Assessing Ammonia and Greenhouse Gas Emissions from Livestock Manure Storage: Comparison of Measurements with Dynamic and Static Chambers
Abstract
:1. Introduction
- The mass balance approach: based on the difference between the total nitrogen manure content before and after the emission period considered.
- Indirect methods: these measure environmental concentrations and use dispersion models to calculate ER. The most common indirect methods are micrometeorological techniques, whereby gas emissions are calculated from a large footprint using tower-based instrumentation (e.g., gradient methods, eddy covariance, and the backward Lagrangian dispersion model).
- Direct methods: when emissions are measured using different devices, for example, wind tunnels, flux chambers (dynamic hoods), or static hoods. Samples are taken and analysed or directly measured using specific sensors.
2. Materials and Methods
2.1. Experiment Set-Up
2.2. Equipment Used
2.3. Emission Sampling and Calculation
2.3.1. Sampling Procedure
2.3.2. Emission Rate (ER) Calculation
2.4. Analytical Methods
3. Results and Discussion
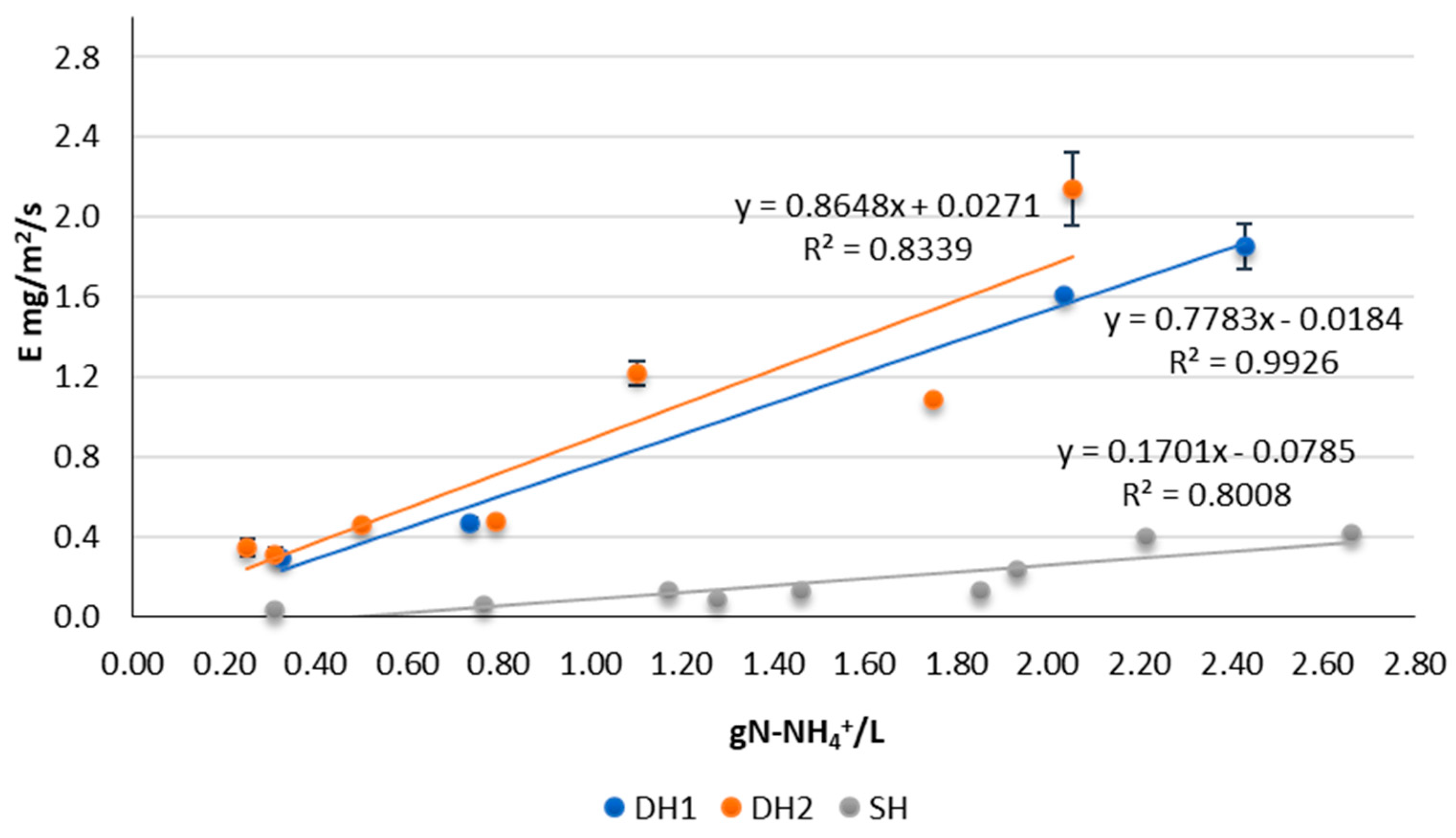
3.1. Ammonia Emissions from Ammonia Solution
3.2. Ammonia and GHG Emissions in Field Conditions
3.2.1. Manure By-Products Characterization
3.2.2. Ammonia Emissions
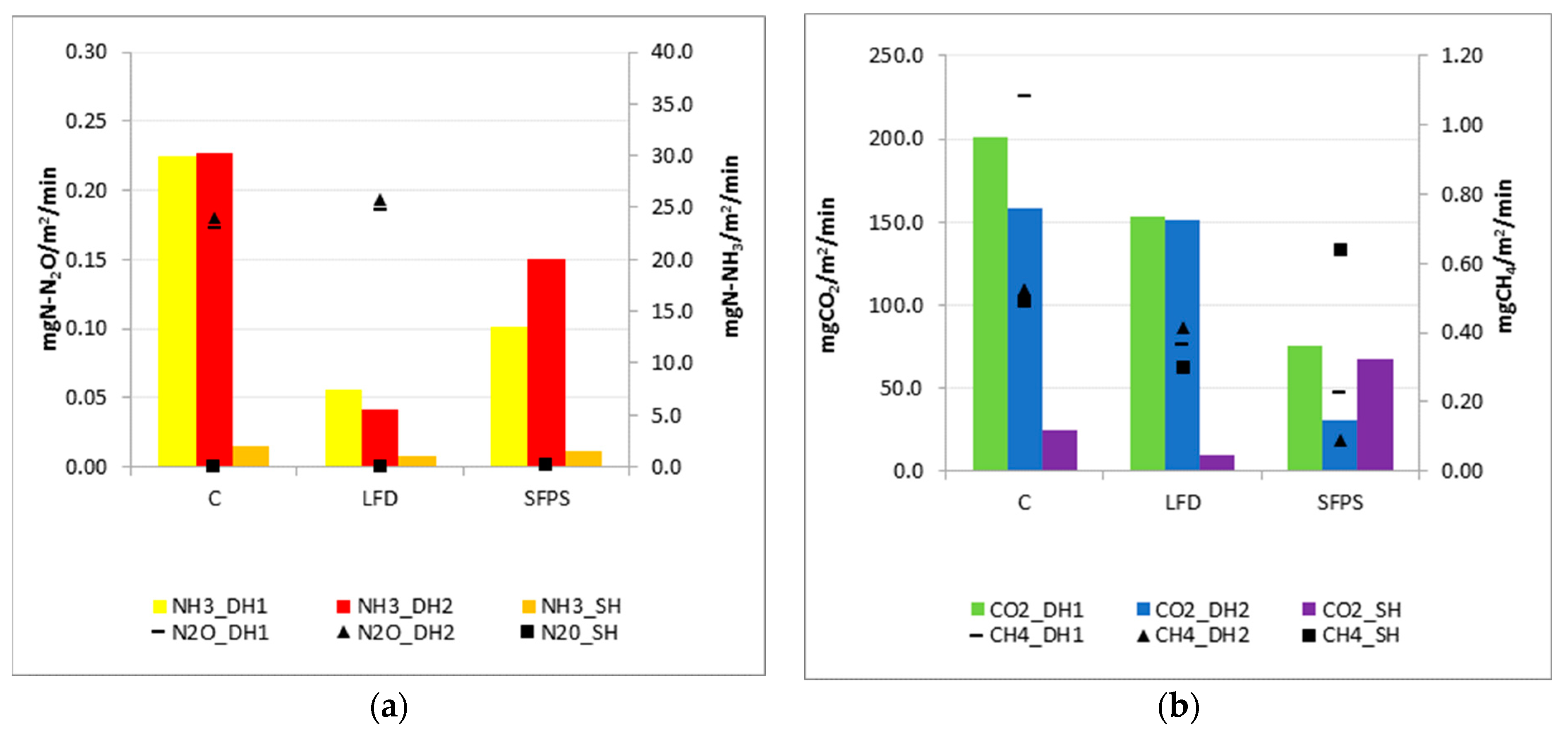
3.2.3. Greenhouse Gas Emissions
3.2.4. Implications of the Study and Future Recommendations
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Qi, J.; Yang, H.; Wang, X.; Zhu, H.; Wang, Z.; Zhao, C.; Li, B.; Liu, Z. State-of-the-art on animal manure pollution control and resource utilization. J. Environ. Chem. Eng. 2023, 11, 110462. [Google Scholar] [CrossRef]
- Kupper, T.; Häni, C.; Neftel, A.; Kincaid, C.; Bühler, M.; Amon, B.; VanderZaag, A. Ammonia and greenhouse gas emissions from slurry storage—A review. Agric. Ecosyst. Environ. 2020, 300, 106963. [Google Scholar] [CrossRef]
