The Case of the Lacking Carbonates and the Emergence of Early Life on Mars
Abstract
:1. Introduction
| Aqueous species | log activity |
| CO2 (aq) | –0.77 |
| SiO2 (aq) | –2.83 |
| Mg2+ | –3.13 |
| Ca2+ | –3.14 |
| Fe2+ | –3.18 |
| HCO3– | –3.61 |
| FeHCO3+ | –4.07 |
| CaHCO3+ | –5.71 |
| MgHCO3+ | –5.71 |
| Mineral saturation | log Q/K |
| Siderite | –3.1 |
| Calcite | –5.1 |
| Aragonite | –5.2 |
| Magnesite | –5.5 |
2. Carbonate Precipitation on Earth
2.1. Geological and Biological Aspects of the Carbon Cycle on Earth
2.2. Carbonate Formation on Earth
3. Control of pH in Surface Waters by Microbial Uptake of CO2
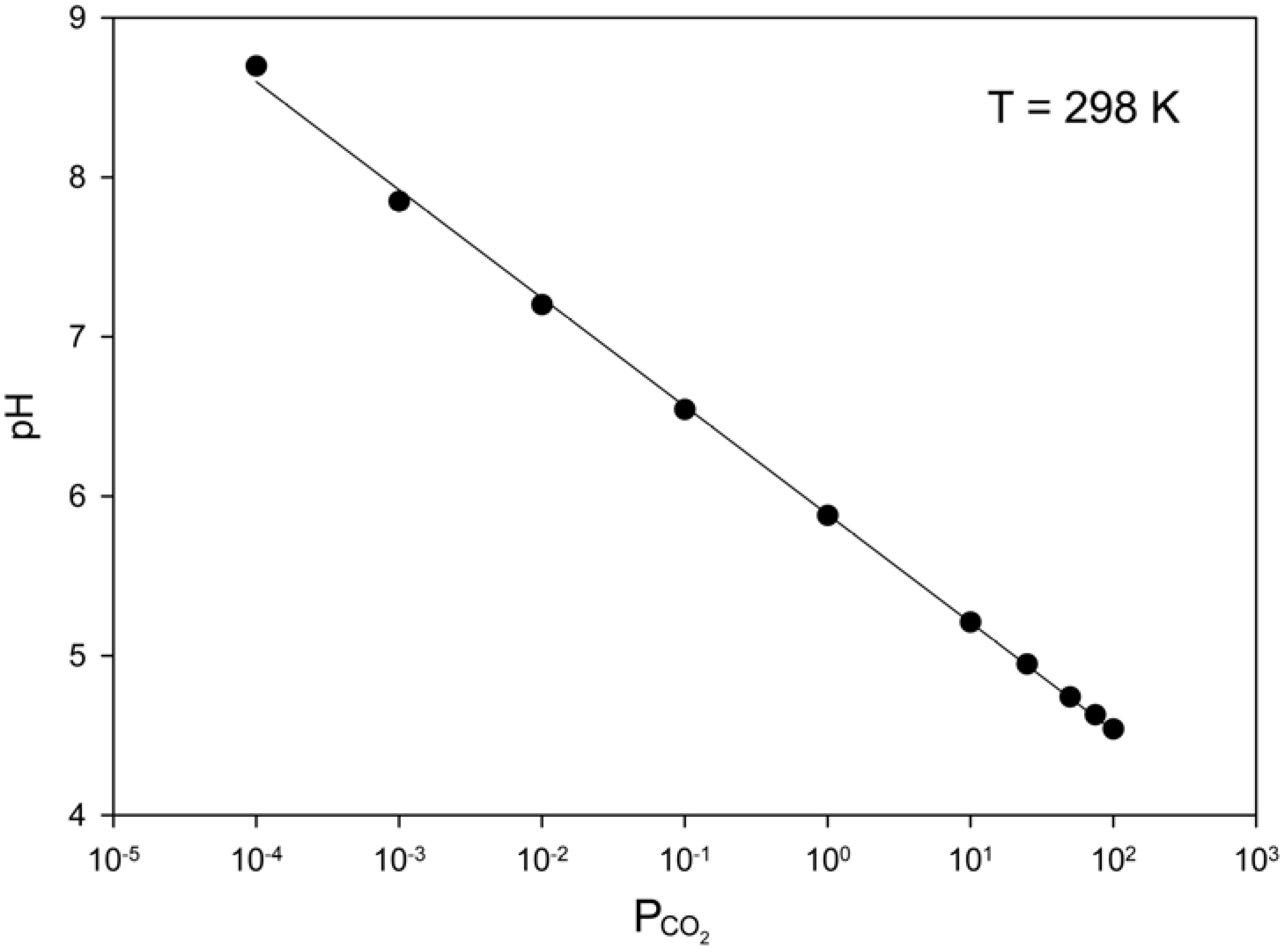
4. Carbonate Distribution on Mars
4.1. Carbonates in a Sterile Mars
4.2. Carbonates on Mars Inhabited with both Surface and Subsurface Life
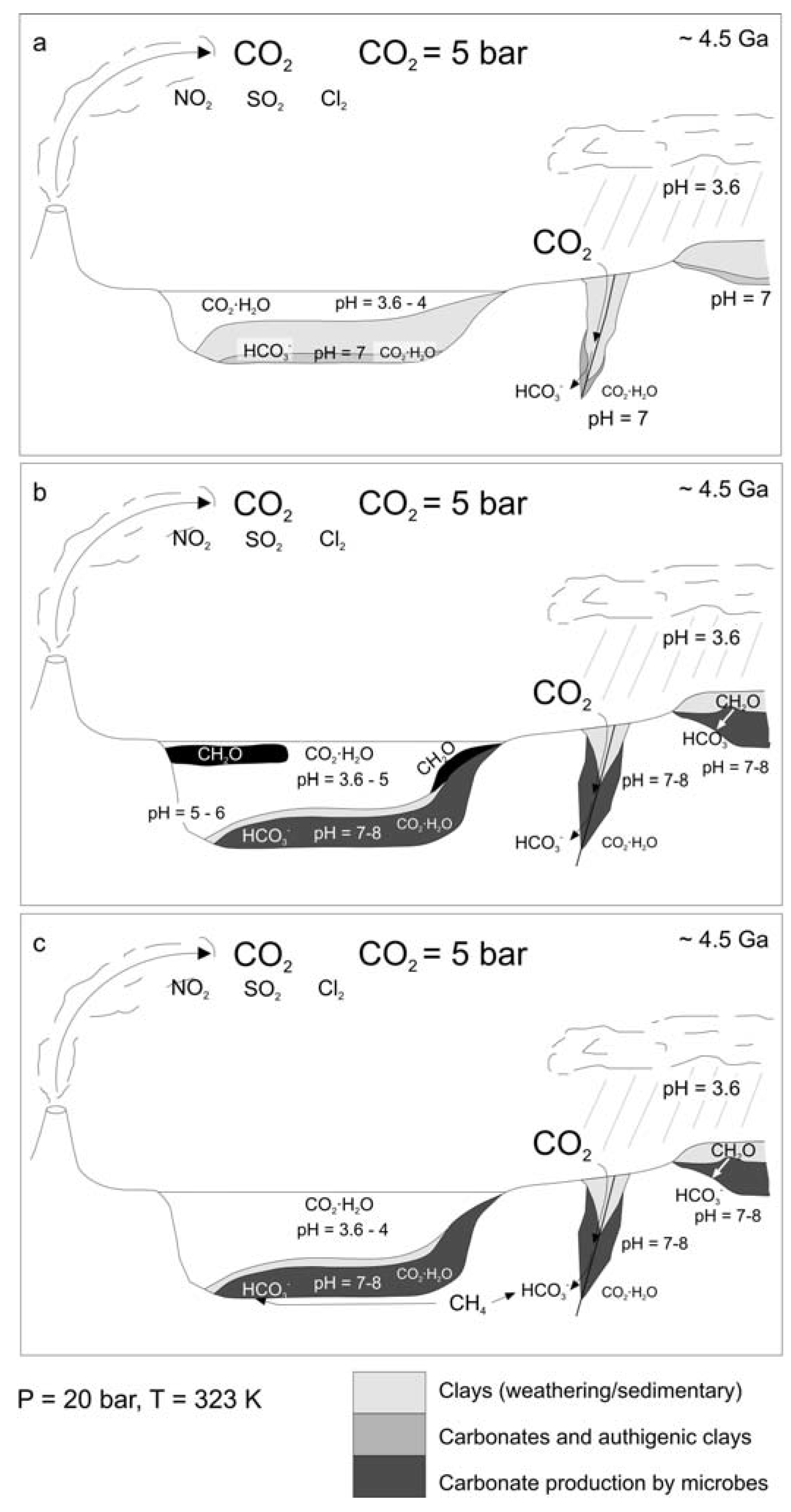
4.3. Carbonates on Mars Inhabited with Subsurface Life
5. Carbonates and the Emergence of Life on Mars
Acknowledgements
References and Notes
- Bibring, J.P.; Langevin, Y.; Mustard, J.F.; Poulet, F.; Arvidson, R.; Gendrin, A.; Gondet, B.; Mangold, N.; Pinet, P.; Forget, F.; The OMEGA Team; Berthe, M.; Gomez, C.; Jouglet, D.; Soufflot, A.; Vincendon, M.; Combes, M.; Drossart, P.; Encrenaz, T.; Fouchet, T.; Merchiorri, R.; Belluci, G.; Altieri, F.; Formisano, V.; Capaccioni, F.; Cerroni, P.; Coradini, A.; Fonti, S.; Korablev, O.; Kottsov, V.; Ignatiev, N.; Moroz, V.; Titov, D.; Zasova, L.; Loiseau, D.; Pinet, P.; Doute, S.; Schmitt, B.; Sotin, C.; Hauber, E.; Hoffmann, H.; Jaumann, R.; Keller, U.; Arvidson, R.; Duxbury, T.; Forget, F.; Neukum, G. Global mineralogical and aqueous Mars history derived from OMEGA/Mars express data. Science 2006, 312, 400–404. [Google Scholar] [CrossRef] [PubMed]
- Squyres, S.W.; Arvidson, R.E.; Bell, J.F.; Bruckner, J.; Cabrol, N.A.; Calvin, W.; Carr, M.H.; Christensen, P.R.; Clark, B.C.; Crumpler, L.; Des