Carbon Sequestration in Fine Aroma Cocoa Agroforestry Systems in Amazonas, Peru
Abstract
:1. Introduction
2. Materials and Methods
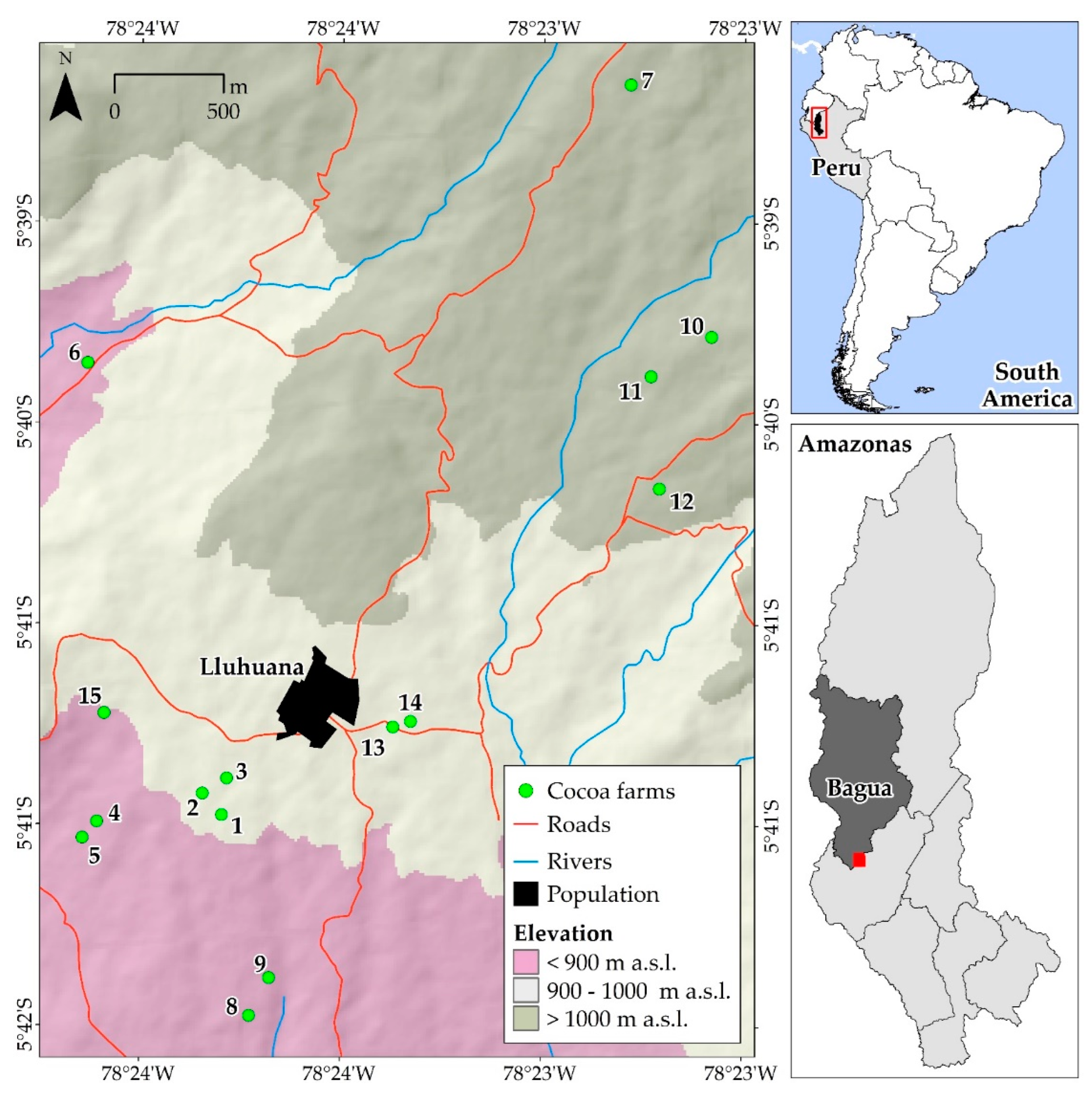
2.1. Study Area
2.2. Estimation of Carbon Pools in Fine Aroma Cocoa Agroforestry System
2.2.1. Selection of Cocoa Farms and Data collection for the Calculation of Aboveground Biomass (Cocoa Trees, Other Tree Species, and Leaf Litter), and Carbon in the Soil
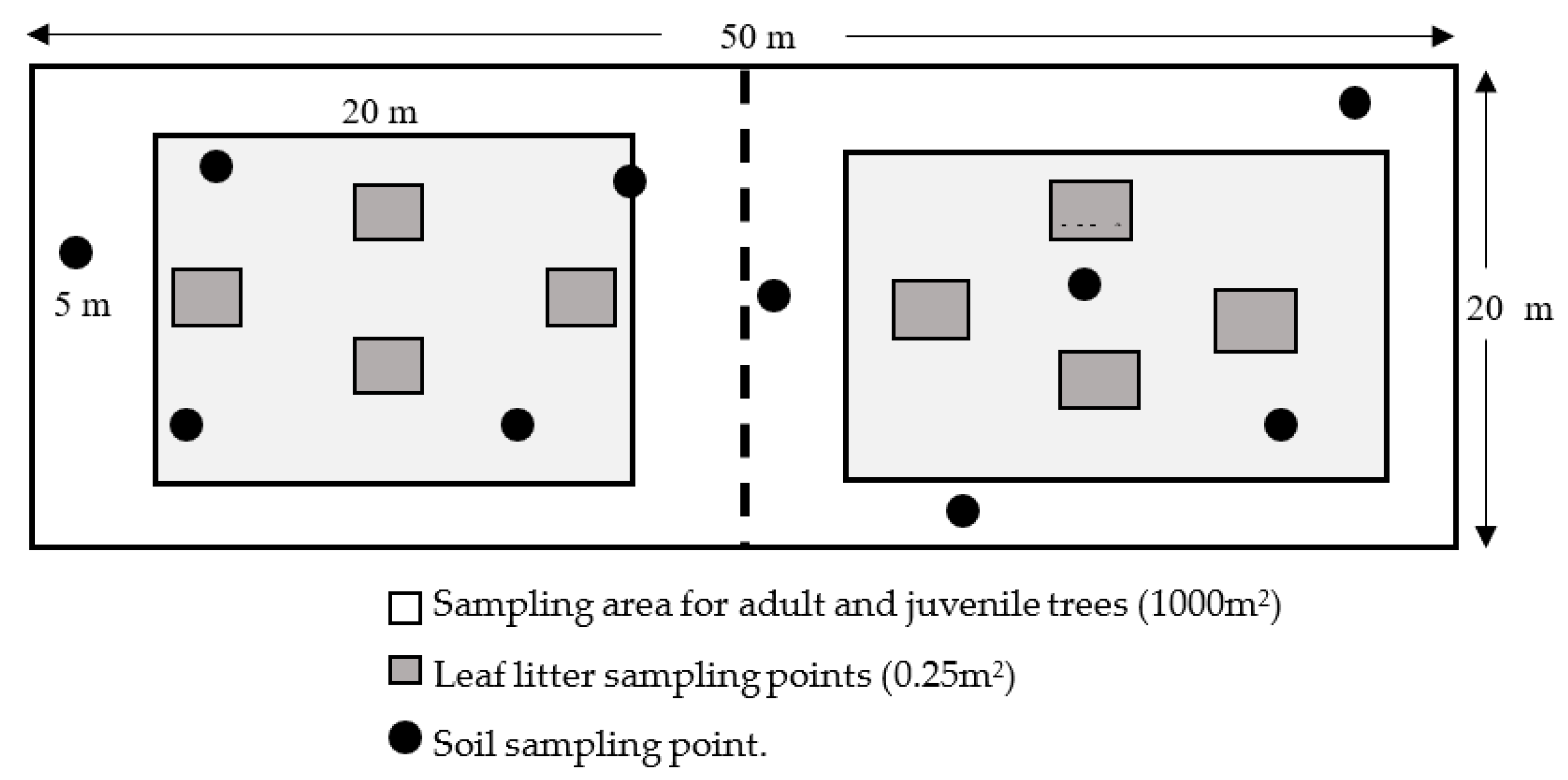
- Only for adult and juvenile trees: In the centre of each plantation system, a rectangle of 1000 m2 with a length of 50.00 m and a width of 20.00 m was delimited, which was divided into 2 smaller rectangles, each measuring a length of 25.00 m and a width of 20.00 m (500 m2) (Figure 2) [21]. Trees with a diameter at breast height (DBH) greater than 2.5 cm within the larger rectangle were measured and the DBH was recorded [20].
- Leaf litter: Within the small rectangles (500 m2), we delimited two rectangles of 5.00 m × 20.00 m (100 m2); within these quadrants, sub-quadrants of 0.5 m × 0.5 m (0.25 m2) were defined at 8 points, where all the plant material (leaf litter) found on the soil surface was weighed at each point. Then, a sub-sample of 500 g was taken in an airtight bag and transported to the laboratory to determine the humidity [22].
