Experimental Investigations of the Thermal Safety of Methyl Ethyl Ketone Oxime Hydrochloride Based on the Flask Method, Thermal Analysis, and GC-MS
Abstract
:1. Introduction
2. Material Analysis
2.1. Characterization
2.2. Preparation of MOS
3. Experimental Process
3.1. Specific Experimental Materials and Equipment
3.2. Experimental Procedure
3.2.1. Flask Method Experiment
3.2.2. Thermal Analysis Experiment
3.2.3. Gas Chromatography-Mass Spectrometry Experiment
4. Results
4.1. Water Solubility Analysis of MEKOH
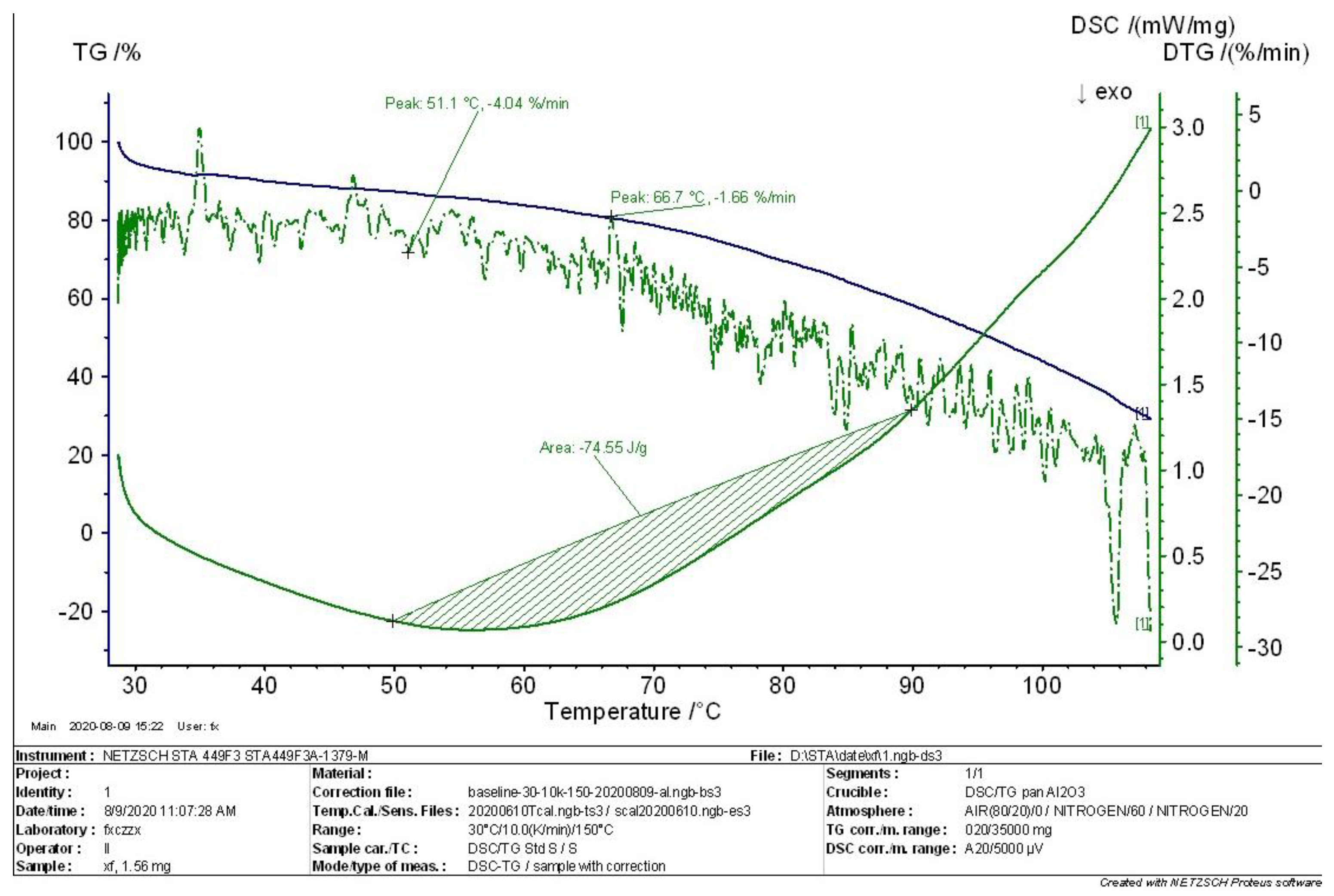
