Biochar Application in Malaysian Sandy and Acid Sulfate Soils: Soil Amelioration Effects and Improved Crop Production over Two Cropping Seasons
Abstract
:1. Introduction
2. Experimental Section
2.1. Selection of Rice Husk Biochar
2.2. Selection of Soils
2.3. Characterizations of Rice Husk Biochar and Soil
2.4. Pot Trial
2.5. Statistical Analysis
3. Results and Discussion
3.1. Biochar and Soil Physico-Chemical Properties
3.1.1. Rice Husk Biochar Properties
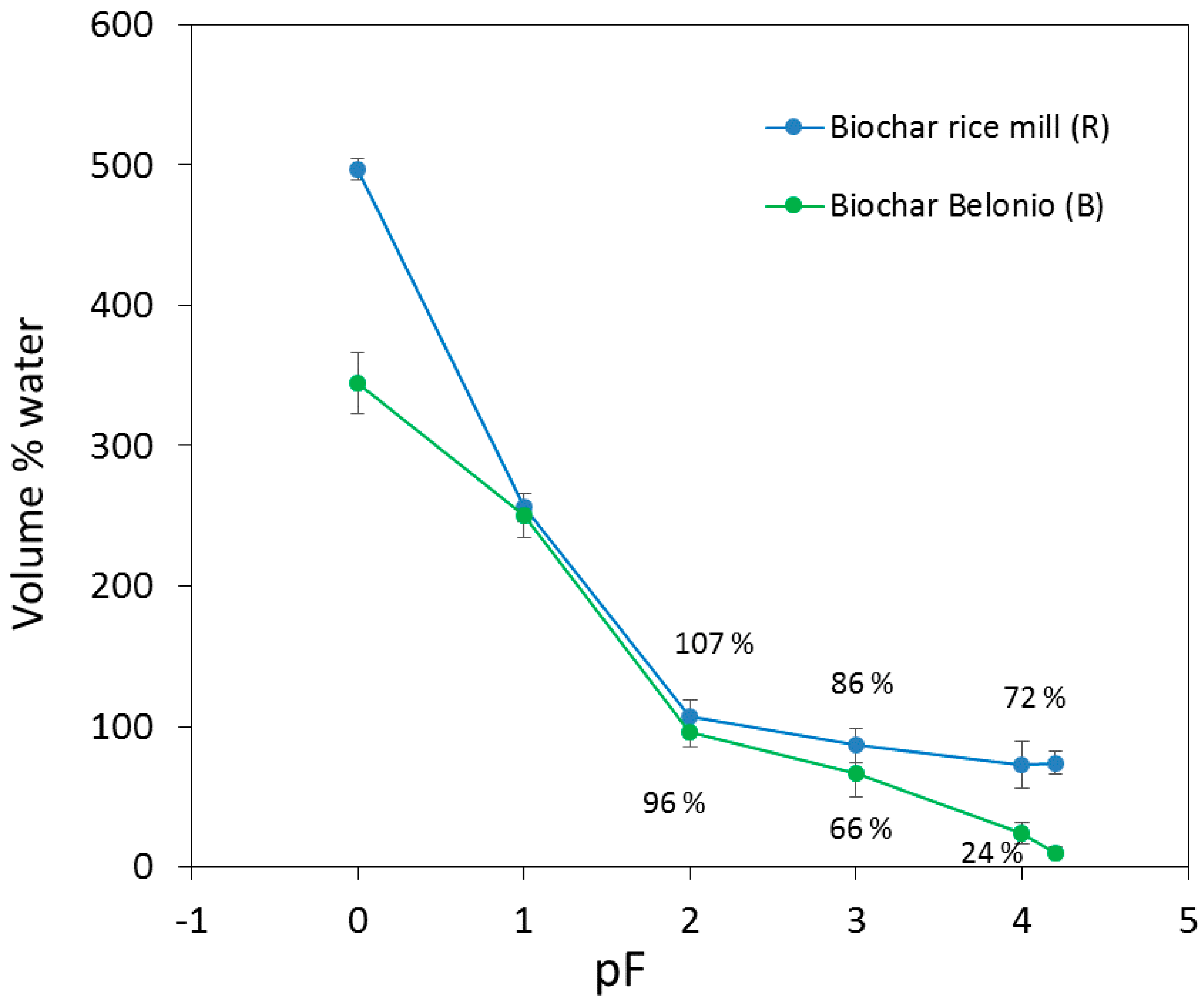
| pH | Total C (%) | Total N (%) | CEC (cmol/kg Soil) | Bulk Density (g/cm3) | Ash (%) | Alkalinity (cmol + kg/Soil) | Ca2+ (mg/kg) | Mg 2+ (mg/kg) | K+ (mg/kg) | Na+ (mg/kg) | Al3+ (m/kg) | Ca/Al Molar Ratio | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rice mill biochar | R | 10.2 | 16 | 0.25 | 13.2 | 0.13 | 75 | 40 | 45 | 980 | 870 | 190 | <0.1 | >100 |
| Belonio Biochar | B | 8.9 | 49 | 0.76 | 10.1 | 0.12 | 38 | 35 | 32 | 50 | 90 | 100 | <0.1 | >100 |
| Crop/Soil | Trt | Pot Trial Season 1 | Pot trial Season 2 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH | CEC (cmol + kg/Soil) | Total Soil Leachate (L) | Water at Field Capacity | Plant Available Water (PAW) | Ca/Al | Total Microbial Count (log Cfu 1 × 105) | pH | CEC (cmol + kg/Soil) | Total Soil Leachate (L) | Water at Field Capacity | Plant Available Water (PAW) | Ca/Al | Total Microbial Count (log Cfu1 × 105) | ||
| Corn/Acid sulfate | C | 5.1 ± 0.33 b | 15.2 ± 0.46 b | * | * | * | 1.29 ± 0.08 b | 4.41 ± 0.42 c | 5.3 ± 0.29 a | 17.9 ± 0.96 a | * | * | * | 2.4 ± 0.34 c | 5.1 ± 0.37 a |
