Fermentative Production of Erythritol from Cane Molasses Using Candida magnoliae: Media Optimization, Purification, and Characterization
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Culture Conditions for Measuring the Growth Curve
2.3. Preparation of Cell Extracts
2.4. Erythrose Reductase Assay
2.5. Fermentative Production of Erythritol
2.6. Response Surface Methodology
2.7. Numerical Optimization
2.8. Analysis of Fermentation Broth
2.9. Purification of Fermentation Broth
2.10. Statistical Analysis
3. Results and Discussion
3.1. Growth Curve of Candida magnoliae
3.2. Screening of Significant Factors Using Plackett–Burman Design
3.3. Response Surface Modeling of Erythritol Yield
3.4. Numerical Optimization of Fermentation Conditions
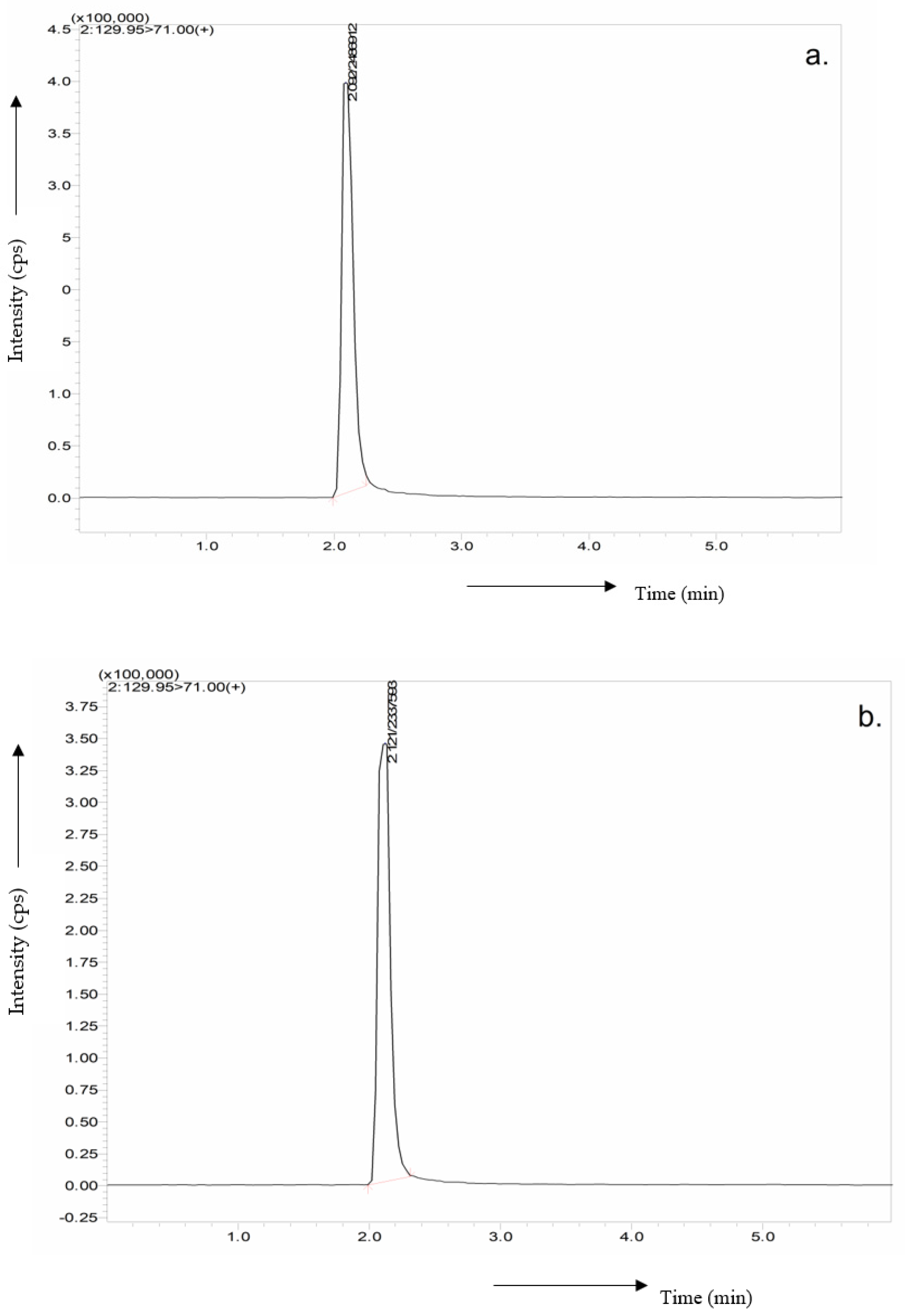
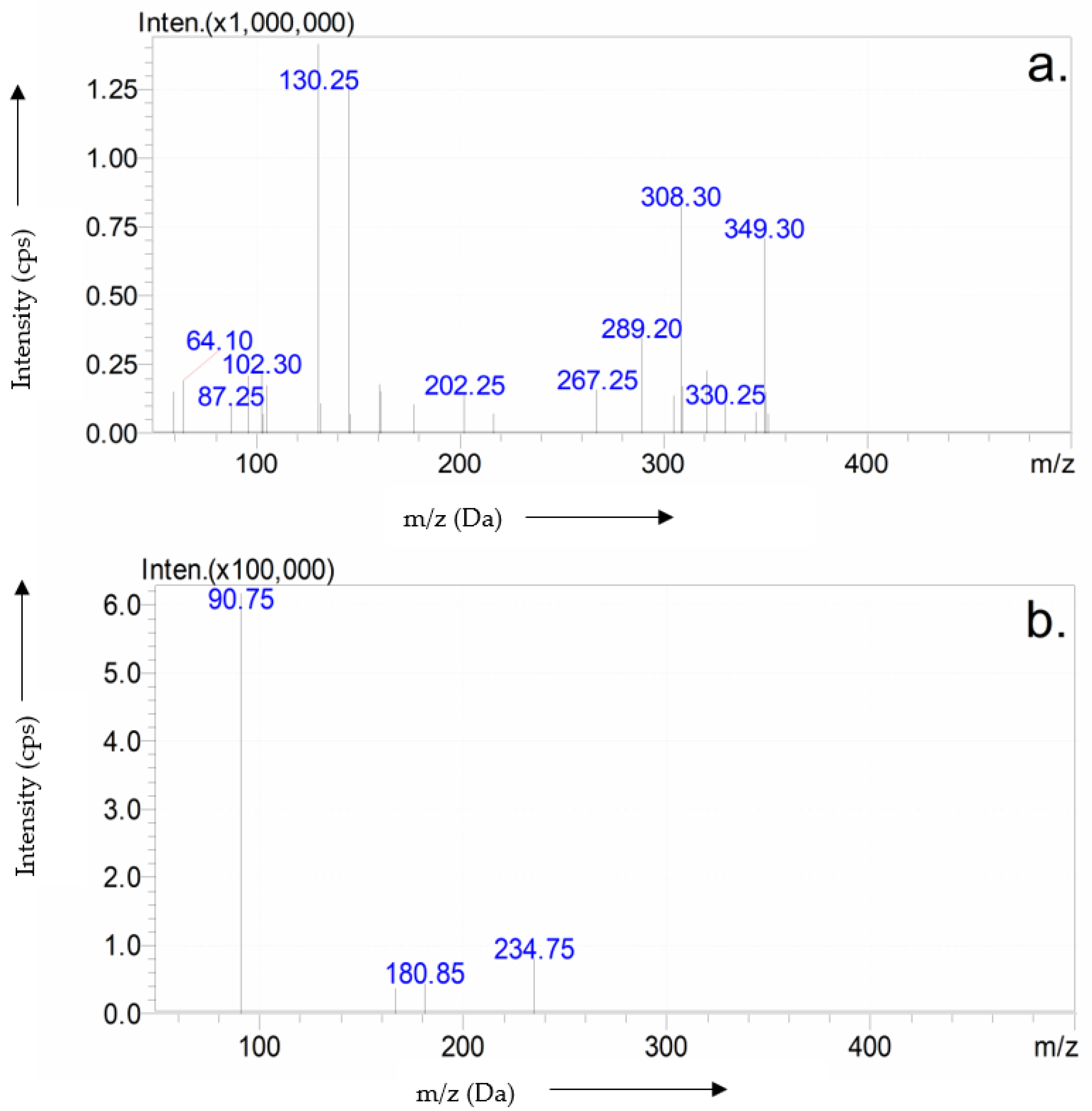
3.5. Purification and Characterization of Erythritol
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lee, K.J.; Lim, J.Y. Optimized conditions for high erythritol production by Penicillium sp. KJ-UV29, mutant of Penicillium sp. KJ81. Biotechnol. Bioprocess Eng. 2003, 8, 173–178. [Google Scholar] [CrossRef]
- Martău, G.A.; Coman, V.; Vodnar, D.C. Recent advances in the biotechnological production of erythritol and mannitol. Crit. Rev. Biotechnol. 2020, 40, 608–622. [Google Scholar] [CrossRef] [PubMed]
- Grembecka, M.; Lebiedzińska, A.; Szefer, P. Simultaneous separation and determination of erythritol, xylitol, sorbitol, mannitol, maltitol, fructose, glucose, sucrose, and maltose in food products by high performance liquid chromatography coupled to charged aerosol detector. Microchem. J. 2014, 117, 77–82. [Google Scholar] [CrossRef]
- Ahuja, K.; Rawat, A. Erythritol Market Trends 2020–2026: Global Industry Report; Global Market Insights Inc.: Selbyville, DE, USA, 2020; Available online: www.gminsights.com/industry-analysis/erythritol-market (accessed on 16 March 2020).
- Daza-Serna, L.; Serna-Loaiza, S.; Masi, A.; Mach, R.L.; Mach-Aigner, A.R.; Friedl, A. From the culture broth to the erythritol crystals: An opportunity for circular economy. Appl. Microbiol. Biotechnol. 2021, 105, 4467–4486. [Google Scholar] [CrossRef]
