Biostimulation and Bioaugmentation of Soils Contaminated with Decachlorobiphenyl (PCB-209) Using Native Bacterial Strains Individually and in Consortia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemical Reagents
2.2. Soil Samples
2.3. Bacterial Strains
2.4. Master Cell Bank
2.5. Biostimulation of Bacterial Strains in a Liquid Medium Contaminated with PCB
2.6. Bioaugmentation Assay
2.7. Measurement of Bacterial Growth in the Bioaugmentation Process
2.8. Physicochemical Characterization of Bioaugmented Soil
2.9. PCB-209 Quantification in Bioaugmented Soil
2.10. Statistic Analysis
3. Results
3.1. Biostimulation of Bacterial Strains
3.2. Bioaugmentation of PCB-Contaminated Soil by Individual Bacterial Strains or in Consortia
3.3. Physicochemical Characteristics of Bioaugmented Soil
3.4. PCB-209 Removal Potential by Bacterial Strains in the Bioaugmentation Process
3.5. Principal Component Analysis (PCA)
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sharma, J.K.; Gautam, R.K.; Nanekar, S.V.; Weber, R.; Singh, B.K.; Singh, S.K.; Juwarkar, A.A. Advances and perspective in bioremediation of polychlorinated biphenyl-contaminated soils. Environ. Sci. Pollut. Res. Int. 2018, 25, 16355–16375. [Google Scholar] [CrossRef] [PubMed]
- Tu, C.; Teng, Y.; Luo, Y.; Li, X.; Sun, X.; Li, Z.; Liu, W.; Christie, P. Potential for biodegradation of polychlorinated biphenyls (PCBs) by Sinorhizobium meliloti. J. Hazard. Mater. 2011, 186, 1438–1444. [Google Scholar] [CrossRef] [PubMed]
- Passatore, L.; Rossetti, S.; Juwarkar, A.A.; Massacci, A. Phytoremediation and bioremediation of polychlorinated biphenyls (PCBs): State of knowledge and research perspectives. J. Hazard. Mater. 2014, 278, 189–202. [Google Scholar] [CrossRef]
- Huo, S.; Li, C.; Xi, B.; Yu, Z.; Yeager, K.M.; Wu, F. Historical record of polychlorinated biphenyls (PCBs) and special occurrence of PCB 209 in a shallow fresh-water lake from eastern China. Chemosphere 2017, 184, 832–840. [Google Scholar] [CrossRef] [PubMed]
- Piazza, R.; Ruiz-Fernández, A.C.; Frignani, M.; Zangrando, R.; Bellucci, L.G.; Moret, I.; Páez-Osuna, F. PCBs and PAHs in surficial sediments from aquatic environments of Mexico City and the coastal states of Sonora, Sinaloa, Oaxaca and Veracruz (Mexico). Environ. Geol. 2008, 54, 1537–1545. [Google Scholar] [CrossRef]
- Zenteno-Rojas, A.; Martínez-Romero, E.; Castañeda-Valbuena, D.; Rincón-Molina, C.I.; Ruiz-Valdiviezo, V.M.; Meza-Gordillo, R.; Villalobos-Maldonado, J.J.; Vences-Guzmán, M.A.; Rincón-Rosales, R. Structure and diversity of native bacterial communities in soils contaminated with polychlorinated biphenyls. AMB Express 2020, 10, 124. [Google Scholar] [CrossRef]
- Luo, W.; D’Angelo, E.M.; Coyne, M. Plant secondary metabolites, biphenyl, and hydroxypropyl-beta-cyclodextrin effects on aerobic polychlorinated biphenyl removal and microbial community structure in soils. Soil Biol. Biochem. 2007, 39, 735–743. [Google Scholar] [CrossRef]
- Pathiraja, G.; Egodawatta, P.; Goonetilleke, A.; Téo, V.S.J. Solubilization and degradation of polychlorinated biphenyls (PCBs) by naturally occurring facultative anaerobic bacteria. Sci. Total Environ. 2019, 651, 2197–2207. [Google Scholar] [CrossRef]