| B2 | 5.2 ± 0.28 b | 17.0 ± 0.38 a | 1.82 ± 0.06 b | 5.82 ± 0.05 a,b | 5.4 ± 0.95 a | 18.0 ± 0.85 a | 4.8 ± 0.92 b | 5.3 ± 0.48 a | |||||||
| B5 | 5.9 ± 0.29 a,b | 16.5 ± 0.46 a | 3.55 ± 0.42 a | 6.30 ± 0.43 a | 5.5 ± 0.46 a | 18.2 ± 0.94 a | 6.9 ± 0.93 a,b | 4.9 ± 1.42 a,b | |||||||
| R2 | 5.5 ± 0.30 a | 16.9 ± 0.66 a | 1.55 ± 0.58 b | 5.51 ± 0.65 b | 5.7 ± 0.45 a | 17.3 ± 0.47 a | 4.5 ± 0.48 b | 4.8 ± 0.38 a,b | |||||||
| R5 | 5.8 ± 0.26 a | 15.7 ± 1.03 b | 2.37 ± 0.37 a,b | 5.84 ± 0.32 a,b | 5.9 ± 0.39 a | 17.7 ± 0.66 a | 6.3 ± 0.30 a,b | 5.2 ± 0.35 a | |||||||
| Corn/Sandy | C | 4.9 ± 0.84 b | 4.4 ± 0.16 c | 1.28 ± 0.11 a | 5.4 ± 0.75 c | 4.3 ± 0.45 b | 12.7 ± 0.54 b | 3.40 ± 0.59 b | 5.5 ± 0.26 a | 4.5 ± 0.15 b | 1.2 ± 0.62 a | 5.5 ± 0.28 c | 4.7 ± 0.24 b | 10.7 ± 0.72 c | 4.7 ± 0.49 b |
| B2 | 5.1 ± 0.26 b | 5.1 ± 0.27 b | 1.01 ± 0.13 b | 13.3 ± 0.16 b | 5.7 ± 0.30 a | 18.0 ± 0.53 b | 6.21 ± 0.27 a | 5.8 ± 0.31 a | 4.7 ± 0.16 a | 1.0 ± 0.13 a,b | 9.4 ± 0.47 b | 6.2 ± 0.23 a,b | 20.9 ± 0.21 b | 5.0 ± 0.37 a,b | |
| B5 | 5.5 ± 0.15 a,b | 6.6 ± 0.44 a | 0.82 ± 0.23 b,c | 15.3 ± 0.42 a | 6.2 ± 0.25 a | 34.4 ± 4.75 a | 6.15 ± 0.34 a | 5.4 ± 0.20 a | 5.2 ± 0.27 a | 0.9 ± 0.19 b | 9.2 ± 0.20 b | 8.2 ± 0.30 a | 26.3 ± 1.72 a,b | 4.8 ± 0.15 b | |
| R2 | 6.0 ± 0.22 a | 4.90 ± 0.33 b | 0.67 ± 0.11 c | 13.3 ± 0.53 b | 5.7 ± 0.32 a | 11.7 ± 0.94 b | 5.52 ± 0.27 a,b | 5.6 ± 0.26 a | 4.9 ± 0.30 a | 1.0 ± 0.15 a,b | 5.7 ± 0.61 c | 5.4 ± 0.36 b | 21.7 ± 2.34 b | 5.4 ± 0.46 a | |
