Assessment of Reclamation Treatments of Abandoned Farmland in an Arid Region of China
Abstract
:1. Introduction
2. Materials and Methods
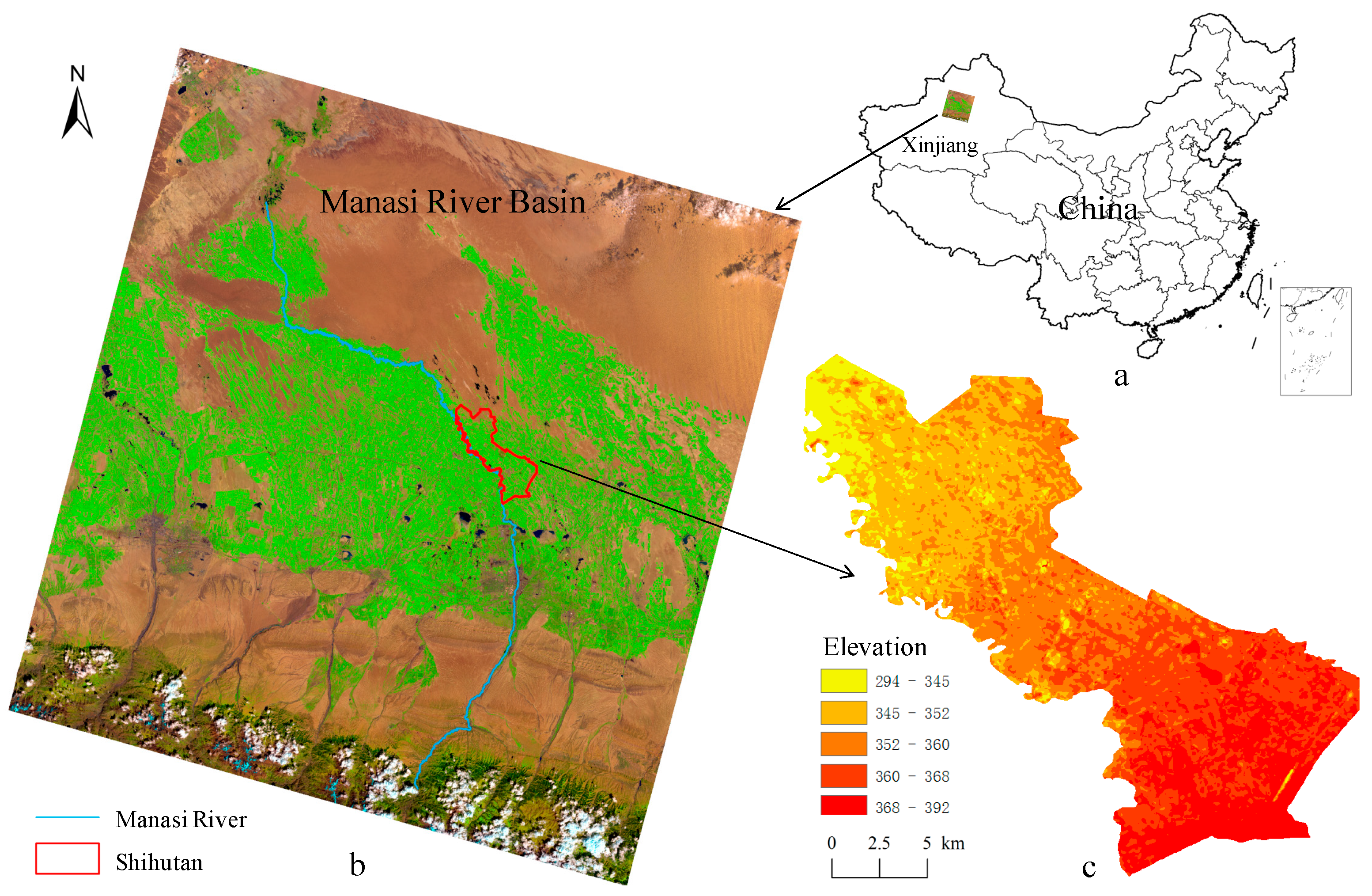
2.1. Study Area
2.2. Experimental Design and Sample Collection
2.3. Soil Sample Test Indicators and Methods
2.4. Statistical Analyses
3. Results
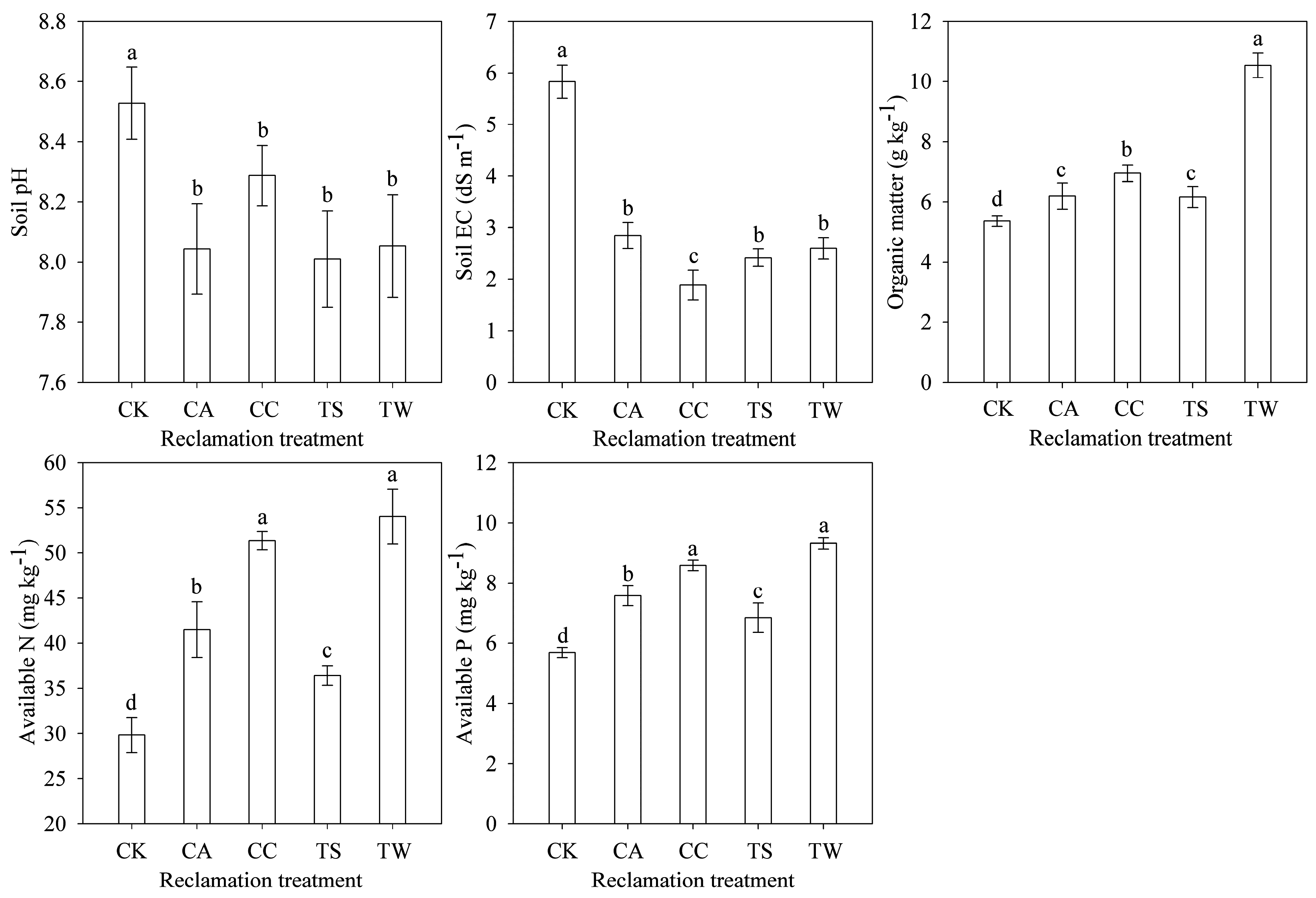
3.1. Soil Nutrients
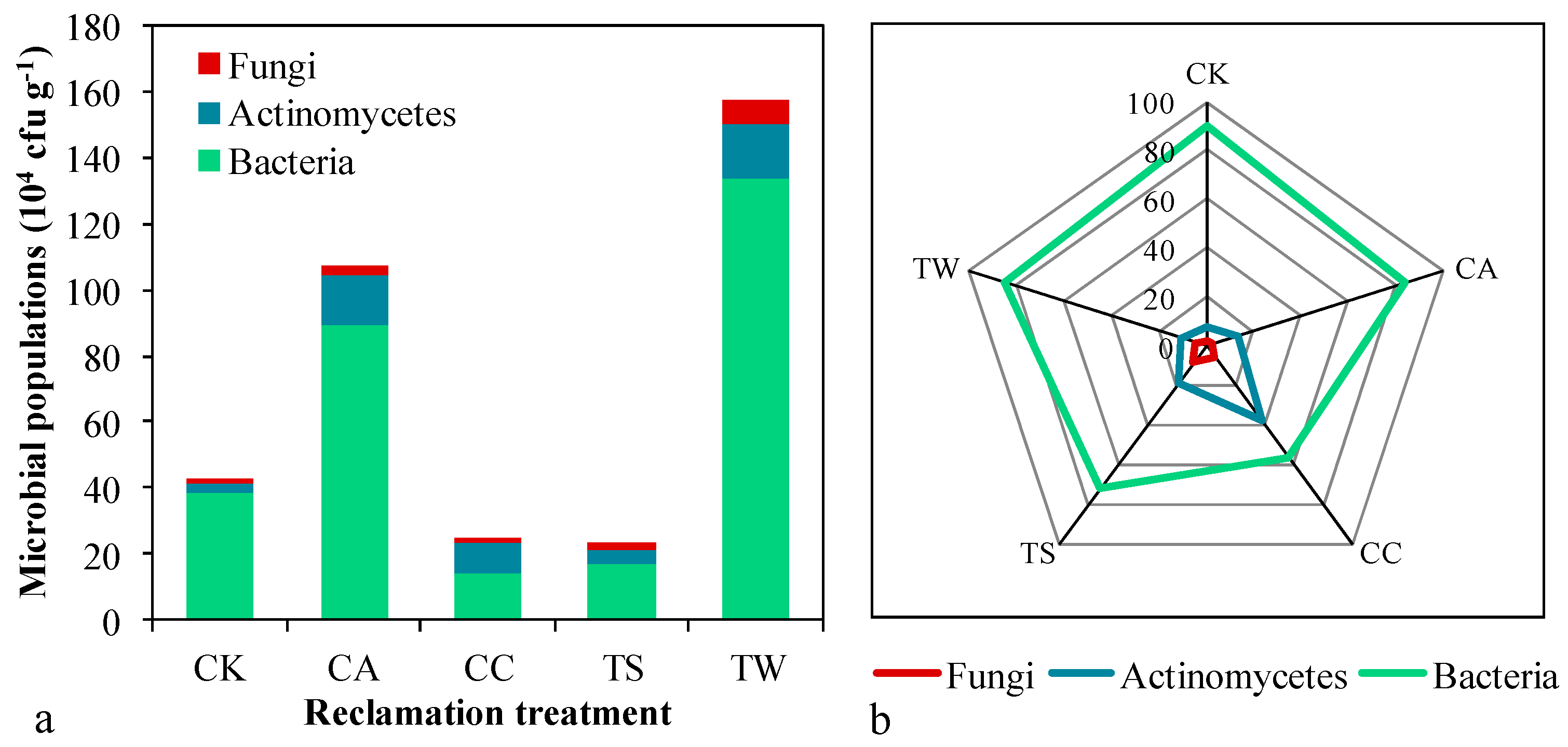
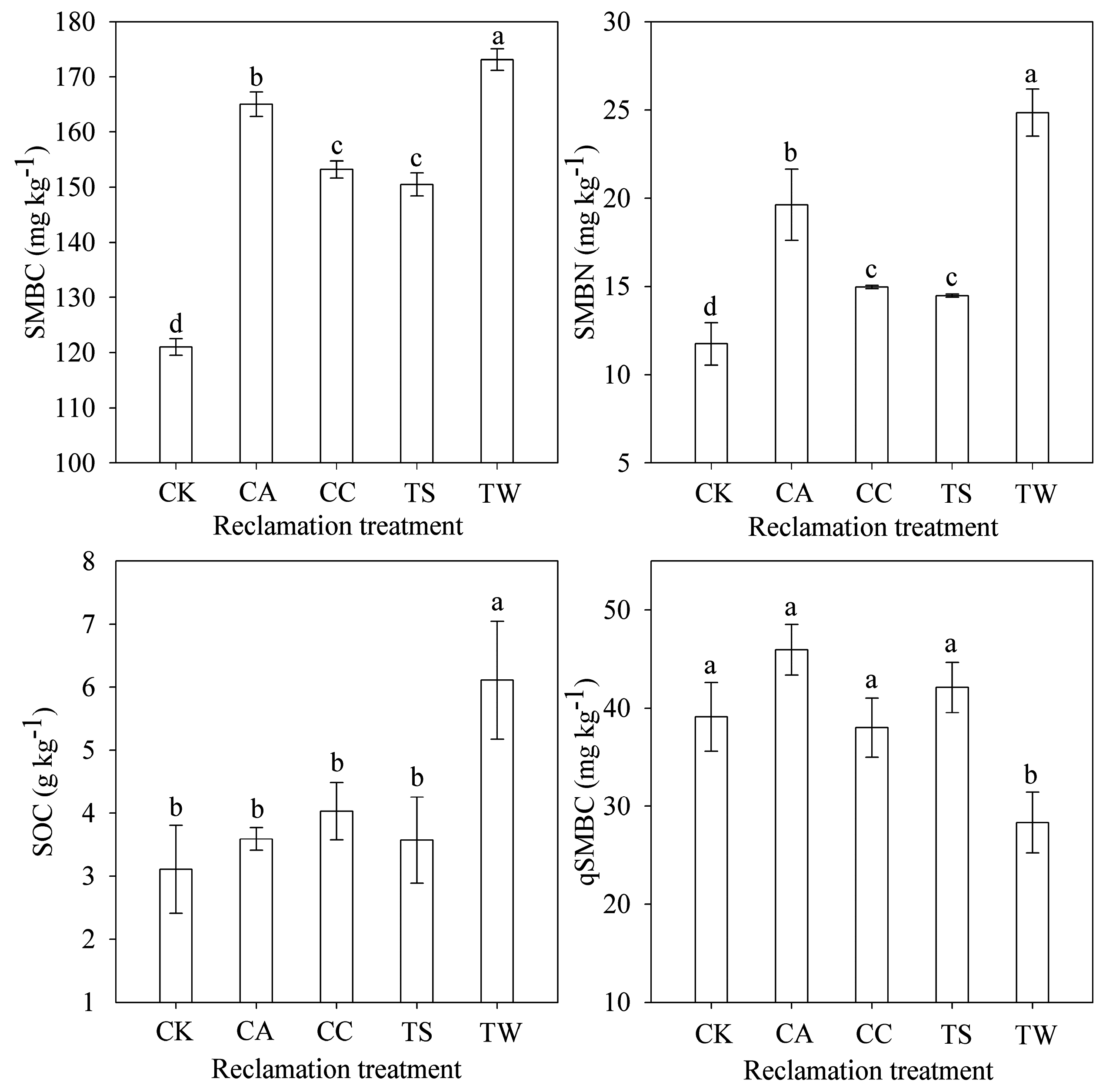
3.2. Soil Microbial Population and Biomass
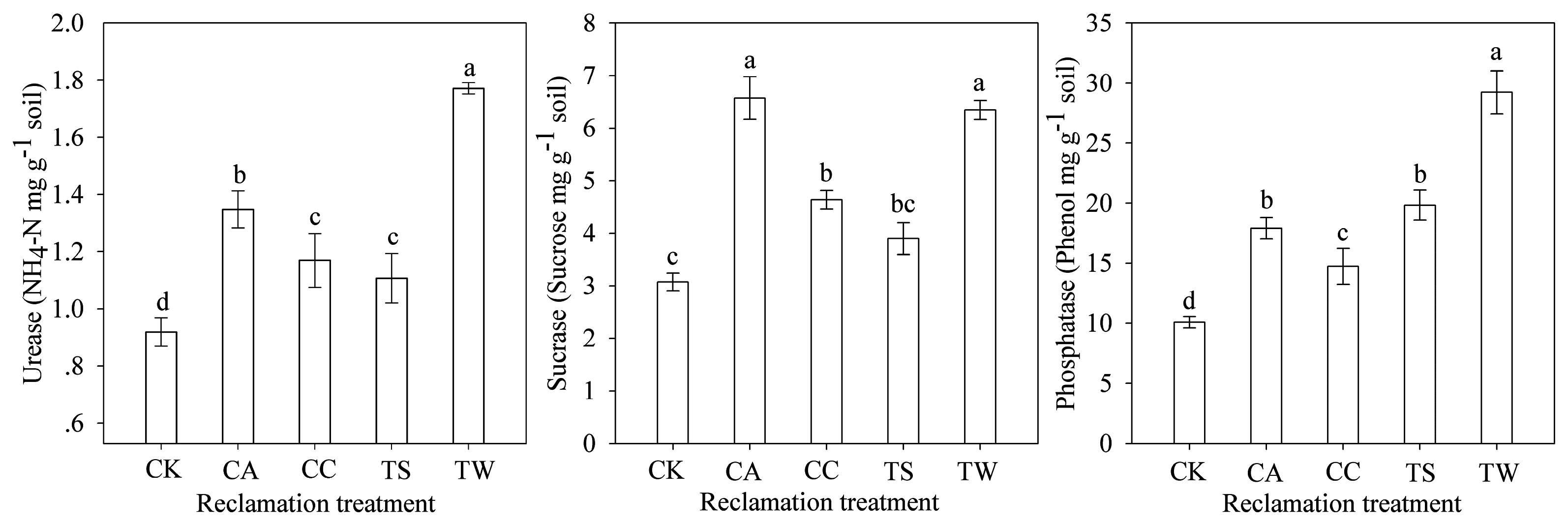
3.3. Soil Enzyme Activity
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lasanta, T.; Garcıa-Ruiz, J.; Pérez-Rontomé, C.; Sancho-Marcén, C. Runoff and sediment yield in a semi-arid environment: The effect of land management after farmland abandonment. Catena 2000, 38, 265–278. [Google Scholar] [CrossRef]
- Moreira, F.; Russo, D. Modelling the impact of agricultural abandonment and wildfires on vertebrate diversity in Mediterranean Europe. Landsc. Ecol. 2007, 22, 1461–1476. [Google Scholar] [CrossRef]
- García-Ruiz, J.M. The effects of land uses on soil erosion in Spain: A review. Catena 2010, 81, 1–11. [Google Scholar] [CrossRef]
- Hou, J.; Fu, B.; Liu, Y.; Lu, N.; Gao, G.; Zhou, J. Ecological and hydrological response of farmlands abandoned for different lengths of time: Evidence from the Loess Hill Slope of China. Glob. Planet. Chang. 2014, 113, 59–67. [Google Scholar] [CrossRef]
