Which Factors Favour Biodiversity in Iberian Dehesas?
Abstract
:1. Introduction
2. Methods
3. Results and Discussion
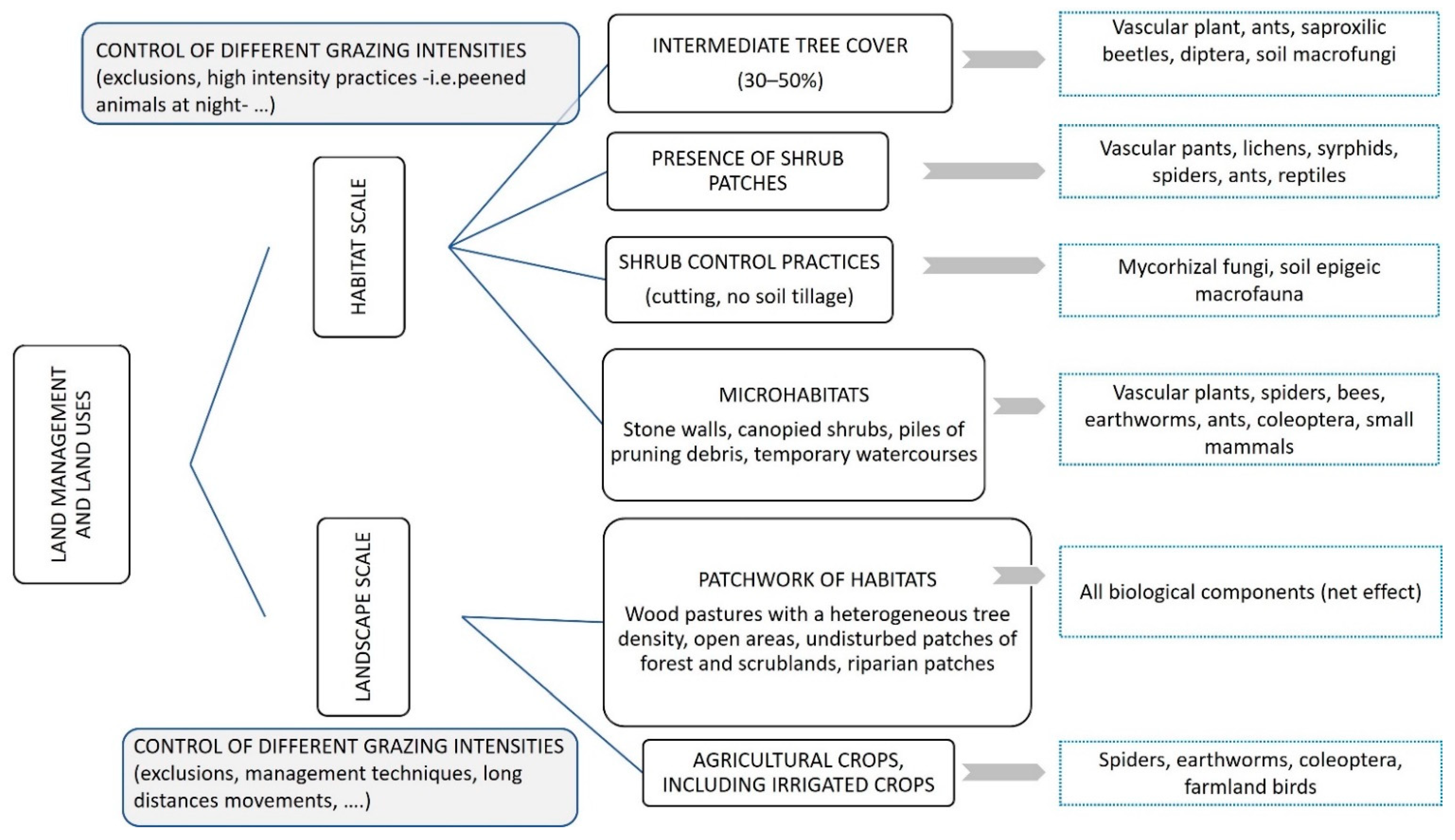
3.1. Tree Factor
3.2. Shrub Factor
3.3. Topography
3.4. Fertilization
3.5. Grazing Management and Land Use
3.6. Habitat Heterogeneity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Keenleyside, C.; Beaufoy, G.; Tucker, G.; Jones, G. High Nature Value farming throughout EU-27 and its financial support under the CAP. In Report Prepared for DG Environment; Contract No ENV B.1/ETU/2012/0035; Institute for European Environmental Policy: London, UK, 2014. [Google Scholar]
- Moreno, G.; Aviron, S.; Berg, S.; Crous-Duran, J.; Franca, A.; de Jalón, S.G.; Hartel, T.; Mirck, J.; Pantera, A.; Palma, J.H.N.; et al. Agroforestry Systems of High Nature and Cultural Value in Europe: Provision of Commercial Goods and Other Ecosystem Services. Agrofor. Syst. 2018, 92, 877–891. [Google Scholar] [CrossRef]
- Moreno, G.; Franca, A.; Pinto-Correia, T.; Godinho, S. Multifunctionality and Dynamics of Silvopastoral Systems View Project. Options Méditerranéennes 2014, 109, 421–436. [Google Scholar]
- European Commission. Interpretation Manual of European Union Habitats–EUR28; European Commission, DG Environment: Brussels, Belgium, 2013. [Google Scholar]
- Manning, A.D.; Fischer, J.; Lindenmayer, D.B. Scattered Trees are Keystone Structures—Implications for Conservation. Biol. Conserv. 2006, 132, 311–321. [Google Scholar] [CrossRef]
- Vogiatzakis, I.N.; Mannion, A.M.; Griffiths, G.H. Mediterranean Ecosystems: Problems and Tools for Conservation. Prog. Phys. Geogr. 2006, 30, 175–200. [Google Scholar] [CrossRef]
- Díaz, M.; Pulido, J.F. 6310 Dehesas Perennifolias de Quercus spp. In Bases Ecológicas Preliminares Para la Conservación de los Tipos de Hábitat de Interés Comunitario en España; Ministerio de Medio Ambiente, Medio Rural y Marino: Madrid, Spain, 2009; pp. 1–69. [Google Scholar]
- Plieninger, T.; Van Der Horst, D.; Schleyer, C.; Bieling, C. Sustaining Ecosystem Services in Cultural Landscapes: Analysis and Management Options Sustaining Ecosystem Services in Cultural Landscapes. Ecol. Soc. 2014, 19, art59. [Google Scholar] [CrossRef]
- Bugalho, M.N.; Dias, F.S.; Briñas, B.; Cerdeira, J.O. Using the High Conservation Value Forest Concept and Pareto Optimization to Identify Areas Maximizing Biodiversity and Ecosystem Services in Cork Oak Landscapes. Agrofor. Syst. 2016, 90, 35–44. [Google Scholar] [CrossRef]
- Oggioni, S.D.; Ochoa-Hueso, R.; Peco, B. Livestock Grazing Abandonment Reduces Soil Microbial Activity and Carbon Storage in a Mediterranean Dehesa. Appl. Soil Ecol. 2020, 153, 103588. [Google Scholar] [CrossRef]
