Temporal Stability of Soil Microbial Properties in Responses to Long-Term Application of Compost Obtained from Tannery Sludge
Abstract
1. Introduction
2. Materials and Methods
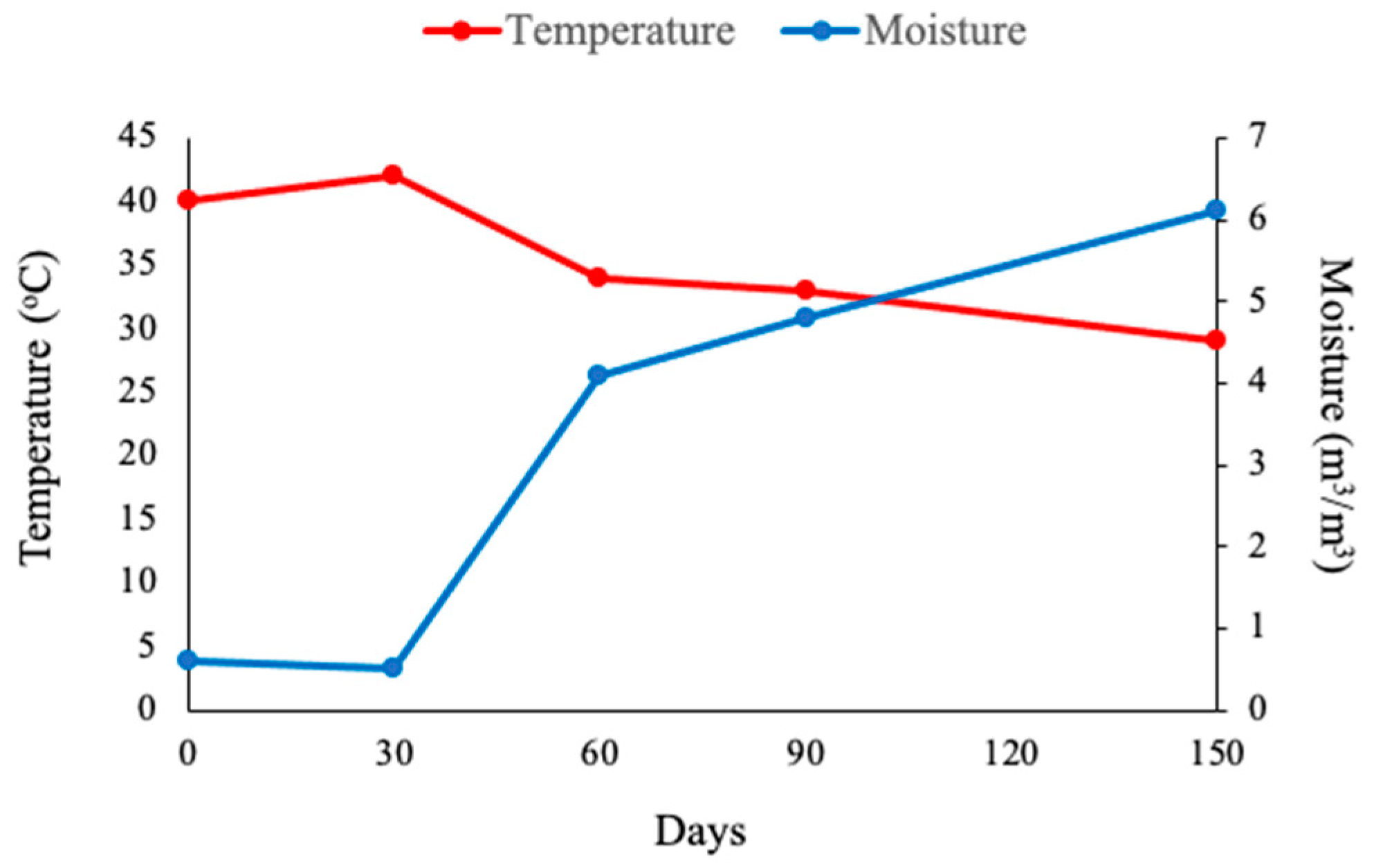
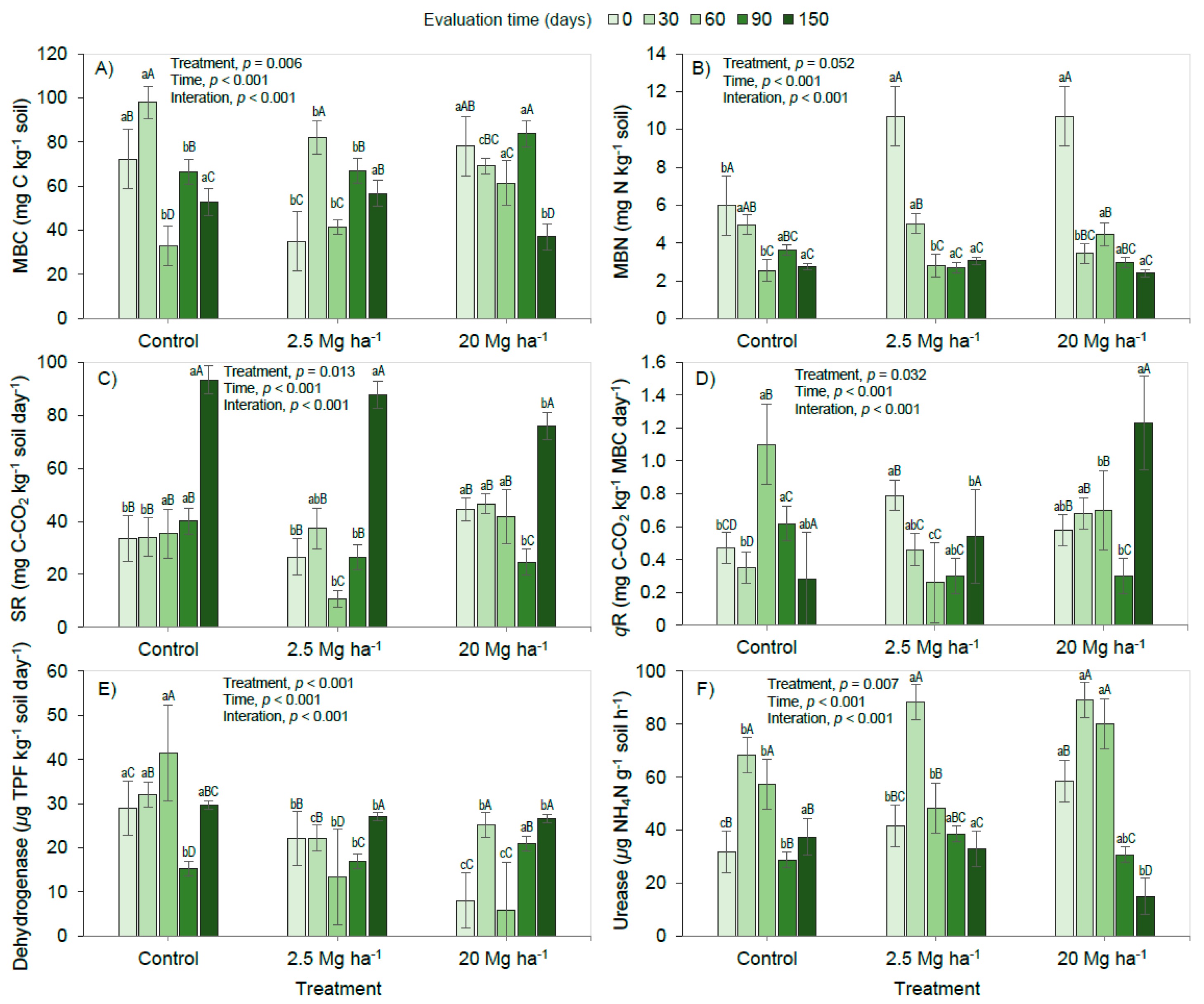
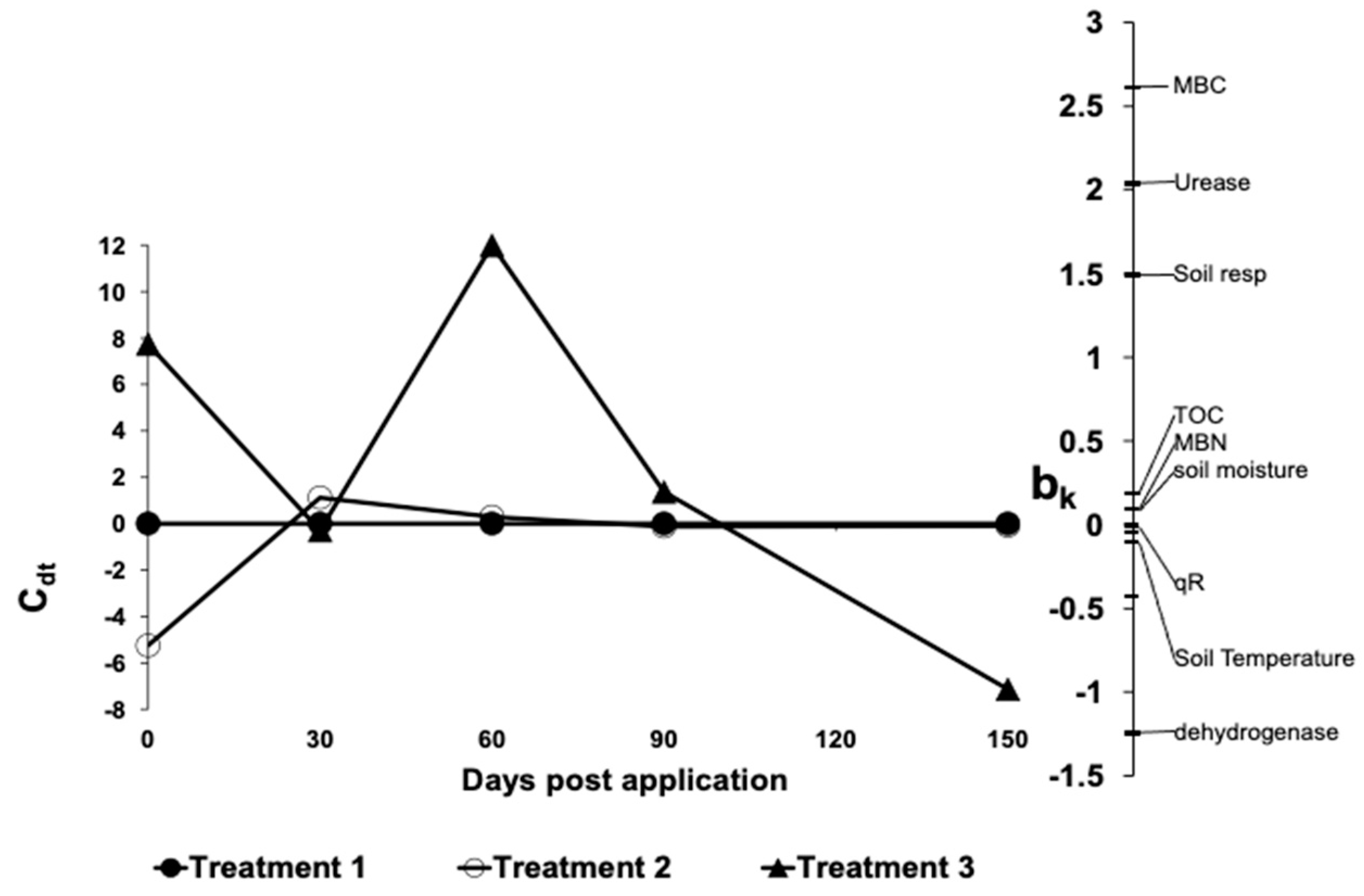
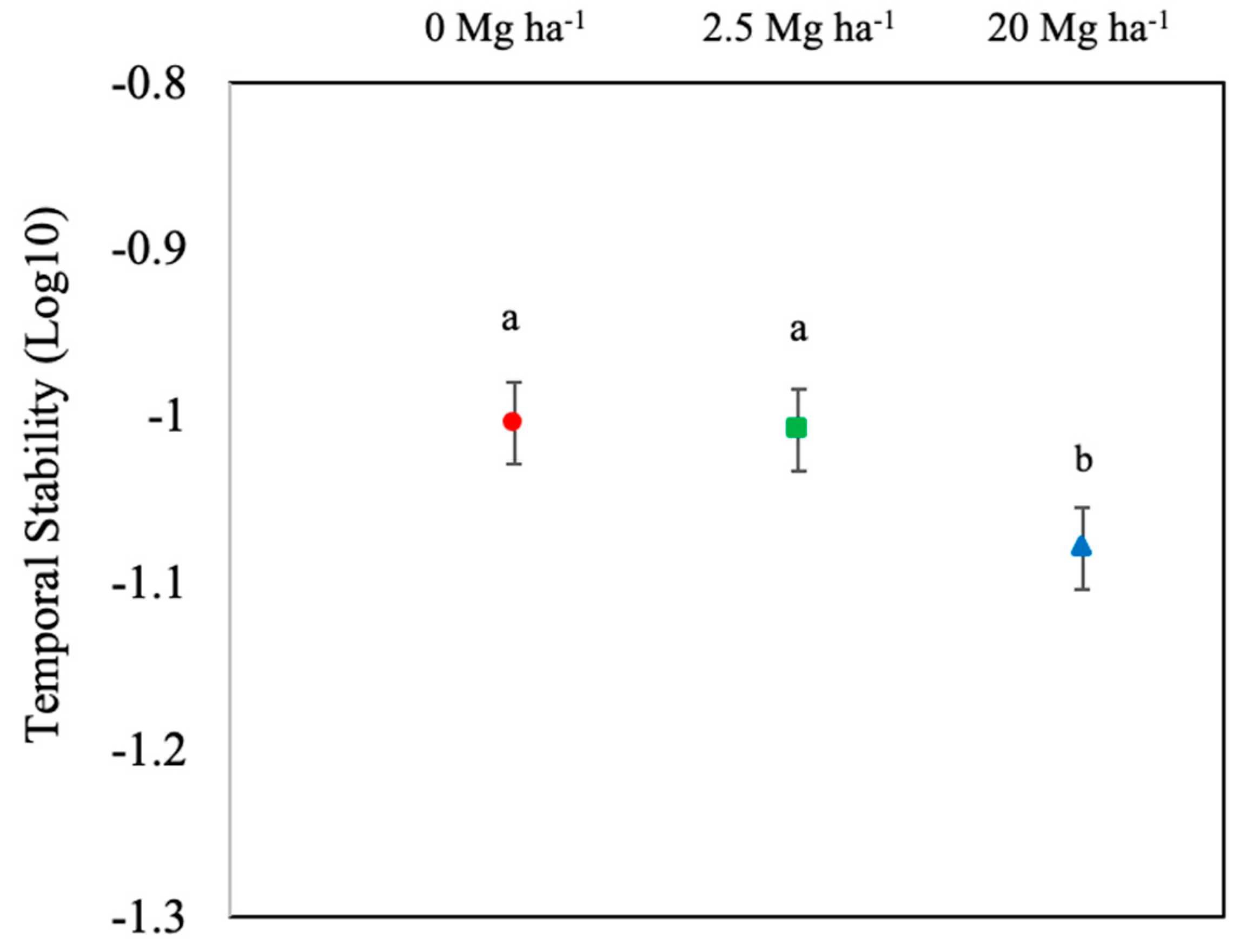
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Araujo, A.S.F.; De Melo, W.J.; Araujo, F.F.; Brink, P.V.D. Long-term effect of composted tannery sludge on soil chemical and biological parameters. Environ. Sci. Pollut. Res. 2020, 27, 41885–41892. [Google Scholar] [CrossRef] [PubMed]