- Günal, H.; Korucu, T.; Birkas, M.; Özgöz, E.; Halbac-Cotoara-Zamfir, R. Threats to sustainability of soil functions in Central and Southeast Europe. Sustainability 2015, 7, 2161–2188. [Google Scholar] [CrossRef]
- Lee, H.F.; Zhang, D.D. Perceiving desertification from the lay perspective in northern China. Land Degrad. Dev. 2004, 15, 529–542. [Google Scholar] [CrossRef]
- Van den Berg, L.; Kellner, K. Restoring degraded patches in a semi-arid rangeland of South Africa. J. Arid Environ. 2005, 61, 497–511. [Google Scholar] [CrossRef]
- Roldán, A.; Albaladejo, J.; Thornes, J. Aggregate stability changes in a semiarid soil after treatment with different organic amendments. Arid Land Res. Manag. 1996, 10, 139–148. [Google Scholar]
- Hinojosa, M.B.; Carreira, J.A.; García-Ruíz, R.; Dick, R.P. Soil moisture pre-treatment effects on enzyme activities as indicators of heavy metal-contaminated and reclaimed soils. Soil Biol. Biochem. 2004, 36, 1559–1568. [Google Scholar] [CrossRef]
- Pérez-de-Mora, A.; Burgos, P.; Madejón, E.; Cabrera, F.; Jaeckel, P.; Schloter, M. Microbial community structure and function in a soil contaminated by heavy metals: Effects of plant growth and different amendments. Soil Biol. Biochem. 2006, 38, 327–341. [Google Scholar] [CrossRef]
- Bošeľa, M.; Petráš, R.; Šebeň, V.; Mecko, J.; Marušák, R. Evaluating competitive interactions between trees in mixed forests in the Western Carpathians: Comparison between long-term experiments and SIBYLA simulations. For. Ecol. Manag. 2013, 310, 577–588. [Google Scholar] [CrossRef]
