Abstract
The background of bioethanol as an alternative to conventional fuels is analyzed with the aim of examining the efficiency of bioethanol production by first (sugar-based) and second (cellulose-based) generation processes. Energy integration is of paramount importance for a complete recovery of the processes’ exergy potential. Based upon literature data and our own findings, exergy analysis is shown to be an important tool in analyzing integrated ethanol production from an efficiency and cost perspective.
1. Introduction
Exergy analysis is a thermodynamic analysis technique, based on the first and second law of thermodynamics, providing an alternative means of energetically assessing and comparing processes by providing a measure of how close the actual process approaches the (thermodynamically) ideal situation. Processes associated with low energy efficiencies will result in a sharp decrease in total exergy. Exergy is clearly better-suited to identifying the causes and locations of thermodynamic losses than a traditional energy balance. The application of exergy analysis has only in recent years been recognized by industry and academics, and the number of research papers dedicated to the exergy analysis of specific processes has been increasing steadily. The present paper will specifically focus on the exergy and CO2 analyses of energy conversion methods to produce ethanol.
The fundamentals of exergy analysis will be summarized and different types of exergetic efficiencies will be defined, being either a simple efficiency (based on irreversibilities when developing energy balances), the exergy ratio (ratio of desired output to the exergy invested in the process), and an efficiency with transiting exergy (modification of simple efficiency through extracting the untransformed exergy components from incoming and outgoing streams).
Numerous papers have been dedicated to exergy analysis in the bioethanol field, as will be discussed. The major trends will be identified. Based on this overview, a preliminary comparison between various systems can already be made.
Exergetic analysis is closely related to a different method of sustainability assessment, i.e., the life cycle assessment (LCA) [1,2,3,4,5,6,7,8,9,10,11,12,13]. A combination of both methods is the so-called exergetic life cycle assessment (ELCA) [14,15,16,17,18]. This type of LCA requires closed material and energy balances. The impact assessment is performed by calculating the exergy destruction during the process. The sum of all destructions leads to the total life cycle irreversibility. The ELCA accounts for all environmental problems, and not only the depletion of natural resources.
The paper will finally conclude by highlighting the most important identified observations, together with an identification of the current shortcomings and the specific topics where further research is recommended.
2. Bioethanol: A Promising Biofuel
Within first (sugar-based), second (lignocellulosic-based) and third (algae-based) generation processes of bio-ethanol [19,20], only the first and second generations are today considered at the industrial and pilot scales, respectively. The traditional feedstocks used for first generation ethanol production are corn, wheat, cassava, sweet sorghum, molasses and sugarcane [21].
The feedstocks for the second generation are based on non-food, cheap and abundant plant waste biomass (agricultural and forest residue, grass, etc.) [22]. In this case, all parts of the plant such as leaves, bark, fruits, and seeds can be transformed into useful products.
Lignocellulosic biomass is the most abundant on earth [21,23]. Consequently, lignocellulosic biomass offers significant advantages over the first generation feedstock for ethanol production (e.g., do not compete with agricultural land needed for food and feed production, lower costs are involved, better engine performance is achieved, and new markers for the agricultural sector are stimulated) [22]. As can be seen in Table 1, the major fraction of such biomass materials is represented by cellulose, followed by hemicellulose and lignin [24,25].

Table 1.
The contents of cellulose, hemicellulose, and lignin in common lignocellulosic biomass [24,25].
| Cellulose % | Hemicellulose % | Lignin % | Others % | |
|---|---|---|---|---|
| Agricultural residues | 38 | 32 | 17 | 13 |
| Hardwoods | ~50 | ~5 | ~22 | ~5 (1) |
| MSW (Municipal Solid Waste) | 45 | 9 | 10 | 36 (2) |
| Waste papers from chemical pulps | 60–70 | 10–20 | 5–10 | - |
| Herbaceous biomass | 45 | 30 | 15 | 10 |
(1): including extractives ~5%; (2): including ash 15%, other carbohydrates 9%, protein 3%, others 9%.
First and second generation bio-ethanol productions follow the same flowsheet, with extra steps involved for lignocellulosic feedstock. This is illustrated in Figure 1 [26], and discussed below.
Ensuring a complete conversion can lead to a substantial increase of the ethanol yield [27]. Moreover, pretreatment methods represent a crucial step in the production of biofuel, since separating the major components of biomass (cellulose, hemicellulose and lignin) improves the digestibility of lignocellulosic material [28]. Pre-treatment involves delignification of the feedstock [24] in order to make cellulose more accessible in the hydrolysis step, using physical, physico-chemical, chemical and biological treatment. Sulphuric acid or carbon dioxide are often added in order to reduce the production of inhibitors and improve the solubilization of hemicellulose [29]. Hemicellulose is largely hydrolyzed, releasing different simple sugars but also other compounds of the cellulosic matrix that can, however, inhibit the enzymatic hydrolysis and fermentation. Pre-treatment is a costly separation, accounting for approximately 33% of the total cost [30]: the economy needs to be improved, and the release of microbial and chemical contamination that possibly reduces the overall yield needs further attention.
