Toxicological Response of Zebrafish Exposed to Cocktails of Polymeric Materials and Valproic Acid
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Note
2.2. Animal Maintenance
2.3. Experimental Design
2.3.1. Novel Tank Test
2.3.2. Social Preference Test
2.3.3. Aggressivity Test
2.3.4. Oxidative Stress Analysis
2.4. Statistical Analysis
3. Results
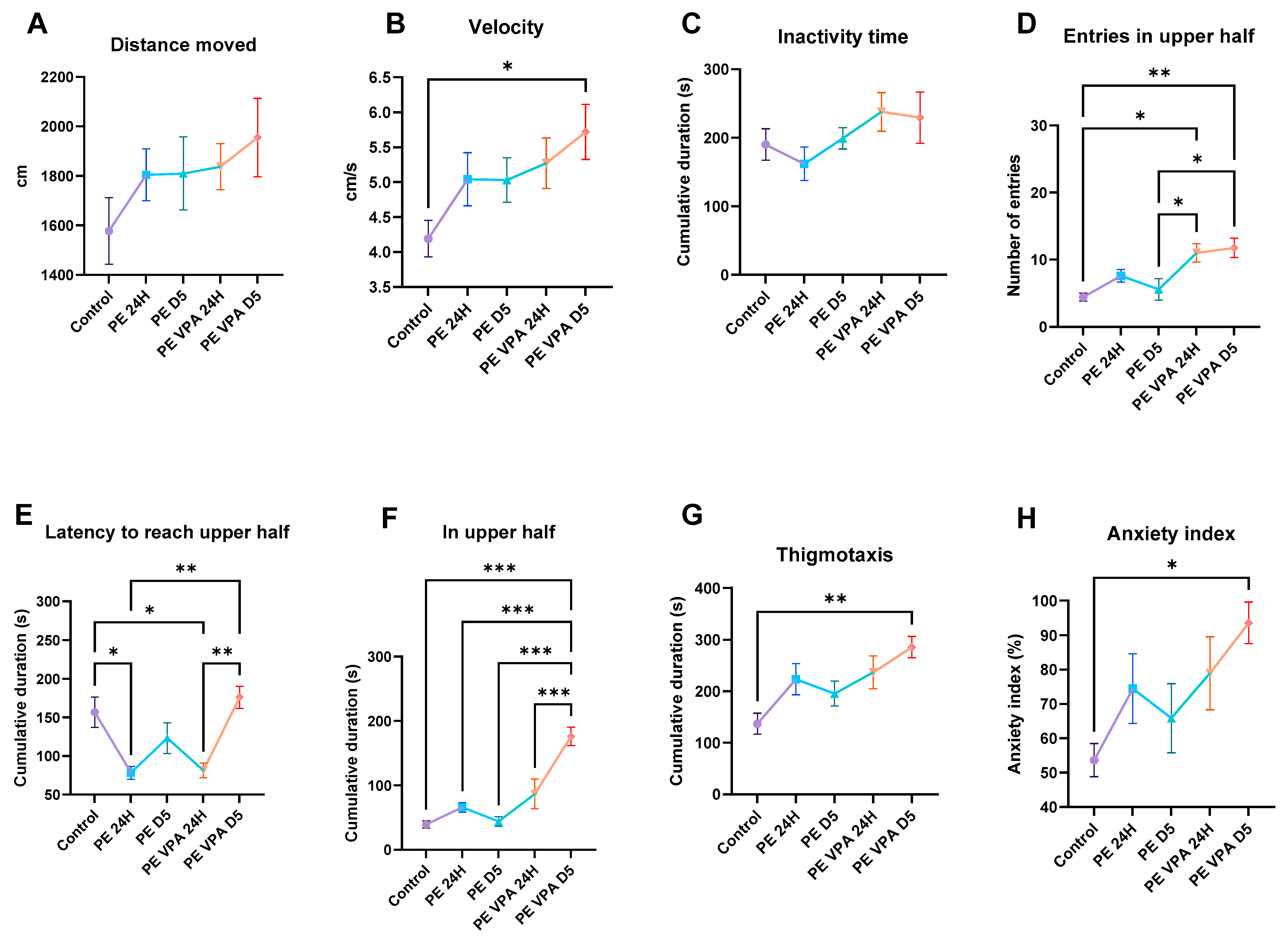
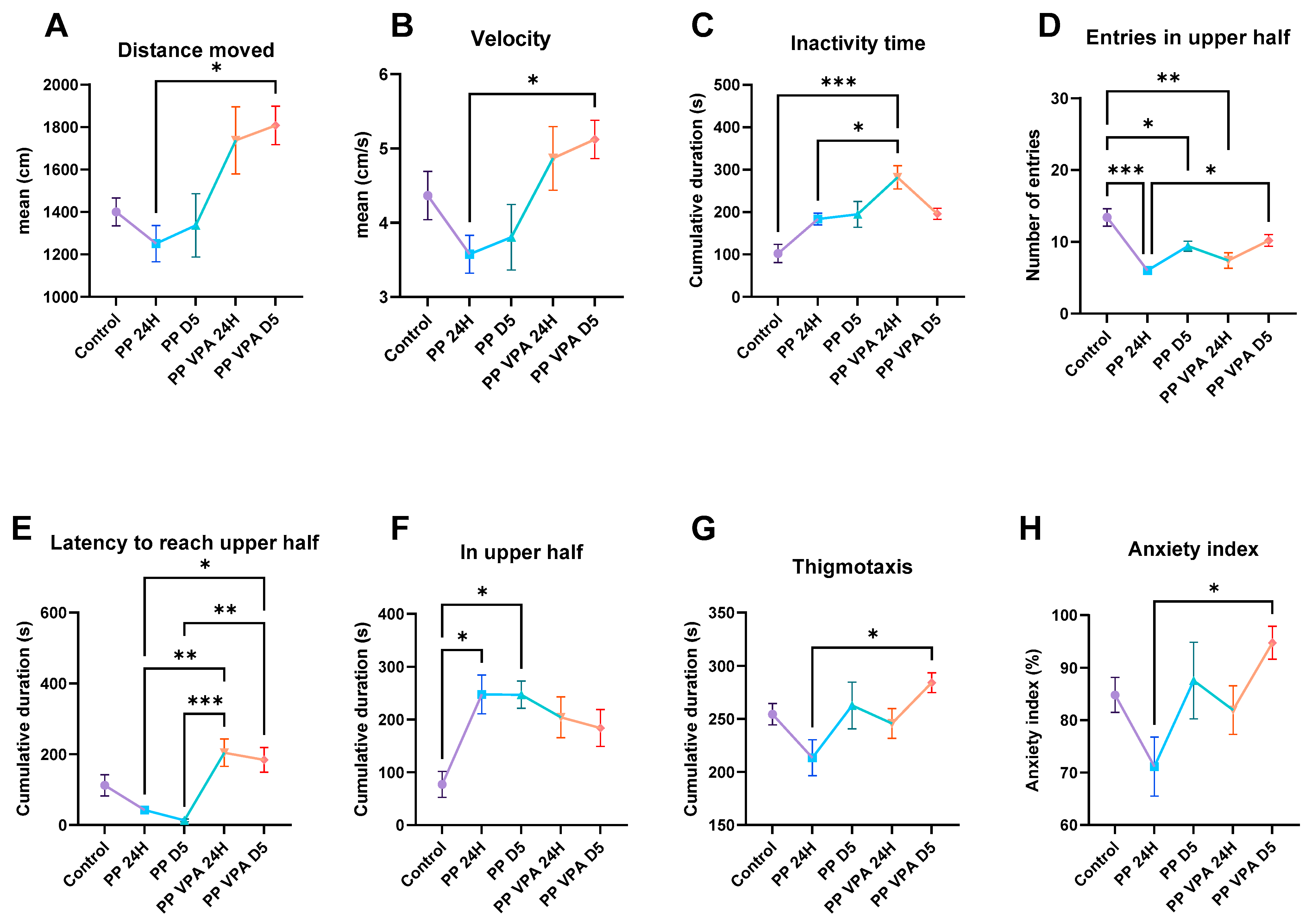
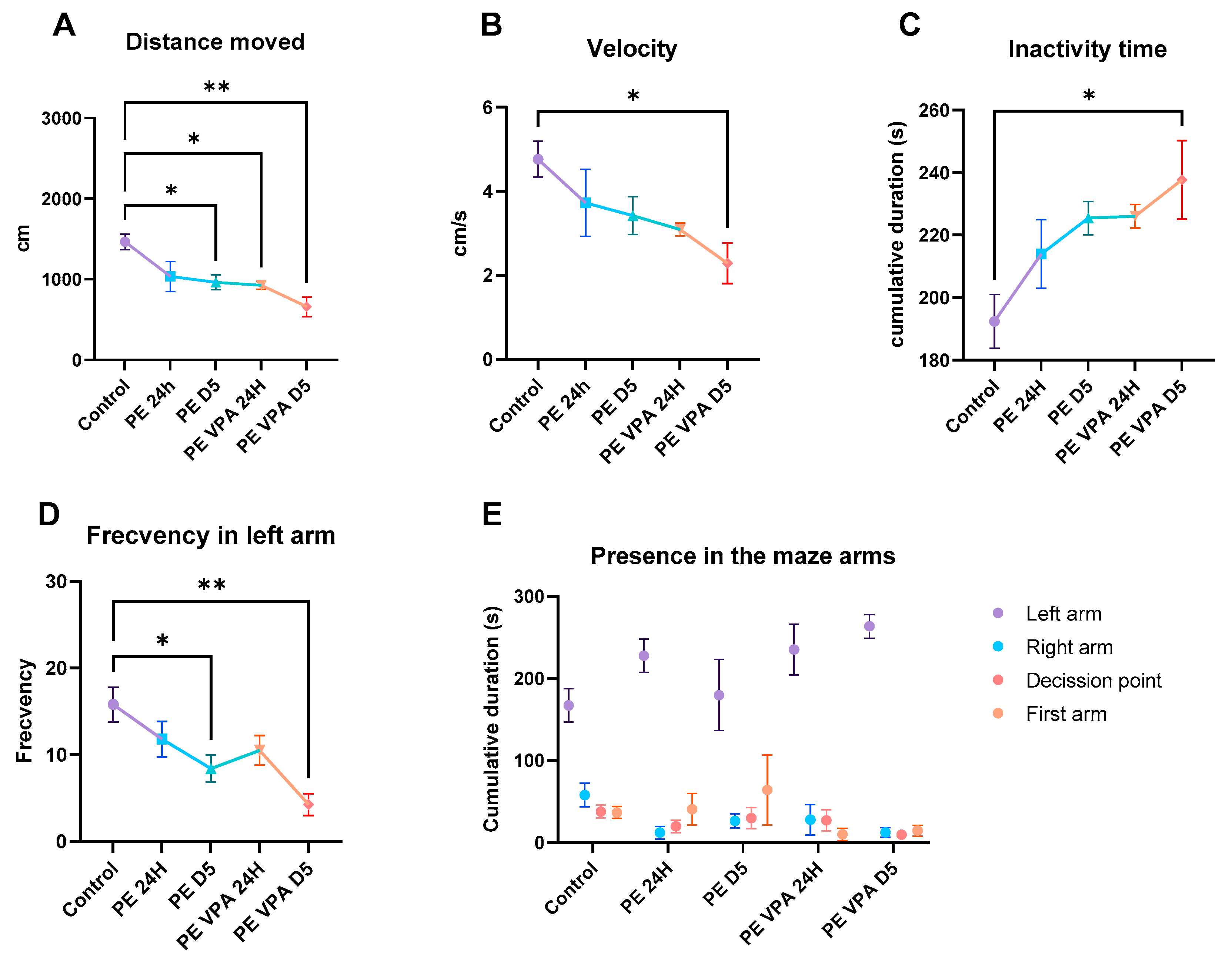
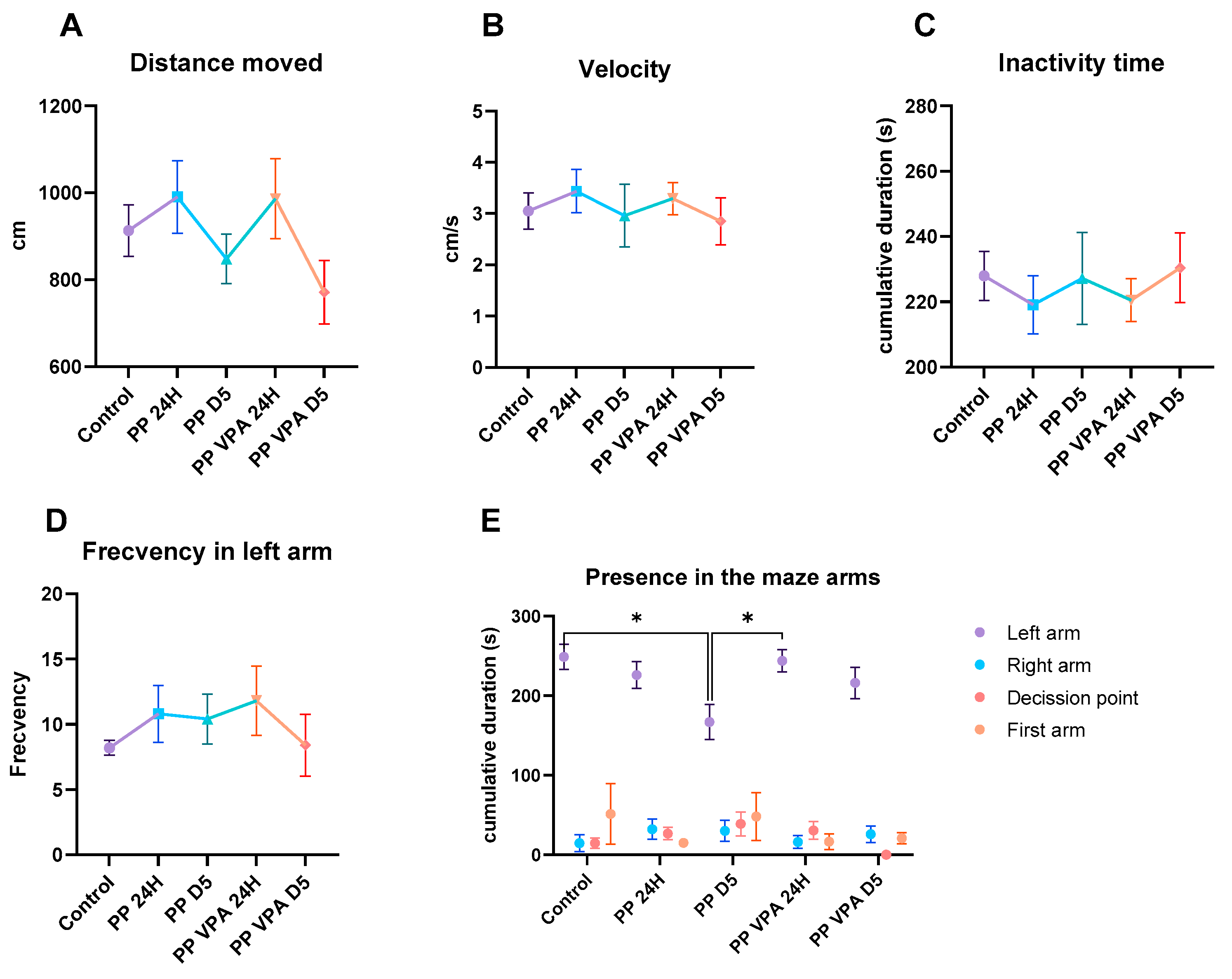
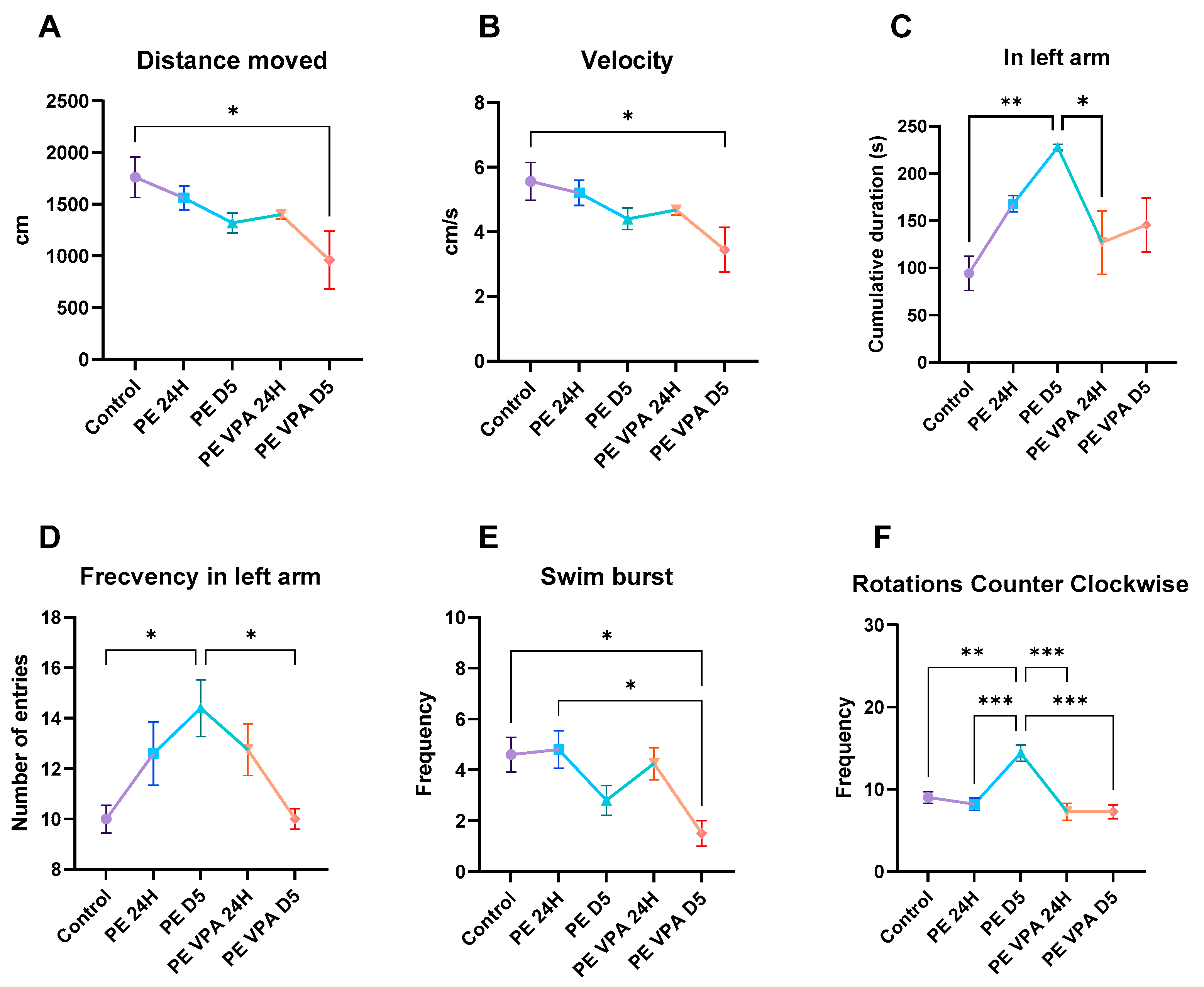
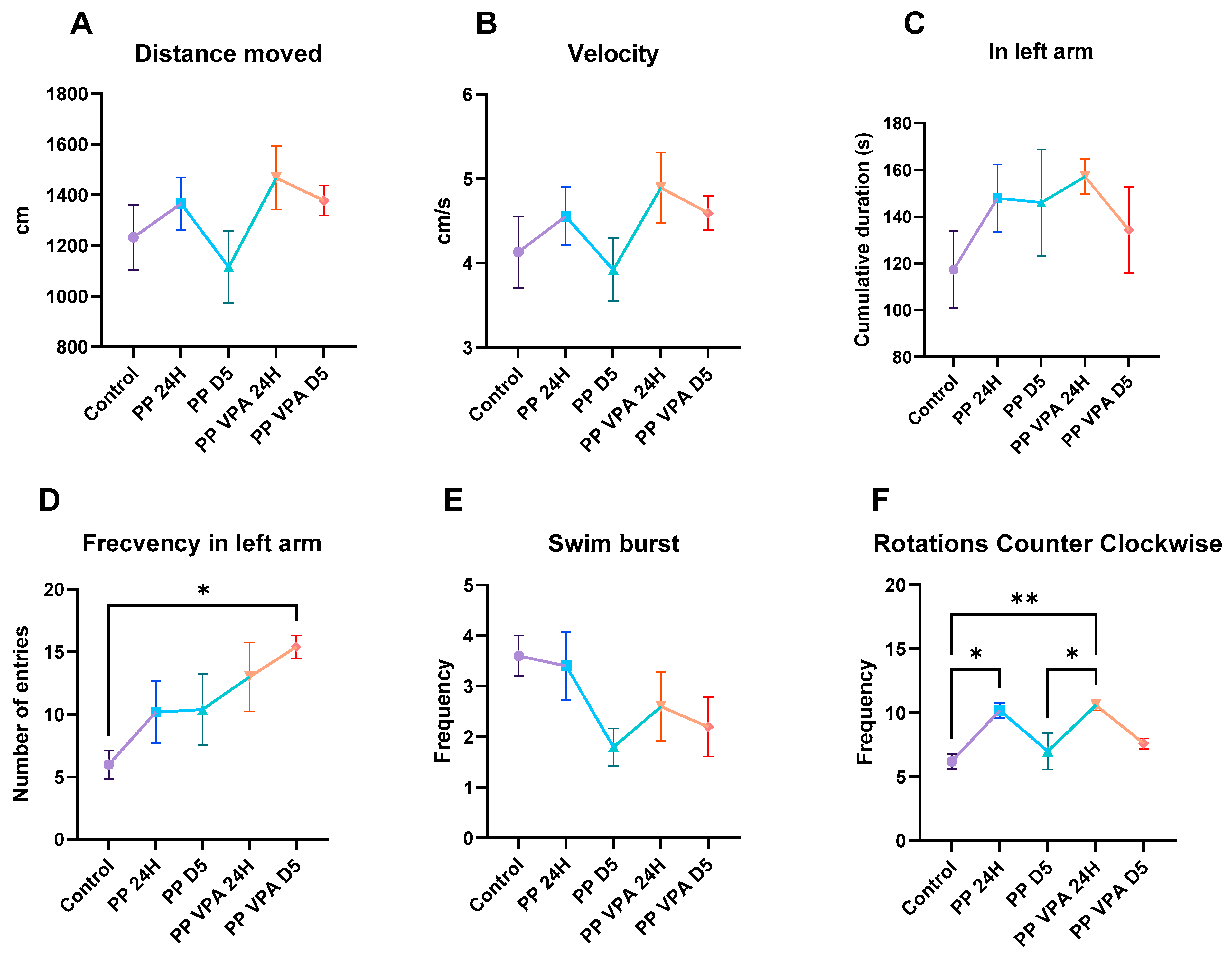
3.1. Behavior Analysis
3.1.1. Novel Tank Test
3.1.2. Social Preference Test
3.1.3. Aggressivity Test
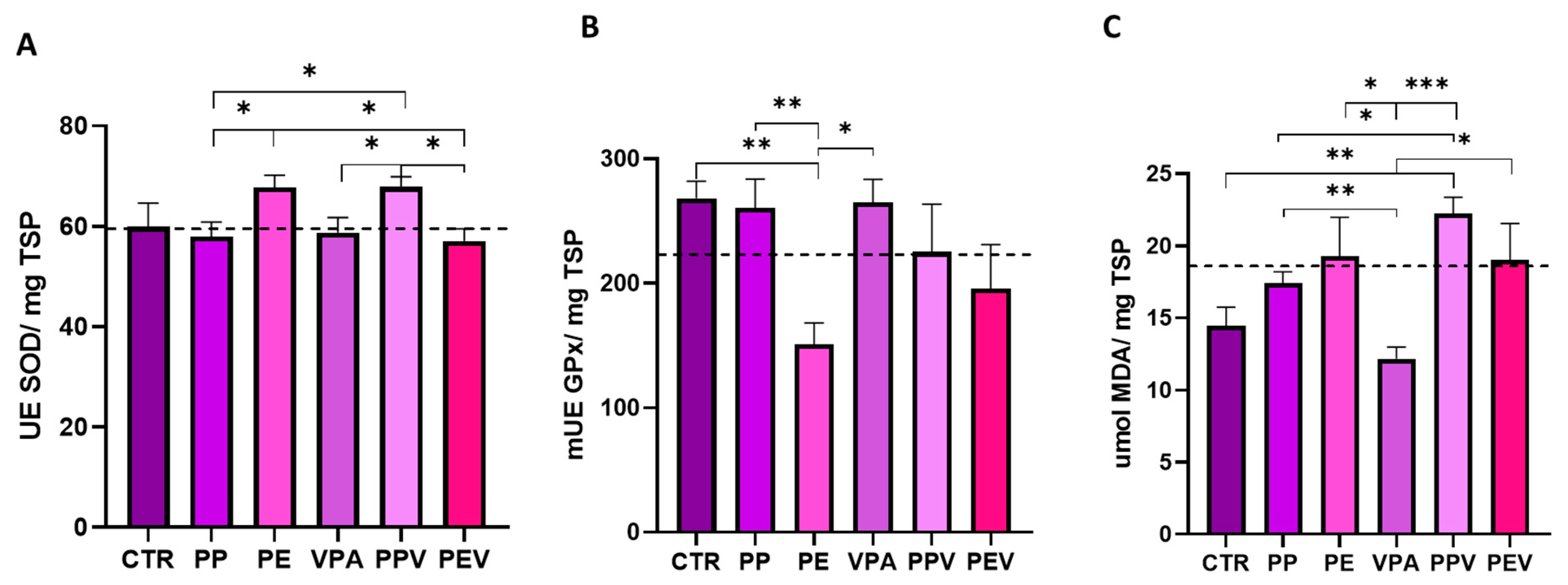
3.2. Oxidative Stress Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Henderson, L.; Green, C. Making Sense of Microplastics? Public Understandings of Plastic Pollution. Mar. Pollut. Bull. 2020, 152, 110908. [Google Scholar] [CrossRef]
- Zhang, B.; Chen, L.; Chao, J.; Yang, X.; Wang, Q. Research Progress of Microplastics in Freshwater Sediments in China. Environ. Sci. Pollut. Res. 2020, 27, 31046–31060. [Google Scholar] [CrossRef] [PubMed]
- Nanthini Devi, K.; Raju, P.; Santhanam, P.; Dinesh Kumar, S.; Krishnaveni, N.; Roopavathy, J.; Perumal, P. Biodegradation of Low-Density Polyethylene and Polypropylene by Microbes Isolated from Vaigai River, Madurai, India. Arch. Microbiol. 2021, 203, 6253–6265. [Google Scholar] [CrossRef]
- Wilkinson, J.L.; Boxall, A.B.A.; Kolpin, D.W.; Leung, K.M.Y.; Lai, R.W.S.; Galbán-Malagón, C.; Adell, A.D.; Mondon, J.; Metian, M.; Marchant, R.A.; et al. Pharmaceutical Pollution of the World’s Rivers. Proc. Natl. Acad. Sci. USA 2022, 119, e2113947119. [Google Scholar] [CrossRef]
- Desai, M.; Njoku, A.; Nimo-Sefah, L. Comparing Environmental Policies to Reduce Pharmaceutical Pollution and Address Disparities. Int. J. Enviorn. Res. Public Health 2022, 19, 8292. [Google Scholar] [CrossRef]