Marais, D.J.; d’Uston, C.; Economou, T.; Farmer, J.; Farrand, W.; Folkner, W.; Golombek, M.; Gorevan, S.; Grant, J. A.; Greeley, R.; Grotzinger, J.; Haskin, L.; Herkenhoff, K.E.; Hviid, S.; Johnson, J.; Klingelhofer, G.; Knoll, A.; Landis, G.; Lemmon, M.; Li, R.; Madsen, M.B.; Malin, M.C.; McLennan, S.M.; McSween, H.Y.; Ming, D.W.; Moersch, J.; Morris, R.V.; Parker, T.; Rice, J.W.; Richter, L.; Rieder, R.; Sims, M.; Smith, M.; Smith, P.; Soderblom, L.A.; Sullivan, R.; Wanke, H.; Wdowiak, T.; Wolff, M.; Yen, A. The Spirit Rover’s Athena science investigation at Gusev Crater, Mars. Science 2004, 305, 794–799. [Google Scholar] [CrossRef] [PubMed]
- Murchie, S.L.; Mustard, J.F.; Ehlmann, B.L.; Miliken, R.E.; Bishop, J.L.; McKeown, N.K.; Noe Dobrea, E.Z.; Seelos, F.P.; Buczkowski, D.L.; Wiseman, S.M.; Arvidson, R.E.; Wray, J.J.; Swayze, G.; Clark, R.N.; Des Marais, D.J.; McEwen, A.S.; Bibring, J.P. A synthesis of Martian aqueous mineralogy after one Mars year of observations from the Mars Reconnaissance Orbiter. J. Geophys. Res. 2009, 114, E00D06. [Google Scholar]
- Wiseman, S.M.; Arvidson, R.A.; Andrews-Hanna, J.C.; Clark, R.N.; Lanza, N.L.; Des Marais, D.; Marzo, G.A.; Morris, R.V.; Murchie, S.L.; Newsom, H.E.; Noe Dobrea, E.Z.; Ollila, A.M.; Poulet, R.; Roush, T.L.; Seelos, F.P.; Swayze, G.A. Phyllosilicate and sulfate-hematite deposits within Miyamoto crater in southern Sinus Meridiani, Mars. Geophys. Res. Let. 2009, 35, L19204. [Google Scholar] [CrossRef]
- Chevrier, V.; Poulet, F.; Bibring, J.P. Early geochemical environment of Mars as determined from thermodynamics of phyllosilicates. Nature 2007, 448, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Skok, J.R.; Mustard, J.F.; Murchie, S.L. Identification of primary Noachian crustal blocks on Mars with CRISM observations. In Proceedings of the 40th Lunar and Planetary Science Conference, The Woodlands, TX, USA, 23–27 March 2009.
- Bibring, J.P.; Ardvison, R.E.; Gendrin, A.; Gondet, B.; Langevin, Y.; Le Mouelic, S.; Mangold, N.; Morris, R.V.; Mustard, J.F.; Poulet, F.; Quantin, C.; Sotin, C. Coupled ferric oxides and sulfates on the Martian surface. Science 2007, 317, 1206–1210. [Google Scholar] [CrossRef] [PubMed]
- Sherlock, E.J.; Lawrence, R.W.; Poulin, R. On the neutralization of acid rock drainage by carbonate and silicates minerals. Environ. Geol. 1995, 25, 43–45. [Google Scholar] [CrossRef]
