Sea Minerals Reduce Dysbiosis, Improve Pasture Productivity and Plant Morphometrics in Pasture Dieback Affected Soils
Abstract
1. Introduction
2. Materials and Methods
2.1. Farm Description and Location
2.2. Experimental Design (Plots Description, Location, Treatments, Sampling)
2.3. Sequencing Data Analysis
3. Results
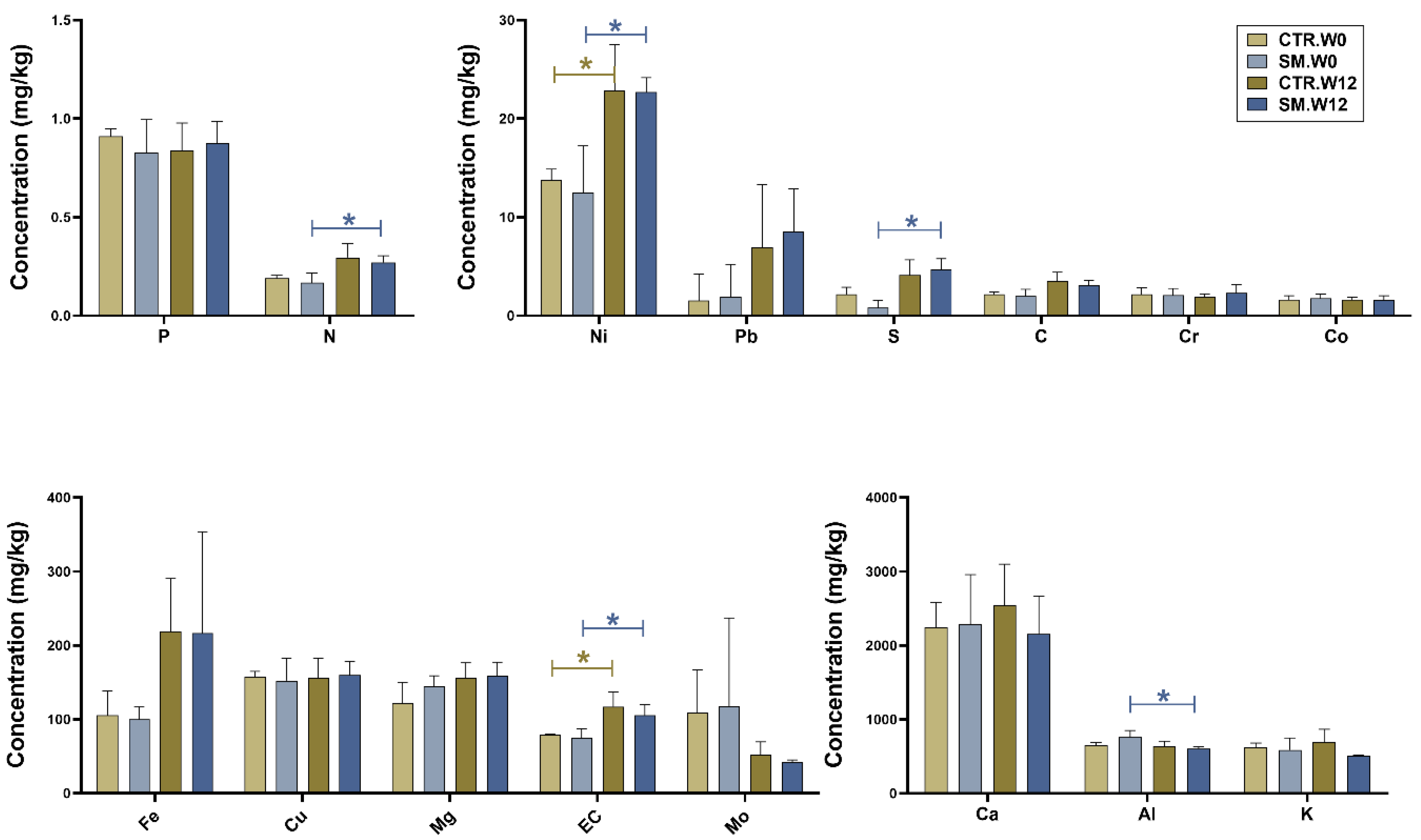
3.1. Soil Chemistry Profile Was Mostly Unaffected
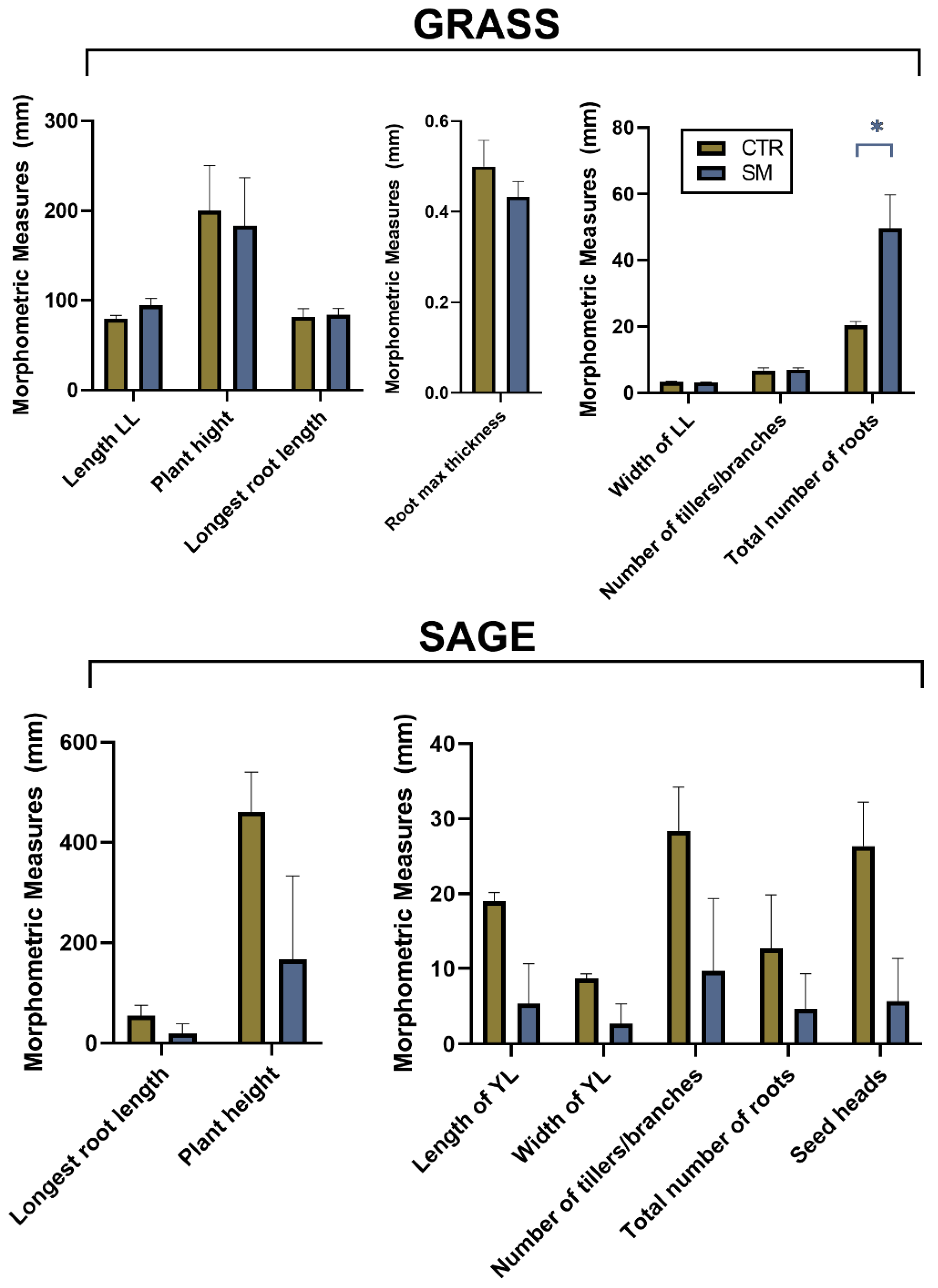
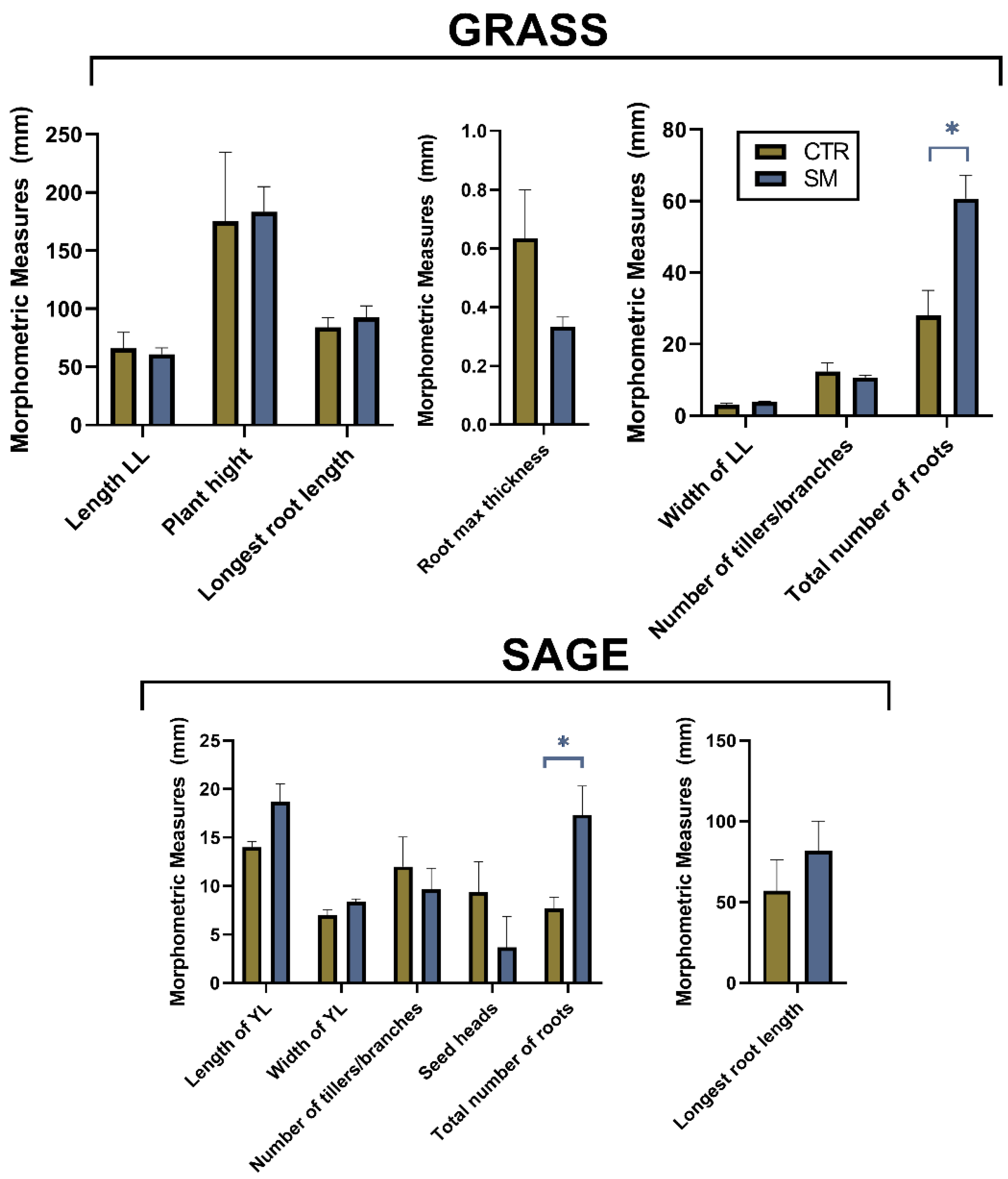
3.2. Impact of SM on Plants
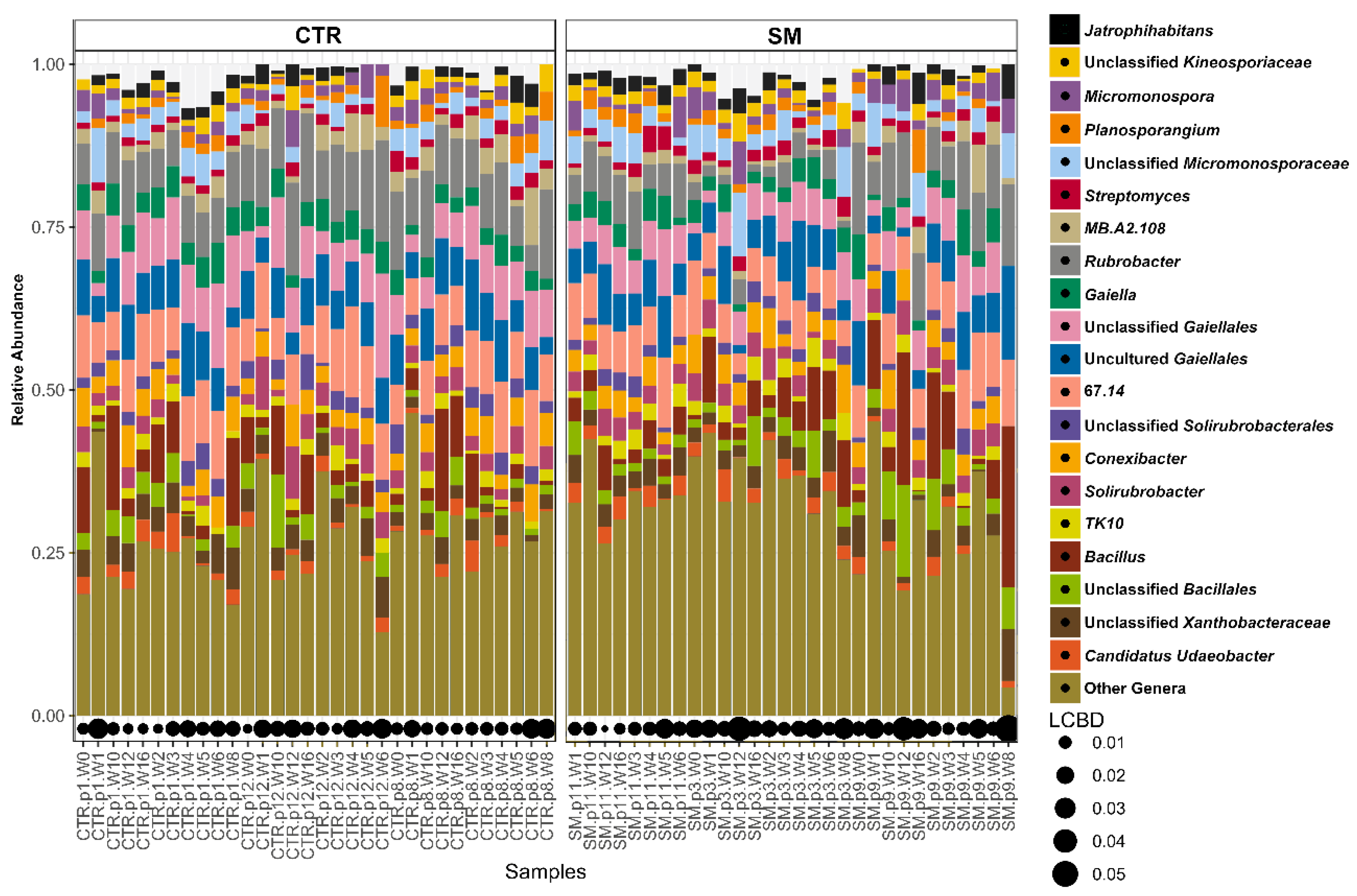
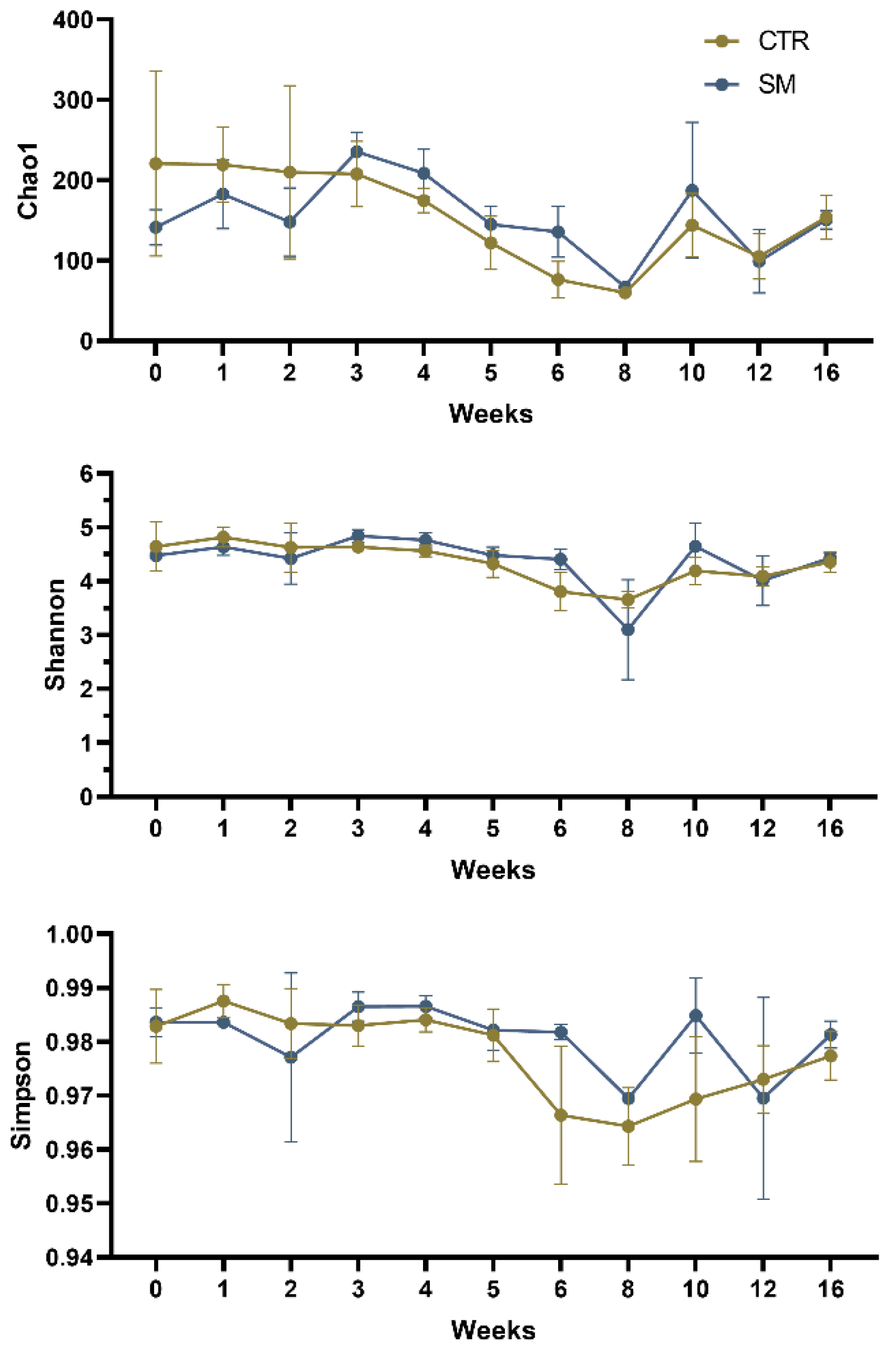
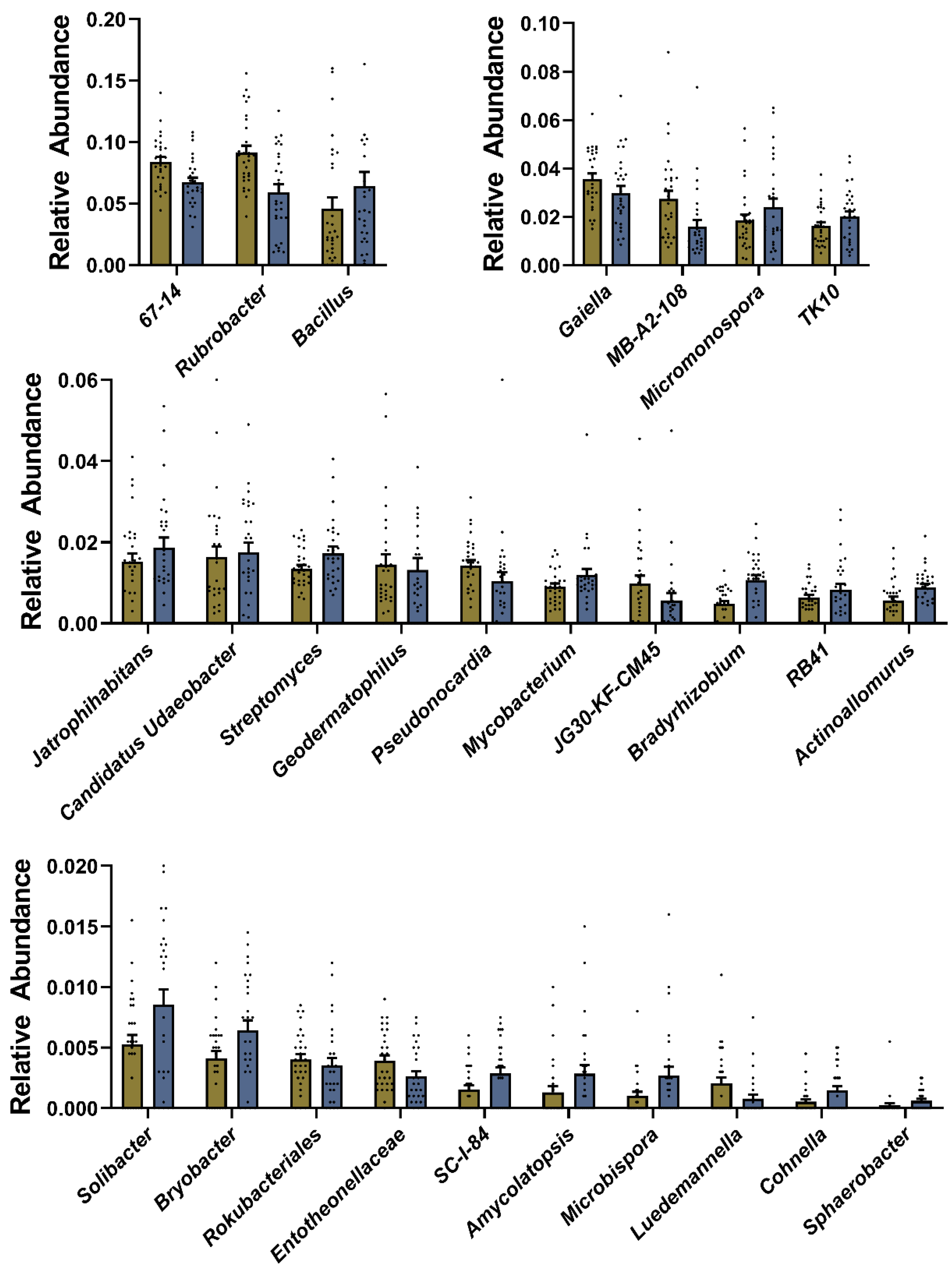
3.3. Soil Microbiota Responded to SM Application
3.4. Multivariate Analysis
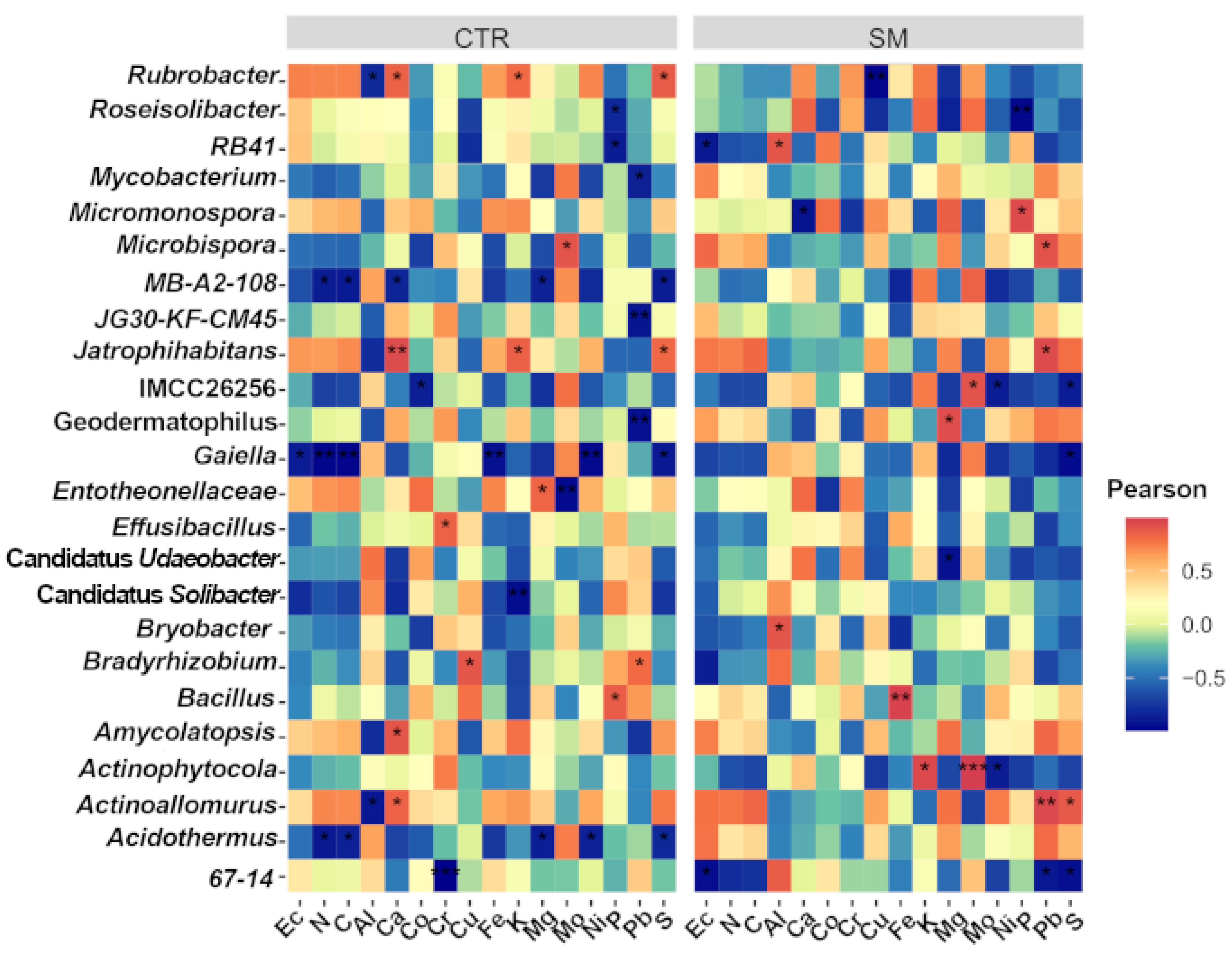
3.5. Microbiota-Environment Interactions
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cerri, C.E.P.; Sparovek, G.; Bernoux, M.; Easterling, W.E.; Melillo, J.M.; Cerri, C.C. Tropical agriculture and global warming: Impacts and mitigation options. Sci. Agric. 2007, 64, 83–99. [Google Scholar] [CrossRef]
- Han, F. Research on the relationship between industrial agglomeration and water pollution and its treatment under the background of green development. Fresenius Environ. Bull. 2022, 31, 258–266. [Google Scholar]
- Liu, Y.; Wang, P.; Gojenko, B.; Yu, J.J.; Wei, L.Z.; Luo, D.G.; Xiao, T.F. A review of water pollution arising from agriculture and mining activities in Central Asia: Facts, causes and effects. Environ. Pollut. 2021, 291, 118209. [Google Scholar] [CrossRef]
