Challenges of Sustainable and Commercial Aquaponics
Abstract
:1. Introduction
2. Principles of Aquaponics
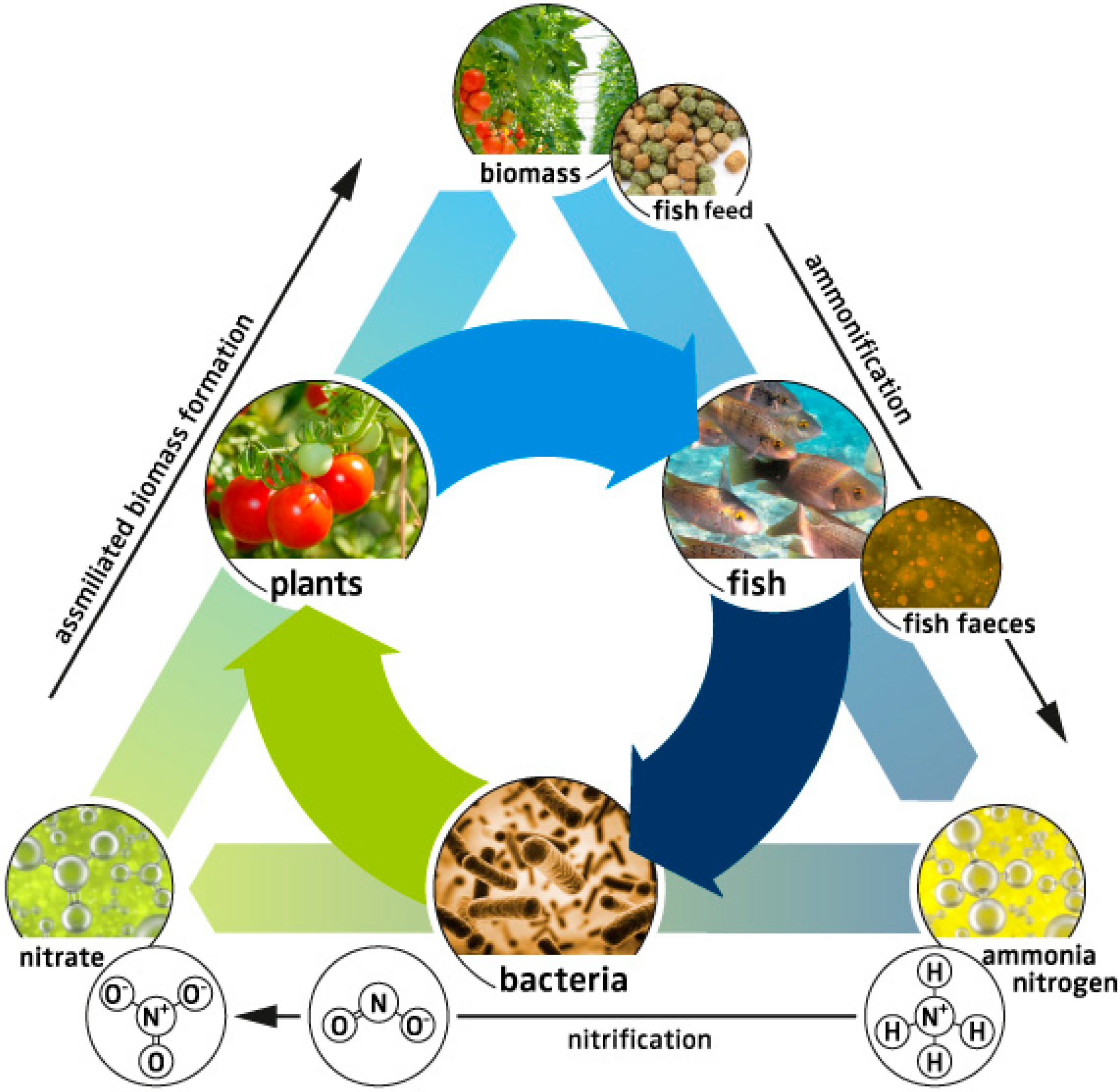
3. System Description

| Media-Based Growing Bed | DWC | NFT | Soil | |
|---|---|---|---|---|
| Advantages | - Biofiltration: media serves as substrate for nitrifying bacteria [32]; - Act as a solids filtering medium; - Mineralization in grow bed; - Colonized by a broad microflora | - Constant water flow; - Small sump tank needed; - Ease of maintenance and cleaning [33] | - Constant water flow - Small sump tank needed; - Ease of maintenance and cleaning; - Require smaller volume of water; - Light hydroponic infrastructure, suits well for roof farming | - Less infrastructure - Natural roots environment; - Colonized by broad microflora and fungi [34]; - Accepted as “organic way of production” |
| Disadvantages | - If flood and drain method: sizing and reliability plus large sump tank needed; - Heavy hydroponic infrastructure; - Maintenance and cleaning difficult; - Clogging leading to water channeling, inefficient biofiltration and inefficient nutrient delivery to plants [33] | - Separate biofilter needs to be added [32]; - Require large volume of water; - Heavy hydroponic infrastructure; - Device for roots aeration mandatory [35] | - Separate biofilter needed; - Lower yields (showed for lettuce by) [32]; - Expensive material; - the system is less stable as there is less water, | - Small control on the soil nutrient solution; - Good soil not available everywhere; - More vulnerable for diseases; - Lower basil and okra yield than in aquaponics [29] |
| Nutrient uptake | - High | - High | - Lower because smaller root-water contact area | - Lower |
| System A | System B | System C | System D | |
|---|---|---|---|---|
| System Type | Nutrient Film Technique (NFT) configured in the conveyor production system. | Deep Water Culture (DWC) | Deep Water Culture (DWC) | Deep Water Culture (DWC) |
| Source | Adler et al. [36] | Roosta and Hamidpour [37] | Rakocy et al. [24,38] | Endut et al. [39] |
| Location | The Conservation Fund’s Freshwater Institute, Shepherdstown, W. Va., USA | University of Rafsanjan, Iran | University of Virgin Islands, USA | University of Malaysia Terengganu |
| Based on | The system was theoretically valuated using data from studies conducted at the Conservation Fund’s Freshwater Institute during 1994 and 1995 [40] | UVI-System | Own setup (UVI-System) | Own Setup |
| Volume RAS (m3) | >38 | 0.848 | 43 | 3 |
| Size Hydroculture (m2) | 498 | Unknown (consisting of 8 plants) | 220 | 2 |
| Plant Density (pcs/m2) | 5.7 per meter of NFT trays | ND | 8 (basil); 2–4 (okra) | ND |
| Fish Density (kg/m3) | 113.4 | 17.69 (Common Carp), 23.58 (Grass Carp), 17.69 (Silver Carp) | 61.5–70.7 | ND |
| Daily feed input/plant growing area (g/day/m2) | ND | ND | 81.4–99.6 | 15–42 |
| Fish:Plant Ratio (kg) | ND | ND | ND | 1:8 |
| Plants Used | Basil (Ocimum basilicum); Lettuce (Lactuca sativa L. “Ostinata”) | Tomato (Lycopersicon esculentum) | Basil (Ocimum basilicum); Okra (Abelmoschus esculentus) | Spinach (Spinacia oleracea) |
| Fish Used | Rainbow Trout (Oncorhynchus mykiss) | Common Carp (Cyprinus carpio), Grass Carp (Ctenopharyngodon idella), Silver Carp (Hypophthalmichthys molitrix) | Nile Tilapia (Oreochromis niloticus L.) | African Catfish (Clarias gariepinus) |
| Hydroculture (Wet) Biomass (kg/m2) | ND | ND | 2 (basil); 2.9 (okra) | 1.16 |