- Andersen, E.; Baldock, D.; Bennett, H.; Beaufoy, G.; Bignal, E.; Brouwer, F.; Elbersen, B.; Eiden, G.; Godeschalk, F.; Jones, G.; et al. Developing a High Nature Value Farming Area Indicator; Internal Report for the European Environment Agency; Institute for European Environmental Policy: Brussels, Belgium, 2003. [Google Scholar]
- Eichhorn, M.P.; Paris, P.; Herzog, F.; Incoll, L.D.; Liagre, F.; Mantzanas, K.; Mayus, M.; Moreno, G.; Papanastasis, V.P.; Pilbeam, D.J.; et al. Silvoarable Systems in Europe-Past, Present and Future Prospects. Agrofor. Syst. 2006, 67, 29–50. [Google Scholar] [CrossRef]
- Acebes, P.; Pereira, D.; Oñate, J.J. Towards the Identification and Assessment of HNV Dehesas: A Meso-Scale Approach. Agrofor. Syst. 2016, 90, 7–22. [Google Scholar] [CrossRef]
- Hernández-Lambraño, R.E.; de la Cruz, D.R.; Sánchez-Agudo, J.Á. Spatial Oak Decline Models to Inform Conservation Planning in the Central-Western Iberian Peninsula. For. Ecol. Manag. 2019, 441, 115–126. [Google Scholar] [CrossRef]
- Plieninger, T.; Rolo, V.; Moreno, G. Large-Scale Patterns of Quercus ilex, Quercus suber, and Quercus pyrenaica Regeneration in Central-Western Spain. Ecosystems 2010, 13, 644–660. [Google Scholar] [CrossRef]
- Simões, M.P.; Belo, A.F.; Fernandes, M.; Madeira, M. Regeneration Patterns of Quercus Suber According to Montado Management Systems. Agrofor. Syst. 2016, 90, 107–115. [Google Scholar] [CrossRef]
- Almeida, M.; Azeda, C.; Guiomar, N.; Pinto-Correia, T. The Effects of Grazing Management in Montado Fragmentation and Heterogeneity. Agrofor. Syst. 2016, 90, 69–85. [Google Scholar] [CrossRef]
- Godinho, S.; Guiomar, N.; Machado, R.; Santos, P.; Sá-Sousa, P.; Fernandes, J.P.; Neves, N.; Pinto-Correia, T. Assessment of Environment, Land Management, and Spatial Variables on Recent Changes in Montado Land Cover in Southern Portugal. Agrofor. Syst. 2016, 90, 177–192. [Google Scholar] [CrossRef]
- Acosta Naranjo, R. Dehesas de la Sobremodernidad. La Cadencia y el Vértigo; Diputación Provincial de Badajoz: Badajoz, Spain, 2008. [Google Scholar]
- Junta de Andalucía. Plan Director de las Dehesas de Andalucía; Technical Report; Junta de Andalucía: Sevilla, Spain, 2017. [Google Scholar]
- Díaz, M.; Campos, P.; Pulido, F.J. The Spanish dehesas: A diversity in land-use and wildlife. In Farming and Birds in Europe: The Common Agricultural Policy and Its Implications for Bird Conservation; Pain, D., Pienkowski, M., Eds.; Academic Press: London, UK, 1997; pp. 178–209. [Google Scholar]
- Moreno, G.; Cáceres, Y. System Report: Iberian Dehesas, Spain. AGFORWARD Project Report, 2016. Available online: www.agforward.eu (accessed on 19 December 2021).