- Khalid, F.; Hashmi, M.Z.; Jamil, N.; Abdul, Q.; Muhammad, I.A. Microbial and enzymatic degradation of PCBs from e-waste-contaminated sites: A review. Environ. Sci. Pollut. Res. 2021, 28, 10474–10487. [Google Scholar] [CrossRef]
- Xiang, Y.; Xing, Z.; Liu, J.; Wei, Q.; Xing, H. Recent advances in the biodegradation of polychlorinated biphenyls. World J. Microbiol. Biotechnol. 2020, 36, 145. [Google Scholar] [CrossRef]
- Egorova, D.O.; Demakov, V.A.; Plotnikova, E.G. Bioaugmentation of a polychlorobiphenyl contaminated soil with two aerobic bacterial strains. J. Hazard. Mater. 2013, 261, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Cervantes-González, E.; Guevara-García, M.A.; García-Mena, J.; Ovando-Medina, V.M. Microbial diversity assessment of polychlorinated biphenyl–contaminated soils and the biostimulation and bioaugmentation processes. Environ. Monit. Assess. 2019, 191, 118. [Google Scholar] [CrossRef] [PubMed]
- Horváthová, H.; Lászlová, K.; Dercová, K. Bioremediation of PCB contaminated shallow river sediments: The efficacy of biodegradation using individual bacterial strains and their consortia. Chemosphere 2018, 193, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Steliga, T.; Wojtowicz, K.; Kapusta, P.; Brzeszcz, J. Assessment of Biodegradation Efficiency of Polychlorinated Biphenyls (PCBs) and Petroleum Hydrocarbons (TPH) in Soil Using Three Individual Bacterial Strains and Their Mixed Culture. Molecules 2020, 25, 709. [Google Scholar] [CrossRef] [Green Version]
- Zenteno-Rojas, A.; Martínez-Romero, E.; Rincón-Molina, C.I.; Ruiz-Valdiviezo, V.M.; Meza-Gordillo, R.; Villalobos-Maldonado, J.J.; Rincón-Rosales, R. Removal of High Concentrations Decachlorobiphenyl of Earthworm Eisenia fetida and its Symbiotic Bacteria in a Vermicomposting System. Water Air Soil Pollut. 2019, 230, 116. [Google Scholar] [CrossRef]
- Sánchez-Pérez, B.N.; Zenteno-Rojas, A.; Rincón-Molina, C.I.; Ruíz-Valdiviezo, V.M.; Gutiérrez-Miceli, F.A.; Vences-Guzmán, M.A.; Villalobos-Maldonado, J.J.; Rincón-Rosales, R. Rhizosphere and Endophytic Bacteria Associated to Ocimum basilicum L. with Decaclorobiphenyl Removal Potential. Water Air Soil Pollut. 2020, 231, 134. [Google Scholar] [CrossRef]
- Gómez, L.M.A.; Zenteno-Rojas, A.; Martinez-Romero, E.; Rincón-Molina, C.I.; Vences-Guzmán, M.A.; Ruíz-Valdiviezo, V.M.; Rincón-Molina, F.A.; Manzano-Gómez, L.A.; Rincón-Rosales, R. Biodegradation and Bioaccumulation of Decachlorobiphenyl (DCB) by Native Strain Pseudomonas extremaustralis ADA-5. Water Air Soil Pollut. 2021, 232, 192. [Google Scholar]
- Del Puerto, C.A.; Iglesias, E.; Morales, T.; Baños, N.; Nocedo, M.D.; Carnota, G.; Martínez, R. Organización y manejo de la colección de cepas de referencia del Instituto Finlay. Vaccimonitor 2009, 18, 20–24. [Google Scholar]
- AOAC. Official Methods of Analysis of AOAC International, 16th ed.; AOAC Interna: Gaithersburg, MD, USA, 1996; Volume 1. [Google Scholar]
- Bremner, J.M. Total nitrogen. In Methods of Soil Analysis Chemical Methods; Sparks, D.L., Ed.; Soil Science Society of America Inc.; American Society of Agronomy Inc.: Madison, WI, USA, 1996; pp. 1085–1122. [Google Scholar]
- USEPA. United States Environmental Protection Agency (USEPA). Method 8270D. Semivolatile Organic Compounds by Gas Chromatography/Mass Spectrometry (GC/MS). Available online: http://www.epa.gov/epawaste/hazard/testmethods/sw846/pdfs/8270d.pdf (accessed on 13 October 2013).