- Onesios-Barry, K.M.; Berry, D.; Proescher, J.B.; Sivakumar, I.K.A.; Bouwer, E.J. Removal of Pharmaceuticals and Personal Care Products during Water Recycling: Microbial Community Structure and Effects of Substrate Concentration. Appl. Enviorn. Microbiol. 2014, 80, 2440–2450. [Google Scholar] [CrossRef] [PubMed]
- Quesada, H.B.; Baptista, A.T.A.; Cusioli, L.F.; Seibert, D.; de Oliveira Bezerra, C.; Bergamasco, R. Surface Water Pollution by Pharmaceuticals and an Alternative of Removal by Low-Cost Adsorbents: A Review. Chemosphere 2019, 222, 766–780. [Google Scholar] [CrossRef]
- Klimaszyk, P.; Rzymski, P. Water and Aquatic Fauna on Drugs: What Are the Impacts of Pharmaceutical Pollution? In Water Management and the Environment: Case Studies; Springer: Cham, Switzerland, 2018; pp. 255–278. [Google Scholar]
- Vatovec, C.; Van Wagoner, E.; Evans, C. Investigating Sources of Pharmaceutical Pollution: Survey of over-the-Counter and Prescription Medication Purchasing, Use, and Disposal Practices among University Students. J. Enviorn. Manag. 2017, 198, 348–352. [Google Scholar] [CrossRef] [PubMed]
- Anjanapriya, S.; SulaimanMumtaz, M.; Mohideen, M.H.A.K.; Radha, A.; Sasirekha, N.; Sawicka, B.; Tamizhazhagan, V. Pharmaceutical Pollution Crisis in the World: A Menace to Ecosystem. Entomol. Appl. Sci. Lett. 2021, 8, 77–89. [Google Scholar] [CrossRef]
- Hsu, H.-C.; Tseng, H.-K.; Wang, S.-C.; Wang, Y.-Y. Valproic Acid-Induced Agranulocytosis. Int. J. Gerontol. 2009, 3, 137–139. [Google Scholar] [CrossRef][Green Version]
- Terbach, N.; Shah, R.; Kelemen, R.; Klein, P.S.; Gordienko, D.; Brown, N.A.; Wilkinson, C.J.; Williams, R.S.B. Identifying an Uptake Mechanism for the Antiepileptic and Bipolar Disorder Treatment Valproic Acid Using the Simple Biomedical Model Dictyostelium. J. Cell Sci. 2011, 124, 2267–2276. [Google Scholar] [CrossRef]
- Triyasakorn, K.; Ubah, U.D.B.; Roan, B.; Conlin, M.; Aho, K.; Awale, P.S. The Antiepileptic Drug and Toxic Teratogen Valproic Acid Alters Microglia in an Environmental Mouse Model of Autism. Toxics 2022, 10, 379. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, Y.-H.; Yun, J.-S.; Lee, C.-J. Valproic Acid Decreases the Proliferation of Telencephalic Cells in Zebrafish Larvae. Neurotoxicol. Teratol. 2013, 39, 91–99. [Google Scholar] [CrossRef]
