Use of Wastewater and Electrogenic Bacteria to Generate Eco-Friendly Electricity through Microbial Fuel Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
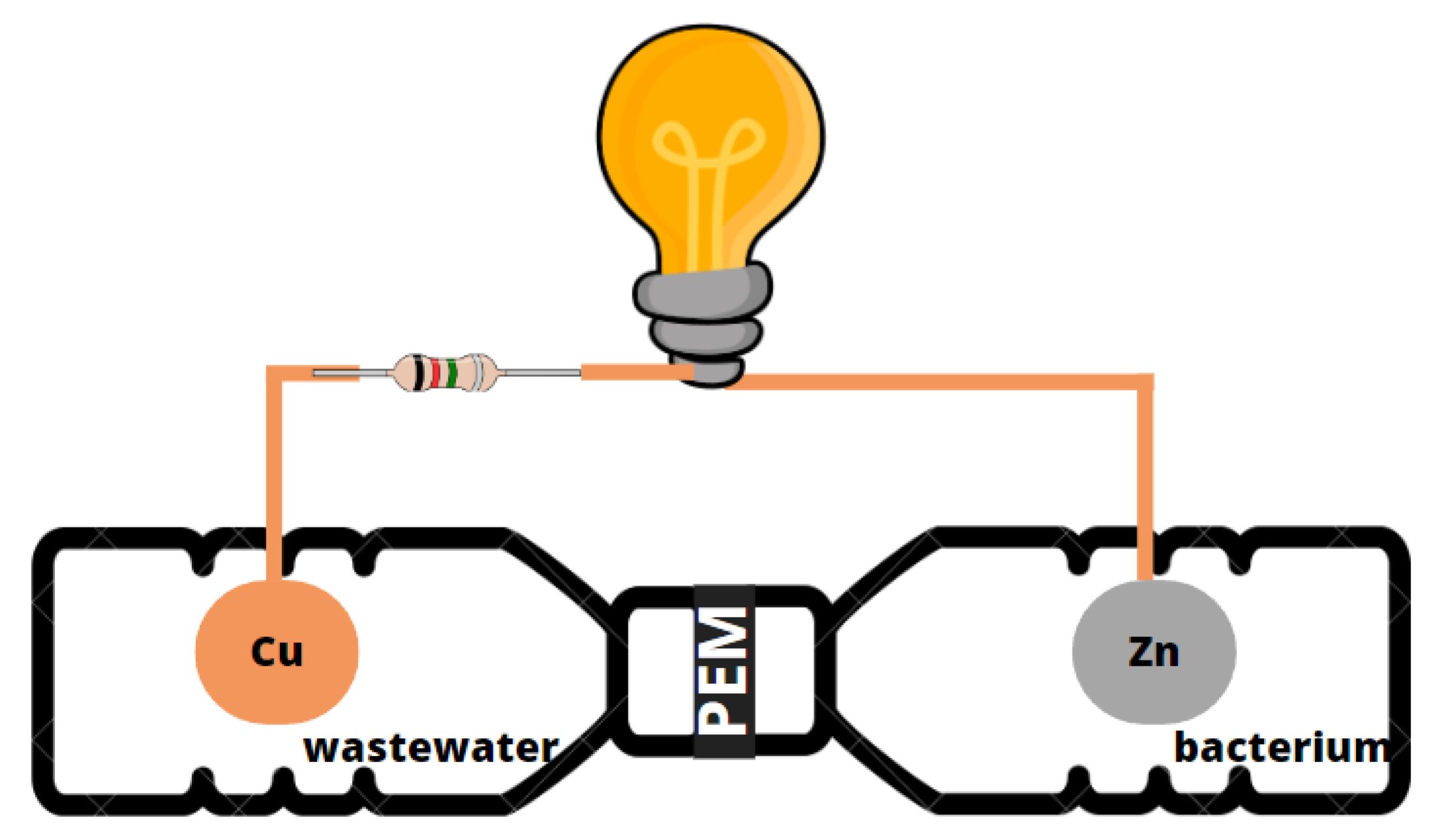
2.2. Dual-Chamber MFC Design and Operation
2.3. Bioelectrochemical Analysis
2.4. Obtaining Electrogenic Strains
2.5. Reactivation of Electrogenic Strains
2.6. Inocula of the Electrogenic Strains in the Cathode Chamber
2.7. Metagenomic Analysis of Wastewater from the PTARC
2.8. Statistical Analysis
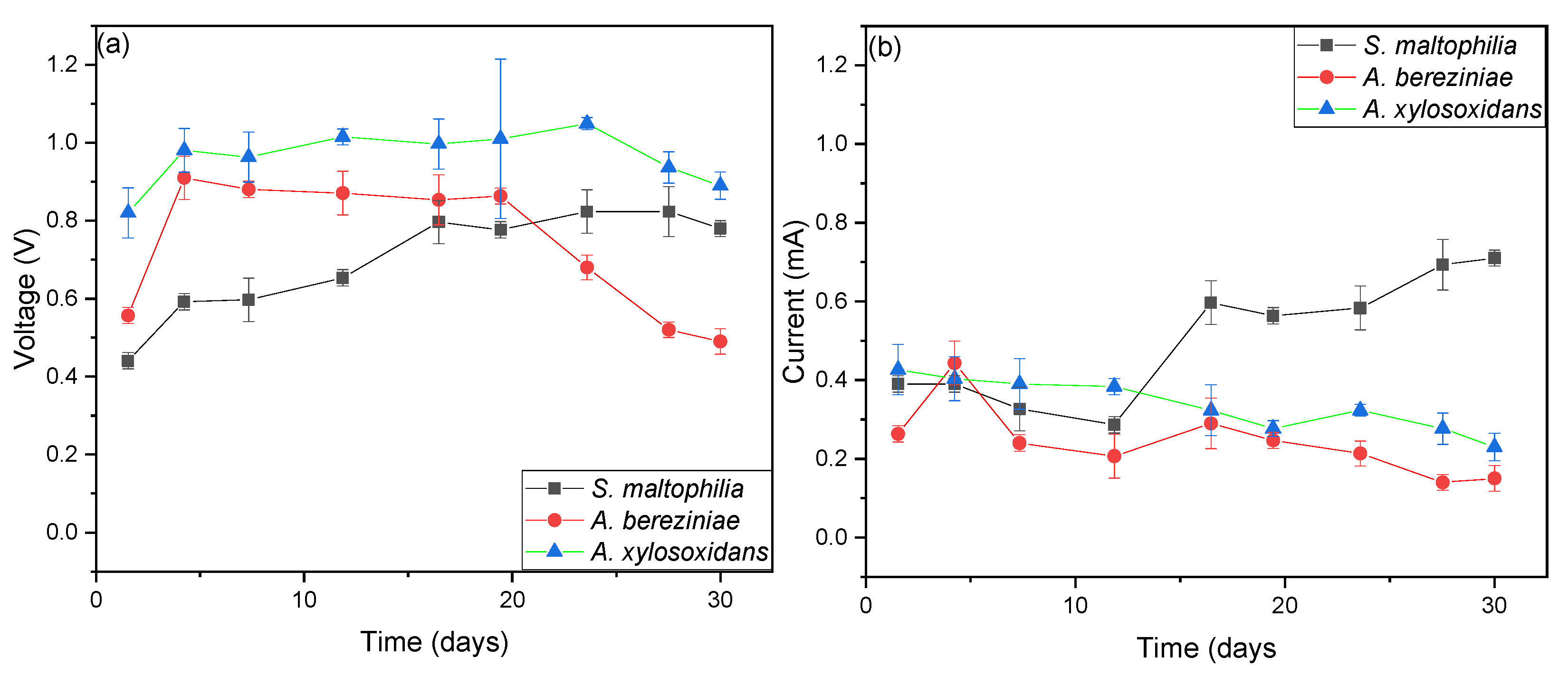
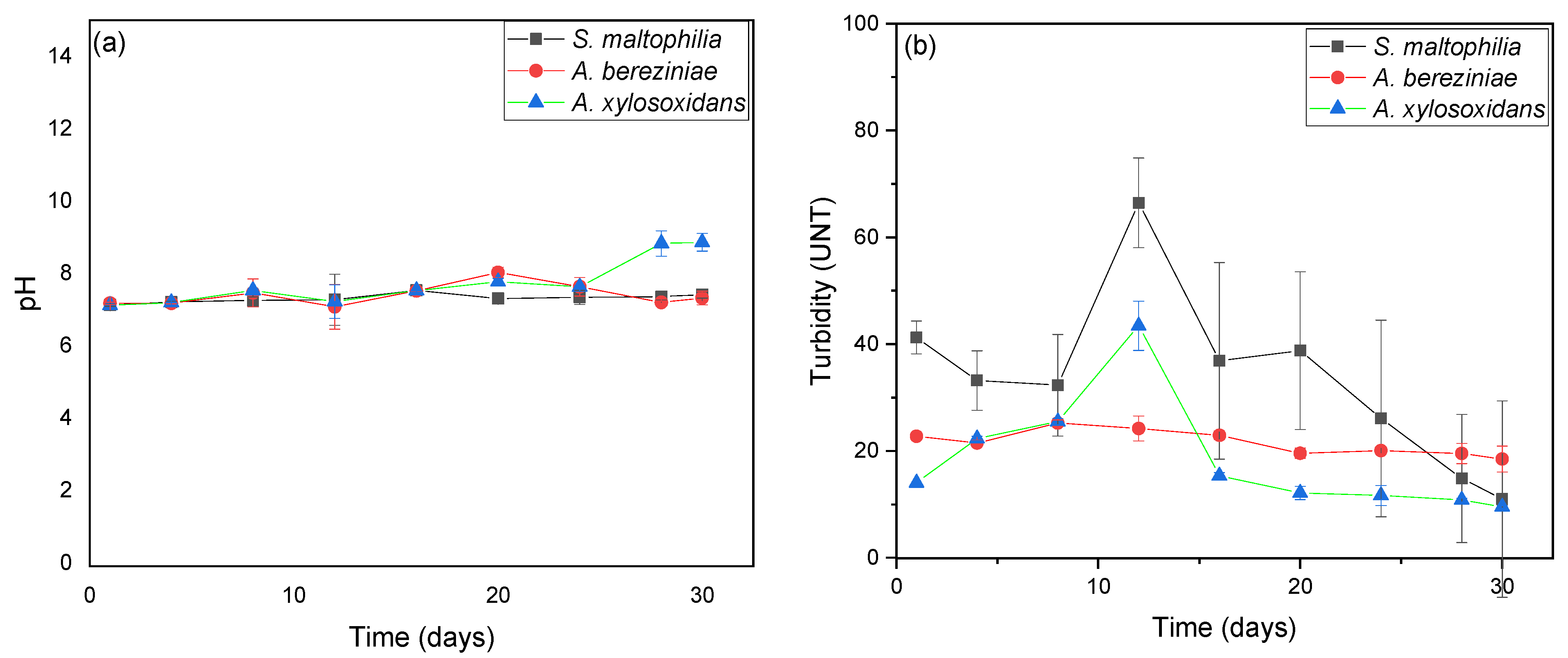
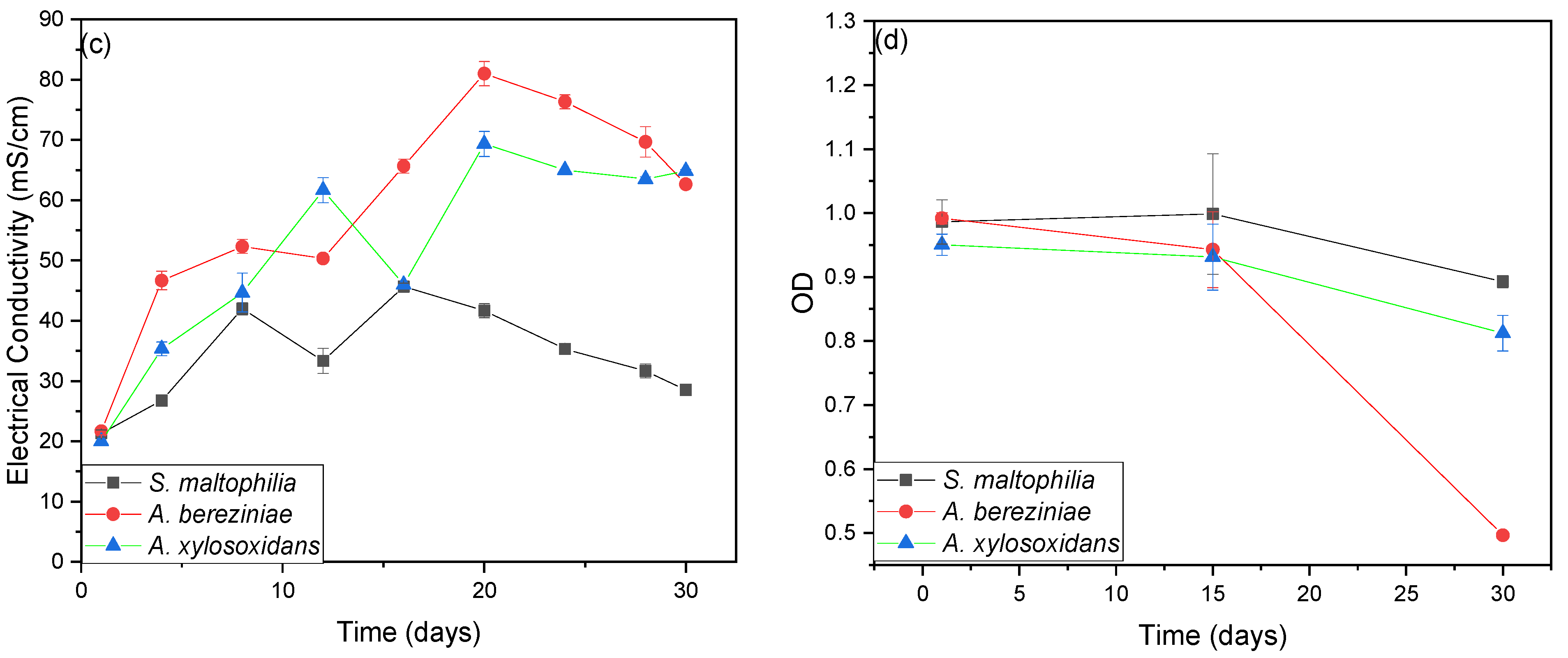
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- van Ruijven, B.J.; De Cian, E.; Sue Wing, I. Amplification of Future Energy Demand Growth Due to Climate Change. Nat. Commun. 2019, 10, 2762. [Google Scholar] [CrossRef]
- International Energy Agency. World Energy Outlook 2021; OECD: Paris, France, 2021; ISBN 9789264654600. [Google Scholar]
- Salehabadi, A.; Umar, M.F.; Ahmad, A.; Ahmad, M.I.; Ismail, N.; Rafatullah, M. Carbon-based Nanocomposites in Solid-state Hydrogen Storage Technology: An Overview. Int. J. Energy Res. 2020, 44, 11044–11058. [Google Scholar] [CrossRef]
- Al-Ghussain, L. Global Warming: Review on Driving Forces and Mitigation: Global Warming: Review on Driving Forces and Mitigation. Environ. Prog. Sustain. Energy 2019, 38, 13–21. [Google Scholar] [CrossRef]
- Guangul, F.M.; Chala, G.T. SWOT Analysis of Wind Energy as a Promising Conventional Fuels Substitute. In Proceedings of the 2019 4th MEC International Conference on Big Data and Smart City (ICBDSC), Muscat, Oman, 15–16 January 2019. [Google Scholar]
- Rathour, R.; Kalola, V.; Johnson, J.; Jain, K.; Madamwar, D.; Desai, C. Treatment of Various Types of Wastewaters Using Microbial Fuel Cell Systems. In Microbial Electrochemical Technology; Mohan, S.V., Varjani, S., Pandey, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 665–692. ISBN 9780444640529. [Google Scholar]