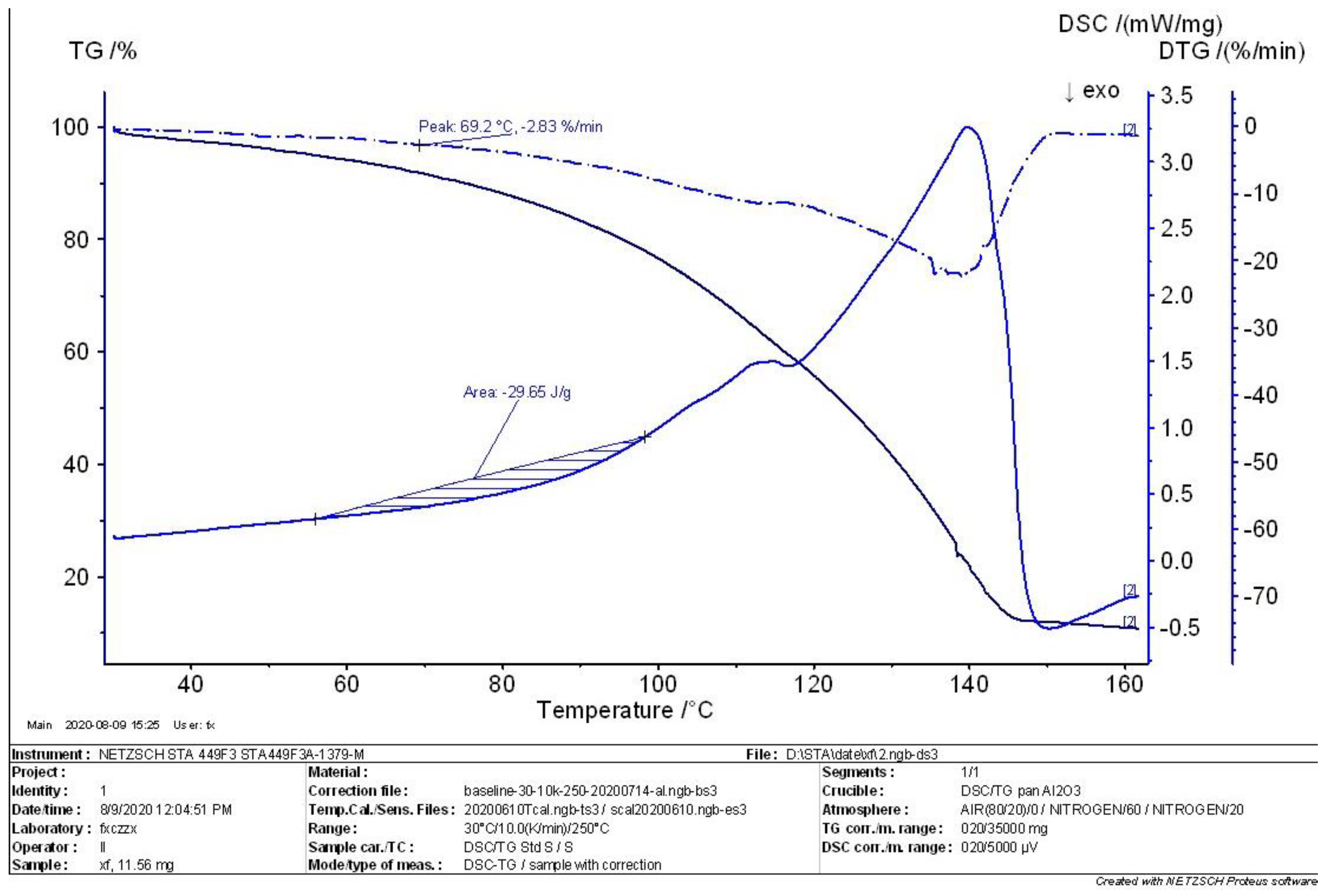
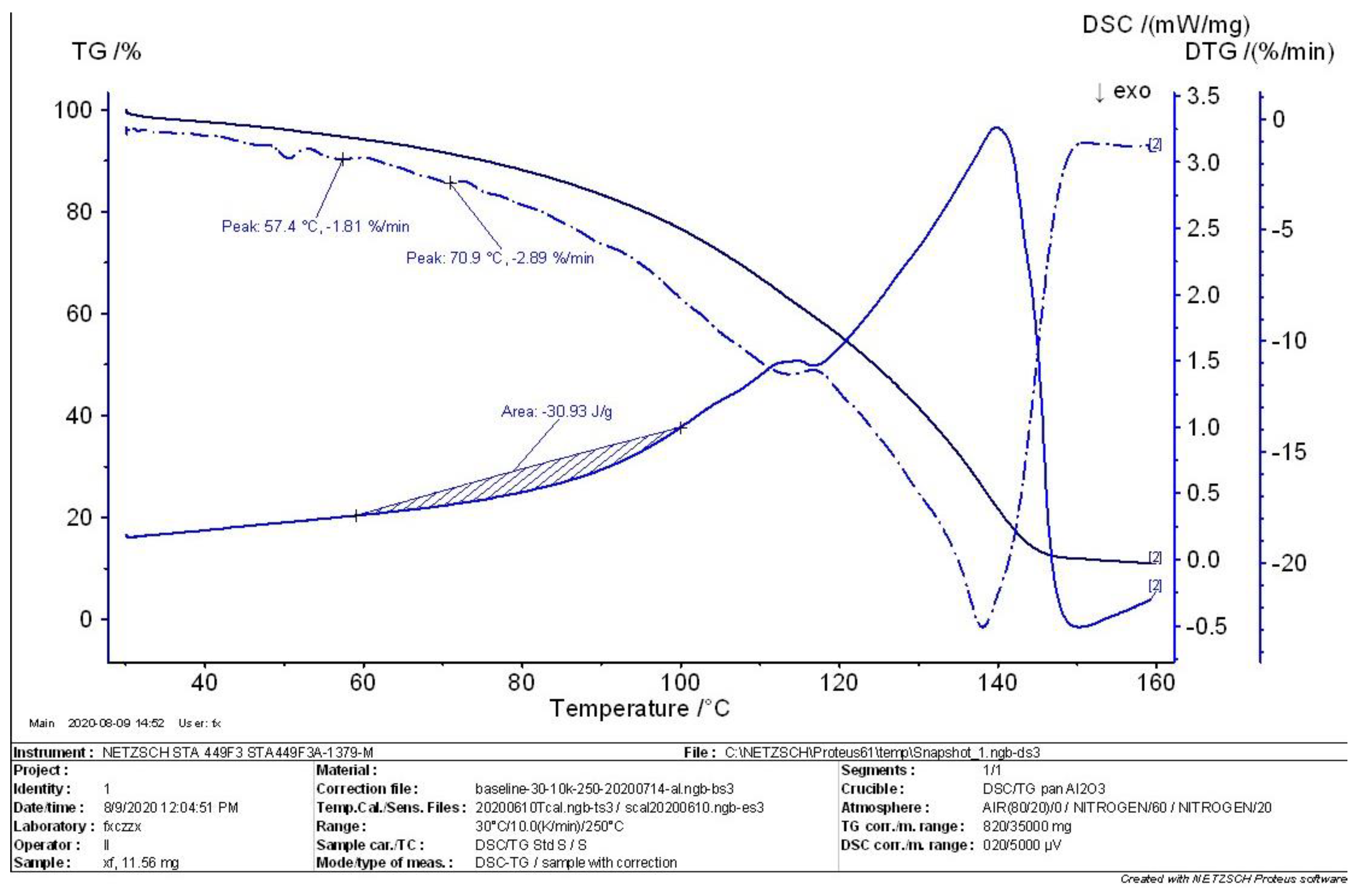
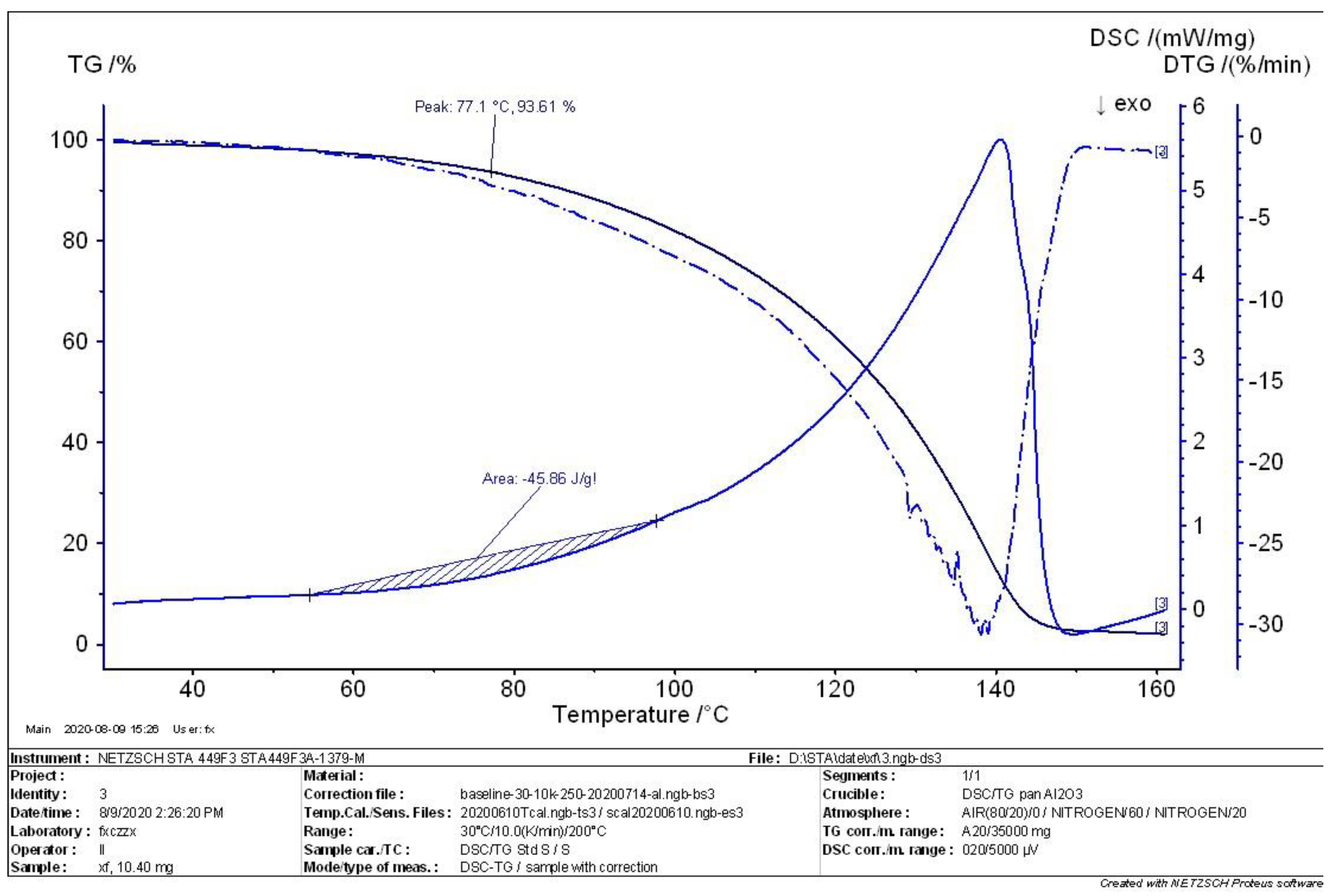
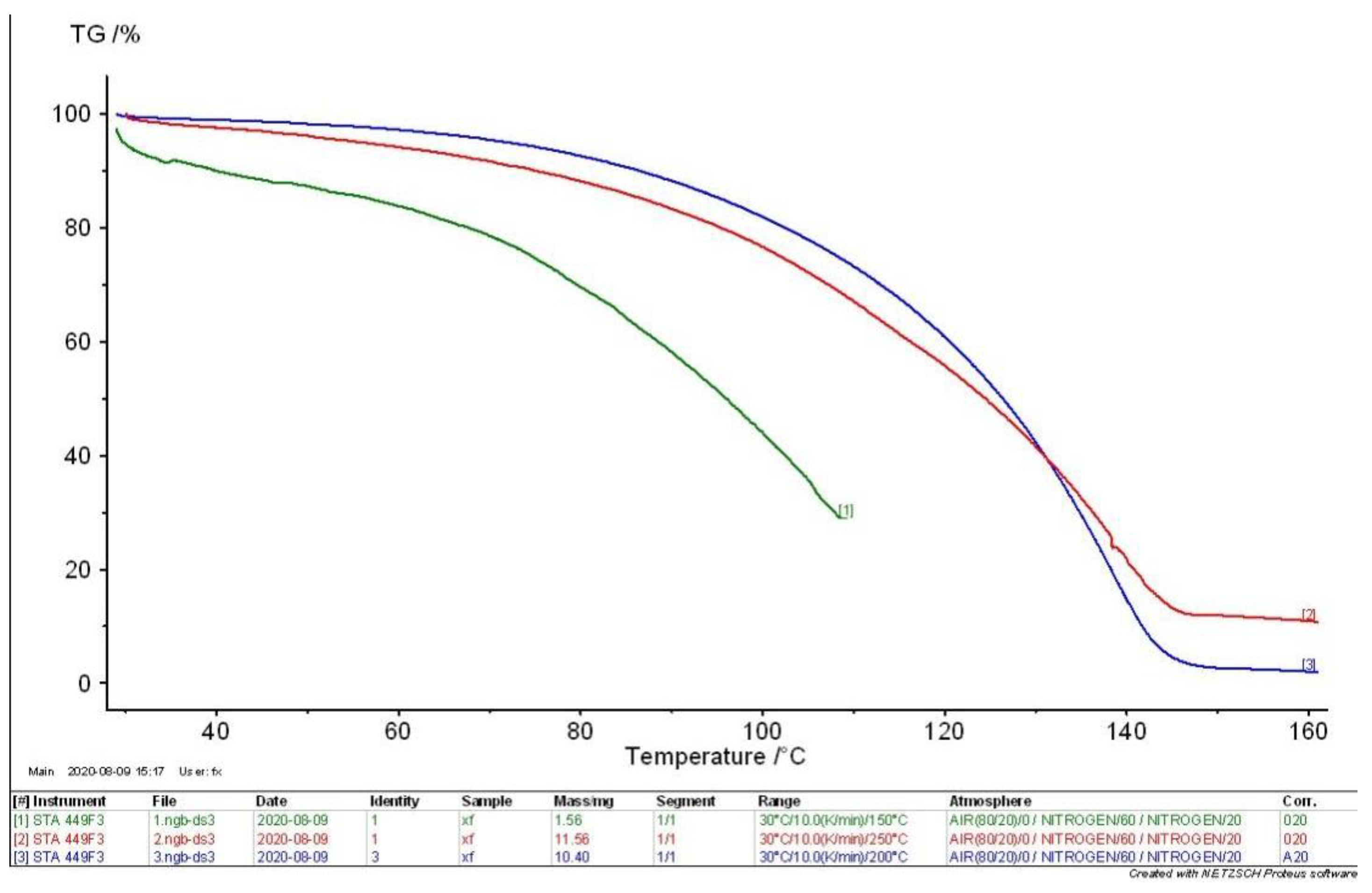
4.2. TG-DSC Analysis of MEKOH
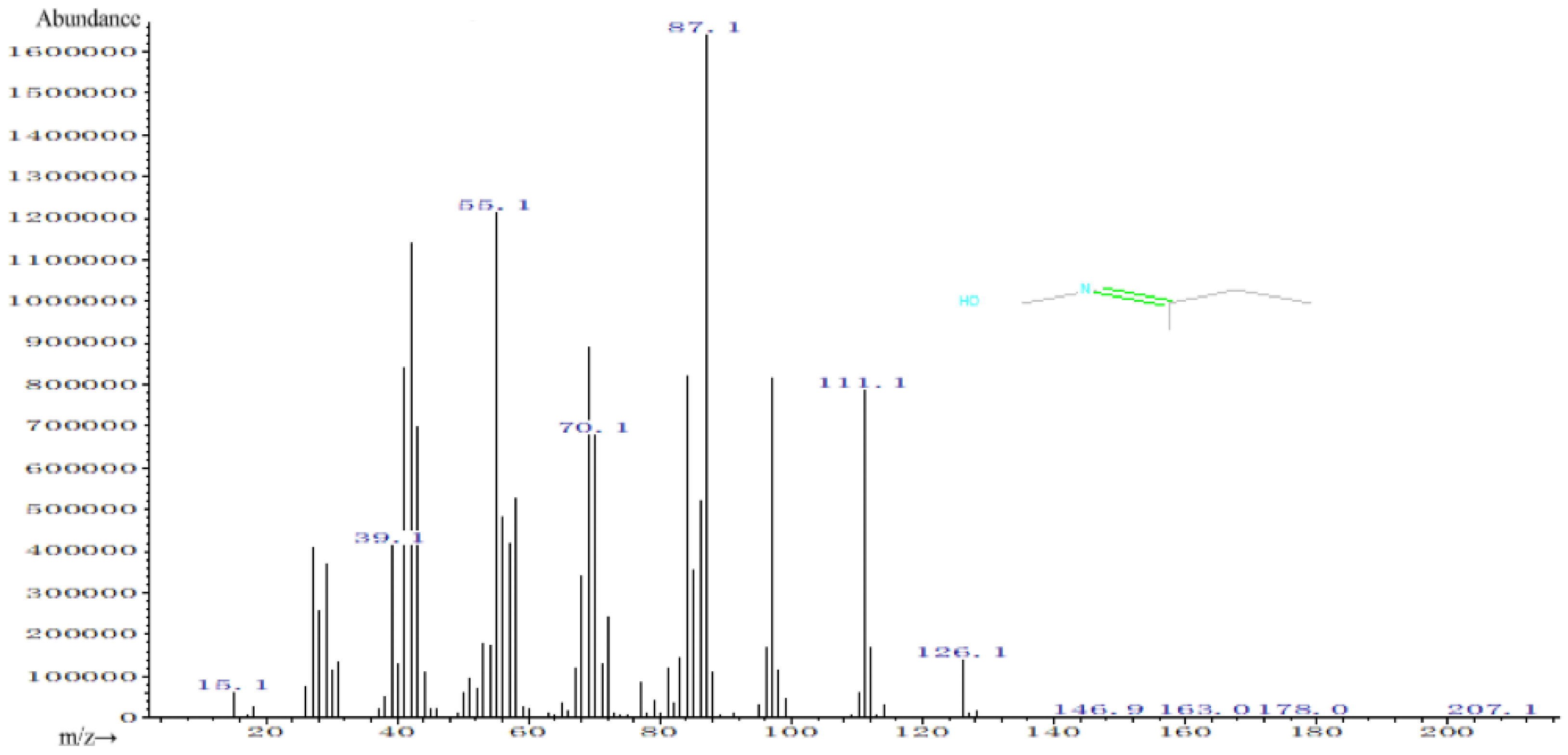
4.3. Analysis of MEKOH Decomposition Substances
5. Discussion and Conclusions
- (1)
- The temperature profiles of MEKOH dissolved in different qualities of deionized water were measured and calculated with flask experiments, and a general warming pattern was determined. In the experimental environment of room temperature (28 °C), the final temperature of the MEKOH solution stabilized at about 33 °C, indicating an increase of approximately 5 °C throughout the entire process. During the process, MEKOH maintained a good thermal stability and was not found to violently decompose, which can be considered an acceptable temperature range. In other words, the dissolution of MEKOH at room temperature can be concluded to be relatively safe with no strong adverse effects on the safety of the production process.
- (2)
- According to the comprehensive analysis results of TG analysis and DSC in the initial, intermediate, and complete states of MEKOH decomposition, MEKOH was found to have a good thermal safety below 50 °C. The substance underwent a violent exothermic decomposition from 51 to 57 °C, and no longer showed any significant change in weight after 145 °C. The enthalpy changes of the weight loss ranged from −29.65 J/g to −45.86 J/g. In actual production, the ambient temperature of MEKOH should be controlled below 50 °C to ensure the thermal safety of MEKOH and thus prevent flash explosion accidents.
