Characterization of Pomegranate (Punica granatum L.) through the Morphoanatomical and Lipid Profile of Seeds for the Valorisation of Italian Accessions
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Morphoanatomical Analysis
2.3. Hardness Analysis
2.3.1. Texturometer Analysis
2.3.2. Gravimetric Chemical Analysis
2.4. Oil Analysis
2.4.1. Extraction of Pomegranate Oil and Chemical Characterization by GC-MS
2.4.2. Fatty Acids Profile
2.4.3. Unsaponifiable Fraction Analysis
2.5. Statistical Analysis
3. Results and Discussions
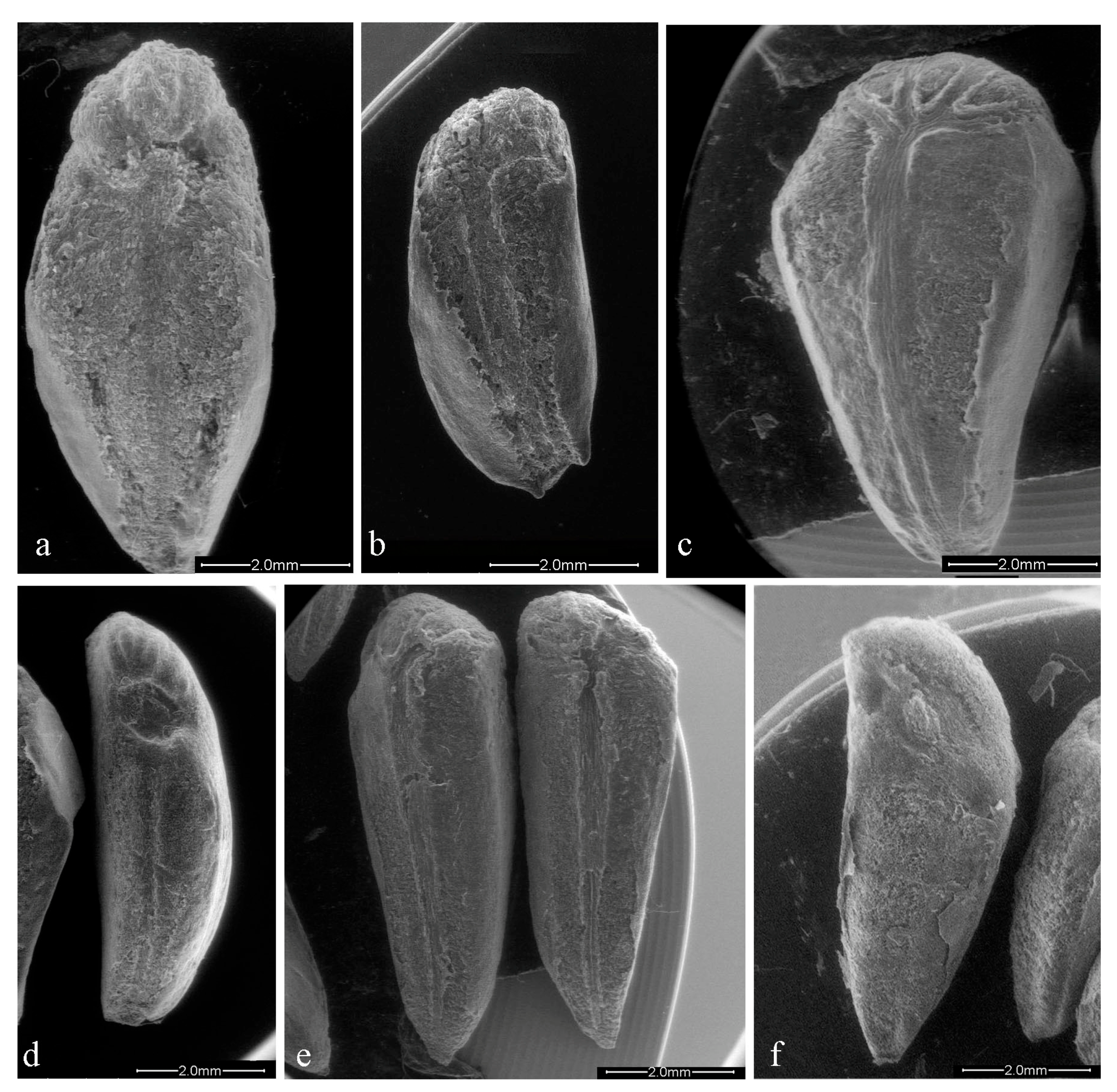
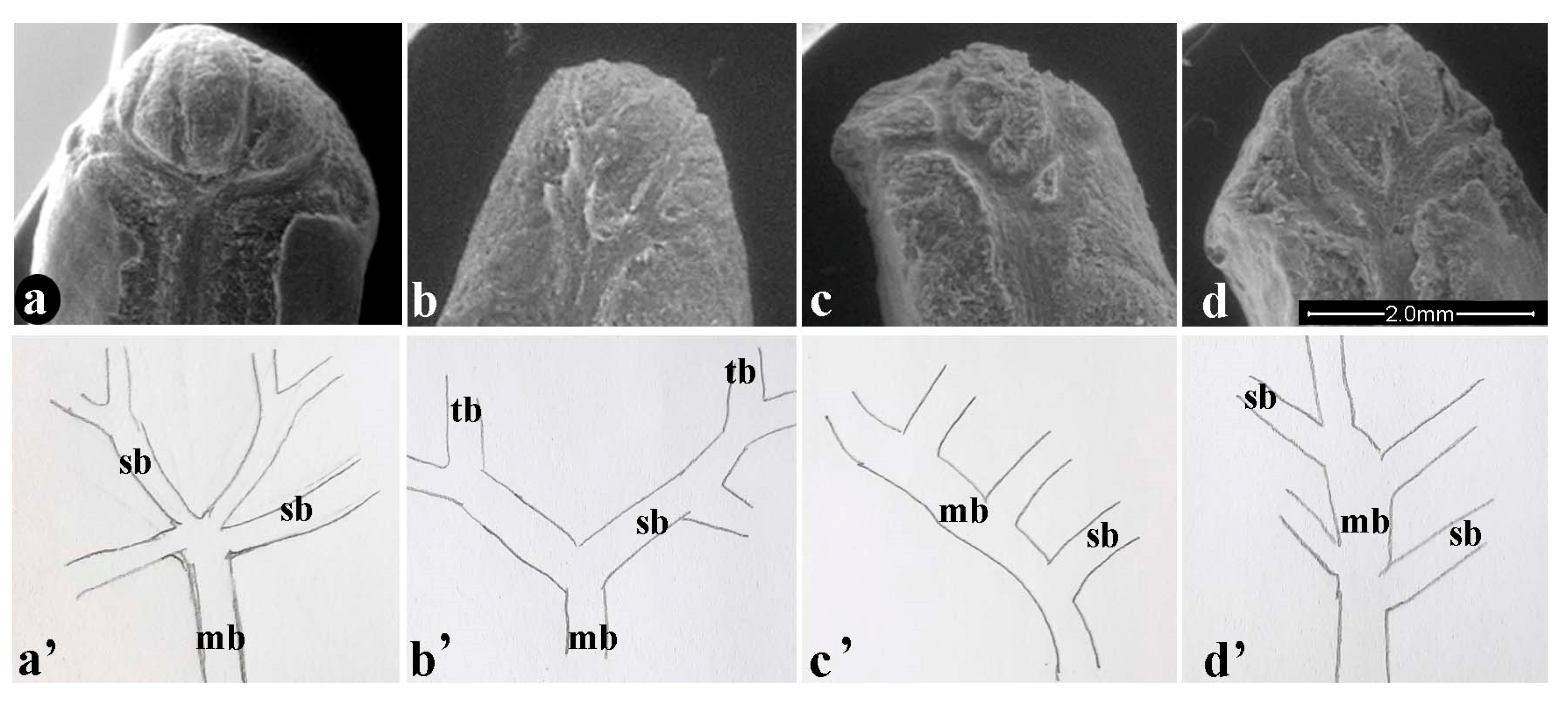