| Biofiltration | Fluidized Sand Filter + Carbon Dioxide Strippers | Net Filter | Net Filter | Rapid Sand Filters |
| Mechanical Filtration | Drum filter | Clarifier plus Net Plastic Filter | Clarifier plus Net Plastic Filter | Rapid Sand Filters |
| Water Parameters (pH; °C) | pH 7.2; Temp: ND | pH 7.0–7.7; Temp: 25.7 °C | pH 7.0–7.5; Temp: 28 °C | pH 5.6–7.3; Temp: 27.5–28.8 °C |
| Temporal length of experiment | ND | 108 days | 28 weeks (basil); 11.7 weeks (okra) | 35 days |
| Cost of setup ($ U.S.) | $100,120 (hydroponic part) * | ND | ND | ND |
| Cost of annual running ($ U.S.) | $204,040 (lettuce); $194,950 (basil) | ND | $24,440 (tilapia + basil) | ND |
| Break-even price ($ U.S.) | $13.80 (per box of 24 lettuces); $0.53 (per basil plant) | ND | $3.23 (per kg of tilapia); $1.66 (per kg of basil) | ND |
| Potential annual profit ($ U.S.) | $12,350–$44,350 (for box of 24 lettuces sold at $14–$16); $27,750–$66,090 (for basil plant sold at $0.60–$0.70) | ND | $116,000 (for tilapia sold at $5.50/kg and basil sold at $22.50/kg) | ND |
4. Technical Challenges
4.1. pH Stabilization
4.2. Nutrient Balance
| Plant Species | System | pH | Ca | Mg | Na | K | TAN | NO3-N | PO4-P | SO4-S | Cl | Fe | Mn | Cu | Zn | B | Mo | Source |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lettuce (Lactuca sativa) | Hydroponic | 5–6.2 | 180 | 24 | 430 | 18 | 266 | 62 | 36 | 2.2 | 0.3 | 0.05 | 0.3 | 0.3 | 0.05 | Sonneveld and Voogt, 2009 [69] | ||
| Lettuce (Lactuca sativa) | Hydroponic | 200 | 50 | 50–90 | 210 | 190 | 50 | 66 | 65–253 | 5 | 0.5 | 0.15 | 0.15 | 0.3 | 0.05 | Resh, 2012 [23] | ||
| Lettuce (Lactuca sativa) | Aquaponic | 8 | 48 | 20 | 10 | Al-Hafedh et al., 2008 [70] | ||||||||||||
| Lettuce (Lactuca sativa) | Aquaponic | 180 | 44 | 17 | 106 | 137 | 9 | Pantanella et al., 2012 [71] | ||||||||||
| Basil (Ocimum basilicum ‘Genovese’) | Aquaponic | 7.4 | 12 | 7 | 45 | 2.20 | 42 | 8 | 2.5 | 0.8 | 0.05 | 0.44 | 0.19 | 0.01 | Rakocy et al., 2004 [24] | |||
| Water spinach (Ipomoea aquatica) | Aquaponic | 5.6–7.3 | 20 | 17 | Endut et al.. 2010 [31] | |||||||||||||
| Tomato (Solanum lycopersicum) | Hydroponic | 5-6.2 | 110 | 24 | 254 | 18 | 151 | 39 | 48 | 0.8 | 0.6 | 0.05 | 0.3 | 0.2 | 0.05 | Sonneveld and Voogt, 2009 [69] | ||
| Tomato (Solanum lycopersicum) | Aquaponic | 7.7 | 34 | 27 | 0.33 | 35 | 8 | 0.2 | 0.04 | 0.37 | Roosta and Hamidpour, 2011 [37] | |||||||
| Okra (Abelmoschus esculentus) | Aquaponic | 7.1 | 24 | 6 | 14 | 64 | 1.58 | 26 | 15 | 6 | 12 | 1.3 | 0.06 | 0.03 | 0.34 | 0.09 | 0.01 | Rakocy et al., 2004 [38] |
4.3. Phosphorous
4.4. Pest and Disease Management
4.5. Other Technical Challenges
5. Socio-Ecological Challenges
5.1. Mineral Recycling
5.2. Water
5.3. Energy
5.4. Overfishing
5.5. Urban Farming and Short Supply Chains
6. Economic Challenges
7. Education as a Necessity
8. Discussion
9. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rakocy, J.E. Island Perspectives; Virgin Islands agricultural Experiment Station: Saint Croix, VI, USA, 1989; pp. 5–10. [Google Scholar]
- Turcios, A.E.; Papenbrock, J. Sustainable Treatment of Aquaculture Effluents-What Can We Learn from the Past for the Future? Sustainability 2014, 6, 836–856. [Google Scholar] [CrossRef]
- Coche, A.G. Fish culture in rice fields a world-wide synthesis. Hydrobiologia 1967, 30, 1–44. [Google Scholar] [CrossRef]
- Love, D.C.; Fry, J.P.; Genello, L.; Hill, E.S.; Frederick, J.A.; Li, X.; Semmens, K. An international survey of aquaponics practitioners. PLoS One 2014, 9, e102662. [Google Scholar] [CrossRef] [PubMed]