- Massarelli, C.; Losacco, D.; Tumolo, M.; Campanale, C.; Uricchio, V.F. Protection of water resources from agriculture pollution: An integrated methodological approach for the Nitrates Directive 91-676-EEC Implementation. Int. J. Environ. Res. Public Health 2021, 18, 13323. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, G.; Bencresciuto, G.F.; Lavrnic, S.; Toscano, A. Diffuse water pollution from agriculture: A review of nature-based solutions for Nitrogen removal and recovery. Water 2021, 13, 1893. [Google Scholar] [CrossRef]
- Zia, M.S.; Khan, M.Q.; Khan, M.J. Waste water use in agriculture and heavy metal pollution in soil-plant system. J. Chem. Soc. Pakistan 2008, 30, 424–430. [Google Scholar]
- Stranak, J. Evaluating of Influence of Soil Pollution by Agriculture in Village Mocenok. Mendelnet. 2013, pp. 372–378. Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwicl8ORrqP7AhWmCrcAHZLYAJ-gQFnoECBQQAQ&url=https%3A%2F%2Fwww.mdpi.com%2F1999-4907%2F13%2F9%2F1445%2Fpdf&usg=AOvVaw2MWdKTbqWplh5sT-um5djB (accessed on 16 August 2020).
- Zhu, D.W.; Wei, Y.; Zhao, Y.H.; Wang, Q.L.; Han, J.C. Heavy metal pollution and ecological risk assessment of the agriculture soil in Xunyang mining area, Shaanxi Province, Northwestern China. Bull. Environ. Contam. Toxicol. 2018, 101, 178–184. [Google Scholar] [CrossRef]
- Moldovan, A.; Torok, A.I.; Kovacs, E.; Cadar, O.; Mirea, I.C.; Micle, V. Metal contents and pollution indices assessment of surface water, soil, and sediment from the Aries river basin mining area, Romania. Sustainability 2022, 14, 8024. [Google Scholar] [CrossRef]