- Matuszkiewicz, J.M.; Kowalska, A.; Kozłowska, A.; Roo-Zielińska, E.; Solon, J. Differences in plant-species composition, richness and community structure in ancient and post-agricultural pine forests in central Poland. For. Ecol. Manag. 2013, 310, 567–576. [Google Scholar] [CrossRef]
- Chen, L.; Gong, J.; Fu, B.; Huang, Z.; Huang, Y.; Gui, L. Effect of land use conversion on soil organic carbon sequestration in the loess hilly area, Loess Plateau of China. Ecol. Res. 2007, 22, 641–648. [Google Scholar] [CrossRef]
- Wei, X.; Shao, M.; Shao, H.; Gao, J.; Xu, G. Fractions and bioavailability of soil inorganic phosphorus in the Loess Plateau of China under different vegetations. Acta Geol. Sin. 2011, 85, 263–270. [Google Scholar]
- Dietrich, A.L.; Lind, L.; Nilsson, C.; Jansson, R. The use of phytometers for evaluating restoration effects on riparian soil fertility. J. Environ. Qual. 2014, 43, 1916–1925. [Google Scholar] [CrossRef] [PubMed]
- Sardinha, M.; Müller, T.; Schmeisky, H.; Joergensen, R.G. Microbial performance in soils along a salinity gradient under acidic conditions. Appl. Soil Ecol. 2003, 23, 237–244. [Google Scholar] [CrossRef]
- Yuan, B.C.; Li, Z.Z.; Liu, H.; Gao, M.; Zhang, Y.Y. Microbial biomass and activity in salt affected soils under arid conditions. Appl. Soil Ecol. 2007, 35, 319–328. [Google Scholar] [CrossRef]