| R5 | 6.1 ± 01.9 a | 6.1 ± 0.23 a,b | 0.69 ± 0.17 c | 15.6 ± 0.39 a | 6.55 ± 0.20 a | 30.6 ± 6.39 a | 5.90 ± 0.15 a | 5.6 ± 0.16 a | 4.9 ± 0.43 a | 0.8 ± 0.25 b | 12.5 ± 0.46 a | 7.0 ± 0.22 b | 43.7 ± 1.12 a | 5.6 ± 0.11 a | |
| Rice/Acid sulfate | C | 4.6 ± 0.35 b | 14.9 ± 0.26 c | * | * | * | 2.28 ± 0.21 b | 3.92 ± 0.42 b | 4.3 ± 0.25 a | 14.6 ± 0.33 b | * | * | * | 2.460.20 b | 4.70.42 b |
| B2 | 4.9 ± 0.07 b | 19.8 ± 2.08 a,b | 2.60 ± 0.31 b | 3.4 ± 0.15 b | 4.5 ± 0.11 a | 15.2 ± 0.46 a,b | 3.14 ± 0.61 a,b | 5.6 ± 0.48 a | |||||||
| B5 | 5.2 ± 0.20 a,b | 23.7 ± 2.71 a | 5.14 ± 0.6 a | 5.11 ± 0.23 a | 4.8 ± 0.69 a | 15.2 ± 0.22 a,b | 4.17 ± 0.19 a | 4.8 ± 0.43 b | |||||||
| R2 | 5.3 ± 0.40 a | 18.1 ± 1.74 a,b | 1.15 ± 0.80 b | 4.53 ± 0.21 a,b | 4.5 ± 0.09 a | 16.9 ± 0.48 a | 2.91 ± 0.61 b | 5.4 ± 0.38 a | |||||||
| R5 | 5.5 ± 0.30 a | 16.9 ± 0.18 b | 5.26 ± 0.79 a | 5.87 ± 0.40 a | 4.7 ± 0.11 a | 17.0 ± 0.28 a | 4.65 ± 0.37 a | 5.0 ± 0.32 a,b | |||||||
3.1.2. Soil Properties
3.2. The Effect of Rice Husk Biochar Amendment on the Yield of Corn and Rice
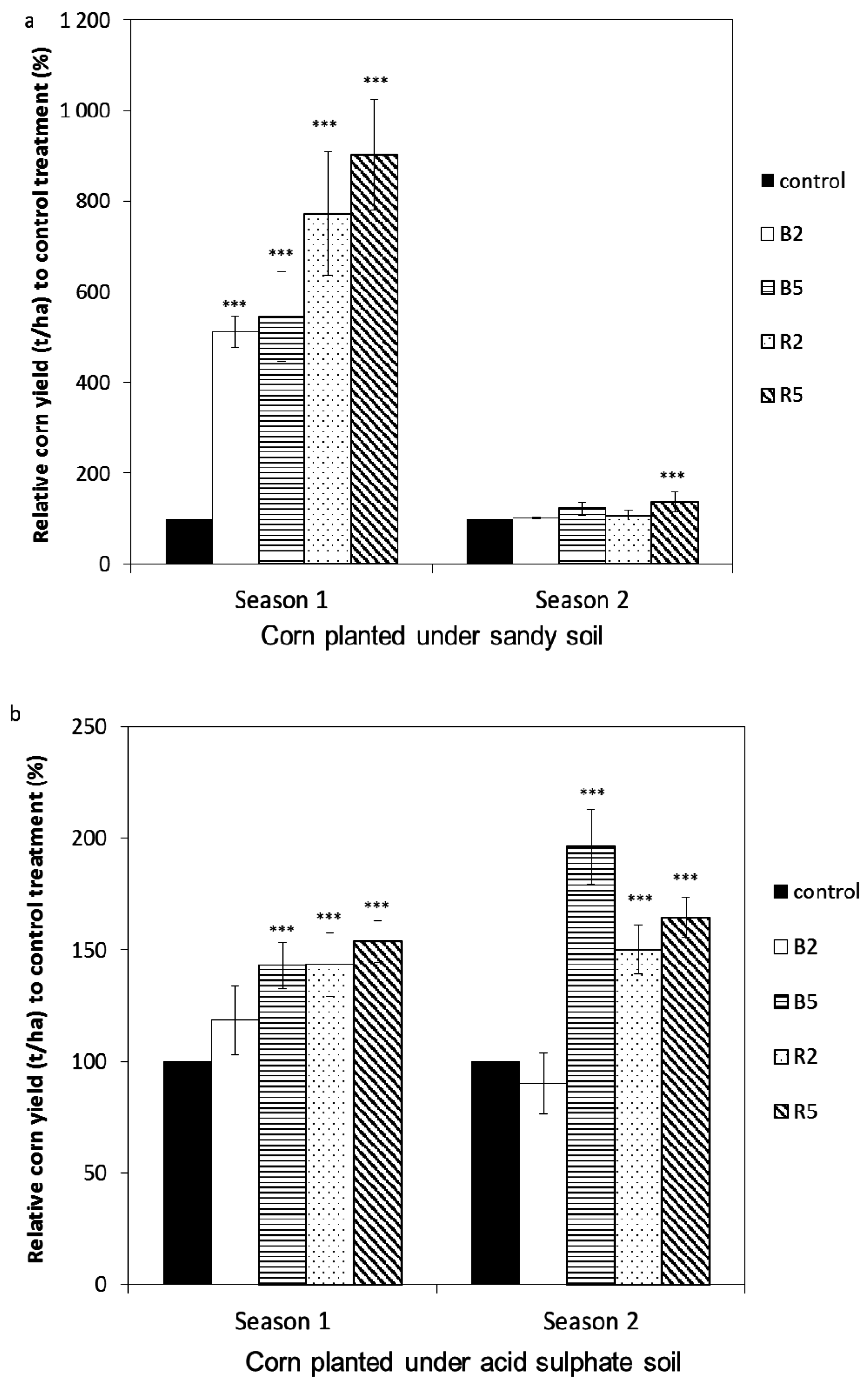
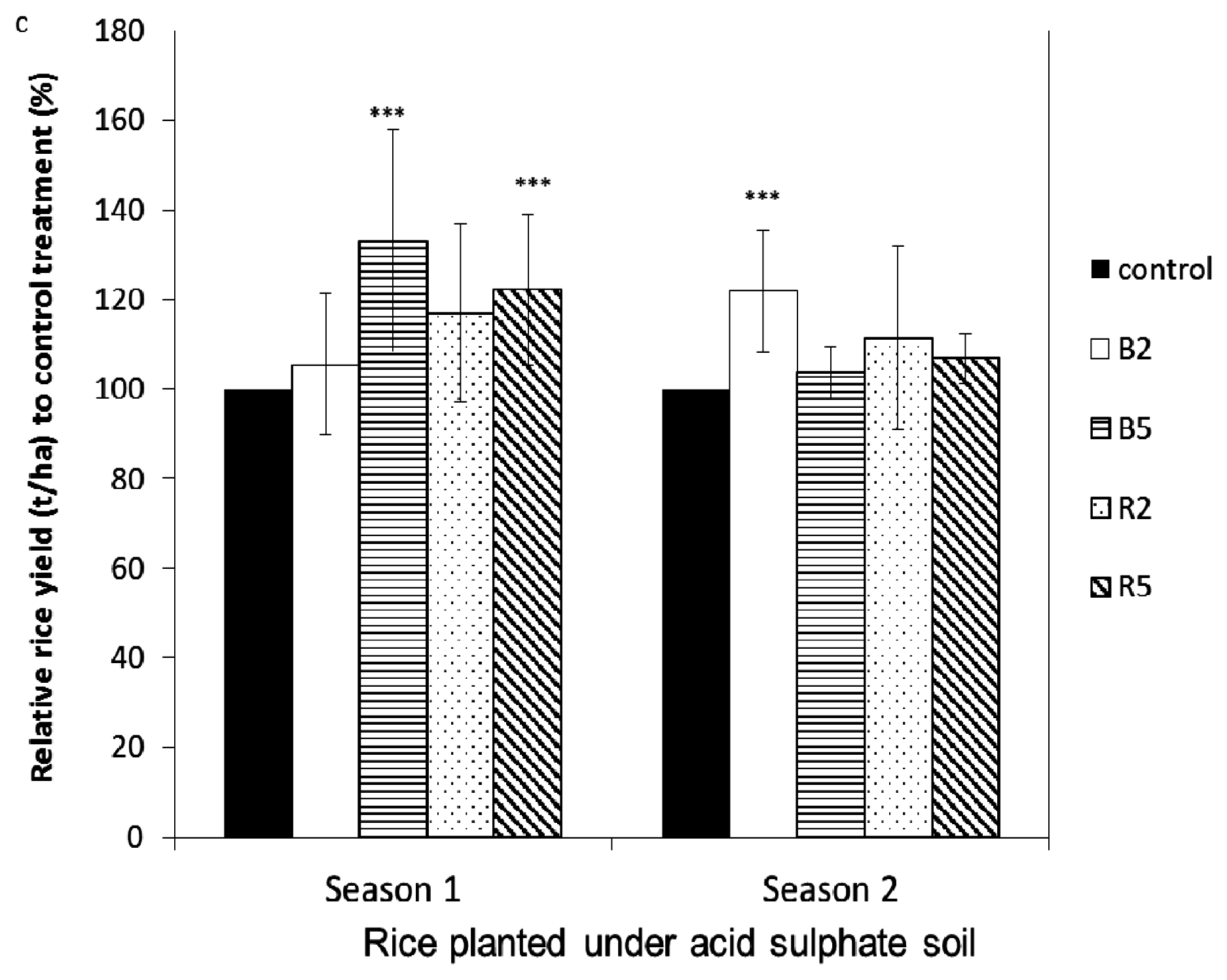
3.2.1. Sandy Soil Cropped with Corn
3.2.2. Acid Sulfate Soil Cropped with Corn and Rice
3.3. The Effect of the Amendment of Rice Husk Biochar on Soil Physical, Chemical and Biological Properties
3.3.1. Soil pH
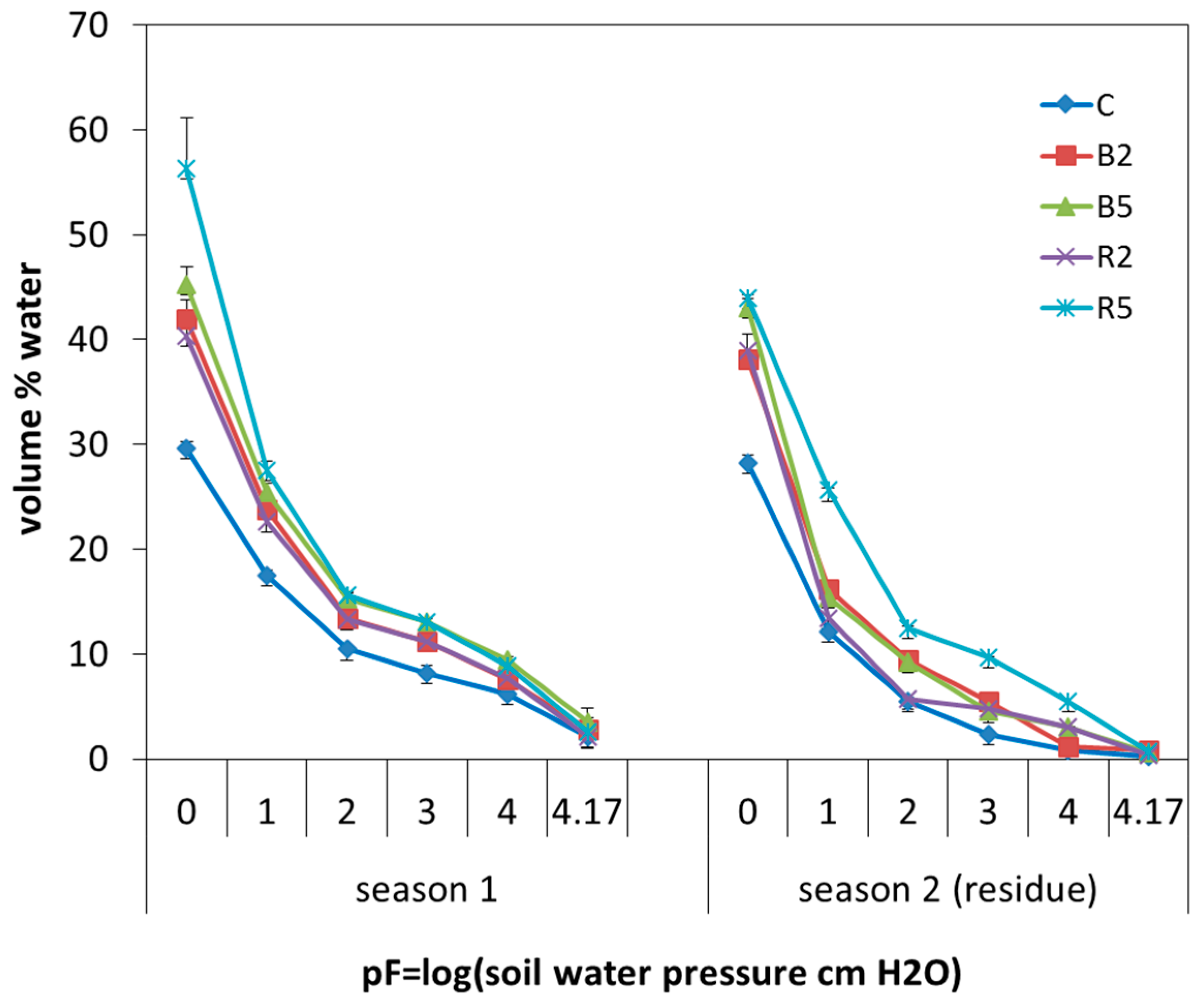
3.3.2. Soil CEC and Exchangeable Bases
3.3.3. Plant-Available Water
3.3.4. Soil Microbial Population
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lehmann, J.; Joseph, S. Biochar for Environmental Management. Science Technology and Implementation, 2nd ed.; Routledge Taylor and Francis Group: London, UK, 2015. [Google Scholar]