- Diver, S.; Rinehart, L. Aquaponics—Integration of Hydroponics with Aquaculture; ATTRA NCAT: Butte, MT, USA, 2010; p. 28. [Google Scholar]
- UN. Human development report 2013. Available online: http://hdr.undp.org/en/media/HDR_2013_EN_complete.pdf (accessed on 13 August 2014).
- Alexandratos, N.; Bruinsma, J. WORLD AGRICULTURE TOWARDS 2030/2050: The 2012 Revision; FAO, Agricultural Development Economics Division: Rome, Italy, 2012. [Google Scholar]
- Bindraban, P.S.; van der Velde, M.; Ye, L.; van den Berg, M.; Materechera, S.; Kiba, D.I.; Tamene, L.; Ragnarsdottir, K.V.; Jongschaap, R.; Hoogmoed, M.; et al. Assessing the impact of soil degradation on food production. Curr. Opin. Environ. Sustain. 2012, 4, 478–488. [Google Scholar] [CrossRef]
- Klinger, D.; Naylor, R. Searching for Solutions in Aquaculture: Charting a Sustainable Course. Annu. Rev. Environ. Resour. 2012, 37, 247–276. [Google Scholar] [CrossRef]
- Lehman, H.; Clark, E.A.; Weise, S.F. Clarifying the definition of Sustainable agriculture. J. Agric. Environ. Ethics 1993, 6, 127–143. [Google Scholar] [CrossRef]
- Francis, C.; Lieblein, G.; Gliessman, S.; Breland, T.A.; Creamer, N.; Harwood, R.; Salomonsson, L.; Helenius, J.; Rickerl, D.; Salvador, R.; et al. Agroecology: The Ecology of Food Systems. J. Sustain. Agric. 2003, 22, 99–118. [Google Scholar] [CrossRef]
- Ragnarsdottir, K.V.; Sverdrup, H.U.; Koca, D. Challenging the planetary boundaries I: Basic principles of an integrated model for phosphorous supply dynamics and global population size. Appl. Geochem. 2011, 26, S303–S306. [Google Scholar] [CrossRef]
- Sonneveld, C.; Voogt, W. Plant Nutrition in Future Greenhouse Production. In Plant Nutrition of Greenhouse Crops; Springer: Heidelberg, The Netherlands, 2009; pp. 393–403. [Google Scholar]
- Sverdrup, H.U.; Ragnarsdottir, K.V. Challenging the planetary boundaries II: Assessing the sustainable global population and phosphate supply, using a systems dynamics assessment model. Appl. Geochem. 2011, 26, S307–S310. [Google Scholar] [CrossRef]
- Gagnon, V.; Maltais-Landry, G.; Puigagut, J.; Chazarenc, F.; Brisson, J. Treatment of hydroponics wastewater using constructed wetlands in winter conditions. Water. Air. Soil Pollut. 2010, 212, 483–490. [Google Scholar] [CrossRef]
- Institution of Mechanical Engineers (IME). Global Food: Waste Not, Want Not; IME: London, UK, 2013; p. 31. [Google Scholar]
- Al-Hafedh, Y.S.; Alam, A.; Alam, M.A. Performance of plastic biofilter media with different configuration in a water recirculation system for the culture of Nile tilapia (Oreochromis niloticus). Aquac. Eng. 2003, 29, 139–154. [Google Scholar] [CrossRef]
- Dalsgaard, J.; Lund, I.; Thorarinsdottir, R.; Drengstig, A.; Arvonen, K.; Pedersen, P.B. Farming different species in RAS in Nordic countries: Current status and future perspectives. Aquac. Eng. 2013, 53, 2–13. [Google Scholar] [CrossRef]
- Martins, C.I.M.; Eding, E.H.; Verdegem, M.C.J.; Heinsbroek, L.T.N.; Schneider, O.; Blancheton, J.P.; D’Orbcastel, E.R.; Verreth, J.A.J. New developments in recirculating aquaculture systems in Europe: A perspective on environmental sustainability. Aquac. Eng. 2010, 43, 83–93. [Google Scholar] [CrossRef]
- Van Rijn, J. Waste treatment in recirculating aquaculture systems. Aquac. Eng. 2013, 53, 49–56. [Google Scholar] [CrossRef]
- Rakocy, J.E. Aquaponics—Integrating Fish and Plant Culture; Wiley-Blackwell: Hoboken, NJ, USA, 2012; pp. 344–386. [Google Scholar]