- Sommer, S.G.; Hafner, S.D.; Laubach, J.; van der Weerden, T.J.; Leytem, A.B.; Pacholski, A. Model for calculating ammonia emission from stored animal liquid manure. Biosyst. Eng. 2022, 223, 41–55. [Google Scholar] [CrossRef]
- European Environment Agency. Air Quality in Europe—2020 Report, EEA Technical Report; Publications Office of the European Union: Luxembourg, 2020. [CrossRef]
- Thangavel, P.; Park, D.; Lee, Y.-C. Recent Insights into Particulate Matter (PM2.5)-Mediated Toxicity in Humans: An Overview. Int. J. Environ. Res. Public Health 2022, 19, 7511. [Google Scholar] [CrossRef]
- Pai, S.J.; Heald, C.L.; Murphy, J.G. Exploring the Global Importance of Atmospheric Ammonia Oxidation. ACS Earth Space Chem. 2021, 5, 1674–1685. [Google Scholar] [CrossRef]
- Mielcarek-Bocheńska, P.; Rzeźnik, W. Greenhouse Gas Emissions from Agriculture in EU Countries—State and Perspectives. Atmosphere 2021, 12, 1396. [Google Scholar] [CrossRef]
- Montemayor, E.; Bonmatí, A.; Torrellas, M.; Camps, F.; Ortiz, C.; Domingo, F.; Riau, V.; Antón, A. Environmental accounting of closed-loop maize production scenarios: Manure as fertilizer and inclusion of catch crops. Resour. Conserv. Recycl. 2019, 146, 395–404. [Google Scholar] [CrossRef]
- Scotto di Perta, E.; Mautone, A.; Oliva, M.; Cervelli, E.; Pindozzi, S. Influence of Treatments and Covers on NH3 Emissions from Dairy Cow and Buffalo Manure Storage. Sustainability 2020, 12, 2986. [Google Scholar] [CrossRef]
- Scotto di Perta, E.; Fiorentino, N.; Carozzi, M.; Cervelli, E.; Pindozzi, S. A Review of Chamber and Micrometeorological Methods to Quantify NH3 Emissions from Fertilisers Field Application. Int. J. Agron. 2020, 2020, 8909784. [Google Scholar] [CrossRef]
- IPCC. 2019 Refinement to the 2006 IPCC Guidelines for National Greenhouse Gas Inventories; Calvo Buendia, E., Tanabe, K., Kranjc, A., Baasansuren, J., Fukuda, M., Ngarize, S., Osako, A., Pyrozhenko, Y., Shermanau, P., Federici, S., Eds.; IPCC: Geneva, Switzerland, 2019. [Google Scholar]