- Manickam, T.; Bachmann, R.T.; Illain, Z.I.; Zulkefli, M.; Husni, M.H.; Samsuri, A.W. Characterization of local mill rice husk charcoal and its effect on compost properties. Malays. J. Soil Sci. 2012, 16, 89–102. [Google Scholar]
- Liang, B.; Lehmann, J.; Solomon, D.; Kinyangi, J.; Grossman, J.; O’Neill, B.; Skjemstad, J.O.; Thies, J.; Luizao, F.J.; Petersen, J.; et al. Black carbon increases cation exchange capacity in soils. Soil Sci. Soc. Am. J. 2006, 70, 1719–1730. [Google Scholar] [CrossRef]
- Cornelissen, G.; Martinsen, V.; Shitumbanuma, V.; Alling, V.; Breedveld, G.D.; Rutherford, D.; Sparrevik, M.; Hale, S.E.; Obia, A.; Mulder, J. Biochar effect on maize yield and soil characteristics in five conservation farming sites in zambia. Agron. J. 2013, 3, 256–274. [Google Scholar] [CrossRef]
- Jeffery, S.; Verheijen, F.G.A.; van der Velde, M.; Bastos, A.C. A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric. Ecosyst. Environ. 2011, 144, 175–187. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, A.; Ji, C.; Joseph, S.; Bian, R.; Li, L.; Pan, G.; Paz-Ferreiro, J. Biochar’s effect on crop productivity and the dependence on experimental conditions-a meta-analysis of literature data. Plant Soil 2013, 373, 583–594. [Google Scholar] [CrossRef]
- Nieuwolt, S. Climate and Agricultural Planning in Peninsular Malaysia (Special Report); Malaysian Agricultural Research & Development Institute: Selangor, Malaysia, 1982; p. 139. [Google Scholar]