- Chowdhury, N.; Marschner, P.; Burns, R.G. Soil microbial activity and community composition: Impact of changes in matric and osmotic potential. Soil Biol. Biochem. 2011, 43, 1229–1236. [Google Scholar] [CrossRef]
- Gu, Y.; Zhang, X.; Tu, S.; Lindström, K. Soil microbial biomass, crop yields, and bacterial community structure as affected by long-term fertilizer treatments under wheat-rice cropping. Eur. J. Soil Biol. 2009, 45, 239–246. [Google Scholar] [CrossRef]
- Wang, B.; Liu, G.B.; Xue, S.; Zhu, B. Changes in soil physico-chemical and microbiological properties during natural succession on abandoned farmland in the Loess Plateau. Environ. Earth Sci. 2011, 62, 915–925. [Google Scholar] [CrossRef]
- Biondini, M.E.; Bonham, C.D.; Redente, E.F. Secondary successional patterns in a sagebrush (Artemisia tridentata) community as they relate to soil disturbance and soil biological activity. Vegetatio 1985, 60, 25–36. [Google Scholar] [CrossRef]
- Wong, V.N.; Dalal, R.C.; Greene, R.S. Salinity and sodicity effects on respiration and microbial biomass of soil. Biol. Fertil. Soils 2008, 44, 943–953. [Google Scholar] [CrossRef]
- Zhang, X.; Dong, W.; Dai, X.; Schaeffer, S.; Yang, F.; Radosevich, M.; Xu, L.; Liu, X.; Sun, X. Responses of absolute and specific soil enzyme activities to long term additions of organic and mineral fertilizer. Sci. Total Environ. 2015, 536, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Sinsabaugh, R.L.; Antibus, R.K.; Linkins, A.E. An enzymic approach to the analysis of microbial activity during plant litter decomposition. Agric. Ecosyst. Environ. 1991, 34, 43–54. [Google Scholar] [CrossRef]