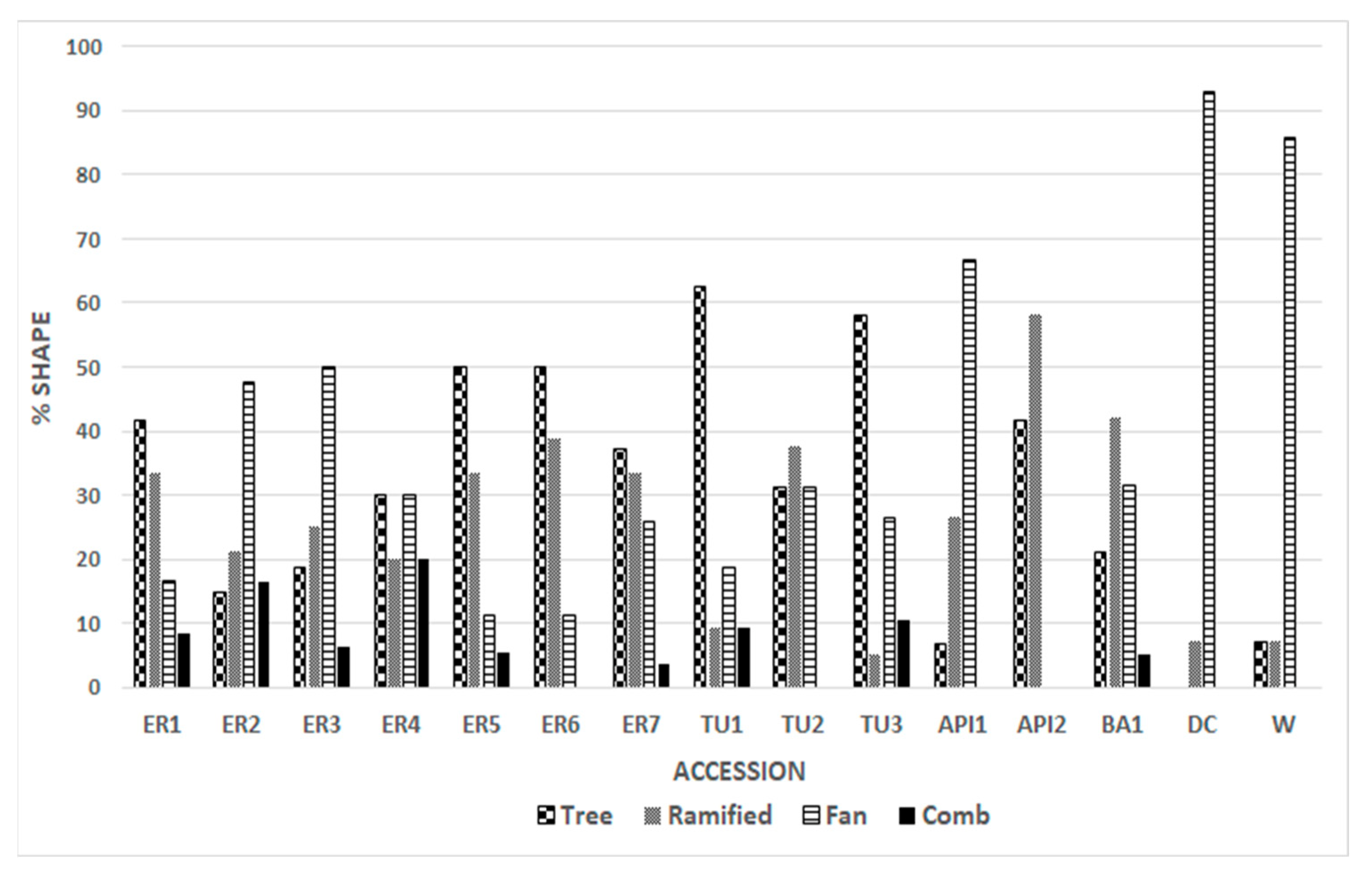
3.1. Morphoanatomical Analysis
3.2. Hardness and Chemical Analysis
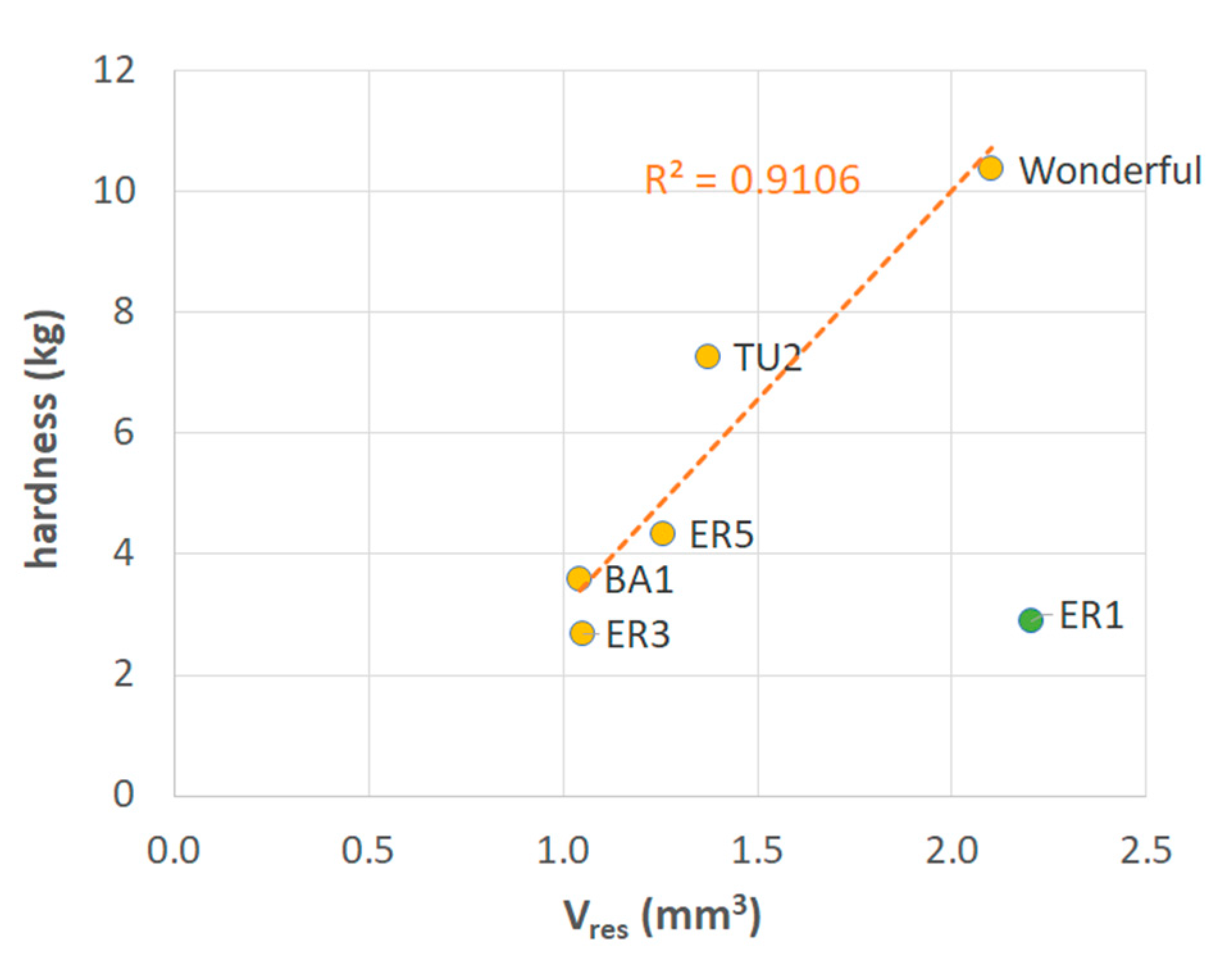
3.3. Mechanical Data
- −
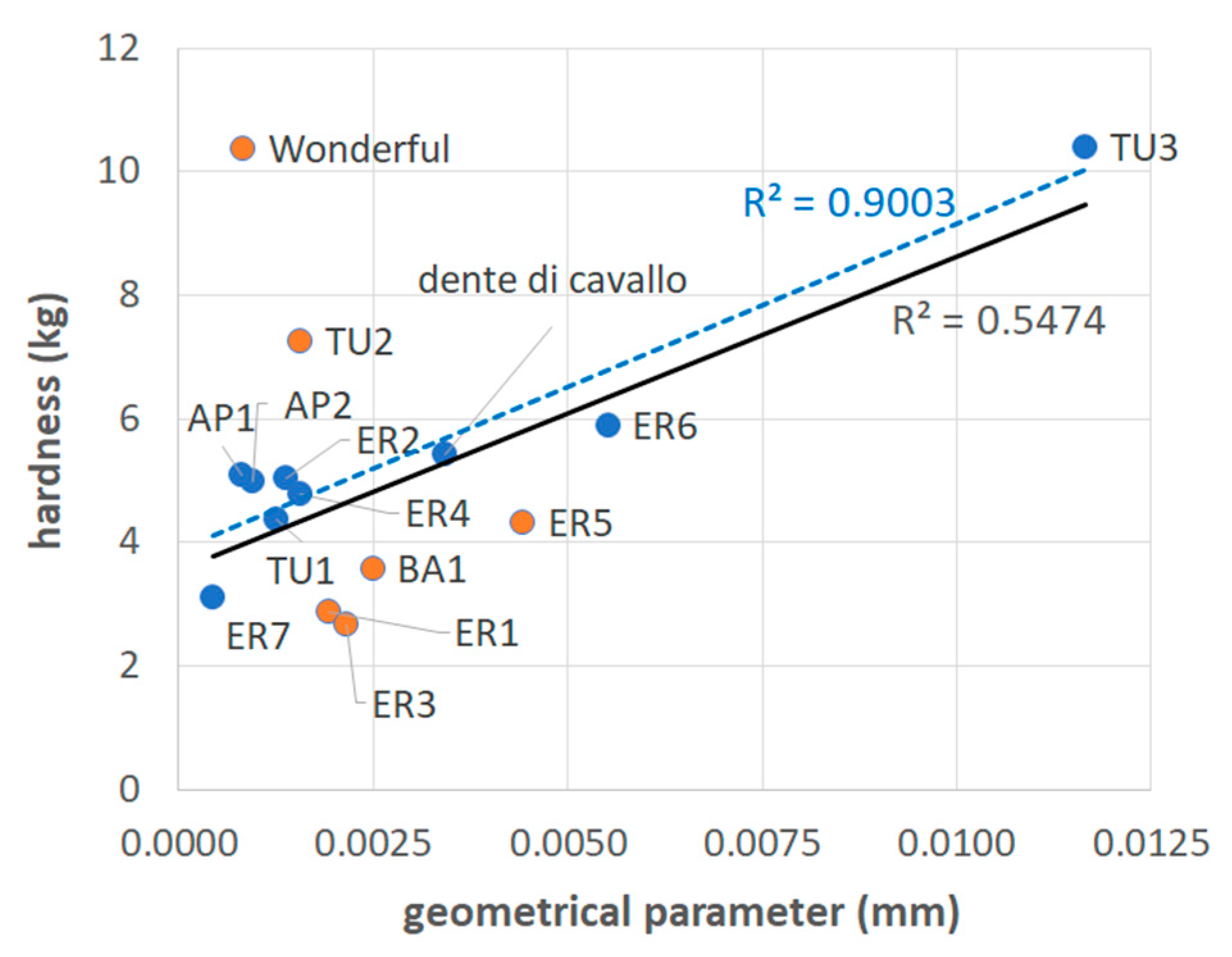
- The existence of the relationship shown in Figure 6 where only lignin is involved in the calculation of Veff implies that the other components, that is, proteins, extractives, ash and polysaccharides, do not appreciably contribute to mechanical strength in seeds, because none of them are structured enough to resist external stresses. The importance of lignin has been also highlighted by Dalimov et al. [12] and Xue et al. [46].
- −
- A hierarchical importance can be attributed to the factors considered in the present analysis, with the geometric characteristics (the thickness and diameter of the sclerotized layer) being the first parameters to be considered for seed hardness analysis. Therefore, a thicker and smaller seed (high value of the geometrical parameter) will be expectedly harder than a thin-walled large seed. In case unexpected values are obtained, a combination of lignin content and sclerotized integument volume should be also considered.