- Villalobos-Maldonado, J.J.; Meza-Gordillo, R.; Mancilla-Margalli, N.A.; Rodríguez-Mendiola, M.; Arias-Castro, C.; Vázquez-Villegas, P.; Gutiérrez-Miceli, F.A.; Ruiz-Valdiviezo, V.M. Removal of decachlorobiphenyl in vermicomposting process amended with rabbit manure and peat moss. Water Air Soil Pollut. 2015, 226, 159. [Google Scholar] [CrossRef]
- Vergani, L.; Mapelli, F.; Suman, J.; Cajthaml, T.; Uhlik, O.; Borin, S. Novel PCB-degrading Rhodococcus strains able to promote plant growth for assisted rhizoremediation of historically polluted soils. PLoS ONE 2019, 14, e0221253. [Google Scholar] [CrossRef] [Green Version]
- Leewis, M.C.; Uhlik, O.; Leigh, M. Synergistic Processing of Biphenyl and Benzoate: Carbon Flow Through the Bacterial Community in Polychlorinated-Biphenyl-Contaminated Soil. Sci. Rep. 2016, 6, 22145. [Google Scholar] [CrossRef] [PubMed]
- Ferrer, M.; Golyshin, P.; Timmis, K. Novel maltotriose esters enhance biodegradation of Aroclor 1242 by Burkholderia cepacia LB400. World J. Microbiol. Biotechnol. 2003, 19, 637–643. [Google Scholar] [CrossRef]
- Egorova, D.O.; Kiryanova, T.; Pyankova, A.; Ananina, L.; Plotnikova, E. Selective pressure of biphenyl/polychlorinated biphenyls on the formation of aerobic bacterial associations and their biodegradative potential. Folia Microbiol. 2021, 66, 659–676. [Google Scholar] [CrossRef] [PubMed]
- Bako, C.M.; Mattes, T.E.; Marek, R.F.; Hornbuckle, K.C.; Schnoor, J.L. Biodegradation of PCB congeners by Paraburkholderia xenovorans LB400 in presence and absence of sediment during lab bioreactor experiments. Environ. Pollut. 2021, 271, 116364. [Google Scholar] [CrossRef]
- Chen, S.C.; Budhraja, R.; Adrian, L.; Calebrese, F.; Stryhanyuk, H.; Musat, N.; Richnow, H.H.; Duan, G.L.; Zhu, Y.G.; Musat, F. Novel clades of soil biphenyl degraders revealed by integrating isotope probing, multi-omics, and single-cell analyses. ISME J. 2021, 15, 3508–3521. [Google Scholar] [CrossRef]
- Field, J.A.; Sierra-Alvarez, R. Microbial transformation and degradation of polychlorinated biphenyls. Environ. Pollut. 2008, 155, 1–12. [Google Scholar] [CrossRef]