- Pineda, F.D.; Nicolas, J.P.; Ruiz, M.; Bernaldez, F.G. Succession, Diversité et Amplitude de Niche Dans Les Pâturages Du Centre de La Péninsule Ibérique. Vegetatio 1981, 46, 267–277. [Google Scholar] [CrossRef]
- Marañón, T. Diversidad Florística y Heterogeneidad Ambiental En Una Dehesa de Sierra Morena. An. Edafol. Abrobiologia 1985, 44, 1183–1197. [Google Scholar]
- Rico, M.; Puerto, A. Estructura Básica Generada Por El Arbolado En Pastos Semiáridos (Ecosistemas de Dehesa). Pastos 1988, 1819, 13–28. [Google Scholar]
- Puerto, A.; Rico, M.; Matías, M.D.; García, J.A. Variation in Structure and Diversity in Mediterranean Grasslands Related to Trophic Status and Grazing Intensity. J. Veg. Sci. 1990, 1, 445–452. [Google Scholar] [CrossRef]
- Díaz, M. Biodiversity in the Dehesa. In Agroforestry Systems as a Technique for Sutainable Land Management; Mosquera-Losada, M.R., Fernández-Lorenzo, J.L., Rigueiro-Rodríguez, A., Eds.; Programme Azahar, AECID: Madrid, Spain, 2009; pp. 209–225. [Google Scholar]
- Bugalho, M.N.; Lecomte, X.; Gonçalves, M.; Caldeira, M.C.; Branco, M. Establishing Grazing and Grazing-Excluded Patches Increases Plant and Invertebrate Diversity in a Mediterranean Oak Woodland. For. Ecol. Manag. 2011, 261, 2133–2139. [Google Scholar] [CrossRef]
- Moreno, G.; Gonzalez-Bornay, G.; Pulido, F.; Lopez-Diaz, M.L.; Bertomeu, M.; Juárez, E.; Diaz, M. Exploring the Causes of High Biodiversity of Iberian Dehesas: The Importance of Wood Pastures and Marginal Habitats. Agrofor. Syst. 2016, 90, 87–105. [Google Scholar] [CrossRef]
- Simonson, W.D.; Allen, H.D.; Parham, E.; de Basto e Santos, E.; Hotham, P. Modelling Biodiversity Trends in the Montado (Wood Pasture) Landscapes of the Alentejo, Portugal. Landsc. Ecol. 2018, 33, 811–827. [Google Scholar] [CrossRef]
- Díaz, M.; González, E.; Muñoz-Pulido, R.; Naveso, M.A. Habitat Selection Patterns of Common Cranes Grus grus Wintering in Holm oak Quercus Ilex Dehesas of Central Spain: Effects of Human Management. Biol. Conserv. 1996, 75, 119–123. [Google Scholar] [CrossRef]
- Avilés, J.M. Common Cranes Grus grus and Habitat Management in Holm Oak Dehesas of Spain. Biodivers. Conserv. 2004, 13, 2015–2025. [Google Scholar] [CrossRef]
- Carrete, M.; Donázar, J.A. Application of Central-Place Foraging Theory Shows the Importance of Mediterranean Dehesas for the Conservation of the Cinereous Vulture, Aegypius Monachus. Biol. Conserv. 2005, 126, 582–590. [Google Scholar] [CrossRef]
- Martin-Díaz, P.; Cortés-Avizanda, A.; Serrano, D.; Arrondo, E.; Sánchez-Zapata, J.A.; Donázar, J.A. Rewilding Processes Shape the Use of Mediterranean Landscapes by an Avian Top Scavenger. Sci. Rep. 2020, 10, 2853. [Google Scholar] [CrossRef] [PubMed]
- Marañón, T. Plant Species Richness and Canopy Effect in the Savanna-like “dehesa” of SW Spain. Ecol. Mediterr. 1986, 12, 131–141. [Google Scholar] [CrossRef]
- Díez, C.; Luis, E.; Tárrega, R. Variación de la diversidad y organización de la comunidad herbácea en robledales adehesados de Quercus pyrenaica. In Proceedings of the 32th Reunión Científica de la SEEP, Pamplona, Spain, 1–5 June 1992; pp. 128–133. [Google Scholar]
- Fernández-Moya, J.; Miguel-Ayanz, A.S.; Cañellas, I.; Gea-Izquierdo, G. Variability in Mediterranean Annual Grassland Diversity Driven by Small-Scale Changes in Fertility and Radiation. Plant Ecol. 2011, 212, 865–877. [Google Scholar] [CrossRef]
- López-Carrasco, C.; López-Sánchez, A.; San Miguel, A.; Roig, S. The Effect of Tree Cover on the Biomass and Diversity of the Herbaceous Layer in a Mediterranean Dehesa. Grass Forage Sci. 2015, 70, 639–650. [Google Scholar] [CrossRef]
- López-Sánchez, A.; San Miguel, A.; Dirzo, R.; Roig, S. Scattered Trees and Livestock Grazing as Keystones Organisms for Sustainable Use and Conservation of Mediterranean Dehesas. J. Nat. Conserv. 2016, 33, 58–67. [Google Scholar] [CrossRef]
- López-Sánchez, A.; San Miguel, A.; López-Carrasco, C.; Huntsinger, L.; Roig, S. The Important Role of Scattered Trees on the Herbaceous Diversity of a Grazed Mediterranean Dehesa. Acta Oecologica 2016, 76, 31–38. [Google Scholar] [CrossRef]
- Rolo, V.; Rivest, D.; López-Díaz, M.L.; Moreno, G. Microhabitat effects on herbaceous nutrient concentrations at the communit [y and species level in Mediterranean open woodlands: The role of species composition. Grass Forage Sci. 2015, 70, 219–228. [Google Scholar] [CrossRef]