3.4. Oil Analysis
3.4.1. Fatty Acids Profile
3.4.2. Unsaponifiable Fraction Analysis
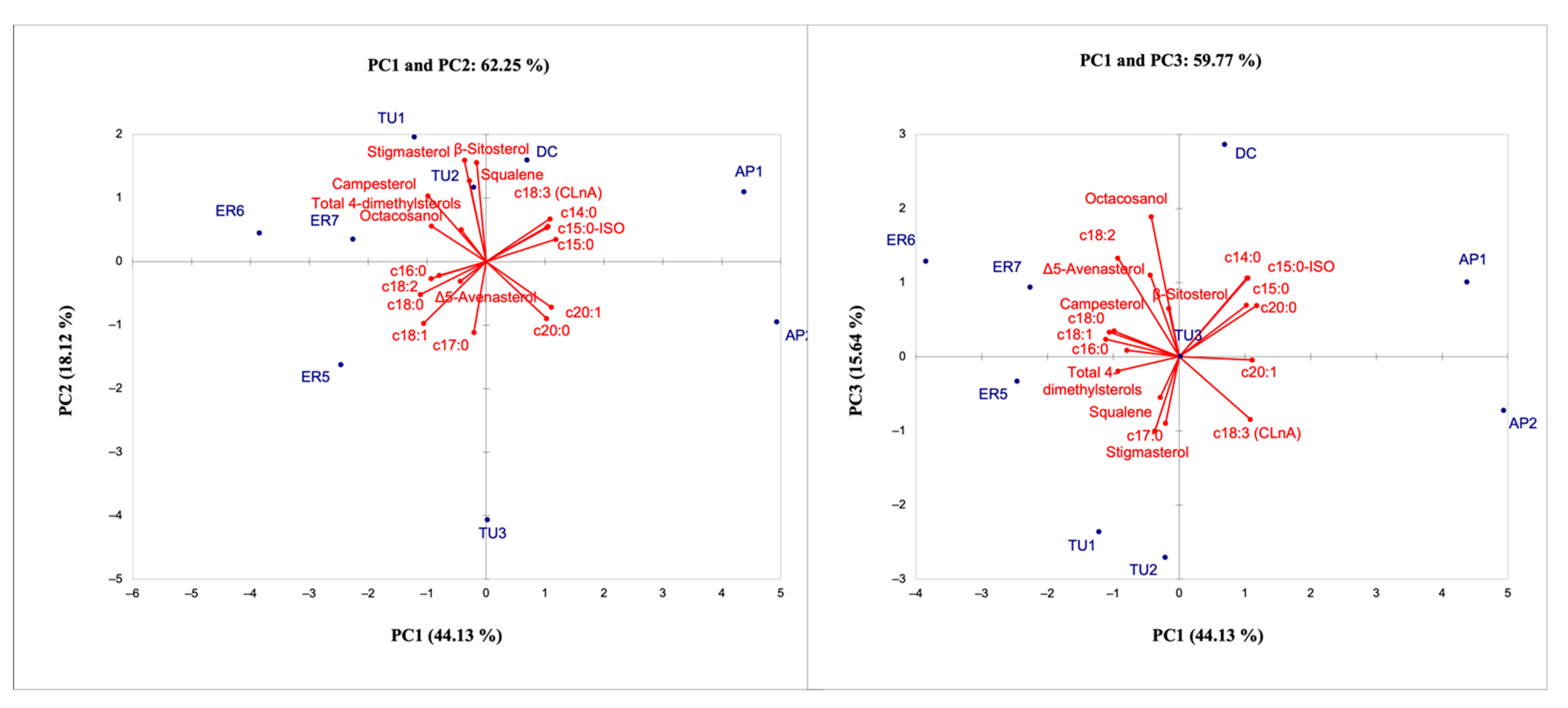
3.4.3. PCA Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tarantino, A.; Difonzo, G.; Disciglio, G.; Frabboni, L.; Paradiso, V.M.; Gambacorta, G.; Caponio, F. Fresh pomegranate juices from cultivars and local ecotypes grown in southeastern Italy: Comparison of physicochemical properties, antioxidant activity and bioactive compounds. J. Sci. Food Agric. 2022, 102, 1185–1192. [Google Scholar] [CrossRef] [PubMed]
- Beghè, D.; Cirlini, M.; Beneventi, E.; Miroslav, Č.; Tatjana, P.; Ganino, T.; Dall’Asta, C. Volatile profile of Italian and Montenegrine pomegranate juices for geographical origin classification. Eur. Food Res. Technol. 2021, 247, 211–220. [Google Scholar] [CrossRef]
- Beghè, D.; Fabbri, A.; Petruccelli, R.; Marieschi, M.; Torelli, A.; Ganino, T. Morphological and molecular characterization of ancient pomegranate (Punica granatum L.) accessions in Northern Italy. Adv. Hortic. Sci. 2019, 33, 581–592. [Google Scholar]
- Marieschi, M.; Torelli, A.; Beghè, D.; Bruni, R. Authentication of Punica granatum L.: Development of SCAR markers for the detection of 10 fruits potentially used in economically motivated adulteration. Food Chem. 2016, 202, 438–444. [Google Scholar] [CrossRef] [PubMed]
- Melgarejo, P.; Núñez-Gómez, D.; Legua, P.; Martínez-Nicolás, J.J.; Almansa, M.S. Pomegranate (Punica granatum L.) a dry pericarp fruit with fleshy seeds. Trends Food Sci. Technol. 2020, 102, 232–236. [Google Scholar] [CrossRef]
- Calani, L.; Beghè, D.; Mena, P.; Del Rio, D.; Bruni, R.; Fabbri, A.; Dall’Asta, C.; Galaverna, G. Ultra-HPLC–MS n (poly) phenolic profiling and chemometric analysis of juices from ancient Punica granatum L. cultivars: A nontargeted approach. J. Agric. Food Chem. 2013, 61, 5600–5609. [Google Scholar] [CrossRef]
- El Moujahed, S.; Dinica, R.M.; Cudalbeanu, M.; Avramescu, S.M.; Msegued Ayam, I.; Ouazzani Chahdi, F.; Rodi, Y.K.; Errachidi, F. Characterizations of Six Pomegranate (Punica granatum L.) Varieties of Global Commercial Interest in Morocco: Pomological, Organoleptic, Chemical and Biochemical Studies. Molecules 2022, 27, 3847. [Google Scholar] [CrossRef]
- Pujari, K.H.; Rane, D.A. Concept of seed hardness in pomegranate-I) Anatomical studies in soft and hard seeds of ‘Muskat’ pomegranate. In Proceedings of the 3rd International Symposium on Pomegranate and Minor Mediterranean Fruits, Tai’an, China, 20 September 2013; pp. 97–104. [Google Scholar]
- Prakash, N. Sexual Reproduction in Seed Plants. An Embryological Approach; University of New England: Arminade, Australia, 1986; p. 84. [Google Scholar]
- Tozzi, F.; Núñez-Gómez, D.; Legua, P.; Del Bubba, M.; Giordani, E.; Melgarejo, P. Qualitative and varietal characterization of pomegranate peel: High-value co-product or waste of production? Sci. Hortic. 2022, 291, 110601. [Google Scholar] [CrossRef]
- Trujillo, I.; Ojeda, M.A.; Urdiroz, N.M.; Potter, D.; Barranco, D.; Rallo, L.; Diez, C.M. Identification of the Worldwide Olive Germplasm Bank of Córdoba (Spain) using SSR and morphological markers. Tree Genet. Genomes 2014, 10, 141–155. [Google Scholar] [CrossRef]
- Dalimov, D.N.; Dalimova, G.N.; Bhatt, M. Chemical composition and lignins of tomato and pomegranate seeds. Chem. Nat. Compd. 2003, 39, 37–40. [Google Scholar] [CrossRef]
- Xia, X.; Li, H.; Cao, D.; Luo, X.; Yang, X.; Chen, L.; Liu, B.; Wang, Q.; Jin, D.; Cao, S. Characterization of a NAC transcription factor involved in the regulation of pomegranate seed hardness (Punica granatum L.). Plant Physiol. Biochem. 2019, 139, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Patil, P.G.; Singh, N.V.; Bohra, A.; Jamma, S.; Manjunatha, N.; Karuppannan, D.B.; Venkatesh, F.C.; Sharma, J.; Marathe, R.A. Novel miRNA-SSRs for improving seed hardness trait of pomegranate (Punica granatum L.). Front. Genet. 2022, 13, 866504. [Google Scholar] [CrossRef] [PubMed]
- Martinez, J.J.; Melgarejo, P.; Hernández, F.A.; Salazar, D.M.; Martinez, R. Seed characterisation of five new pomegranate (Punica granatum L.) varieties. Sci. Hortic. 2006, 110, 241–246. [Google Scholar] [CrossRef]
- Zarei, A.; Zamani, Z.; Fatahi, R.; Mousavi, A.; Salami, S.A. A mechanical method of determining seed-hardness in pomegranate. J. Crop Improv. 2013, 27, 444–459. [Google Scholar] [CrossRef]
- Zarei, A.; Zamani, Z.; Fatahi, R.; Mousavi, A.; Salami, S.A.; Avila, C.; Cánovas, F.M. Differential expression of cell wall related genes in the seeds of soft-and hard-seeded pomegranate genotypes. Sci. Hortic. 2016, 205, 7–16. [Google Scholar] [CrossRef]