- Carbon in the soil: A homogeneous sample of 500 g of soil was collected from 10 random points per farm, sampling was done at a depth of 0–30 cm, and this sample was used to calculate the organic matter of the soil at the laboratory [21]. These data were used to calculate the amount of carbon that can be accumulated by the studied AFS.
2.2.2. Measurement of Total Aboveground Biomass (TAB) and Calculation of the Total Carbon of the Cocoa Agroforestry Systems
2.3. Statistical Analysis
3. Results
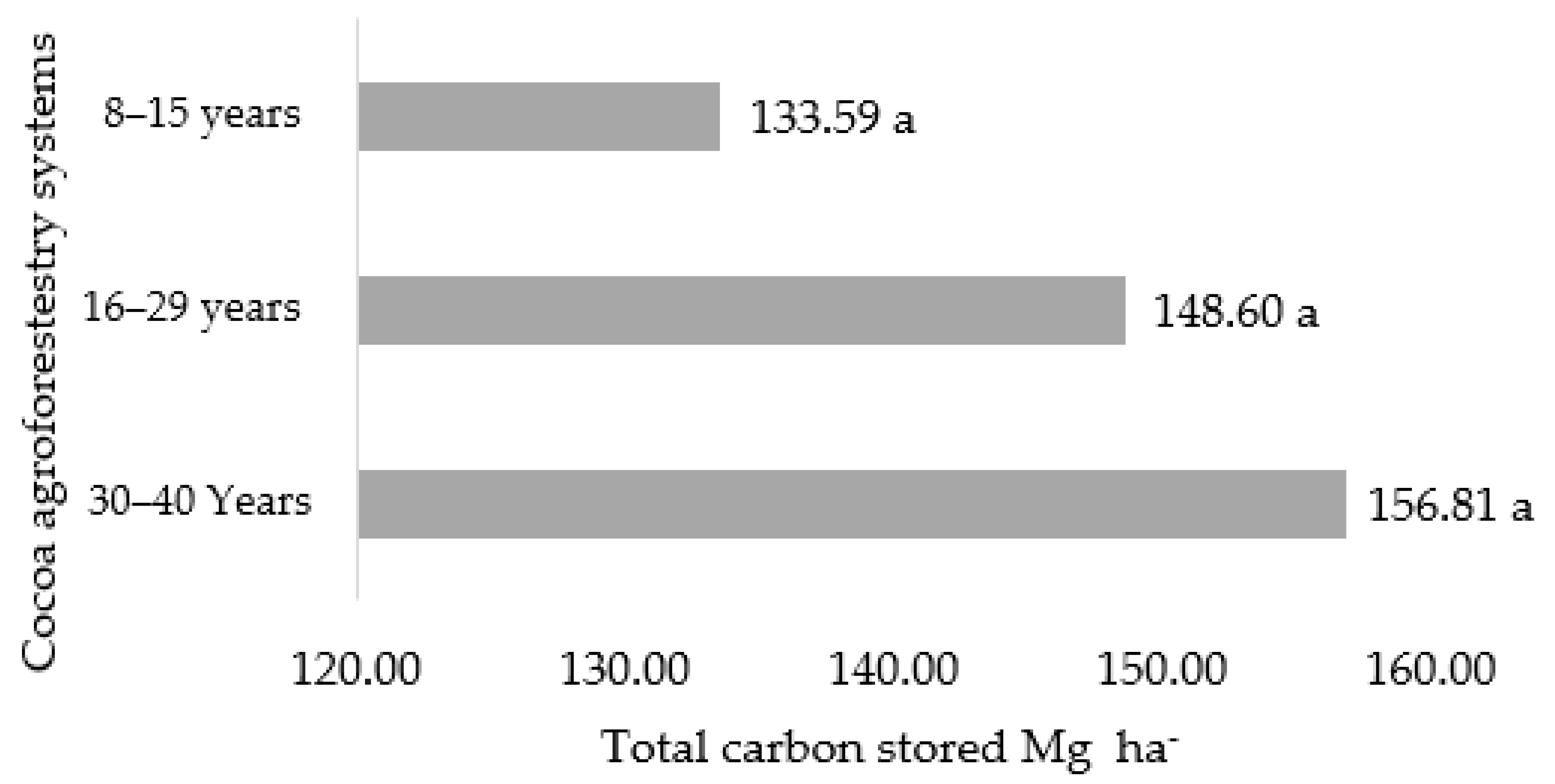
3.1. Aboveground Biomass Capture and Carbon Stored in Cocoa Agroforestry Systems
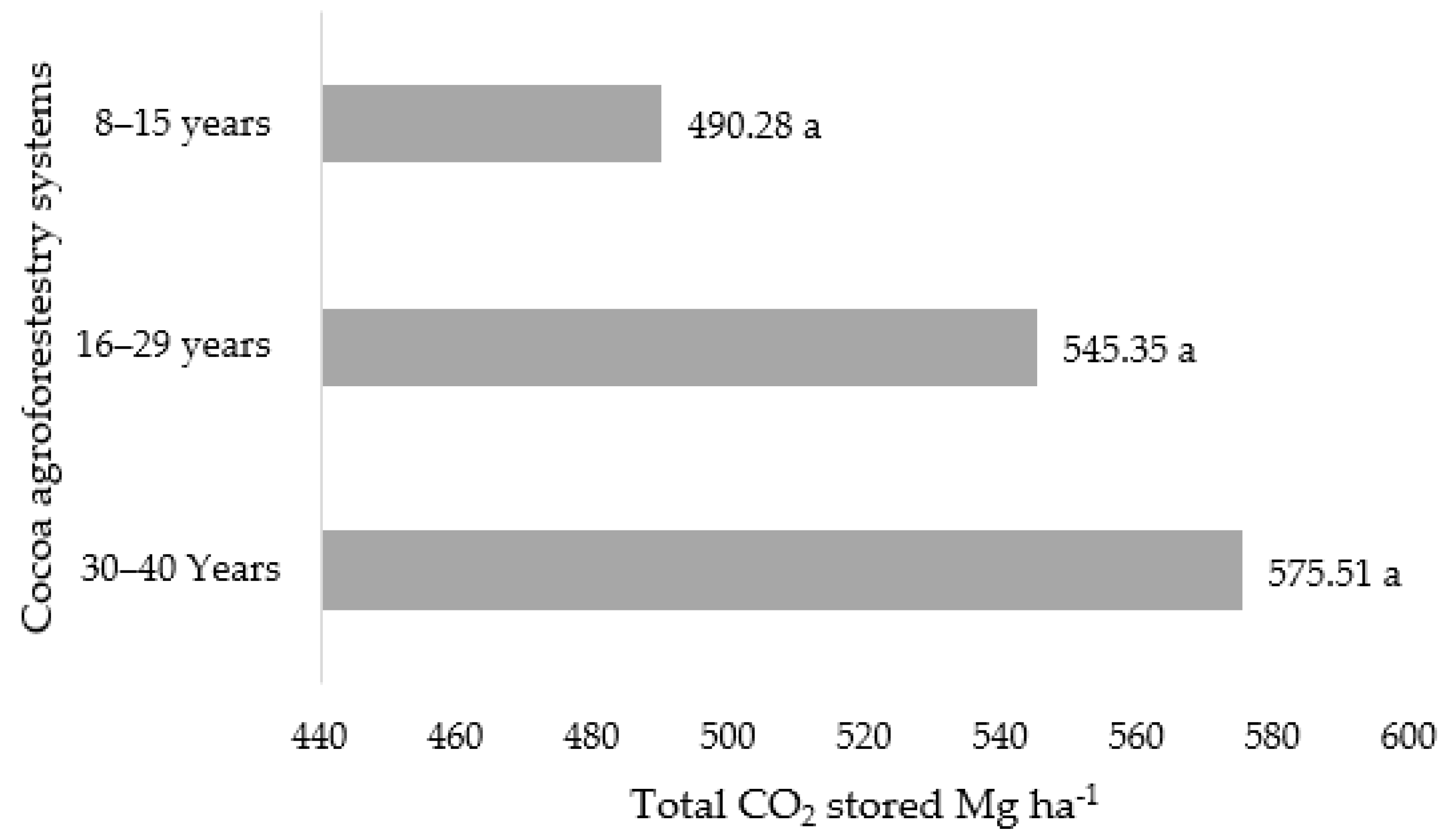
3.2. CO2 Sequestration of Cocoa Agroforestry Systems
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Janiola, M.D.C.; Marin, R.A. Carbon Sequestration Potential of Fruit Tree Plantations in Southern Philippines. J. Biodivers. Environ. Sci. 2016, 8, 164–174. [Google Scholar]
- Andrade, H.J.; Ibrahim, M. ¿Cómo monitorear el secuestro de carbono en los sistemas silvopastoriles? Agrofor. Am. 2003, 10, 109–116. [Google Scholar]
- FAO. Emissions due to Agriculture. Global, Regional and Country Trends 2000–2018; FAOSTAT Analytical Brief Series No 18; FAO: Rome, Italy, 2020. [Google Scholar]