- Kus, K.; Burda, K.; Nowakowska, E.; Czubak, A.; Metelska, J.; Łancucki, M.; Brodowska, K.; Nowakowska, A. Effect of Valproic Acid and Environmental Enrichment on Behavioral Functions in Rats. Arzneimittelforschung 2011, 60, 471–478. [Google Scholar] [CrossRef]
- Brotzmann, K.; Escher, S.E.; Walker, P.; Braunbeck, T. Potential of the Zebrafish (Danio rerio) Embryo Test to Discriminate between Chemicals of Similar Molecular Structure—A Study with Valproic Acid and 14 of Its Analogues. Arch. Toxicol. 2022, 96, 3033–3051. [Google Scholar] [CrossRef]
- Romoli, M.; Mazzocchetti, P.; D’Alonzo, R.; Siliquini, S.; Rinaldi, V.E.; Verrotti, A.; Calabresi, P.; Costa, C. Valproic Acid and Epilepsy: From Molecular Mechanisms to Clinical Evidences. Curr. Neuropharmacol. 2019, 17, 926–946. [Google Scholar] [CrossRef]
- Lee, S.; Chun, H.-S.; Lee, J.; Park, H.-J.; Kim, K.-T.; Kim, C.-H.; Yoon, S.; Kim, W.-K. Plausibility of the Zebrafish Embryos/Larvae as an Alternative Animal Model for Autism: A Comparison Study of Transcriptome Changes. PLoS ONE 2018, 13, e0203543. [Google Scholar] [CrossRef]
- Al-Khazrajy, O.S.A.; Boxall, A.B.A. Risk-Based Prioritization of Pharmaceuticals in the Natural Environment in Iraq. Environ. Sci. Pollut. Res. 2016, 23, 15712–15726. [Google Scholar] [CrossRef] [PubMed]
- Perazzolo, C.; Morasch, B.; Kohn, T.; Magnet, A.; Thonney, D.; Chèvre, N. Occurrence and Fate of Micropollutants in the Vidy Bay of Lake Geneva, Switzerland. Part I: Priority List for Environmental Risk Assessment of Pharmaceuticals. Enviorn. Toxicol. Chem. 2010, 29, 1649–1657. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.T.; Bisceglia, K.J.; Bouwer, E.J.; Roberts, A.L.; Coelhan, M. Determination of Pharmaceuticals and Antiseptics in Water by Solid-Phase Extraction and Gas Chromatography/Mass Spectrometry: Analysis via Pentafluorobenzylation and Stable Isotope Dilution. Anal. Bioanal. Chem. 2012, 403, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.T.; Bouwer, E.J.; Coelhan, M. Occurrence and Biodegradability Studies of Selected Pharmaceuticals and Personal Care Products in Sewage Effluent. Agric. Water Manag. 2006, 86, 72–80. [Google Scholar] [CrossRef]