- Tomaszewska-Hetman, L.; Rymowicz, W.; Rywińska, A. Waste conversion into a sweetener—development of an innovative strategy for erythritol production by Yarrowia lipolytica. Sustainability 2020, 12, 7122. [Google Scholar] [CrossRef]
- Tomaszewska, L.; Rymowicz, W.; Rywińska, A. Mineral supplementation increases erythrose reductase activity in erythritol biosynthesis from glycerol by Yarrowia lipolytica. Appl. Biochem. Biotechnol. 2014, 172, 3069–3078. [Google Scholar] [CrossRef]
- Tomaszewska, L.; Rywińska, A.; Rymowicz, W. High selectivity of erythritol production from glycerol by Yarrowia lipolytica. Biomass Bioenergy 2014, 64, 309–320. [Google Scholar] [CrossRef]
- Yang, L.B.; Zhan, X.B.; Zheng, Z.Y.; Wu, J.R.; Gao, M.J.; Lin, C.C. A novel osmotic pressure control fed-batch fermentation strategy for improvement of erythritol production by Yarrowia lipolytica from glycerol. Bioresour. Technol. 2014, 151, 120–127. [Google Scholar] [CrossRef]
- Yu, J.H.; Lee, D.H.; Oh, Y.J.; Han, K.C.; Ryu, Y.W.; Seo, J.H. Selective utilization of fructose to glucose by Candida magnoliae, an erythritol producer. Appl. Biochem. Biotechnol. 2006, 131, 870–879. [Google Scholar] [CrossRef]
- Guo, J.; Li, J.; Chen, Y.; Guo, X.; Xiao, D. Improving erythritol production of Aureobasidium pullulans from xylose by mutagenesis and medium optimization. Appl. Biochem. Biotechnol. 2016, 180, 717–727. [Google Scholar] [CrossRef]
- Mirończuk, A.M.; Rakicka, M.; Biegalska, A.; Rymowicz, W.; Dobrowolski, A. A two-stage fermentation process of erythritol production by yeast Y. lipolytica from molasses and glycerol. Bioresour. Technol. 2015, 198, 445–455. [Google Scholar] [CrossRef] [PubMed]
- Rzechonek, D.A.; Dobrowolski, A.; Rymowicz, W.; Mirończuk, A.M. Recent advances in biological production of erythritol. Crit. Rev. Biotechnol. 2018, 38, 620–633. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, X.; Wang, Y.; Li, Q.; Yan, S.; He, N. The production of bioflocculants by Bacillus licheniformis using molasses and its application in the sugarcane industry. Biotechnol. Bioprocess Eng. 2012, 17, 1041–1047. [Google Scholar] [CrossRef]
- Clarke, M.A. Syrups. In Encyclopedia of Food Sciences and Nutrition, 2nd ed.; Academic Press: Cambridge, MA, USA, 2003; pp. 5711–5717. [Google Scholar] [CrossRef]