The conversion reaction for hexoses (C6) and pentoses (C5) is as follows:
The overall theoretical ethanol yield (at 20°C) hence becomes 0.719 and 0.736 liter per kg of glucan (or/and other 6C structures) and xylan (or/and other 5C structures), respectively. S. cerevisiae, the yeast commonly used for first generation ethanol production, cannot metabolize xylose. Other yeasts and bacteria are under investigation to ferment xylose and other pentoses into ethanol. Additional research tried to find microorganisms that can effectively ferment both types of sugars into ethanol with Escherichia coli, Klebisiella oxytoca and Zymomonas mobilis as promising candidates [31,32].
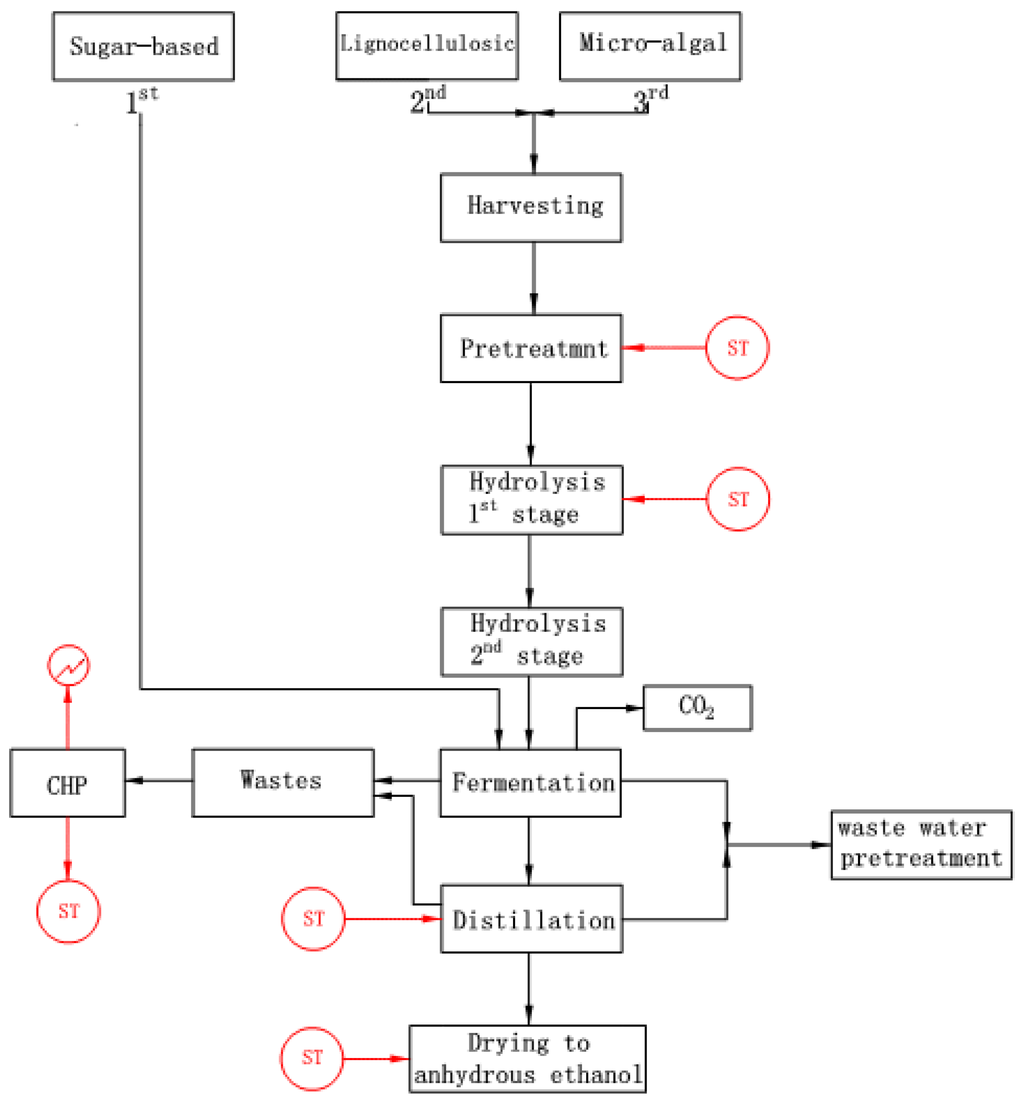
Figure 1.
Bio-ethanol production and potential membrane applications (ST: steam).
Process integration can be viewed as a key for reducing costs in the ethanol industry. Furthermore, the poly-generation that yields fuel, heat, and electricity can more effectively reduce the amount of waste produced in the process, minimize wasted heat, and increase the ethanol concentration in the feed to the distillation or otherwise minimize unwanted products [33].
Exergy analysis is viewed as a primary tool to analyze the ethanol production process from an integrated point of view [34,35]: it will identify the energy and exergy losses within the system, which decrease the system performance, and determine the most efficient process to convert biological matter to an energy carrier [33].
3. Exergy-Based Performance Analysis
Exergy is defined as “the maximum (theoretical) work that can be extracted from a mass or energy stream when it flows from a given thermodynamic state to one in chemical, mechanical and thermal equilibrium with the environment in a reversible way, interacting only with the components in the environment” [35] or in other words: “exergy is the maximum work that can be produced when a heat or material stream is brought to equilibrium in relation to a reference environment” [36]. The main objectives of an exergy analysis are [36,37]: (1) to identify the location, the source, and the magnitude of true thermodynamic inefficiencies in a given process; (2) to quantify the exergy losses in each process step; (3) to compare different process configurations.