- Alcaraz-Mármol, F.; Nuncio-Jáuregui, N.; Calín-Sánchez, Á.; Carbonell-Barrachina, Á.A.; Martínez, J.J.; Hernández, F. Determination of fatty acid composition in arils of 20 pomegranates cultivars grown in Spain. Sci. Hortic. 2015, 197, 712–718. [Google Scholar] [CrossRef]
- Montefusco, A.; Durante, M.; Migoni, D.; de Caroli, M.; Ilahy, R.; Pék, Z.; Helyes, L.; Fanizzi, F.P.; Mita, G.; Piro, G.; et al. Analysis of the phytochemical composition of pomegranate fruit juices, peels and kernels: A comparative study on four cultivars grown in southern Italy. Plants 2021, 10, 2521. [Google Scholar] [CrossRef]
- Caligiani, A. Pomegranate seed oil conjugated linoleic acids: Characterization and health effects. In Pomegranate: Chemistry, Processing and Health Benefits; Caligiani, A., Ed.; Nova Science Publishers: New York, NY, USA, 2016; pp. 105–120. [Google Scholar]
- Liu, N.; Ren, G.; Faiza, M.; Li, D.; Cui, J.; Zhang, K.; Yao, X.; Zhao, M. Comparison of conventional and green extraction methods on oil yield, physicochemical properties, and lipid compositions of pomegranate seed oil. J. Food Compos. Anal. 2022, 114, 104747. [Google Scholar] [CrossRef]
- Caligiani, A.; Bonzanini, F.; Palla, G.; Cirlini, M.; Bruni, R. Characterization of a potential nutraceutical ingredient: Pomegranate (Punica granatum L.) seed oil unsaponifiable fraction. Plant Foods Hum. Nutr. 2010, 65, 277–283. [Google Scholar] [CrossRef]
- Fernandes, L.; Pereira, J.A.; Lopéz-Cortés, I.; Salazar, D.M.; Ramalhosa, E.; Casal, S. Fatty acid, vitamin E and sterols composition of seed oils from nine different pomegranate (Punica granatum L.) cultivars grown in Spain. J. Food Compos. Anal. 2015, 39, 13–22. [Google Scholar] [CrossRef]
- Melgarejo-Sánchez, P.; Nunez-Gomez, D.; Martínez-Nicolás, J.J.; Hernández, F.; Legua, P.; Melgarejo, P. Pomegranate variety and pomegranate plant part, relevance from bioactive point of view: A review. Bioresour. Bioprocess. 2021, 8, 2. [Google Scholar] [CrossRef]
- Bonzanini, F.; Bruni, R.; Palla, G.; Serlataite, N.; Caligiani, A. Identification and distribution of lignans in Punica granatum L. fruit endocarp, pulp, seeds, wood knots and commercial juices by GC–MS. Food Chem. 2009, 117, 745–749. [Google Scholar] [CrossRef]
- International Union for the Protection of New Varieties of Plants. Guidelines for the Conduct of Tests for Distinctness, Uniformity and Stability: Pomegranate; International Union for the Protection of New Varieties of Plants: Geneva, Switzerland, 2013. [Google Scholar]
- Hernández, F.; Legua, P.; Martínez, R.; Melgarejo, P.; Martínez, J.J. Fruit quality characterization of seven pomegranate accessions (Punica granatum L.) grown in Southeast of Spain. Sci. Hortic. 2014, 175, 174–180. [Google Scholar] [CrossRef]
- Calín-Sánchez, Á.; Cano-Lamadrid, M.; Alcaraz-Mármol, F.; García-Sánchez, F.; Hernández, F.; Martínez-Nicolás, J.J. A new combined sensory-instrumental tool for pomegranate seed hardness determination. J. Sci. Food Agric. 2021, 101, 1355–1363. [Google Scholar] [CrossRef]
- TAPPI T 211 om-02; Ash in Wood, Pulp, Paper and Paperboard: Combustion at 525 °C. Technical Association of the Pulp and Paper Industry (TAPPI):: Peachtree Corners, GA, USA, 2002.
- TAPPI T 222 om-02; Acid Insoluble Lignin in Wood and Pulp. Technical Association of the Pulp and Paper Industry (TAPPI): Peachtree Corners, GA, USA, 2002.
- Santi, C.A.; Cortes, S.; D’Acqui, L.P.; Sparvoli, E.; Pushparaj, B. Reduction of organic pollutants in olive mill wastewater by using different mineral substrates as adsorbents. Bioresour. Technol. 2008, 99, 1945–1951. [Google Scholar] [CrossRef]
- Mariotti, F.; Tomé, D.; Mirand, P.P. Converting nitrogen into protein—beyond 6.25 and Jones’ factors. Crit. Rev. Food Sci. Nutr. 2008, 48, 177–184. [Google Scholar] [CrossRef]
- ISO 12966-2:2017; Animal and Vegetable Fats and OILS–Gas Chromatography of Fatty Acid Methyl Esters-Part 2: Preparation of Methyl Esters of Fatty Acids. International Organization for Standardization: Geneva, Switzerland, 2017.
- ISO 12228-1:2014; Determination of Individual and Total Sterols Contents–Gas Chromatographic Method–Part 1: Animal and Vegetable Fats and Oils. International Organization for Standardization: Geneva, Switzerland, 2014.
- Ferrara, G.; Cavoski, I.; Pacifico, A.; Tedone, L.; Mondelli, D. Morpho-pomological and chemical characterization of pomegranate (Punica granatum L.) genotypes in Apulia region, Southeastern Italy. Sci. Hortic. 2011, 130, 599–606. [Google Scholar] [CrossRef]
- Barone, E.; Sottile, F.; Caruso, T.; Marra, F.P. Preliminary Observations on Some Sicilian pomegranate (Punica granatum l.) Varieties. In Production, Processing and Marketing of Pomegranate in the Mediterranean Region: Advances in Research and Technology; Melgarejo, P., Martínez-Nicolás, J.J., Martínez-Tomé, J., Ciheam, Z., Eds.; Centre International de Hautes Etudes Agronomiques Méditerranéennes: Paris, France, 2000; pp. 137–141. [Google Scholar]