- Collier, A.D.; Khan, K.M.; Caramillo, E.M.; Mohn, R.S.; Echevarria, D.J. Zebrafish and Conditioned Place Preference: A Translational Model of Drug Reward. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2014, 55, 16–25. [Google Scholar] [CrossRef]
- de Abreu, M.S.; Costa, F.; Giacomini, A.C.V.V.; Demin, K.A.; Zabegalov, K.N.; Maslov, G.O.; Kositsyn, Y.M.; Petersen, E.V.; Strekalova, T.; Rosemberg, D.B.; et al. Towards Modeling Anhedonia and Its Treatment in Zebrafish. Int. J. Neuro-Psychopharmacol. 2022, 25, 293–306. [Google Scholar] [CrossRef]
- Cueto-Escobedo, J.; German-Ponciano, L.J.; Guillén-Ruiz, G.; Soria-Fregozo, C.; Herrera-Huerta, E.V. Zebrafish as a Useful Tool in the Research of Natural Products With Potential Anxiolytic Effects. Front. Behav. Neurosci. 2022, 15, 795285. [Google Scholar] [CrossRef]
- Chateauvieux, S.; Morceau, F.; Dicato, M.; Diederich, M. Molecular and Therapeutic Potential and Toxicity of Valproic Acid. J. Biomed. Biotechnol. 2010, 2010, 479364. [Google Scholar] [CrossRef]
- Singh, D.; Gupta, S.; Verma, I.; Morsy, M.A.; Nair, A.B.; Ahmed, A.-S.F. Hidden Pharmacological Activities of Valproic Acid: A New Insight. Biomed. Pharmacother. 2021, 142, 112021. [Google Scholar] [CrossRef]
- Gago-Ferrero, P.; Bletsou, A.A.; Damalas, D.E.; Aalizadeh, R.; Alygizakis, N.A.; Singer, H.P.; Hollender, J.; Thomaidis, N.S. Wide-Scope Target Screening of >2000 Emerging Contaminants in Wastewater Samples with UPLC-Q-ToF-HRMS/MS and Smart Evaluation of Its Performance through the Validation of 195 Selected Representative Analytes. J. Hazard. Mater. 2020, 387, 121712. [Google Scholar] [CrossRef]
- Gorovits, R.; Sobol, I.; Akama, K.; Chefetz, B.; Czosnek, H. Pharmaceuticals in Treated Wastewater Induce a Stress Response in Tomato Plants. Sci. Rep. 2020, 10, 1856. [Google Scholar] [CrossRef] [PubMed]