- Vermeulen, T.; Kamstra, A. The need for systems design for robust aquaponic systems in the urban environment. 2013, 1004, 71–78. [Google Scholar]
- Resh, H.M. Hydroponic Food Production: A Definitive Guidebook for the Advanced Home Gardener and the Commercial Hydroponic Grower. CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Rakocy, J.E.; Shultz, R.C.; Bailey, D.S.; Thoman, E.S. Aquaponic production of tilapia and basil: Comparing a batch and staggered cropping system. Acta Hortic. 2004, 648, 63–69. [Google Scholar]
- Damon, E.; Seawright, R.B.; Walker, R.R.S. Nutrient dynamics in integrated aquaculture-hydroponics systems. Aquaculture 1998, 160, 215–237. [Google Scholar] [CrossRef]
- Tyson, R.V.; Simonne, E.H.; Treadwell, D.D.; White, J.M.; Simonne, A. Reconciling pH for ammonia biofiltration and cucumber yield in a recirculating aquaponic system with perlite biofilters. HortScience 2008, 43, 719–724. [Google Scholar]
- Endut, A.; Jusoh, A.; Ali, N. Nitrogen budget and effluent nitrogen components in aquaponics recirculation system. Desalin. Water Treat. 2014, 52, 744–752. [Google Scholar] [CrossRef]
- Graber, A.; Junge, R. Aquaponic Systems: Nutrient recycling from fish wastewater by vegetable production. Desalination 2009, 246, 147–156. [Google Scholar] [CrossRef]
- Rakocy, J.E.; Masser, M.P.; Losordo, T.M. Recirculating Aquaculture Tank Production Systems: Aquaponics—Integrating Fish and Plant Culture; Southern Regional Aquaculture Center: Stoneville, MS, USA, 2006; pp. 1–16. [Google Scholar]
- Rakocy, J.E. Ten Guidelines for Aquaponic Systems. Aquaponics J. 2007, 1, 14–17. [Google Scholar]
- Endut, A.; Jusoh, A.; Ali, N.; Wan Nik, W.B.; Hassan, A. A study on the optimal hydraulic loading rate and plant ratios in recirculation aquaponic system. Bioresour. Technol. 2010, 101, 1511–1517. [Google Scholar] [CrossRef] [PubMed]
- Lennard, W.A.; Leonard, B.V. A Comparison of Three Different Hydroponic Sub-systems (gravel bed, floating and nutrient film technique) in an Aquaponic Test System. Aquac. Int. 2006, 14, 539–550. [Google Scholar] [CrossRef]
- Rakocy, J.A.; Hargreaves, J.E. Integration of vegetable hydroponics with fish culture: A review. In Techniques for Modern Aquaculture, Proceedings Aquacultural Engineering Conference; Wang, J.K., Ed.; American Society for Agricultural Engineers: St. Joseph, MI, USA, 1993; pp. 112–136. [Google Scholar]
- Bulgarelli, D.; Schlaeppi, K.; Spaepen, S.; van Themaat, E.V.L.; Schulze-Lefert, P. Structure and Functions of the Bacterial Microbiota of Plants. Annu. Rev. Plant Biol. 2013, 64, 807–838. [Google Scholar] [CrossRef] [PubMed]
- Nicola, S.; Hoeberechts, J.; Fontana, E. Ebb-and-Flow and Floating systems to grow leafy vegetables: A review for rocket, corn salad, garden cress and purslane. Acta Hortic 2007, 747, 585–592. [Google Scholar]
- Adler, P.R.; Harper, J.K.; Wade, E.M. Economic analysis of an aquaponic system for the integrated production of rainbow trout and plants. Int. J. 2000, 1, 15–34. [Google Scholar]
- Roosta, H.R.; Hamidpour, M. Effects of foliar application of some macro- and micro-nutrients on tomato plants in aquaponic and hydroponic systems. Sci. Hortic. 2011, 129, 396–402. [Google Scholar] [CrossRef]
- Rakocy, J.E.; Bailey, D.S.; Shultz, R.C.; Thoman, E.S. Update on tilapia and vegetable production in the UVI aquaponic system. In New Dimensions on Farmed Tilapia, Proceedings of the 6th International Symposium on Tilapia in Aquaculture, Manila, Philippines, 12–16 September 2004; Volume 6, pp. 1–15.