- Kalantary, R.R.; Mohseni-Bandpi, A.; Esrafili, A.; Nasseri, S.; Ashmagh, F.R.; Jorfi, S.; Ja’fari, M. Effectiveness of biostimulation through nutrient content on the bioremediation of phenanthrene contaminated soil. J. Environ. Health Sci. Eng. 2014, 12, 143. [Google Scholar] [CrossRef] [Green Version]
- Chun, C.L.; Payne, R.B.; Sowers, K.R.; May, H.D. Electrical stimulation of microbial PCB degradation in sediment. Water Res. 2013, 47, 141–152. [Google Scholar] [CrossRef] [Green Version]
- Bhattacharya, A.S.; Khare, K. Biodegradation of 4-chlorobiphenyl by using induced cells and cell extract of Burkholderia xenovorans. Bioremediation J. 2017, 21, 109–118. [Google Scholar] [CrossRef]
- Bedard, D.L. A case study for microbial biodegradation: Anaerobic bacterial reductive dechlorination of polychlorinated biphenyls-from sediment to defined medium. Annu. Rev. Microbiol. 2008, 62, 253–270. [Google Scholar] [CrossRef]
- Saavedra, J.M.; Acevedo, F.; González, M.; Seeger, M. Mineralization of PCBs by the genetically modified strain Cupriavidus necator JMS34 and its application for bioremediation of PCBs in soil. Appl. Microbiol. Biotechnol. 2010, 87, 1543–1554. [Google Scholar] [CrossRef]
- Su, X.; Li, S.; Cai, J.; Xiao, Y.; Tao, L.; Hashmi, M.Z.; Lin, H.; Chen, J.; Mei, R.; Sun, F. Aerobic degradation of 3,3′,4,4′-tetrachlorobiphenyl by a resuscitated strain Castellaniella sp. SPC4: Kinetics model and pathway for biodegradation. Sci. Total Environ. 2019, 688, 917–925. [Google Scholar] [CrossRef] [PubMed]
- Wardle, D.A. The influence of biotic interactions on soil biodiversity. Ecol. Lett. 2006, 9, 870–886. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Baquerizo, M.; Trivedi, P.; Trivedi, C.; Eldridge, D.; Reich, P.; Jeffries, T.; Singh, B. Microbial richness and composition independently drive soil multifunctionality. Funct. Ecol. 2017, 31, 2330–2343. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Mera, E.; García-Paredes, J.D.; Corrales-Paternina, A.; Torregroza-Espinosa, A.C. Variability of Nitrogen Mineralization from Organic Matter in Agricultural Soils in the North of Colombia. Chiang Mai Univ. J. Nat. Sci. 2021, 20, e2021073. [Google Scholar]