- Lackner, K.S. Carbonate chemistry for sequestering fossil carbon. Annu. Rev. Energ. Environ. 2002, 27, 197–232. [Google Scholar] [CrossRef]
- Ehlmann, B.L.; Mustard, J.F.; Murchie, S.L.; Poulet, F.; Bishop, J.L.; Brown, A.J.; Calvin, W.M.; Clark, R.N.; Des Marais, D.; Milliken, R.E.; Roach, L.H.; Roush, T.L.; Swayze, G.A.; Wray, J.J. Orbital identification of carbonate-bearing rocks on Mars. Science 2008, 322, 1828–1832. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, F.; Scaillet, B. The sulfur content of volcanic gases on Mars. Earth Planet. Sci. Let. 2009, 279, 34–43. [Google Scholar] [CrossRef]
- Elkins-Tanton, L.T. Linked magma ocean solidification and atmospheric growth for Earth and Mars. Earth Planet. Sci. Let. 2008, 271, 181–191. [Google Scholar] [CrossRef]
- Duan, Z.; Sun, R.; Zhu, C.; Chou, I.M. An improved model for the calculation of CO2 solubility in aqueous solutions containing Na+, K+, Ca2+, Mg2+, Cl–, and SO42–. Mar. Chem. 2006, 98, 131–139. [Google Scholar] [CrossRef]
- Orr, J.C.; Fabry, V.J.; Aumont, O.; Bopp, L.; Doney, S.C.; Feely, R.A.; Gnanadesikan, A.; Gruber, N.; Ishida, A.; Joos, F.; Key, R.M.; Lindsay, K.; Maier-Reimer, E.; Matear, R.; Monfray, P.; Mouchet, A.; Najjar, R.G.; Plattner, G.K.; Rodgers, K.B.; Sabine, C.L.; Sarmiento, J.L.; Schlitzer, R.; Slater, R.D.; Totterdell, I.J.; Weirig, M.F.; Yamanaka, Y.; Yool, A. Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 2005, 437, 681–686. [Google Scholar] [CrossRef] [PubMed]
- Squyres, S.W.; Kasting, J.F. Early Mars: How warm and how wet? Science 1994, 265, 744–749. [Google Scholar] [CrossRef] [PubMed]
- Hirschmann, M.M.; Withers, A.C. Ventilation of CO2 from a reduced mantle and consequences for the early Martian greenhouse. Earth Planet. Sci. Let. 2008, 270, 147–155. [Google Scholar] [CrossRef]
- Robert, F.; Chaussidon, M. A palaeotemperature curve for the Precambrian oceans based on silicon isotopes in cherts. Nature 2006, 443, 969–972. [Google Scholar] [CrossRef] [PubMed]
- Carr, M.H. Retention of an atmosphere on Early Mars. J. Geophys. Res. 1999, 104, 21897–21909. [Google Scholar]