- Nakatani, A.S.; Nogueira, M.A.; Martines, A.M.; Dos Santos, C.A.; Baldesin, L.F.; Marschner, P.; Cardoso, E.J. Effects of tannery sludge application on physiological and fatty acid profiles of the soil microbial community. Appl. Soil Ecol. 2012, 61, 92–99. [Google Scholar] [CrossRef]
- Santos, A.J.M.; Backes, C.; Rodrigues, L.M.; Teodoro, A.G.; De Godoy, L.J.G.; Tomazello, D.A.; Campos, L.F.C.; Ribon, A.A.; Lopes, T.A.; Boas, R.L.V. Chemical characteristics of soil after application of tannery sludge as fertilizer in the sugarcane plant crop. Aust. J. Crop Sci. 2020, 14, 641–648. [Google Scholar] [CrossRef]
- Blanco-Canqui, H.; Shapiro, C.A.; Wortmann, C.S.; Drijber, R.A.; Mamo, M.; Shaver, T.M.; Ferguson, R.B. Soil organic carbon: The value to soil properties. J. Soil Water Conserv. 2013, 68, 129A–134A. [Google Scholar] [CrossRef]
- Gentili, R.; Ambrosini, R.; Montagnani, C.; Caronni, S.; Citterio, S. Effect of Soil pH on the Growth, Reproductive Investment and Pollen Allergenicity of Ambrosia artemisiifolia L. Front. Plant Sci. 2018, 9, 1335. [Google Scholar] [CrossRef]
- Kandziora-Ciupa, M.; Nadgórska-Socha, A.; Barczyk, G. The influence of heavy metals on biological soil quality assessments in the Vaccinium myrtillus L. rhizosphere under different field conditions. Ecotoxicology 2021, 30, 292–310. [Google Scholar] [CrossRef]
- Strecker, T.; Macé, O.G.; Scheu, S.; Eisenhauer, N. Functional composition of plant communities determines the spatial and