- Chen, L.; Zhou, M.X.; Wang, J.Z.; Zhang, Z.Q.; Duan, C.J.; Wang, X.X.; Zhao, S.L.; Bai, X.H.; Li, Z.J.; Li, Z.M.; et al. A global meta-analysis of heavy metal(loid)s pollution in soils near copper mines: Evaluation of pollution level and probabilistic health risks. Sci. Total Environ. 2022, 835, 155441. [Google Scholar] [CrossRef]
- Ran, H.Z.; Deng, X.G.; Guo, Z.H.; Hu, Z.H.; An, Y.N.; Xiao, X.Y.; Yi, L.W.; Xu, R. Pollution characteristics and environmental availability of toxic elements in soil from an abandoned arsenic-containing mine. Chemosphere 2022, 303, 135189. [Google Scholar] [CrossRef]
- Lal, R. Regenerative agriculture for food and climate. J. Soil Water Conserv. 2020, 75, 123a–124a. [Google Scholar] [CrossRef]
- Giller, K.E.; Hijbeek, R.; Andersson, J.A.; Sumberg, J. Regenerative agriculture: An agronomic perspective. Outlook Agric. 2021, 50, 13–25. [Google Scholar] [CrossRef]
- Kreyling, J.; Dengler, J.; Walter, J.; Velev, N.; Ugurlu, E.; Sopotlieva, D.; Ransijn, J.; Picon-Cochard, C.; Nijs, I.; Hernandez, P.; et al. Species richness effects on grassland recovery from drought depend on community productivity in a multisite experiment. Ecol. Lett. 2017, 20, 1405–1413. [Google Scholar] [CrossRef]
- Wright, A.J.; Mommer, L.; Barry, K.; van Ruijven, J. Stress gradients and biodiversity: Monoculture vulnerability drives stronger biodiversity effects during drought years. Ecology 2021, 102, e03193. [Google Scholar] [CrossRef]
- Padilla, F.M.; Mommer, L.; de Caluwe, H.; Smit-Tiekstra, A.E.; Visser, E.J.W.; de Kroon, H. Effects of extreme rainfall events are independent of plant species richness in an experimental grassland community. Oecologia 2019, 191, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.Q.; Wang, J.; Shen, C.; He, J.Z.; Ge, Y. Plant evenness modulates the effect of plant richness on soil bacterial diversity. Sci. Total Environ. 2019, 662, 8–14. [Google Scholar] [CrossRef]