- Rolo, V.; Rivest, D.; Lorente, M.; Kattge, J.; Moreno, G. Taxonomic and functional diversity in Mediterranean pastures: Insights on the biodiversity-productivity trade-off. J. Appl. Ecol. 2016, 53, 1575–1584. [Google Scholar] [CrossRef]
- Reyes-López, J.; Ruiz, N.; Fernández-Haeger, J. Community Structure of Ground-Ants: The Role of Single Trees in a Mediterranean Pastureland. Acta Oecologica 2003, 24, 195–202. [Google Scholar] [CrossRef]
- Gaytán, Á.; Bautista, J.L.; Bonal, R.; Moreno, G.; González-Bornay, G. Trees Increase Ant Species Richness and Change Community Composition in Iberian Oak Savannahs. Diversity 2021, 13, 115. [Google Scholar] [CrossRef]
- Martins da Silva, P.; Aguiar, C.A.S.; Niemelä, J.; Sousa, J.P.; Serrano, A.R.M. Cork-Oak Woodlands as Key-Habitats for Biodiversity Conservation in Mediterranean Landscapes: A Case Study Using Rove and Ground Beetles (Coleoptera: Staphylinidae, Carabidae). Biodivers. Conserv. 2009, 18, 605–619. [Google Scholar] [CrossRef]
- Ramírez-Hernández, A.; Micó, E.; de los Ángeles Marcos-García, M.; Brustel, H.; Galante, E. The “dehesa”, a Key Ecosystem in Maintaining the Diversity of Mediterranean SaproxylicIinsects (Coleoptera and Diptera: Syrphidae). Biodivers. Conserv. 2014, 23, 2069–2086. [Google Scholar] [CrossRef]
- García-Tejero, S.; Taboada, A. Microhabitat Heterogeneity Promotes Soil Fertility and Ground-Dwelling Arthropod Diversity in Mediterranean Wood-Pastures. Agric. Ecosyst. Environ. 2016, 233, 192–201. [Google Scholar] [CrossRef]
- Cárdenas, A.M.; Gallardo, P.; Salido, Á.; Márquez, J. Effects of Environmental Traits and Landscape Management on the Biodiversity of Saproxylic Beetles in Mediterranean Oak Forests. Diversity 2020, 12, 451. [Google Scholar] [CrossRef]
- Ricarte, A.; Nencioni, A.; Tubić, N.K.; Grković, A.; Vujić, A.; Marcos-García, M.Á. The Hoverflies of an Oak Dehesa from Spain, with a New Species and Other Insights into the Taxonomy of the Eumerus Tricolor Group (Diptera: Syrphidae). Ann. Zool. 2018, 68, 259–280. [Google Scholar] [CrossRef]
- Santos-Silva, C.; Gonçalves, A.; Louro, R. Canopy Cover Influence on Macrofungal Richness and Sporocarp Production in Montado Ecosystems. Agrofor. Syst. 2011, 82, 149–159. [Google Scholar] [CrossRef]
- Canteiro, C.; Pinto-Cruz, C.; Simões, M.P.; Gazarini, L. Conservation of Mediterranean Oak Woodlands: Understorey Dynamics under Different Shrub Management. Agrofor. Syst. 2011, 82, 161–171. [Google Scholar] [CrossRef]
- Martín, J.; Lopez, P. The Effect of Mediterranean Dehesa Management on Lizard Distribution and Conservation. Biol. Conserv. 2002, 108, 213–219. [Google Scholar] [CrossRef]
- Godinho, S.; Santos, A.P.; Sá-Sousa, P. Montado Management Effects on the Abundance and Conservation of Reptiles in Alentejo, Southern Portugal. Agrofor. Syst. 2011, 82, 197–207. [Google Scholar] [CrossRef]
- Oksuz, D.P.; Aguiar, C.A.S.; Tápia, S.; Llop, E.; Lopes, P.; Serrano, A.R.M.; Leal, A.I.; Branquinho, C.; Correia, O.; Rainho, A.; et al. Increasing Biodiversity in Wood-Pastures by Protecting Small Shrubby Patches. For. Ecol. Manag. 2020, 464, 118041. [Google Scholar] [CrossRef]
- Slancarova, J.; Garcia-Pereira, P.; Fric, Z.F.; Romo, H.; Garcia-Barros, E. Butterflies in Portuguese ‘Montados’: Relationships between Climate, Land Use and Life-History Traits. J. Insect Conserv. 2015, 19, 823–836. [Google Scholar] [CrossRef]
- Azul, A.M.; Sousa, J.P.; Agerer, R.; Martín, M.P.; Freitas, H. Land Use Practices and Ectomycorrhizal Fungal Communities from Oak Woodlands Dominated by Quercus suber L. Considering Drought Scenarios. Mycorrhiza 2010, 20, 73–88. [Google Scholar] [CrossRef]
- Azul, A.M.; Mendes, S.M.; Sousa, J.P.; Freitas, H. Fungal Fruitbodies and Soil Macrofauna as Indicators of Land Use Practices on Soil Biodiversity in Montado. Agrofor. Syst. 2011, 82, 121–138. [Google Scholar] [CrossRef]
- Barrico, L.; Rodríguez-Echeverría, S.; Freitas, H. Diversity of Soil Basidiomycete Communities Associated with Quercus suber L. in Portuguese Montados. Eur. J. Soil Biol. 2010, 46, 280–287. [Google Scholar] [CrossRef]
- Santos-Silva, C.; Louro, R. Assessment of the Diversity of Epigeous Basidiomycota under Different Soil-Management Systems in a Montado Ecosystem: A Case Study Conducted in Alentejo. Agrofor. Syst. 2016, 90, 117–126. [Google Scholar] [CrossRef]
- Mendes, S.M.; Santos, J.; Freitas, H.; Sousa, J.P. Assessing the Impact of Understory Vegetation Cut on Soil Epigeic Macrofauna from a Cork-Oak Montado in South Portugal. Agrofor. Syst. 2011, 82, 139–148. [Google Scholar] [CrossRef]