- (3)
- By analyzing the pyrolysis products, a large number of hydrocarbon compounds and other flammable substances were detected in the GC-MS experiments, but MEKOH was not detected. Once the temperature exceeded the temperature threshold for the thermal safety of MEKOH, the physicochemical properties of the pyrolysis products need to be promptly monitored and controlled while cooling. Moreover, a large amount of MEKO was found in the pyrolysis products of MEKOH, which is one of the reactants for preparing MOS. Therefore, MEKO from MEKOH decomposition can be recovered to be reused as a reactant, which can reduce a company’s production costs and waste disposal hazards to some extent.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, Q.M. A study on the focus and key points of chemical safety management. China Pet. Chem. Stand. Qual. 2022, 42, 84–86. [Google Scholar]
- Liu, D.; Wang, K.; Dai, F.X.; Wang, Z. Network Analysis on Key Causes of Chemical Accidents Considering Structural Characteristics. J. Saf. Sci. Technol. 2021, 17, 71–76. [Google Scholar]
- Xu, P. Investigation and Countermeasures of Common Fire Accidents in Coal Chemical Enterprises. Mod. Chem. Res. 2023, 16, 182–184. [Google Scholar]
- Sun, Y.L.; Li, H.B.; Liu, X.F.; Wang, J.M.; Jiang, Q.Q. Research on Quantitative Analysis Method for Causal Factors of Chemical Accidents Based on AcciMap Model. J. Saf. Environ. 2021, 21, 1670–1675. [Google Scholar]
- Pei, J.K.; Wang, F.Y.; Guo, H.H.; Yang, C.Y.; Pei, C.W. Cause Analysis of Chemical Accidents based on Improved Cusp Catastrophe Model. China Saf. Sci. J. 2019, 29, 20–25. [Google Scholar]
- Yuan, B.H.; Yang, J.Q.; Luo, X.Y.; Su, X.Y. Evaluation Method of Safety Resilience of Chemical Industry Park Based on Fuzzy Matter Element. J. Saf. Environ. 2023, in press. [Google Scholar] [CrossRef]
- Mathew, E. Producing Oximinosilanes, Oximinogermanes and Oximinostannanes. EP Patent EP19820110766, 22 November 1982. [Google Scholar]
- Nelson, G.O.; Priante, S.J.; Strong, M.; Anderson, D.; Fallon-Carine, J. Permeation of Substituted Silanes and Siloxanes through Selected Gloves and Protective Clothing. AIHAJ 2000, 61, 709–723. [Google Scholar] [CrossRef]
- F739-96; ASTM (The American Society for Testing and Materials): Standard Test Method for Resistance of Protective Clothing Materials to Permeation by Liquids or Gases Under Conditions of Continuous Contact. ASTM: Philadelphia, PA, USA, 1996.
- Li, M.; Liu, J.D.; Na, H.Z. Application and Advancement in Synthesis of 2-Butanone Oxime. Chem. Eng. 2006, 07, 42–43. [Google Scholar]
- Zhao, Q.; Liu, Q.Y.; Xu, H.H.; Bei, Y.L.; Feng, S.Y. Preparation and Characterization of Room Temperature Vulcanized Silicone Rubber Using Alpha-Amine Ketoximesilanes as Auto-Catalyzed Cross-Linkers. RSC Adv. 2016, 44, 38447–38453. [Google Scholar] [CrossRef]
- Chen, K.L.; Guo, W.W.; Xu, L.; Zhao, Y. Low Cost and Facile Preparation of Robust Multifunctional Coatings with Self-Healing Superhydrophobicity and High Conductivity. Compos. Sci. Technol. 2018, 156, 177–185. [Google Scholar] [CrossRef]
- Xu, G.J.; Huang, L.; Lu, C.L.; Zhou, X.H.; Cui, G.L. Revealing the Multilevel Thermal Safety of Lithium Batteries. Energy Storage Mater. 2020, 31, 72–86. [Google Scholar] [CrossRef]
- Niu, H.; Chen, S.S.; Shu, Q.H.; Li, L.J.; Jin, S.H. Preparation, Characterization and Thermal Risk Evaluation of Dihydroxylammonium 5,5‘-Bistetrazole-1,1‘-Diolate based Polymer Bonded Explosive. J. Hazard. Mater. 2017, 338, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.B.; Gao, X.F.; Wang, Y.; Tong, Y.J. Thermal Safety Analysis of On-Site Emulsion Explosives Mixed with Waste Engine Oil. Energies 2022, 15, 895. [Google Scholar] [CrossRef]
- Xu, C.; Cheng, Z. Thermal Stability of Ionic Liquids: Current Status and Prospects for Future Development. Processes 2021, 9, 337. [Google Scholar] [CrossRef]
- Zheng, Q.J.; Zhang, Y.F.; Montazerian, M.; Gulbiten, O.; Mauro, J.C.; Zanotto, E.D.; Yue, Y.Z. Understanding Glass through Differential Scanning Calorimetry. Chem. Rev. 2019, 119, 7848–7939. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.; Ali, A.; Alzahrani, B.; Sanada, K. Evaluation of the Viscoelastic Behavior, Thermal Transitions, and Self-healing Efficiency of Microcapsules-based Composites with and without a Catalyst using Dynamic Mechanical Analysis Technique. J. Appl. Polym. Sci. 2023, 140, e54323. [Google Scholar] [CrossRef]
- Liu, H.X. Advances in Thermal Analysis—Instruments and Applications. China Instrum. 1984, 06, 17–19+13. [Google Scholar]
- Sava, I.; Burescu, A.; Lisa, G. Study of Thermal Behavior of Polyimides Containing Pendent-Substituted Azobenzene Units. Polym. Bull. 2014, 71, 1359–1373. [Google Scholar] [CrossRef]