- Vignesh Kumar, B.; Muthumari, B.; Kavitha, M.; John Praveen Kumar, J.K.; Thavamurugan, S.; Arun, A.; Jothi Basu, M. Studies on optimization of sustainable Lactic Acid production by Bacillus amyloliquefaciens from sugarcane molasses through microbial fermentation. Sustainability 2022, 14, 7400. [Google Scholar] [CrossRef]
- Carly, F.; Fickers, P. Erythritol production by yeasts: A snapshot of current knowledge. Yeast 2018, 35, 455–463. [Google Scholar] [CrossRef]
- Savergave, L.S. Microbial Production of Erythritol and Mannitol: Strain Improvement and Process Optimization. Ph.D. Thesis, National Chemical Laboratory, Pune, India, 2011. [Google Scholar]
- Ghezelbash, G.R.; Nahvi, I.; Malekpour, A. Erythritol production with minimum By-product using Candida magnoliae mutant. Appl. Biochem. Microbiol. 2014, 50, 292–296. [Google Scholar] [CrossRef]
- Gassara, F.; Brar, S.K.; Tyagi, R.D.; John, R.P.; Verma, M.; Valero, J.R. Parameter optimization for production of ligninolytic enzymes using agro-industrial wastes by response surface method. Biotechnol. Bioprocess Eng. 2011, 16, 343–351. [Google Scholar] [CrossRef]
- Borkar, V.; Chakraborty, S.; Gokhale, J.S. Fermentative production of naringinase from Aspergillus niger van Tieghem MTCC 2425 using citrus wastes: Process optimization, partial purification, and characterization. Appl. Biochem. Biotechnol. 2021, 193, 1321–1337. [Google Scholar] [CrossRef]
- Chen, L.; Wei, X.; Chaves, B.D.; Jones, D.; Ponder, M.A.; Subbiah, J. Inactivation of Salmonella enterica and Enterococcus faecium NRRL B2354 on cumin seeds using gaseous ethylene oxide. Food Microbiol. 2021, 94, 103656. [Google Scholar] [CrossRef]
- Yan, Y.; Zhao, X.; Wang, C.; Fang, Q.; Zhong, L.; Wei, Q. Preparation, optimization, and characterization of inclusion complexes of Cinnamomum longepaniculatum essential oil in β-Cyclodextrin. Sustainability 2022, 14, 9513. [Google Scholar] [CrossRef]
- Ryu, Y.W.; Park, C.Y.; Park, J.B.; Kim, S.Y.; Seo, J.H. Optimization of erythritol production by Candida magnoliae in fed-batch culture. J. Ind. Microbiol. Biotechnol. 2000, 25, 100–103. [Google Scholar] [CrossRef]