A comprehensive literature review on exergy studies (>2005) is presented in Table 2.

Table 2.
Recent literature about exergy analysis of bio-ethanol production (>2005).
| Reference | Topics |
|---|---|
| [38] | Thermodynamic analysis and evaluation of bioethanol manufacture |
| [39] | Environmental, economic, and exergetic costs and benefits of biodiesel and ethanol biofuels |
| [40] | Exergetic analysis of biofuels production |
| [41] | Life-cycle analysis and the ecology of biofuels |
| [42] | Exergy analysis of enzymatic hydrolysis reactors for transformation of lignocellulosic biomass to bioethanol |
| [43] | The GHG (Greenhouse Gas) emissions of cellulosic ethanol supply chains in Europe |
| [35] | Exergy and renewability analysis of the ethanol production from banana fruit and its lignocellulosic residues |
| [36] | Energy and exergy analysis of an ethanol-fueled solid oxide fuel cell power plant |
| [44] | Second-generation bio-ethanol (SGB) from Malaysian palm empty fruit bunch: Energy and exergy analysis |
| [45] | Comparative exergy analysis of NREL (National Renewable Energy Laboratory)thermochemical biomass-to-ethanol conversion process designs |
| [46] | Improving bioethanol production from sugarcane: Evaluation of distillation, thermal integration and cogeneration systems |
| [47] | Exergy analysis and process integration of bioethanol production from acid pre-treated biomass: SHF (Saccharification hydrolysis and fermentation), SSF (Simultaneous saccharification and fermentation) and SSCF (Simultaneous saccharification and cofernentation) pathways |
| [48] | Sustainable ethanol production from lignocellulosic biomass-Application of exergy analysis |
| [49] | Thermodynamic analysis of lignocellulosic biofuel production via a biochemical process: technology selection and research focus |
| [33] | Comparison of combined ethanol and biogas polygeneration facilities using exergy analysis |
| [50] | Land-use change and GHG emissions from corn and cellulosic ethanol |
| [51] | Possibilities for sustainable biorefineries based on agricultural residues-potential straw-based ethanol production in Sweden |
| [52] | Energy and exergy analysis of the combined production process of sugar and ethanol from sugarcane |
| [53] | Comparing life cycle assessments of different biofuel options |
| [54] | Thermodynamic assessment of lignocellulosic pretreatment methods for bioethanol production via exergy analysis |
| [55] | Thermodynamic evaluation of biomass-to-biofuels production systems |
| [56] | Exergy analysis of pretreatment processes of bioethanol production based on sugarcane bagasse |
| [57] | Energy and exergy analysis of ethanol reforming process |
| [58] | Pinch and exergy analysis of lignocellulosic ethanol |
| [59] | An energy-, exergy-, and emergy-based thermodynamic system analysis of bioethanol |
| [60] | Life cycle analysis of biofuels under different environmental aspects |
Based upon this literature survey, the following conclusions can be drawn: (1) Most of the studies focus on specific parts of the process, e.g., water reduction and recycling, integrated energy supply using stillage and/or residual biomass, the use of co-generation systems (CHP (Combined heat and power), gasification, Rankine cycle); (2) Due to the energy-intensive nature of lignocellulosic ethanol production, different studies investigate improvements of the efficiency of ethanol production; (3) The production facilities (e.g., power, heat, lignocellulosic ethanol, and syngas (SNG)) are often integrated in poly-generation systems [61]; (4) The exergy analysis is usually applied to assess the efficiency of an integrated system; (5) Exergy analysis has been performed mainly for thermochemical pathways while fewer studies involve applications of exergy analysis to biochemical pathways [47,62]; (6) Understanding the complex structure of lignocellulose represents an important key to design a sustainable pretreatment process for the production of bioethanol [28].
4. Environmental Benefits
To be a sustainable alternative for fossil fuels, a biofuel should be economically viable, have environmental benefits by assuring energy security, reduce greenhouse emissions, and not compete with land use, food supplies, water consumption and biodiversity [39]. The interest for second generation biofuels gained a special focus in recent years [51,63].
A number of studies have focused on environmental impacts of various biofuels, but such studies are still rare for second generation biofuels at a commercial scale [51]. Based on a review of recent articles on biofuel LCA, Kendall and Yuan (2013) [53] concluded that LCAs of biofuels exhibit great variability and uncertainty, leading to inconclusive results for the performance of particular pathways. This is mainly caused by different processing and pretreatment strategies required for each raw material that has a different composition and may require different pretreatment methods [28].
Essential conclusions emerging from the literature are important: (1) Although concerns about first generation raw materials have drawn international attention due to the associated impacts, the majority of biofuels are still produced from food crops; (2) Although numerous studies address the conversion of cellulosic biomass to ethanol, the high costs involved, particularly at large-scale production, currently prohibit commercialization of such processes [28]; (3) Reports on environmental and energy benefits of biofuels are controversial and exhibit great variability and uncertainty [28,41]; (4) LCA studies attributed to biofuels have some limitations: production systems are inherently complex, different and subjective methods are used to quantify savings, the empirical data are limited, and impacts of, e.g., land use and topography should also be taken into account.
As solutions for some problems mentioned above, different authors suggested: (1) integration of the production of first and second generation ethanol as a more successful strategy [51]; (2) co-locating ethanol production, electricity generation and enzyme production in a single facility [43]; (3) the application of LCA in research and development for future fuel pathways [64]; (4) technological innovations on effective and low-cost enzymes, feedstocks and pretreatment methods [28,51].