- Chandra, R.; Babu, K.D.; Jadhav, V.T.; Jaime, A.; Silva, T.D. Origin, history and domestication of pomegranate. Fruit Veg. Cereal Sci. Biotechnol. 2010, 2, 1–6. [Google Scholar]
- Damigella, P. La coltura del melograno nella Sicilia orientale. Tec. Agric. 1960, 6, 1–15. [Google Scholar]
- Talekar, S.; Patti, A.F.; Singh, R.; Vijayraghavan, R.; Arora, A. From waste to wealth: High recovery of nutraceuticals from pomegranate seed waste using a green extraction process. Ind. Crops Prod. 2018, 112, 790–802. [Google Scholar] [CrossRef]
- Pizzo, B.; Pometti, C.L.; Charpentier, J.P.; Boizot, N.; Saidman, B.O. Relationships involving several types of extractives of five native argentine wood species of genera Prosopis and Acacia. Ind. Crops Prod. 2011, 34, 851–859. [Google Scholar] [CrossRef]
- Xue, H.; Cao, S.; Li, H.; Zhang, J.; Niu, J.; Chen, L.; Zhao, D. De novo transcriptome assembly and quantification reveal differentially expressed genes between soft-seed and hard-seed pomegranate (Punica granatum L.). PLoS ONE 2017, 12, e0178809. [Google Scholar] [CrossRef] [PubMed]
- Melgarejo, P.; Sánchez, M.; Hernández, F.; Martínez, J.J.; Amorós, A. Parameters for determining the hardness and pleasantness of pomegranates (Punica granatum L.). Options Méditerranéennes Série A Séminaires Méditerranéens 2000, 42, 225–230. [Google Scholar]
- Wang, C.H.; Mai, Y.W. Deformation and fracture of Macadamia nuts. Part 2: Microstructure and fracture mechanics analysis of nutshell. Int. J. Fract. 1994, 69, 67–85. [Google Scholar] [CrossRef]
- Lu, Y.; Carroll, J.D.; Long, K.N.; Long, R. Failure of brittle micro-spherical shells embedded in elastomer matrix under indentation. Compos. Part B Eng. 2019, 173, 106870. [Google Scholar] [CrossRef]
- Young, W.C.; Budynas, R.C. Roark’s Formulas for Stress and Strain, 7th ed.; McGraw-Hill: New York, NY, USA, 2002. [Google Scholar]
- Xue, H.; Cao, S.Y.; Li, H.; Zhang, J.; Niu, J.; Chen, L.; Zhao, D. De novo assembly and quantification reveal differentially expressed genes between soft-seed and hard-seed pomegranate (Punica granatum L.). In II Asian Horticultural Congress; ISHS Acta Horticulturae: Chengdu, China, 2016; Volume 1, pp. 131–146. [Google Scholar]
- Kýralan, M.; Gölükcü, M.; Tokgöz, H. Oil and conjugated linolenic acid contents of seeds from important pomegranate cultivars (Punica granatum L.) grown in Turkey. J. Am. Oil Chem. Soc. 2009, 86, 985–990. [Google Scholar] [CrossRef]
- Ferrara, G.; Giancaspro, A.; Mazzeo, A.; Giove, S.L.; Matarrese, A.M.S.; Pacucci, C.; Punzi, R.; Trani, A.; Gambacorta, G.; Blanco, A.; et al. Characterization of pomegranate (Punica granatum L.) genotypes collected in Puglia region, Southeastern Italy. Sci. Hortic. 2014, 178, 70–78. [Google Scholar] [CrossRef]
- Khoddami, A.; Man, Y.B.C.; Roberts, T.H. Physico-chemical properties and fatty acid profile of seed oils from pomegranate (Punica granatum L.) extracted by cold pressing. Eur. J. Lipid Sci. Technol. 2014, 116, 553–562. [Google Scholar] [CrossRef]
- Paul, A.; Radhakrishnan, M. Pomegranate seed oil in food industry: Extraction, characterization, and applications. Trends Food Sci. Technol. 2020, 105, 273–283. [Google Scholar] [CrossRef]
- Habibnia, M.; Ghavami, M.; Ansaripour, M.; Vosough, S. Chemical evaluation of oils extracted from five different varieties of Iranian pomegranate seeds. J. Food Sci. Technol. 2012, 35–40. [Google Scholar]
- Fadavi, A.; Barzegar, M.; Azizi, M.H. Determination of fatty acids and total lipid content in oilseed of 25 pomegranates varieties grown in Iran. J. Food Compos. Anal. 2006, 19, 676–680. [Google Scholar] [CrossRef]
- Zhang, T.; Xie, L.; Liu, R.; Chang, M.; Zhang, H.; Jin, Q.; Wang, X. Revisiting the 4, 4-dimethylsterols profile from different kinds of vegetable oils by using GC-MS. LWT 2020, 124, 109163. [Google Scholar] [CrossRef]
- Verardo, V.; Garcia-Salas, P.; Baldi, E.; Segura-Carretero, A.; Fernandez-Gutierrez, A.; Caboni, M.F. Pomegranate seeds as a source of nutraceutical oil naturally rich in bioactive lipids. Food Res. Int. 2014, 65, 445–452. [Google Scholar] [CrossRef]
- Zhou, Y.; Cao, F.; Luo, F.; Lin, Q. Octacosanol and health benefits: Biological functions and mechanisms of action. Food Biosci. 2022, 101632. [Google Scholar] [CrossRef]
- Costa, A.M.M.; Silva, L.O.; Torres, A.G. Chemical composition of commercial cold-pressed pomegranate (Punica granatum) seed oil from Turkey and Israel, and the use of bioactive compounds for samples’ origin preliminary discrimination. J. Food Compos. Anal. 2019, 75, 8–16. [Google Scholar] [CrossRef]

| ID | Accession Name | Location |
|---|---|---|
| ER1 | Venturini | Parma (Emilia Romagna, Italy) |