- Faria, A.M.; Marcos, C.A.; Vigna, C.R.M.; Collins, C.H. Determination of Polymeric Layer Thickness on Immobilized Stationary Phases for High-Performance Liquid Chromatography Using Thermogravimetric Analyses. Química Nova 2007, 31, 417–420. [Google Scholar] [CrossRef]
- Zhorin, V.A.; Kiselev, M.R.; Roldugin, V.I. Thermogravimetric Analysis of the Aluminum-Polypropylene Mixtures After Plastic Deformation Under High Pressure. Russ. J. Appl. Chem. 2013, 86, 15–19. [Google Scholar] [CrossRef]
- Kumar, P.; Subbarao, P.M.V.; Kala, L.D.; Vijay, V.K. Thermogravimetric Devolatilization Behavior of Agricultural Residue for Generation of Syngas. J. Energy Resour. Technol. 2021, 143, 112106–112113. [Google Scholar]
- Yin, C.Y.; Jiang, Z.T. Thermogravimetric and Kinetic Analyses of Degradation of Groundnut (Arachis hypogaea) Shell in Air and Nitrogen. J. Energy Inst. 2012, 85, 57–60. [Google Scholar] [CrossRef]
- Earnest, C.M.; Chun, D. Modern Thermogravimetric Analysis. Mod. Sci. Instrum. 1986, 1, 40–44. [Google Scholar]
- Zou, T.; Zhao, J.; Guo, S.; Chen, Y.D. Development and application of thermal analysis technology in China. Anal. Instrum. 2019, 227, 9–16. [Google Scholar]
- Yang, Y.H. Implementation and Application of Gas Chromatography Technology in Food Safety Detection. China Food Saf. Mag. 2022, 344, 177–179. [Google Scholar]
- Chen, W.Q.; Peng, H.; Deng, B.W.; Wei, W.; Liu, K.H.; Ding, X.W.; Xie, X.C. Application of Gas Chromatography in Food Safety Testing. Jiangsu Agric. Sci. 2009, 04, 356–358. [Google Scholar]
- Gao, J.W. Application of Gas Chromatography in Food Safety Detection. China Food Saf. Mag. 2022, 350, 175–177. [Google Scholar]
- Liu, D.N. Application of Gas Chromatography in Chemical Analysis. Chem. Eng. Manag. 2022, 640, 48–50. [Google Scholar]
- Zhang, G.L. Application of Gas Chromatography in Environmental Monitoring Engineering. Shanxi Chem. Ind. 2023, 43, 93–95. [Google Scholar]
- Chen, X.; Cai, B.L.; Feng, J.; Cai, Z.Y. Gas Chromatography-Mass Spectrometry and its Application. Deterg. Cosmet. 2023, 46, 50–53. [Google Scholar]
- Chen, X.S.; Hou, H.W.; Bian, Z.Y.; Tang, G.L.; Hu, Q.Y. Research Progress on Application of GC-MS/MS. J. Chin. Mass Spectrom. Soc. 2013, 34, 308–320. [Google Scholar]
- Teng, Y. Simulation and Research on the Production of MOS. Master’s Thesis, East China University of Science and Technology, Shanghai, China, 2015. [Google Scholar]
- Lin, X.F.; Lan, C.X.; Hu, G.X.; Huang, Y.J.; Chen, T.G. 107 Silicone Rubber Branch Methyl MQ Silicone Resin and its Application in Silicone Resin. Silicone Mater. 2018, 32, 46–49. [Google Scholar]
- Zhang, F.; Zhang, Z.X.; Gong, X.A.; Xiang, S.L.; Zhao, S.L. Preparation and Performance of Silicone Insulation Coatings, New Chemical Materials. New Chem. Mater. 2019, 47, 80–82. [Google Scholar]
- Yang, X.B.; Yu, C.Q.; Yu, Y.R. Study on the Preparation and Adhesion of Silicone Heat-Resistant Coatings. Mod. Chem. Ind. 2019, 39, 168–172. [Google Scholar]
- Muiller, H.; Schmidt, E.; Frey, V.; Riedle, R. Process for Preparing Oxime Containing Siliconcompound. U.S. Patent 4,126,630, 16 March 1978. [Google Scholar]
- Jiang, P. Study on the Synthesis of Several Functional Silane Compounds and Their Gas-Liquid Equilibrium. Master’s Thesis, Hangzhou Normal University, Hangzhou, China, 2021. [Google Scholar]
- Yamato, K.; Sato, M.; Oya, I.; Nawamaki, H. Imidazolesul Fonamide Deriviatives and Herbicides. U.S. Patent 4,830,660, 16 May 1987. [Google Scholar]
- Zheng, Y.B. Basic Research on Application of Methyl Ethyl Ketoxime and Its Product Chain Design. Master’s Thesis, Zhejiang University, Hangzhou, China, 2010. [Google Scholar]
- Liu, T.Y. Study on New Process of TS-1 Catalyzed Butanone Ammoximation. Master’s Thesis, Beijing University of Chemical Technology, Beijing, China, 2020. [Google Scholar]
- Li, W.D.; Zhou, D.H.; Xiao, Z.H.; Chang, L.; Wang, N.; Zhang, M. Experimental Analysis of Thermal Stability of MEKOH. Shandong Chem. Ind. 2021, 50, 85–88. [Google Scholar]
- GB/T 27841-2011; Chemical Products for Industrial Use—Determination of Hydrosolubility of Solids and Liquids with High Solubility—Flask Method. General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China, Standardization Administration of the People’s Republic of China: Beijing, China, 2012.
- ISO 4788; Laboratory Glassware—Graduated Measuring Cylinders. Technical Committee ISO/TC 48, Subcommittee SC 6: Geneva, Switzerland, 2005.
- GB/T 13464-2008; Thermal Analysis Test Methods for Thermal Stability of Materials. Inspection and Quarantine of the People’s Republic of China, Standardization Administration of the People’s Republic of China: Beijing, China, 2009.
- GB/T 9722-2006; Chemical Reagent-General Rules for the Gas Chromatography. Inspection and Quarantine of the People’s Republic of China, Standardization Administration of the People’s Republic of China: Beijing, China, 2006.