- Delgado-Baquerizo, M.; Trivedi, P.; Trivedi, C.; Eldridge, D.J.; Reich, P.B.; Jeffries, T.C.; Singh, B.K. Microbial richness and composition independently drive soil multifunctionality. Funct. Ecol. 2017, 31, 2330–2343. [Google Scholar] [CrossRef]
- Zandalinas, S.I.; Fritschi, F.B.; Mittler, R. Global warming, climate change, and environmental pollution: Recipe for a multifactorial stress combination disaster. Trends Plant Sci. 2021, 26, 588–599. [Google Scholar] [CrossRef] [PubMed]
- Zandalinas, S.I.; Sengupta, S.; Fritschi, F.B.; Azad, R.K.; Nechushtai, R.; Mittler, R. The impact of multifactorial stress combination on plant growth and survival. New Phytol. 2021, 230, 1034–1048. [Google Scholar] [CrossRef] [PubMed]
- Dineen, M.; McCarthy, B.; Ross, D.; Ortega, A.; Dillon, P.; Van Amburgh, M.E. Characterization of the nutritive value of perennial ryegrass (Lolium perenne L.) dominated pastures using updated chemical methods with application for the Cornell Net Carbohydrate and Protein System. Anim. Feed. Sci. Technol. 2021, 272, 114752. [Google Scholar] [CrossRef]
- Statistics, A.B.O. Value of Agricultural Commodities Produced, Australia. Available online: https://www.abs.gov.au/statistics/industry/agriculture/value-agricultural-commodities-produced-australia/latest-release#:~:text=Key%20livestock%20results%20for%202020,for%20poultry%20(up%204%25) (accessed on 16 August 2020).