- Endut, A.; Jusoh, A.; Ali, N.; Nik, W.B.W. Nutrient removal from aquaculture wastewater by vegetable production in aquaponics recirculation system. Desalin. Water Treat. 2011, 32, 422–430. [Google Scholar] [CrossRef]
- Adler, P.R.; Summerfelt, S.T.; Glenn, D.M.; Takeda, F. Evaluation of the Effect of a Conveyor Production Strategy on Lettuce and Basil Productivity and Phosphorus Removal from Aquaculture Wastewater. In Recycling the Resource Ecological Engineering for Wastewater Treatment. Proceedings of the Second International Conference, Switzerland; Environmental Research Forum, Ed.; Trans Tech Publications Ltd: Zurich, 1996; pp. 131–136. [Google Scholar]
- Adler, P.R.; Harper, J.K.; Takeda, F.; Wade, E.M.; Summerfelt, S.T. Economic Evaluation of Hydroponics and Other Treatment Options for Phosphorus Removal in Aquaculture Effluent. HortScience 2000, 35, 993–999. [Google Scholar]
- Love, D.C.; Fry, J.P.; Li, X.; Hill, E.S.; Genello, L.; Semmens, K.; Thompson, R.E. Ommercial aquaponics production and profitability: Findings from an international survey. Aquaculture 2015, 435, 67–74. [Google Scholar] [CrossRef]
- McAndrew, B.J.; Turner, G.F.; Robinson, R.L.; Beveridge, M.C.M.; Baird, D.J.; Ross, L.G.; Lowe-McConnell, R.H.; Lorenzen, K.; Penman, D.J.; Little, D.C.; et al. Tilapias: Biology and Exploitation; Beveridge, M.C.M., McAndrew, B.J., Eds.; Springer: Heidelberg, The Netherlands, 2000; p. 508. [Google Scholar]
- Villaverde, S. Influence of pH over nitrifying biofilm activity in submerged biofilters. Water Res. 1997, 31, 1180–1186. [Google Scholar] [CrossRef]
- Antoniou, P.; Hamilton, J.; Koopman, B.; Jain, R.; Holloway, B.; Lyberatos, G.; Svoronos, S.A. Effect of temperature and ph on the effective maximum specific growth rate of nitrifying bacteria. Water Res. 1990, 24, 97–101. [Google Scholar] [CrossRef]
- Keen, G.A.; Prosser, J.I. Interrelationship between pH and surface growth of Nitrobacter. Soil Biol. Biochem. 1987, 19, 665–672. [Google Scholar] [CrossRef]
- Hatayama, R.; Takahashi, R.; Ohshima, M.; Shibasaki, R.; Tokuyama, T. Ribulose-1,5-bisphosphate carboxylase/oxygenase from an ammonia-oxidizing bacterium, Nitrosomonas sp. K1: Purification and properties. J. Biosci. Bioeng. 2000, 90, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Blackburne, R.; Vadivelu, V.M.; Yuan, Z.; Keller, J. Kinetic characterisation of an enriched Nitrospira culture with comparison to Nitrobacter. Water Res. 2007, 41, 3033–3042. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, D.E.; Lamb, J.A.; Bloom, P.R. Managing Iron Deficiency Chlorosis in Soybean; Regents of the University of Minnesota: Saint Paul, MN, USA, 2011. [Google Scholar]
- Sverdrup, H.; Eklund, H.; Bjerle, I. Kalkning av rinnande vatten-Erfarenheter från en fluidiserad kalkbrunn. Mover. Vatten 1981, 37, 388–394. [Google Scholar]
- Gelfand, I.; Barak, Y.; Even-Chen, Z.; Cytryn, E.; van Rijn, J.; Krom, M.D.; Neori, A. A novel zero discharge intensive Seawater recirculating system for the culture of marine fish. J. World Aquac. Soc. 2003, 34, 344–358. [Google Scholar] [CrossRef]
- Neori, A.; Krom, M.D.; van Rijn, J. Biogeochemical processes in intensive zero-effluent marine fish culture with recirculating aerobic and anaerobic biofilters. J. Exp. Mar. Bio. Ecol. 2007, 349, 235–247. [Google Scholar] [CrossRef]
- Neto, R.M.; Ostrensky, A. Nutrient load estimation in the waste of Nile tilapia Oreochromis niloticus (L.) reared in cages in tropical climate conditions. Aquac. Res. 2013. [Google Scholar] [CrossRef]
- Chen, S.; Ling, J.; Blancheton, J.P. Nitrification kinetics of biofilm as affected by water quality factors. Aquac. Eng. 2006, 34, 179–197. [Google Scholar] [CrossRef]
- Lekang, O.-I.; Kleppe, H. Efficiency of nitrification in trickling filters using different filter media. Aquac. Eng. 2000, 21, 181–199. [Google Scholar] [CrossRef]