- Vallero, D.A. Wastewater. In Waste; Letcher, T.M., Vallero, D.A., Eds.; Elsevier: San Diego, CA, USA, 2019; pp. 259–290. ISBN 9780128150603. [Google Scholar]
- Häder, D.-P. Ecotoxicological Monitoring of Wastewater. In Bioassays; Häder, D.-P., Erzinger, G.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 369–386. ISBN 9780128118610. [Google Scholar]
- Slate, A.J.; Whitehead, K.A.; Brownson, D.A.C.; Banks, C.E. Microbial Fuel Cells: An Overview of Current Technology. Renew. Sustain. Energy Rev. 2019, 101, 60–81. [Google Scholar] [CrossRef]
- Revelo, D.M.; Hurtado, N.H.; Ruiz, J.O. Celdas de combustible microbianas (CCMs): Un reto para la remoción de materia orgánica y la generación de energía eléctrica. Inf. Tecnológica 2013, 24, 17–28. [Google Scholar] [CrossRef]
- Smida, H.; Flinois, T.; Lebègue, E.; Lagrost, C.; Barrière, F. Microbial Fuel Cells—Wastewater Utilization. In Encyclopedia of Interfacial Chemistry; Wandelt, K., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 328–336. ISBN 9780128098943. [Google Scholar]
- Dutta, K.; Kundu, P.P. Introduction to Microbial Fuel Cells. In Progress and Recent Trends in Microbial Fuel Cells; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1–6. ISBN 9780444640178. [Google Scholar]
- Cao, Y.; Mu, H.; Liu, W.; Zhang, R.; Guo, J.; Xian, M.; Liu, H. Electricigens in the Anode of Microbial Fuel Cells: Pure Cultures versus Mixed Communities. Microb. Cell Fact. 2019, 18, 39. [Google Scholar] [CrossRef] [PubMed]
- Nandy, A.; Kundu, P.P. Configurations of Microbial Fuel Cells. In Progress and Recent Trends in Microbial Fuel Cells; Elsevier: Amsterdam, The Netherlands, 2018; pp. 25–45. ISBN 9780444640178. [Google Scholar]
- Lu, N.; Zhou, S.-G.; Zhuang, L.; Zhang, J.-T.; Ni, J.-R. Electricity Generation from Starch Processing Wastewater Using Microbial Fuel Cell Technology. Biochem. Eng. J. 2009, 43, 246–251. [Google Scholar] [CrossRef]