- Gorovtsov, A.V.; Sazykin, I.S.; Sazykina, M.A. The influence of heavy metals, polyaromatic hydrocarbons, and polychlorinated biphenyls pollution on the development of antibiotic resistance in soils. Environ. Sci. Pollut. Res. 2018, 25, 9283–9292. [Google Scholar] [CrossRef] [PubMed]
- Méndez, V.; Fuentes, S.; Morgante, V.; Hernández, M.; González, M.; Moore, E.; Seeger, M. Novel hydrocarbonoclastic metal-tolerant Acinetobacter and Pseudomonas strains from Aconcagua river oil-polluted soil. J. Soil Sci. Plant Nutr. 2017, 17, 1074–1087. [Google Scholar] [CrossRef] [Green Version]
- Vaishnavi, J.; Devanesan, S.; AlSalhi, M.S.; Rajasekar, A.; Selvi, A.; Srinivasan, P.; Govarthanan, M. Biosurfactant mediated bioelectrokinetic remediation of diesel contaminated environment. Chemosphere 2021, 264, 128337. [Google Scholar] [CrossRef]
- Polak, L.; Demšar, L.; Kirinčič, S.; Kozolc, B.; Polak, T. Degradation of PCBs in liquid media: Effects of commercial meat starters. J. Food Sci. Technol. 2016, 65, 1087–1092. [Google Scholar] [CrossRef]
- Chang-Hyun, C.; Jaeyoon, L.; Bong-Gun, K.; Sung-Kuk, K.; Jong-Soo, C. Staphylococcus sp. KW-07 contains nahH gene encoding catechol 2,3-dioxygenase for phenanthrene degradation and a test in soil microcosm. Int. Biodeterior Biodegrad. 2011, 65, 198–203. [Google Scholar] [CrossRef]
- Eddouaouda, K.; Mnif, S.; Badis, A.; Younes, S.B.; Cherif, S.; Ferhat, S.; Mhiri, N.; Chamkha, M.; Sayadi, S. Characterization of a novel biosurfactant produced by Staphylococcus sp. strain 1E with potential application on hydrocarbon bioremediation. J. Basic Microbiol. 2012, 4, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Naeim, A.H.; Baharlouei, J.; Ataabadi, M. Biochemical tests to determine the biodegradability potential of bacterial strains in PAH polluted sites. World J. Microbiol. Biotechnol. 2020, 36, 181. [Google Scholar] [CrossRef] [PubMed]
- Samadi, A.; Sharifi, H.; Ghobadi, N.Z.; Yaghmaei, S. Biodegradation of Polychlorinated Biphenyls by Lysinibacillus macrolides and Bacillus firmus Isolated from Contaminated Soil. Int. J. Eng. 2019, 32, 628–633. [Google Scholar] [CrossRef] [Green Version]