- Koh, E.S.; Lee, T.H.; Lee, D.Y. Scale-up of erythritol production by an osmophilic mutant of Candida magnoliae. Biotechnol. Lett. 2003, 25, 2103–2105. [Google Scholar] [CrossRef] [PubMed]
- Savergave, L.S.; Gadre, R.V.; Vaidya, B.K.; Narayanan, K. Strain improvement and statistical media optimization for enhanced erythritol production with minimal by-products from Candida magnoliae mutant R23. Biochem. Eng. J. 2011, 55, 92–100. [Google Scholar] [CrossRef]
- Krohn, R.I. The colorimetric detection and quantitation of total protein. Curr. Protoc. Toxicol. 2005, 23, A-3I. [Google Scholar] [CrossRef]
- Kim, K.A.; Noh, B.S.; Lee, J.K.; Kim, S.Y.; Park, Y.C.; Oh, D.K. Optimization of culture conditions for erythritol production by Torula sp. J. Microbiol. Biotechnol. 2000, 10, 69–74. [Google Scholar]
- Chang, C.S.; Yeh, T.S. Detection of 10 sweeteners in various foods by liquid chromatography/tandem mass spectrometry. J. Food Drug Anal. 2014, 22, 318–328. [Google Scholar] [CrossRef]
- Hijosa-Valsero, M.; Garita-Cambronero, J.; Paniagua-García, A.; Diez Antolinez, R. By-products of sugar factories and wineries as feedstocks for erythritol generation. Food Bioprod. Process. 2021, 126, 345–355. [Google Scholar] [CrossRef]
- Park, J.; Seo, B.; Kim, J.; Park, Y. Production of erythritol in fed-batch cultures of Trichosporon sp. J. Ferment. Bioeng. 1998, 86, 577–580. [Google Scholar] [CrossRef]
- Deshpande, M.S.; Kulkarni, P.P.; Kumbhar, P.S.; Ghosalkar, A.R. Erythritol production from sugar based feedstocks by Moniliella pollinis using lysate of recycled cells as nutrients source. Process Biochem. 2022, 112, 45–52. [Google Scholar] [CrossRef]
- Rakicka, M.; Biegalska, A.; Rymowicz, W.; Dobrowolski, A.; Mirończuk, A.M. Polyol production from waste materials by genetically modified Yarrowia lipolytica. Bioresour. Technol. 2017, 243, 393–399. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, J.; Jiang, H. Microbial production of value-added bioproducts and enzymes from molasses, a by-product of sugar industry. Food Chem. 2021, 346, 128860. [Google Scholar] [CrossRef] [PubMed]