- Naina Mohamed, S.; Thota Karunakaran, R.; Manickam, M. Enhancement of Bioelectricity Generation from Treatment of Distillery Wastewater Using Microbial Fuel Cell. Environ. Prog. Sustain. Energy 2018, 37, 663–668. [Google Scholar] [CrossRef]
- Li, M.; Zhou, M.; Tian, X.; Tan, C.; McDaniel, C.T.; Hassett, D.J.; Gu, T. Microbial Fuel Cell (MFC) Power Performance Improvement through Enhanced Microbial Electrogenicity. Biotechnol. Adv. 2018, 36, 1316–1327. [Google Scholar] [CrossRef] [PubMed]
- Logan, B.E.; Rossi, R.; Ragab, A.; Saikaly, P.E. Electroactive Microorganisms in Bioelectrochemical Systems. Nat. Rev. Microbiol. 2019, 17, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Clark, D.P.; Pazdernik, N.J. Environmental Biotechnology. In Biotechnology; Clark, D.P., Pazdernik, N.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 393–418. ISBN 9780123850157. [Google Scholar]
- Zhao, N.; Jiang, Y.; Alvarado-Morales, M.; Treu, L.; Angelidaki, I.; Zhang, Y. Electricity Generation and Microbial Communities in Microbial Fuel Cell Powered by Macroalgal Biomass. Bioelectrochemistry 2018, 123, 145–149. [Google Scholar] [CrossRef]
- Segundo, R.-F.; De La Cruz-Noriega, M.; Milly Otiniano, N.; Benites, S.M.; Esparza, M.; Nazario-Naveda, R. Use of Onion Waste as Fuel for the Generation of Bioelectricity. Molecules 2022, 27, 625. [Google Scholar] [CrossRef] [PubMed]
- Bose, D.; Gopinath, M.; Vijay, P.; Sridharan, S.; Rawat, R.; Bahuguna, R. Bioelectricity Generation and Biofilm Analysis from Sewage Sources Using Microbial Fuel Cell. Fuel 2019, 255, 115815. [Google Scholar] [CrossRef]
- Ryan, R.P.; Monchy, S.; Cardinale, M.; Taghavi, S.; Crossman, L.; Avison, M.B.; Berg, G.; van der Lelie, D.; Dow, J.M. The Versatility and Adaptation of Bacteria from the Genus Stenotrophomonas. Nat. Rev. Microbiol. 2009, 7, 514–525. [Google Scholar] [CrossRef]
- Adegoke, A.; Stenström, T.; Okoh, A. Stenotrophomonas maltophilia as an Emerging Ubiquitous Pathogen: Looking Beyond Contemporary Antibiotic Therapy. Front. Microbiol. 2017, 8, 2276. [Google Scholar] [CrossRef]
