that increasing Si uptake had some effect on decreasing powdery mildew development (R
2 = 0.49) (
Figure 2). The treatments that were effective at increasing plant uptake of Si tended to confer the greatest benefits on younger plants and with diminishing effectiveness with plant age.
4. Discussion and Conclusions
This study demonstrates that wollastonite is both an effective liming material and an effective source of plant available Si. Of the various Si sources, wollastonite tends to be better for enhancing Si uptake by pumpkin plants and for offering more protection from powdery mildew disease. CaMg Silicate Slag also tended to be effective in this regard but it did not reach the level of statistical significance. MontanaGrow™, wood ash, and glacial rock flour did little to improve Si uptake or suppress powdery mildew disease on pumpkin. Wood ash increased soil pH and supplied an abundance of K but little Si.
A short term greenhouse study may not allow for the disease suppression responses to be translated in pumpkin vein biomass yield. However, in full season field research suppression of early season powdery mildew disease development in association with Si uptake has been shown to be reflected in a pumpkin fruit yield increase in previous work [
8].
Organic growers need to check with their certifier before applying questionable amendments to certified farmland. MontanaGrow™, might appeal to organic growers because it is an OMRI listed product, but the current study does not indicate that this is an effective source of Si nor effective as a liming material. It may, however, have other values as a soil amendment. The practical findings from this research suggest that organic growers could use wollastonite as a soil amendment and liming material to achieve multiple benefits for pumpkin production. Potential benefits of applying wollastonite to soil include delayed onset of powdery mildew disease, increased yield, and improved soil fertility. The use of wollastonite for pumpkin production may be a better value than limestone in that they may provide more agronomic benefits. Besides the initial crop being grown, there may be residual benefits from the applied Si to other crops following in the rotation. Long term field studies would be needed to evaluate the residual benefits of Si soil amendments. On acid soils, a dual purpose Si fertilizer/liming agent may also improve the economics of Si fertilization.