5. The Exergy-Efficiency of the Process
Brazil produces ethanol from domestically grown sugar cane. The US feed stock is mostly corn and wheat [65]. In developing economies, sweet sorghum or cassava is preferably used as raw materials.The world’s first sweet sorghum-based ethanol production distillery began commercial ethanol production in 2007 in Andhra Pradesh, India [66]. Sorghum is now also an important feedstock for bio-ethanol production in the US Great Plains states. In France and Italy, grapes have become a feedstock for fuel ethanol from wine surplus. In Japan, it has been proposed to use rice, normally converted into sake. The world’s first large-scale cassava ethanol plant was built in Guangxi (China) by COFCO in 2007 [67,68]. The Canadian company Iogen started the first cellulose-based ethanol plant in 2004 [69]. Development of this technology could deal with a number of cellulose-containing agricultural byproducts, such as, e.g., straw, wood trimmings, sawdust, bamboo and others.
One of the key parameters that can be evaluated in an exergy analysis is the overall thermodynamic efficiency. The thermodynamic efficiency of a system is defined as:
In the case of lignocellulosic ethanol, the overall efficiency of ethanol production via the biochemical process can be written as:
Here, EX,bm is the input chemical exergy of biomass, ∑EX,ch is the sum of the chemical exergies of all inputs to the process, EX,LT is the exergy of a potential low temperature heat source supplied to the system, EX,et is the chemical exergy of ethanol, EX,res is the exergy of the lignin-enriched residue and Pnet is the net electricity produced by the system.
Exergy analysis can also be used to evaluate thermodynamic losses in each unit process of the system. The exergy balance applied to the system boundary of a unit operation of a process gives
where is the total input exergy flow, is the total output exergy flow in the products, is the total output exergy flow in the waste products from the unit processes and I is the exergy destruction due to internal irreversibility. The last two terms in the exergy balance represent the total exergy loss associated with the unit process. For an irreversible process, I ≠ 0 and Equation (3) express the fundamental property that, unlike energy, exergy is not conserved.
Applied to the widely studied test case of corn-based bioethanol, the exergy and CO2 emissions are illustrated in Table 3 and Table 4. The corn production aspect includes seeds, fertilizers, herbicides, insecticides and irrigation. Corn transportation involves fuel use only. The ethanol conversion includes all process steps from biomass delivery to anhydrous ethanol. The ethanol distribution to further processing again includes fuel only. Co-products involve processing steps that generate process waste-based energy. The efficiency η is calculated as the ratio of the lower heating value (LHV) of ethanol and the non-renewable exergy input.

Table 3.
The main four stages in ethanol production (in kJ/L).
| Production | Transportation | Ethanol Conversion | Ethanol Distribution | Total Non-Renewable | |
|---|---|---|---|---|---|
| Corn-1 | 5260 | 596 | 13,100 −7320 (Co-products) | 414 | 12,080 |
| Corn-2 | 10,500 | 1350 | 15,700 −1860 (Co-products) | 1380 | 27,140 |
With the LHV of ethanol as 21,200 kJ/L, the efficiency is 0.78 in the case of the Pimentel and Patzek (Corn-2) [70] assumptions, but 1.75 when using the Shapouri et al. (Corn-1) [64] values. The predictions by Shapouri et al. are far more optimistic than the predictions by Pimentel and Patzek [70], hence stressing the wide variability in basic data for exergy analysis.
Table 4 expresses the calculated CO2 values for each of the process steps, from corn cultivation to final product distribution.

Table 4.
Net CO2 emissions calculation results (g/MJ ethanol).
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | Total | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Equivalent CO2 emissions | 0.3 | 0.5 | 0.6 | 0.8 | 0.8 | 0.9 | 1 | 1.2 | 1.3 | 2 | 3 | 3.5 | 4 | 5.2 | 6.2 | 18 | 29.5 | 43.5 | ~120 |
1: P2O5; 2: KCl; 3: NG (Natural Gas); 4: Herbicides; 5: Transportation; 6: Electricity; 7:Gasoline; 8: Custom work; 9: LPG (Liquefied Petroleum Gas); 10: Seeds; 11: Diesel; 12: CaO; 13: Wastewater BOD (Biochemical Oxygen Demand); 14: Ethanol Plant Transport; 15: Machinery; 16: Nitrogen as Ammonia; 17: Humus Oxid. 18: Ethanol Plant Fuel.
Comparing the values with the CO2 emissions of common fossil fuels (Table 5), it is clear that the production of bio-ethanol from corn cannot be considered as CO2-advantageous.

Table 5.
CO2 emissions compared to other fuels (g/MJ in Fuel).
| NRR (Negative Regulator of Resistance) in Ethanol Corn Cycle | Diesel Fuel | Gasoline Fuel | Methane Fuel | ||
|---|---|---|---|---|---|
| Equivalent CO2 emissions | Fossil Fuels | 82 | 78 | 58 | |
| Corn Farming | 68 | ||||
| Ethanol Plant | 40 | ||||
| BOD Treatment | 12 | ||||
| Total | 120 | 82 | 78 | 58 |
This picture is positively affected if other raw materials are considered, e.g., cassava. Corn has a starch content of 66% only, whereas cassava contains 90% starch [71,72]. Since this starch is the real reaction component in the transformation to glucose and ethanol, conversion efficiencies are significantly higher, thus also reducing energy needs (and CO2 burden) in the ethanol plant fuel part of the analysis. Cassava, moreover, needs less cultivation aids and irrigation, thus reducing the production inputs. As a result, η considerably exceeds the corn value whereas the total CO2 balance is reduced by ≥50 g/MJ ethanol. Cassava-based bio-ethanol hence meets objectives of CO2 competitiveness and exergy efficiency.