- Makiela, S.; Harrower, K.M. Overview of the current status of buffel grass dieback. Australas. Plant Dis. Notes 2008, 3, 12–16. [Google Scholar] [CrossRef]
- Thomson, M. Hidden Gems: An Epidemiological Investigation into the Association of Ground Pearls with Pasture Dieback; University of Queensland: Brisbane, QLD, Australia, 2019. [Google Scholar]
- Hauxwell, C.; McNicholl, D. Mealybugs and Pasture Dieback; Queensland University of Technology: Brisbane, QLD, Australia; Meat and Livestock Australia: Sydney, NSW, Australia, 2018. [Google Scholar]
- Schjoerring, J.K.; Cakmak, I.; White, P.J. Plant nutrition and soil fertility: Synergies for acquiring global green growth and sustainable development. Plant Soil 2019, 434, 1–6. [Google Scholar] [CrossRef]
- Etienne, P.; Diquelou, S.; Prudent, M.; Salon, C.; Maillard, A.; Ourry, A. Macro and micronutrient storage in plants and their remobilization when facing scarcity: The case of drought. Agriculture 2018, 8, 14. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, M.; Zhou, X.; Li, S.; Zhao, Y.; Li, L.; Hu, X. Effect of the mineral-microbial complexes on the quality, soil nutrients, and microbial community of tailing substrates for growing potted Rorippa. Microbiol. Res. 2022, 262, 127084. [Google Scholar] [CrossRef]
- El-Ramady, H.; El-Henawy, A.; Amer, M.; Omara, A.E.D.; Elsakhawy, T.; Salama, A.M.; Ezzat, A.; Elsherif, A.R.; Elmahrouk, M.; Shalaby, T.A. Agro-Pollutants and their Nano-Remediation from Soil and Water: A Mini-Review. Environ. Biodivers. Soil Secur. 2020, 4, 361–375. [Google Scholar] [CrossRef]
- Kandpal, G. Review on impact of chemical fertilizers on environment. Int. J. Mod. Agric. 2021, 10, 758–763. [Google Scholar]
- Li, W.; Guo, S.; Liu, H.; Zhai, L.; Wang, H.; Lei, Q. Comprehensive environmental impacts of fertilizer application vary among different crops: Implications for the adjustment of agricultural structure aimed to reduce fertilizer use. Agric. Water Manag. 2018, 210, 1–10. [Google Scholar] [CrossRef]
- Liu, S.; Xie, Z.; Zeng, Y.; Liu, B.; Li, R.; Wang, Y.; Wang, L.; Qin, P.; Jia, B.; Xie, J. Effects of anthropogenic nitrogen discharge on dissolved inorganic nitrogen transport in global rivers. Glob. Chang. Biol. 2019, 25, 1493–1513. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.; Valentine, T. Sea Energy Agriculture; Valentine Books: London, UK, 1976. [Google Scholar]
- Perera, S.S.; Wasalathilake, K.C.; Wijayarathne, U.P.L.; Vidanage, P.W. Development of a multi-nutrient fertilizer from liquid waste of solar salt manufacturing process. In Proceedings of the Moratuwa Engineering Research Conference (MerCon), Moratuwa, Sri Lanka, 7–8 April 2015; pp. 24–28. [Google Scholar]
- Loganathan, P.; Naidu, G.; Vigneswaran, S. Mining valuable minerals from seawater: A critical review. Environ. Sci. Water Res. Technol. 2017, 3, 37–53. [Google Scholar] [CrossRef]
- Durgo, K.; Orescanin, V.; Lulic, S.; Kopjar, N.; Eljezic, D.Z.; Colic, J.F. The assessment of genotoxic effects of wastewater from a fertilizer factory. J. Appl. Toxicol. 2009, 29, 42–51. [Google Scholar] [CrossRef]
- Radic, S.; Gregorovic, G.; Stipanicev, D.; Cvjetko, P.; Srut, M.; Vujcic, V.; Orescanin, V.; Vinko Klobucar, G.I. Assessment of surface water in the vicinity of fertilizer factory using fish and plants. Ecotoxicol. Environ. Saf. 2013, 96, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Weisenburger, D.D. Human health effects of agrichemical use. Hum. Pathol. 1993, 24, 571–576. [Google Scholar] [CrossRef]
- Paltineanu, C.; Domnariu, H.; Marica, D.; Lacatusu, A.R.; Popa, G.A.; Grafu, I.A.; Neagoe, A.D. Fertilizers’ leaching from the root system zone—A potential environmental risk for groundwater pollution in coarse and medium-textured soils. Carpathian J. Earth Environ. Sci. 2021, 16, 139–150. [Google Scholar] [CrossRef]
- Aoun, M.; Arnaudguilhem, C.; El Samad, O.; Khozam, R.B.; Lobinski, R. Impact of a phosphate fertilizer plant on the contamination of marine biota by heavy elements. Environ. Sci. Pollut. Res. 2015, 22, 14940–14949. [Google Scholar] [CrossRef] [PubMed]
- Dal Molin, S.J.; Ernani, P.R.; Gerber, J.M. Soil acidification and nitrogen release following application of nitrogen ertilizers. Commun. Soil Sci. Plant Anal. 2020, 51, 2551–2558. [Google Scholar] [CrossRef]
- Tkaczyk, P.; Mocek-Plociniak, A.; Skowronska, M.; Bednarek, W.; Kusmierz, S.; Zawierucha, E. The mineral fertilizer-dependent chemical parameters of soil acidification under field conditions. Sustainability 2020, 12, 7165. [Google Scholar] [CrossRef]
- Schroder, J.L.; Zhang, H.L.; Girma, K.; Raun, W.R.; Penn, C.J.; Payton, M.E. Soil acidification from long-term use of Nitrogen fertilizers on winter wheat. Soil Sci. Soc. Am. J. 2011, 75, 957–964. [Google Scholar] [CrossRef]
- Pavlidis, G.; Karasali, H.; Tsihrintzis, V.A. Pesticide and fertilizer pollution reduction in two alley cropping agroforestry cultivating systems. Water Air Soil Pollut. 2020, 231, 241. [Google Scholar] [CrossRef]
- Fouda, S.E.; Niel, E.M. Influence of compost tea and potassium humate on soil properties and plant growth. Asian J. Soil Sci. Plant Nutr. 2021, 29–40. [Google Scholar] [CrossRef]
- Ferguson, R.B.; Hergert, G.W.; Shapiro, C.A.; Wortmann, C.S. Guidelines for Soil Sampling; University of Nebraska–Lincoln: Lincoln, NE, USA, 2007. [Google Scholar]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Underwood, A.J. (Ed.) Experiments in Ecology: Their Logical Design and Interpretation Using Analysis of Variance; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Bill, M.; Chidamba, L.; Gokul, J.K.; Labuschagne, N.; Korsten, L. Bacterial community dynamics and functional profiling of soils from conventional and organic cropping systems. Appl. Soil Ecol. 2021, 157, 103734. [Google Scholar] [CrossRef]
- Guo, Y.X.; Chen, Q.J.; Qin, Y.; Yang, Y.R.; Yang, Q.Z.; Wang, Y.X.; Cheng, Z.A.; Cao, N.; Zhang, G.Q. Succession of the microbial communities and function prediction during short-term peach sawdust-based composting. Bioresour. Technol. 2021, 332, 125079. [Google Scholar] [CrossRef]