- Krom, M.D.; Ben David, A.; Ingall, E.D.; Benning, L.G.; Clerici, S.; Bottrell, S.; Davies, C.; Potts, N.J.; Mortimer, R.J.G.; van Rijn, J. Bacterially mediated removal of phosphorus and cycling of nitrate and sulfate in the waste stream of a “zero-discharge” recirculating mariculture system. Water Res. 2014, 56, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Cripps, S.J.; Bergheim, A. Solids management and removal for intensive land-based aquaculture production systems. Aquac. Eng. 2000, 22, 33–56. [Google Scholar] [CrossRef]
- Jung, I.S.; Lovitt, R.W. Leaching techniques to remove metals and potentially hazardous nutrients from trout farm sludge. Water Res. 2011, 45, 5977–5986. [Google Scholar] [CrossRef] [PubMed]
- Bajsa, O.; Nair, J.; Mathew, K.; Ho, G.E. Vermiculture as a tool for domestic wastewater management. Water Sci. Technol. 2003, 48, 125–132. [Google Scholar] [PubMed]
- Qi, Y. Vermiculture Technology: Earthworms, Organic Wastes, and Environmental Management. Int. J. Environ. Stud. 2012, 69, 173–174. [Google Scholar] [CrossRef]
- Torri, S.I.; Puelles, M.M. Use of vermiculture technology for waste management and environmental remediation in Argentina. Int. J. Glob. Environ. Issues 2010, 10, 239–254. [Google Scholar] [CrossRef]
- Jorgensen, B.; Meisel, E.; Schilling, C.; Swenson, D.; Thomas, B. Developing food production systems in population centers. Biocycle 2009, 50, 27–29. [Google Scholar]
- Martins, C.I.M.; Eding, E.H.; Verreth, J.A.J. The effect of recirculating aquaculture systems on the concentrations of heavy metals in culture water and tissues of Nile tilapia Oreochromis niloticus. Food Chem. 2011, 126, 1001–1005. [Google Scholar] [CrossRef]
- Gilbert, N. Environment: The disappearing nutrient. Nature 2009, 461, 716–718. [Google Scholar] [CrossRef] [PubMed]
- Amadori, M.; Daley, D. An Engineered Ecosystem for Waste Management and Food Production. Master’s Thesis, State University of New York College of Environmental Science and Forestry, Syracuse, NY, USA, 2012; p. 44. [Google Scholar]
- Mandal, R.N.; Datta, A.K.; Sarangi, N.; Mukhopadhyay, P.K. Diversity of aquatic macrophytes as food and feed components to herbivorous fish-A review. Indian J. Fish. 2010, 57, 65–73. [Google Scholar]
- Van Huis, A. Potential of Insects as Food and Feed in Assuring Food Security. Annu. Rev. Entomol. 2011, 58, 563–583. [Google Scholar] [CrossRef]
- Böhme, M. Effects of lactate, humate and bacillus subtilis on the growth of tomato plants in hydroponic systems. Acta Hortic. 1999, 481, 231–239. [Google Scholar]
- Sonneveld, C.; Voogt, W. Nutrient Management in Substrate Systems. In Plant Nutrition of Greenhouse Crops; Springer: Heidelberg, The Netherlands, 2009; pp. 277–312. [Google Scholar]
- Al-Hafedh, Y.S.; Alam, A.; Beltagi, M.S. Food Production and Water Conservation in a Recirculating Aquaponic System in Saudi Arabia at Different Ratios of Fish Feed to Plants. J. World Aquac. Soc. 2008, 39, 510–520. [Google Scholar] [CrossRef]
- Pantanella, E.; Cardarelli, M.; Colla, G.; Rea, E.; Marcucci, A. Aquaponics vs. Hydroponics: Production and Quality of Lettuce Crop. Acta Hortic. 2012, 927, 887–893. [Google Scholar]
- Lennard, W.; Leonard, B. A comparison of reciprocating flow versus constant flow in an integrated, gravel bed, aquaponic test system. Aquac. Int. 2005, 12, 539–553. [Google Scholar] [CrossRef]
- Nichols, M.; Lennard, W. Aquaponics in New Zealand. Pract. Hydroponics Greenh. 2010, 115, 46–51. [Google Scholar]
- Savidov, N.A.; Hutchings, E.; Rakocy, J.E. Fish and plant production in a recirculating aquaponic system: A new approach to sustainable agriculture in Canada. 2007, 742, 209–222. [Google Scholar]
- Savidov, N. Evaluation and Development or Aquaponics Production and Product Market Capabilities in Alberta. Available online: http://www.ecogrow.ca/pdf/CDC_Report_Phase_II.pdf (accessed on 10 July 2014).