- Tang, Y.M.; Ou, S.P.; Ye, L.H.; Huang, Y.P.; Wang, S. Determination of Equilibrium Solubility and Oil-Water Partition Coefficient of Glycycoumarin. Cent. South Pharm. 2023, 21, 959–962. [Google Scholar]
- Song, B. Study on the Solubility in Water and N-Octanol/Water Partation Coefficients of Phenol Compounds. Master’s Thesis, Qingdao University of Science and Technology, Qingdao, China, 2012. [Google Scholar]
- Vuyyuru, N.R.; Reddy, A.M.; Ramadevi, B.; Kumar, Y.R.; Prasad, B.D. A Simple, Specific, Mass Compatible and Validated Gas Chromatographic Method for the Estimation of Piperidine-3-amine Content in Linagliptin Finished and Stability Samples without Derivatization. Asian J. Chem 2020, 32, 2567–2572. [Google Scholar] [CrossRef]
- Zhang, H.; Jiang, J.C.; Yan, T.Y.; Ni, L.; Liu, S.H. Thermal Hazard Risk and Decomposition Mechanism Identification of 1-Hexyl-2,3-Dimethylimidazolium Nitrate: Combined Thermal Analysis Experiment and DFT Emulation. Process Saf. Environ. Prot. 2023, 172, 38–47. [Google Scholar] [CrossRef]
- Colman, M.D.; Lazzarotto, S.R.D.; Lazzarotto, M.; Hansel, F.A.; Colman, T.A.D.; Schnitzler, E. Evolved Gas Analysis (TG-DSC-FTIR) and (Pyr-GC-MS) in the Disposal of Medicines (Aceclofenac). J. Anal. Appl. Pyrolysis 2016, 119, 157–161. [Google Scholar] [CrossRef]
- Riley, J.T.; Marsh, M. Macro Thermogravimetric Analyzers: Versatile and Underutilized Analytical Instruments. J. Test. Eval. 2021, 49, 4357–4363. [Google Scholar] [CrossRef]
- Liu, L.; Xu, Y.P.; Chen, X.; Hong, M.; Tong, J. Thermogravimetric Analysis of Enthalpy Variation of 1-Alkyl-3-methylimidazole Chloride. Acta Phys. Chim. Sin. 2020, 36, 98–106. [Google Scholar] [CrossRef]
- Xie, Q.Y.; Chen, D.D.; Ding, Y.W. Thermogravimetric Analysis and Its Applications in Polymer Characterization. Acta Polym. Sin. 2022, 53, 193–210. [Google Scholar]
- Song, Y.W.; Hou, J.B.; Mao, H.K.; Liu, H.B. Study on Heat Treatment Process of Al-Si-Cu-Mg Alloy Based on DSC Analysis. Hot Work. Technol. 2022, 51, 139–142+145. [Google Scholar]
- Jia, C.X.; Liu, H.P.; Bai, J.R.; Qin, H.; Wang, Q. Investigation of Oil Sand Combustion by Simultaneous Thermal Analyzer. Chem. Ind. Eng. Prog. 2013, 32, 1273–1277. [Google Scholar]
- Song, F.; Wang, H.; Huang, Y.L.; Zhang, Y.F.; Chen, W.J.; Zhao, S.L.; Zhang, M. TG-DSC method applied to drying characteristics of areca inflorescence during drying. Heat Mass Transf. 2017, 53, 3181–3188. [Google Scholar] [CrossRef]
- Zhao, L.H.; Liu, X.F.; Wang, Y.J.; Ren, T.S. Thermal Analysis Determining Soil Organic Matter Content and Thermal Stability. Trans. CSAE 2016, 32, 105–114. [Google Scholar]
- Zhang, Z.H.; Tian, S.B.; Zhu, S.Q. Analysis of Hydrocarbon Constituents in Heavy Oil by Chromatography Mass Spectrometry and Chromatography-Mass Spectrometry. Chem. Eng. Oil Gas 2005, 4, 315–320+228. [Google Scholar]
- Zhu, Y.; Chen, X.T.; Qiao, K.; Li, S.G.; Liu, S.J.; Pan, N.; Liu, Z.Y.; Shi, W.Z. Progress in Research on Gas Chromatography and Hyphenated Technology for Studies on Food Flavor. Food Res. Dev. 2022, 43, 200–210. [Google Scholar]
- Zhang, Y.M. Principle of Gas Chromatography and its Application in the Detection of Pesticide Residues in Food. China Food Saf. Mag. 2021, 35, 187–189. [Google Scholar]
- Li, J.J.; Lei, C.Y.; Huang, C.H.; Zhou, L.; Yan, F.M. Chemical Analysis Techniques for Infochemicals. Chin. J. Appl. Entomol. 2014, 51, 586–590. [Google Scholar]
- Donval, J.P.; Guyader, V.; Boissy, E. A Simple Method for the Preparation and Injection of Gas Mixtures into a Gas Chromatograph using a Two-component Device. J. Chromatogr. A 2020, 1631, 461579. [Google Scholar] [CrossRef]
- Zhu, L.X.; Chen, S.; Lin, Q.B.; Zhong, H.N.; Li, Z.; Liao, J. Detection and Discrimination of Volatile Compounds in Recycled and Virgin Paper by HS-GC-MS. Chin. J. Anal. Lab. 2020, 39, 1405–1411. [Google Scholar]
- Wang, S. Kinetics of Thermal Decompositionof Cerium Carbonate. Master’s Thesis, Inner Mongolia University of Science and Technology, Baotou, China, 2020. [Google Scholar]
- Huo, X.B.; Jia, G.G.; Li, Z.Q.; Zhang, P.; He, X.H. Study on Fragmentation Modes of Two Kinds of NCS Liquid Crystalline Compounds. J. Hebei Norm. Univ. Nat. Sci. 2020, 44, 521–525. [Google Scholar]





| Experiment | Relevant Bas | Equipment | Type | Grade | Number |
|---|---|---|---|---|---|
| Flask method | GB/T 27841-2011 Chemical products for industrial use—Determination of hydrosolubility of solids and liquids with high solubility—Flask method [44] | Graduated flask with ground mouth | 100 mL | Compliant with ISO 4788 [45] | A01-A10 |
| Thermal analysis | GB/T 13464-2008 Thermal analysis test methods for thermal stability of materials [46] | NETZSCH synchronous thermal analyzer | STA449F3 | Qualified | 1379-M |