- Borah, A.; Hazarika, S.N.; Thakur, D. Potentiality of actinobacteria to combat against biotic and abiotic stresses in tea [Camellia sinensis (L) O. Kuntze]. J. Appl. Microbiol. 2022, 133, 2314–2330. [Google Scholar] [CrossRef]
- Charalambous, A.; Grivogiannis, E.; Dieronitou, I.; Michael, C.; Rahme, L.; Apidianakis, Y. Proteobacteria and Firmicutes secreted factors exert distinct effects on Pseudomonas aeruginosa infection under normoxia or mild hypoxia. Metabolites 2022, 12, 449. [Google Scholar] [CrossRef]
- Coumes-Florens, S.; Brochier-Armanet, C.; Guiseppi, A.; Denizot, F.; Foglino, M. A new highly conserved antibiotic sensing/resistance pathway in Firmicutes Involves an ABC transporter interplaying with a Signal Transduction System. PLoS ONE 2011, 6, e15951. [Google Scholar] [CrossRef]
- Nkosi, B.V.Z.; Padayachee, T.; Gront, D.; Nelson, D.R.; Syed, K. Contrasting health effects of Bacteroidetes and Firmicutes lies in their genomes: Analysis of P450s, ferredoxins, and secondary metabolite clusters. Int. J. Mol. Sci. 2022, 23, 5057. [Google Scholar] [CrossRef]
- Wagi, S.; Ahmed, A. Bacillus spp.: Potent microfactories of bacterial IAA. PeerJ 2019, 7, e7258. [Google Scholar] [CrossRef]
- Idris, E.E.; Iglesias, D.J.; Talon, M.; Borriss, R. Tryptophan-dependent production of indole-3-acetic acid (IAA) affects level of plant growth promotion by Bacillus amyloliquefaciens FZB42. Mol. Plant-Microbe Interact. 2007, 20, 619–626. [Google Scholar] [CrossRef]
- Kim, S.T.; Yoo, S.J.; Weon, H.Y.; Song, J.; Sang, M.K. Bacillus butanolivorans KJ40 contributes alleviation of drought stress in pepper plants by modulating antioxidant and polyphenolic compounds. Sci. Hortic. 2022, 301, 111111. [Google Scholar] [CrossRef]
- Da Fonseca, M.D.; Bossolani, J.W.; de Oliveira, S.L.; Moretti, L.G.; Portugal, J.R.; Scudeletti, D.; de Oliveira, E.F.; Crusciol, C.A.C. Bacillus subtilis inoculation improves nutrient uptake and physiological activity in sugarcane under drought stress. Microorganisms 2022, 10, 809. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.Z.; Zhang, Z.F.; Zhou, N.; Jiang, C.Y.; Wang, B.J.; Cai, L.; Liu, S.J. Diversity, distribution and co-occurrence patterns of bacterial communities in a Karst cave system. Front. Microbiol. 2019, 10, 1726. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, M.; Ma, Y.; Freitas, H. Characterization of metal-resistant plant-growth promoting Bacillus weihenstephanensis isolated from serpentine soil in Portugal. J. Basic Microbiol. 2008, 48, 500–508. [Google Scholar] [CrossRef]
- Jang, S.W.; Yoou, M.H.; Hong, W.J.; Kim, Y.J.; Lee, E.J.; Jung, K.H. Re-analysis of 16S amplicon sequencing data reveals soil microbial population shifts in rice fields under drought condition. Rice 2020, 13, 44. [Google Scholar] [CrossRef]
- Yang, S.; Xiao, J.; Liang, T.; He, W.; Tan, H. Response of soil biological properties and bacterial diversity to different levels of nitrogen application in sugarcane fields. AMB Express 2021, 11, 1–11. [Google Scholar] [CrossRef]
- Martinez-Hidalgo, P.; Garcia, J.M.; Pozo, M.J. Induced systemic resistance against Botrytis cinerea by Micromonospora strains isolated from root nodules. Front. Microbiol. 2015, 6, 922. [Google Scholar] [CrossRef]
- Ismet, A.; Vikineswary, S.; Paramaswari, S.; Wong, W.H.; Ward, A.; Seki, T.; Fiedler, H.P.; Goodfellow, M. Production and chemical characterization of antifungal metabolites from Micromonospora sp M39 isolated from mangrove rhizosphere soil. World J. Microb. Biot. 2004, 20, 523–528. [Google Scholar] [CrossRef]
- El-Tarabily, K.A.; AlKhajeh, A.S.; Ayyash, M.M.; Alnuaimi, L.H.; Sham, A.; ElBaghdady, K.Z.; Tariq, S.; AbuQamar, S.F. Growth promotion of salicornia bigelovii by Micromonospora chalcea UAE1, an endophytic 1-Aminocyclopropane-1-Carboxylic Acid Deaminase-Producing Actinobacterial isolate. Front. Microbiol. 2019, 10, 1694. [Google Scholar] [CrossRef]
- Zhao, S.S.; Liu, C.X.; Zheng, W.W.; Ma, Z.X.; Cao, T.T.; Zhao, J.W.; Yan, K.; Xiang, W.S.; Wang, X.J. Micromonospora parathelypteridis sp nov., an endophytic actinomycete with antifungal activity isolated from the root of Parathelypteris beddomei (Bak.) Ching. Int. J. Syst. Evol. Micr. 2017, 67, 268–274. [Google Scholar] [CrossRef]
- Kisil, O.V.; Efimenko, T.A.; Efremenkova, O.V. Looking back to amycolatopsis: History of the antibiotic discovery and future prospects. Antibiotics 2021, 10, 1254. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.Q.; Xu, T.C.; Wang, J.F.; Hou, Y.G.; Liu, C.S.; Liu, S.S.; Wu, S.H. Secondary metabolites of the genus Amycolatopsis: Structures, bioactivities and biosynthesis. Molecules 2021, 26, 1884. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Han, Y.; Li, X.; Li, M.; Wang, C.; Li, Z.; Wang, Y.; Wang, W. A salt-tolerant Streptomyces paradoxus D2-8 from rhizosphere soil of phragmites communis augments soybean tolerance to soda saline-alkali stress. Pol. J. Microbiol. 2022, 71, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Das, P.; Sarkar, B.; Ghati, A.; Mondal, R.; Dam, P.; Franco, O.L.; Cardoso, M.H.; Sharma, A.; Swarnakar, S.; Miere Groza, F.; et al. Draft genome sequence of Streptomyces sp. Strain PSAA01, isolated from the soil of eastern Himalayan foothills. Microbiol. Resour. Announc. 2022, 11, e00370-22. [Google Scholar] [CrossRef] [PubMed]
- Carlucci, A.; Raimondo, M.L.; Colucci, D.; Lops, F. Streptomyces albidoflavus Strain CARA17 as a biocontrol agent against fungal soil-borne pathogens of fennel plants. Plants 2022, 11, 1420. [Google Scholar] [CrossRef] [PubMed]
- Eida, M.F.; Nagaoka, T.; Wasaki, J.; Kouno, K. Isolation and characterization of cellulose-decomposing bacteria inhabiting sawdust and coffee residue composts. Microbes Environ. 2012, 27, 226–233. [Google Scholar] [CrossRef]