- Enio, M.S.K.; Shamshuddin, J.; Fauziah, C.I.; Husni, M.H.A. Pyritization of the coastal sediments in the kelantan plains in the malay peninsula during the holocene. Am. J. Agric. Biol. Sci. 2011, 6, 393–402. [Google Scholar] [CrossRef]
- Jusop, S. Acid Sulfate Soils. Occurrence, Properties and Utilisation for Rice Production; Akademi Sains Malaysia: Kuala Lumpur, Malaysia, 2012. [Google Scholar]
- Shamshuddin, J.; Muhrizal, S.; Fauziah, I.; van Ranst, E. A laboratory study of pyrite oxidation in acid sulfate soils. Commun. Soil Sci. Plant Anal. 2004, 35, 117–129. [Google Scholar] [CrossRef]
- Panhwar, Q.A.; Naher, U.A.; Radziah, O.; Shamshuddin, J.; Razi, I.M. Bio-fertilizer, ground magnesium limestone and basalt applications may improve chemical properties of malaysian acid sulfate soils and rice growth. Pedosphere 2014, 24, 827–835. [Google Scholar] [CrossRef]
- Shamshuddin, J.; Fauziah, C.I.; Arifin, A.; Rosenami, A.B. Effects of biochar application on the nutrients uptake of corn grown on sandy spodsols. Personal communication, 2015. [Google Scholar]
- Hale, S.E.; Lehmann, J.; Rutherford, D.; Zimmerman, A.R.; Bachmann, R.T.; Shitumbanuma, V.; O’Toole, A.; Sundqvist, K.L.; Arp, H.P.H.; Cornelissen, G. Quantifying the total and bioavailable polycyclic aromatic hydrocarbons and dioxins in biochars. Environ. Sci. Technol. 2012, 46, 2830–2838. [Google Scholar] [CrossRef] [PubMed]
- Carter, S.; Shackley, S.; Sohi, S.; Suy, T.B.; Haefele, S.M. The impact of biochar application on soil properties and plant growth of pot grown lettuce (Lactuca sativa) and cabbage (Brassica chinensis). Agron. J. 2013, 3, 404–418. [Google Scholar] [CrossRef]