| GC-MS | GB/T 9722-2006 Chemical reagent—General rules for gas chromatography [47] | Agilent gas chromatography-mass spectrometry | GC 7890A MSD 5975C | Qualified | CN10481054 |
| No. | Percentage of Deionized Water/% | Initial Temperature/°C | Stable Temperature/°C | Start Stable Time/min | Temperature Contrast/°C |
|---|---|---|---|---|---|
| Sample 01 | 5 | 28.0 | 32.5 | 2.5 | 4.5 |
| 10 | 28.0 | 32.0 | 4 | 4.0 | |
| 15 | 28.0 | 31.0 | 8 | 3.0 | |
| Sample 02 | 5 | 28.0 | 33.5 | 7.5 | 5.5 |
| 10 | 28.0 | 33.5 | 9 | 5.5 | |
| 15 | 28.0 | 33.0 | 9 | 5.0 | |
| Sample 03 | 5 | 28.0 | 33.5 | 10 | 5.5 |
| 10 | 28.0 | 33.2 | 10 | 5.2 | |
| 15 | 28.0 | 32.7 | 10 | 4.7 | |
| Sample 04 | 5 | 28.0 | 33.5 | 4 | 5.5 |
| 10 | 28.0 | 33.5 | 7.5 | 5.5 | |
| 15 | 28.0 | 33.5 | 9 | 5.5 |
| Peak | Retention Time | Area | Material | Formula | CAS | Mass |
|---|---|---|---|---|---|---|
| 2 | 3.694 | 11.30 | Pyrrolidine | C4H9N | 123-75-1 | 46 |
| Hexane, 3,3,4-trimethyl | C9H20 | 16747-31-2 | 43 | |||
| Heptane | C7H16 | 142-82-5 | 43 | |||
| 4 | 4.362 | 10.05 | Cycloheptane | C7H14 (isomer) | 291-64-5 | 55 |
| 1-Heptene | C7H14 (isomer) | 592-76-7 | 50 | |||
| Methylcyclohexane | C7H14 | 108-87-2 | 46 | |||
| 8 | 5.378 | 10.28 | Octane, 4-methyl- | C9H20 | 2216-34-4 | 46 |
| Hexane, 2,3,4-trimethyl- | C9H20 | 921-47-1 | 43 | |||
| Heptane, 2-methyl- | C8H18 | 592-27-8 | 38 | |||
| 55 | 18.746 | 11.25 | Azetidine, n-propyl- | C3H7N(isomer) | \ | 16 |
| 1-Methoxy-2,3-cis-dimethylaziridine(sin) | C5H11NO | 61593-25-7 | 12 | |||
| Phosphine oxide, methyldiphenyl- | C13H13OP | 2129-89-7 | 12 |
| Group | Peaks Belonging to MEKO | Retention Time | Area | Mass (MEKO) |
|---|---|---|---|---|
| 1 | 23 | 6.688 | 0.28 | 46 |
| 46 | ||||
| 46 | ||||
| 24 | 6.744 | 0.38 | 93 | |
| 86 | ||||
| 64 | ||||
| 25 | 6.832 | 0.83 | 60 | |
| 60 | ||||
| 27 | 6.986 | 0.29 | 78 | |
| 60 | ||||
| 53 | ||||
| 28 | 7.037 | 1.51 | 42 | |
| 27 | ||||
| 2 | 25 | 6.693 | 0.28 | 46 |
| 46 | ||||
| 46 | ||||
| 26 | 6.750 | 0.38 | 93 | |
| 86 | ||||
| 64 | ||||
| 27 | 6.837 | 0.83 | 90 | |
| 90 | ||||
| 60 | ||||
| 28 | 6.955 | 1.82 | 90 | |
| 89 | ||||
| 60 | ||||
| 29 | 6.991 | 0.28 | 78 | |
| 60 | ||||
| 53 | ||||
| 30 | 7.042 | 1.54 | 38 | |
| 27 | ||||
| 3 | 24 | 6.693 | 0.29 | 46 |
| 46 | ||||
| 46 | ||||
| 25 | 6.750 | 0.39 | 89 | |
| 76 | ||||
| 26 | 6.837 | 0.85 | 90 | |
| 90 | ||||
| 60 | ||||
| 28 | 7.032 | 1.40 | 30 | |
| 30 | ||||
| 4 | 23 | 6.693 | 0.30 | 46 |
| 46 | ||||
| 46 | ||||
| 24 | 6.750 | 0.40 | 86 | |
| 70 | ||||
| 25 | 6.837 | 0.87 | 60 | |
| 60 | ||||
| 60 | ||||
| 27 | 6.986 | 0.39 | 83 | |
| 58 | ||||
| 5 | 24 | 6.688 | 0.30 | 46 |
| 46 | ||||
| 46 | ||||
| 25 | 6.745 | 0.41 | 86 | |
| 64 | ||||
| 26 | 6.832 | 0.88 | 60 | |
| 60 | ||||
| 60 | ||||
| 28 | 6.996 | 0.56 | 83 | |
| 64 | ||||
| 6 | 24 | 6.693 | 0.39 | 46 |
| 46 | ||||
| 46 | ||||
| 25 | 6.750 | 0.52 | 86 | |
| 64 | ||||
| 26 | 6.837 | 1.12 | 90 | |
| 78 | ||||
| 60 | ||||
| 27 | 6.955 | 2.89 | 90 | |
| 89 | ||||
| 60 | ||||
| 7 | 25 | 6.688 | 0.31 | 46 |
| 46 | ||||
| 46 | ||||
| 26 | 6.745 | 0.43 | 93 | |
| 90 | ||||
| 76 | ||||
| 27 | 6.832 | 0.91 | 90 | |
| 78 | ||||
| 60 | ||||
| 28 | 6.924 | 1.34 | 52 | |
| 52 | ||||
| 46 | ||||
| 30 | 6.981 | 0.62 | 70 | |
| 55 | ||||
| 49 | ||||
| 8 | 24 | 6.657 | 0.40 | 46 |
| 46 | ||||
| 46 | ||||
| 25 | 6.714 | 0.54 | 93 | |
| 86 | ||||
| 64 | ||||
| 26 | 6.801 | 1.16 | 90 | |
| 60 | ||||
| 27 | 6.904 | 2.68 | 64 | |
| 50 | ||||
| 50 | ||||
| 9 | 17 | 6.411 | 0.49 | 42 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, D.; Peng, S.; Xie, B.; Wang, L.; Li, H. Experimental Investigations of the Thermal Safety of Methyl Ethyl Ketone Oxime Hydrochloride Based on the Flask Method, Thermal Analysis, and GC-MS. Sustainability 2023, 15, 14598. https://doi.org/10.3390/su151914598
Zhou D, Peng S, Xie B, Wang L, Li H. Experimental Investigations of the Thermal Safety of Methyl Ethyl Ketone Oxime Hydrochloride Based on the Flask Method, Thermal Analysis, and GC-MS. Sustainability. 2023; 15(19):14598. https://doi.org/10.3390/su151914598
Chicago/Turabian StyleZhou, Dehong, Shiyu Peng, Bin Xie, Lunping Wang, and Haochen Li. 2023. "Experimental Investigations of the Thermal Safety of Methyl Ethyl Ketone Oxime Hydrochloride Based on the Flask Method, Thermal Analysis, and GC-MS" Sustainability 15, no. 19: 14598. https://doi.org/10.3390/su151914598
APA StyleZhou, D., Peng, S., Xie, B., Wang, L., & Li, H. (2023). Experimental Investigations of the Thermal Safety of Methyl Ethyl Ketone Oxime Hydrochloride Based on the Flask Method, Thermal Analysis, and GC-MS. Sustainability, 15(19), 14598. https://doi.org/10.3390/su151914598