- Cachat, J.M.; Canavello, P.R.; Elkhayat, S.I.; Bartels, B.K.; Hart, P.C.; Elegante, M.F.; Beeson, E.C.; Laffoon, A.L.; Haymore, W.A.M.; Tien, D.H.; et al. Video-Aided Analysis of Zebrafish Locomotion and Anxiety-Related Behavioral Responses. Neuromethods 2011, 51, 1–14. [Google Scholar] [CrossRef]
- Freitas, Í.N.; Dourado, A.V.; Araújo, A.P.D.C.; de Souza, S.S.; da Luz, T.M.; Guimarães, A.T.B.; Gomes, A.R.; Islam, A.R.M.D.T.; Rahman, M.D.M.; Arias, A.H.; et al. Toxicity Assessment of SARS-CoV-2-Derived Peptides in Combination with a Mix of Pollutants on Zebrafish Adults: A Perspective Study of Behavioral, Biometric, Mutagenic, and Biochemical Toxicity. Sci. Total Environ. 2023, 858, 159838. [Google Scholar] [CrossRef] [PubMed]
- Saverino, C.; Gerlai, R. The Social Zebrafish: Behavioral Responses to Conspecific, Heterospecific, and Computer Animated Fish. Behav. Brain Res. 2008, 191, 77–87. [Google Scholar] [CrossRef]
- Ogi, A.; Licitra, R.; Naef, V.; Marchese, M.; Fronte, B.; Gazzano, A.; Santorelli, F.M. Social Preference Tests in Zebrafish: A Systematic Review. Front. Vet. Sci. 2021, 7, 590057. [Google Scholar] [CrossRef] [PubMed]
- Ilie, O.-D.; Duta, R.; Jijie, R.; Nita, I.-B.; Nicoara, M.; Faggio, C.; Dobrin, R.; Mavroudis, I.; Ciobica, A.; Doroftei, B. Assessing Anti-Social and Aggressive Behavior in a Zebrafish (Danio rerio) Model of Parkinson’s Disease Chronically Exposed to Rotenone. Brain Sci. 2022, 12, 898. [Google Scholar] [CrossRef] [PubMed]
- Way, G.P.; Southwell, M.; McRobert, S.P. Boldness, Aggression, and Shoaling Assays for Zebrafish Behavioral Syndromes. J. Vis. Exp. 2016, 2016, 54049. [Google Scholar] [CrossRef]
- Balmus, I.-M.; Lefter, R.; Ciobica, A.; Cojocaru, S.; Guenne, S.; Timofte, D.; Stanciu, C.; Trifan, A.; Hritcu, L. Preliminary Biochemical Description of Brain Oxidative Stress Status in Irritable Bowel Syndrome Contention-Stress Rat Model. Medicina 2019, 55, 776. [Google Scholar] [CrossRef] [PubMed]