- Berg, B. Litter decomposition and organic matter turnover in northern forest soils. For. Ecol. Manag. 2000, 133, 13–22. [Google Scholar] [CrossRef]
- Caldwell, B.A. Enzyme activities as a component of soil biodiversity: A review. Pedobiologia 2005, 49, 637–644. [Google Scholar] [CrossRef]
- Paul, E.A. Soil Microbiology, Ecology and Biochemistry; Academic Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Goudie, A.S. The Human Impact on the Natural Environment: Past, Present, and Future; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Fan, H.; Pan, X.; Li, Y.; Chen, F.; Zhang, F. Evaluation of soil environment after saline soil reclamation of Xinjiang Oasis, China. Agron. J. 2008, 100, 471–476. [Google Scholar]
- Zhang, F.; Hanjra, M.A.; Hua, F.; Shu, Y.; Li, Y. Analysis of climate variability in the Manas River Valley, North-Western China (1956–2006). Mitig. Adapt. Strategies Glob. Chang. 2014, 19, 1091–1107. [Google Scholar] [CrossRef]
- Rasapoor, M.; Nasrabadi, T.; Kamali, M.; Hoveidi, H. The effects of aeration rate on generated compost quality, using aerated static pile method. Waste Manag. 2009, 29, 570–573. [Google Scholar] [CrossRef] [PubMed]
- Allen, O. Experiments in Soil Bacteriology. Soil Sci. 1958, 85, 172. [Google Scholar] [CrossRef]