- Sommer, S.G.; Webb, J.; Hutchings, N.D. New Emission Factors for Calculation of Ammonia Volatilization From European Livestock Manure Management Systems. Front. Sustain. Food Syst. 2019, 3, 101. [Google Scholar] [CrossRef]
- Liu, L.; Abdala Prata Junior, A.; Fisher, R.M.; Stuetz, R.M. Measuring volatile emissions from biosolids: A critical review on sampling methods. J. Environ. Manag. 2022, 317, 115290. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ni, X.; Liu, B.; Tao, L.; Yu, L.; Wang, Q.; Yang, Y.; Liu, J.; Wu, Y. Measuring field ammonia emissions and canopy ammonia fluxes in agriculture using portable ammonia detector method. J. Clean. Prod. 2019, 216, 542–551. [Google Scholar] [CrossRef]
- Pihlatie, M.K.; Christiansen, J.R.; Aaltonen, H.; Korhonen, J.F.J.; Nordbo, A.; Rasilo, T.; Benanti, G.; Giebels, M.; Helmy, M.; Sheehy, J.; et al. Comparison of static chambers to measure CH4 emissions from soils. Agric. For. Meteorol. 2013, 171–172, 124–136. [Google Scholar] [CrossRef]
- Sabrina, J.; Nurulhuda, K.; Amin, A.M.; Sulaiman, M.F.; Man, H.C. Exploring use of a commercial passive sampler in a closed static chamber to measure ammonia volatilization. Environ. Pollut. 2022, 315, 120282. [Google Scholar] [CrossRef]
- Guo, C.; Ren, T.; Li, P.; Wang, B.; Zou, J.; Hussain, S.; Cong, R.; Wu, L.; Lu, J.; Li, X. Producing more grain yield of rice with less ammonia volatilization and greenhouse gases emission using slow/controlled-release urea. Environ. Sci. Pollut. Res. 2019, 26, 2569–2579. [Google Scholar] [CrossRef]
- Verdi, L.; Kuikman, P.J.; Orlandini, S.; Mancini, M.; Napoli, M.; Dalla Marta, A. Does the use of digestate to replace mineral fertilizers have less emissions of N2O and NH3? Agric. For. Meteorol. 2019, 269–270, 112–118. [Google Scholar] [CrossRef]
- Chen, J.; Li, W.; Qiao, P.; Li, Y.; Zheng, K.; Wang, Y.; Dong, X.; Wang, S.; Tan, L.; Chu, F.; et al. Characterizing ammonia emissions from water bodies using dynamic floating chambers. Sci. Total Environ. 2021, 796, 148978. [Google Scholar] [CrossRef]