- Meshalkina, D.A.; Kizlyk, M.N.; Kysil, E.V.; Collier, A.D.; Echevarria, D.J.; Abreu, M.S.; Barcellos, L.J.G.; Song, C.; Warnick, J.E.; Kyzar, E.J.; et al. Zebrafish Models of Autism Spectrum Disorder. Exp. Neurol. 2018, 299, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, P. Zebrafish as a Pharmacological Tool: The How, Why and When. Curr. Opin. Pharmacol. 2004, 4, 504–512. [Google Scholar] [CrossRef] [PubMed]
- Belo, M.A.A.; Oliveira, M.F.; Oliveira, S.L.; Aracati, M.F.; Rodrigues, L.F.; Costa, C.C.; Conde, G.; Gomes, J.M.M.; Prata, M.N.L.; Barra, A.; et al. Zebrafish as a Model to Study Inflammation: A Tool for Drug Discovery. Biomed. Pharmacother. 2021, 144, 112310. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, F.F.; Gaspary, K.V.; Leite, C.E.; De Paula Cognato, G.; Bonan, C.D. Embryological Exposure to Valproic Acid Induces Social Interaction Deficits in Zebrafish (Danio rerio): A Developmental Behavior Analysis. Neurotoxicol. Teratol. 2015, 52, 36–41. [Google Scholar] [CrossRef]
- Robea, M.A.; Ciobica, A.; Curpan, A.-S.; Plavan, G.; Strungaru, S.; Lefter, R.; Nicoara, M. Preliminary Results Regarding Sleep in a Zebrafish Model of Autism Spectrum Disorder. Brain Sci. 2021, 11, 556. [Google Scholar] [CrossRef]
- Joseph, T.P.; Zhou, F.; Sai, L.Y.; Chen, H.; Lin, S.L.; Schachner, M. Duloxetine Ameliorates Valproic acid-Induced Hyperactivity, Anxiety-like Behavior, and Social Interaction Deficits in Zebrafish. Autism. Res. 2022, 15, 27–41. [Google Scholar] [CrossRef]
- Li, L.; Bonneton, F.; Tohme, M.; Bernard, L.; Chen, X.Y.; Laudet, V. In Vivo Screening Using Transgenic Zebrafish Embryos Reveals New Effects of HDAC Inhibitors Trichostatin A and Valproic Acid on Organogenesis. PLoS ONE 2016, 11, e0149497. [Google Scholar] [CrossRef]
- Bailey, J.M.; Oliveri, A.N.; Karbhari, N.; Brooks, R.A.J.; De La Rocha, A.J.; Janardhan, S.; Levin, E.D. Persistent Behavioral Effects Following Early Life Exposure to Retinoic Acid or Valproic Acid in Zebrafish. Neurotoxicology 2016, 52, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Helinski, O.K.; Poor, C.J.; Wolfand, J.M. Ridding Our Rivers of Plastic: A Framework for Plastic Pollution Capture Device Selection. Mar. Pollut. Bull. 2021, 165, 112095. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shi, H.; Li, T.; Yu, L.; Qi, Y.; Tian, G.; He, F.; Li, X.; Sun, N.; Liu, R. Size-Dependent Effects of Nanoplastics on Structure and Function of Superoxide Dismutase. Chemosphere 2022, 309, 136768. [Google Scholar] [CrossRef]