- Shackley, S.; Carter, S.; Knowles, T.; Middelink, E.; Haefele, S.M.; Sohi, S.; Cross, A.; Haszeldine, S. Sustainable gasification–biochar systems? A case-study of rice-husk gasification in cambodia, part I: Context, chemical properties, environmental and health and safety issues. Energy Policy 2012, 42, 49–58. [Google Scholar] [CrossRef]
- Igarashi, T. Soil Improvement Effect of FMP & CRH in Indonesia; JICA Pamphlet; JICA: Tokoy, Japan, 1996; p. 30. [Google Scholar]
- Oka, H.; Rungrattanakasin, W. Improvement of sandy soil in the northeast by using carbonized rice husks. ICA Tech. Rep. 1993, 13, 40–42. [Google Scholar]
- Jaafar, N.M.; Clode, P.L.; Abbott, L.K. Soil microbial responses to biochars varying in particle size, surface and pore properties. Pedosphere 2015, 25, 770–780. [Google Scholar] [CrossRef]
- Kameyama, K.; Miyamoto, T.; Shinogi, Y. Increases in available water content of soils by applying bagasse-charcoals. In Proceedings of the 19th World Congress of Soil Science, Soil Solutions for a Changing World, Brisbane, Australia, 1–6 August 2010.
- Steiner, C.; Teixeira, W.G.; Lehmann, J.; Nehls, T.; de Macedo, J.L.V.; Blum, W.E.H.; Zech, W. Long term effects of manure, charcoal and mineral fertilization on crop production and fertility on a highly weathered central amazonian upland soil. Plant Soil 2007, 291, 275–290. [Google Scholar] [CrossRef]
- Kisiki, N.H.; Hale, S.E.; Cornelissen, G.; Bachmann, R.T. Designing and performance evaluation of biochar production in a top-lit updraft up-scaled gasifier. J. Sustain. Bioenergy Syst. 2015, 5, 41–55. [Google Scholar]