- Adeuya, A.; Price, N. Rapid characterization of polyalcohols by silylation and matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. Int. J. Devoted Rapid Dissem. Up-Minute Res. Mass Spectrom. 2007, 21, 3977–3981. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.B.; Zhan, X.B.; Zhu, L.; Gao, M.J.; Lin, C.C. Optimization of a low-cost hyperosmotic medium and establishing the fermentation kinetics of erythritol production by Yarrowia lipolytica from crude glycerol. Prep. Biochem. Biotechnol. 2016, 46, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.W.; Jeon, E.J.; Jung, I.H.; Min, M.J.; Lee, H.Y.; Kim, M.J.; Moon, T.W. Maltosyl-erythritol, a major transglycosylation product of erythritol by Bacillus stearothermophilus maltogenic amylase. Biosci. Biotechnol. Biochem. 2003, 67, 525–531. [Google Scholar] [CrossRef]
- Rakicka, M.; Rywińska, A.; Lazar, Z.; Rymowicz, W. Two-stage continuous culture–technology boosting erythritol production. J. Clean. Prod. 2017, 168, 420–427. [Google Scholar] [CrossRef]
- Forbes, T.P.; Sisco, E. Trace detection and competitive ionization of erythritol tetranitrate in mixtures using direct analysis in real time mass spectrometry. Anal. Methods 2015, 7, 3632–3636. [Google Scholar] [CrossRef]
- AOAC International. Official Methods of Analysis, 15th ed.; AOAC International: Gaithersburg, MD, USA, 1990; Volume 1. [Google Scholar]
- Daza-Serna, L.; Knežević, K.; Kreuzinger, N.; Mach-Aigner, A.R.; Mach, R.L.; Krampe, J.; Friedl, A. Recovery of salts from synthetic erythritol culture broth via electrodialysis: An alternative strategy from the bin to the loop. Sustainability 2022, 14, 734. [Google Scholar] [CrossRef]


| Run | Independent Variables (Coded Value) | Response | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| X1 | X2 | X3 | X4 | X5 | X6 | X7 | X8 | X9 | X10 | X11 | Y1 | Y2 | Y3 | |
| g·L−1 | g·L−1 | g·L−1 | g·L−1 | - | mL | °C | h | rpm | mL | mg·L−1 | g·L−1 | mg | - | |
| 1 | 300 | 12 | 2 | 0.5 | 7 | 3 | 25 | 48 | 180 | 30 | 80 | 37.2 ± 1.8 | 350 ± 3 | 4.6 ± 0.0 |
| 2 | 200 | 12 | 5 | 0.1 | 7 | 3 | 30 | 48 | 180 | 20 | 120 | 32.6 ± 1.5 | 451 ± 4 | 4.3 ± 0.1 |
| 3 | 300 | 9 | 5 | 0.5 | 4 | 3 | 30 | 96 | 180 | 20 | 80 | 105.7 ± 4.3 | 482 ± 4 | 3.0 ± 0.1 |
| 4 | 200 | 12 | 2 | 0.5 | 7 | 1 | 30 | 96 | 240 | 20 | 80 | 22.5 ± 1.5 | 733 ± 5 | 5.2 ± 0.0 |
| 5 | 200 | 9 | 5 | 0.1 | 7 | 3 | 25 | 96 | 240 | 30 | 80 | 35.1 ± 1.6 | 934 ± 8 | 4.2 ± 0.0 |