For cassava, items 1 to 4, and 12, 16, and 17 of Table 4 are negligible. Whereas other items remain about the same, except for item 18 where heat recovery is the distillation, the use of Very High Fermentation and the use of hydrophilic membranes to prepare anhydrous ethanol reduce the fuel consumption and equivalent CO2 emissions to 50% of the corn-based example [26,73]. The CO2 emissions decrease to about 70 g/MJ ethanol, a reduction of 42%. For woody biomass, the situation is comparable with cassava, although the machinery and ethanol plant fuel will increase by 30%–40%.For lignocellulosic bio-ethanol, with its low conversion efficiency and widely variable raw materials, insufficient data are currently available to establish reliable exergy and CO2 balances. This will, however, soon change when pilot plant results of NREL (Golden, CO, USA), Iogen Corporation (Ottawa, ON, Canada) and ETEK (Viksjö, Sweden) are made available.
Cassava-based bioethanol is certainly approaching the current fossil fuel findings. The intensive pre-treatment required for sugarcane will negatively reflect in the exergy and CO2 efficiency.
6. Conclusions
The corn-based ethanol production has doubtful positive exergy results, and a negative CO2 balance. Improvements largely depend upon agricultural needs, upon the content of starch or glucose, and upon the energy integration of the system. Corn, with its 66% starch content, scores less than, e.g., cassava (90% starch), and cassava is also less demanding towards agricultural needs. This reflects in the cassava-based efficiency, η, which is largely in excess of 2, and a CO2 emission balance equivalent to the combustion of traditional fuels. Lignocellulosic-based ethanol requires additional data prior to establishing a reliable exergy and CO2 balance.
Acknowledgments
This work was supported in part by the National Basic Research Program of China (973 program) (2013CB733600, 2012CB725200), the National Nature Science Foundation of China (21436002, 21390202), the National High-Tech R&D Program of China (863 Program) (2012AA021404, 2014AA021903, 2014AA021904), the Key Projects in the National Science & Technology Pillar Program during the 12th Five-year Plan Period (2011BAD22B04).
Author Contributions
Both authors contributed equally to the research.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Wang, L.; Littlewood, J.; Murphy, R.J. An economic and environmental evaluation for bamboo-derived bioethanol. RSC Adv. 2014, 4, 29604–29611. [Google Scholar] [CrossRef]
- Piemonte, V.; Paola, L.D.; Russo, V. An LCA study on feedstocks and process for biofuels production. Chem. Eng. Trans. 2014, 37, 517–522. [Google Scholar]
- Wang, L.; Littlewood, J.; Murphy, R.J. Environmental Sustainability of bioethanol production from wheat straw in the UK. Renew. Sust. Energ. Rev. 2013, 28, 715–725. [Google Scholar] [CrossRef]
- Amores, M.J.; Mele, F.D.; Jiménez, L.; Castells, F. Life cycle assessment of fuel ethanol from sugarcane in Argentina. Int. J. Life Cycle Assess. 2013, 18, 1344–1357. [Google Scholar] [CrossRef]
- Borrion, A.L.; McManus, M.C.; Hammond, G.P. Environmental life cycle assessment for bioethanol production from wheat straw. Biomass Bioenerg. 2012, 47, 9–19. [Google Scholar] [CrossRef]
- Wiloso, E.I.; Heijungs, R.; de Snoo, G.R. LCA of second generation bioethanol: A review and some issues to be resolved for good LCA practice. Renew. Sust. Energ. Rev. 2012, 16, 5295–5308. [Google Scholar] [CrossRef]
- Gallejones, P.; Pardo, G.; Aizpurua, A.; del Prado, A. Life cycle assessment of first-generation biofuels using a nitrogen crop model. Sci. Total Environ. 2015, 505, 1191–1201. [Google Scholar] [CrossRef] [PubMed]
- Shadbahr, J.; Zhang, Y.; Khan, F. Life cycle assessment of bioethanol production from woodchips with modifications in the pretreatment process. Appl. Biochem. Biotech. 2014, 175, 1080–1091. [Google Scholar] [CrossRef] [PubMed]
- Saga, K.; Kawahara, T.; Sekino, H.; Lmou, K. LCA of small-scale ethanol production system from high-yield rice. J. Jpn. Inst. Eng. 2014, 93, 278–291. [Google Scholar] [CrossRef] [Green Version]