| ER2 | Lago Costanza | Parma (Emilia Romagna, Italy) |
| ER3 | Reggiolo | Parma (Emilia Romagna, Italy) |
| ER4 | Marzapello | Parma (Emilia Romagna, Italy) |
| ER5 | Ecotype1 Stuard16 | Parma (Emilia Romagna, Italy) |
| ER6 | Ecotype2 Stuard1 | Parma (Emilia Romagna, Italy) |
| ER7 | Ecotype3 Stuard2 | Parma (Emilia Romagna, Italy) |
| TU1 | Melograno di Firenze | Firenze (Tuscany, Italy) |
| TU2 | Melograno di Buti | Pisa (Tuscany, Italy) |
| TU3 | Melograno di Lucca | Lucca (Tuscany, Italy) |
| AP1 | Precoce di Puglia | Lecce (Apulia, Italy) |
| AP2 | Tardiva di Puglia | Lecce (Apulia, Italy) |
| BA1 | Melograno di Matera | Matera (Basilicata, Italy) |
| DC | Dente di Cavallo | Catania (Sicily, Italy) |
| W | Wonderful | Catania (Sicily, Italy) |
| Genotype | Length (mm) | Width (mm) | Length/ Width | Perimeter (mm) | Weight (g) | Wood Index (%) |
|---|---|---|---|---|---|---|
| ER1 | 6.1 ± 0.7 fg | 2.9 ± 0.2 bc | 2.1 ± 0.3 g | 15.4 ± 1.2 cd | 0.028 ± 0.001 c–e | 7.6 ± 0.4 b–e |
| ER2 | 7.2 ± 0.6 a | 3.5 ± 0.2 a | 2.1 ± 0.2 g | 15.2 ± 2.0 cd | 0.025 ± 0.002 d–f | 8.2 ± 0.2 b–d |
| ER3 | 6.2 ± 0.4 ef | 2.6 ± 0.3 de | 2.4 ± 0.2 c–f | 16.0 ± 1.1 bc | 0.029 ± 0.004 c | 11.8 ± 0.1 a |
| ER4 | 6.9 ± 0.4 a–c | 2.9 ± 0.2 bc | 2.3 ± 0.2 d–f | 14.3 ± 0.9 d | 0.017 ± 0.001 g | 11.9 ± 0.1 a |
| ER5 | 6.2 ± 0.3 e–f | 2.2 ± 0.2 f | 2.8 ± 0.3 ab | 16.3 ± 1.8 bc | 0.028 ± 0.004 c–e | 11.7 ± 0.4 a |
| ER6 | 5.6 ± 0.2 h | 2.3 ± 0.2 f | 2.5 ± 0.2 cd | 18.9 ± 1.0 a | 0.026 ± 0.004 d–f | 6.2 ± 0.4 e–g |
| ER7 | 5.3 ± 0.2 h | 2.4 ± 0.2 ef | 2.2 ± 0.2 d–g | 14.9 ± 0.7 cd | 0.028 ± 0.004 cd | 7.4 ± 0.2 c–f |
| TU1 | 6.7 ± 0.5 b–d | 3.1 ± 0.7 b | 2.1 ± 0.4 e–g | 17.1 ± 1.5 b | 0.024 ± 0.004 f | 6.1 ± 1.0 f–g |
| TU2 | 5.6 ± 0.4 h | 2.6 ± 0.2 de | 2.2 ± 0.3 e–g | 11.0 ± 0.5 e | 0.015 ± 0.004 g | 7.0 ± 0.9 d–f |
| TU3 | 5.7 ± 0.6 gh | 2.4 ± 0.3 ef | 2.4 ± 0.3 c–e | 14.0 ± 2.1 d | 0.016 ± 0.002 g | 7.5 ± 0.7 b–f |
| AP1 | 6.9 ± 0.5 a–c | 2.6 ± 0.2 de | 2.6 ± 0.3 bc | 16.2 ± 0.7 bc | 0.017 ± 0.002 g | 4.2 ± 0.5 h |
| AP2 | 6.3 ± 0.6 e–f | 3.0 ± 0.2 bc | 2.2 ± 0.2 fg | 16.2 ± 1.3 bc | 0.021 ± 0.003 fg | 4.8 ± 0.7 gh |
| BA1 | 7.1 ± 0.5 ab | 2.5 ± 0.3 d–f | 2.9 ± 0.4 a | 19.9 ± 1.0 a | 0.025 ± 0.004 ef | 8.8 ± 0.6 bc |
| DC | 6.6 ± 0.3 d–f | 2.7 ± 0.2 cd | 2.4 ± 0.2 c–e | 17.2 ± 0.8 b | 0.038 ± 0.004 a | 7.3 ± 0.6 d–f |
| W | 7.1 ± 0.3 ab | 3.6 ± 0.3 a | 2.0 ± 0.2 g | 19.7 ± 1.1 a | 0.032 ± 0.002 b | 8.9 ± 0.8 b |
| Genotype | OE (%) | AE (%) | L (%) | A (%) | P (%) | OS (%) | T (µm) | Geometrical Parameter (mm) | Hardness (Kg/cm2) |
|---|---|---|---|---|---|---|---|---|---|
| ER1 | 10.5 ± 0.5 f | 2.7 ± 0.1 d–f | 26.1 ± 0.1 cd | 1.0 ± 0.1 d | 9.2 ± 0.3 ij | 50.6 ± 0.2 a | 419.7 ± 30.1 ab | 0.0019 ± 0.00 cd | 2.88 ± 0.16 fg |
| ER2 | 23.7 ± 1.1 ab | 4.4 ± 0.2 bc | 19.2 ± 0.6 g | 0.9 ± 0.07 d | 18.3 ± 0.2 e | 33.8 ± 0.1 e | 445.8 ± 32.6 ab | 0.0014 ± 0.00 de | 5.05 ± 0.35 c–e |
| ER3 | 18.8 ± 1.0 cd | 8.3 ± 0.2 a | 16.6 ± 0.2 h | 0.6 ± 0.1 d | 17.0 ± 0.1 f | 38.8 ± 0.09 d | 394.5 ± 27.4 a-c | 0.0022 ± 0.00 cd | 2.68 ± 0.21 g |
| ER4 | 8.5 ± 0.6 fg | 5.4 ± 0.3 b | 29.3 ± 0.3 b | 0.8 ± 0.1 d | 10.7 ± 0.2 h | 45.6 ± 0.2 b | 399.5 ± 30.2 a-c | 0.0016 ± 0.00 d | 4.79 ± 0.29 c–e |
| ER5 | 13.1 ± 0.4 e | 1.6 ± 0.2 fg | 29.6 ± 0.4 b | 1.0 ± 0.09 d | 23.4 ± 0.2 c | 31.6 ± 0.2 f | 403.7 ± 28.2 ab | 0.0044 ± 0.00 b | 4.34 ± 0.42 d–g |
| ER6 | 8.8 ± 0.5 fg | 4.8 ± 0.1 bc | 24.8 ± 0.3 de | 0.7 ± 0.1 d | 19.9 ± 0.1 d | 41.1 ± 0.1 c | 432.6 ± 25.4 ab | 0.0055 ± 0.00 b | 5.90 ± 0.36 bc |
| ER7 | 18.1 ± 0.6 d | 4.9 ± 0.2 bc | 26.6 ± 0.2 c | 1.0 ± 0.2 d | 22.6 ± 0.2 c | 27.0 ± 0.2 h | 256.1 ± 16.4 d | 0.0004 ± 0.00 f | 3.11 ± 0.29 fg |
| TU1 | 24.5 ± 0.4 ab | 1.4 ± 0.2 fg | 17.6 ± 0.2 h | 1.9 ± 0.09 c | 8.6 ± 0.2 ij | 46.2 ± 0.1 b | 397.4 ± 28.2 a–c | 0.0013 ± 0.00 de | 4.37 ± 0.72 d–f |
| TU2 | 17.1 ± 0.5 d | 0.3 ± 0.3 g | 24.1 ± 0.3 e | 1.6 ± 0.1 cd | 27.7 ± 0.1 a | 29.7 ± 0.2 g | 362.5 ± 24.6 bc | 0.0016 ± 0.00 d | 7.26 ± 0.27 b |
| TU3 | 7.1 ± 0.6 g | 1.2 ± 0.1 fg | 31.1 ± 0.3 a | 0.9 ± 0.1 d | 9.8 ± 0.2 hi | 50.0 ± 0.3 a | 514.1 ± 24.6 a | 0.0117 ± 0.00 a | 10.41 ± 0.37 a |
| AP1 | 22.9 ± 0.3 b | 3.6 ± 0.2 cd | 20.3 ± 0.2 fg | 2.6 ± 0.1 c | 27.2 ± 0.1 ab | 24.1 ± 0.1 j | 317.6 ± 36.8 c | 0.0008 ± 0.00 ef | 5.09 ± 0.47 c–e |
| AP2 | 20.6 ± 0.4 c | 4.3 ± 0.1 bc | 21.6 ± 0.3 f | 2.0 ± 0.1 c | 9.2 ± 0.3 ij | 42.7 ± 0.2 c | 359.8 ± 22.3 bc | 0.0010 ± 0.00 de | 4.93 ± 0.29 c–e |
| BA1 | 25.2 ± 0.4 a | 1.9 ± 0.1 e–g | 19.5 ± 0.3 g | 1.8 ± 0.07 c | 26.2 ± 0.1 b | 25.8 ± 0.1 i | 387.5 ± 25.1 a-c | 0.0025 ± 0.00 cd | 3.58 ± 0.25 e–g |
| DC | 22.9 ± 0.3 b | 4.6 ± 0.3 c–e | 19.4 ± 0.2 g | 2.9 ± 0.2 b | 6.8 ± 0.1 g | 43.6 ± 0.2 bc | 450.3 ± 43.7 ab | 0.0034 ± 0.00 bc | 5.43 ± 0.51 cd |