- Albarède, F. Volatile accretion history of the terrestrial planets and dynamic implications. Nature 2009, 461, 1227–1233. [Google Scholar] [CrossRef] [PubMed]
- Morse, J.W.; Mackenzie, F.T. Geochemistry of sedimentary carbonates. In Developments in Sedimentology; Elsevier: Amsterdam, The Netherlands, 1990. [Google Scholar]
- Morse, J.W.; Arvidson, R.S.; Lüttge, A. Calcium carbonate formation and dissolution. Chem. Rev. 2007, 107, 342–381. [Google Scholar]
- McKenzie, J.A. The dolomite problem: An outstanding controversy. In Controversies in Modern Geology: Evolution of Geochemical Theories in Sedimentology, Earth History and Tectonics; Müller, D.W., McKenzie, J.A., Weissert, H., Eds.; Academic Press: London, UK, 1991; pp. 37–54. [Google Scholar]
- Sánchez-Román, M.; Vasconcelos, C.; Schmid, T.; Dittrich, M.; McKenzie, J.A.; Zenobi, R.; Rivadeneyra, M.A. Aerobic microbial dolomite at the nanometer scale: Implications for the geologic record. Geology 2008, 36, 879–882. [Google Scholar] [CrossRef]
- Morrows, D.W.; Rickets, B.D. Experimental investigation of sulphate inhibition of dolomite and its mineral analogues. In Sedimentology and Geochemistry of Dolostones; Shukla, V., Baker, P.A., Eds.; SEPM: Tulsa, OK, USA, 1988; Volume 43, pp. 25–38. [Google Scholar]
- Gaines, A.M. Dolomiotization kinetics: Recent experimental studies. In Concepts and Models of Dolomitization; Zenger, D.H., Dunhan, J.B., Ethington, R.L., Eds.; SEPM: Tulsa, OK, USA, 1980; Volume 28, pp. 81–86. [Google Scholar]
- Lippman, F. Sedimentary Carbonate Minerals; Springer-Verlag: New York, NY, USA, 1973; p. 228. [Google Scholar]
- Folk, R.L.; Land, L.S. Mg/Ca ratio and salinity: Two controls over crystallization of dolomite. AAPGB 1975, 59, 60–68. [Google Scholar]
- Baker, P.A.; Kastner, M. Constraints on the formation of sedimentary dolomite. Science 1981, 213, 214–216. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, C.; McKenzie, J.A.; Bernasconi, S.; Grujic, D.; Tien, A.J. Microbial mediation as a possible mechanism for natural dolomite formation at low temperatures. Nature 1995, 377, 220–222. [Google Scholar] [CrossRef]