- Guo, M.; Li, C.; Facciotto, G.; Bergante, S.; Bhatia, R.; Comolli, R.; Ferré, C.; Murphy, R. Bioethanol from poplar clone Imola: An environmentally viable alternative to fossil fuel? Biotechnol. Biofuels 2015, 8, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Gnansounou, E.; Vaskan, P.; Pachón, E.R. Comparative techno-economic assessment and LCA of selected integrated sugarcane-based biorefineries. Bioresour. Technol. 2015, 196, 364–375. [Google Scholar] [CrossRef] [PubMed]
- McManus, M.C.; Taylor, C.M.; Mohr, A.; Whittaker, C.; Scown, C.D.; Borrion, A.L.; Glithero, N.; Yin, Y. Challenge clusters facing LCA in environmental decision-making-what we can learn from biofuels. Int. J. Life Cycle Assess. 2015, 20, 1399–1414. [Google Scholar] [CrossRef]
- Ren, J.; Manzardo, A.; Mazzi, A.; Zuliani, F.; Scipioni, A. Prioritization of bioethanol production pathways in China based of life cycle sustainability assessment and multicriteria decision-making. Int. J. Life Cycle Assess. 2015, 20, 842–853. [Google Scholar] [CrossRef]
- Morales, M.; Quintero, J.; Conejeros, R.; Aroca, G. Life cycle assessment of lignocellulosic bioethanol: Environmental impacts and energy balance. Renew. Sust. Energ. Rev. 2015, 42, 1349–1361. [Google Scholar] [CrossRef]
- Brunet, R.; Boer, D.; Guillén-Gosálbez, G.; Jiménez, L. Reducing the cost, environmental impact and energy consumption of biofuel processes through heat integration. Chem. Eng. Res. Des. 2015, 93, 203–212. [Google Scholar] [CrossRef]
- Miret, C.; Chazara, P.; Montastruc, L.; Negny, S.; Domenech, S. Design of bioethanol green supply chain: Comparison between first and second generation biomass concerning economic, environmental and social criteria. Comput. Chem. Eng. 2016, 85, 16–35. [Google Scholar] [CrossRef]
- Valle, C.R.; Perales, A.L.V.; Vidal-Barrero, F.; Ollero, P. Integrated economic and life cycle assessment of thermochemical production of bioethanol to reduce production cost by exploiting excess of greenhouse gas savings. Appl. Energ. 2015, 148, 466–475. [Google Scholar] [CrossRef]
- Dong, J.; Chi, Y.; Tang, Y.; Wang, F.; Huang, Q. Combined life cycle environmental and exergetic assessment of four typical sewage sludge treatment techniques in China. Energ. Fuel 2014, 28, 2114–2122. [Google Scholar] [CrossRef]
- Kang, Q.; Appels, L.; Baeyens, J.; Dewil, R.; Tan, T.W. Energy-Efficient Production of Cassava-Based Bio-Ethanol. Adv. Biosci. Biotechnol. 2014, 5, 925–939. [Google Scholar] [CrossRef]
- Kang, Q.; Appels, L.; Tan, T.W.; Dewil, R. Bioethanol from Lignocellulosic Biomass: Current Findings Determine Research Priorities. Sci. World J. 2014, 2014, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Singh, S.P.; Mishra, I.M.; Adhikari, D.K. Recent Advances in Production of Bioethanol from Lignocellulosic Biomass. Chem. Eng. Technol. 2009, 32, 517–526. [Google Scholar] [CrossRef]
- Naik, S.N.; Goud, V.V.; Rout, R.K.; Dalai, A.K. Production of first and second generation biofuels: A comprehensive review. Renew. Sust. Energ. Rev. 2010, 14, 578–597. [Google Scholar] [CrossRef]
- Giri, S.S.; Sen, S.S.; Sukumaran, V. Effects of dietary supplementation of potential probiotic Pseudomonas aeruginosa VSG-2 on the innate immunity and disease resistance. Fish Shellfish Immunol. 2012, 32, 1135–1140. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Cheng, J. Hydrolysis of lignocellulosic materials for ethanol production: A review. Bioresour. Technol. 2002, 83, 1–11. [Google Scholar] [CrossRef]
- Wyman, C.E. Ethanol from lignocellulosic biomass: Technology, economics, and opportunities. Bioresour. Technol. 1994, 50, 3–16. [Google Scholar] [CrossRef]
- Baeyens, J.; Kang, Q.; Appels, L.; Dewil, R.; Lv, Y.Q.; Tan, T.W. Challenges and opportunities in improving the production of bio-ethanol. Prog. Energ. Combust. Sci. 2015, 47, 60–88. [Google Scholar] [CrossRef]
- Gassner, M.; Maréchal, F. Thermo-economic Evaluation of the Thermochemical Production of Liquid Fuels from Biomass. In Proceedings of the 23rd International Conference on Efficiency, Cost, Optimization, Simulation and Environmental Impact of Energy Systerms (ECOS), Lausanne, Switzerland, 14–17 June 2010.