- Masika, W.S.; Moonsamy, G.; Mandree, P.; Ramchuran, S.; Lalloo, R.; Kudanga, T. Biodegradation of petroleum hydrocarbon waste using consortia of Bacillus sp. Bioremediat. J. 2020, 25, 72–79. [Google Scholar] [CrossRef]
- Shimura, M.; Mukerjee-Dhar, G.; Kimbara, K.; Nagato, H.; Kiyohara, H.; Hatta, T. Isolation and characterization of a thermophilic Bacillus sp. JF8 capable of degrading polychlorinated biphenyls and naphthalene. FEMS Microbiol. Lett. 1999, 178, 87–93. [Google Scholar] [CrossRef] [Green Version]
- Sun, J.; Pan, L.; Zhu, L. Formation of hydroxylated and methoxylated polychlorinated biphenyls by Bacillus subtilis: New insights into microbial metabolism. Sci. Total Environ. 2018, 613–614, 54–61. [Google Scholar] [CrossRef]
- Leaes, F.L.; Daniel, A.P.; Mello, G.B.; Battisti, V.; Bogusz, S., Jr.; Emanuelli, T.; Fries, L.L.M.; Costabeber, I. Degradation of polychlorinated biphenyls (PCBs) by Staphylococcus xylosus in liquid media and meat mixture. Food Chem. Toxicol. 2006, 44, 847–854. [Google Scholar] [CrossRef]
- Jing, R.; Fusi, S.; Kjellerup, B.V. Remediation of Polychlorinated Biphenyls (PCBs) in Contaminated Soils and Sediment: State of Knowledge and Perspectives. Front. Environ. Sci. 2018, 6, 2296–2665X. [Google Scholar] [CrossRef]
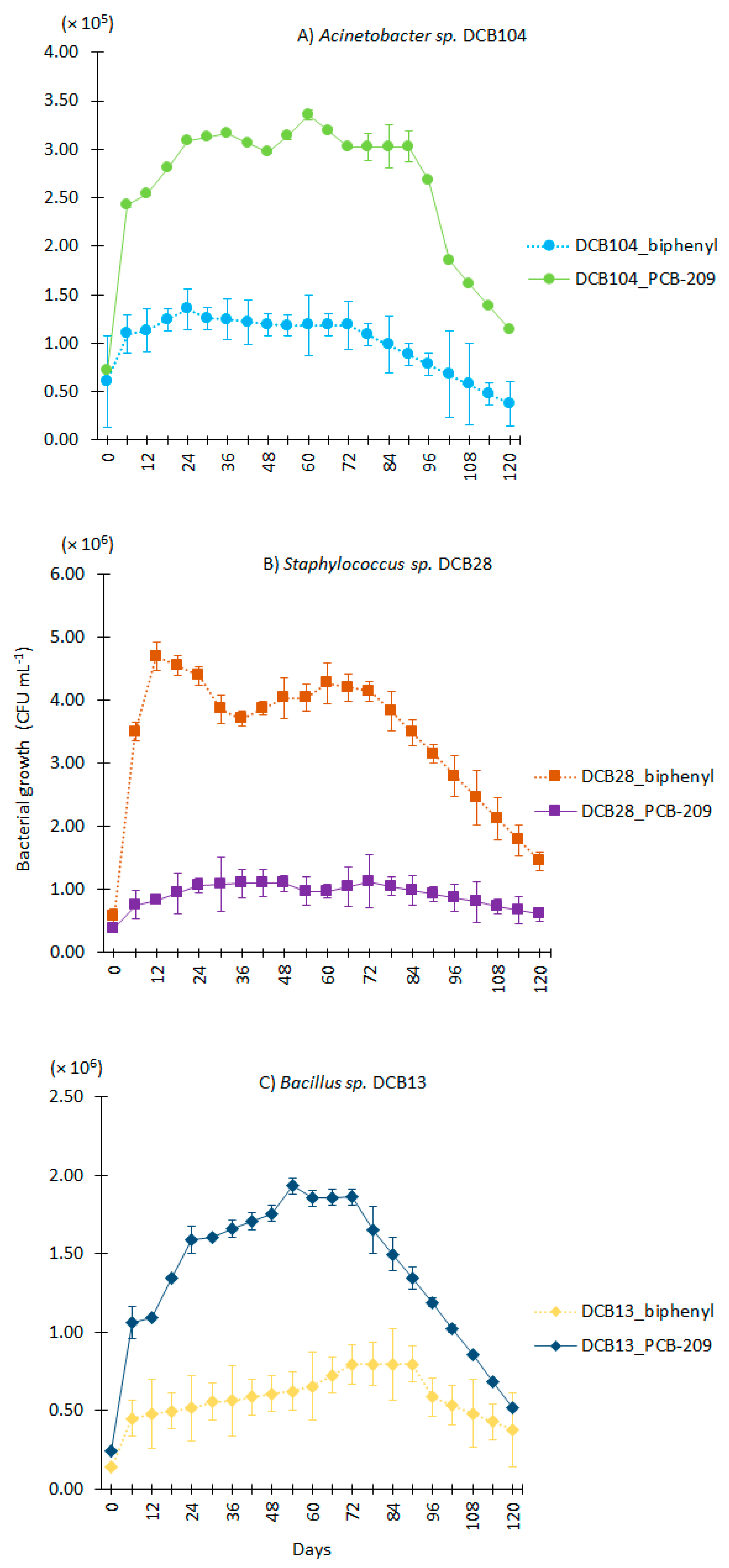
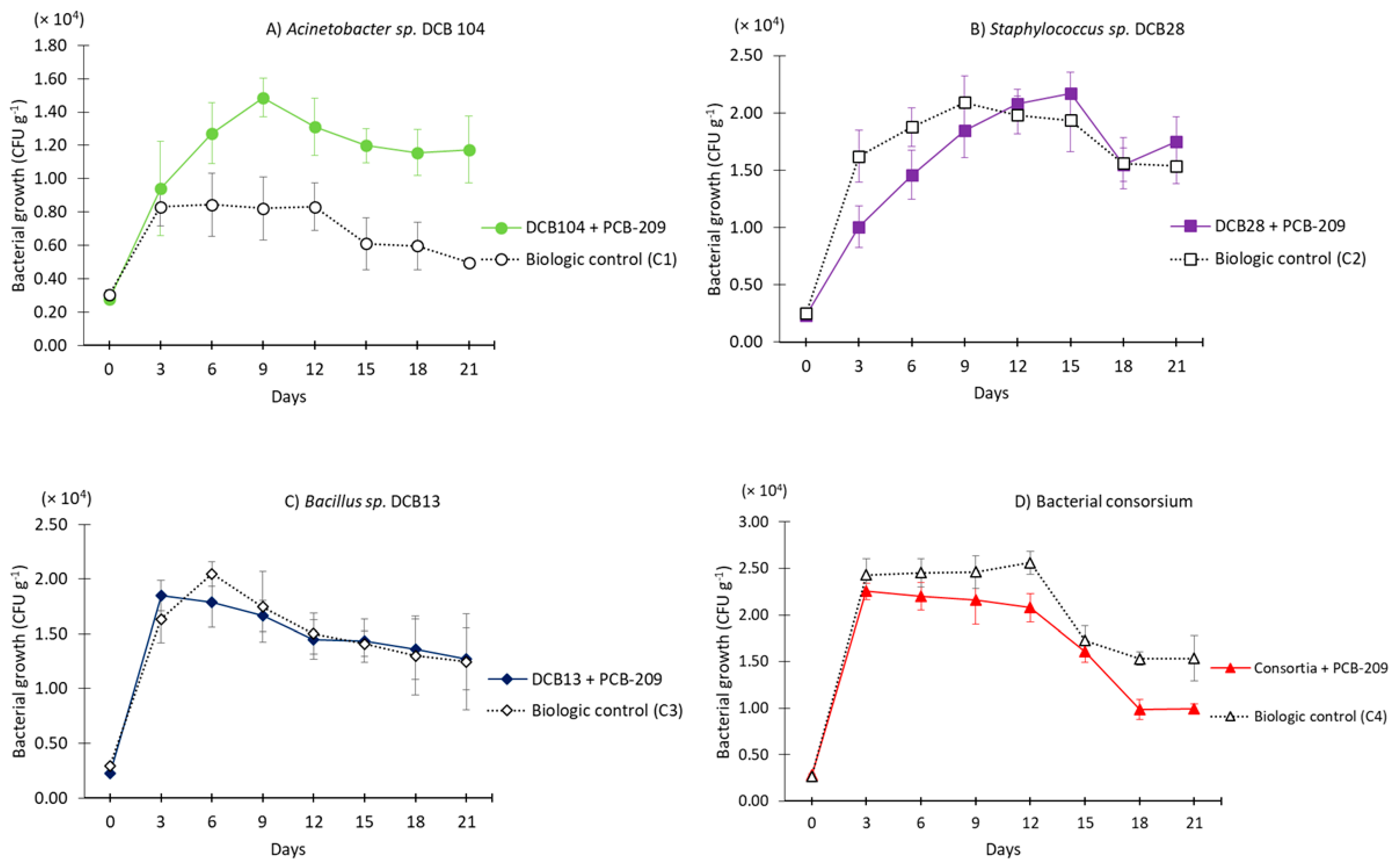
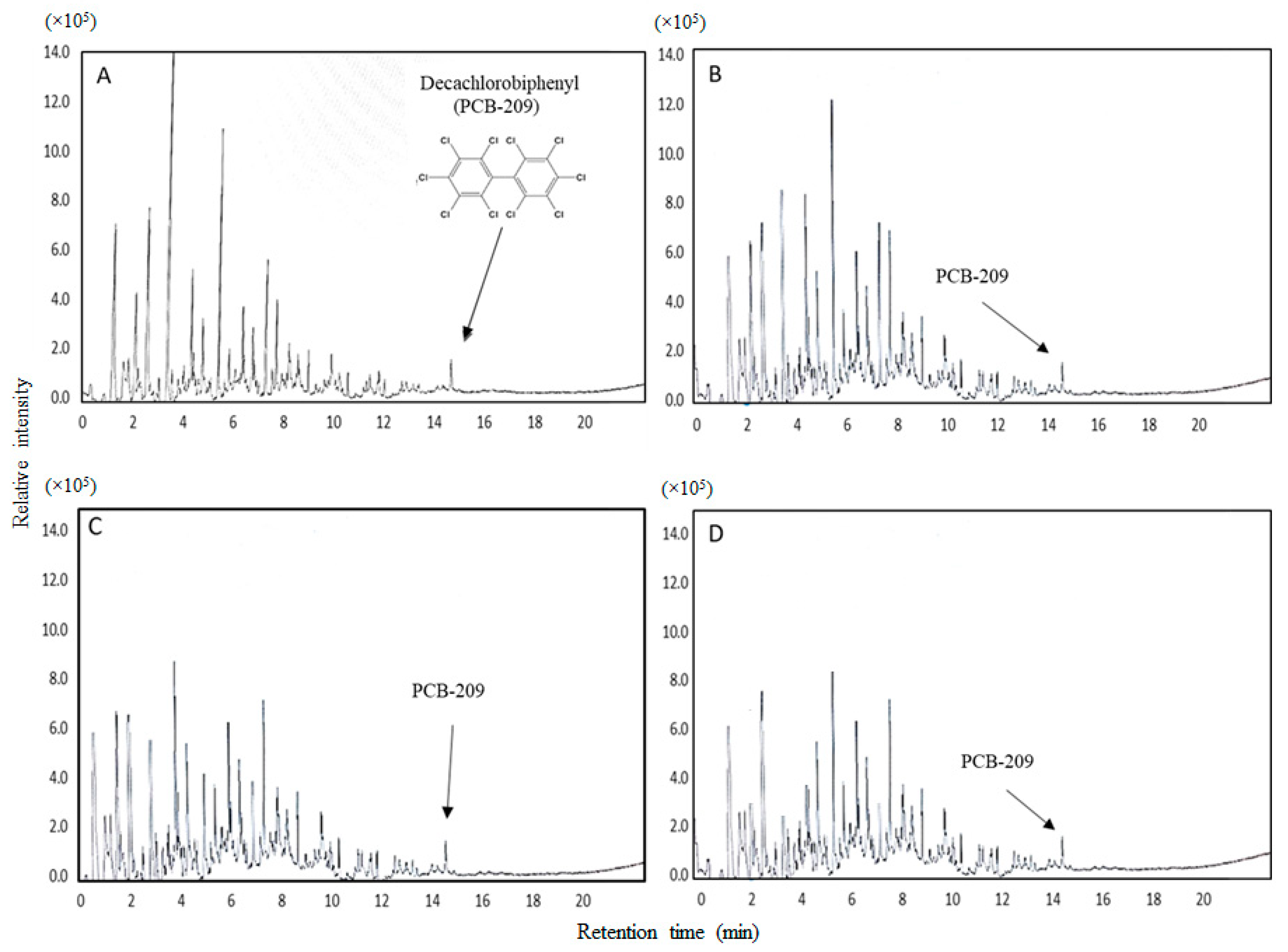

| Strain | Closest NCBI Match/Similarity (%) * | Accession Number | Phylum | Soil Collection Site |
|---|---|---|---|---|
| DCB13 | Bacillus thuringiensis L2.TYA/(95.6) | MZ544391 | Firmicutes | Tropical forest |
| DCB28 | Staphylococcus argenteus MSHR1132/(100) | MZ544389 | Firmicutes | Agricultural rhizosphere |
| CB104 | Acinetobacter baumannii B8342/(99.0) | MZ544388 | Proteobacteria | Hydroelectric power station |
| Treatment | Inoculated Strain | Treatment Characteristic |
|---|---|---|
| T1 | Acinetobacter sp. DCB104 | 200 mg/kg of PCB-209 |
| T2 | Staphylococcus sp. DCB28 | 200 mg/kg of PCB-209 |