- Peco, B.; Espigares, T.; Levassor, C. Trends and fluctuations in species abundance and richness in Mediterranean annual pastures. Appl. Veg. Sci. 1998, 1, 21–28. [Google Scholar] [CrossRef]
- Peco, B.; Sánchez, A.M.; Azcárate, F.M. Abandonment in Grazing Systems: Consequences for Vegetation and Soil. Agric. Ecosyst. Environ. 2006, 113, 284–294. [Google Scholar] [CrossRef]
- Carmona, C.P.; Azcárate, F.M.; de Bello, F.; Ollero, H.S.; Lepš, J.; Peco, B. Taxonomical and Functional Diversity Turnover in Mediterranean Grasslands: Interactions between Grazing, Habitat Type and Rainfall. J. Appl. Ecol. 2012, 49, 1084–1093. [Google Scholar] [CrossRef]
- del Barrio, J.M.G.; Ponce, R.A.; Benavides, R.; Roig, S. Species Richness and Similarity of Vascular Plants in the Spanish Dehesas at Two Spatial Scales. For. Syst. 2014, 23, 111–119. [Google Scholar] [CrossRef]
- Rico Rodríguez, M.; García Criado, L.; García Criado, B.; García Ciudad, A. Efecto de Fertilizantes Fosfatados Sobre La Composición Florística de Pastizales Seminaturales En Suelos Ácidos. Pastos 1985, 15, 139–158. [Google Scholar]
- López-Carrasco, C.; Gómez, M.; Carpintero, J.; Brañas, J.; Roig, S. Efectos de la aplicación de fertilizantes de nueva generación en la dehesa toledana: Producción y diversidad de pastos herbáceos. In Nuevos Retos de la Ganadería Extensiva: Un Agente de Conservación en Peligro de Extinción; Canals, R.M., San Emeterio, L., Eds.; Sociedad Española para el Estudio de Pastos: Pamplona, Spain, 2012; pp. 29–35. [Google Scholar]
- López-Carrasco, C.; López-Sánchez, A.; Gómez, M.; Carpintero, J.; Brañas, J.; Roig, S. Efectos de la aplicación de fertilizantes de nueva generación en la dehesa toledana: Producción, diversidad y composición florística de pastos herbáceos. In Los Pastos: Nuevos Retos, Nuevas Oportunidades; Olea, L., Poblaciones, M.J., Rodrigo, S.M., Santamaría, O., Eds.; Sociedad Española para el Estudio de Pastos: Pamplona, Spain, 2013; pp. 545–552. [Google Scholar]
- López-Carrasco, C.; Gómez, M.J.; Carpintero, J.M.; Brañas, J.; Roig, S. Efecto residual de la aplicación de fertilizantes de nueva generación sobre la producción, composición y diversidad de pastos herbáceos en una dehesa toledana. In Proceedings of the 53th Reunión Científica de la SEEP, Potes, Spain, 9–12 June 2014; pp. 215–222. [Google Scholar]
- Tárrega, R.; Calvo, L.; Taboada, Á.; García-Tejero, S.; Marcos, E. Abandonment and Management in Spanish Dehesa Systems: Effects on Soil Features and Plant Species Richness and Composition. For. Ecol. Manag. 2009, 257, 731–738. [Google Scholar] [CrossRef]
- Castro, H.; Lehsten, V.; Lavorel, S.; Freitas, H. Functional Response Traits in Relation to Land Use Change in the Montado. Agric. Ecosyst. Environ. 2010, 137, 183–191. [Google Scholar] [CrossRef]
- Rueda, M.; Rebollo, S.; García-Salgado, G. Contrasting Impacts of Different-Sized Herbivores on Species Richness of Mediterranean Annual Pastures Differing in Primary Productivity. Oecologia 2013, 172, 449–459. [Google Scholar] [CrossRef]
- Köbel, M.; Listopad, C.M.C.S.; Príncipe, A.; Nunes, A.; Branquinho, C. Temporary Grazing Exclusion as a Passive Restoration Strategy in a Dryland Woodland: Effects over Time on Tree Regeneration and on the Shrub Community. For. Ecol. Manag. 2021, 483, 118732. [Google Scholar] [CrossRef]
- Aragón, G.; López, R.; Martínez, I. Effects of Mediterranean Dehesa Management on Epiphytic Lichens. Sci. Total Environ. 2010, 409, 116–122. [Google Scholar] [CrossRef]
- Azcárate, F.M.; Peco, B. Abandonment of Grazing in a Mediterranean Grassland Area: Consequences for Ant Assemblages. Insect Conserv. Divers. 2012, 5, 279–288. [Google Scholar] [CrossRef]
- García-Tejero, S.; Taboada, Á.; Tárrega, R.; Salgado, J.M. Land Use Changes and Ground Dwelling Beetle Conservation in Extensive Grazing Dehesa Systems of North-West Spain. Biol. Conserv. 2013, 161, 58–66. [Google Scholar] [CrossRef]
- Barriga, J.C.; Lassaletta, L.; Moreno, A.G. Ground-Living Spider Assemblages from Mediterranean Habitats under Different Management Conditions. J. Arachnol. 2010, 38, 258–269. [Google Scholar] [CrossRef]
- Arosa, M.L.; Freitas, H.; Costa, S.R. Temporal Effects Dominate Land Use as Factors Affecting Soil Nematode Communities in Mediterranean Oak Woodlands. Agrofor. Syst. 2016, 90, 127–136. [Google Scholar] [CrossRef]
- Pulido, F.J.; Díaz, M. Relaciones entre la estructura de la vegetación y las comunidades de aves nidificantes en las dehesas: Influencia del manejo humano. Ardeola 1992, 39, 63–72. [Google Scholar]