temporal stability of soil microbial properties in a long-term plant diversity experiment. Oikos 2016, 125, 1743–1754. [Google Scholar] [CrossRef]
- Proulx, R.; Wirth, C.; Voigt, W.; Weigelt, A.; Roscher, C.; Attinger, S.; Baade, J.; Barnard, R.; Buchmann, N.; Buscot, F.; et al. Diversity Promotes Temporal Stability across Levels of Ecosystem Organization in Experimental Grasslands. PLoS ONE 2010, 5, e13382. [Google Scholar] [CrossRef]
- Atlas, R.M.; Horowitz, A.; Krichevsky, M.; Bej, A.K. Response of microbial populations to environmental disturbance. Microb. Ecol. 1991, 22, 249–256. [Google Scholar] [CrossRef]
- Kostin, J.E.; Cesarz, S.; Lochner, A.; Schädler, M.; Macdonald, C.A.; Eisenhauer, N. Land-use drives the temporal stability and magnitude of soil microbial functions and modulates climate effects. Ecol. Appl. 2021, 31, e2325. [Google Scholar] [CrossRef]
- Araújo, A.S.F.; Cesarz, S.; Leite, L.F.C.; Borges, C.D.; Tsai, S.M.; Eisenhauer, N. Soil microbial properties and temporal stability in degraded and restored lands of Northeast Brazil. Soil Biol. Biochem. 2013, 66, 175–181. [Google Scholar] [CrossRef]
- Yeomans, J.C.; Bremner, J.M. A rapid and precise method for routine determination of organic carbon in soil. Commun. Soil Sci. Plant Anal. 1988, 19, 1467–1476. [Google Scholar] [CrossRef]
- USEPA. Test Method for Evaluating Solid Waste; USEPA: Washington, DC, USA, 1986.