- Ministerio del Ambiente. El Perú y el Cambio Climático Tercera Comunicación Nacional del Perú; Ministerio del Ambiente: Lima, Peru, 2016. [Google Scholar]
- Chou, S.W.; Gutiérrez-Espeleta, E.E. Ecuación para estimar la biomasa arbórea en los bosques tropicales de Costa Rica. Rev. Tecnol. Marcha 2013, 26, 41. [Google Scholar] [CrossRef]
- Jiménez-Herrera, E. Estimación de la Cantidad de Carbono Almacenado en un Agroecosistema de Cacao (Theobroma cacao L.) en el Sector Shupishiña-San Martín. Ph.D. Thesis, Universidad Nacional de San Martin, Tarapoto, Peru, 2019. [Google Scholar]
- Montagnini, F.; Nair, P.K.R. Carbon sequestration: An underexploited environmental benefit of agroforestry systems. Agrofor. Syst. 2004, 61–62, 281–295. [Google Scholar] [CrossRef]
- Ram, N.; Dhyani, S.K. Agroforestry systems for carbon sequestration: Present status and scope. Ind. J. Agrofor. 2008, 10, 1–9. [Google Scholar]
- FAO; CIAT; CCAFS. Retos del Cambio Climático para la Agricultura en América Latina y el Caribe. Centro Internacional de Agricultura Tropical (CIAT), Cali. CO. 2018. Available online: https://hdl.handle.net/10568/98282 (accessed on 21 July 2022).