- Knorre, H.; Krumbein, W.E. Bacterial calcification. In Microbial Sediments; Riding, R.E., Awramik, S.M., Eds.; Springer: Berlin, Germany, 2000; pp. 25–31. [Google Scholar]
- Thompson, J.B.; Ferris, F.G. Cyanobacterial precipitation of gypsum, calcite, and magnesite from natural alkaline lake water. Geology 1990, 18, 995–998. [Google Scholar] [CrossRef]
- Roberts, J.A.; Bennett, P.C.; Gonzalez, L.A.; Macpherson, G.L.; Miliken, K.L. Microbial precipitation of dolomite in methanogenic groundwater. Geology 2004, 32, 277–280. [Google Scholar] [CrossRef]
- Warthmann, R.; van Lith, Y.; Vasconcelos, C.; McKenzie, J.A.; Karpoff, A.M. Bacterially induced dolomite precipitation in anoxic culture experiments. Geology 2000, 28, 1991–1094. [Google Scholar] [CrossRef]
- Ehrlich, H.L. Geomicrobiology, 4th ed.; Marcel Dekker: New York, NY, USA, 2002; p. 768. [Google Scholar]
- Schultze-Lam, S.; Beveridge, T.J. Nucleation of celestite and strontianite on a cyanobacterial S-layer. Appl. Environ. Microbiol. 1994, 60, 447–453. [Google Scholar] [PubMed]
- Sánchez-Román, M.; Rivadeneyra, M.; Vasconcelos, C.; McKenzie, J.A. Biomineralization of carbonate and phosphate by halophilic bacteria: Influence of Ca2+ and Mg2+ ions. FEMS Microbiol. Ecol. 2007, 61, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Díaz-del-Río, V.; Somoza, L.; Martínez-Frías, J.; Mata, M.P.; Delgado, A.; Hernandez-Molina, F.J.; Lunar, R.; Martín-Rubí, J.A.; Maestro, A.; Fernández-Puga, M.C.; León, R.; Llave, E.; Medialdea, T.; Vázquez, J.T. Vast fields of hydrocarbon-derived carbonate chimneys related to the accretionary wedge/olistostrome of the Gulf of Cádiz. Mar. Geol. 2003, 195, 177–200. [Google Scholar] [CrossRef]
- Merinero, R.; Lunar, R.; Martínez-Frías, J.; Somoza, L.; Díaz-del-Río, V. Iron oxyhydroxide and sulphide mineralization in hydrocarbon seep-related carbonate submarine chimneys, Gulf of Cadiz (SW Iberian Peninsula). Mar. Petrol. Geol. 2008, 25, 706–713. [Google Scholar] [CrossRef]