- Torrellas, M.; Burgos, L.; Tey, L.; Noguerol, J.; Riau, V.; Palatsi, J.; Antón, A.; Flotats, X.; Bonmatí, A. Different approaches to assess the environmental performance of a cow manure biogas plant. Atmos. Environ. 2018, 177, 203–213. [Google Scholar] [CrossRef]
- Kim, M.-S.; Kim, J.-G. Effects of Thickness of Solid Media, Ventilation Rate, and Chamber Volume on Ammonia Emission from Liquid Fertilizers Using Dynamic Chamber-Capture System (DCS). Agriculture 2020, 10, 226. [Google Scholar] [CrossRef]
- Kim, M.-S.; Lee, Y.-S.; Min, H.-G.; Kim, J.-G. Applicability of the dynamic chamber-capture system (DCS) for estimating the flux of ammonia emission during liquid fertilizer spreading. Atmos. Pollut. Res. 2020, 11, 723–729. [Google Scholar] [CrossRef]
- Yang, J.; Jiao, Y.; Yang, W.Z.; Gu, P.; Bai, S.G.; Liu, L.J. Review of methods for determination of ammonia volatilization in farmland. IOP Conf. Ser. Earth Environ. Sci. 2018, 113, 12022. [Google Scholar] [CrossRef]
- Alexander, J.R.; Spackman, J.A.; Wilson, M.L.; Fernández, F.G.; Venterea, R.T. Capture efficiency of four chamber designs for measuring ammonia emissions. Agrosyst. Geosci. Environ. 2021, 4, e20199. [Google Scholar] [CrossRef]
- EPA. Odour Impacts and Odour Emission Control Measures for Intensive Agriculture; Environmental Research R&D Report Series No 14. Johnstown Castle Estate: Wexford, Ireland, 2001. [Google Scholar]
- Hernandez-Ramirez, G.; Brouder, S.M.; Smith, D.R.; Van Scoyoc, G.E. Carbon and Nitrogen Dynamics in an Eastern Corn Belt Soil: Nitrogen Source and Rotation. Soil Sci. Soc. Am. J. 2009, 73, 128–137. [Google Scholar] [CrossRef]
- Duchemin, E.; Lucotte, M.; Canuel, R. Comparison of static chamber and thin boundary layer equation methods for measuring greenhouse gas emissions from large water bodies. Environ. Sci. Technol. 1999, 33, 350–357. [Google Scholar] [CrossRef]
- Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2017.