- Brookes, P.C.; Joergensen, R.G. Microbial biomass measurement by fumigation-extraction. In Microbiological Methods for Assessing Soil Quality; Bloem, J., Hopkins, W., Benedetti, A., Eds.; CABI: Wallingford, UK, 2006; pp. 77–83. [Google Scholar]
- Alef, K.; Nannipieri, P. Methods in Soil Microbiology and Biochemistry; Academic Press: New York, NY, USA, 1995. [Google Scholar]
- Casida, L.E., Jr.; Klein, D.A.; Santoro, T. Soil dehydrogenase activity. Soil Sci. 1964, 98, 371–376. [Google Scholar] [CrossRef]
- Kandeler, E.; Gerber, H. Short-term assay of soil urease activity using colorimetric determination of ammonium. Biol. Fertil. Soils 1988, 6, 68–72. [Google Scholar] [CrossRef]
- Van den Brink, P.J.; Ter Braak, C.J.F. Principal response curves: Analysis of time-dependent multivariate responses of biological community to stress. Environ. Toxicol. Chem. 1999, 18, 138–148. [Google Scholar] [CrossRef]
- Brink, P.J.V.D.; Ter Braak, C.J. Multivariate analysis of stress in experimental ecosystems by Principal Response Curves and similarity analysis. Aquat. Ecol. 1998, 32, 163–178. [Google Scholar] [CrossRef]
- Ter Braak, C.J.F.; Šmilauer, P. Canoco Reference Manual and User’s Guide: Software for Ordination, version 5.1x; Microcomputer Power: Ithaca, NY, USA, 2018; p. 536. [Google Scholar]
- Eisenhauer, N.; Milcu, A.; Allan, E.; Nitschke, N.; Scherber, C.; Temperton, V.; Weigelt, A.; Weisser, W.W.; Scheu, S. Impact of above- and below-ground invertebrates on temporal and spatial stability of grassland of different diversity. J. Ecol. 2011, 99, 572–582. [Google Scholar] [CrossRef]
- Araujo, A.S.F.; Pereira, A.P.D.A.; Mendes, L.W. Applications of Cr-rich composted tannery sludge in the soil decrease microbial biomass and select specific bacterial groups. Environ. Sci. Pollut. Res. 2022, 29, 75113–75118. [Google Scholar] [CrossRef]
- Lima, J.R.D.S.; Souza, R.M.S.; Dos Santos, E.S.; De Souza, E.S.; Oliveira, J.E.D.S.; De Medeiros, E.V.; Pessoa, L.G.M.; Antonino, A.C.D.; Hammecker, C. Impacts of land-use changes on soil respiration in the semi-arid region of Brazil. Rev. Bras. Ciênc. Solo 2020, 44, e0200092. [Google Scholar] [CrossRef]
- Lepcha, N.T.; Devi, N.B. Effect of land use, season, and soil depth on soil microbial biomass carbon of Eastern Himalayas. Ecol. Process. 2020, 9, 65. [Google Scholar] [CrossRef]
- Malik, A.A.; Puissant, J.; Buckeridge, K.; Goodall, T.; Jehmlich, N.; Chowdhury, S.; Gweon, H.S.; Peyton, J.M.; Mason, K.E.; Van Agtmaal, M.; et al. Land use driven change in soil pH affects microbial carbon cycling processes. Nat. Commun. 2018, 9, 3591. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, X.; Li, L.; Fu, G.; Liu, X.; Xing, S.; Feng, H.; Chen, B. The toxicity of hexavalent chromium to soil microbial processes concerning soil properties and aging time. Environ. Res. 2021, 204, 111941. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Hu, Y.; Zhang, S.; Noll, L.; Böckle, T.; Richter, A.; Wanek, W. Growth explains microbial carbon use efficiency across soils differing in land use and geology. Soil Biol. Biochem. 2018, 128, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Batool, M.; Rahman, S.U.; Ali, M.; Nadeem, F.; Ashraf, M.N.; Harris, M.; Du, Z.; Khan, W.-U. Microbial-assisted soil chromium immobilization through zinc and iron-enriched rice husk biochar. Front. Microbiol. 2022, 13, 3401. [Google Scholar] [CrossRef]