- Morris, R.V.; Ruff, S.W.; Gellert, R.; Ming, D.W.; Arvidson, R.E.; Clark, B.C.; Golden, D.C.; Siebach, K.; Klingelhöfer, G.; Schröder, C.; Fleischer, I.; Yen, A.S.; Squyres, S.W. Identification of carbonate-rich outcrops on Mars by the Spirit Rover. Science 2010, 329, 421–424. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Frias, J.; Delgado-Huertas, A.; García-Moreno, F.; Reyes, E.; Lunar, R; Rull, F. Oxygen and carbon isotopic signatures of extinct low-temperature hydrothermal chimneys in the Jaroso Mars analog. Planet. Space Sci. 2007, 55, 441–448. [Google Scholar] [CrossRef]
- Martínez-Frías, J.; Lunar, R.; Rodríguez-Losada, J.A.; Delgado, A. The volcanism-related multistage hydrothermal system of El Jaroso (SE Spain): Implications for the exploration of Mars Earth. Planet. Space 2004, 56, 5–8. [Google Scholar] [CrossRef]
- Sundquist, E.T.; Visser, K. The geologic history of the carbon cycle. In Treatise on Geochemistry; Holland, H.D., Turekian, K.K., Eds.; Elsevier: Amsterdam, The Netherlands, 2003; Volume 8, pp. 425–472. [Google Scholar]
- Morse, J.W. Sediments, Diagenesis and Sedimentary Rocks; Mackenzie, F.T., Ed.; Elsevier: Amsterdam, The Netherlands, 2005; Volume 7, pp. 67–85. [Google Scholar]
- Shitashima, K. CO2 supply from deep-sea hydrothermal systems. Waste Manag. 1998, 17, 385–390. [Google Scholar] [CrossRef]
- Millero, F.J. The Marine inorganic carbon cycle. Chem. Rev. 2007, 107, 308–341. [Google Scholar] [CrossRef] [PubMed]
- Krumbein, W.E. How and where on Earth were and are carbonate minerals formed? Geochim. Cosmochim. Acta 2009, 73, A699. [Google Scholar]
- Lodders, K.; Feggley, B., Jr. The Planetary Scientist’s Companion; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Loáiciga, H.A. Modern-age buildup of CO2 and its effects on seawater acidity and salinity. Geophys. Res. Let. 2006, 33, L10605. [Google Scholar] [CrossRef]
- Schultz, R.; Lin, J. Three-dimensional normal faulting models of the Valles Marineris, Mars, and geodynamic implications. J. Geophys. Res. 2001, 106, 16549–16566. [Google Scholar] [CrossRef]
- McKay, C.P. The deep biosphere: Lessons for planetary exploration. In Subsurface Microbiology and Biogeochemistry; Fredickson, J.K., Fletcher, M., Eds.; Wiley-Liss: New York, NY, USA, 2001; pp. 315–328. [Google Scholar]