| T3 | Bacillus sp. DCB13 | 200 mg/kg of PCB-209 |
| T4 | Consorsium (DCB104 + DCB28 + DCB13) | 200 mg/kg of PCB-209 |
| C1 | Strain DCB104 | Without contaminant |
| C2 | Strain DCB28 | Without contaminant |
| C3 | Strain DCB13 | Without contaminant |
| C4 | Consorsium (DCB104 + DCB28 + DCB13) | Without contaminant |
| CQ | Without inoculum | 200 mg/kg of PCB-209 (chemical control) |
| CN | Without inoculum | Without contaminant (negative control) |
| Treatments | Treatment Characteristics | pH | EC (dS m−1) | SOM (%) | TP (mg kg−1) | TC (mg kg−1) | TN (mg kg−1) | C:N Ratio |
|---|---|---|---|---|---|---|---|---|
| T1 | Strain DCB104 + decachlorobiphenyl, PCB-209 | 6.1 B * | 0.400 A | 13.0 C | 4.4 CD | 7.1 B | 0.84 AB | 8.76 C |
| T2 | Strain DCB28 + decachlorobiphenyl, PCB-209 | 6.1 B | 0.083 A | 12.6 CD | 5.1 B | 7.5 AB | 0.64 CD | 11.6 BC |
| T3 | Strain DCB13 + decachlorobiphenyl, PCB-209 | 6.2 AB | 0.077 A | 14.1 B | 4.8 BC | 7.8 AB | 0.74 BC | 10.6 BC |
| T4 | Consortium (DCB104, DCB28, and DCB13) + decachlorobiphenyl, PCB-209 | 5.9 B | 0.384 A | 15.4 A | 4.7 BC | 7.5 AB | 0.71 BCD | 10.4 BC |
| C1 | Strain DCB104 | 6.6 AB | 0.093 A | 11.0 E | 4.7 BC | 8.3 AB | 0.71 BCD | 11.7 BC |
| C2 | Strain DCB28 | 6.4 AB | 0.082 A | 12.0 D | 4.1 DE | 9.1 A | 0.90 A | 10.0 BC |
| C3 | Strain DCB13 | 6.3 AB | 0.062 A | 11.0 E | 4.1 DE | 8.7 AB | 0.58 D | 16.4 A |
| C4 | Consortium (DCB104, DCB28, and DCB13) | 6.5 AB | 0.080 A | 14.0 B | 3.7 E | 9.1 A | 0.69 BCD | 13.2 AB |
| CQ | Decachlorobiphenyl, PCB-209 | 6.8 A | 0.082 A | 11.1 E | 3.9 E | 7.9 AB | 0.72 BCD | 10.9 BC |
| CN | Uninoculated + without PCB-209 | 6.8 A | 0.090 A | 14.1 B | 5.6 A | 7.1 B | 0.57 D | 12.6 ABC |
| p-value = | 0.0010 | 0.5566 | 0.0000 | 0.0000 | 0.0024 | 0.0059 | 0.0693 | |
| Tukey HSD (p < 0.05) = | 0.6488 | 0.7005 | 0.6903 | 0.3970 | 1.7573 | 0.1577 | 4.19937 | |
| Treatments | Final Concentration of PCB-209 Remaining at Day 21 (mg/kg) | Estimated Amount Removed during Assay (mg/kg) | % of PCB-209 Remaining at Day 21 | Estimated Amount Removed During Assay (%) |
|---|---|---|---|---|