- Rueda, X.; Garrett, R.D.; Lambin, E.F. Corporate investments in supply chain sustainability: Selecting instruments in the agri-food industry. J. Clean. Prod. 2017, 142, 2480–2492. [Google Scholar] [CrossRef]
- Rodriguez Guevara, E.G. La gestión de la cadena de suministro sostenible en la industria alimenticia. AD-Minister 2018, 113–134. [Google Scholar] [CrossRef]
- Toledo-Hernández, M.; Tscharntke, T.; Tjoa, A.; Anshary, A.; Cyio, B.; Wanger, T.C. Hand pollination, not pesticides or fertilizers, increases cocoa yields and farmer income. Agric. Ecosyst. Environ. 2020, 304, 107160. [Google Scholar] [CrossRef]
- Anchundia, D.M.; Herrada, M.R.; Segovia, E.L. Sistemas agroforestales con cultivo de cacao fino de aroma: Entorno socio-económico y productivo. Agroforestry systems with fine aroma cocoa cultivation: Socio-economic and productive environment. Rev. Cuba. Cienc. For. 2018, 6, 103–115. [Google Scholar]
- Murthy, I.K.; Gupta, M.; Tomar, S.; Munsi, M.; Hegde, R.T.G.; Nh, R. Carbon Sequestration Potential of Agroforestry Systems in India. J. Earth Sci. Clim. Chang. 2013, 4, 1–7. [Google Scholar] [CrossRef]
- Sanial, E.; Fountain, A.; Hoefsloot, H.; Jezeer, R. Agroforestry in Cocoa, a Need for Ambitious Collaborative Landscape Approaches. The Cocoa Barometer Consortium. 2020. Available online: https://hdl.handle.net/10568/111621 (accessed on 21 July 2022).