- Chapman, H.D. Cation exchange capacity. In Methods of Soil Analysis; Black, C.A., Ed.; American Society of Agronomy: Madison, WI, USA, 1965; Part 2; pp. 891–901. [Google Scholar]
- Zimmerman, A.R. Abiotic and microbial oxidation of laboratory-produced black carbon (biochar). Environ. Sci. Technol. 2010, 44, 1295–1301. [Google Scholar] [CrossRef] [PubMed]
- Krishnakumar, S.; Kumar, S.R.; Mariappan, N.; Surendar, K.K. Biochar-boon to soil health and crop production. Afr. J. Agric. Res. 2013, 8, 4726–4739. [Google Scholar]
- Petter, F.A.; Madari, B.E.; Silva, M.A.S.D.; Carneiro, M.A.C.; Carvalho, M.T.D.M.; Marimon Júnior, B.H.; Pacheco, L.P. Soil fertility and upland rice yield after biochar application in the cerrado. Pesqui. Agropecu. Bras. 2012, 47, 699–706. [Google Scholar] [CrossRef]
- Zhang, A.; Bian, R.; Pan, G.; Cui, L.; Hussain, Q.; Li, L.; Zheng, J.; Zheng, J.; Zhang, X.; Han, X.; et al. Effects of biochar amendment on soil quality, crop yield and greenhouse gas emission in a chinese rice paddy: A field study of 2 consecutive rice growing cycles. Field Crops Res. 2012, 127, 153–160. [Google Scholar] [CrossRef]
- Alling, V.; Hale, S.E.; Martinsen, V.; Mulder, J.; Smebye, A.; Breedveld, G.D.; Cornelissen, G. The role of biochar in retaining nutrients in amended tropical soils. J. Plant Nutr. Soil Sci. 2014, 177, 671–680. [Google Scholar] [CrossRef]
- Fernández, F.G.; Hoeft, R.G. Managing soil pH and crop nutrients. Available online: http://extension.cropsciences.illinois.edu/handbook/pdfs/chapter08.pdf (accessed on 16 December 2015).