- Ding, R.; Tong, L.; Zhang, W. Microplastics in Freshwater Environments: Sources, Fates and Toxicity. Water Air Soil Pollut. 2021, 232, 181. [Google Scholar] [CrossRef]
- Karuppasamy, P.K.; Ravi, A.; Vasudevan, L.; Elangovan, M.P.; Dyana Mary, P.; Vincent, S.G.T.; Palanisami, T. Baseline Survey of Micro and Mesoplastics in the Gastro-Intestinal Tract of Commercial Fish from Southeast Coast of the Bay of Bengal. Mar. Pollut. Bull. 2020, 153, 110974. [Google Scholar] [CrossRef] [PubMed]
- Olexová, L.; Štefánik, P.; Kršková, L. Increased Anxiety-like Behaviour and Altered GABAergic System in the Amygdala and Cerebellum of VPA Rats—An Animal Model of Autism. Neurosci. Lett. 2016, 629, 9–14. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Y.; Lin, J.; Xia, Q.; Guo, N.; Li, Q. Social Preference Deficits in Juvenile Zebrafish Induced by Early Chronic Exposure to Sodium Valproate. Front. Behav. Neurosci. 2016, 10, 201. [Google Scholar] [CrossRef]
- Younus, H. Therapeutic Potentials of Superoxide Dismutase. Int. J. Health Sci. 2018, 12, 88–93. [Google Scholar]
- Félix, L.; Carreira, P.; Peixoto, F. Effects of Chronic Exposure of Naturally Weathered Microplastics on Oxidative Stress Level, Behaviour, and Mitochondrial Function of Adult Zebrafish (Danio rerio). Chemosphere 2023, 310, 136895. [Google Scholar] [CrossRef]
- Lubos, E.; Loscalzo, J.; Handy, D.E. Glutathione Peroxidase-1 in Health and Disease: From Molecular Mechanisms to Therapeutic Opportunities. Antioxid. Redox. Signal. 2011, 15, 1957. [Google Scholar] [CrossRef] [PubMed]
- Mak, C.W.; Ching-Fong Yeung, K.; Chan, K.M. Acute Toxic Effects of Polyethylene Microplastic on Adult Zebrafish. Ecotoxicol. Enviorn. Saf. 2019, 182, 109442. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Stewart, A.J.; Pellegrini, N. A Review of Recent Studies on Malondialdehyde as Toxic Molecule and Biological Marker of Oxidative Stress. Nutr. Metab. Cardiovasc. Dis. 2005, 15, 316–328. [Google Scholar] [CrossRef] [PubMed]