| T1 | 158.12 A * | 10.21 B | 94.15 B | 5.84 B |
| T2 | 160.43 A | 9.58 B | 94.28 B | 5.71 B |
| T3 | 160.02 A | 12.62 B | 93.33 B | 6.66 B |
| T4 | 143.66 B | 17.33 A | 89.48 C | 10.51 A |
| CQ | 163.10 A | 3.62 C | 97.53 A | 1.87 C |
| p-value | 0.0017 | 0.0000 | 0.0000 | 0.0000 |
| HSD (p < 0.05) | 11.9458 | 2.08816 | 1.93987 | 1.26814 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zenteno-Rojas, A.; Rincón-Molina, C.I.; Martinez-Romero, E.; Manzano-Gomez, L.A.; Rincón-Molina, F.A.; Ruiz-Valdiviezo, V.M.; Rodriguez, R.I.C.; Maldonado, J.J.V.; Rincón-Rosales, R. Biostimulation and Bioaugmentation of Soils Contaminated with Decachlorobiphenyl (PCB-209) Using Native Bacterial Strains Individually and in Consortia. Sustainability 2022, 14, 9068. https://doi.org/10.3390/su14159068
Zenteno-Rojas A, Rincón-Molina CI, Martinez-Romero E, Manzano-Gomez LA, Rincón-Molina FA, Ruiz-Valdiviezo VM, Rodriguez RIC, Maldonado JJV, Rincón-Rosales R. Biostimulation and Bioaugmentation of Soils Contaminated with Decachlorobiphenyl (PCB-209) Using Native Bacterial Strains Individually and in Consortia. Sustainability. 2022; 14(15):9068. https://doi.org/10.3390/su14159068
Chicago/Turabian StyleZenteno-Rojas, Adalberto, Clara Ivette Rincón-Molina, Esperanza Martinez-Romero, Luis Alberto Manzano-Gomez, Francisco Alexander Rincón-Molina, Victor Manuel Ruiz-Valdiviezo, Rosa Isela Cruz Rodriguez, Juan Jose Villalobos Maldonado, and Reiner Rincón-Rosales. 2022. "Biostimulation and Bioaugmentation of Soils Contaminated with Decachlorobiphenyl (PCB-209) Using Native Bacterial Strains Individually and in Consortia" Sustainability 14, no. 15: 9068. https://doi.org/10.3390/su14159068
APA StyleZenteno-Rojas, A., Rincón-Molina, C. I., Martinez-Romero, E., Manzano-Gomez, L. A., Rincón-Molina, F. A., Ruiz-Valdiviezo, V. M., Rodriguez, R. I. C., Maldonado, J. J. V., & Rincón-Rosales, R. (2022). Biostimulation and Bioaugmentation of Soils Contaminated with Decachlorobiphenyl (PCB-209) Using Native Bacterial Strains Individually and in Consortia. Sustainability, 14(15), 9068. https://doi.org/10.3390/su14159068