- Gong, X.J.; Xiang, W.S.; Cao, X.; Yu, Y.; Hao, Y.B.; Li, L.; Wang, Q.J.; Zou, H.T.; Qian, C.R. Microbispora cellulosiformans sp. nov., a novel actinomycete with cellulase activity isolated from soil in the cold region. Antonie Van Leeuwenhoek 2020, 113, 2053–2062. [Google Scholar] [CrossRef]
- Ningsih, F.; Sari, D.C.A.F.; Rachmania, M.K.; Yabe, S.; Yokota, A.; Oetari, A.; Sjamsuridzal, W. Isolation and 16S rRNA gene sequences analysis of thermophilic Actinobacteria isolated from soil in Cisolok geothermal area, West Java, Indonesia. IOP Conf. Ser. Earth Environ. Sci. 2020, 457, 012015. [Google Scholar] [CrossRef]
- Pozzi, R.; Simone, M.; Mazzetti, C.; Maffioli, S.; Monciardini, P.; Cavaletti, L.; Bamonte, R.; Sosio, M.; Donadio, S. The genus Actinoallomurus and some of its metabolites. J. Antibiot. 2011, 64, 133–139. [Google Scholar] [CrossRef]
- Mazzetti, C.; Ornaghi, M.; Gaspari, E.; Parapin, S.; Maffioli, S.; Sosio, M.; Donadio, S. Halogenated Spirotetronates from Actinoallomurus. J. Nat. Prod. 2012, 75, 1044–1050. [Google Scholar] [CrossRef]
- Cheung, P.Y.; Kinkle, B.K. Mycobacterium diversity and pyrene mineralization in petroleum-contaminated soils. Appl. Environ. Microb. 2001, 67, 2222–2229. [Google Scholar] [CrossRef] [PubMed]
- Leys, N.M.; Bastiaens, L.; Verstraete, W.; Springael, D. Influence of the carbon/nitrogen/phosphorus ratio on polycyclic aromatic hydrocarbon degradation by Mycobacterium and Sphingomonas in soil. Appl. Microbiol. Biot. 2005, 66, 726–736. [Google Scholar] [CrossRef] [PubMed]
- Walsh, C.M.; Gebert, M.J.; Delgado-Baquerizo, M.; Maestre, F.T.; Fierer, N. A global survey of mycobacterial diversity in soil. Appl. Environ. Microb. 2019, 85. [Google Scholar] [CrossRef] [PubMed]
- Foesel, B.U.; Rohde, M.; Overmann, J. Blastocatella fastidiosa gen. nov., sp. nov., isolated from semiarid savanna soil—The first described species of Acidobacteria subdivision 4. Syst. Appl. Microbiol. 2013, 36, 82–89. [Google Scholar] [CrossRef]
- Stone, B.W.; Li, J.H.; Koch, B.J.; Blazewicz, S.J.; Dijkstra, P.; Hayer, M.; Hofmockel, K.S.; Liu, X.J.A.; Mau, R.L.; Morrissey, E.M.; et al. Nutrients cause consolidation of soil carbon flux to small proportion of bacterial community. Nat. Commun. 2021, 12, 3381. [Google Scholar] [CrossRef]
- Wang, M.; Chen, S.B.; Chen, L.; Wang, D. Responses of soil microbial communities and their network interactions to saline-alkaline stress in Cd-contaminated soils. Environ. Pollut. 2019, 252, 1609–1621. [Google Scholar] [CrossRef]
- Challacombe, J.F.; Eichorst, S.A.; Hauser, L.; Land, M.; Xie, G.; Kuske, C.R. Biological consequences of ancient gene acquisition and duplication in the large genome of Candidatus Solibacter usitatus Ellin6076. PLoS ONE 2011, 6, e24882. [Google Scholar] [CrossRef]
- Zhang, R.H.; Li, R.; Zhang, L.L. Soil nutrient variability mediates the effects of erosion on soil microbial communities: Results from a modified topsoil removal method in an agricultural field in Yunnan plateau, China. Environ. Sci. Pollut. Res. 2022, 29, 3659–3671. [Google Scholar] [CrossRef]
- Wang, B.; Wang, Y.; Wei, Y.; Chen, W.; Ding, G.; Zhan, Y.; Liu, Y.; Xu, T.; Xiao, J.; Li, J. Impact of inoculation and turning for full-scale composting on core bacterial community and their co-occurrence compared by network analysis. Bioresour. Technol. 2022, 345, 126417. [Google Scholar] [CrossRef]
- Zhan, Y.B.; Chang, Y.; Tao, Y.Y.; Zhang, H.; Lin, Y.F.; Deng, J.; Ma, T.T.; Ding, G.C.; Wei, Y.Q.; Li, J. Insight into the dynamic microbial community and core bacteria in composting from different sources by advanced bioinformatics methods. Environ. Sci. Pollut. Res. 2022, 1–11. [Google Scholar] [CrossRef]
- Wang, L.Y.; Chen, S.F.; Wang, L.; Zhou, Y.G.; Liu, H.C. Cohnella plantaginis sp. nov., a novel nitrogen-fixing species isolated from plantain rhizosphere soil. Antonie Van Leeuwenhoek 2012, 102, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Maeng, S.; Kim, M.K.; Jang, J.H.; Yi, H.; Subramani, G. Cohnella candidum sp. nov., radiation-resistant bacterium from soil. Antonie Van Leeuwenhoek 2019, 112, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Ormeno-Orrillo, E.; Martinez-Romero, E. A genomotaxonomy view of the Bradyrhizobium genus. Front. Microbiol. 2019, 10, 1334. [Google Scholar] [CrossRef] [PubMed]
- Vetrovsky, T.; Baldrian, P. An in-depth analysis of actinobacterial communities shows their high diversity in grassland soils along a gradient of mixed heavy metal contamination. Biol. Fert. Soils 2015, 51, 827–837. [Google Scholar] [CrossRef]
- Madhaiyan, M.; Hu, C.J.; Kim, S.J.; Weon, H.Y.; Kwon, S.W.; Ji, L.H. Jatrophihabitans endophyticus gen. nov., sp nov., an endophytic actinobacterium isolated from a surface-sterilized stem of Jatropha curcas L. Int. J. Syst. Evol. Microbiol. 2013, 63, 1241–1248. [Google Scholar] [CrossRef] [PubMed]
- Li, C.W.; Chen, G.Z.; Zhang, J.L.; Zhu, P.; Bai, X.F.; Hou, Y.P.; Zhang, X.X. The comprehensive changes in soil properties are continuous cropping obstacles associated with American ginseng (Panax quinquefolius) cultivation. Sci. Rep. 2021, 11, 5068. [Google Scholar] [CrossRef]
- Goswami, A.; Adkins-Jablonsky, S.; Barreto Filho, M.M.; Schilling, M.; Dawson, A.; Heiser, S.; O’Connor, A.; Walker, M.; Roberts, Q.; Morris, J. Heavy metal pollution impacts soil bacterial community structure and antimicrobial resistance at the Birmingham 35th Avenue Superfund Site. bioRxiv 2022. [Google Scholar] [CrossRef]
- Karpinska, A.; Ryan, D.; Germaine, K.; Dowling, D.; Forrestal, P.; Kakouli-Duarte, T. Soil microbial and nematode community response to the field application of recycled bio-based fertilisers in Irish grassland. Sustainability 2021, 13, 12342. [Google Scholar] [CrossRef]
- Egas, C.; Barroso, C.; Froufe, H.J.C.; Pacheco, J.; Albuquerque, L.; da Costa, M.S. Complete genome sequence of the Radiation-Resistant bacterium Rubrobacter radiotolerans RSPS-4. Stand. Genom. Sci. 2014, 9, 1062–1075. [Google Scholar] [CrossRef]
- Zhang, C.F.; Tayyab, M.; Abubakar, A.Y.; Yang, Z.Q.; Pang, Z.Q.; Islam, W.; Lin, Z.L.; Li, S.Y.; Luo, J.; Fan, X.L.; et al. Bacteria with different assemblages in the soil profile drive the diverse nutrient cycles in the sugarcane straw retention ecosystem. Diversity 2019, 11, 194. [Google Scholar] [CrossRef]