- Voogt, W. Potassium Management of Vegetables Under Intensive Growth Conditions; Pasricha, N.S., Bansal, S.K., Eds.; The International Potash Institute: Bern, Switzerland, 2002; pp. 347–362. [Google Scholar]
- De Kreij, C.; Voogt, W.; Baas, R. Nutrient Solutions and Water Quality for Soilless Cultures; Brochure; Research Station for Floriculture and Glasshouse Vegetables (PBG): Naaldwijk, The Netherlands, 1999; Volume 196. [Google Scholar]
- López-Arredondo, D.L.; Leyva-González, M.A.; Alatorre-Cobos, F.; Herrera-Estrella, L. Biotechnology of nutrient uptake and assimilation in plants. Int. J. Dev. Biol. 2013, 57, 595–610. [Google Scholar] [CrossRef] [PubMed]
- Schneider, O.; Sereti, V.; Eding, E.H.; Verreth, J.A.J. Analysis of nutrient flows in integrated intensive aquaculture systems. Aquac. Eng. 2005, 32, 379–401. [Google Scholar] [CrossRef]
- Villarroell, M.; Alvarino, J.M.R.; Duran, J.M. Aquaponics: Integrating fish feeding rates and ion waste production for strawberry hydroponics. Spanish J. Agric. Res. 2011, 9, 537–545. [Google Scholar]
- Sikawa, D.C.; Yakupitiyage, A. The hydroponic production of lettuce (Lactuca sativa L) by using hybrid catfish (Clarias macrocephalus × C. gariepinus) pond water: Potentials and constraints. Agric. Water Manag. 2010, 97, 1317–1325. [Google Scholar] [CrossRef]
- Nichols, M.A.; Savidov, N.A. Aquaponics: A nutrient and water efficient production system. Acta Hortic. 2012, 947, 129–132. [Google Scholar]
- Cordell, D.; Rosemarin, A.; Schröder, J.J.; Smit, A.L. Towards global phosphorus security: A systems framework for phosphorus recovery and reuse options. Chemosphere 2011, 84, 747–758. [Google Scholar] [CrossRef] [PubMed]
- Shu, L.; Schneider, P.; Jegatheesan, V.; Johnson, J. An economic evaluation of phosphorus recovery as struvite from digester supernatant. Bioresour. Technol. 2006, 97, 2211–2216. [Google Scholar] [CrossRef] [PubMed]
- Jijakli, H.M. Pichia anomala in biocontrol for apples: 20 years of fundamental research and practical applications. Antonie Van Leeuwenhoek 2011, 99, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Saraf, M.; Pandya, U.; Thakkar, A. Role of allelochemicals in plant growth promoting rhizobacteria for biocontrol of phytopathogens. Microbiol. Res. 2014, 169, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Kouassi, K.H.S.; Bajji, M.; Jijakli, H. The control of postharvest blue and green molds of citrus in relation with essential oil–wax formulations, adherence and viscosity. Postharvest Biol. Technol. 2012, 73, 122–128. [Google Scholar] [CrossRef]
- Lang, G.; Buchbauer, G. A review on recent research results (2008–2010) on essential oils as antimicrobials and antifungals. A review. Flav. Fragr. J. 2012, 27, 13–39. [Google Scholar] [CrossRef]
- Fujiwara, K.; Iida, Y.; Iwai, T.; Aoyama, C.; Inukai, R.; Ando, A.; Ogawa, J.; Ohnishi, J.; Terami, F.; Takano, M.; et al. The rhizosphere microbial community in a multiple parallel mineralization system suppresses the pathogenic fungus Fusarium oxysporum. Microbiologyopen 2013, 2, 997–1009. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, K.; Aoyama, C.; Takano, M.; Shinohara, M. Suppression of Ralstonia solanacearum bacterial wilt disease by an organic hydroponic system. J. Gen. Plant Pathol. 2012, 78, 217–220. [Google Scholar] [CrossRef]
- Crinnion, W.J. Organic foods contain higher levels of certain nutrients, lower levels of pesticides, and may provide health benefits for the consumer. Altern. Med. Rev. 2010, 15, 4–12. [Google Scholar] [PubMed]
- Davidson, J.; Good, C.; Welsh, C.; Summerfelt, S.T. Comparing the effects of high vs. low nitrate on the health, performance, and welfare of juvenile rainbow trout Oncorhynchus mykiss within water recirculating aquaculture systems. Aquac. Eng. 2014, 59, 30–40. [Google Scholar] [CrossRef]
- Schram, E.; Roques, J.A.C.; Abbink, W.; Yokohama, Y.; Spanings, T.; de Vries, P.; Bierman, S.; van de Vis, H.; Flik, G. The impact of elevated water nitrate concentration on physiology, growth and feed intake of African catfish Clarias gariepinus (Burchell 1822). Aquac. Res. 2012, 45, 1499–1511. [Google Scholar] [CrossRef]
- Webster, C.D.; Lim, C. Tilapia; CRC-Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Kampschreur, M.J.; Temmink, H.; Kleerebezem, R.; Jetten, M.S.M.; van Loosdrecht, M.C.M. Nitrous oxide emission during wastewater treatment. Water Res. 2009, 43, 4093–4103. [Google Scholar] [CrossRef] [PubMed]
- Losordo, T.M.; Masser, M.P.; Rakocy, J.E. Recirculating Aquaculture Tank Production Systems-A Review of Component Options; Southern Regional Aquaculture Center: Stoneville, MS, USA, 1999. [Google Scholar]