- Maslova, O.; Mindlin, S.; Beletsky, A.; Mardanov, A.; Petrova, M. Plasmids as Key Players in Acinetobacter Adaptation. Int. J. Mol. Sci. 2022, 23, 10893. [Google Scholar] [CrossRef]
- Bramhachari, P.V.; Reddy, D.R.S.; Kotresha, D. Biodegradation of Catechol by Free and Immobilized Cells of Achromobacter Xylosoxidans Strain 15DKVB Isolated from Paper and Pulp Industrial Effluents. Biocatal. Agric. Biotechnol. 2016, 7, 36–44. [Google Scholar] [CrossRef]
- Rossi, R.; Hur, A.Y.; Page, M.A.; Thomas, A.O.; Butkiewicz, J.J.; Jones, D.W.; Baek, G.; Saikaly, P.E.; Cropek, D.M.; Logan, B.E. Pilot Scale Microbial Fuel Cells Using Air Cathodes for Producing Electricity While Treating Wastewater. Water Res. 2022, 215, 118208. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, P.; Dong, S.; Yu, Y.; Chen, H.; Xiao, J. Using Watermelon Rind and Nitrite-Containing Wastewater for Electricity Production in a Membraneless Biocathode Microbial Fuel Cell. J. Clean. Prod. 2021, 307, 127306. [Google Scholar] [CrossRef]
- Obileke, K.; Onyeaka, H.; Meyer, E.L.; Nwokolo, N. Microbial Fuel Cells, a Renewable Energy Technology for Bio-Electricity Generation: A Mini-Review. Electrochem. Commun. 2021, 125, 107003. [Google Scholar] [CrossRef]
- Konovalova, E.Y.; Stom, D.I.; Zhdanova, G.O.; Yuriev, D.A.; Li, Y.; Barbora, L.; Goswami, P. The Microorganisms Used for Working in Microbial Fuel Cells. AIP Conf. Proc. 2018, 1952, 020017. [Google Scholar]
- Prathiba, S.; Kumar, P.S.; Vo, D.-V.N. Recent Advancements in Microbial Fuel Cells: A Review on Its Electron Transfer Mechanisms, Microbial Community, Types of Substrates and Design for Bio-Electrochemical Treatment. Chemosphere 2022, 286, 131856. [Google Scholar] [CrossRef]
- Ramírez, B.; Jesfredt, J.; Laiza, C.; Antonio, M. Obtención de Combustible Líquido a Partir de Lodos Residuales de las Lagunas de Oxidación de la Planta de Tratamiento de Agua Residual de Covicorti-Trujillo. Available online: https://renati.sunedu.gob.pe/handle/sunedu/2697195 (accessed on 18 September 2006).