- Misselbrook, T.; Hunt, J.; Perazzolo, F.; Provolo, G. Greenhouse Gas and Ammonia Emissions from Slurry Storage: Impacts of Temperature and Potential Mitigation through Covering (Pig Slurry) or Acidification (Cattle Slurry). J. Environ. Qual. 2016, 45, 1520–1530. [Google Scholar] [CrossRef]
- Sommer, S.G.; Christensen, M.L.; Schmidt, T.; Jensen, L.S. Animal Manure Recycling: Treatment and Management; John Wiley & Sons Ltd.: Chichester, UK, 2013; ISBN 9781118676677. [Google Scholar]
- Sommer, S.G.; Génermont, S.; Cellier, P.; Hutchings, N.J.; Olesen, J.E.; Morvan, T. Processes controlling ammonia emission from livestock slurry in the field. Eur. J. Agron. 2003, 19, 465–486. [Google Scholar] [CrossRef]
- Shah, S.B.; Westerman, P.W.; Arogo, J. Measuring ammonia concentrations and emissions from agricultural land and liquid surfaces: A review. J. Air Waste Manag. Assoc. 2006, 56, 945–960. [Google Scholar] [CrossRef]
- Sommer, S.G.; McGinn, S.M.; Hao, X.; Larney, F.J. Techniques for measuring gas emissions from a composting stockpile of cattle manure. Atmos. Environ. 2004, 38, 4643–4652. [Google Scholar] [CrossRef]
- Miola, E.C.C.; Aita, C.; Rochette, P.; Chantigny, M.H.; Angers, D.A.; Bertrand, N.; Gasser, M.-O. Static Chamber Measurements of Ammonia Volatilization from Manured Soils: Impact of Deployment Duration and Manure Characteristics. Soil Sci. Soc. Am. J. 2015, 79, 305–313. [Google Scholar] [CrossRef]
- Livingston, G.P.; Hutchinson, G.L.; Spartalian, K. Trace Gas Emission in Chambers. Soil Sci. Soc. Am. J. 2006, 70, 1459–1469. [Google Scholar] [CrossRef]
- Dinuccio, E.; Berg, W.; Balsari, P. Gaseous emissions from the storage of untreated slurries and the fractions obtained after mechanical separation. Atmos. Environ. 2008, 42, 2448–2459. [Google Scholar] [CrossRef]
- Baldé, H.; VanderZaag, A.C.; Burtt, S.D.; Wagner-Riddle, C.; Evans, L.; Gordon, R.; Desjardins, R.L.; MacDonald, J.D. Ammonia emissions from liquid manure storages are affected by anaerobic digestion and solid-liquid separation. Agric. For. Meteorol. 2018, 258, 80–88. [Google Scholar] [CrossRef]
- Tang, R.; Liu, Y.; Ma, R.; Zhang, L.; Li, Y.; Li, G.; Wang, D.; Lin, J.; Li, Q.; Yuan, J. Effect of moisture content, aeration rate, and C/N on maturity and gaseous emissions during kitchen waste rapid composting. J. Environ. Manag. 2023, 326, 116662. [Google Scholar] [CrossRef] [PubMed]
- Perazzolo, F.; Mattachini, G.; Tambone, F.; Misselbrook, T.; Provolo, G. Effect of mechanical separation on emissions during storage of two anaerobically codigested animal slurries. Agric. Ecosyst. Environ. 2015, 207, 1–9. [Google Scholar] [CrossRef]
- Petersen, S.O.; Dorno, N.; Lindholst, S.; Feilberg, A.; Eriksen, J. Emissions of CH4, N2O, NH3 and odorants from pig slurry during winter and summer storage. Nutr. Cycl. Agroecosyst. 2013, 95, 103–113. [Google Scholar] [CrossRef]
- Sommer, S.G.; Møller, H.B. Emission of greenhouse gases during composting of deep litter from pig production—Effect of straw content. J. Agric. Sci. 2000, 134, 327–335. [Google Scholar] [CrossRef]
- Bao, M.; Cui, H.; Lv, Y.; Wang, L.; Ou, Y.; Hussain, N. Greenhouse gas emission during swine manure aerobic composting: Insight from the dissolved organic matter associated microbial community succession. Bioresour. Technol. 2023, 373, 128729. [Google Scholar] [CrossRef]
- Pumpanen, J.; Kolari, P.; Ilvesniemi, H.; Minkkinen, K.; Vesala, T.; Niinistö, S.; Lohila, A.; Larmola, T.; Morero, M.; Pihlatie, M.; et al. Comparison of different chamber techniques for measuring soil CO2 efflux. Agric. For. Meteorol. 2004, 123, 159–176. [Google Scholar] [CrossRef]
- Ba, S.; Qu, Q.; Zhang, K.; Groot, J.C.J. Meta-analysis of greenhouse gas and ammonia emissions from dairy manure composting. Biosyst. Eng. 2020, 193, 126–137. [Google Scholar] [CrossRef]
- de Klein, C.; Harvey, M. (Eds.) Nitrous Oxide Chamber Methodology Guidelines; Ministry for Primary Industries: Wellington, New Zealand, 2015; ISBN 978-0-478-40585-9.