| W | 25.6 ± 0.3 a | 4.6 ± 0.1 bc | 16.5 ± 0.2 h | 7.7 ± 0.2 a | 12.6 ± 0.2 g | 33.9 ± 0.2 e | 402.0 ± 33.5 ab | 0.0008 ± 0.00 ef | 10.37 ± 0.61 a |
| Genotype | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| ER5 | ER6 | ER7 | TU1 | TU2 | TU3 | AP1 | AP2 | DC | |
| Lipid Content | 16 ± 3 a,b | 17 ± 1 b | 15 ± 3 a | 14 ± 1 a | 22.7 ± 0.8 d | 19 ± 1 c | 14 ± 1 a | 25 ± 1 e | 21 ± 2 c |
| c14:0 | 0.018 ± 0.001 a | n.d. | 0.006 ± 0.001 a | n.d. | 0.03 ± 0.01 a | 0.027 ± 0.007 a | 0.33 ± 0.03 b | 0.18 ± 0.02 b | 1.6 ± 0.2 c |
| c15:0-iso | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.22 ± 0.02 a | 0.16 ± 0.02 a | 0.9 ± 0.1 b |
| c15:0 | n.d. | n.d. | 0.004 ± 0.001 a | n.d. | 0.014 ± 0.003 a | 0.013 ± 0.005 a | 0.20 ± 0.02 b | 0.15 ± 0.01 b | 0.7 ± 0.1 c |
| c16:0 | 7.2 ± 0.5 d | 5.8 ± 0.1 c | 5.1 ± 0.7 c | 4.7 ± 0.2 b,c | 3.2 ± 0.1 a | 3.5 ± 0.5 a | 4.2 ± 0.4 b | 3.5 ± 0.3 a | 12 ± 1 e |
| c17:0 | 0.012 ± 0.001 a | 0.008 ± 0.001 a | 0.009 ± 0.001 a | 0.009 ± 0.001 a | 0.056 ± 0.006 b | 0.073 ± 0.007 c | n.d. | n.d. | n.d. |
| c18:0 | 3.5 ± 0.1 d | 3.6 ± 0.2 d | 2.9 ± 0.5 c | 3.3 ± 0.1 d | 2.3 ± 0.3 b | 3.3 ± 0.3 d | 2.1 ± 0.2 a | 2.0 ± 0.2 a | 6.2 ± 0.6 e |
| c18:1 | 8.7 ± 0.3 c | 10.7 ± 0.3 e | 7.7 ± 0.2 c | 5.4 ± 0.2 b | 4.2 ± 0.7 b | 9.1 ± 0.3 d | 2.9 ± 0.3 a | 3.1 ± 0.3 a | 7.7 ± 0.8 c |
| c18:2 | 8.5 ± 0.3 d | 11.72 ± 0.05 e | 9.2 ± 0.4 d | 4.7 ± 0.2 b | 5.0 ± 0.3 b | 8.2 ± 0.4 d | 3.0 ± 0.3 a | 3.7 ± 0.4 a | 7.0 ± 0.7 c |
| c18:3 (CLnA) | 71.8 ± 0.7 c | 68.0 ± 0.5 b | 74.9 ± 0.4 c | 81 ± 1 d | 85 ± 1 e | 74.9 ± 0.9 c | 86 ± 4 e | 86 ± 4 e | 63 ± 3 a |
| c20:0 | 0.08 ± 0.02 a | 0.05 ± 0.001 a | 0.059 ± 0.001 a | 0.062 ± 0.005 a | 0.04 ± 0.01 a | 0.43 ± 0.02 b | 0.37 ± 0.04 b | 0.41 ± 0.04 b | 1.3 ± 0.1 c |
| c20:1 | 0.08 ± 0.02 a | 0.05 ± 0.01 a | 0.057 ± 0.001 a | 0.064 ± 0.005 a | 0.04 ± 0.02 a | 0.43 ± 0.03 b | 0.67 ± 0.07 c | 0.83 ± 0.08 d | n.d. |
| ΣSFA | 11 c | 9.0 c | 8.2 b | 8.4 b | 6.0 a | 7.3 a | 7.5 a | 6.0 a | 22 d |
| ΣMUFA | 9.0 d | 11 e | 7.8 c | 5.6 b | 4.0 a | 9.7 d | 3.5 a | 4.0 a | 8.0 c |
| ΣPUFA | 80 b | 80 b | 84 c | 86 c | 90 d | 83 c | 89 d | 90 d | 70 a |
| Sterols | ER5 | ER6 | ER7 | TU1 | TU2 | TU3 | AP1 | AP2 | DC |
|---|---|---|---|---|---|---|---|---|---|
| Campesterol | 430 ± 40 | 655 ± 65 | 480 ± 48 | 484 ± 17 | 470 ± 50 | 320 ± 30 | 365 ± 40 | 304 ± 30 | 470 ± 50 |
| Stigmasterol | 180 ± 20 | 230 ± 20 | 245 ± 20 | 303 ± 15 | 265 ± 30 | 145 ± 10 | 194 ± 20 | 201 ± 20 | 210 ± 20 |
| β-Sitosterol | 3800 ± 40 | 3850 ± 190 | 3745 ± 40 | 4040 ± 19 | 3520 ± 180 | 1670 ± 20 | 2960 ± 150 | 1770 ± 90 | 5370 ± 27 |
| Δ5-Avenasterol | 200 ± 20 | 371 ± 37 | 580 ± 60 | 281 ± 8 | 250 ± 20 | 570 ± 60 | 365 ± 40 | 75 ± 7 | 440 ± 40 |
| Total 4-desmethylsterols | 1190 ± 120 | 5106 ± 312 | 5050 ± 168 | 5108 ± 59 | 4505 ± 280 | 2705 ± 110 | 3884 ± 250 | 2350 ± 147 | 6490 ± 38 |
| Total 4,4-dimethylsterols | 1760 ± 90 | 1185 ± 60 | 1420 ± 70 | 1224 ± 35 | 1820 ± 90 | 830 ± 80 | 631 ± 63 | 195 ± 20 | 1517 ± 75 |
| Octacosanol | 52 ± 7 | 70 ± 10 | 76 ± 7 | 22.2 ± 0.4 | 25 ± 1 | 30 ± 1 | 57 ± 6 | 24 ± 1 | 77 ± 7 |
| Squalene | 890 ± 90 | 1025 ± 50 | 830 ± 80 | 1173 ± 40 | 940 ± 90 | 725 ± 70 | 1100 ± 55 | 690 ± 70 | 800 ± 59 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beghè, D.; Giordano, C.; Pizzo, B.; Lolli, V.; Caligiani, A.; Bonzanini, F.; Anichini, M.; Fiorentino, L.; Rodolfi, M.; Ganino, T.; et al. Characterization of Pomegranate (Punica granatum L.) through the Morphoanatomical and Lipid Profile of Seeds for the Valorisation of Italian Accessions. Sustainability 2022, 14, 14780. https://doi.org/10.3390/su142214780
Beghè D, Giordano C, Pizzo B, Lolli V, Caligiani A, Bonzanini F, Anichini M, Fiorentino L, Rodolfi M, Ganino T, et al. Characterization of Pomegranate (Punica granatum L.) through the Morphoanatomical and Lipid Profile of Seeds for the Valorisation of Italian Accessions. Sustainability. 2022; 14(22):14780. https://doi.org/10.3390/su142214780
Chicago/Turabian StyleBeghè, Deborah, Cristiana Giordano, Benedetto Pizzo, Veronica Lolli, Augusta Caligiani, Francesca Bonzanini, Monica Anichini, Luigi Fiorentino, Margherita Rodolfi, Tommaso Ganino, and et al. 2022. "Characterization of Pomegranate (Punica granatum L.) through the Morphoanatomical and Lipid Profile of Seeds for the Valorisation of Italian Accessions" Sustainability 14, no. 22: 14780. https://doi.org/10.3390/su142214780
APA StyleBeghè, D., Giordano, C., Pizzo, B., Lolli, V., Caligiani, A., Bonzanini, F., Anichini, M., Fiorentino, L., Rodolfi, M., Ganino, T., & Petruccelli, R. (2022). Characterization of Pomegranate (Punica granatum L.) through the Morphoanatomical and Lipid Profile of Seeds for the Valorisation of Italian Accessions. Sustainability, 14(22), 14780. https://doi.org/10.3390/su142214780