- Huarachi-Olivera, R.; Dueñas-Gonza, A.; Yapo-Pari, U.; Vega, P.; Romero-Ugarte, M.; Tapia, J.; Molina, L.; Lazarte-Rivera, A.; Pacheco-Salazar, D.G.; Esparza, M. Bioelectrogenesis with Microbial Fuel Cells (MFCs) Using the Microalga Chlorella Vulgaris and Bacterial Communities. Electron. J. Biotechnol. 2018, 31, 34–43. [Google Scholar] [CrossRef]
- Rojas-Flores, S.; De La Cruz-Noriega, M.; Cabanillas-Chirinos, L.; Benites, S.M.; Nazario-Naveda, R.; Delfín-Narciso, D.; Rojas-Villacorta, W. Use of Kiwi Waste as Fuel in MFC and Its Potential for Use as Renewable Energy. Fermentation 2023, 9, 446. [Google Scholar] [CrossRef]
- Santiago, B.; Rojas-Flores, S.; De La Cruz Noriega, M.; Cabanillas-Chirinos, L.; Otiniano, N.M.; Silva-Palacios, F.; Luis, A.S. Bioelectricity from Saccharomyces cerevisiae yeast through low-cost microbial fuel cells. In Proceedings of the 18th LACCEI International Multi-Conference for Engineering, Education, and Technology: Engineering, Integration, and Alliances for a Sustainable Development, Virtual, 27 July 2020. [Google Scholar]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, J.; Shinde, S.; Wang, X.; Li, Y.; Dai, Y.; Liu, X. Simultaneous wastewater treatment and energy harvesting in microbial fuel cells: An update on the biocatalysts. RSC Adv. 2020, 10, 25874–25887. [Google Scholar] [CrossRef]
- Arkatkar, A.; Mungray, A.K.; Sharma, P. Bioelectrochemical behaviour of a sequentially added biocatalytic coculture in a microbial fuel cell. J. Basic Microbiol. 2020, 60, 562–573. [Google Scholar] [CrossRef]
- Aftab, S.; Shah, A.; Nisar, J.; Ashiq, M.N.; Akhter, M.S.; Shah, A.H. Marketability prospects of microbial fuel cells for sustainable energy generation. Energy Fuels 2020, 34, 9108–9136. [Google Scholar] [CrossRef]
- Breheny, M.; Bowman, K.; Farahmand, N.; Gomaa, O.; Keshavarz, T.; Kyazze, G. Biocatalytic electrode improvement strategies in microbial fuel cell systems. J. Chem. Technol. Biotechnol. 2019, 94, 2081–2091. [Google Scholar] [CrossRef]
- Qiu, S.; Guo, Z.; Naz, F.; Yang, Z.; Yu, C. An overview in the development of cathode materials for the improvement in power generation of microbial fuel cells. Bioelectrochemistry 2021, 141, 107834. [Google Scholar] [CrossRef]
- Mousavi, M.R.; Ghasemi, S.; Sanaee, Z.; Nejad, Z.G.; Mardanpour, M.M.; Yaghmaei, S.; Ghorbanzadeh, M. Improvement of the microfluidic microbial fuel cell using a nickel nanostructured electrode and microchannel modifications. J. Power Sources 2019, 437, 226891. [Google Scholar] [CrossRef]
- Li, C.; He, W.; Liang, D.; Tian, Y.; Yadav, R.S.; Li, D.; Feng, Y. The anaerobic and starving treatment eliminates filamentous bulking and recovers biocathode biocatalytic activity with residual organic loading in microbial electrochemical system. Chem. Eng. J. 2021, 404, 127072. [Google Scholar] [CrossRef]