| 6 | 200 | 9 | 2 | 0.5 | 4 | 3 | 30 | 48 | 240 | 30 | 120 | 36.5 ± 1.7 | 592 ± 4 | 3.1 ± 0.0 |
| 7 | 300 | 9 | 2 | 0.1 | 7 | 1 | 30 | 96 | 180 | 30 | 120 | 96.6 ± 4.9 | 174 ± 2 | 4.6 ± 0.1 |
| 8 | 300 | 12 | 2 | 0.1 | 4 | 3 | 25 | 96 | 240 | 20 | 120 | 36.1 ± 1.8 | 664 ± 3 | 3.7 ± 0.1 |
| 9 | 300 | 12 | 5 | 0.1 | 4 | 1 | 30 | 48 | 240 | 30 | 80 | 94.5 ± 4.7 | 613 ± 3 | 3.9 ± 0.1 |
| 10 | 200 | 12 | 5 | 0.5 | 4 | 1 | 25 | 96 | 180 | 30 | 120 | 23.8 ± 1.6 | 742 ± 4 | 3.4 ± 0.0 |
| 11 | 300 | 9 | 5 | 0.5 | 7 | 1 | 25 | 48 | 240 | 20 | 120 | 125.9 ± 4.9 | 585 ± 3 | 4.7 ± 0.1 |
| 12 | 200 | 9 | 2 | 0.1 | 4 | 1 | 25 | 48 | 180 | 20 | 80 | 33.5 ± 1.5 | 464 ± 3 | 3.0 ± 0.0 |
| t-Stat (Y1) | 52.02 | −31.13 | 25.88 | 3.89 | 3.31 | −18.94 | 16.08 | −6.75 | 3.5 | −5.45 | 3.83 | |||
| %Cont (Y1) | 53.15 | 19.04 | 13.16 | 0.30 | 0.22 | 7.04 | 5.08 | 0.89 | 0.24 | 0.58 | 0.29 | |||
| Rank (Y1) | I | II | III | VIII | XI | IV | V | VI | X | VII | IX | |||
| Run | Independent Variable (Coded Value) | Erythritol Yield | Error | |||
|---|---|---|---|---|---|---|
| Molasses | Yeast Extract | KH2PO4 | Actual | Predicted | ||
| g·L−1 | g·L−1 | g·L−1 | g·L−1 | g·L−1 | % | |
| 1 | 200 (−1) | 9 (−1) | 2 (−1) | 35.1 ± 1.8 | 33.5 | +4.5 |
| 2 | 200 (−1) | 9 (−1) | 5 (+1) | 96.6 ± 3.9 | 88.1 | +8.7 |
| 3 | 200 (−1) | 12 (+1) | 2 (−1) | 63.1 ± 2.3 | 60.0 | +4.9 |
| 4 | 200 (−1) | 12 (+1) | 5 (+1) | 72.5 ± 2.7 | 76.1 | −4.9 |
| 5 | 300 (+1) | 9 (−1) | 2 (−1) | 59.2 ± 2.5 | 51.6 | +12.7 |
| 6 | 300 (+1) | 9 (−1) | 5 (+1) | 89.4 ± 3.5 | 86.0 | +3.8 |
| 7 | 300 (+1) | 12 (+1) | 2 (−1) | 62.3 ± 2.4 | 64.3 | −3.2 |
| 8 | 300 (+1) | 12 (+1) | 5 (+1) | 65.2 ± 2.9 | 60.2 | +7.6 |
| 9 | 166 (−1.68) | 10.5 (0) | 3.5 (0) | 28.2 ± 1.7 | 30.9 | −9.6 |
| 10 | 334 (+1.68) | 10.5 (0) | 3.5 (0) | 68.7 ± 3.1 | 73.4 | −6.8 |
| 11 | 250 (0) | 7.98 (−1.68) | 3.5 (0) | 57.8 ± 3.2 | 63.5 | −9.8 |
| 12 | 250 (0) | 13.02 (+1.68) | 3.5 (0) | 65.8 ± 3.6 | 64.1 | +2.6 |
| 13 | 250 (0) | 10.5 (0) | 0.98 (−1.68) | 80.8 ± 3.3 | 83.3 | −3.1 |
| 14 | 250 (0) | 10.5 (0) | 6.02 (+1.68) | 78.6 ± 3.1 | 85.2 | −8.4 |
| 15 | 250 (0) | 10.5 (0) | 3.5 (0) | 89.4 ± 3.6 | 96.6 | −8.0 |
| 16 | 250 (0) | 10.5 (0) | 3.5 (0) | 105.6 ± 4.6 | 96.6 | +8.5 |