- Gong, S.; Zhang, T.; Guo, R.; Cao, H.; Shi, L.; Guo, J.; Sun, W. Response of soil enzyme activity to warming and nitrogen addition in a meadow steppe. Soil Res. 2015, 53, 242–252. [Google Scholar] [CrossRef]
- Munns, R. Strategies for Crop Improvement in Saline Soils. Salinity and Water Stress; Springer: New York, NY, USA, 2009; pp. 99–110. [Google Scholar]
- Wang, K.; Fan, H.; Tariq, R.; Hanjrac, M.A.; Deng, B.; Liu, H.; Zhang, F. Changes in soil carbon and nitrogen under long-term cotton plantations in China. J. Agric. Sci. 2011, 149, 497–505. [Google Scholar]
- Li, J.H.; Fang, X.W.; Jia, J.J.; Wang, G. Effect of legume species introduction to early abandoned field on vegetation development. Plant Ecol. 2007, 191, 1–9. [Google Scholar] [CrossRef]
- Jouve, L.; Hoffmann, L.; Hausman, J.F. Polyamine, carbohydrate, and proline content changes during salt stress exposure of aspen (Populus tremula L.): Involvement of oxidation and osmoregulation metabolism. Plant Biol. 2004, 6, 74–80. [Google Scholar] [PubMed]
- Zeller, V.; Bardgett, R.D.; Tappeiner, U. Site and management effects on soil microbial properties of subalpine meadows: A study of land abandonment along a north–south gradient in the European Alps. Soil Biol. Biochem. 2001, 33, 639–649. [Google Scholar] [CrossRef]
- Saggar, S.; Yeates, G.W.; Shepherd, T.G. Cultivation effects on soil biological properties, microfauna and organic matter dynamics in Eutric Gleysol and Gleyic Luvisol soils in New Zealand. Soil Tillage Res. 2001, 58, 55–68. [Google Scholar] [CrossRef]