- Godinho, C.; Rabaça, J.E. Birds like It Corky: The Influence of Habitat Features and Management of “montados” in Breeding Bird Communities. Agrofor. Syst. 2011, 82, 183–195. [Google Scholar] [CrossRef]
- Catarino, L.; Godinho, C.; Pereira, P.; Luís, A.; Rabaça, J.E. Can Birds Play a Role as High Nature Value Indicators of Montado System? Agrofor. Syst. 2016, 90, 45–56. [Google Scholar] [CrossRef]
- Oksuz, D.P.; Palmeirim, J.M.; Correia, R.A. Bird Taxonomic and Functional Responses to Land Abandonment in Wood-Pastures. Agrofor. Syst. 2021, 95, 1167–1176. [Google Scholar] [CrossRef]
- Díaz, M.; González, E.; Muñoz-Pulido, R.; Naveso, M.A. Abundance, seed predation rates, and body condition of rodents wintering in Spanish Holm-oak Quercus ilex L. dehesas and cereal croplands: Effects of food abundance and habitat structure. Mamm. Biol. 1993, 58, 302–311. [Google Scholar]
- Jiménez-Carmona, F.; Carpintero, S.; Reyes-López, J.L. Ants (Hymenoptera: Formicidae) as Surrogates for Epigeic Arthropods in Northern Andalusian “dehesas” (Spain). Sociobiology 2020, 67, 201–212. [Google Scholar] [CrossRef]
- Martins da Silva, P.; Aguiar, C.A.S.; de e Silva, I.F.; Serrano, A.R.M. Orchard and Riparian Habitats Enhance Ground Dwelling Beetle Diversity in Mediterranean Agro-Forestry Systems. Biodivers. Conserv. 2011, 20, 861–872. [Google Scholar] [CrossRef]
- Tellería, J.L. Passerine Bird Communities of Iberian Dehesas: A Review. Anim. Biodivers. Conserv. 2001, 24, 67–78. [Google Scholar]
- Díaz, M.; Pulido, F.J.; Marañón, T. Diversidad Biológica y Sostenibilidad Ecológica y Económica de Los Sistemas Adehesados. Ecosistemas 2003, 12, 1–10. [Google Scholar] [CrossRef]
- Rosalino, L.M.; do Rosário, J.; Santos-Reis, M. The Role of Habitat Patches on Mammalian Diversity in Cork Oak Agroforestry Systems. Acta Oecol. 2009, 35, 507–512. [Google Scholar] [CrossRef]
- Gonçalves, P.; Alcobia, S.; Simões, L.; Santos-Reis, M. Effects of Management Options on Mammal Richness in a Mediterranean Agro-Silvo-Pastoral System. Agrofor. Syst. 2012, 85, 383–395. [Google Scholar] [CrossRef]
- Curveira-Santos, G.; Marques, T.A.; Björklund, M.; Santos-Reis, M. Mediterranean Mesocarnivores in Spatially Structured Managed Landscapes: Community Organisation in Time and Space. Agric. Ecosyst. Environ. 2017, 237, 280–289. [Google Scholar] [CrossRef]
- Costa, P.; Medinas, D.; Silva, B.M.; Mira, A.; Guiomar, N.; Sales-Baptista, E.; Ferraz-de-Oliveira, M.I.; Simões, M.P.; Belo, A.D.F.; Herrera, J.M. Cattle-Driven Forest Disturbances Impact Ensemble Composition and Activity Levels of Insectivorous Bats in Mediterranean Wood Pastures. Agrofor. Syst. 2019, 93, 1687–1699. [Google Scholar] [CrossRef]
- Hernández-Esteban, A.; Rolo, V.; López-Díaz, M.L.; Moreno, G. Long-Term Implications of Sowing Legume-Rich Mixtures for Plant Diversity of Mediterranean Wood Pastures. Agric. Ecosyst. Environ. 2019, 286, 106686. [Google Scholar] [CrossRef]
- Leal, A.I.; Correia, R.A.; Granadeiro, J.P.; Palmeirim, J.M. Impact of Cork Extraction on Birds: Relevance for Conservation of Mediterranean Biodiversity. Biol. Conserv. 2011, 144, 1655–1662. [Google Scholar] [CrossRef]
- Leal, A.I.; Correia, R.A.; Palmeirim, J.M.; Granadeiro, J.P. Does Canopy Pruning Affect Foliage-Gleaning Birds in Managed Cork Oak Woodlands? Agrofor. Syst. 2013, 87, 355–363. [Google Scholar] [CrossRef]
- López-Sánchez, A.; Perea, R.; Roig, S. Are Large-Nested Bird Colonies Compatible with the Conservation of Grazed Woodlands? Glob. Ecol. Conserv. 2019, 20, e00705. [Google Scholar] [CrossRef]
- Margalef, R. Ecología; Omega: Barcelona, Spain, 1974. [Google Scholar]
- Grime, J.P. Plant Strategies and Vegetation Processes; Wiley: Chichester, UK, 1979. [Google Scholar]
- San Miguel, A.; Roig, S.; Perea, R. The pastures of Spain. Pastos 2016, 46, 6–39. [Google Scholar]
- Olea, L.; San Miguel, A. The Spanish dehesa. A traditional Mediterranean silvopastoral system linking production and nature conservation. Grassl. Sci. Eur. 2006, 11, 3–13. [Google Scholar]
- Murillo, M.; González, F. Gestión de la dehesa en Extremadura. Recursos pascícolas y mejoras. Pastos 2008, 38, 5–7. [Google Scholar]
- Hernando Varela, J. El proyecto de investigación agrícola del Banco Mundial y la modernización de la investigación agraria en España en la década de los setenta. Estud. Agrosoc. Pesq. 2001, 190, 43–63. [Google Scholar]
- Katznelson, J. Aspectos económicos de la aplicación de fertilizantes en pastos de secano naturales y artificiales. Pastos 1975, 5, 84–98. [Google Scholar]