- Oelbermann, M.; Voroney, R.P.; Gordon, A.M. Carbon sequestration in tropical and temperate agroforestry systems: A review with examples from Costa Rica and southern Canada. Agric. Ecosyst. Environ. 2004, 104, 359–377. [Google Scholar] [CrossRef]
- Borden, K.A.; Anglaaere, L.C.N.; Adu-Bredu, S.; Isaac, M.E. Root biomass variation of cocoa and implications for carbon stocks in agroforestry systems. Agrofor. Syst. 2019, 93, 369–381. [Google Scholar] [CrossRef]
- Chave, J.; Andalo, C.; Brown, S.; Cairns, M.A.; Chambers, J.Q.; Eamus, D.; Fölster, H.; Fromard, F.; Higuchi, N.; Kira, T.; et al. Tree allometry and improved estimation of carbon stocks and balance in tropical forests. Oecologia 2005, 145, 87–99. [Google Scholar] [CrossRef]
- Mohammed, A.M.; Robinson, J.S.; Midmore, D.; Verhoef, A. Biomass stocks in Ghanaian cocoa ecosystems: The effects of region, management and stand age of cocoa trees. Eur. J. Agric. For. Res. 2015, 3, 22–43. [Google Scholar]
- Trinidad, F.W.; Sol-Sánchez, Á.; Galindo-Alcántara, A. Evaluación de la rentabilidad económica y captura de carbono en plantaciones de cacao en el plan Chontalpa, Tabasco. Rev. Iberoam. Bioecon. Cambio Clim. 2016, 2, 53–71. [Google Scholar] [CrossRef] [Green Version]
- Cerda-Bustillos, R.; Espin-Chion, T.; Cifuentes, M. Carbono en sistemas agroforestales de cacao de la Reserva Indígena Bribri de Talamanca, Costa Rica. Agrofor. Am. 2013, 49, 33–41. [Google Scholar]
- Acosta-Mireles, M.; Quednow, K.; Etchevers-Barra, J.; Monreal, C. Un método para la medición del carbono almacenado en la parte aérea de sistemas con vegetación natural e inducida en terrenos de ladera en México. In Proceedings of the Simposio Internacional Medición y Monitoreo de la Captura de Carbono en Ecosistemas Forestales, Valdivia, Chile, 18–20 October 2001; pp. 1–11. [Google Scholar]
- Arévalo, L.; Alegre, J.C.; Palm, C.A. Manual de las Reservas Totales de Carbono en los Diferentes Sistemas de Uso de la Tierra en Perú; STC-CGIAR Ministerio de Agricultura: Pucallpa, Perú, 2003. [Google Scholar]
- Yuliasmara, F.; Wibawa, A.; Prawoto, A. Carbon stock in different ages and plantation system of cocoa: Allometric approach. Pelita Perkeb. Coffee Cocoa Res. J. 2009, 26, 86–100. [Google Scholar] [CrossRef]
- Hairiah, K.; Dewi, S.; Agus, F.; van Noordwijk, M.; Rahayu, S. Measuring Carbon Stocks Across Land Use Systems: A Manual; World Agroforestry Centre (ICRAF), SEA Regional Office: Bogor, Indonesia, 2009. [Google Scholar]
- Andrade, H.; Segura, M.; Somarriba, E.; Villalobos, M. Valoración biofísica y financiera de la fijación de carbono por uso del suelo en fincas cacaoteras indígenas de Talamanca, Costa Rica. Agrofor. Am. 2008, 46, 45–50. [Google Scholar]
- Vásquez, H.; Valqui, L.; Alegre, J.C.; Gómez, C.; Maicelo, J. Analysis of four silvopastoral systems in Peru: Physical and nutritional characterization of pastures, floristic composition, carbon and CO2 reserves. Sci. Agropecu. 2020, 11, 167–176. [Google Scholar] [CrossRef]
- IPCC. Good Practice Guidance for Land Use, Land-Use Change and Forestry, 1st ed.; Intergovernmental Panel on Climate Change: Hayama, Japan, 2003; Volume 338, ISBN 4887880030. [Google Scholar]
- Salamanca-Jiménez, A.; Sadeghian-Khalajabadi, S. La densidad aparente y su realción con otras propiedades en suelos de la zona cafetera colombiana. Cenicafé 2005, 56, 381–397. [Google Scholar]
- Vasquez, A.; Vasquez, I.; Vasquez, C.; Cañamero, M. Fundamentos de la Ingeniería de Riegos; Universidad Nacional Agraria La Molina: Lima, Peru, 2017; Volume 1, ISBN 9786124147715. [Google Scholar]
- United States Department of Agriculture (USDA). Soil Bulk Density/Moisture/Aeration. Available online: https://www.nrcs.usda.gov/Internet/FSE_DOCUMENTS/nrcs142p2_053260.pdf (accessed on 19 August 2021).