- Timmons, M.B.; Ebeling, J.M. Recirculating Aquaculture, 3rd ed.; Ithaca Publishing Company LLC: Ithaca, NY, USA, 2013. [Google Scholar]
- Stark, J.M. Modeling the temperature response of nitrification. Biogeochemistry 1996, 35, 433–445. [Google Scholar] [CrossRef]
- Zhu, S.; Chen, S. The impact of temperature on nitrification rate in fixed film biofilters. Aquac. Eng. 2002, 26, 221–237. [Google Scholar] [CrossRef]
- World Water Assessment Programme (WWAP). The United Nations World Water Development Report 4: Managing Water under Uncertainty and Risk; United Nations Educational: Paris, France, 2012; p. 380. [Google Scholar]
- FAO. AQUASTAT Survey, Irrigation in Africa in Figures; FAO: Rome, Italy, 2005. [Google Scholar]
- Bernstein, S. Aquaponic Gardening: A Step-by-Step Guide to Raising Vegetables and Fish Together; New Society Publishers: Gabriola Island, BC, Canada, 2011; p. 256. [Google Scholar]
- Fraunhofer Gesellschaft. Research News; Fraunhofer-Gesellschaft München, Fraunhofer Press: München, Germany, 2009. [Google Scholar]
- Duriau, Y. Desalination by reverse osmosis. Desalination 1968, 5, 120–121. [Google Scholar] [CrossRef]
- Greenlee, L.F.; Lawler, D.F.; Freeman, B.D.; Marrot, B.; Moulin, P. Reverse osmosis desalination: Water sources, technology, and today’s challenges. Water Res. 2009, 43, 2317–2348. [Google Scholar] [CrossRef] [PubMed]
- Chan, H.-Y.; Riffat, S.B.; Zhu, J. Review of passive solar heating and cooling technologies. Renew. Sustain. Energy Rev. 2010, 14, 781–789. [Google Scholar] [CrossRef]
- Bakos, G.C.; Fidanidis, D.; Tsagas, N.F. Greenhouse heating using geothermal energy. Geothermics 1999, 28, 759–765. [Google Scholar] [CrossRef]
- Ragnarsson, Á. Utilization of geothermal energy in Iceland. In Proceedings of the International Geothermal Conference, Reykjavík, Iceland, 14–17 September 2003.
- Ismail, B.; Ahmed, W. Thermoelectric Power Generation Using Waste-Heat Energy as an Alternative Green Technology. Recent Patents Electr. Eng. 2009, 2, 27–39. [Google Scholar] [CrossRef]
- FAO. Statistical Yearbook; FAO: Rome, Italy, 2012; p. 366. [Google Scholar]
- Belal, I.E.H. A review of some fish nutrition methodologies. Bioresour. Technol. 2005, 96, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Millennium Ecosystem Assessment. Ecosystems and Human Well-Being: Synthesi; Island Press: Washington, DC, USA, 2005; p. 137. [Google Scholar]
- EcoPlan International (EPI). Global Assessment of Closed System Aquaculture; EPI: Vancouver, BC, Canada, 2008; p. 79. [Google Scholar]
- Dybas, C.L. Dead Zones Spreading in World Oceans. Bioscience 2005, 55, 552–557. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture; FAO: Rome, Italy, 2014; pp. 1–243. [Google Scholar]
- Hui, S.C.M. Green roof urban farming for buildings in high-density urban cities. In Proceedings of the World Green Roof Conference, Hainan, China, 8–21 March 2011; pp. 1–9.
- Wohlgenant, M.K. Chapter 16 Marketing margins: Empirical analysis. Handb. Agric. Econ. 2001, 1, 933–970. [Google Scholar]
- Bon, H.; Parrot, L.; Moustier, P. Sustainable urban agriculture in developing countries. A review. Agron. Sustain. Dev. 2010, 30, 21–32. [Google Scholar] [CrossRef]
- Toumi, S.; Vidal, R. A Comparison of Urban Agriculture and Short Food Chains in Paris and Tunis. Urban Agric. Mag. 2010, 24, 31–34. [Google Scholar]
- Rupasinghe, J.W.; Kennedy, J.O.S. Economic benefits of integrating a hydroponic-lettuce system into a barramundi fish production system. Aquac. Econ. Manag. 2010, 14, 81–96. [Google Scholar] [CrossRef]
- Hochmuth, G.J.; Hanlon, E.A. Commercial Vegetable Fertilization Principles; #SL319; Horticultural Sciences Department: Gainesville, USA, 2010; pp. 1–17. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goddek, S.; Delaide, B.; Mankasingh, U.; Ragnarsdottir, K.V.; Jijakli, H.; Thorarinsdottir, R. Challenges of Sustainable and Commercial Aquaponics. Sustainability 2015, 7, 4199-4224. https://doi.org/10.3390/su7044199
Goddek S, Delaide B, Mankasingh U, Ragnarsdottir KV, Jijakli H, Thorarinsdottir R. Challenges of Sustainable and Commercial Aquaponics. Sustainability. 2015; 7(4):4199-4224. https://doi.org/10.3390/su7044199
Chicago/Turabian StyleGoddek, Simon, Boris Delaide, Utra Mankasingh, Kristin Vala Ragnarsdottir, Haissam Jijakli, and Ragnheidur Thorarinsdottir. 2015. "Challenges of Sustainable and Commercial Aquaponics" Sustainability 7, no. 4: 4199-4224. https://doi.org/10.3390/su7044199
APA StyleGoddek, S., Delaide, B., Mankasingh, U., Ragnarsdottir, K. V., Jijakli, H., & Thorarinsdottir, R. (2015). Challenges of Sustainable and Commercial Aquaponics. Sustainability, 7(4), 4199-4224. https://doi.org/10.3390/su7044199