- Rustad, L.E.J.L.; Campbell, J.; Marion, G.; Norby, R.; Mitchell, M.; Hartley, A.; Cornelissen, J.; Gurevitch, J. A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 2001, 126, 543–562. [Google Scholar]


| Reclamation Treatment | Plant Density (Plants ha−1) | Irrigation | Fertilizer | |||
|---|---|---|---|---|---|---|
| Type | Times | Quantity (m3·ha−1) | Fertilizer Amount (ha−1) | Mode | ||
| Natural succession (CK) | 2.89 × 104 | No irrigation | 0 | 0 | No fertilizer | — |
| Continuous alfalfa (CA) | 5.5 × 106 | Drip irrigation | 8–10 | 4500 | 300 kg N | Drip fertigation |
| Continuous cotton (CC) | 1.95 × 105 | Drip irrigation under plastic mulch | 8–10 | 4500 | 300 kg N, 120 kg P2O5, 60 kg K2O | Basal application + drip fertigation |
| Trees (TS) | 3.3 × 103 | Drip irrigation | 6 | 300 | 270 kg N | Drip fertigation |
| Tree-wheat intercropping (TW) | 1.02 × 103 + 4.8 × 106 | Drip irrigation | 2 + 6 | 5250 | 300 kg N, 120 kg P2O5, 60 kg K2O | Basal application + drip fertigation |
| Indicator | Continuous Alfalfa (CA) | Continuous Cotton (CC) | Trees (TS) | Tree–Wheat Intercropping (TW) |
|---|---|---|---|---|
| Soil pH | −4.7 | −1.8 | −5.1 | −4.6 |
| Soil EC | −51.1 | −67.6 | −58.5 | −55.4 |
| Soil organic matter | 26.6 | 42.1 | 26.0 | 115.5 |
| Available N | 39.1 | 72.2 | 22.1 | 81.1 |
| Available P | 33.2 | 51.0 | 20.4 | 63.8 |
| Indicator | Continuous Alfalfa (CA) | Continuous Cotton (CC) | Trees (TS) | Tree–Wheat Intercropping (TW) |
|---|---|---|---|---|