- Lan, J.; Wang, S.; Wang, J.; Qi, X.; Long, Q.; Huang, M. The shift of soil bacterial community after afforestation influence soil organic carbon and aggregate stability in Karst region. Front. Microbiol. 2022, 13, 901126. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Wang, J.; Xie, M.; Zhao, F.; Doughty, R. Increasing temperature can modify the effect of straw mulching on soil C fractions, soil respiration, and microbial community composition. PLoS ONE 2020, 15, e0237245. [Google Scholar] [CrossRef] [PubMed]
- Montero-Calasanz Mdel, C.; Goker, M.; Broughton, W.J.; Cattaneo, A.; Favet, J.; Potter, G.; Rohde, M.; Sproer, C.; Schumann, P.; Klenk, H.P.; et al. Geodermatophilus tzadiensis sp. nov., a UV radiation-resistant bacterium isolated from sand of the Saharan desert. Syst. Appl. Microbiol. 2013, 36, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Castro, J.F.; Nouioui, I.; Sangal, V.; Trujillo, M.E.; Montero-Calasanz, M.D.C.; Rahmani, T.; Bull, A.T.; Asenjo, J.A.; Andrews, B.A.; Goodfellow, M. Geodermatophilus chilensis sp. nov., from soil of the Yungay core-region of the Atacama Desert, Chile. Syst. Appl. Microbiol. 2018, 41, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.Z.; Li, J.; Qin, Y.L.; Miao, C.P.; Wei, D.Q.; Zhang, S.; Xu, L.H.; Li, W.J. Pseudonocardia antimicrobica sp nov., a novel endophytic actinomycete associated with Artemisia annua L. (sweet wormwood). J. Antibiot. 2012, 65, 469–472. [Google Scholar] [CrossRef]
- Goldstein, S.L.; Klassen, J.L. Pseudonocardia symbionts of fungus-growing ants and the evolution of defensive secondary metabolism. Front. Microbiol. 2020, 11, 621041. [Google Scholar] [CrossRef]
- Shen, M.C.; Zhang, Y.Z.; Bo, G.D.; Yang, B.; Wang, P.; Ding, Z.Y.; Wang, Z.B.; Yang, J.M.; Zhang, P.; Yuan, X.L. Microbial responses to the reduction of chemical fertilizers in the rhizosphere soil of flue-cured tobacco. Front. Bioeng. Biotechnol. 2021, 9, 812316. [Google Scholar] [CrossRef]
- Li, H.; Wang, X.M.; Wu, Y.; Zhang, D.R.; Xu, H.Y.; Xu, H.R.; Xing, X.G.; Qi, Z.L. Relationships among bedding materials, bedding bacterial composition and lameness in dairy cows. Anim. Biosci. 2021, 34, 1559–1568. [Google Scholar] [CrossRef]
- Zhang, X.C.; Myrold, D.D.; Shi, L.L.; Kuzyakov, Y.; Dai, H.C.; Hoang, D.T.T.; Dippold, M.A.; Meng, X.T.; Song, X.N.; Li, Z.Y.; et al. Resistance of microbial community and its functional sensitivity in the rhizosphere hotspots to drought. Soil Biol. Biochem. 2021, 161, 108360. [Google Scholar] [CrossRef]
| Mineral | Concentration |
|---|---|
| Total dissolved ions | 32% |
| Sodium | 2.87% |
| Chloride | 19% |
| Potassium | 1% |
| Aluminium | 0.1 mg/L |
| Copper | 0.09 mg/L |
| Manganese | 6.78 mg/L |
| Boron | 469.51 mg/L |
| Molybdenum | 0.05 mg/L |
| Barium | 0.015 mg/L |
| Beryllium | 0.01 mg/L |
| Calcium | 10 mg/L |
| Phosphorus | 12 mg/L |
| Sulphur | 17429 mg/L |
| Magnesium | 12% |
| Zinc | 1.85 mg/L |
| Lithium | 17.1 mg/L |
| Titanium | 0.13 mg/L |
| Uranium | 0.041 mg/L |
| Vanadium | 0.1 mg/L |
| Arsenic | 0.01 mg/L |
| Mercury | 0.01 mg/L |
| Selenium | 0.01 mg/L |
| Cadmium | 1.43 mg/L |
| Chromium | 4.7 mg/L |
| Lead | 0.6 mg/L |
| Nickel | 0.6 mg/L |
| Silver | 0.35 mg/L |
| Tin | 0.01 mg/L |
| Nitrates-Nitrogen | 0.1 mg/L |
| Nitrogen | 0.011% w/w |
| Sulphate | 7.01% |
| Salt from chloride | 31% |
| Genus | Contribution% | Cumulative% |
|---|---|---|
| Bacillus | 7.14 | 7.14 |
| Rubrobacter | 5.86 | 13 |
| Unknown Gaiellales | 4.14 | 17.14 |
| Unclassified Gaiellales | 3.7 | 20.83 |
| Unknown Bacillales | 3.35 | 24.18 |
| 67-14 | 3.23 | 27.41 |
| Unclassified Micromonosporaceae | 2.83 | 30.24 |
| Acidothermus | 2.41 | 32.64 |
| MB-A2-108 | 2.37 | 35.01 |
| Micromonospora | 2.24 | 37.25 |
| Unclassified Xanthobacteraceae | 2.2 | 39.45 |
| Solirubrobacter | 2.19 | 41.64 |
| Gaiella | 2.18 | 43.82 |
| Conexibacter | 2.03 | 45.85 |
| Planosporangium | 1.87 | 47.72 |
| Candidatus Udaeobacter | 1.83 | 49.55 |
| Geodermatophilus | 1.82 | 51.37 |
| Unclassified Gaiellales | 1.71 | 53.08 |
| Jatrophihabitans | 1.65 | 54.73 |
| TK10 | 1.47 | 56.2 |
| Unclassified Kineosporiaceae | 1.45 | 57.65 |
| Pseudonocardia | 1.3 | 58.95 |
| JG30-KF-CM45 | 1.27 | 60.22 |
| Unclassified Actinobacteriota | 1.16 | 61.38 |
| Unclassified Elsterales | 1.1 | 62.47 |
| Unclassified IMCC26256 | 1.08 | 63.55 |
| Streptomyces | 1.08 | 64.63 |
| Bradyrhizobium | 0.96 | 65.59 |
| RB41 | 0.95 | 66.54 |
| Rhodoplanes | 0.88 | 67.43 |
| Solibacter | 0.87 | 68.3 |
| Unclassified Vicinamibacterales | 0.84 | 69.14 |
| Unclassified Gemmatimonadaceae | 0.82 | 69.96 |
| Kribbella | 0.82 | 70.78 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Whitton, M.M.; Ren, X.; Yu, S.J.; Irving, A.D.; Trotter, T.; Bajagai, Y.S.; Stanley, D. Sea Minerals Reduce Dysbiosis, Improve Pasture Productivity and Plant Morphometrics in Pasture Dieback Affected Soils. Sustainability 2022, 14, 14873. https://doi.org/10.3390/su142214873
Whitton MM, Ren X, Yu SJ, Irving AD, Trotter T, Bajagai YS, Stanley D. Sea Minerals Reduce Dysbiosis, Improve Pasture Productivity and Plant Morphometrics in Pasture Dieback Affected Soils. Sustainability. 2022; 14(22):14873. https://doi.org/10.3390/su142214873
Chicago/Turabian StyleWhitton, Maria M., Xipeng Ren, Sung J. Yu, Andrew D. Irving, Tieneke Trotter, Yadav S. Bajagai, and Dragana Stanley. 2022. "Sea Minerals Reduce Dysbiosis, Improve Pasture Productivity and Plant Morphometrics in Pasture Dieback Affected Soils" Sustainability 14, no. 22: 14873. https://doi.org/10.3390/su142214873
APA StyleWhitton, M. M., Ren, X., Yu, S. J., Irving, A. D., Trotter, T., Bajagai, Y. S., & Stanley, D. (2022). Sea Minerals Reduce Dysbiosis, Improve Pasture Productivity and Plant Morphometrics in Pasture Dieback Affected Soils. Sustainability, 14(22), 14873. https://doi.org/10.3390/su142214873