- Puig, S.; Serra, M.; Coma, M.; Cabré, M.; Balaguer, M.D.; Colprim, J. Effect of PH on Nutrient Dynamics and Electricity Production Using Microbial Fuel Cells. Bioresour. Technol. 2010, 101, 9594–9599. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, V.B.; Simões, M.; Melo, L.F.; Pinto, A.M.F.R. Overview on the Developments of Microbial Fuel Cells. Biochem. Eng. J. 2013, 73, 53–64. [Google Scholar] [CrossRef]
- Bose, D.; Dhawan, H.; Kandpal, V.; Vijay, P.; Gopinath, M. Bioelectricity Generation from Sewage and Wastewater Treatment Using Two-Chambered Microbial Fuel Cell. Int. J. Energy Res. 2018, 42, 4335–4344. [Google Scholar] [CrossRef]
- De La Cruz Noriega, M.; Rojas-Flores, S.; Benites, S.M.; Otiniano, N.M.; Cabanillas-Chirinos, L.; Rodriguez-Yupanqui, M.; Valdiviezo-Dominguez, F.; Rojas-Villacorta, W. Generación bioelectricidad a partir de aguas residuales mediante celdas de combustible. In Proceedings of the 19th LACCEI International Multi-Conference for Engineering, Education, and Technology, Virtual, 19–23 July 2021. [Google Scholar]
- Heddam, S. Bat Algorithm Optimized Extreme Learning Machine: A New Modeling Strategy for Predicting River Water Turbidity at the United States. In Handbook of Hydroinformatics; Eslamian, S., Eslamian, F., Eds.; Elsevier: Amsterdam, The Netherlands, 2023; pp. 39–55. ISBN 9780128212851. [Google Scholar]
- Tyler, A.; Hunter, P.; De Keukelaere, L.; Ogashawara, I.; Spyrakos, E. Remote Sensing of Inland Water Quality. In Encyclopedia of Inland Waters; Mehner, T., Tockner, K., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 570–584. ISBN 9780128220412. [Google Scholar]
- Mardanpour, M.M.; Nasr Esfahany, M.; Behzad, T.; Sedaqatvand, R. Single Chamber Microbial Fuel Cell with Spiral Anode for Dairy Wastewater Treatment. Biosens. Bioelectron. 2012, 38, 264–269. [Google Scholar] [CrossRef]
- Ma, P.; Ma, H.; Sabatino, S.; Galia, A.; Scialdone, O. Electrochemical Treatment of Real Wastewater. Part 1: Effluents with Low Conductivity. Chem. Eng. J. 2018, 336, 133–140. [Google Scholar] [CrossRef]
- de Sousa, D.N.R.; Mozeto, A.A.; Carneiro, R.L.; Fadini, P.S. Electrical Conductivity and Emerging Contaminant as Markers of Surface Freshwater Contamination by Wastewater. Sci. Total Environ. 2014, 484, 19–26. [Google Scholar] [CrossRef]
- Rahmani, A.R.; Navidjouy, N.; Rahimnejad, M.; Alizadeh, S.; Samarghandi, M.R.; Nematollahi, D. Effect of different concentrations of substrate in microbial fuel cells toward bioenergy recovery and simultaneous wastewater treatment. Environ. Technol. 2020, 43, 1–9. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Ibrahim, M.N.; Yaakop, A.S.; Umar, K.; Ahmad, A. Modified graphene oxide anode: A bioinspired waste material for bioremediation of Pb2+ with energy generation through microbial fuel cells. Chem. Eng. J. 2021, 417, 128052. [Google Scholar] [CrossRef]