- Di Rienzo, J.; Balzarini, M.; Robledo, C.; Casanoves, F.; Gonzales, L.; Tablada, E. InfoStat, Versión 2008. Manual del Usuario; Universidad Nacional de Córdoba: Córdoba, Argentina, 2008. [Google Scholar]
- Dean, C.B.; Nielsen, J.D. Generalized linear mixed models: A review and some extensions. Lifetime Data Anal. 2007, 13, 497–512. [Google Scholar] [CrossRef] [PubMed]
- Bolker, B.M.; Brooks, M.E.; Clark, C.J.; Geange, S.W.; Poulsen, J.R.; Stevens, M.H.H.; White, J.-S.S. Generalized linear mixed models: A practical guide for ecology and evolution. Trends Ecol. Evol. 2009, 24, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Patiño, S.; Suearez, L.N.; Andrade, H.J.; Segura, M.a. Captura de carbono en biomasa en plantaciones forestales y sistemas agroforestales en Armero-Guayabal, Tolima, Colombia. Capture of carbon in biomass in forestry plantations and agroforestry systems in Armero-Guayabal, Tolima, Colombia. Rev. Investig. Agrar. Ambient. 2018, 9, 14. [Google Scholar] [CrossRef]
- Ramírez-Vela, C.A.; Panduro-Pisco, G.; Miranda-Ruiz, E. Captura de carbono en un sistema agroforestal con Theobroma cacao en el campus universitario nacional de Ucayali; Pucallpa-Perú. Tzhoecoen Rev. Cient. 2014, 6, 165–180. [Google Scholar]
- Caro-Paza, D.M. Secuestro de Carbono en Sistemas Agroforestales de los Distritos de las Piedras y Tambopata, Provincia Tambopata, Departamento de Madre de Dios-Peru. Engineer’s Thesis, Universidad Nacional Amazónica de Madre de Dios, Puerto Maldonado, Peru, 2018. [Google Scholar]
- Concha, J.Y.; Alegre, J.C.; Pocomucha, V. Determincación e las reservas de carbono en la biomasa aérea de sistemas agroforestales de Theobroma cacao L. en el departamento de San Matín, Perú. Ecol. Apl. 2007, 6, 75–82. [Google Scholar] [CrossRef] [Green Version]
- Bohre, P.; Chaubey, O.P.; Singhal, P.K. Biomass accumulation and carbon sequestration in Tectona grandis Linn. f. and Gmelina arborea Roxb. Int. J. Bio-Sci. Bio-Technol. 2013, 5, 153–174. [Google Scholar]
- Ortiz, A.; Lorena, R.; Eduardo, S. Almacenamiento y tasas de fijación de biomasa y carbono en sistemas agroforestales de cacao (Theobroma cacao) y laurel (Cordia alliodora). Agrofor. Am. 2008, 2008, 26–29. [Google Scholar]
- Alcudia-Aguilar, A.; Martínez-Zurimendi, P.; Van Der Wal, H.; Castillo-Uzcanga, M.M.; Suárez-Sánchez, J. Allometric estimation of the biomass of Musa spp. in homegardens of Tabasco, Mexico. Trop. Subtrop. Agroecosyst. 2019, 22, 143–152. [Google Scholar]
- Zake, J.; Pietsch, S.A.; Friedel, J.K.; Zechmeister-Boltenstern, S. Can agroforestry improve soil fertility and carbon storage in smallholder banana farming systems? J. Plant Nutr. Soil Sci. 2015, 178, 237–249. [Google Scholar] [CrossRef]
- Danarto, S.A.; Hapsari, L. Biomass and Carbon Stock Estimation Inventory of Indonesian Bananas (Musa spp.) and Its Potential Role for Land Rehabilitation. Biotropia 2015, 22, 102–108. [Google Scholar] [CrossRef] [Green Version]
- Nijmeijer, A.; Lauri, P.-É.; Harmand, J.-M.; Saj, S. Carbon dynamics in cocoa agroforestry systems in Central Cameroon: Afforestation of savannah as a sequestration opportunity. Agrofor. Syst. 2019, 93, 851–868. [Google Scholar] [CrossRef]
- Post, W.M.; Kwon, K.C. Soil carbon sequestration and land-use change: Processes and potential. Glob. Chang. Biol. 2000, 6, 317–327. [Google Scholar] [CrossRef] [Green Version]
- United States Environmental Protection Agency. Carbon Storage by Tree Biomass; EnviroAtlas National Data Fact Sheet; EPA: Washington, DC, USA, 2018; pp. 1–2.
- Lal, R. Soil carbon sequestration to mitigate climate change. Geoderma 2004, 123, 1–22. [Google Scholar] [CrossRef]
- Albrecht, A.; Kandji, S.T. Carbon sequestration in tropical agroforestry systems. Agric. Ecosyst. Environ. 2003, 99, 15–27. [Google Scholar] [CrossRef]