- Sheng, C.; Zhang, S.; Zhang, Y. The Influence of Different Polymer Types of Microplastics on Adsorption, Accumulation, and Toxicity of Triclosan in Zebrafish. J. Hazard. Mater. 2021, 402, 123733. [Google Scholar] [CrossRef]
- Bobori, D.C.; Feidantsis, K.; Dimitriadi, A.; Datsi, N.; Ripis, P.; Kalogiannis, S.; Sampsonidis, I.; Kastrinaki, G.; Ainali, N.M.; Lambropoulou, D.A.; et al. Dose-Dependent Cytotoxicity of Polypropylene Microplastics (PP-MPs) in Two Freshwater Fishes. Int. J. Mol. Sci. 2022, 23, 13878. [Google Scholar] [CrossRef]
- Xiao, S.; Cui, Y.; Brahney, J.; Mahowald, N.M.; Li, Q. Long-Distance Atmospheric Transport of Microplastic Fibres Influenced by Their Shapes. Nat. Geosci. 2023, 16, 863–870. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savuca, A.; Chelaru, I.-A.; Balmus, I.-M.; Curpan, A.-S.; Nicoara, M.N.; Ciobica, A.S. Toxicological Response of Zebrafish Exposed to Cocktails of Polymeric Materials and Valproic Acid. Sustainability 2024, 16, 2057. https://doi.org/10.3390/su16052057
Savuca A, Chelaru I-A, Balmus I-M, Curpan A-S, Nicoara MN, Ciobica AS. Toxicological Response of Zebrafish Exposed to Cocktails of Polymeric Materials and Valproic Acid. Sustainability. 2024; 16(5):2057. https://doi.org/10.3390/su16052057
Chicago/Turabian StyleSavuca, Alexandra, Ionut-Alexandru Chelaru, Ioana-Miruna Balmus, Alexandrina-Stefania Curpan, Mircea Nicusor Nicoara, and Alin Stelian Ciobica. 2024. "Toxicological Response of Zebrafish Exposed to Cocktails of Polymeric Materials and Valproic Acid" Sustainability 16, no. 5: 2057. https://doi.org/10.3390/su16052057
APA StyleSavuca, A., Chelaru, I.-A., Balmus, I.-M., Curpan, A.-S., Nicoara, M. N., & Ciobica, A. S. (2024). Toxicological Response of Zebrafish Exposed to Cocktails of Polymeric Materials and Valproic Acid. Sustainability, 16(5), 2057. https://doi.org/10.3390/su16052057