- Olea, L.; Paredes, J.; González, J.D.; Santos, A. Influencia de la sequía en la persistencia de leguminosas anuales y en la fertilidad de los suelos de pastos del SO de España. In Proceedings of the 24th Reunión Científica de la SEEP, Santander, Spain, 30 May–3 June 1994; pp. 103–109. [Google Scholar]
- Perea, R.; López-Sánchez, A.; Roig, S. The Use of Shrub Cover to Preserve Mediterranean Oak Dehesas: A Comparison between Sheep, Cattle and Wild Ungulate Management. Appl. Veg. Sci. 2016, 19, 244–253. [Google Scholar] [CrossRef]
- Moreno, G.; Bartolome, J.W.; Gea-Izquierdo, G.; Cañellas, I. Overstory–understorey relationships. In Mediterranean oak Woodland Working Landscapes; Campos, P., Huntsinger, L., Oviedo, J.L., Starrs, P.F., Diaz, M., Standiford, R.B., Montero, G., Eds.; Springer: Dordrecht, The Netherlands, 2013; pp. 145–179. [Google Scholar]
- Rossetti, I.; Bagella, S.; Cappai, C.; Caria, M.C.; Lai, R.; Roggero, P.P.; da Silva, P.M.; Sousa, J.P.; Querner, P.; Seddaiu, G. Isolated Cork Oak Trees Affect Soil Properties and Biodiversity in a Mediterranean Wooded Grassland. Agric. Ecosyst. Environ. 2015, 202, 203–216. [Google Scholar] [CrossRef]
- García Moreno, A.M.; Fernández Rebollo, P.; Muñoz Espejo, M.L.; Carbonero Muñoz, M.D. Gestión de los Pastos en la Dehesa; Instituto de Investigación y Formación Agraria y Pequera: Sevilla, Spain, 2016; pp. 1–138. [Google Scholar]
- Pereira, M.; Rodríguez, A. Conservation Value of Linear Woody Remnants for Two Forest Carnivores in a Mediterranean Agricultural Landscape. J. Appl. Ecol. 2010, 47, 611–620. [Google Scholar] [CrossRef]
- Ribeiro, S.; Fernandes, J.P.; Espírito-Santo, M.D. Diversity and Floristic Patterns of Mediterranean Grasslands: The Relative Influence of Environmental and Land Management Factors. Biodivers. Conserv. 2014, 23, 2903–2921. [Google Scholar] [CrossRef]
- Faria, N. Predicting Agronomical and Ecological Effects of Shifting from Sheep to Cattle Grazing in Highly Dynamic Mediterranean Dry Grasslands. Land Degrad. Dev. 2019, 30, 300–314. [Google Scholar] [CrossRef]
- Faria, N.; Morales, M.B. Farmland Management Regulates Ecosystem Services in Mediterranean Drylands: Assessing the Sustainability of Agri-Environmental Payments for Bird Conservation. J. Nat. Conserv. 2020, 58, 125913. [Google Scholar] [CrossRef]
- Ramos, R.F.; Diogo, J.A.; Santana, J.; Silva, J.P.; Reino, L.; Schindler, S.; Beja, P.; Lomba, A.; Moreira, F. Impacts of Sheep versus Cattle Livestock Systems on Birds of Mediterranean Grasslands. Sci. Rep. 2021, 11, 10827. [Google Scholar] [CrossRef] [PubMed]
- Peco, B.; Navarro, E.; Carmona, C.P.; Medina, N.G.; Marques, M.J. Effects of Grazing Abandonment on Soil Multifunctionality: The Role of Plant Functional Traits. Agric. Ecosyst. Environ. 2017, 249, 215–225. [Google Scholar] [CrossRef]
- Moreno, G.; Hernández-Esteban, A.; Rolo, V.; Igual, J.M. The enduring effects of sowing legume-rich mixtures on the soil microbial community and soil carbon in semi-arid wood pastures. Plant Soil 2021, 465, 563–582. [Google Scholar] [CrossRef]
- Hevia, V.; Carmona, C.P.; Azcárate, F.M.; Torralba, M.; Alcorlo, P.; Ariño, R.; Lozano, J.; Castro-Cobo, S.; González, J.A. Effects of Land Use on Taxonomic and Functional Diversity: A Cross-Taxon Analysis in a Mediterranean Landscape. Oecologia 2016, 181, 959–970. [Google Scholar] [CrossRef] [PubMed]
- Rocha, B.; Pinho, P.; Branquinho, C.; Boieiro, M.; Matos, P. Bringing the Concept of Ammonia Critical Levels into Managing Cork-Oak Woodland for Conservation. For. Ecol. Manag. 2019, 453, 117566. [Google Scholar] [CrossRef]
- Oksuz, D.P.; Aguiar, C.A.S.; Tápia, S.; Llop, E.; Lopes, P.; Serrano, A.R.M.; Leal, A.I.; Correia, O.; Matos, P.; Rainho, A.; et al. The Contribution of Small Shrubby Patches to the Functional Diversity of Wood-Pastures. Acta Oecol. 2020, 108, 103626. [Google Scholar] [CrossRef]
- Díaz, M.; Tietje, W.D.; Barret, R.H. Effects of Management on Biological Diversity and Endangered Species. In Mediterranean oak Woodland Working Landscapes; Campos, P., Huntsinger, L., Oviedo, J.L., Starrs, P.F., Diaz, M., Standiford, R.B., Montero, G., Eds.; Springer: Dordrecht, The Netherlands, 2013; pp. 213–243. [Google Scholar]
- Mupepele, A.-C.; Bruelheide, H.; Brühl, C.; Dauber, J.; Fenske, M.; Freibauer, A.; Gerowitt, B.; Kruß, A.; Lankner, S.; Plieninger, T.; et al. Biodiversity in agricultural landscapes: Transformative societal changes needed. Trends Ecol. Evol. 2021, 36, 1067–1070. [Google Scholar] [CrossRef]