- Williams, J.P.; El Hachem, K.; Kang, M. Controlled-release testing of the static chamber methodology for direct measurements of methane emissions. Atmos. Meas. Tech. Discuss. 2023, 16, 3421–3435. [Google Scholar] [CrossRef]

| L1 Length (m) | L2 Width (m) | H Height (m) | Emitting Surface (m2) | |
|---|---|---|---|---|
| DH1 | 0.96 | 0.96 | 0.15 | 0.92 |
| DH2 | 1.03 | 0.53 | 0.25 | 0.55 |
| SH | 0.4 | 0.405 | 0.75 | 0.16 |
| Buffer | Nº Rep. DH1 | Nº Rep DH2 | Nº Rep. SH | Environmental T (°C) | Slurry T (°C) | TAN (gN-NH4+/L) | pH | ER_DH1 (mg N-NH3/m2/s) | ER_DH2 (mg N-NH3/m2/s) | ER_ SH (mg N-NH3/m2/s) |
|---|---|---|---|---|---|---|---|---|---|---|
| N | 3 | 3 | 3 | 10.96 ± 1.99 | 11.33 ± 1.39 | 2.26 ± 0.3 | 11.32 ± 0.08 | 1.85 ± 0.12 | 2.14 ± 0.18 | 0.35 |
| N | 3 | 3 | 1 | 12.37 ± 0.2 | 10.52 ± 0.08 | 1.88 ± 0.14 | 10.72 ± 0.12 | 1.6 ± 0.02 | 1.1 ± 0.02 | 0.1 |
| N | - | - | 3 | 13.02 ± 3.06 | 10.8 ± 1.43 | 1.26 ± 0.27 | 11.67 ± 0.31 | - | - | 0.12 |
| N | - | 3 | - | 13.25 ± 0.07 | 13.3 ± 0.14 | 1.1 ± 0.01 | 10.92 ± 0.04 | - | 1.2 ± 0.06 | - |
| N | 3 | 3 | 1 | 13.72 ± 0.37 | 11.72 ± 0.47 | 0.77 ± 0.03 | 10.33 ± 0.08 | 0.5 ± 0.03 | 0.5 ± 0.01 | 0.1 |
| N | 3 | 3 | 1 | 14.17 ± 0.1 | 12.05 ± 0.14 | 0.29 ± 0.06 | 11.1 ± 0.06 | 0.3 ± 0.03 | 0.3 ± 0.04 | 0 |
| N | - | 3 | - | 14.2 ± 1.13 | 13.5 ± 0.42 | 0.5 ± 0.01 | 10.56 ± 0.03 | - | 0.5 ± 0.004 | - |
| N | - | 3 | - | 17.35 ± 0.92 | 13.5 ± 0.28 | 0.31 ± 0.01 | 10.38 ± 0.05 | - | 0.3± 0.03 | - |
| Y | 3 | 3 | - | 9.03 ± 0.76 | 13.77 ± 0.83 | 2.36 ± 0.05 | 8.3 ± 0.13 | 0.35 ± 0.07 | 0.37 ± 0.09 | - |
| Y | - | - | 1 | 18.94 ± 3.00 | 15.96 ± 2.05 | 1.76 | 8.7 | - | - | 0.01 |
| Y | - | - | 3 | 13.38 ± 1.49 | 12.72 ± 0.94 | 1.44 ± 0.04 | 8.53 ± 0.06 | - | - | 0 |
| Parameter | Units | LFD | C | SFPS |
|---|---|---|---|---|
| pH | - | 7.86 | 8.62 | 8.80 |
| EC | mS/cm | 20.60 | 4.10 | 2.73 |
| ST | % | 5.43 | 28.45 | 27.75 |
| SV | % | 3.68 | 24.91 | 23.96 |
| NH4+ | gN/kg | 3.05 | 2.89 | 1.96 |
| TKN | gN/kg | 4.33 | 8.36 | 3.68 |
| NO2− | mgN/kg | 0.00 | 0.00 | 763.00 |
| NO3− | mgN/kg | 0.00 | 0.00 | 578.00 |
| COD | mg O2/kg | 58,546 | 459,854 | 412,196 |
| SO42− | mg/kg | - | 313.00 | - |
| PO43− | mg/kg | 62.00 | 73.00 | - |
| Ptot | g/kg | 0.43 | 0.85 | 1.15 |