- Zafar, Z.; Ayaz, K.; Nasir, H.; Yousaf, S.; Sharafat, I.; Ali, N. Electrochemical performance of biocathode microbial fuel cells using petroleum-contaminated soil and hot water spring. Int. J. Environ. Sci. Technol. 2019, 16, 1487–1500. [Google Scholar] [CrossRef]
- Li, S.; Cheng, C.; Thomas, A. Carbon-Based Microbial-Fuel-Cell Electrodes: From Conductive Supports to Active Catalysts. Adv. Mater. 2017, 29, 1602547. [Google Scholar] [CrossRef] [PubMed]
- An, J.; Li, N.; Wan, L.; Zhou, L.; Du, Q.; Li, T.; Wang, X. Electric field induced salt precipitation into activated carbon air-cathode causes power decay in microbial fuel cells. Water Res. 2017, 123, 369–377. [Google Scholar] [CrossRef]
- Akinwumi, O.D.; Aremu, M.O.; Agarry, S.E. Enhanced microbial fuel cell-bioelectricity generation and pollutant removal from brewery wastewater and modelling the kinetics. Biomass Conv. Bioref. 2022, 1–18. [Google Scholar] [CrossRef]
- Bose, D.; Bhattacharya, R.; Gopinath, M.; Vijay, P.; Krishnakumar, B. Bioelec-tricity production and bioremediation from sugarcane industry wastewater using mi-crobial fuel cells with activated carbon cathodes. Results Eng. 2023, 18, 101052. [Google Scholar] [CrossRef]
- SUNASS—Superintendencia Nacional de Servicios de Saneamiento. Diagnóstico de las Plantas de Tratamiento de Aguas Residuales (PTAR) en el ámbito de las Empresas Prestadoras. Primera Edición, Perú. 2022, p. 278. Available online: https://www.sunass.gob.pe/wp-content/uploads/2022/06/Informe-de-diagnostico-de-las-Plantas-de-Tratamiento-de-Aguas-Residuales-PTAR_VdigitalConcomentario.pdf (accessed on 15 April 2023).
- Shanks, O.C.; Newton, R.J.; Kelty, C.A.; Huse, S.M.; Sogin, M.L.; McLellan, S.L. Comparison of the microbial community structures of untreated wastewaters from different geographic locales. Appl. Environ. Microbiol. 2013, 79, 2906–2913. [Google Scholar] [CrossRef]
- Yasir, M. Analysis of microbial communities and pathogen detection in domestic sewage using metagenomic sequencing. Diversity 2020, 13, 6. [Google Scholar] [CrossRef]
- Zhang, D.; Peng, Y.; Chan, C.L.; On, H.; Wai, H.K.F.; Shekhawat, S.S.; Tun, H.M. Metagenomic survey reveals more diverse and abundant antibiotic resistance genes in municipal wastewater than hospital wastewater. Front. Microbiol. 2021, 12, 712843. [Google Scholar] [CrossRef]
- Huijbers, P.M.; Blaak, H.; de Jong, M.C.; Graat, E.A.; Vandenbroucke-Grauls, C.M.; de Roda Husman, A.M. Role of the environment in the transmission of antimicrobial resistance to humans: A review. Environ. Sci. Technol. 2015, 49, 11993–12004. [Google Scholar] [CrossRef]
- Ferreira, C.; Otani, S.; Aarestrup, F.; Manaia, C. Quantitative PCR versus metagenomics for monitoring antibiotic resistance genes: Balancing high sensitivity and broad coverage. FEMS Microbes 2023, 4, xtad008. [Google Scholar] [CrossRef]