- Vicente-Vicente, J.L.; García-Ruiz, R.; Francaviglia, R.; Aguilera, E.; Smith, P. Soil carbon sequestration rates under Mediterranean woody crops using recommended management practices: A meta-analysis. Agric. Ecosyst. Environ. 2016, 235, 204–214. [Google Scholar] [CrossRef] [Green Version]
- Dhillon, G.S.; Van Rees, K.C.J. Soil organic carbon sequestration by shelterbelt agroforestry systems in Saskatchewan. Can. J. Soil Sci. 2017, 97, 1–16. [Google Scholar] [CrossRef] [Green Version]
| Tree Species | Allometric Equations | Author |
|---|---|---|
| Timber trees | BA = 0.1184DBH2.53 | [23] |
| Cacao trees | Y = 0.1208 DBH1.98 | [24] |
| Musaceae | Y = 0.0303DBH2.13 | [25] |
| Fruit trees | [26] |
| Soil Texture | Total Pore Space or Total Porosity (%) | Bulk Density BD (g/cm3) |
|---|---|---|
| Sandy | 38 (32–42) | 1.65 (1.55–1.80) |
| Sandy loam | 43 (40–47) | 1.50 (1.40–1.60) |
| loam | 47 (43–49) | 1.40 (1.35–1.50) |
| Clay loam | 49 (47–51) | 1.35 (1.30–1.40) |
| Sandy clay | 51 (49–53) | 1.30 (1.25–1.35) |
| Clay | 53 (51–55) | 1.25 (1.20–1.30) |
| AFS | Theobroma cacao Mg ha−1 | Mussa sp. Mg ha−1 | Cordia sp. Mg ha−1 | Persea sp. Mg ha−1 | Other Species * Mg ha−1 | Leaf Litter Mg ha−1 | Total Aboveground Biomass Mg ha−1 |
|---|---|---|---|---|---|---|---|
| 1 | 9.35 ± 0.05 | 0.71 ± 0.03 | 13.65.04 ± 4.65 | 0.63 ± 0.04 | 27.64 ± 7.01 | 1.11 ± 0.10 | 53.09 ± 10.68 |
| 2 | 13.24 ± 0.10 | 0.85 ± 0.09 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 0.83 ± 0.41 | 14.92 ± 05.28 |
| 3 | 14.15 ± 0.22 | 00 ± 00 | 1.58 ± 0.00 | 00 ± 00 | 4.72 ± 0.34 | 1.14 ± 0.22 | 21.59 ± 05.45 |
| 4 | 9.35 ± 0.04 | 1.81 ± 0.14 | 27.17 ± 4.96 | 00 ± 00 | 10.99 ± 4.87 | 0.98 ± 0.14 | 50.30 ± 02.66 |
| 5 | 14.47 ± 0.07 | 00 ± 00 | 13.96 ± 1.11 | 00 ± 00 | 5.08 ± 1.69 | 1.05 ± 0.15 | 34.56 ± 06.81 |
| 6 | 13.11 ± 0.04 | 1.14 ± 0.07 | 00 ± 00 | 00 ± 00 | 2.92 ± 0.38 | 1.03 ± 0.14 | 18.20 ± 05.05 |
| 7 | 11.59 ± 0.04 | 4.77 ± 0.05 | 11.24 ± 1.08 | 00 ± 00 | 00 ± 00 | 0.87 ± 0.21 | 28.47 ± 05.46 |
| 8 | 30.64 ± 0.05 | 3.61 ± 0.05 | 11.24 ± 1.08 | 3.68 ± 0.41 | 1.42 ± 0.27 | 1.06 ± 0.07 | 51.65 ± 11.40 |
| 9 | 24.02 ± 0.13 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 18.79 ± 5.27 | 0.91 ± 0.10 | 43.72 ± 11.07 |
| 10 | 24.16 ± 0.11 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 0.02 ± 0.00 | 1.07 ± 0.27 | 25.25 ± 09.78 |
| 11 | 17.76 ± 0.04 | 1.54 ± 0.03 | 16.34 ± 11.28 | 00 ± 00 | 0.61 ± 0.00 | 1.23 ± 0.26 | 37.48 ± 08.40 |
| 12 | 12.91 ± 0.09 | 0.06 ± 0.00 | 20.22 ± 1.06 | 00 ± 00 | 5.51 ± 0.59 | 0.87 ± 0.20 | 39.57 ± 08.57 |
| 13 | 07.35 ± 0.13 | 4.75 ± 0.04 | 4.03 ± 1.38 | 3.04 ± 1.32 | 14.25 ± 3.25 | 0.78 ± 0.14 | 34.20 ± 04.71 |
| 14 | 9.33 ± 0.06 | 0.19 ± 0.02 | 95.70 ± 22.52 | 0.25 ± 0.00 | 9.90 ± 3.63 | 0.61 ± 0.31 | 115.98 ± 37.69 |
| 15 | 10.01 ± 0.05 | 3.92 ± 0.04 | 00 ± 00 | 2.55 ± 0.00 | 5.63 ± 0.16 | 0.73 ± 0.37 | 22.84 ± 01.79 |
| 8–15 years | 12.13 ± 2.90 a | 2.13 ± 2.44 a | 6.16 ± 6.13 a | 0.61 ± 1.36 a | 5.39 ± 5.34 a | 0.97 ± 0.15 ab | 27.40 ± 7.37 a |
| 16–29 years | 14.71 ± 6.31 a | 0.82 ± 8.83 a | 15.48 ± 10.04 a | 0.13 ± 0.28 a | 8.95 ± 11.34 a | 1.05 ± 0.14 a | 41.14 ± 11.12 a |
| 30–40 years | 17.45 ± 9.44 a | 1.71 ± 1.90 a | 21.39 ± 41.83 a | 1.30 ± 1.71 a | 7.15 ± 7.57 a | 0.83 ± 0.17 b | 49.82 ± 39.89 a |
| AFS | Theobroma cacao Mg ha−1 | Mussa sp. Mg ha−1 | Cordia sp. Mg ha−1 | Persea sp. Mg ha−1 | Other Species * Mg ha−1 | Litter Leaf Mg ha−1 | Total Aboveground Biomass Mg ha−1 |
|---|---|---|---|---|---|---|---|
| 1 | 4.69 ± 0.03 | 0.35 ± 0.01 | 6.83 ± 2.33 | 0.32 ± 0.02 | 13.82 ± 3.50 | 0.50 ± 0.05 | 26.51 ± 5.35 |
| 2 | 6.62 ± 0.05 | 0.43 ± 0.05 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 0.37 ± 0.19 | 7.42 ± 2.64 |
| 3 | 7.08 ± 0.11 | 00 ± 00 | 0.79 ± 0.00 | 00 ± 00 | 2.36 ± 0.17 | 0.51 ± 0.10 | 10.74 ± 2.73 |