| Emission Source | Nº Rep. | Environmental T (°C) | Slurry T (°C) | TAN (gN-NH4+/L) | pH | ER_NH3 (mg N-NH3/m2/s) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| DH1 | DH2 | SH | DH1 | DH2 | SH | |||||
| C | 3 | 3 | 2 | 16.04 ± 2.27 | 25.56 ± 3.32 | 3.05 | 8.62 | 0.5 ± 0.05 | 0.51 ± 0.23 | 0.03 |
| LFD | 3 | 3 | 2 | 17.21 ± 2.93 | 14.39 ± 3.06 | 2.95 ± 0.04 | 7.86 | 0.12 ± 0.02 | 0.09 ± 0.02 | 0.02 |
| SFPS | 3 | 3 | 2 | 6.43 ± 1.59 | 13.70 | 1.96 | 8.8 | 0.22 ± 0.03 | 0.33 ± 0.03 | 0.02 |
| Emission Source | ER_N2O (mg/m2/min) | ER_CO2 (mg/m2/min) | ER_CH4 (mg/m2/min) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| DH1 | DH2 | SH | DH1 | DH2 | SH | DH1 | DH2 | SH | |
| C | 0.2 | 0.2 | 0.0 | 201.2 | 158.7 | 24.1 | 1.1 | 0.5 | 0.5 |
| LFD | 0.2 | 0.2 | 0.0 | 152.8 | 150.3 | 9.2 | 0.4 | 0.4 | 0.3 |
| SFPS | 0.0 | 0.0 | 0.0 | 75.4 | 30.3 | 67.4 | 0.2 | 0.1 | 0.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cattaneo, M.; Tayà, C.; Burgos, L.; Morey, L.; Noguerol, J.; Provolo, G.; Cerrillo, M.; Bonmatí, A. Assessing Ammonia and Greenhouse Gas Emissions from Livestock Manure Storage: Comparison of Measurements with Dynamic and Static Chambers. Sustainability 2023, 15, 15987. https://doi.org/10.3390/su152215987
Cattaneo M, Tayà C, Burgos L, Morey L, Noguerol J, Provolo G, Cerrillo M, Bonmatí A. Assessing Ammonia and Greenhouse Gas Emissions from Livestock Manure Storage: Comparison of Measurements with Dynamic and Static Chambers. Sustainability. 2023; 15(22):15987. https://doi.org/10.3390/su152215987
Chicago/Turabian StyleCattaneo, Martina, Carlota Tayà, Laura Burgos, Lluis Morey, Joan Noguerol, Giorgio Provolo, Míriam Cerrillo, and August Bonmatí. 2023. "Assessing Ammonia and Greenhouse Gas Emissions from Livestock Manure Storage: Comparison of Measurements with Dynamic and Static Chambers" Sustainability 15, no. 22: 15987. https://doi.org/10.3390/su152215987
APA StyleCattaneo, M., Tayà, C., Burgos, L., Morey, L., Noguerol, J., Provolo, G., Cerrillo, M., & Bonmatí, A. (2023). Assessing Ammonia and Greenhouse Gas Emissions from Livestock Manure Storage: Comparison of Measurements with Dynamic and Static Chambers. Sustainability, 15(22), 15987. https://doi.org/10.3390/su152215987