- Lira, F.; Vaz-Moreira, I.; Tamames, J.; Manaia, C.M.; Martínez, J.L. Metagenomic analysis of an urban resistome before and after wastewater treatment. Sci. Rep. 2020, 10, 8174. [Google Scholar] [CrossRef] [PubMed]
- Chu, B.; Petrovich, M.; Chaudhary, A.; Wright, D.; Murphy, B.; Wells, G.; Poretsky, R. Metagenomics reveals the impact of wastewater treatment plants on the dispersal of microorganisms and genes in aquatic sediments. Appl. Environ. Microbiol. 2018, 84, e02168-17. [Google Scholar] [CrossRef] [PubMed]
- Jankowski, P.; Gan, J.; Le, T.; McKennitt, M.; Garcia, A.; Yanaç, K.; Uyaguari-Diaz, M. Metagenomic community composition and resistome analysis in a full-scale cold climate wastewater treatment plant. Environ. Microbiome 2022, 17, 3. [Google Scholar] [CrossRef] [PubMed]
- Logan, B.E.; Regan, J.M. Electricity-producing bacterial communities in microbial fuel cells. Trends Microbiol. 2006, 14, 512–518. [Google Scholar] [CrossRef]
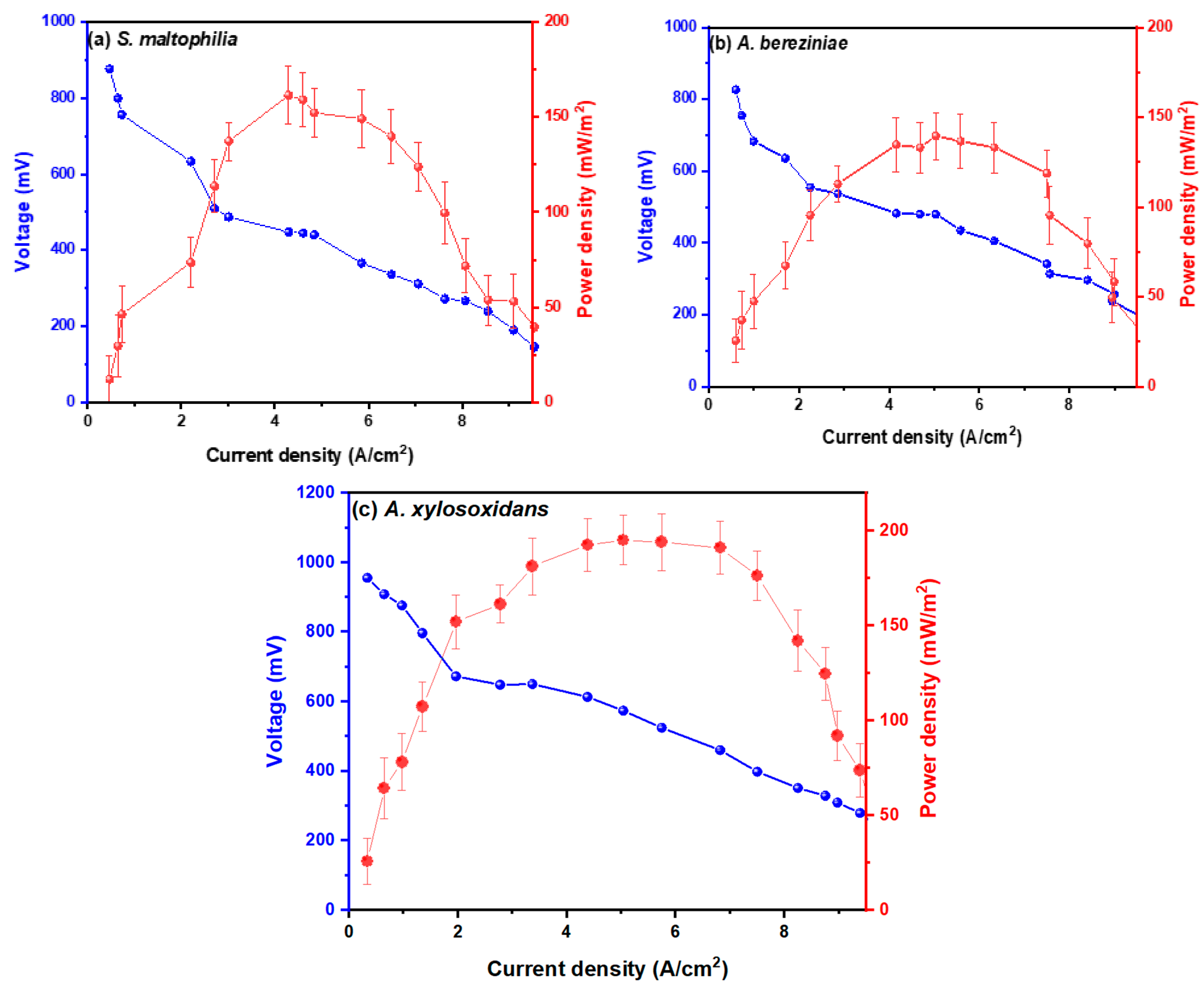
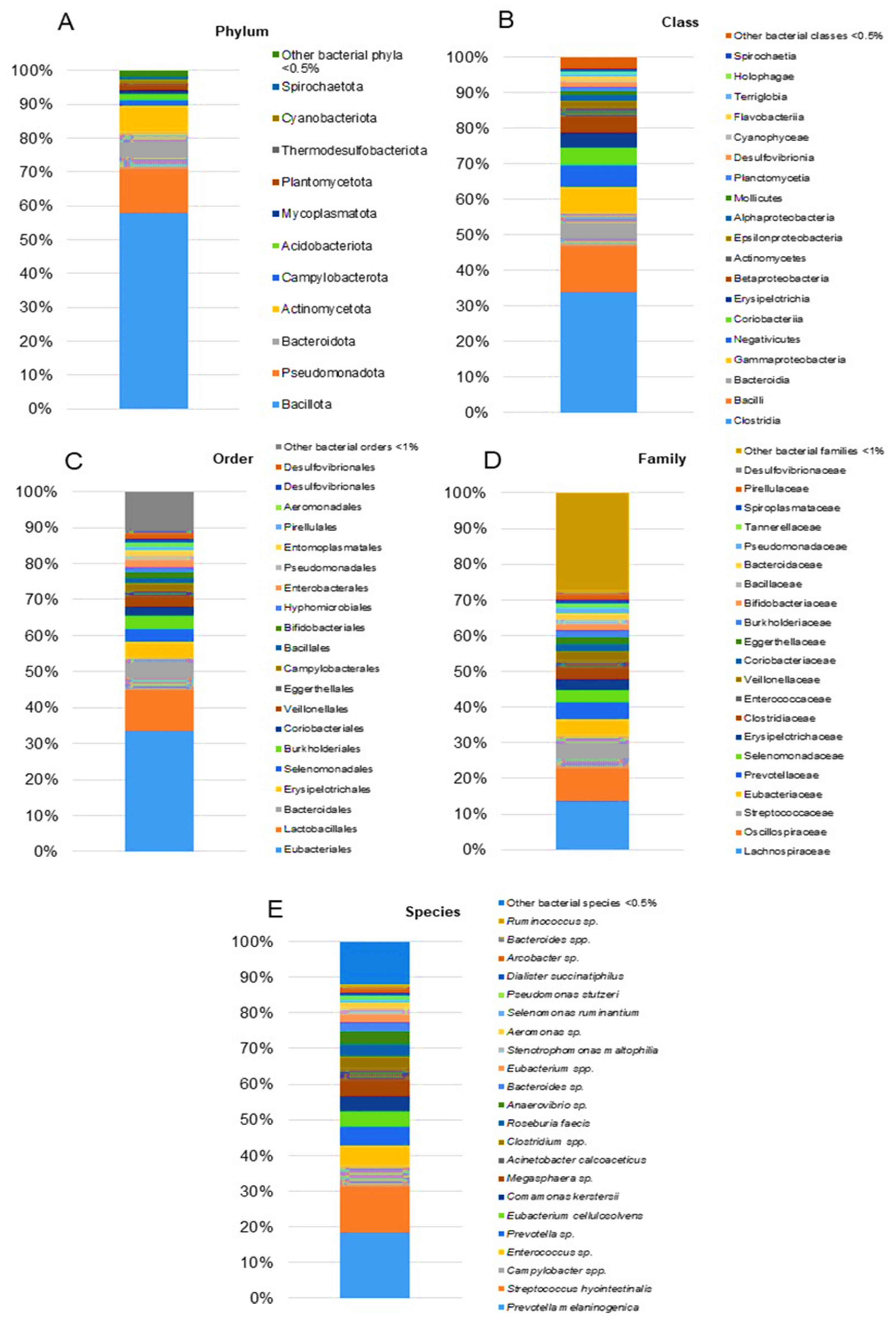
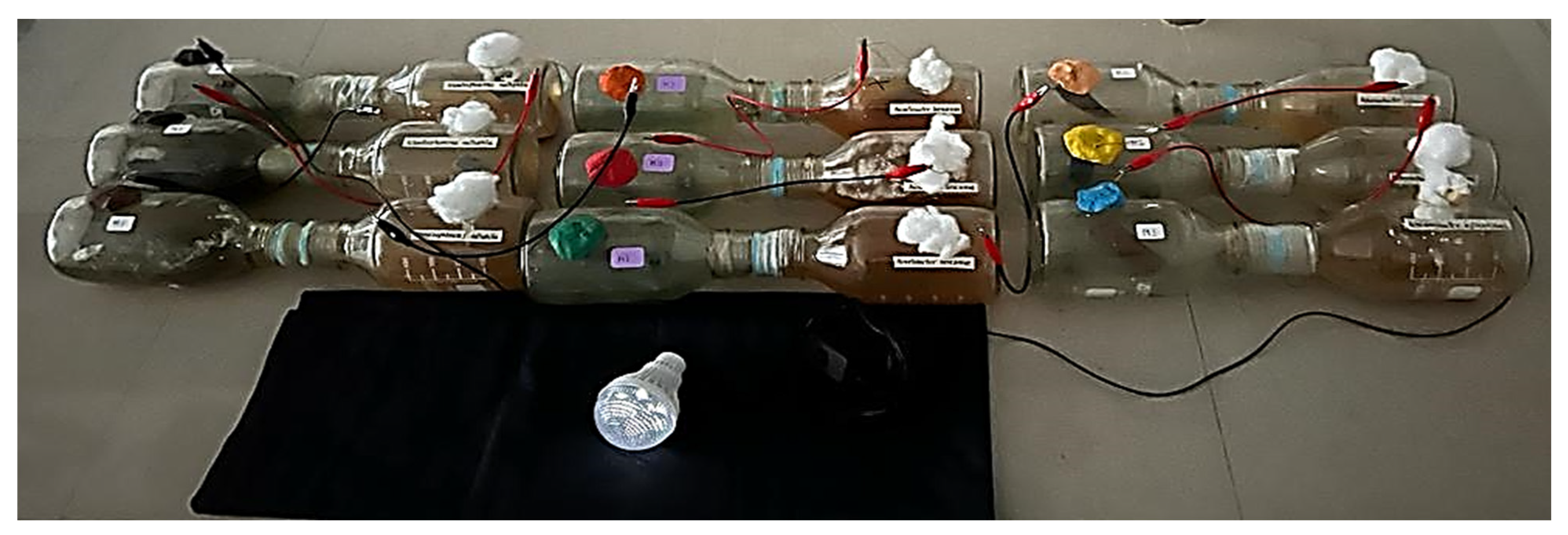
| Day 1 | Day 15 | Day 30 | |
|---|---|---|---|
| System * | DBO (mg O2/L) | DBO (mg O2/L) | DBO (mg O2/L) |
| MFC-1 | 640 | 65 | 5 |
| MFC-2 | 640 | 20 | 3 |
| MFC-3 | 640 | 20 | 3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De La Cruz-Noriega, M.; Benites, S.M.; Rojas-Flores, S.; Otiniano, N.M.; Sabogal Vargas, A.M.; Alfaro, R.; Cabanillas-Chirinos, L.; Rojas-Villacorta, W.; Nazario-Naveda, R.; Delfín-Narciso, D. Use of Wastewater and Electrogenic Bacteria to Generate Eco-Friendly Electricity through Microbial Fuel Cells. Sustainability 2023, 15, 10640. https://doi.org/10.3390/su151310640
De La Cruz-Noriega M, Benites SM, Rojas-Flores S, Otiniano NM, Sabogal Vargas AM, Alfaro R, Cabanillas-Chirinos L, Rojas-Villacorta W, Nazario-Naveda R, Delfín-Narciso D. Use of Wastewater and Electrogenic Bacteria to Generate Eco-Friendly Electricity through Microbial Fuel Cells. Sustainability. 2023; 15(13):10640. https://doi.org/10.3390/su151310640
Chicago/Turabian StyleDe La Cruz-Noriega, Magaly, Santiago M. Benites, Segundo Rojas-Flores, Nelida M. Otiniano, Ana M. Sabogal Vargas, Rubén Alfaro, Luis Cabanillas-Chirinos, Walter Rojas-Villacorta, Renny Nazario-Naveda, and Daniel Delfín-Narciso. 2023. "Use of Wastewater and Electrogenic Bacteria to Generate Eco-Friendly Electricity through Microbial Fuel Cells" Sustainability 15, no. 13: 10640. https://doi.org/10.3390/su151310640
APA StyleDe La Cruz-Noriega, M., Benites, S. M., Rojas-Flores, S., Otiniano, N. M., Sabogal Vargas, A. M., Alfaro, R., Cabanillas-Chirinos, L., Rojas-Villacorta, W., Nazario-Naveda, R., & Delfín-Narciso, D. (2023). Use of Wastewater and Electrogenic Bacteria to Generate Eco-Friendly Electricity through Microbial Fuel Cells. Sustainability, 15(13), 10640. https://doi.org/10.3390/su151310640