- Chibuzor, N.E.; Chuks, K.O.; Emmanuel, A.E.; Paul, I.O.; Simeon, C.E.; Uchenna, J.O. Chromium (III) and its effects on soil microbial activities and phytoremediation potentials of Arachis hypogea and Vigna unguiculata. Afr. J. Biotechnol. 2018, 17, 1207–1214. [Google Scholar] [CrossRef]
- Lellei-Kovács, E.; Kovács-Láng, E.; Botta-Dukát, Z.; Kalapos, T.; Emmett, B.; Beier, C. Thresholds and interactive effects of soil moisture on the temperature response of soil respiration. Eur. J. Soil Biol. 2011, 47, 247–255. [Google Scholar] [CrossRef]
- Geyer, K.M.; Kyker-Snowman, E.; Grandy, A.S.; Frey, S.D. Microbial carbon use efficiency: Accounting for population, community, and ecosystem-scale controls over the fate of metabolized organic matter. Biogeochemistry 2016, 127, 173–188. [Google Scholar] [CrossRef]
- Saifuddin, M.; Bhatnagar, J.M.; Segrè, D.; Finzi, A.C. Microbial carbon use efficiency predicted from genome-scale metabolic models. Nat. Commun. 2019, 10, 3568. [Google Scholar] [CrossRef]
- Qu, J.; Ren, G.; Chen, B.; Fan, J.; E, Y. Effects of lead and zinc mining contamination on bacterial community diversity and enzyme activities of vicinal cropland. Environ. Monit. Assess. 2011, 182, 597–606. [Google Scholar] [CrossRef]
- Madejón, E.; Burgos, P.; López, R.; Cabrera, F. Soil enzymatic response to addition of heavy metals with organic residues. Biol. Fertil. Soils 2001, 34, 144–150. [Google Scholar] [CrossRef]
- Stêpniewska, Z.; Woliñska, A. Soil dehydrogenase activity in the presence of chromium (III) and (VI). Int. Agrophysics 2005, 19, 79–83. [Google Scholar]
- Łukowski, A.; Dec, D. Fractions of Ni, Pb, Cr, and Their Impact on Enzyme Activities of Arable Land Cultivated by the Simplified Method. Minerals 2021, 11, 584. [Google Scholar] [CrossRef]
| Treatment | Soil pH (H2O) | Organic C (g kg−1) | Cr Content (mg kg−1) |
|---|---|---|---|
| 0 Mg ha−1 | 6.3 | 8.7 | 2.5 |
| 2.5 Mg ha−1 | 7.8 | 21.5 | 24.9 |
| 20 Mg ha−1 | 8.9 | 41.8 | 158.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saraiva, T.C.d.S.; Ventura, S.H.; Brito Junior, E.S.; Rocha, S.M.B.; Costa, R.M.; Pereira, A.P.d.A.; De Sousa, R.S.; Verma, J.P.; Van den Brink, P.J.; Araujo, A.S.F. Temporal Stability of Soil Microbial Properties in Responses to Long-Term Application of Compost Obtained from Tannery Sludge. Sustainability 2022, 14, 16736. https://doi.org/10.3390/su142416736
Saraiva TCdS, Ventura SH, Brito Junior ES, Rocha SMB, Costa RM, Pereira APdA, De Sousa RS, Verma JP, Van den Brink PJ, Araujo ASF. Temporal Stability of Soil Microbial Properties in Responses to Long-Term Application of Compost Obtained from Tannery Sludge. Sustainability. 2022; 14(24):16736. https://doi.org/10.3390/su142416736
Chicago/Turabian StyleSaraiva, Talyta Carine da Silva, Sabrina Hermelindo Ventura, Eudemio Sousa Brito Junior, Sandra Mara Barbosa Rocha, Romario Martins Costa, Arthur Prudencio de Araujo Pereira, Ricardo Silva De Sousa, Jay Prakash Verma, Paul J. Van den Brink, and Ademir Sergio Ferreira Araujo. 2022. "Temporal Stability of Soil Microbial Properties in Responses to Long-Term Application of Compost Obtained from Tannery Sludge" Sustainability 14, no. 24: 16736. https://doi.org/10.3390/su142416736
APA StyleSaraiva, T. C. d. S., Ventura, S. H., Brito Junior, E. S., Rocha, S. M. B., Costa, R. M., Pereira, A. P. d. A., De Sousa, R. S., Verma, J. P., Van den Brink, P. J., & Araujo, A. S. F. (2022). Temporal Stability of Soil Microbial Properties in Responses to Long-Term Application of Compost Obtained from Tannery Sludge. Sustainability, 14(24), 16736. https://doi.org/10.3390/su142416736