- Reeburgh, W.S. Methane consumption in Cariaco Trench waters and sediments. Earth Planet. Sci. Let. 1976, 28, 337–344. [Google Scholar] [CrossRef]
- Konhauser, K. Introduction to Geomicrobiology; Blackwell Publishing: Malden, MA, USA, 2007; Chapter 2. [Google Scholar]
- Fernández-Remolar, D.C.; Prieto-Ballesteros, O.; Rodríguez, N.; Gómez, F.; Amils, R.; Gómez-Elvira, J.; Stoker, C.R. Underground habitats in the Río Tinto Basin: A model for subsurface life habitats on Mars. Astrobiology 2008, 8, 1023–1047. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Remolar, D.C.; Sánchez-Román, M.; Rodríguez, N.; Amils, R.; Romanek, C.S. The association of carbonate minerals to acidic environments: A possible biosignature for Mars 2009. In Proceedings of the 40th Lunar and Planetary Science Conference, The Woodlands, TX, USA, 23–27 March 2009.
- Tosca, N.J.; Knoll, A.H. Juvenile chemical sediments and the long term persistence of water at the surface of Mars. Earth Planet. Sci. Let. 2009, 286, 379–386. [Google Scholar] [CrossRef]
- Allwood, A.C.; Walter, M.R.; Kamber, B.S.; Marshall, C.P.; Burch, I.W. Stromatolite reef from the Early Archaean era of Australia. 2006, 441, 714–718. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fernández-Remolar, D.C.; Sánchez-Román, M.; Amils, R. The Case of the Lacking Carbonates and the Emergence of Early Life on Mars. Sustainability 2010, 2, 2541-2554. https://doi.org/10.3390/su2082541
Fernández-Remolar DC, Sánchez-Román M, Amils R. The Case of the Lacking Carbonates and the Emergence of Early Life on Mars. Sustainability. 2010; 2(8):2541-2554. https://doi.org/10.3390/su2082541
Chicago/Turabian StyleFernández-Remolar, David Carlos, Mónica Sánchez-Román, and Ricardo Amils. 2010. "The Case of the Lacking Carbonates and the Emergence of Early Life on Mars" Sustainability 2, no. 8: 2541-2554. https://doi.org/10.3390/su2082541
APA StyleFernández-Remolar, D. C., Sánchez-Román, M., & Amils, R. (2010). The Case of the Lacking Carbonates and the Emergence of Early Life on Mars. Sustainability, 2(8), 2541-2554. https://doi.org/10.3390/su2082541