- Menon, V.; Rao, M. Trends in bioconversion of lignocellulose: Biofuels, platform chemicals & biorefinery concept. Prog. Energ. Combust. Sci. 2012, 38, 578–597. [Google Scholar]
- Hendriks, A.T.W.M.; Zeeman, G. Pretreatments to enhance the digestibility of lignocellulosic biomass. Bioresour. Technol. 2009, 100, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Tomas-Pejo, E.; Olivia, J.M.; Ballesteros, M. Realistic approach for full-scale bioethanol production from lignocelluloses. Rev. J. Sci. Ind. Res. 2008, 67, 874–884. [Google Scholar]
- Dien, B.S.; Cotta, M.A.; Jefferies, T.W. Bacteria engineered for fuel ethanol production: Current status. Appl. Microbiol. Biotechnol. 2003, 63, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Hahn-Hägerdal, B.; Karhumaa, H.B.K.; Fonseca, C.; Spencer-Martins, I.; Gorwa-Grauslund, M.F. Toward industrial pentose-fermenting yeast strains. Appl. Microbiol. Biotechnol. 2007, 74, 937–953. [Google Scholar] [CrossRef] [PubMed]
- Bösch, P.; Modarresi, A.; Friedl, A. Comparison of combined ethanol and biogas polygeneration facilities using exergy analysis. Appl. Therm. Eng. 2012, 37, 19–29. [Google Scholar]
- Kravanja, P.; Friedl, A. Process simulation of ethanol from straw-validation of scenarios for Austria. Chem. Eng. Trans. 2011, 25, 635–640. [Google Scholar]
- Arredondo, H.I.V.; Colorado, A.A.R.; Junior, S.D.O. Ethanol Production from Banana Fruit and its Lignocellulosic Residues: Exergy and Renewability Analysis. Int. J. Thermodyn. 2009, 12, 155–162. [Google Scholar]
- Casas, Y.; Arteaga, L.E.; Morales, M.; Rose, E.; Peralta, L.M.; Dewulf, J. Energy and exergy analysis of an ethanol fueled solid oxide fuel cell power plant. Chem. Eng. J. 2010, 162, 1057–1066. [Google Scholar] [CrossRef]
- Asprion, N.; Rumpf, B.; Gritsch, A. Work Flow In Process Development For Energy Efficient Processes. Appl. Therm. Eng. 2011, 31, 2067–2072. [Google Scholar] [CrossRef]
- Schulz, M.; Hebecker, D. Thermodynamische Analyse und Bewertung der Bioethanolherstellung. Chem. Ing. Tech. 2005, 77, 792–798. [Google Scholar] [CrossRef]
- Hill, J.; Nelson, E.; Tilman, D.; Polasky, S.; Tiffany, D. Environmental, economic, and energetic costs and benefits of biodiesel and ethanol biofuels. Proc. Natl. Acad. Sci. USA 2006, 103, 11206–11210. [Google Scholar] [CrossRef] [PubMed]
- Ptasinski, K.J. Thermodynamic efficiency of biomass gasification and biofuels conversion. Bioprod. Biorefin. 2008, 239–253. [Google Scholar] [CrossRef]
- Davis, S.C.; Anderson-Teixeira, K.; DeLucia, E.H. Life-cycle analysis and the ecology of biofuels. Trends Plant Sci. 2009, 14, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Ojeda, K.; Kafarov, V. Exergy analysis of enzymatic hydrolysis reactors for transformation of lignocellulosic biomass to bioethanol. Chem. Eng. J. 2009, 154, 390–395. [Google Scholar] [CrossRef]
- Slade, R.; Bauen, A.; Shah, N. The greenhouse gas emissions performance of cellulosic ethanol supply chains in Europe. Biotechnol. Biofuels 2009, 2, 50–52. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.T.; Lee, K.T.; Mohamed, A.R. Second-generation bio-ethanol (SGB) from Malaysian palm empty fruit bunch: Energy and exergy analyses. Bioresour. Technol. 2010, 101, 5719–5727. [Google Scholar] [CrossRef] [PubMed]
- Teh, K.Y.; Lutz, A.E.; Shaddix, C.R.; Dutta, A.; Biddy, M.; Aden, A. Comparative exergy analysis of NREL thermo-chemical biomass-to-ethanol process designs. In Proceedings of the ACS National Meeting & Exposition, San Francisco, CA, USA, 22–25 March 2010.
- Davis, M.O.S.; Modesto, M.; Ensinas, A.V.; Nebra, S.A.; Filho, R.M.; Rossell, C.E.V. Improving bioethanol production from sugarcane: Evaluation of distillation, thermal integration and cogeneration systems. Energy 2011, 36, 3691–3703. [Google Scholar]
- Ojeda, K.; Sánchez, E.; El-Hawagic, M.; Kafarov, V. Exergy analysis and process integration of bioethanol production from acid pre-treated biomass: Comparison of SHF, SSF and SSCF pathways. Chem. Eng. J. 2011, 176, 195–201. [Google Scholar] [CrossRef]
- Ojeda, K.; Sánchez, E.; Kafarov, V. Sustainable ethanol production from lignocellulosic biomass-pplication of exergy analysis. Energy 2011, 36, 2119–2128. [Google Scholar] [CrossRef]
- Sohel, M.I.; Jack, M.W. Thermodynamic analysis of lignocellulosic biofuel production via a biochemical process: Guiding technology selection and research focus. Bioresour. Technol. 2011, 102, 2617–2622. [Google Scholar] [CrossRef] [PubMed]
- Dunn, J.B.; Mueller, S.; Kwon, H.Y.; Wang, M.Q. Land-use change and greenhouse gas emissions from corn and cellulosic ethanol. Biotechnol. Biofuel 2013, 6, 204–205. [Google Scholar] [CrossRef] [PubMed]
- Ekman, A.; Wallberg, O.; Joelsson, E.; Börjesson, P. Possibilities for sustainable biorefineries based on agricultural residues—A case study of potential straw-based ethanol production in Sweden. Appl. Energ. 2013, 102, 299–308. [Google Scholar] [CrossRef]
- Héctor, I.V.A.; Carolina Mesa, M.; Sergio, A.G.A. Energy and exergy analysis of the combined production process of sugar and ethanol from sugarcane (a Colombian case study). In Proceedings of the International Conference on Efficiency, Cost, Optimization, Simulation and Environmental Impact of Energy Systerms (ECOS), Guilin, China, 15–19 July 2013.