| 17 | 250 (0) | 10.5 (0) | 3.5 (0) | 104.2 ± 4.5 | 96.6 | +7.3 |
| 18 | 250 (0) | 10.5 (0) | 3.5 (0) | 89.4 ± 4.8 | 96.6 | −8.0 |
| 19 | 250 (0) | 10.5 (0) | 3.5 (0) | 97.1 ± 5.0 | 96.6 | +0.5 |
| 20 | 250 (0) | 10.5 (0) | 3.5 (0) | 95.0 ± 4.9 | 96.6 | −1.6 |
| Constituents (Unit) | Quantity |
|---|---|
| Moisture (g/100 g) | 22.5 ± 4.3 |
| Total sugar (g/100 g) | 53.3 ± 5.6 |
| Reducing sugar (g/100 g) | 18.1 ± 2.5 |
| Non-reducing sugar (g/100 g) | 35.2 ± 3.2 |
| Crude protein (g/100 g) | 0.35 ± 0.1 |
| Crude fat (g/100 g) | 0.42 ± 0.1 |
| Ash (g/100 g) | 11.5 ± 4.0 |
| Brix value (°Bx) | 78.1 ± 0.2 |
| pH (-) | 5.9 ± 0.1 |
| Parameter | Coefficient ± 95% CI | p-Value |
|---|---|---|
| Constant | 96.58 ± 8.41 | 0.0005 |
| x1 | 12.63 ± 5.61 | 0.0005 |
| x2 | 0.18 ± 2.52 | 0.9433 |
| x3 | 0.55 ± 2.51 | 0.8301 |
| x1x2 | −9.61 ± 3.29 | 0.0153 |
| x1x3 | −5.05 ± 3.66 | 0.1004 |
| x2x3 | −3.44 ± 3.28 | 0.3200 |
| x1x1 | −15.68 ± 5.47 | <0.0001 |
| x2x2 | −11.57 ± 5.46 | 0.0008 |
| x3x3 | −4.34 ± 2.05 | 0.0093 |
| F-value | 10.52 | - |
| p-value (model) | - | 0.0002 |
| p-value (lack of fit) | - | 0.4539 |
| R2 | 0.92 | - |
| Adj R2 | 0.84 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seshadrinathan, S.; Chakraborty, S. Fermentative Production of Erythritol from Cane Molasses Using Candida magnoliae: Media Optimization, Purification, and Characterization. Sustainability 2022, 14, 10342. https://doi.org/10.3390/su141610342
Seshadrinathan S, Chakraborty S. Fermentative Production of Erythritol from Cane Molasses Using Candida magnoliae: Media Optimization, Purification, and Characterization. Sustainability. 2022; 14(16):10342. https://doi.org/10.3390/su141610342
Chicago/Turabian StyleSeshadrinathan, Shruthy, and Snehasis Chakraborty. 2022. "Fermentative Production of Erythritol from Cane Molasses Using Candida magnoliae: Media Optimization, Purification, and Characterization" Sustainability 14, no. 16: 10342. https://doi.org/10.3390/su141610342
APA StyleSeshadrinathan, S., & Chakraborty, S. (2022). Fermentative Production of Erythritol from Cane Molasses Using Candida magnoliae: Media Optimization, Purification, and Characterization. Sustainability, 14(16), 10342. https://doi.org/10.3390/su141610342