- Sohi, S.P.; Krull, E.; Lopez-Capel, E.; Bol, R. A review of biochar and its use and function in soil. Adv. Agron. 2010, 105, 47–82. [Google Scholar]
- Havlin, J.L.; Beaton, J.D.; Tisdale, S.L.; Nelson, W.R. Soil Fertility and Fertilizers, an Introduction to Nutrient Management, 6th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 1999. [Google Scholar]
- Laird, D.A.; Fleming, P.; Davis, D.D.; Horton, R.; Wang, B.; Karlen, D.L. Impact of biochar amendments on the quality of a typical midwestern agricultural soil. Geoderma 2010, 158, 443–449. [Google Scholar] [CrossRef]
- Martinsen, V.; Mulder, J.; Shitumbanuma, V.; Sparrevik, M.; Borresen, T.; Cornelissen, G. Farmer-led maize biochar trials: Effect on crop yield and soil nutrients under conservation farming. J. Plant Nutr. Soil Sci. 2014, 177, 681–695. [Google Scholar] [CrossRef]
- Masulili, A.; Utomo, W.H.; Syechfani, M.S. Rice husk biochar for rice based cropping system in acid soil 1. The characteristics of rice husk biochar and its influence on the properties of acid sulfate soils and rice growth in west kalimantan, indonesia. J. Agric. Sci. 2010, 2, 39–47. [Google Scholar] [CrossRef]
- De Melo Carvalho, M.T.; de Holanda Nunes Maia, A.; Madari, B.E.; Bastiaans, L.; van Oort, P.A.J.; Heinemann, A.B.; Soler da Silva, M.A.; Petter, F.A.; Marimon, B.H., Jr.; Meinke, H. Biochar increases plant-available water in a sandy loam soil under an aerobic rice crop system. Solid Earth 2014, 5, 939–952. [Google Scholar] [CrossRef]
- Kammann, C.I.; Schmidt, H.-P.; Messerschmidt, N.; Linsel, S.; Steffens, D.; Mueller, C.; Koyro, H.-W.; Conte, P.; Stephen, J. Plant growth improvement mediated by nitrate capture in co-composted biochar. Sci. Rep. 2015. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manickam, T.; Cornelissen, G.; Bachmann, R.T.; Ibrahim, I.Z.; Mulder, J.; Hale, S.E. Biochar Application in Malaysian Sandy and Acid Sulfate Soils: Soil Amelioration Effects and Improved Crop Production over Two Cropping Seasons. Sustainability 2015, 7, 16756-16770. https://doi.org/10.3390/su71215842
Manickam T, Cornelissen G, Bachmann RT, Ibrahim IZ, Mulder J, Hale SE. Biochar Application in Malaysian Sandy and Acid Sulfate Soils: Soil Amelioration Effects and Improved Crop Production over Two Cropping Seasons. Sustainability. 2015; 7(12):16756-16770. https://doi.org/10.3390/su71215842
Chicago/Turabian StyleManickam, Theeba, Gerard Cornelissen, Robert T. Bachmann, Illani Z. Ibrahim, Jan Mulder, and Sarah E. Hale. 2015. "Biochar Application in Malaysian Sandy and Acid Sulfate Soils: Soil Amelioration Effects and Improved Crop Production over Two Cropping Seasons" Sustainability 7, no. 12: 16756-16770. https://doi.org/10.3390/su71215842
APA StyleManickam, T., Cornelissen, G., Bachmann, R. T., Ibrahim, I. Z., Mulder, J., & Hale, S. E. (2015). Biochar Application in Malaysian Sandy and Acid Sulfate Soils: Soil Amelioration Effects and Improved Crop Production over Two Cropping Seasons. Sustainability, 7(12), 16756-16770. https://doi.org/10.3390/su71215842