- Kendall, A.; Yuan, J. Comparing life cycle assessments of different biofuel options. Curr. Opin. Chem. Biol. 2013, 17, 439–443. [Google Scholar] [CrossRef] [PubMed]
- Ofiori-Boateng, C.; Lee, K.T. Comparative thermodynamic sustainability assessment of lignocellulosic pretreatment methods for bioethanol production via exergy analysis. Chem. Eng. J. 2013, 228, 162–171. [Google Scholar] [CrossRef]
- Piekarczyk, W.; Czarnowska, L.; Ptasinski, K.; Stanek, W. Thermodynamic evaluation of biomass-to-biofuels production systems. Energy 2013, 62, 95–104. [Google Scholar] [CrossRef]
- Silva Ortiz, P.; de Oliveira, S. Exergy analysis of pretreatment processes of bioethanol production based on sugarcane bagasse. Energy 2014, 76, 130–138. [Google Scholar] [CrossRef]
- Tippawan, P.; Arpornwichanop, A. Energy and exergy analysis of an ethanol reforming process for solid oxide fuel cell applications. Bioresour. Technol. 2014, 157, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Moderassi, A.; Kravanja, P.; Friedl, A. Pinch and exergy analysis of lignocellulosic ethanol, biomethane, heat and power production from straw. Appl. Therm. Eng. 2012, 43, 20–28. [Google Scholar] [CrossRef]
- Liao, W.; Heijungs, R.; Huppes, G. Is bioethanol a sustainable energy source? An energy-, exergy-, and emergy-based thermodynamic system analysis. Renew. Energ. 2011, 36, 3479–3487. [Google Scholar] [CrossRef]
- Kaltschmitt, M.; Reinhardt, G.A.; Stelzer, T. Life cycle analysis of biofuels under different environmental aspects. Biomass Bioenerg. 1997, 12, 121–134. [Google Scholar] [CrossRef]
- Lythcke-JØrgensen, C.; Haglind, F.; Clausen, L.R. Exergy analysis of a combined heat and power plant with integrated lignocellulosic ethanol production. In Proceedings of the International Conference on Efficiency, Cost, Optimization, Simulation and Environmental Impact of Energy Systerms (ECOS), Guilin, China, 15–19 July 2013.
- Sohel, M.I.; Jack, M.W. Thermodynamic analysis of a lignocellulosic biorefinery based on a biochemical process. In Proceedings of the World Renewable Energy Congress, Linköping, Sweden, 7–13 May 2011.
- Ciubota-Rosie, C.; Gavrilescu, M.; Macoveanu, M. Biomass—An important renewable source of energy in Romania. Environ. Eng. Manag. J. 2008, 7, 559–568. [Google Scholar]
- Shapouri, H.; Duffield, J.A.; Wang, M. The Energy Balance of Corn Ethanol: An Update; Agricultural Economic Report 813; U.S. Department of Agriculture: Washington, DC, USA, 2002.
- Renewable Fuels Association, World Fuel Ethanol Production. Available online: http://www.ethanolrfa.org/resources/industry/statistics/ (accessed on 4 June 2014).
- Balat, M. Production of bioethanol from lignocellulosic materials via the biochemical pathway: A review. Energ. Convers. Manag. 2011, 52, 858–875. [Google Scholar] [CrossRef]
- Liu, S.H.; Liu, X.F. Technological development of non-grain based fuel ethanol production. Liquor. Mak. 2010, 37, 9–11. [Google Scholar]
- Jansson, C.; Westerbergh, A.; Zhang, J.; Hu, X.; Sun, C. Cassava, a potential biofuel crop in the People’s Republic of China. Appl. Energ. 2009, 86, 595–599. [Google Scholar] [CrossRef]
- Mousdale, D.M. Biofuels: Biotechnology, Chemistry and Sustainable Development; CRC Press, Taylor & Francis Group: New York, NY, USA, 2008. [Google Scholar]
- Pimentel, D.; Patzek, T.W. Ethanol Production Using Corn, Switchgrass, and Wood; Biodiesel Production Using Soybean and Sunflower. Nat. Resour. Res. 2005, 14, 65–76. [Google Scholar] [CrossRef]
- Zhang, H.L.; Baeyens, J.; Tan, T.W. The bubble-induced mixing in starch-to-ethanol fermenters. Chem. Eng. Res. Des. 2012, 90, 2122–2128. [Google Scholar] [CrossRef]
- Zhang, H.L.; Baeyens, J.; Tan, T.W. Mixing phenomena in a large-scale fermenter of starch to bio-ethanol. Energy 2012, 48, 380–391. [Google Scholar] [CrossRef]
- Kang, Q.; Huybrechts, J.; van der Bruggen, B.; Baeyens, J.; Tan, T.W.; Dewil, R. Hydrophilic membranes to replace molecular sieves in dewatering the bio-ethanol/water azeotropic mixture. Sep. Purif. Techonol. 2014, 136, 144–149. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).