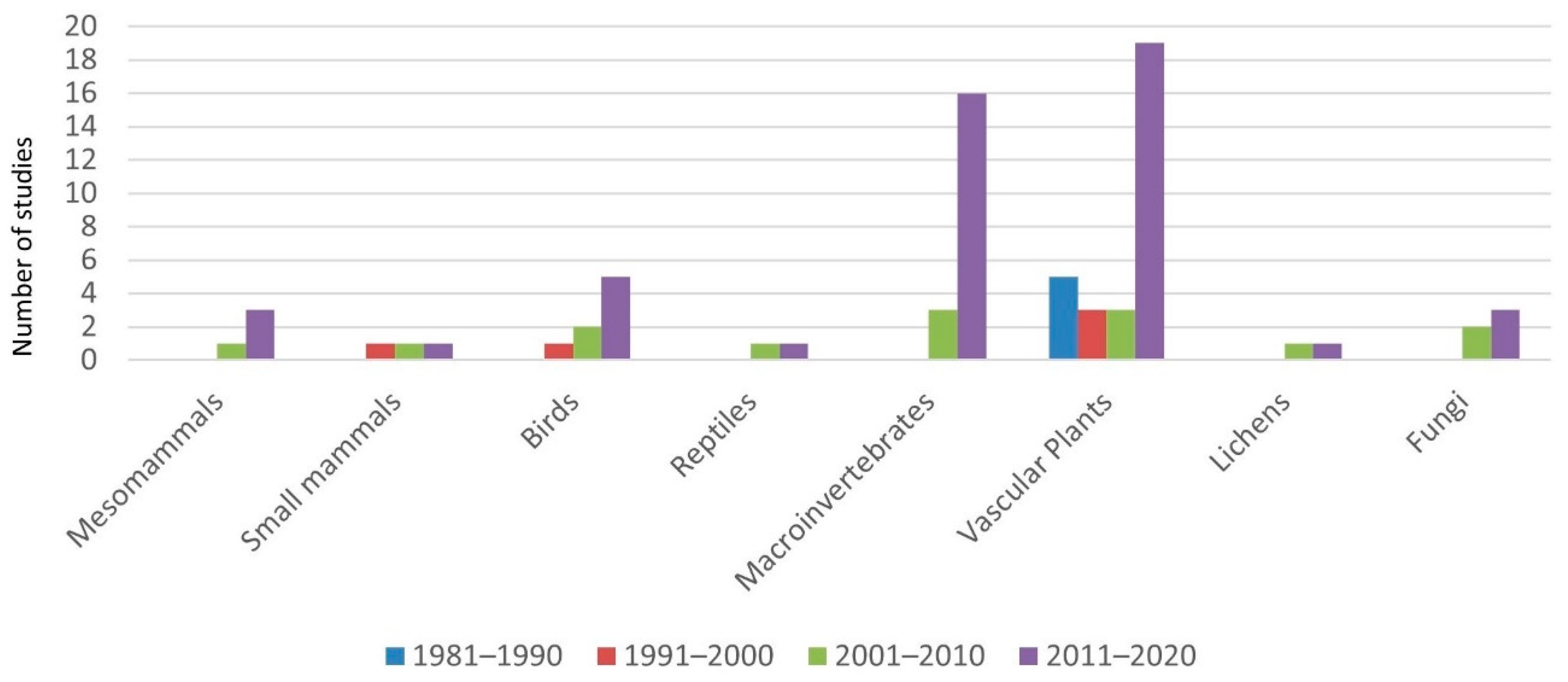
| Land Factors | Publications |
|---|---|
| Tree factor (tree presence and density) | Marañón (1985, 1986) [24,35]; Rico and Puerto (1988) [25]; Díez et al., (1992) [36]; Fernández-Moya et al., (2011) [37]; López-Carrasco et al., (2015) [38]; López-Sánchez et al., (2016a, 2016b) [39,40]; Rolo et al., (2015) [41]; Rolo et al., (2016) [42]-vascular plants- Reyes-López et al., (2003) [43]; Gaytán et al., (2021) [44]-ants- Martins da Silva et al., (2009) [45]; Ramirez-Hernández et al., (2014) [46]; García-Tejero and Taboada (2016) [47]; Cárdenas et al., (2020) [48]-coleoptera- Ricarte et al., (2018) [49]; Ramirez-Hernández et al., (2014) [46]-syrphids- Santos-Silva et al., (2011) [50]-macrofungi- |
| Shrub (shrub cover, shrub clearing practices) | Canteiro et al., (2011) [51]-vascular plants- Martín and Lopez (2002) [52]; Godinho et al., (2011) [53]-reptiles- García-Tejero and Taboada (2016) [47]; Cárdenas et al., (2020) [48] -coleoptera- Ramirez-Hernández et al., (2014) [46]-coleoptera, syrphids- Oksuz et al., (2020) [54] -vascular plants, lichens, coleoptera- Slancarova et al., (2015) [55]-butterflies- Simonson et al., (2018) [30]-vascular plants, butterflies, birds- Azul et al., (2010, 2011) [56,57]; Barrico et al., (2010) [58]; Santos-Silva and Louro (2016) [59]-macrofungi- Mendes et al., (2011) [60]-soil macroinvertebrates- |
| Topography | Pineda et al., (1981) [23]; Puerto et al., (1990) [26]; Peco et al., (1998) [61]; Peco et al., (2006) [62]; Carmona et al., (2012) [63]; Garcia del Barrio et al., (2014) [64]; López-Carrasco et al., (2015) [38]-vascular plants- |
| Phosphorus fertilization | Rico et al., (1985) [65]; López-Carrasco et al., (2012, 2013, 2014) [66,67,68]-vascular plants- |
| Grazing management and land use | Peco et al., (2006) [62]; Tárrega et al., (2009) [69]; Castro et al., (2010) [70]; Rueda et al., (2013) [71]; Carmona et al., (2012) [63]; López-Sanchez et al., (2016a, 2016b) [39,40]; Köbel et al., (2021) [72]-vascular plants- Aragón et al., (2010) [73]-epiphytic lichens- Azcárate and Peco (2012) [74]-ants- García-Tejero et al., (2013) [75]-coleoptera- Martins da Silva et al., (2009) [45]; Barriga et al., (2010) [76]-spiders- Arosa et al., (2016) [77]-soil nematodes- Bugalho et al., (2011) [28]-vascular plants, soil macroinvertebrates- Pulido and Díaz (1992) [78]; Godinho and Rabaça (2011) [79]; Catarino et al. (2016) [80]; Oksuz et al., (2021) [81]-birds- Díaz et al., (1993) [82]–small mammals- |
| Habitat heterogeneity and microhabitat variability | Marañón (1985) [24]-vascular plants Moreno et al., (2016) [29]-vascular plants, bees, spiders, nematodes- Simonson et al., (2018) [30]-vascular plants, butterflies, birds- Jiménez-Carmona et al., (2020) [83]-ants- Godinho et al., (2011) [53]-reptiles- Martins da Silva et al., (2011) [84]; García-Tejero and Taboada (2016) [47]-coleoptera- Pulido and Díaz (1992) [78]; Tellería (2001) [85]; Díaz et al., (2003) [86]; Godinho and Rabaça (2011) [79]; Catarino et al., (2016) [80]-birds- Rosalino et al., (2009) [87]; Gonçalves et al., (2012) [88]; Curveira-Santos et al., (2017) [89] -small mammals, mesomammals- Costa et al., (2019) [90] -bats- |
| Others | Legume sowing: Hernández-Esteban (2019) [91]-vascular plants- Cork removal: Godinho and Rabaça (2011) [79]; Leal et al., (2011) [92]-birds- Canopy pruning: Leal et al., (2013) [93]-birds- Large-nested bird colonies: López-Sánchez et al., (2019) [94]-vascular plants- |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Rojo, M.P.; Roig, S.; López-Carrasco, C.; Redondo García, M.M.; Sánchez-Mata, D. Which Factors Favour Biodiversity in Iberian Dehesas? Sustainability 2022, 14, 2345. https://doi.org/10.3390/su14042345
Rodríguez-Rojo MP, Roig S, López-Carrasco C, Redondo García MM, Sánchez-Mata D. Which Factors Favour Biodiversity in Iberian Dehesas? Sustainability. 2022; 14(4):2345. https://doi.org/10.3390/su14042345
Chicago/Turabian StyleRodríguez-Rojo, Maria Pilar, Sonia Roig, Celia López-Carrasco, María Manuela Redondo García, and Daniel Sánchez-Mata. 2022. "Which Factors Favour Biodiversity in Iberian Dehesas?" Sustainability 14, no. 4: 2345. https://doi.org/10.3390/su14042345
APA StyleRodríguez-Rojo, M. P., Roig, S., López-Carrasco, C., Redondo García, M. M., & Sánchez-Mata, D. (2022). Which Factors Favour Biodiversity in Iberian Dehesas? Sustainability, 14(4), 2345. https://doi.org/10.3390/su14042345