| 4 | 4,69 ± 0.02 | 0.9 ± 0.07 | 13.58 ± 2.48 | 00 ± 00 | 5.5 ± 2.43 | 0.44 ± 0.06 | 25.11 ± 5.15 |
| 5 | 7.24 ± 0.03 | 00 ± 00 | 6.98 ± 0.56 | 00 ± 00 | 2.55 ± 0.80 | 0.47 ± 0.06 | 17.24 ± 3.34 |
| 6 | 6,52 ± 0.02 | 0.56 ± 0.04 | 00 ± 00 | 00 ± 00 | 1.46 ± 0.20 | 0.47 ± 0.07 | 9.01 ± 2.52 |
| 7 | 5.81 ± 0.02 | 2.36 ± 0.02 | 5.62 ± 0.54 | 00 ± 00 | 00 ± 00 | 0.39 ± 0.09 | 14.18 ± 2.74 |
| 8 | 15.30 ± 0.06 | 1.79 ± 0.02 | 5.62 ± 0.54 | 1.84 ± 0.21 | 0.7 ± 0.13 | 0.48 ± 0.03 | 25.73 ± 5.70 |
| 9 | 11.98 ± 0.06 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 9.39 ± 2.63 | 0.41 ± 0.05 | 21.78 ± 5.53 |
| 10 | 11.92 ± 0.06 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 0.01 ± 0.00 | 0.48 ± 0.12 | 12.41 ± 4.83 |
| 11 | 8.86 ± 0.02 | 0.77 ± 0.02 | 8.17 ± 0.64 | 00 ± 00 | 0.31 ± 0.00 | 0.55 ± 0.12 | 18.66 ± 4.27 |
| 12 | 6.50 ± 0.05 | 0.03 ± 0.00 | 10.12 ± 0.53 | 00 ± 00 | 2.75 ± 0.29 | 0.39 ± 0.09 | 19.79 ± 4.30 |
| 13 | 3.64 ± 0.07 | 2.38 ± 0.02 | 2.01 ± 0.69 | 1.52 ± 0.66 | 7.11 ± 1.62 | 0.35 ± 0.06 | 17.01 ± 2.35 |
| 14 | 4.62 ± 0.03 | 0.09 ± 0.02 | 47.86 ± 11.26 | 0.12 ± 0.00 | 4.93 ± 1.82 | 0.27 ± 0.14 | 57.89 ± 18.860 |
| 15 | 5.04 ± 0.02 | 2.00 ± 0.02 | 00 ± 00 | 1.28 ± 0.00 | 2.81 ± 0.08 | 0.33 ± 0.17 | 11.46 ± 1.79 |
| 8–15 years | 6.06 ± 1.46 a | 1.06 ± 1.22 a | 3.08 ± 3.06 a | 0.30 ± 0.68 a | 2.70 ± 2.67 a | 0.44 ± 0.07 ab | 13.64 ± 3.69 a |
| 16–29 years | 7.33 ± 3.08 a | 0.41 ± 0.41 a | 7.74 ± 5.02 a | 0.06 ± 0.14 a | 4.48 ± 5.67 a | 0.47 ± 0.06 a | 20.50 ± 5.63 a |
| 30–40 years | 8.71 ± 4.71 a | 0.86 ± 0.96 a | 10.70 ± 20.92 a | 0.65 ± 0.86 a | 3.57 ± 7.78 a | 0.37 ± 0.08 b | 24.86 ± 19.91 a |
| AFS | SVW Mg ha−1 | Texture of Soil | Bulk Density (g/cm3) | % Carbono | Total Carbon in Soil Mg ha−1 |
|---|---|---|---|---|---|
| 1 | 3750 | Clay | 1.25 | 1.60 | 60.00 |
| 2 | 3900 | Sandy clay | 1.30 | 2.13 | 83.07 |
| 3 | 4200 | Loam | 1.40 | 2.13 | 89.46 |
| 4 | 3750 | Clay | 1.25 | 3.15 | 118.13 |
| 5 | 3750 | Clay | 1.25 | 2.75 | 103.13 |
| 6 | 4200 | Sandy clay loam | 1.40 | 3.50 | 147.00 |
| 7 | 3900 | Sandy loam | 1.30 | 3.57 | 139.23 |
| 8 | 4200 | Sandy clay loam | 1.40 | 3.65 | 153.30 |
| 9 | 4200 | Sandy clay loam | 1.40 | 3.31 | 139.02 |
| 10 | 3750 | Clay | 1.25 | 4.50 | 168.75 |
| 11 | 3750 | Clay | 1.25 | 4.10 | 153.75 |
| 12 | 3750 | Clay | 1.25 | 3.73 | 139.88 |
| 13 | 4200 | Sandy clay loam | 1.40 | 2.88 | 120.96 |
| 14 | 4500 | Sandy loam | 1.50 | 3.55 | 159.75 |
| 15 | 4500 | Sandy clay | 1.50 | 2.77 | 124.65 |
| 8–15 years | — | — | 2.97 ± 0.59 | 119.96 ± 24.07 | |
| 16–29 years | — | — | 2.42 ± 1.13 | 128.10 ± 42.39 | |
| 30–40 years | — | — | 3.08 ± 0.63 | 131.96 ± 30.50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goñas, M.; Rojas-Briceño, N.B.; Culqui-Gaslac, C.; Arce-Inga, M.; Marlo, G.; Pariente-Mondragón, E.; Oliva-Cruz, M. Carbon Sequestration in Fine Aroma Cocoa Agroforestry Systems in Amazonas, Peru. Sustainability 2022, 14, 9739. https://doi.org/10.3390/su14159739
Goñas M, Rojas-Briceño NB, Culqui-Gaslac C, Arce-Inga M, Marlo G, Pariente-Mondragón E, Oliva-Cruz M. Carbon Sequestration in Fine Aroma Cocoa Agroforestry Systems in Amazonas, Peru. Sustainability. 2022; 14(15):9739. https://doi.org/10.3390/su14159739
Chicago/Turabian StyleGoñas, Malluri, Nilton B. Rojas-Briceño, Cristian Culqui-Gaslac, Marielita Arce-Inga, Gladys Marlo, Elí Pariente-Mondragón, and Manuel Oliva-Cruz. 2022. "Carbon Sequestration in Fine Aroma Cocoa Agroforestry Systems in Amazonas, Peru" Sustainability 14, no. 15: 9739. https://doi.org/10.3390/su14159739
APA StyleGoñas, M., Rojas-Briceño, N. B., Culqui-Gaslac, C., Arce-Inga, M., Marlo, G., Pariente-Mondragón, E., & Oliva-Cruz, M. (2022). Carbon Sequestration in Fine Aroma Cocoa Agroforestry Systems in Amazonas, Peru. Sustainability, 14(15), 9739. https://doi.org/10.3390/su14159739