| Fungi | 172.3 | 38.6 | 95.1 | 572.5 |
| Actinomycetes | 359.4 | 196.9 | 37.5 | 428.1 |
| Bacteria | 134.2 | 63.2 | 55.3 | 250.0 |
| SOC | 15.4 | 29.6 | 14.8 | 96.5 |
| SMBC | 36.4 | 26.4 | 24.0 | 43.0 |
| SMBN | 48.5 | 13.6 | 9.8 | 88.6 |
| qSMBC | 17.6 | 2.8 | 7.7 | 27.6 |
| Indicator | Continuous Alfalfa (CA) | Continuous Cotton (CC) | Trees (TS) | Tree–Wheat Intercropping (TW) |
|---|---|---|---|---|
| Urease | 51.7 | 31.5 | 24.7 | 98.9 |
| Sucrase | 105.0 | 44.5 | 21.2 | 97.8 |
| Phosphatase | 73.8 | 42.7 | 92.2 | 183.5 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, H.; Zhang, F.; Chen, Y.; Xu, T.; Cheng, Z.; Liang, J. Assessment of Reclamation Treatments of Abandoned Farmland in an Arid Region of China. Sustainability 2016, 8, 1183. https://doi.org/10.3390/su8111183
Yang H, Zhang F, Chen Y, Xu T, Cheng Z, Liang J. Assessment of Reclamation Treatments of Abandoned Farmland in an Arid Region of China. Sustainability. 2016; 8(11):1183. https://doi.org/10.3390/su8111183
Chicago/Turabian StyleYang, Haichang, Fenghua Zhang, Yun Chen, Tingbao Xu, Zhibo Cheng, and Jing Liang. 2016. "Assessment of Reclamation Treatments of Abandoned Farmland in an Arid Region of China" Sustainability 8, no. 11: 1183. https://doi.org/10.3390/su8111183
APA StyleYang, H., Zhang, F., Chen, Y., Xu, T., Cheng, Z., & Liang, J. (2016). Assessment of Reclamation Treatments of Abandoned Farmland in an Arid Region of China. Sustainability, 8(11), 1183. https://doi.org/10.3390/su8111183









